The Application of Chemometrics in Metabolomic and Lipidomic Analysis Data Presentation for Halal Authentication of Meat Products
Abstract
1. Introduction
2. Halal Meat Products
3. Chemometrics
4. Chemometrics Applications in Metabolomic Studies for Halal Authentication of Meat Products
5. Chemometrics Applications in Lipidomic Analysis for Halal Authentication of Meat Products
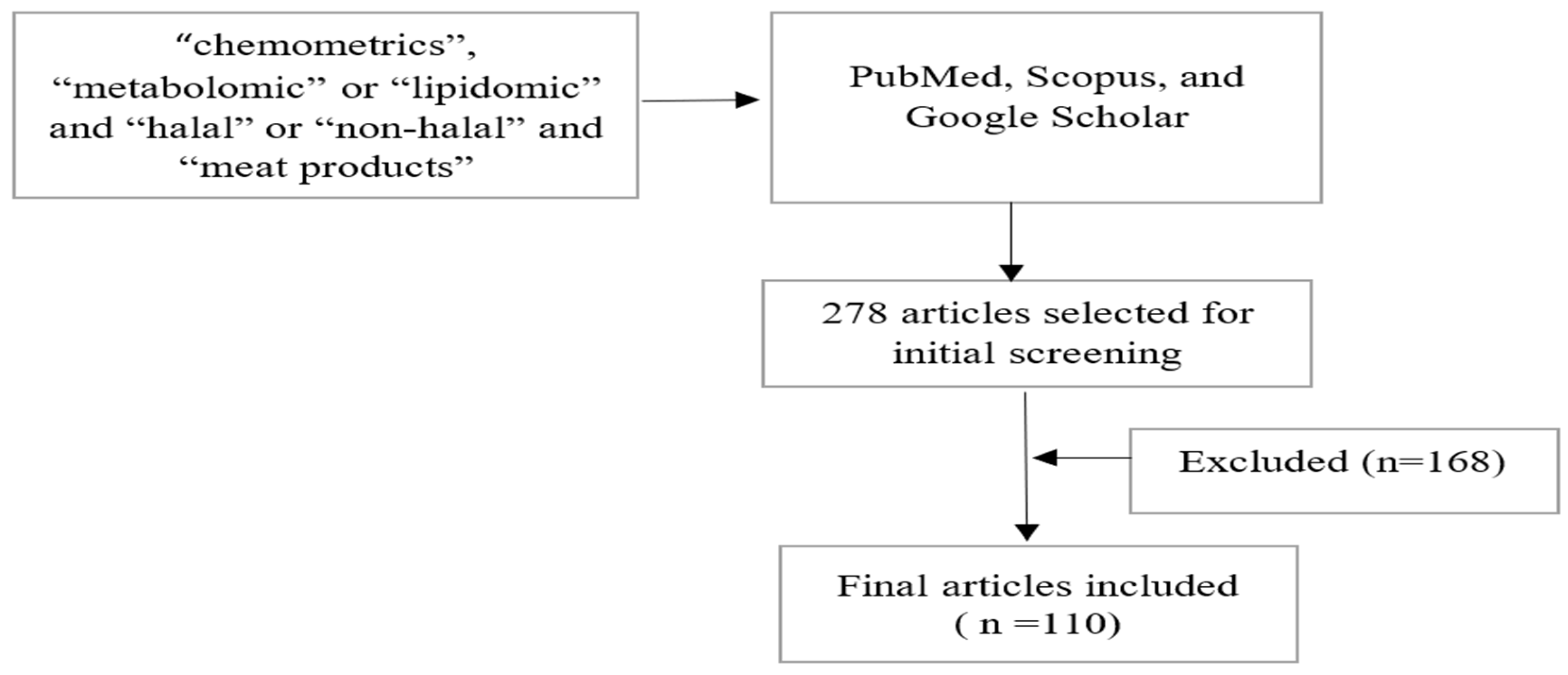
6. Methods
7. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nakyinsige, K.; Man, Y.B.C.; Sazili, A.Q. Halal Authenticity Issues in Meat and Meat Products. Meat Sci. 2012, 91, 207–214. [Google Scholar] [CrossRef]
- Hossain, M.A.M.; Uddin, S.M.K.; Sultana, S.; Wahab, Y.A.; Sagadevan, S.; Johan, M.R.; Ali, M.E. Authentication of Halal and Kosher Meat and Meat Products: Analytical Approaches, Current Progresses and Future Prospects. Crit. Rev. Food Sci. Nutr. 2022, 62, 285–310. [Google Scholar] [CrossRef] [PubMed]
- Martuscelli, M.; Serio, A.; Capezio, O.; Mastrocola, D. Safety, Quality and Analytical Authentication of Ḥalāl Meat Products, with Particular Emphasis on Salami: A Review. Foods 2020, 9, 1111. [Google Scholar] [CrossRef] [PubMed]
- Farouk, M.M.; Al-Mazeedi, H.M.; Sabow, A.B.; Bekhit, A.E.D.; Adeyemi, K.D.; Sazili, A.Q.; Ghani, A. Halal and Kosher Slaughter Methods and Meat Quality: A Review. Meat Sci. 2014, 98, 505–519. [Google Scholar] [CrossRef] [PubMed]
- Amir, S.H.; Yuswan, M.H.; Aizat, W.M.; Mansor, M.K.; Desa, M.N.M.; Yusof, Y.A.; Song, L.K.; Mustafa, S. Comparative Database Search Engine Analysis on Massive Tandem Mass Spectra of Pork-Based Food Products for Halal Proteomics. J. Proteom. 2021, 241, 104240. [Google Scholar] [CrossRef] [PubMed]
- Alikord, M.; Keramat, J.; Kadivar, M.; Momtaz, H.; Eshtiaghi, M.N.; Homayouni-Rad, A. Multiplex-PCR As a Rapid and Sensitive Method for Identification of Meat Species in Halal-Meat Products. Recent Pat. Food. Nutr. Agric. 2017, 8, 175–182. [Google Scholar] [CrossRef]
- Lou, X.; Zhai, D.; Yang, H. Changes of Metabolite Profiles of Fish Models Inoculated with Shewanella Baltica during Spoilage. Food Control 2021, 123, 107697. [Google Scholar] [CrossRef]
- Chen, H.; Wei, F.; Dong, X.; Xiang, J.; Quek, S.; Wang, X. Lipidomics in Food Science. Curr. Opin. Food Sci. 2017, 16, 80–87. [Google Scholar] [CrossRef]
- Bo, Y.; Feng, J.; Xu, J.J.; Huang, Y.; Cai, H.; Cui, X.; Dong, J.; Ding, S.; Chen, Z. High-Resolution Pure Shift NMR Spectroscopy Offers Better Metabolite Discrimination in Food Quality Analysis. Food Res. Int. 2019, 125, 108574. [Google Scholar] [CrossRef]
- Lin, W.J.; Shen, P.C.; Liu, H.C.; Cho, Y.C.; Hsu, M.K.; Lin, I.C.; Chen, F.H.; Yang, J.C.; Ma, W.L.; Cheng, W.C. LipidSig: A Web-Based Tool for Lipidomic Data Analysis. Nucleic Acids Res. 2021, 49, W336–W345. [Google Scholar] [CrossRef]
- Sethi, S.; Brietzke, E. Recent Advances in Lipidomics: Analytical and Clinical Perspectives. Prostaglandins Other Lipid Mediat. 2017, 128–129, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Harlina, P.W.; Maritha, V.; Musfiroh, I.; Huda, S.; Sukri, N.; Muchtaridi, M. Possibilities of Liquid Chromatography Mass Spectrometry (LC-MS)-Based Metabolomics and Lipidomics in the Authentication of Meat Products: A Mini Review. Food Sci. Anim. Resour. 2022, 42, 744–761. [Google Scholar] [CrossRef] [PubMed]
- Rohman, A.; Ghazali, M.A.I.B.; Windarsih, A.; Irnawati; Riyanto, S.; Yusof, F.M.; Mustafa, S. Comprehensive Review on Application of FTIR Spectroscopy Coupled with Chemometrics for Authentication Analysis of Fats and Oils in the Food Products. Molecules 2020, 25, 5485. [Google Scholar] [CrossRef] [PubMed]
- Pavlidis, D.E.; Mallouchos, A.; Ercolini, D.; Panagou, E.Z.; Nychas, G.J.E. A Volatilomics Approach for Off-Line Discrimination of Minced Beef and Pork Meat and Their Admixture Using HS-SPME GC/MS in Tandem with Multivariate Data Analysis. Meat Sci. 2019, 151, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Nurani, L.H.; Riswanto, F.D.O.; Windarsih, A.; Edityaningrum, C.A.; Guntarti, A.; Rohman, A. Use of chromatographic-based techniques and chemometrics for halal authentication of food products: A review. Int. J. Food Prop. 2022, 25, 1399–1416. [Google Scholar] [CrossRef]
- Xu, L.; Cai, C.B.; Cui, H.F.; Ye, Z.H.; Yu, X.P. Rapid Discrimination of Pork in Halal and Non-Halal Chinese Ham Sausages by Fourier Transform Infrared (FTIR) Spectroscopy and Chemometrics. Meat Sci. 2012, 92, 506–510. [Google Scholar] [CrossRef]
- Granato, D.; Putnik, P.; Kovačević, D.B.; Santos, J.S.; Calado, V.; Rocha, R.S.; Da Cruz, A.G.; Jarvis, B.; Rodionova, O.Y.; Pomerantsev, A. Trends in Chemometrics: Food Authentication, Microbiology, and Effects of Processing. Compr. Rev. Food Sci. Food Saf. 2018, 17, 663–677. [Google Scholar] [CrossRef]
- Sohaib, M.; Jamil, F. An Insight of Meat Industry in Pakistan with Special Reference to Halal Meat: A Comprehensive Review. Korean J. Food Sci. Anim. Resour. 2017, 37, 329. [Google Scholar] [CrossRef]
- Khan, M.I.; Haleem, A.; Khan, S. Defining Halal Supply Chain Management. Supply Chain Forum: Int. J. 2018, 19, 122–131. [Google Scholar] [CrossRef]
- Suzana Jaafar, H.; Faisol, N.; Abd Rahman, F.; Muhammad, A. Halal Logistics Versus Halal Supply Chain: A Preliminary Insight. Contemp. Issues Dev. Glob. Halal Ind. 2016, 579–588. [Google Scholar] [CrossRef]
- Nurhayati, T.; Hendar, H. Personal Intrinsic Religiosity and Product Knowledge on Halal Product Purchase Intention: Role of Halal Product Awareness. J. Islam. Mark. 2020, 11, 603–620. [Google Scholar] [CrossRef]
- Verbeka, W.; Rutseart, P.; Bonne, K.; Vermeir, I. Credence quality coordination and consumer’s willigness-to-pay-for certified halal labelled meat. Meat Sci. 2013, 95, 790–797. [Google Scholar] [CrossRef] [PubMed]
- Izberk-Bilgin, E.; Nakata, C.C. A New Look at Faith-Based Marketing: The Global Halal Market. Bus. Horiz. 2016, 59, 285–292. [Google Scholar] [CrossRef]
- Aghwan, Z.A.; Bello, A.U.; Abubakar, A.A.; Imlan, J.C.; Sazili, A.Q. Efficient Halal Bleeding, Animal Handling, and Welfare: A Holistic Approach for Meat Quality. Meat Sci. 2016, 121, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Anwar, M.K. The Urgency of Halal Assurance System for Product Reliability. Int. J. Islam. Bus. Econ. 2018, 2, 119–125. [Google Scholar] [CrossRef]
- Bonne, K.; Vermeir, I.; Verbeke, W. Impact of Religion on Halal Meat Consumption Decision Making in Belgium. J. Int. Food Agribus. Mark. 2009, 21, 5–26. [Google Scholar] [CrossRef]
- Bonne, K.; Verbeke, W. Religious Values Informing Halal Meat Production and the Control and Delivery of Halal Credence Quality. Agric. Hum. Values 2007, 25, 35–47. [Google Scholar] [CrossRef]
- Zaharah, T.; Rahimi Osman, M.; Normalina Omar, E. A Review of Regulatory Framework for Halal Meat Supply Chain: The Case of Halal Meat Based Food Products in Mala... Related Papers Halalan Toyyiban Supply Chain of t He Food Industry. J. Appl. Environ. Biol. Sci. 2016, 6, 14–21. [Google Scholar]
- Fuseini, A.; Hadley, P.; Knowles, T. Halal Food Marketing: An Evaluation of UK Halal Standards. J. Islam. Mark. 2020, 12, 977–991. [Google Scholar] [CrossRef]
- Denyingyhot, A.; Srinulgray, T.; Mahamad, P.; Ruangprach, A.; Sa-I, S.; Saerae, T.; Vesaratchavest, M.; Dahlan, W.; Keeratipibul, S. Modern On-Site Tool for Monitoring Contamination of Halal Meat with Products from Five Non-Halal Animals Using Multiplex Polymerase Chain Reaction Coupled with DNA Strip. Food Control 2022, 132, 108540. [Google Scholar] [CrossRef]
- Siddiqui, M.A.; Khir, M.H.M.; Witjaksono, G.; Ghumman, A.S.M.; Junaid, M.; Magsi, S.A.; Saboor, A. Multivariate Analysis Coupled with M-SVM Classification for Lard Adulteration Detection in Meat Mixtures of Beef, Lamb, and Chicken Using FTIR Spectroscopy. Foods 2021, 10, 2405. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, F.A.A.; Borilova, G.; Steinhauserova, I. Halal Criteria Versus Conventional Slaughter Technology. Animals 2019, 9, 530. [Google Scholar] [CrossRef] [PubMed]
- Fuseini, A.; Knowles, T.G.; Hadley, P.J.; Wotton, S.B. Halal Stunning and Slaughter: Criteria for the Assessment of Dead Animals. Meat Sci. 2016, 119, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Fuseini, A.; Wotton, S.B.; Knowles, T.G.; Hadley, P.J. Halal Meat Fraud and Safety Issues in the UK: A Review in the Context of the European Union. Food Ethics 2017, 1, 127–142. [Google Scholar] [CrossRef]
- Anam, J.; Sany Sanuri, B.M.M.; Ismail, B.L.O. Conceptualizing the Relation between Halal Logo, Perceived Product Quality and the Role of Consumer Knowledge. J. Islam. Mark. 2018, 9, 727–746. [Google Scholar] [CrossRef]
- Kumar, N.; Bansal, A.; Sarma, G.S.; Rawal, R.K. Chemometrics Tools Used in Analytical Chemistry: An Overview. Talanta 2014, 123, 186–199. [Google Scholar] [CrossRef]
- Kanginejad, A.; Mani-Varnosfaderani, A. Chemometrics Advances on the Challenges of the Gas Chromatography–Mass Spectrometry Metabolomics Data: A Review. J. Iran. Chem. Soc. 2018, 15, 2733–2745. [Google Scholar] [CrossRef]
- Rohman, A.; Windarsih, A. The Application of Molecular Spectroscopy in Combination with Chemometrics for Halal Authentication Analysis: A Review. Int. J. Mol. Sci. 2020, 21, 5155. [Google Scholar] [CrossRef]
- Martynko, E.; Kirsanov, D. Application of Chemometrics in Biosensing: A Brief Review. Biosensors 2020, 10, 100. [Google Scholar] [CrossRef]
- Feizi, N.; Hashemi-Nasab, F.S.; Golpelichi, F.; Sabouruh, N.; Parastar, H. Recent Trends in Application of Chemometric Methods for GC-MS and GC×GC-MS-Based Metabolomic Studies. TrAC-Trends Anal. Chem. 2021, 138, 116239. [Google Scholar] [CrossRef]
- Navarro-Reig, M.; Jaumot, J.; Tauler, R. An Untargeted Lipidomic Strategy Combining Comprehensive Two-Dimensional Liquid Chromatography and Chemometric Analysis. J. Chromatogr. A 2018, 1568, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Sahu, P.K.; Ramisetti, N.R.; Cecchi, T.; Swain, S.; Patro, C.S.; Panda, J. An Overview of Experimental Designs in HPLC Method Development and Validation. J. Pharm. Biomed. Anal. 2018, 147, 590–611. [Google Scholar] [CrossRef] [PubMed]
- Karabagias, I.K. Advances of Spectrometric Techniques in Food Analysis and Food Authentication Implemented with Chemometrics. Foods 2020, 9, 1550. [Google Scholar] [CrossRef]
- Kumar, R.; Sharma, V. Chemometrics in Forensic Science. TrAC Trends Anal. Chem. 2018, 105, 191–201. [Google Scholar] [CrossRef]
- Wu, B.; Wei, F.; Xu, S.; Xie, Y.; Lv, X.; Chen, H.; Huang, F. Mass Spectrometry-Based Lipidomics as a Powerful Platform in Foodomics Research. Trends Food Sci. Technol. 2021, 107, 358–376. [Google Scholar] [CrossRef]
- Andre, C.M.; Soukoulis, C. Food Quality Assessed by Chemometrics. Foods 2020, 9, 897. [Google Scholar] [CrossRef]
- Kalogiouri, N.P.; Aalizadeh, R.; Dasenaki, M.E.; Thomaidis, N.S. Application of High Resolution Mass Spectrometric Methods Coupled with Chemometric Techniques in Olive Oil Authenticity Studies—A Review. Anal. Chim. Acta 2020, 1134, 150–173. [Google Scholar] [CrossRef] [PubMed]
- Hassan, N.; Ahmad, T.; Zain, N.M. Chemical and Chemometric Methods for Halal Authentication of Gelatin: An Overview. J. Food Sci. 2018, 83, 2903–2911. [Google Scholar] [CrossRef] [PubMed]
- Karamizadeh, S.; Abdullah, S.M.; Manaf, A.A.; Zamani, M.; Hooman, A. An Overview of Principal Component Analysis. J. Signal Inf. Process. 2013, 4, 173–175. [Google Scholar] [CrossRef]
- Windarsih, A.; Suratno; Warmiko, H.D.; Indrianingsih, A.W.; Rohman, A.; Ulumuddin, Y.I. Untargeted Metabolomics and Proteomics Approach Using Liquid Chromatography-Orbitrap High Resolution Mass Spectrometry to Detect Pork Adulteration in Pangasius Hypopthalmus Meat. Food Chem. 2022, 386, 132856. [Google Scholar] [CrossRef]
- Harlina, P.W.; Ma, M.; Shahzad, R. Quantification of Lipidomics Profiling Using UPLC-QE-HESI- Lipid Analysis on the Salted Duck Egg Incorporated with Clove Extract. Eur. J. Lipid Sci. Technol. 2021, 123, 2000284. [Google Scholar] [CrossRef]
- Dirong, G.; Nematbakhsh, S.; Selamat, J.; Chong, P.P.; Idris, L.H.; Nordin, N.; Fatchiyah, F.; Razis, A.F.A. Omics-Based Analytical Approaches for Assessing Chicken Species and Breeds in Food Authentication. Molecules 2021, 26, 6502. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.; Zhang, Y.; Zhang, M.; Qi, J.; Zhao, W.; Gu, J.; Guo, W.; Li, Y. Screening of Specific Quantitative Peptides of Beef by LC–MS/MS Coupled with OPLS-DA. Food Chem. 2022, 387, 132932. [Google Scholar] [CrossRef] [PubMed]
- Cebi, N.; Dogan, C.E.; Mese, A.E.; Ozdemir, D.; Arıcı, M.; Sagdic, O. A Rapid ATR-FTIR Spectroscopic Method for Classification of Gelatin Gummy Candies in Relation to the Gelatin Source. Food Chem. 2019, 277, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Panchuk, V.; Yaroshenko, I.; Legin, A.; Semenov, V.; Kirsanov, D. Application of Chemometric Methods to XRF-Data—A Tutorial Review. Anal. Chim. Acta 2018, 1040, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Azizan, N.I.; Mokhtar, N.F.K.; Arshad, S.; Sharin, S.N.; Mohamad, N.; Mustafa, S.; Hashim, A.M. Detection of Lard Adulteration in Wheat Biscuits Using Chemometrics-Assisted GCMS and Random Forest. Food Anal. Methods 2021, 14, 2276–2287. [Google Scholar] [CrossRef]
- Jia, W.; Wu, X.; Li, R.; Liu, S.; Shi, L. Effect of Nisin and Potassium Sorbate Additions on Lipids and Nutritional Quality of Tan Sheep Meat. Food Chem. 2021, 365, 130535. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, C.; Xie, K.; Wang, J.; Pan, Z. Current State of Metabolomics Research in Meat Quality Analysis and Authentication. Foods 2021, 10, 2388. [Google Scholar] [CrossRef]
- Mi, S.; Shang, K.; Li, X.; Zhang, C.H.; Liu, J.Q.; Huang, D.Q. Characterization and Discrimination of Selected China’s Domestic Pork Using an LC-MS-Based Lipidomics Approach. Food Control 2019, 100, 305–314. [Google Scholar] [CrossRef]
- Mishra, P.; Biancolillo, A.; Roger, J.M.; Marini, F.; Rutledge, D.N. New Data Preprocessing Trends Based on Ensemble of Multiple Preprocessing Techniques. TrAC-Trends Anal. Chem. 2020, 132, 6502. [Google Scholar] [CrossRef]
- Houhou, R.; Bocklitz, T. Trends in Artificial Intelligence, Machine Learning, and Chemometrics Applied to Chemical Data. Anal. Sci. Adv. 2021, 2, 128–141. [Google Scholar] [CrossRef]
- Stefanuto, P.H.; Smolinska, A.; Focant, J.F. Advanced Chemometric and Data Handling Tools for GC×GC-TOF-MS: Application of Chemometrics and Related Advanced Data Handling in Chemical Separations. TrAC Trends Anal. Chem. 2021, 139, 116251. [Google Scholar] [CrossRef]
- Teye, E.; Amuah, C.L.Y.; McGrath, T.; Elliott, C. Innovative and Rapid Analysis for Rice Authenticity Using Hand-Held NIR Spectrometry and Chemometrics. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 217, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.H.; Li, H.D.; Deng, B.C.; Cao, D.S. An Overview of Variable Selection Methods in Multivariate Analysis of Near-Infrared Spectra. TrAC Trends Anal. Chem. 2019, 113, 102–115. [Google Scholar] [CrossRef]
- Mishra, P.; Roger, J.M.; Jouan-Rimbaud-Bouveresse, D.; Biancolillo, A.; Marini, F.; Nordon, A.; Rutledge, D.N. Recent Trends in Multi-Block Data Analysis in Chemometrics for Multi-Source Data Integration. TrAC Trends Anal. Chem. 2021, 137, 116206. [Google Scholar] [CrossRef]
- Truong, V.K.; Dupont, M.; Elbourne, A.; Gangadoo, S.; Pathirannahalage, P.R.; Cheeseman, S.; Chapman, J.; Cozzolino, D. From Academia to Reality Check: A Theoretical Framework on the Use of Chemometric in Food Sciences. Foods 2019, 8, 164. [Google Scholar] [CrossRef]
- Utpott, M.; Rodrigues, E.; de Oliveira Rios, A.; Mercali, G.D.; Flôres, S.H. Metabolomics: An Analytical Technique for Food Processing Evaluation. Food Chem. 2022, 366, 130685. [Google Scholar] [CrossRef]
- Sani, N.A.; Akbar, H.; Yaacob, O.; Ahmmad, S.; Rafee, Y.M.; Nuriz, A.; Hafsah, B.; Dahlan, H.A. Current Trend for Food Safety and Halal Measures. In Proceedings of the ASEAN Community Conference 2015, Bangi, Malaysia, 11–12 November 2015. [Google Scholar]
- Cambiaghi, A.; Ferrario, M.; Masseroli, M. Analysis of Metabolomic Data: Tools, Current Strategies and Future Challenges for Omics Data Integration. Brief. Bioinform. 2017, 18, 498–510. [Google Scholar] [CrossRef]
- Ebrahimi, P.; Larsen, F.H.; Jensen, H.M.; Vogensen, F.K.; Engelsen, S.B. Real-Time Metabolomic Analysis of Lactic Acid Bacteria as Monitored by in Vitro NMR and Chemometrics. Metabolomics 2016, 12, 1–17. [Google Scholar] [CrossRef]
- Khakimov, B.; Jespersen, B.M.; Engelsen, S.B. Comprehensive and Comparative Metabolomic Profiling of Wheat, Barley, Oat and Rye Using Gas Chromatography-Mass Spectrometry and Advanced Chemometrics. Foods 2014, 3, 569–585. [Google Scholar] [CrossRef]
- Dawid, C.; Hille, K. Functional Metabolomics—A Useful Tool to Characterize Stress-Induced Metabolome Alterations Opening New Avenues towards Tailoring Food Crop Quality. Agronomy 2018, 8, 138. [Google Scholar] [CrossRef]
- Dashti, A.; Müller-Maatsch, J.; Weesepoel, Y.; Parastar, H.; Kobarfard, F.; Daraei, B.; Aliabadi, M.H.S.; Yazdanpanah, H. The Feasibility of Two Handheld Spectrometers for Meat Speciation Combined with Chemometric Methods and Its Application for Halal Certification. Foods 2022, 11, 71. [Google Scholar] [CrossRef] [PubMed]
- Chong, J.; Wishart, D.S.; Xia, J. Using MetaboAnalyst 4.0 for Comprehensive and Integrative Metabolomics Data Analysis. Curr. Protoc. Bioinforma. 2019, 68, e86. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.C. Chemometrics Methods and Strategies in Metabolomics. Adv. Exp. Med. Biol. 2017, 965, 163–190. [Google Scholar] [CrossRef]
- Ferreira, S.L.C.; Silva Junior, M.M.; Felix, C.S.A.; da Silva, D.L.F.; Santos, A.S.; Santos Neto, J.H.; de Souza, C.T.; Cruz Junior, R.A.; Souza, A.S. Multivariate Optimization Techniques in Food Analysis—A Review. Food Chem. 2019, 273, 3–8. [Google Scholar] [CrossRef]
- Rudd, T.R.; Mauri, L.; Marinozzi, M.; Stancanelli, E.; Yates, E.A.; Naggi, A.; Guerrini, M. Multivariate Analysis Applied to Complex Biological Medicines. Faraday Discuss. 2019, 218, 303–316. [Google Scholar] [CrossRef]
- Dailey, A.L. Metabolomic Bioinformatic Analysis. Methods Mol. Biol. 2017, 1606, 341–352. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, M.; Wang, S.; Kang, C.; Zhang, M.; Li, Y. Identification and Quantification of Fox Meat in Meat Products by Liquid Chromatography–Tandem Mass Spectrometry. Food Chem. 2022, 372, 131336. [Google Scholar] [CrossRef]
- Ueda, S.; Iwamoto, E.; Kato, Y.; Shinohara, M.; Shirai, Y.; Yamanoue, M. Comparative Metabolomics of Japanese Black Cattle Beef and Other Meats Using Gas Chromatography–Mass Spectrometry. Biosci. Biotechnol. Biochem. 2019, 83, 137–147. [Google Scholar] [CrossRef]
- Gromski, P.S.; Muhamadali, H.; Ellis, D.I.; Xu, Y.; Correa, E.; Turner, M.L.; Goodacre, R. A Tutorial Review: Metabolomics and Partial Least Squares-Discriminant Analysis--a Marriage of Convenience or a Shotgun Wedding. Anal. Chim. Acta 2015, 879, 10–23. [Google Scholar] [CrossRef]
- Shi, H.; Yuan, J.; Zhang, Y.; Feng, S.; Wang, J. Discovering Significantly Different Metabolites between Han and Uygur Two Racial Groups Using Urinary Metabolomics in Xinjiang, China. J. Pharm. Biomed. Anal. 2019, 164, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Shikh Zahari, S.M.S.N.; Mohamed Ali, N.S.; Zabidi, A.R.; Rosli, D.; Abdul Manap, M.N.; Yahaya, N. Influence of Neck Slaughtering in Broiler Chicken Meat on Physicochemical Analysis and Metabolites ‘Fingerprinting’ to Enhance Meat Quality. Arab. J. Chem. 2021, 14, 103042. [Google Scholar] [CrossRef]
- Ali, N.S.M.; Zabidi, A.R.; Manap, M.N.A.; Zahari, S.M.S.N.S.; Yahaya, N. Identification of Metabolite Profile in Halal and Non-Halal Broiler Chickens Using Fourier-Transform Infrared Spectroscopy (Ftir) and Ultra High Performance Liquid Chromatography-Time of Flight-Mass Spectrometry (Uhplc-Tof-Ms). Malays. Appl. Biol. 2020, 49, 87–93. [Google Scholar] [CrossRef]
- Pranata, A.W.; Yuliana, N.D.; Amalia, L.; Darmawan, N. Volatilomics for Halal and Non-Halal Meatball Authentication Using Solid-Phase Microextraction–Gas Chromatography–Mass Spectrometry. Arab. J. Chem. 2021, 14, 103146. [Google Scholar] [CrossRef]
- Akhtar, M.T.; Samar, M.; Shami, A.A.; Mumtaz, M.W.; Mukhtar, H.; Tahir, A.; Shahzad-Ul-hussan, S.; Chaudhary, S.U.; Kaka, U. 1 H-NMR-Based Metabolomics: An Integrated Approach for the Detection of the Adulteration in Chicken, Chevon, Beef and Donkey Meat. Molecules 2021, 26, 4643. [Google Scholar] [CrossRef] [PubMed]
- Ahda, M.; Guntarti, A.; Kusbandari, A.; Melianto, Y. Authenticity Analysis of Beef Meatball Adulteration with Wild Boar Using Ftir Spectroscopy Combined with Chemometrics. J. Microbiol. Biotechnol. Food Sci. 2020, 9, 937–940. [Google Scholar] [CrossRef]
- Yuswan, M.H.; Aizat, W.M.; Lokman, A.A.; Desa, M.N.M.; Mustafa, S.; Junoh, N.M.; Yusof, Z.N.B.; Mohamed, R.; Mohmad, Z.; Lamasudin, D.U. Chemometrics-Assisted Shotgun Proteomics for Establishment of Potential Peptide Markers of Non-Halal Pork (Sus Scrofa) among Halal Beef and Chicken. Food Anal. Methods 2018, 11, 3505–3515. [Google Scholar] [CrossRef]
- Heidari, M.; Talebpour, Z.; Abdollahpour, Z.; Adib, N.; Ghanavi, Z.; Aboul-Enein, H.Y. Discrimination between Vegetable Oil and Animal Fat by a Metabolomics Approach Using Gas Chromatography-Mass Spectrometry Combined with Chemometrics. J. Food Sci. Technol. 2020, 57, 3415–3425. [Google Scholar] [CrossRef]
- Abbas, N.; Ali, A.; Kumari, S.; Iqbal, A.; Husain, A.; Saeed, T.; AbdulAmer Al-Ballam, Z.; Ahmed, N.; El-Seedi, H.R.; Musharraf, S.G. Untargeted-Metabolomics Differentiation between Poultry Samples Slaughtered with and without Detaching Spinal Cord. Arab. J. Chem. 2020, 13, 9081–9089. [Google Scholar] [CrossRef]
- Zhang, M.; Li, Y.; Zhang, Y.; Kang, C.; Zhao, W.; Ren, N.; Guo, W.; Wang, S. Rapid LC-MS/MS Method for the Detection of Seven Animal Species in Meat Products. Food Chem. 2022, 371, 131075. [Google Scholar] [CrossRef]
- Züllig, T.; Trötzmüller, M.; Köfeler, H.C. Lipidomics from Sample Preparation to Data Analysis: A Primer. Anal. Bioanal. Chem. 2020, 412, 2191–2209. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, Z.; Zhang, H.; Wang, Y.; Liu, X.; Wang, Q.; Xue, J.; Zhao, Y.; Yang, S. Meat Differentiation between Pasture-Fed and Concentrate-Fed Sheep/Goats by Liquid Chromatography Quadrupole Time-of-Flight Mass Spectrometry Combined with Metabolomic and Lipidomic Profiling. Meat Sci. 2021, 173, 108374. [Google Scholar] [CrossRef] [PubMed]
- Che Man, Y.B.; Rohman, A. Detection of Lard in Vegetable Oils. Lipid Technol. 2011, 23, 180–182. [Google Scholar] [CrossRef]
- Cajka, T.; Showalter, M.R.; Riddellova, K.; Fiehn, O. Advances in Mass Spectrometry for Food Authenticity Testing: An Omics Perspective. Adv. Food Authent. Test. 2016, 171–200. [Google Scholar] [CrossRef]
- Syed Idrus, S.I.; Latiff, A.A.; Ismail, M.N. Determination of Triacylglycerols in Food by High-Performance Liquid Chromatography. Instrum. Sci. Technol. 2017, 45, 577–591. [Google Scholar] [CrossRef]
- Avela, H.F.; Sirén, H. Advances in Lipidomics. Clin. Chim. Acta. 2020, 510, 123–141. [Google Scholar] [CrossRef]
- Wu, Z.; Bagarolo, G.I.; Thoröe-Boveleth, S.; Jankowski, J. “Lipidomics”: Mass Spectrometric and Chemometric Analyses of Lipids. Adv. Drug Deliv. Rev. 2020, 159, 294–307. [Google Scholar] [CrossRef]
- Zhao, Y.Y.; Wu, S.P.; Liu, S.; Zhang, Y.; Lin, R.C. Ultra-Performance Liquid Chromatography–Mass Spectrometry as a Sensitive and Powerful Technology in Lipidomic Applications. Chem. Biol. Interact. 2014, 220, 181–192. [Google Scholar] [CrossRef]
- Artegoitia, V.M.; Foote, A.P.; Lewis, R.M.; Freetly, H.C. Metabolomics Profile and Targeted Lipidomics in Multiple Tissues Associated with Feed Efficiency in Beef Steers. ACS Omega 2019, 4, 3973–3982. [Google Scholar] [CrossRef]
- Novák, M.; Palya, D.; Bodai, Z.; Nyiri, Z.; Magyar, N.; Kovács, J.; Eke, Z. Combined Cluster and Discriminant Analysis: An Efficient Chemometric Approach in Diesel Fuel Characterization. Forensic Sci. Int. 2017, 270, 61–69. [Google Scholar] [CrossRef]
- Yi, L.; Dong, N.; Yun, Y.; Deng, B.; Ren, D.; Liu, S.; Liang, Y. Chemometric Methods in Data Processing of Mass Spectrometry-Based Metabolomics: A Review. Anal. Chim. Acta 2016, 914, 17–34. [Google Scholar] [CrossRef] [PubMed]
- Cajka, T.; Smilowitz, J.T.; Fiehn, O. Validating Quantitative Untargeted Lipidomics Across Nine Liquid Chromatography-High-Resolution Mass Spectrometry Platforms. Anal. Chem. 2017, 89, 12360–12368. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.D.; McCombie, G.; Titman, C.M.; Griffin, J.L. A Matter of Fat: An Introduction to Lipidomic Profiling Methods. J. Chromatogr. B 2008, 871, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Taylan, O.; Cebi, N.; Tahsin Yilmaz, M.; Sagdic, O.; Bakhsh, A.A. Detection of Lard in Butter Using Raman Spectroscopy Combined with Chemometrics. Food Chem. 2020, 332, 127344. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, L.; Xu, Z.; Wang, Y.; Niu, C.; Yang, S. Liquid Chromatography Quadrupole Time-of-Flight Mass Spectrometry and Rapid Evaporative Ionization Mass Spectrometry Were Used to Develop a Lamb Authentication Method: A Preliminary Study. Foods 2020, 9, 1723. [Google Scholar] [CrossRef]
- Trivedi, D.K.; Hollywood, K.A.; Rattray, N.J.W.; Ward, H.; Trivedi, D.K.; Greenwood, J.; Ellis, D.I.; Goodacre, R. Meat, the Metabolites: An Integrated Metabolite Profiling and Lipidomics Approach for the Detection of the Adulteration of Beef with Pork. Analyst 2016, 141, 2155–2164. [Google Scholar] [CrossRef]
- Kumar, P.; Rani, A.; Singh, S.; Kumar, A. Recent Advances on DNA and Omics-Based Technology in Food Testing and Authentication: A Review. J. Food Saf. 2022, 42, e12986. [Google Scholar] [CrossRef]
- Mabood, F.; Boqué, R.; Alkindi, A.Y.; Al-Harrasi, A.; Al Amri, I.S.; Boukra, S.; Jabeen, F.; Hussain, J.; Abbas, G.; Naureen, Z.; et al. Fast Detection and Quantification of Pork Meat in Other Meats by Reflectance FT-NIR Spectroscopy and Multivariate Analysis. Meat Sci. 2020, 163, 108084. [Google Scholar] [CrossRef]
- Windarsih, A.; Rohman, A.; Riswanto, F.D.O.; Dachriyanus; Yuliana, N.D.; Bakar, N.K.A. The Metabolomics Approaches Based on LC-MS/MS for Analysis of Non-Halal Meats in Food Products: A Review. Agriculture 2022, 12, 984. [Google Scholar] [CrossRef]

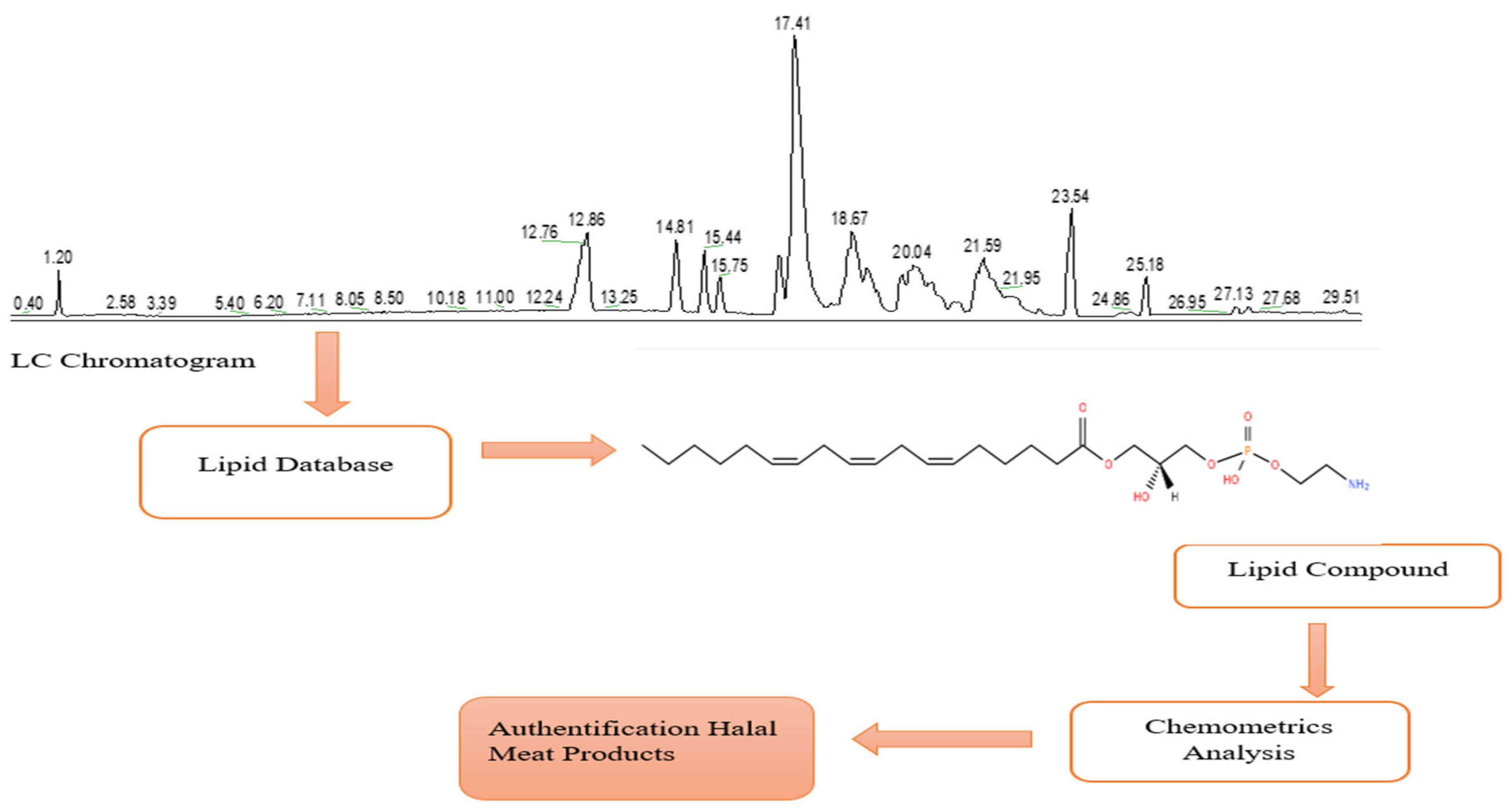
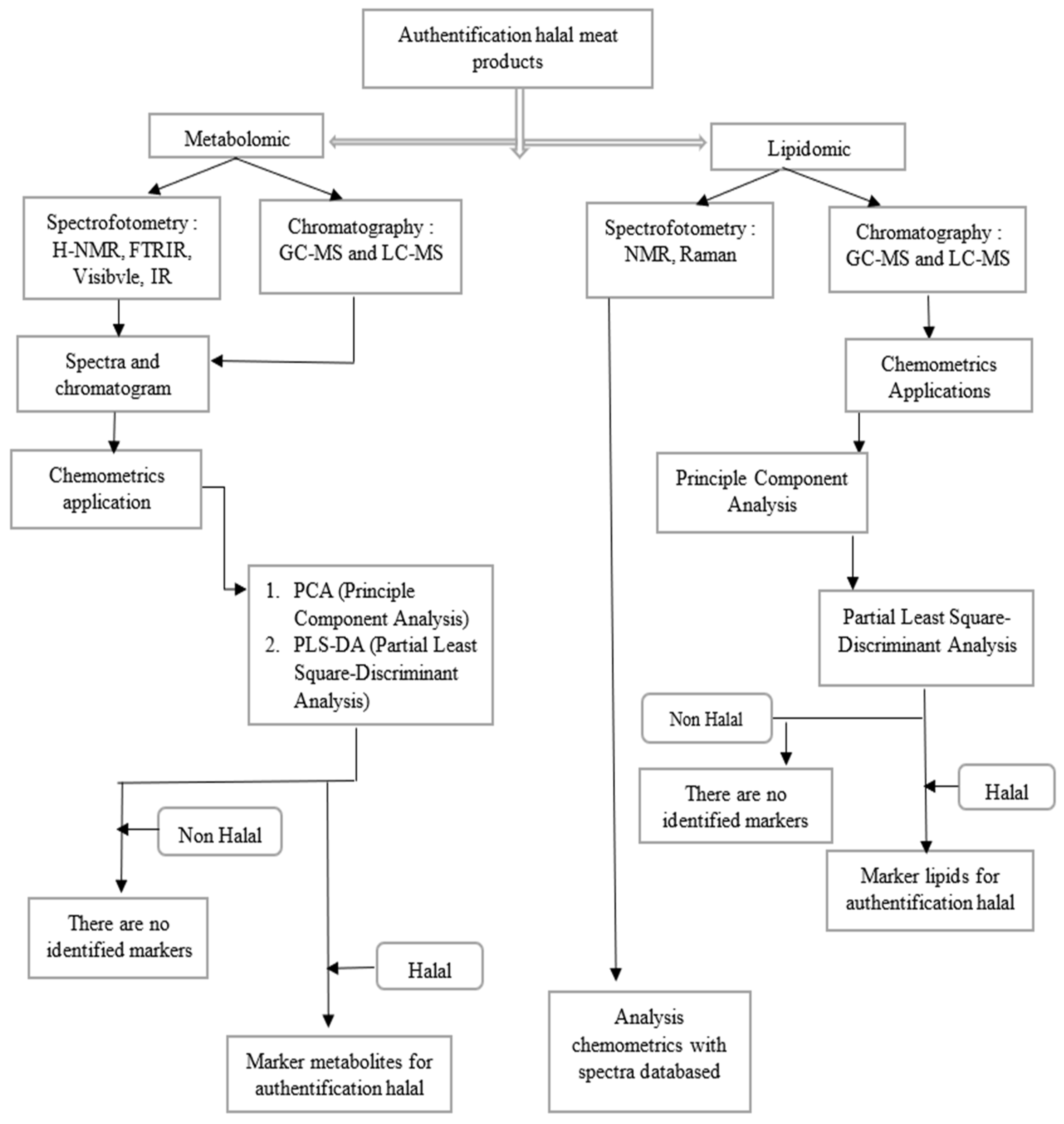
| No | Title | Objectives | Equipment | Chemometrics Techniques | Results | Ref. |
|---|---|---|---|---|---|---|
| 1 | Volatilomics for halal and non halal meatball authentication using solid-phase microextraction-gas chromatography-mass spectrometry | Meatball | GC-MS | PLS-DA | PLS-DA was able to distinguish volatile compounds in samples | [85] |
| 2 | The Feasibility of Two Handheld Spectrometers for Meat Specification Combined with Chemometric Method and Its Application for Halal Certification | Meat (lamb, beef, chicken, pork) | (Vis-NIR) and (NIR) spectroscopy | PLS-DA | PLS-DA was able to classify meat types with an accuracy value of 88.3% | [73] |
| 3 | 1H-NMR-Based Metabolomic: An Integrated Approach for the Detection of the Adulteration in Chicken, Chevon, Beef and Donkey Meat | Meat (chicken, chevon, beef and donkey) | 1H-NMR | PCA and OPLS-DA | PCA was able to identify 37 metabolites while OPLS-DA was able to distinguish the types of chicken, chevon, beef, and donkey meats | [86] |
| 4 | Authenticity Analysis of Beef Meatball Adulteration With Wild Boar Using FTIR Spectroscopy Combined With Chemometrics | Beef meatball and wild boar | FTIR | PCA and PLS | PCA would differentiate wild boar meatball and beef meatball products. PLS gave the value of determination coefficient (R2) of 0.9991 | [87] |
| 5 | Chemometrics-Assisted Shotgun Proteomics for Establishment of Potential Peptide Markers of Non-Halal Pork (Sus Scrofa) among Halal Beef and Chicken | Beef and chicken | LC-MS | PCA and OPLS-DA | PCA was able to reduce the data of metabolites that have similarities. OPLS-DA differentiated the results from PCA of beef and chicken based on the slaughter process | [88] |
| 6 | Discrimination between vegetable oil and animal fat by a metabolomics approach using gas chromatography-mass spectrometry combined with chemometrics | Lard | GC-MS | PCA | PCA was able to distinguish types of fat | [89] |
| 7 | Identification of Metabolomic Profile in Halal and Non-Halal Broiler Chicken Using Fourier-Transform Infrared Spectroscopy (FTIR) and Ultra High-Performance Liquid Chromatography-Time of Flight-Mass Spectrometry (UHPLC-TOF-MS) | Chicken broiler | FTIR and UHPLC-TOF-MS | PCA and PLS-DA | PCA was able to classify the metabolites found in broiler chickens based on the method of slaughter. PLS-DA distinguished non-halal chicken by the presence of high histidine and inosine | [84] |
| 8 | Untargeted-metabolomics different between poultry samples slaughtered with and without detaching spinal cord | Chicken meat | LC-ESI-MS/MS | PCA and OPLS-DA | PCA characterized metabolites based on the mode of slaughter. OPLS-DA was able to classify halal and non-halal samples | [90] |
| No | Title | Objectives | Equipment | Chemometrics Techniques | Results | Ref. |
|---|---|---|---|---|---|---|
| 1 | Detection of lard in butter using Raman spectroscopy combined with chemometrics | Lard | Spectroscopy Raman | HCA and PCA | HCA and PCA were successfully performed for the classification and discrimination of butter and lard-adulterated samples. | [105] |
| 2 | Liquid Chromatography Quadrupole Time-of-Flight Mass Spectrometry and Rapid Evaporative Ionization Mass Spectrometry Were Used to Develop a Lamb Authentication Method | Lamb | LC-QTOF-MS | PCA and OPLS-DA | PCA was performed to identify. OPLS-DA was carried out to separate the samples to the largest extent | [106] |
| 3 | The Metabolites: An Integrated metabolite Profiling and Lipidomic Approach for The Detection of The Adulteration of Beef With Pork. Analyst | Beef and pork | GC-MS | PCA and PLS-DA | PCA and PLS-DA was able to distinguish beef that contained pork | [107] |
| 4 | Multivariate Analysis Coupled with M-SVM Classification for Lard Adulteration Detection in Meat Mixtures of Beef, Lamb, and Chicken Using FTIR Spectroscopy | Lard | FTIR | PCA | PCA was able to classify with an accuracy value of 85% | [31] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maritha, V.; Harlina, P.W.; Musfiroh, I.; Gazzali, A.M.; Muchtaridi, M. The Application of Chemometrics in Metabolomic and Lipidomic Analysis Data Presentation for Halal Authentication of Meat Products. Molecules 2022, 27, 7571. https://doi.org/10.3390/molecules27217571
Maritha V, Harlina PW, Musfiroh I, Gazzali AM, Muchtaridi M. The Application of Chemometrics in Metabolomic and Lipidomic Analysis Data Presentation for Halal Authentication of Meat Products. Molecules. 2022; 27(21):7571. https://doi.org/10.3390/molecules27217571
Chicago/Turabian StyleMaritha, Vevi, Putri Widyanti Harlina, Ida Musfiroh, Amirah Mohd Gazzali, and Muchtaridi Muchtaridi. 2022. "The Application of Chemometrics in Metabolomic and Lipidomic Analysis Data Presentation for Halal Authentication of Meat Products" Molecules 27, no. 21: 7571. https://doi.org/10.3390/molecules27217571
APA StyleMaritha, V., Harlina, P. W., Musfiroh, I., Gazzali, A. M., & Muchtaridi, M. (2022). The Application of Chemometrics in Metabolomic and Lipidomic Analysis Data Presentation for Halal Authentication of Meat Products. Molecules, 27(21), 7571. https://doi.org/10.3390/molecules27217571







