Bimetallic Nanomaterials: A Promising Nanoplatform for Multimodal Cancer Therapy
Abstract
1. Introduction
2. Synthesis Methods of BMNs
2.1. Co-Reduction Method
2.2. Hydrothermal Method
2.3. Seed-Mediated Growth Method
2.4. Electrodeposition Method

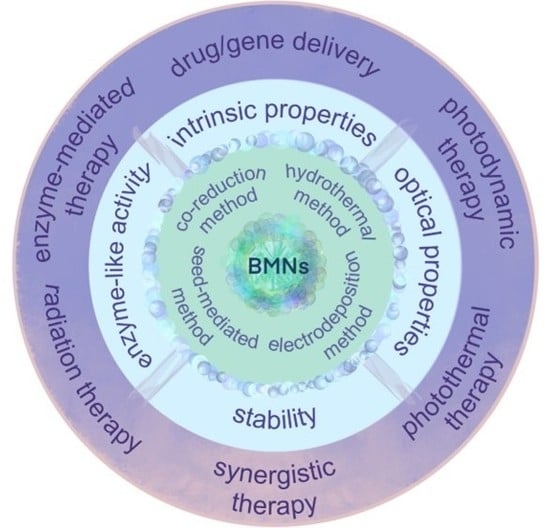
3. Unique Properties of BMNs Relevant to Cancer Therapy
3.1. Intrinsic Properties of BMNs

3.2. Optical Properties
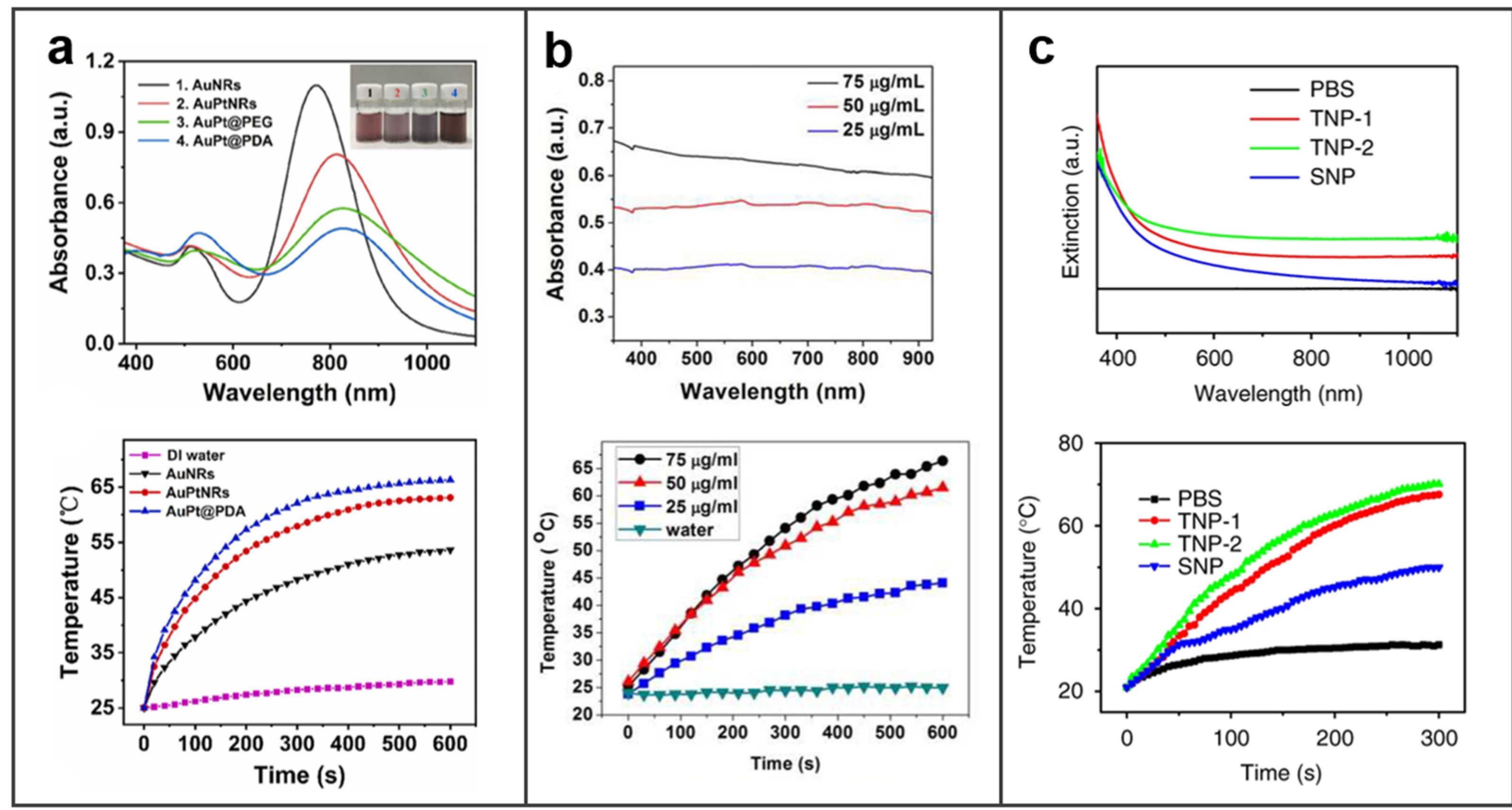
3.2.1. Photothermal Properties
3.2.2. Photocatalytic Properties
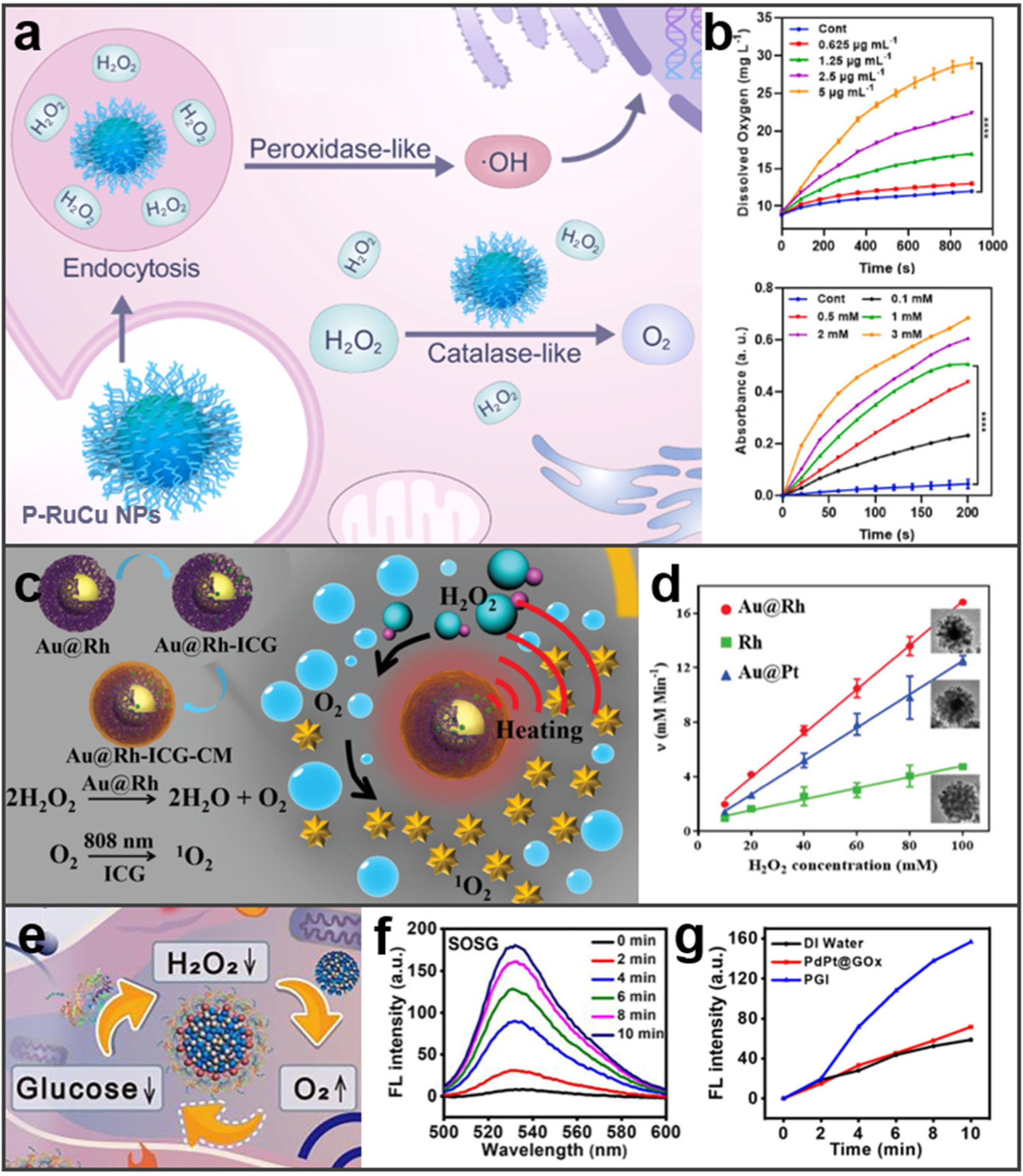
3.3. Enzyme-Like Activity
3.4. Stability

4. Application of BMNs in Cancer Therapy
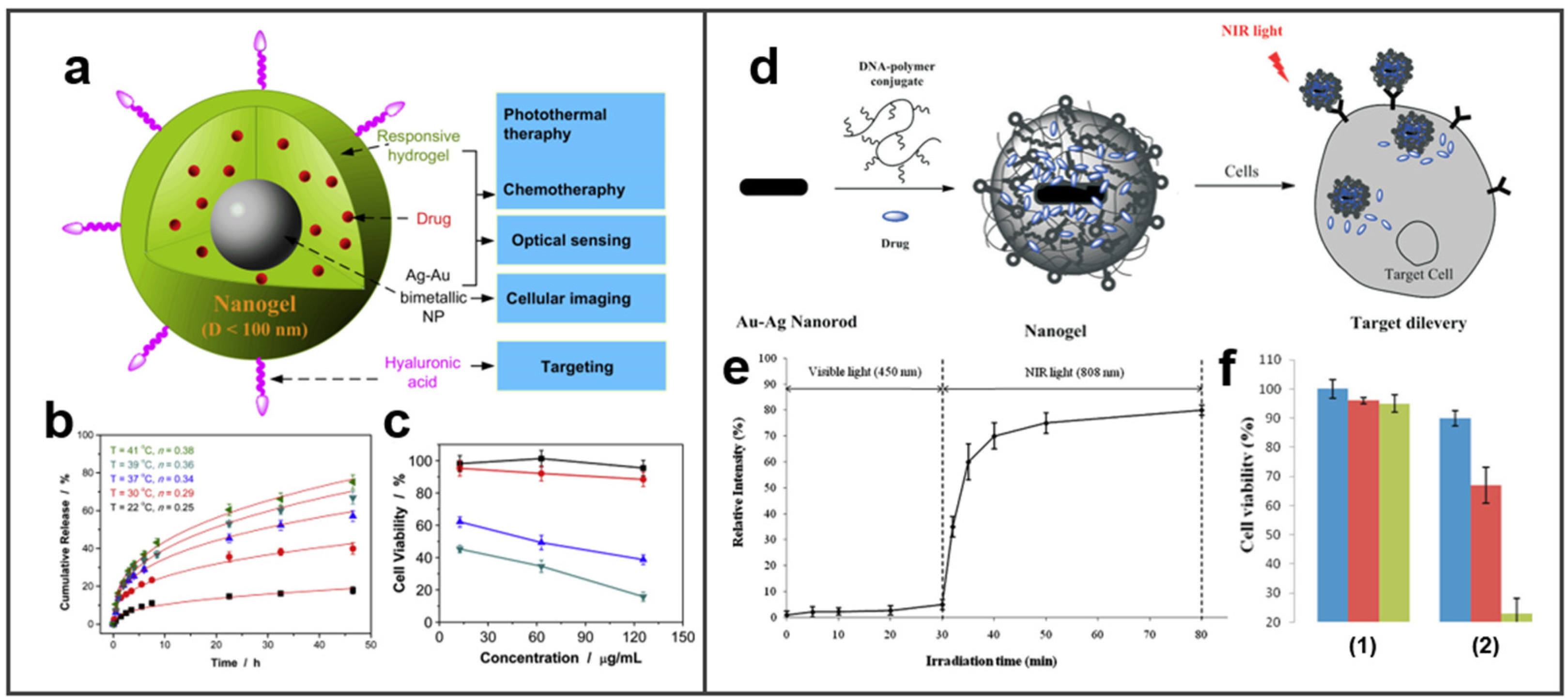
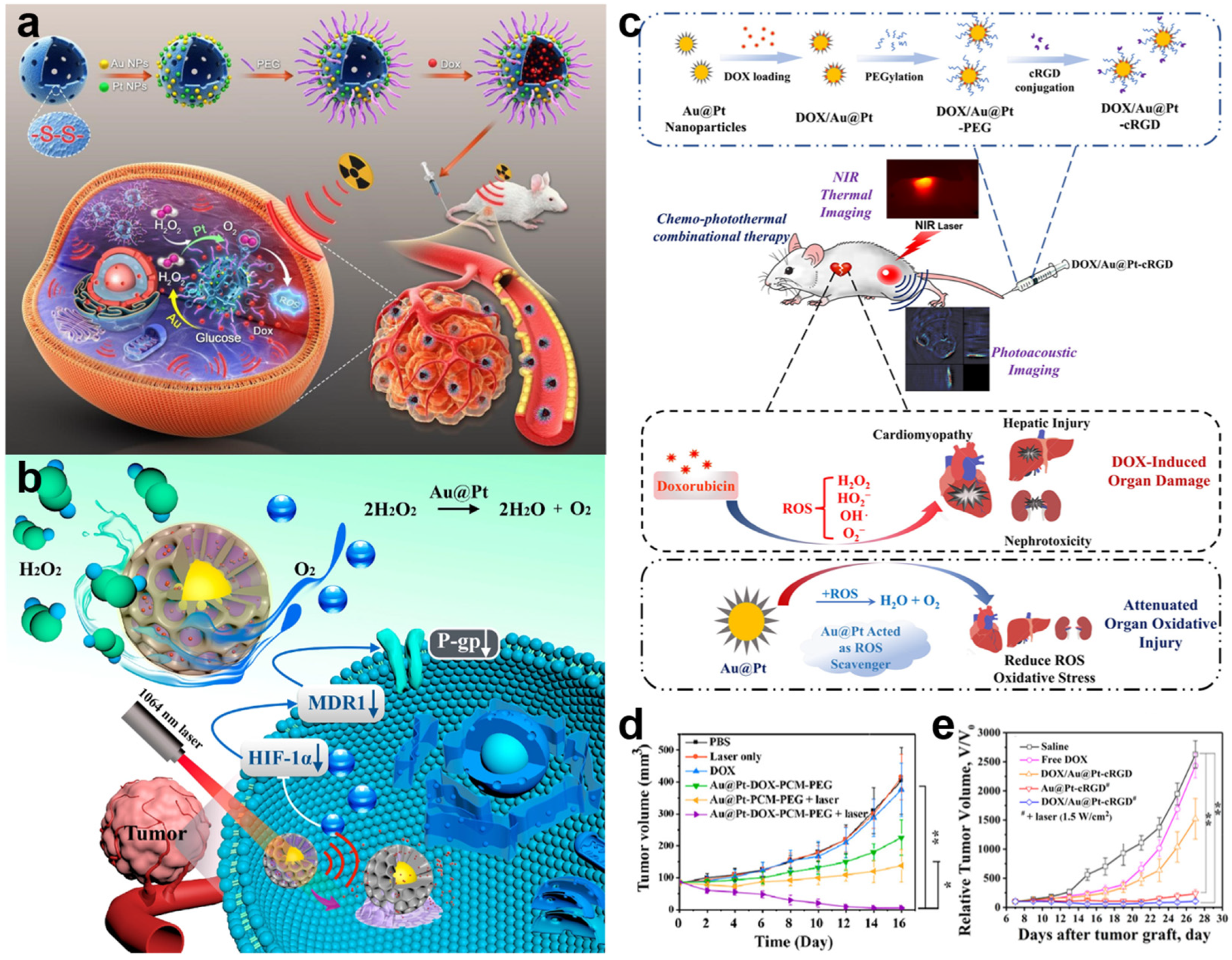
4.1. Drug/Gene Delivery
4.2. Enzyme-Mediated Tumor Therapy
4.3. Radiation Therapy
4.4. Photodynamics Therapy
4.5. Photothermal Therapy
4.6. Synergistic Therapy
5. Conclusions and Prospects
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BMNs | Bimetallic nanomaterials |
| MNs | Metallic nanomaterials |
| PTT | Photothermal therapy |
| PDT | Photodynamic therapy |
| GT | Gene therapy |
| SPR | Surface plasmon resonance |
| POD | Peroxidase |
| CAT | Catalase |
| SOD | Superoxide dismutase |
| AgCM | Monodispersed Ag cubic to mesh nanostructures |
| NPs | Nanoparticles |
| Au@Ag@Pt core@multishell | Ag-Pt double-shell on Au-core with a hollow-granular shell structure |
| ATP@Au-Cu NP | Adenosine 5′-triphosphate @Au-Cu nanoparticle |
| NDs | Nanodendrites |
| FCC | Face-centered cubic |
| AuNS | Gold nanostar |
| CP | A metal–drug coordination polymer |
| TPL | Two-photon luminescence |
| TNP | Cu-Pd alloy tetrapod nanoparticles |
| MOF | Metal–organic framework |
| CNDs | Carbon nanodots |
| NWs | Nanowires |
| NIR | Near-infrared |
| LSPR | Localized surface plasmon resonance |
| AuPtNRs | Dumbbell-shaped Au-Pt bimetallic nanorods |
| AuNRs | Gold nanorods |
| SNP | Spherical nanoparticle |
| CR | Charge separation |
| ROS | Reactive oxygen species |
| •O2− | Superoxide anion |
| O•− | Singlet oxygen |
| •OH | Hydroxyl radical |
| RhB | Rhodamine B |
| GOx | Glucose oxidase |
| GSH | Glutathione peroxidase |
| DFT | Density functional theory |
| Au@Rh-ICG-CM | Porous Au@Rh core–shell nanostructures loaded indocyanine green in the pores and coating with cancer cell membrane |
| US | Ultrasound |
| SDT | Sonodynamic therapy |
| PGI | PdPt@GOx/IR780 |
| Ico | Icosahedrons |
| CDT | Chemodynamic therapy |
| RT | Radiotherapy |
| FA | Folic acid |
| DOX | Doxorubicin |
| PEG | Polyethylene glycol |
| HA | Hyaluronic acid |
| TMZ | Targeting ligands and temozolomide |
| DOX-loaded sgc8c NGs | DOX-loaded sgc8c conjugated core–shell nanogels |
| BMOF-DMR | Bimetal metal-organic framework domino micro-reactor |
| E. coli | Escherichia coli |
| S. aureus | Staphylococcus aureus |
| S-AuNC | Snowflake-like Au nanocarriers |
| aPD-L1 | Anti-programmed death ligand 1 |
| DMSN | Dendritic mesoporous silica |
| 5-ALA/Au-Ag-PEG-Ab NC | Nanoconjugate combined with PEG functionalized Au-Ag nanoparticles, 5-aminolevulinic acid and antibodies |
| Au@Pt nanostructures | Au nanorods coated with a shell of Pt nanodots |
| HAAA-NUs | Hollow Au-Ag alloy nanourchins |
| CeO2/Au@Pt-PEG | ceria-loaded gold@platinum nanospheres modified with polyethylene glycol |
| PCN | Porphyrin metal–organic frameworks |
| CuCo NS | Single-site bimetallic (copper hexacyanocobaltate) nanosheet |
| HMOTP@Pt@Au@DOX | Hollow mesoporous organotantalum nanospheres modified with Au and Pt dual nanoenzymes |
| DOX/Au@Pt-cRGD | cRGD-modified DOX-loaded Au@Pt nanoparticles |
References
- Davis, M.E.; Chen, Z.; Shin, D.M. Nanoparticle therapeutics: An emerging treatment modality for cancer. Nat. Rev. Drug Discov. 2008, 7, 771–782. [Google Scholar] [CrossRef] [PubMed]
- Evans, E.R.; Bugga, P.; Asthana, V.; Drezek, R. Metallic nanoparticles for cancer immunotherapy. Mater. Today 2018, 21, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Wang, T.Y.; Bai, S.W.; Wan, Y.F.; Zhu, S.; Li, T.; Peng, N.; Qiu, T.; Liu, Y. Multifunction in one nanoparticle for anticancer therapy: Bowl-shaped Au@PDA yolk-shell NPs. ACS Appl. Mater. Interfaces 2022, 14, 27733–27742. [Google Scholar] [CrossRef] [PubMed]
- Couvreur, P. Nanoparticles in drug delivery: Past, present and future. Adv. Drug Deliv. Rev. 2013, 65, 21–23. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, C.; Yang, X.L.; Pu, K.Y. Photoactivatable protherapeutic nanomedicine for cancer. Adv. Mater. 2020, 32, 2002661. [Google Scholar] [CrossRef]
- Alamdari, S.G.; Amini, M.; Jalilzadeh, N.; Baradaran, B.; Mohammadzadeh, R.; Mokhtarzadeh, A.; Oroojalian, F. Recent advances in nanoparticle-based photothermal therapy for breast cancer. J. Control. Release 2022, 349, 269–303. [Google Scholar] [CrossRef]
- Song, H.Q.; Cai, Z.H.; Li, J.Y.; Xiao, H.H.; Qi, R.G.; Zheng, M.H. Light triggered release of a triple action porphyrin-cisplatin conjugate evokes stronger immunogenic cell death for chemotherapy, photodynamic therapy and cancer immunotherapy. J. Nanobiotechnol. 2022, 20, 329. [Google Scholar] [CrossRef]
- Ouyang, Z.J.; Gao, Y.; Yang, R.; Shen, M.W.; Shi, X.Y. Genetic engineering of dendritic cells using partially zwitterionic dendrimer-entrapped gold nanoparticles boosts efficient tumor immunotherapy. Biomacromolecules 2022, 23, 1326–1336. [Google Scholar] [CrossRef]
- Johnson, H.M.; Dasher, A.M.; Monahan, M.; Seifert, S.; Moreau, L.M. Mapping the effects of physical and chemical reduction parameters on local atomic distributions within bimetallic nanoparticles. Nanoscale 2022, 14, 4519–4530. [Google Scholar] [CrossRef]
- Yuan, Y.; Diao, S.C.; Ni, X.Y.; Zhang, D.; Yi, W.R.; Jian, C.; Hu, X.; Li, D.F.; Yu, A.X.; Zhou, W.; et al. Peptide-based semiconducting polymer nanoparticles for osteosarcoma-targeted NIR-II fluorescence/NIR-I photoacoustic dual-model imaging and photothermal/photodynamic therapies. J. Nanobiotechnol. 2022, 20, 44. [Google Scholar] [CrossRef]
- Zhai, J.M.; Gao, J.P.; Zhang, J.B.; Liu, D.C.; Gao, S.G.; Yan, Y.Y.; Zhang, K.K.; Cai, K.Y.; Yu, F.B.; Lin, M.P.; et al. Concave octahedral PtCu nanoframes mediated synergetic photothermal and chemodynamic tumor therapy. Chem. Eng. J. 2022, 442, 136172. [Google Scholar] [CrossRef]
- Liu, C.; Xing, J.; Akakuru, O.U.; Luo, L.J.; Sun, S.; Zou, R.F.; Yu, Z.S.; Fang, Q.L.; Wu, A.G. Nanozymes-engineered metal-organic frameworks for catalytic cascades-enhanced synergistic cancer therapy. Nano Lett. 2019, 19, 5674–5682. [Google Scholar] [CrossRef] [PubMed]
- Amoli-Diva, M.; Sadighi-Bonabi, R.; Pourghazi, K.; Hadilou, N. Tunable surface plasmon resonance-based remote actuation of bimetallic core-shell nanoparticle-coated stimuli responsive polymer for switchable chemo-photothermal synergistic cancer therapy. J. Pharm. Sci. 2018, 107, 2618–2627. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, D.; Mohan, L.; Alex, S.A.; Chandrasekaran, N.; Mukherjee, A. Bimetallic gold nanorods with enhanced biocorona formation for doxorubicin loading and sustained release. Biomater. Sci. 2019, 7, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Baijal, G.; Somashekar, R.; Iyer, S.; Nayak, V. One pot synthesis of PEGylated bimetallic gold-silver nanoparticles for imaging and radiosensitization of oral cancers. Int. J. Nanomed. 2021, 16, 7103–7121. [Google Scholar] [CrossRef] [PubMed]
- Sytwu, K.; Vadai, M.; Dionne, J.A. Bimetallic nanostructures: Combining plasmonic and catalytic metals for photocatalysis. Adv. Phys. X 2019, 4, 1619480. [Google Scholar] [CrossRef]
- He, J.H.; Ichinose, I.; Kunitake, T.; Nakao, A.; Shiraishi, Y.; Toshima, N. Facile fabrication of Ag-Pd bimetallic nanoparticles in ultrathin TiO2-gel films: Nanoparticle morphology and catalytic activity. J. Am. Chem. Soc. 2003, 125, 11034–11040. [Google Scholar] [CrossRef]
- Zhang, H.; Jin, M.S.; Xia, Y.N. Enhancing the catalytic and electrocatalytic properties of Pt-based catalysts by forming bimetallic nanocrystals with Pd. Chem. Soc. Rev. 2012, 41, 8035–8049. [Google Scholar] [CrossRef]
- Somu, P.; Paul, S. Protein assisted one pot controlled synthesis of monodispersed and multifunctional colloidal silver-gold alloy nanoparticles. J. Mol. Liq. 2019, 291, 111303. [Google Scholar] [CrossRef]
- Scaria, J.; Nidheesh, P.V.; Kumar, M.S. Synthesis and applications of various bimetallic nanomaterials in water and wastewater treatment. J. Environ. Manag. 2020, 259, 110011. [Google Scholar] [CrossRef]
- Filez, M.; Redekop, E.A.; Dendooven, J.; Ramachandran, R.K.; Solano, E.; Olsbye, U.; Weckhuysen, B.M.; Galvita, V.V.; Poelman, H.; Detavernier, C.; et al. Formation and functioning of bimetallic nanocatalysts: The power of X-ray probes. Angew. Chem. Int. Ed. 2019, 58, 13220–13230. [Google Scholar] [CrossRef] [PubMed]
- Gilroy, K.D.; Ruditskiy, A.; Peng, H.C.; Qin, D.; Xia, Y.N. Bimetallic nanocrystals: Syntheses, properties, and applications. Chem. Rev. 2016, 116, 10414–10472. [Google Scholar] [CrossRef] [PubMed]
- Yt, A.; Yc, A.; Mei, C.B.; Zls, C.; Bx, A.; Xbz, A. Peroxidase-like [emailprotected] nanozyme as an integrated nanosensor for Ag+ detection by lspr spectroscopy. Talanta 2021, 221, 121627. [Google Scholar]
- Chiu, T.H.; Liao, J.H.; Gam, F.; Wu, Y.Y.; Wang, X.P.; Kahlal, S.; Saillard, J.Y.; Liu, C.W. Hydride-containing eight-electron Pt/Ag superatoms: Structure, bonding, and multi-nmr studies. J. Am. Chem. Soc. 2022, 144, 10599–10607. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhang, A.Q.; Li, H.J.; Qian, D.J.; Chen, M. Synthesis, study, and discrete dipole approximation simulation of Ag-Au bimetallic nanostructures. Nanoscale Res. Lett. 2016, 11, 1701751. [Google Scholar] [CrossRef]
- Liu, Y.; Qing, Y.L.; Jing, L.C.; Zou, W.T.; Guo, R. Platinum-copper bimetallic colloid nanoparticle cluster nanozymes with multiple enzyme-like activities for scavenging reactive oxygen species. Langmuir 2021, 37, 7364–7372. [Google Scholar] [CrossRef]
- Joo, J.H.; Kim, B.H.; Lee, J.S. Synthesis of gold nanoparticle-embedded silver cubic mesh nanostructures using AgCl nanocubes for plasmonic photocatalysis. Small 2017, 13, 1701751. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Lee, H.J.; Dao, A.; Kasai, H.; Teranishi, R.; Kaneko, K. Formation of multishell Au@Ag@Pt nanoparticles by coreduction method: A microscopic study. Mater. Today Chem. 2021, 21, 100515. [Google Scholar] [CrossRef]
- Radwan, A.B.; Paramparambath, S.; Cabibihan, J.J.; Al-Ali, A.K.; Kasak, P.; Shakoor, R.A.; Malik, R.A.; Mansour, S.A.; Sadasivuni, K.K. Superior non-invasive glucose sensor using bimetallic CuNi nanospecies coated mesoporous carbon. Biosensors 2021, 11, 463. [Google Scholar] [CrossRef]
- Motlak, M.; Barakat, N.; El-Deen, A.G.; Hamza, A.M.; Obaid, M.; Yang, O.B.; Akhtar, M.S.; Khalil, K.A. NiCu bimetallic nanoparticle-decorated graphene as novel and cost-effective counter electrode for dye-sensitized solar cells and electrocatalyst for methanol oxidation. Appl. Catal. A Gen. 2015, 501, 41–47. [Google Scholar] [CrossRef]
- Gai, C.; Zhang, F.; Yang, T.X.; Liu, Z.G.; Jiao, W.T.; Peng, N.N.; Liu, T.T.; Lang, Q.Q.; Xia, Y. Hydrochar supported bimetallic Ni-Fe nanocatalysts with tailored composition, size and shape for improved biomass steam reforming performance. Green Chem. 2018, 20, 2788–2800. [Google Scholar] [CrossRef]
- Wei, L.G.; Chen, W.; Jia, C.Y.; Wang, D.; Li, M.; Dong, Y.L.; Song, W.N.; Liu, L.L.; Yang, Y.L. Facile synthesis of CoNi bimetallic nanoparticle decorated reduced graphene oxide as efficient and low-cost counter electrode for dye-sensitized solar cells. J. Nanosci. Nanotechnol. 2019, 19, 7790–7798. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.J.; Cheng, Y.R.; Chen, M.T.; Ye, Y.H.; Liu, Y.X. Design of low-loaded nire bimetallic catalyst on N-doped mesoporous carbon for highly selective deoxygenation of oleic acid to n-heptadecane. Korean J. Chem. Eng. 2022, 39, 1753–1761. [Google Scholar] [CrossRef]
- Xu, B.; Zhang, Z.C.; Wang, X. Formamide: An efficient solvent to synthesize water-soluble and sub-ten-nanometer nanocrystals. Nanoscale 2013, 5, 4495–4505. [Google Scholar] [CrossRef]
- Habibi, A.H.; Hayes, R.E.; Semagina, N. Evaluation of hydrothermal stability of encapsulated PdPt@SiO2 catalyst for lean CH4 combustion. Appl. Catal. A Gen. 2018, 556, 129–136. [Google Scholar] [CrossRef]
- Shen, L.Q.; Zhou, X.; Wang, A.L.; Yin, H.B.; Yin, H.X.; Cui, W.J. Hydrothermal conversion of high-concentrated glycerol to lactic acid catalyzed by bimetallic CuAux (x = 0.01–0.04) nanoparticles and their reaction kinetics. RSC Adv. 2017, 7, 30725–30739. [Google Scholar] [CrossRef]
- Yuan, M.; Liu, A.P.; Zhao, M.; Dong, W.J.; Zhao, T.Y.; Wang, J.J.; Tang, W.H. Bimetallic PdCu nanoparticle decorated three-dimensional graphene hydrogel for non-enzymatic amperometric glucose sensor. Sens. Actuator B Chem. 2014, 190, 707–714. [Google Scholar] [CrossRef]
- Feng, J.; Xu, D.D.; Yang, F.; Chen, J.X.; Wu, C.; Yin, Y.D. Surface engineering and controlled ripening for seed-mediated growth of Au islands on Au nanocrystals. Angew. Chem. Int. Ed. 2021, 60, 16958–16964. [Google Scholar] [CrossRef]
- Scala, A.; Neri, G.; Micale, N.; Cordaro, M.; Piperno, A. State of the art on green route synthesis of gold/silver bimetallic nanoparticles. Molecules 2022, 27, 1134. [Google Scholar] [CrossRef]
- Orouji, A.; Abbasi-Moayed, S.; Ghasemi, F.; Hormozi-Nezhad, M.R. A wide-range pH indicator based on colorimetric patterns of gold@silver nanorods. Sens. Actuator B Chem. 2022, 358, 131479. [Google Scholar] [CrossRef]
- Zhang, Q.F.; Jing, H.; Li, G.G.; Lin, Y.; Blom, D.A.; Wang, H. Intertwining roles of silver ions, surfactants, and reducing agents in gold nanorod overgrowth: Pathway switch between silver underpotential deposition and gold-silver codeposition. Chem. Mater. 2016, 28, 2728–2741. [Google Scholar] [CrossRef]
- Xue, C.; Millstone, J.E.; Li, S.Y.; Mirkin, C.A. Plasmon-driven synthesis of triangular core-shell nanoprisms from gold seeds. Angew. Chem. Int. Ed. 2007, 46, 8436–8439. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.Y.; Li, W.Y.; Cho, E.C.; Li, Z.Y.; Yu, T.K.; Zeng, J.; Xie, Z.X.; Xia, Y.N. Au@Ag core-shell nanocubes with finely tuned and well-controlled sizes, shell thicknesses, and optical properties. ACS Nano 2010, 4, 6725–6734. [Google Scholar] [CrossRef] [PubMed]
- Tahsini, N.; Yang, A.C.; Streibel, V.; Werghi, B.; Goodman, E.D.; Aitbekova, A.; Bare, S.R.; Li, Y.J.; Abild-Pedersen, F.; Cargnello, M. Colloidal platinum-copper nanocrystal alloy catalysts surpass platinum in low-temperature propene combustion. J. Am. Chem. Soc. 2022, 144, 1612–1621. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wu, Z.; Li, F.X.; Gan, X.P.; Tao, J.M.; Yi, J.H.; Liu, Y.C. A combination of enhanced mechanical and electromagnetic shielding properties of carbon nanotubes reinforced Cu-Ni composite foams. Nanomaterials 2021, 11, 1772. [Google Scholar] [CrossRef]
- Simunkova, H.; Lednicky, T.; Whitehead, A.H.; Kalina, L.; Simunek, P.; Hubalek, J. Tantalum-based nanotube arrays via porous-alumina-assisted electrodeposition from ionic liquid: Formation and electrical characterization. Appl. Surf. Sci. 2021, 548, 149264. [Google Scholar] [CrossRef]
- Katarkar, A.S.; Pingale, A.D.; Belgamwar, S.U.; Bhaumik, S. Experimental study of pool boiling enhancement using a two-step electrodeposited Cu-GNPs nanocomposite porous surface with R-134a. J. Heat Transf. Trans. ASME 2021, 143, 121601. [Google Scholar] [CrossRef]
- Sanguansap, Y.; Karn-Orachai, K.; Laocharoensuk, R. Tailor-made porous striped gold-silver nanowires for surface enhanced raman scattering based trace detection of beta-hydroxybutyric acid. Appl. Surf. Sci. 2020, 500, 144049. [Google Scholar] [CrossRef]
- Jia, H.Y.; Gao, P.C.; Ma, H.M.; Wu, D.; Du, B.; Wei, Q. Preparation of Au-Pt nanostructures by combining top-down with bottom-up strategies and application in label-free electrochemical immunosensor for detection of nmp22. Bioelectrochemistry 2015, 101, 22–27. [Google Scholar] [CrossRef]
- Farquhar, A.K.; Brooksby, P.A.; Dryfe, R.; Downard, A.J. Controlled electrodeposition of gold nanoparticles onto copper-supported few-layer graphene in non-aqueous conditions. Electrochim. Acta 2017, 237, 54–60. [Google Scholar] [CrossRef]
- Chebrolu, V.T.; Balakrishnan, B.; Raman, V.; Cho, I.; Bak, J.S.; Prabakar, K.; Kim, H.J. Co-electrodeposition of NiCu(OH)(2)@Ni-Cu-Se hierarchical nanoparticle structure for supercapacitor application with enhanced performance. Appl. Surf. Sci. 2020, 506, 145015. [Google Scholar] [CrossRef]
- Wang, C.Y.; Chen, D.P.; Sang, X.H.; Unocic, R.R.; Skrabalak, S.E. Size-dependent disorder-order transformation in the synthesis of monodisperse intermetallic PdCu nanocatalysts. ACS Nano 2016, 10, 6345–6353. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.L.; Ai, K.L.; Liu, J.H.; Deng, M.; He, Y.Y.; Lu, L.H. Dopamine-melanin colloidal nanospheres: An efficient near-infrared photothermal therapeutic agent for in vivo cancer therapy. Adv. Mater. 2013, 25, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Lei, Q.; Qiu, W.X.; Liu, L.H.; Zheng, D.W.; Fan, J.X.; Rong, L.; Sun, Y.X.; Zhang, X.Z. Mitochondria-targeting “nanoheater” for enhanced photothermal/chemo-therapy. Biomaterials 2017, 117, 92–104. [Google Scholar] [CrossRef]
- Salimi, M.; Shokrgozar, M.A.; Hamid, D.H.; Vossoughi, M. Photothermal properties of two-dimensional molybdenum disulfide (mos2) with nanoflower and nanosheet morphology. Mater. Res. Bull. 2022, 152, 111837. [Google Scholar] [CrossRef]
- Mirabi, A.; Gilaki, M.; Ghorbani, H.; Rad, A.S.; Binaeian, E. Thermal and physical properties of Pt nanorods. Asian J. Chem. 2011, 23, 5149–5152. [Google Scholar]
- Deng, X.R.; Liang, S.; Cai, X.C.; Huang, S.S.; Cheng, Z.Y.; Shi, Y.S.; Pang, M.L.; Ma, P.A.; Lin, J. Yolk-shell structured Au nanostar@metal-organic framework for synergistic chemo-photothermal therapy in the second near-infrared window. Nano Lett. 2019, 19, 6772–6780. [Google Scholar] [CrossRef]
- Li, B.; Wang, Y.; He, J. Gold nanorods-based smart nanoplatforms for synergic thermotherapy and chemotherapy of tumor metastasis. ACS Appl. Mater. Interfaces 2019, 11, 7800–7811. [Google Scholar] [CrossRef]
- Song, Q.; Liu, Y.; Zhang, P.; Feng, W.; Shi, S.; Zhou, N.; Chu, X.; Shen, J. Au–cu bimetallic nanostructures for photothermal antibacterial and wound healing promotion. ACS Appl. Nano Mater. 2022, 5, 8621–8630. [Google Scholar] [CrossRef]
- Li, Y.J.; Ding, W.C.; Li, M.R.; Xia, H.B.; Wang, D.Y.; Tao, X.T. Synthesis of core-shell au-pt nanodendrites with high catalytic performance via overgrowth of platinum on in situ gold nanoparticles. J. Mater. Chem. A 2015, 3, 368–376. [Google Scholar] [CrossRef]
- Li, M.Y.; Li, L.L.; Zhan, C.Y.; Kohane, D.S. Core-shell nanostars for multimodal therapy and imaging. Theranostics 2016, 6, 2306–2313. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, X.; Zhu, M.; Lin, G.H.; Liu, J.; Zhou, Z.F.; Tian, X.; Pan, Y. PEGylated Au@Pt nanodendrites as novel theranostic agents for computed tomography imaging and photothermal/radiation synergistic therapy. ACS Appl. Mater. Interfaces 2017, 9, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Sha, R.; Zhang, L.; Zhang, W.B.; Jin, P.P.; Xu, W.G.; Ding, J.X.; Lin, J.; Qian, J.; Yao, G.Y.; et al. Harnessing copper-palladium alloy tetrapod nanoparticle-induced pro-survival autophagy for optimized photothermal therapy of drug-resistant cancer. Nat. Commun. 2018, 9, 4236. [Google Scholar] [CrossRef] [PubMed]
- Klebowski, B.; Stec, M.; Depciuch, J.; Gałuszka, A.; Pajor-Swierzy, A.; Baran, J.; Parlinska-Wojtan, M. Gold-decorated platinum and palladium nanoparticles as modern nanocomplexes to improve the effectiveness of simulated anticancer proton therapy. Pharmaceutics 2021, 13, 1726. [Google Scholar] [CrossRef] [PubMed]
- Depciuch, J.; Stec, M.; Klebowski, B.; Baran, J.; Parlinska-Wojtan, M. Platinum-gold nanoraspberries as effective photosensitizer in anticancer photothermal therapy. J. Nanobiotechnol. 2019, 17, 107. [Google Scholar] [CrossRef]
- Zhang, J.; Feng, Y.H.; Mi, J.L.; Shen, Y.T.; Tu, Z.G.; Liu, L. Photothermal lysis of pathogenic bacteria by platinum nanodots decorated gold nanorods under near infrared irradiation. J. Hazard. Mater. 2018, 342, 121–130. [Google Scholar] [CrossRef]
- Pavithra, S.; Jothi, V.K.; Rajaram, A.; Ingavale, S.; Natarajan, A. A facile synthesis of a fusiform-shaped three-dimensional Co/Mn@cnds-MOF nanocomposite as an efficient electrocatalyst for oxygen evolution reaction in alkaline medium. Energy Fuels 2022, 36, 6409–6419. [Google Scholar] [CrossRef]
- Choi, J.R.; Oh, S.J.; Ju, H.; Cheon, J. Massive fabrication of free-standing one-dimensional Co/Pt nanostructures and modulation of ferromagnetism via a programmable barcode layer effect. Nano Lett. 2005, 5, 2179–2183. [Google Scholar] [CrossRef]
- Hada, A.; Potara, M.; Astilean, S.; Cordaro, A.; Neri, G.; Malanga, M.; Nostro, A.; Mazzaglia, A.; Scala, A.; Piperno, A. Linezolid nanoantiobiotics and sers-nanotags based on polymeric cyclodextrin bimetallic core-shell nanoarchitectures. Carbohydr. Polym. 2022, 293, 119736. [Google Scholar] [CrossRef]
- Wang, J.; Sun, J.; Hu, W.; Wang, Y.; Chou, T.; Zhang, B.; Zhang, Q.; Ren, L.; Wang, H. A porous au@rh bimetallic core–shell nanostructure as an h2o2-driven oxygenerator to alleviate tumor hypoxia for simultaneous bimodal imaging and enhanced photodynamic therapy. Adv. Mater. 2020, 32, 2001862. [Google Scholar] [CrossRef]
- Liz-Marzan, L.M.; Murphy, C.J.; Wang, J.F. Nanoplasmonics. Chem. Soc. Rev. 2014, 43, 3820–3822. [Google Scholar] [CrossRef] [PubMed]
- Linic, S.; Aslam, U.; Boerigter, C.; Morabito, M. Photochemical transformations on plasmonic metal nanoparticles. Nat. Mater. 2015, 14, 744. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.H.; El-Sayed, I.H.; Qian, W.; El-Sayed, M.A. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc. 2006, 128, 2115–2120. [Google Scholar] [CrossRef] [PubMed]
- Duan, Q.Q.; Wang, J.L.; Zhang, B.Y.; Wang, X.Y.; Xue, J.J.; Zhang, W.D.; Sang, S.B. Polydopamine coated Au-Pt nanorods: Enhanced photothermal properties and efficient reactive oxygen scavengers. Colloid Surf. B Biointerfaces 2022, 210, 112247. [Google Scholar] [CrossRef] [PubMed]
- Kalisman, P.; Houben, L.; Aronovitch, E.; Kauffmann, Y.; Bar-Sadan, M.; Amirav, L. The golden gate to photocatalytic hydrogen production. J. Mater. Chem. A 2015, 3, 19679–19682. [Google Scholar] [CrossRef]
- Mizukoshi, Y.; Sato, K.; Konno, T.J.; Masahashi, N. Dependence of photocatalytic activities upon the structures of Au/Pd bimetallic nanoparticles immobilized on TiO2 surface. Appl. Catal. B Environ. 2010, 94, 248–253. [Google Scholar] [CrossRef]
- Zheng, Z.K.; Tachikawa, T.; Majima, T. Single-particle study of Pt-modified Au nanorods for plasmon-enhanced hydrogen generation in visible to near-infrared region. J. Am. Chem. Soc. 2014, 136, 6870–6873. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Wang, L.L.; Kong, X.Y.; Jiang, H.Y.; Zhang, F.; Shi, J.S. Novel Ag-Cu bimetallic alloy decorated near-infrared responsive three-dimensional rod-like architectures for efficient photocatalytic water purification. J. Colloid Interface Sci. 2018, 522, 29–39. [Google Scholar] [CrossRef]
- Bian, H.D.; Nguyen, N.T.; Yoo, J.; Hejazi, S.; Mohajernia, S.; Muller, J.; Spiecker, E.; Tsuchiya, H.; Tomanec, O.; Sanabria-Arenas, B.E.; et al. Forming a highly active, homogeneously alloyed AuPt co-catalyst decoration on TiO2 nanotubes directly during anodic growth. ACS Appl. Mater. Interfaces 2018, 10, 18220–18226. [Google Scholar] [CrossRef]
- Xiao, J.D.; Han, L.L.; Luo, J.; Yu, S.H.; Jiang, H.L. Integration of plasmonic effects and schottky junctions into metal-organic framework composites: Steering charge flow for enhanced visible-light photocatalysis. Angew. Chem. Int. Edit. 2018, 57, 1103–1107. [Google Scholar] [CrossRef]
- Ding, J.J.; Li, X.Y.; Chen, L.; Zhang, X.; Tian, X.Y. Photocatalytic hydrogen production over plasmonic AuCu/cain2s4 composites with different AuCu atomic arrangements. Appl. Catal. B Environ. 2018, 224, 322–329. [Google Scholar] [CrossRef]
- Cybula, A.; Priebe, J.B.; Pohl, M.M.; Sobczak, J.W.; Schneider, M.; Zielhiska-Jurek, A.; Bruckner, A.; Zaleska, A. The effect of calcination temperature on structure and photocatalytic properties of Au/Pd nanoparticles supported on TiO2. Appl. Catal. B Environ. 2014, 152, 202–211. [Google Scholar] [CrossRef]
- Jiang, T.T.; Jia, C.C.; Zhang, L.C.; He, S.R.; Sang, Y.H.; Li, H.D.; Li, Y.Q.; Xu, X.H.; Liu, H. Gold and gold-palladium alloy nanoparticles on heterostructured TiO2 nanobelts as plasmonic photocatalysts for benzyl alcohol oxidation. Nanoscale 2015, 7, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.Q.; Xu, H.T.; Su, Y.Q.; Xu, Z.L.; Wang, K.F.; Wang, W.Z. Noble metal (Pt, Au@Pd) nanoparticles supported on metal organic framework (MOF-74) nanoshuttles as high-selectivity CO2 conversion catalysts. J. Catal. 2019, 370, 70–78. [Google Scholar] [CrossRef]
- Misra, M.; Singh, N.; Gupta, R.K. Enhanced visible-light-driven photocatalytic activity of Au@Ag core-shell bimetallic nanoparticles immobilized on electrospun TiO2 nanofibers for degradation of organic compounds. Catal. Sci. Technol. 2017, 7, 570–580. [Google Scholar] [CrossRef]
- Tanaka, A.; Hashimoto, K.; Kominami, H. Gold and copper nanoparticles supported on cerium(IV) oxide-a photocatalyst mineralizing organic acids under red light irradiation. ChemCatChem 2011, 3, 1619–1623. [Google Scholar] [CrossRef]
- Zielinska-Jurek, A.; Kowalska, E.; Sobczak, J.W.; Lisowski, W.; Ohtani, B.; Zaleska, A. Preparation and characterization of monometallic (Au) and bimetallic (Ag/Au) modified-titania photocatalysts activated by visible light. Appl. Catal. B Environ. 2011, 101, 504–514. [Google Scholar] [CrossRef]
- Luneau, M.; Guan, E.J.; Chen, W.; Foucher, A.C.; Marcella, N.; Shirman, T.; Verbart, D.; Aizenberg, J.; Aizenberg, M.; Stach, E.A.; et al. Enhancing catalytic performance of dilute metal alloy nanomaterials. Commun. Chem. 2020, 3, 46. [Google Scholar] [CrossRef]
- Venkatesan, P.; Santhanalakshmi, J. Kinetics of oxidation of l-leucine by mono- and bimetallic gold and silver nanoparticles in hydrogen peroxide solution. Chin. J. Catal. 2012, 33, 1306–1311. [Google Scholar] [CrossRef]
- Sun, H.H.; Qing, T.P.; He, X.X.; Shangguan, J.F.; Jia, R.C.; Bu, H.C.; Huang, J.; Wang, K.M. Rapid synthesis of Au/Ag bimetallic nanoclusters with highly biochemical stability and its applications for temperature and ratiometric pH sensing. Anal. Chim. Acta 2019, 1070, 88–96. [Google Scholar] [CrossRef]
- Nosrati, H.; Baghdadchi, Y.; Abbasi, R.; Barsbay, M.; Ghaffarlou, M.; Abhari, F.; Mohammadi, A.; Kavetskyy, T.; Bochani, S.; Rezaeejam, H.; et al. Iron oxide and gold bimetallic radiosensitizers for synchronous tumor chemoradiation therapy in 4t1 breast cancer murine model. J. Mater. Chem. B 2021, 9, 4510–4522. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Cheng, L.; Yang, N.L.; Betzer, O.; Feng, L.Z.; Zhou, Q.; Li, Y.G.; Chen, R.H.; Popovtzer, R.; Liu, Z. Ultrasmall oxygen-deficient bimetallic oxide mnwox nanoparticles for depletion of endogenous GSH and enhanced sonodynamic cancer therapy. Adv. Mater. 2019, 31, 1900730. [Google Scholar] [CrossRef] [PubMed]
- Aghahosseini, H.; Rezaei, S.; Abdolahnjadian, D.; Maleki, M.; Ramazani, A. Ag-Pd alloy immobilized on semi-heterogeneous support as a novel highly efficient artificial nitroreductase: Experimental design optimization and kinetic study. Catal. Lett. 2021, 151, 1262–1272. [Google Scholar] [CrossRef]
- Gong, F.; Yang, N.; Wang, Y.; Zhuo, M.; Zhao, Q.; Wang, S.; Li, Y.; Liu, Z.; Chen, Q.; Cheng, L. Oxygen-deficient bimetallic oxide fewox nanosheets as peroxidase-like nanozyme for sensing cancer via photoacoustic imaging. Small 2020, 16, 2003496. [Google Scholar] [CrossRef] [PubMed]
- Niu, B.Y.; Liao, K.X.; Zhou, Y.X.; Wen, T.; Quan, G.L.; Pan, X.; Wu, C.B. Application of glutathione depletion in cancer therapy: Enhanced ros-based therapy, ferroptosis, and chemotherapy. Biomaterials 2021, 277, 121110. [Google Scholar] [CrossRef]
- Hu, B.; Xiao, X.; Chen, P.; Qian, J.; Yuan, G.; Ye, Y.; Zeng, L.; Zhong, S.; Wang, X.; Qin, X.; et al. Enhancing anti-tumor effect of ultrasensitive bimetallic rucu nanoparticles as radiosensitizers with dual enzyme-like activities. Biomaterials 2022, 290, 121811. [Google Scholar] [CrossRef]
- Zhou, Z.; Huang, J.; Zhang, Z.; Zhang, L.; Cao, Y.; Xu, Z.; Kang, Y.; Xue, P. Bimetallic PdPt-based nanocatalysts for photothermal-augmented tumor starvation and sonodynamic therapy in NIR-II biowindow assisted by an oxygen self-supply strategy. Chem. Eng. J. 2022, 435, 135085. [Google Scholar] [CrossRef]
- Chookajorn, T.; Murdoch, H.A.; Schuh, C.A. Design of stable nanocrystalline alloys. Science 2012, 337, 951–954. [Google Scholar] [CrossRef]
- Lopez, G.E.; Freeman, D.L. A study of low temperature heat capacity anomalies in bimetallic alloy clusters using j-walking Monte Carlo methods. J. Chem. Phys. 1993, 98, 1428. [Google Scholar] [CrossRef]
- Larsen, P.M.; Jacobsen, K.W.; Schiotz, J. Rich ground-state chemical ordering in nanoparticles: Exact solution of a model for Ag-Au clusters. Phys. Rev. Lett. 2018, 120, 256101. [Google Scholar] [CrossRef]
- Dean, J.; Cowan, M.J.; Estes, J.; Ramadan, M.; Mpourmpakis, G. Rapid prediction of bimetallic mixing behavior at the nanoscale. ACS Nano 2020, 14, 8171–8180. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.S.; Wang, G.; Qian, P.; Su, Y.J. Element segregation and thermal stability of Ni-Pd nanoparticles. J. Mater. Sci. 2022, 57, 7384–7399. [Google Scholar] [CrossRef]
- Huang, R.; Shao, G.F.; Zhang, Y.; Wen, Y.H. Thermal stability of platinum-cobalt bimetallic nanoparticles: Chemically disordered alloys, ordered intermetallics, and core-shell structures. ACS Appl. Mater. Interfaces 2017, 9, 12486–12493. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.F.; Jackson, G.S.; Eichhorn, B.W. Tuning the co-tolerance of Pt-Fe bimetallic nanoparticle electrocatalysts through architectural control. Energy Environ. Sci. 2011, 4, 1900–1903. [Google Scholar] [CrossRef]
- Yao, Q.F.; Feng, Y.; Fung, V.; Yu, Y.; Jiang, D.E.; Yang, J.; Xie, J.P. Precise control of alloying sites of bimetallic nanoclusters via surface motif exchange reaction. Nat. Commun. 2017, 8, 1555. [Google Scholar] [CrossRef]
- Maney, V.; Singh, M. The synergism of platinum-gold bimetallic nanoconjugates enhances 5-fluorouracil delivery in vitro. Pharmaceutics 2019, 11, 439. [Google Scholar] [CrossRef]
- Barretina, J.; Caponigro, G.; Stransky, N.; Venkatesan, K.; Margolin, A.A.; Kim, S.; Wilson, C.J.; Lehar, J.; Kryukov, G.V.; Sonkin, D.; et al. The cancer cell line encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 2012, 483, 603–607. [Google Scholar] [CrossRef]
- Al-Lazikani, B.; Banerji, U.; Workman, P. Combinatorial drug therapy for cancer in the post-genomic era. Nat. Biotechnol. 2012, 30, 679–691. [Google Scholar] [CrossRef]
- Jia, Y.P.; Song, Y.; Qu, Y.; Peng, J.R.; Shi, K.; Du, D.; Li, H.; Lin, Y.H.; Qian, Z.Y. Mesoporous ptpd nanoparticles for ligand-mediated and imaging-guided chemo-photothermal therapy of breast cancer. Nano Res. 2020, 13, 1739–1748. [Google Scholar] [CrossRef]
- Wu, W.T.; Shen, J.; Banerjee, P.; Zhou, S.Q. Core-shell hybrid nanogels for integration of optical temperature-sensing, targeted tumor cell imaging, and combined chemo-photothermal treatment. Biomaterials 2010, 31, 7555–7566. [Google Scholar] [CrossRef]
- Kang, H.Z.; Trondoli, A.C.; Zhu, G.Z.; Chen, Y.; Chang, Y.J.; Liu, H.P.; Huang, Y.F.; Zhang, X.L.; Tan, W.H. Near-infrared light-responsive core-shell nanogels for targeted drug delivery. ACS Nano 2011, 5, 5094–5099. [Google Scholar] [CrossRef] [PubMed]
- Dutta, D.; Chattopadhyay, A.; Ghosh, S.S. Cationic BSA templated Au-Ag bimetallic nanoclusters as a theranostic gene delivery vector for hela cancer cells. ACS Biomater. Sci. Eng. 2016, 2, 2090–2098. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.H.; Qi, C.; Lin, J.; Huang, P. Catalytic chemistry of glucose oxidase in cancer diagnosis and treatment. Chem. Soc. Rev. 2018, 47, 6454–6472. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.H.; Qi, C.; Hu, Y.R.; Lin, J.; Huang, P. Glucose oxidase-instructed multimodal synergistic cancer therapy. Adv. Mater. 2019, 31, 1808325. [Google Scholar] [CrossRef] [PubMed]
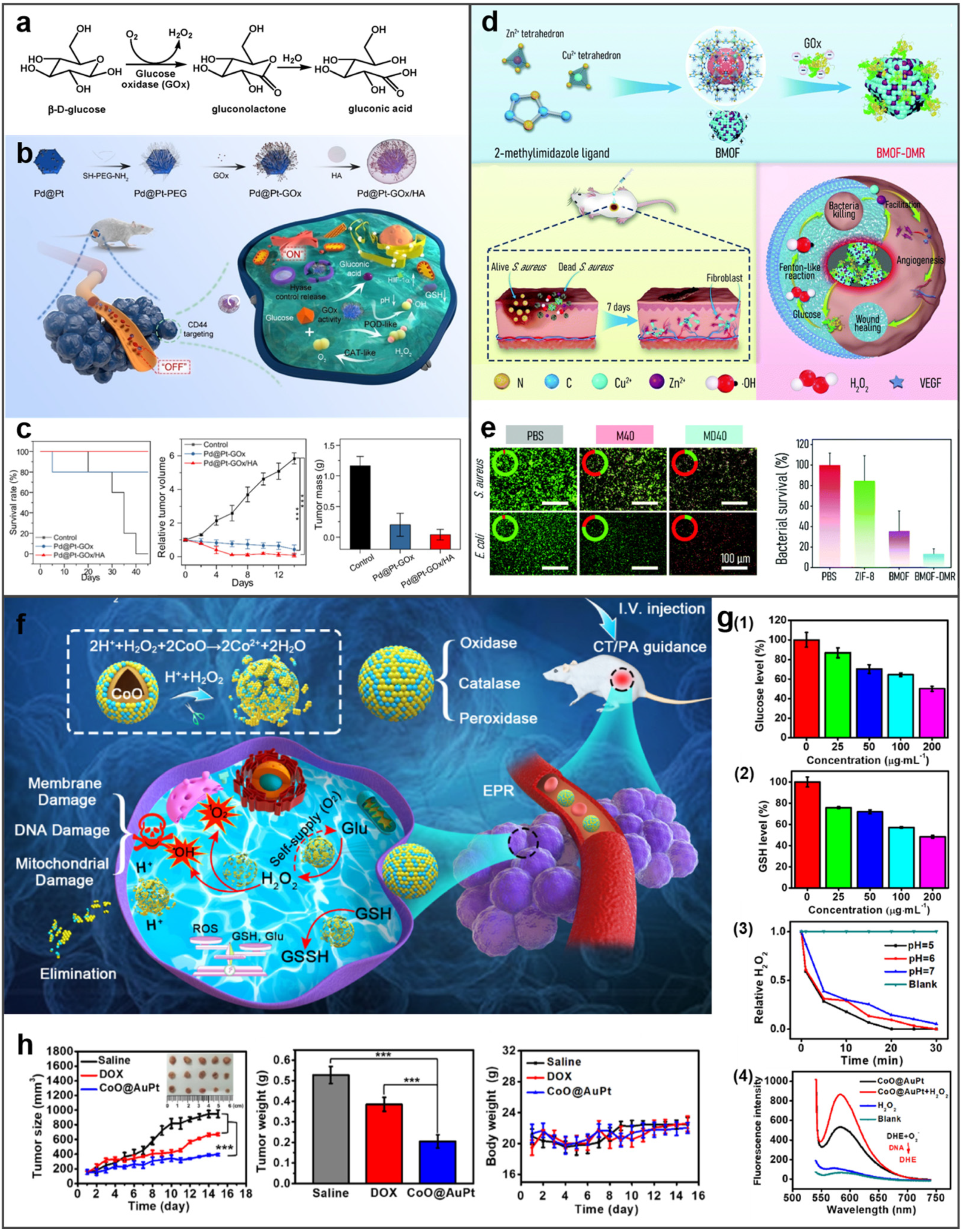
- Ming, J.; Zhu, T.B.; Yang, W.H.; Shi, Y.R.; Huang, D.D.; Li, J.C.; Xiang, S.J.; Wang, J.J.; Chen, X.L.; Zheng, N.F. Pd@Pt-GOx/HA as a novel enzymatic cascade nanoreactor for high-efficiency starving-enhanced chemodynamic cancer therapy. ACS Appl. Mater. Interfaces 2020, 12, 51249–51262. [Google Scholar] [CrossRef]
- Peng, L.M.; Yang, X.Y.; Wang, S.; Chan, Y.K.; Chen, Y.; Yang, Z.P.; Mao, Y.R.; Li, L.M.; Yang, W.Z.; Deng, Y. Bimetal metal-organic framework domino micro-reactor for synergistic antibacterial starvation/chemodynamic therapy and robust wound healing. Nanoscale 2022, 14, 2052–2064. [Google Scholar] [CrossRef]
- Fu, S.Y.; Yang, R.H.; Zhang, L.; Liu, W.W.; Du, G.Y.; Cao, Y.; Xu, Z.G.; Cui, H.J.; Kang, Y.J.; Xue, P. Biomimetic CoO@AuPt nanozyme responsive to multiple tumor microenvironmental clues for augmenting chemodynamic therapy. Biomaterials 2020, 257, 120279. [Google Scholar] [CrossRef]
- Cho, S.; Park, W.; Kim, H.; Jokisaari, J.R.; Roth, E.W.; Lee, S.; Klie, R.F.; Lee, B.; Kim, D. Gallstone-formation-inspired bimetallic supra-nanostructures for computed-tomography-image-guided radiation therapy. ACS Appl. Nano Mater. 2018, 1, 4602–4611. [Google Scholar] [CrossRef]
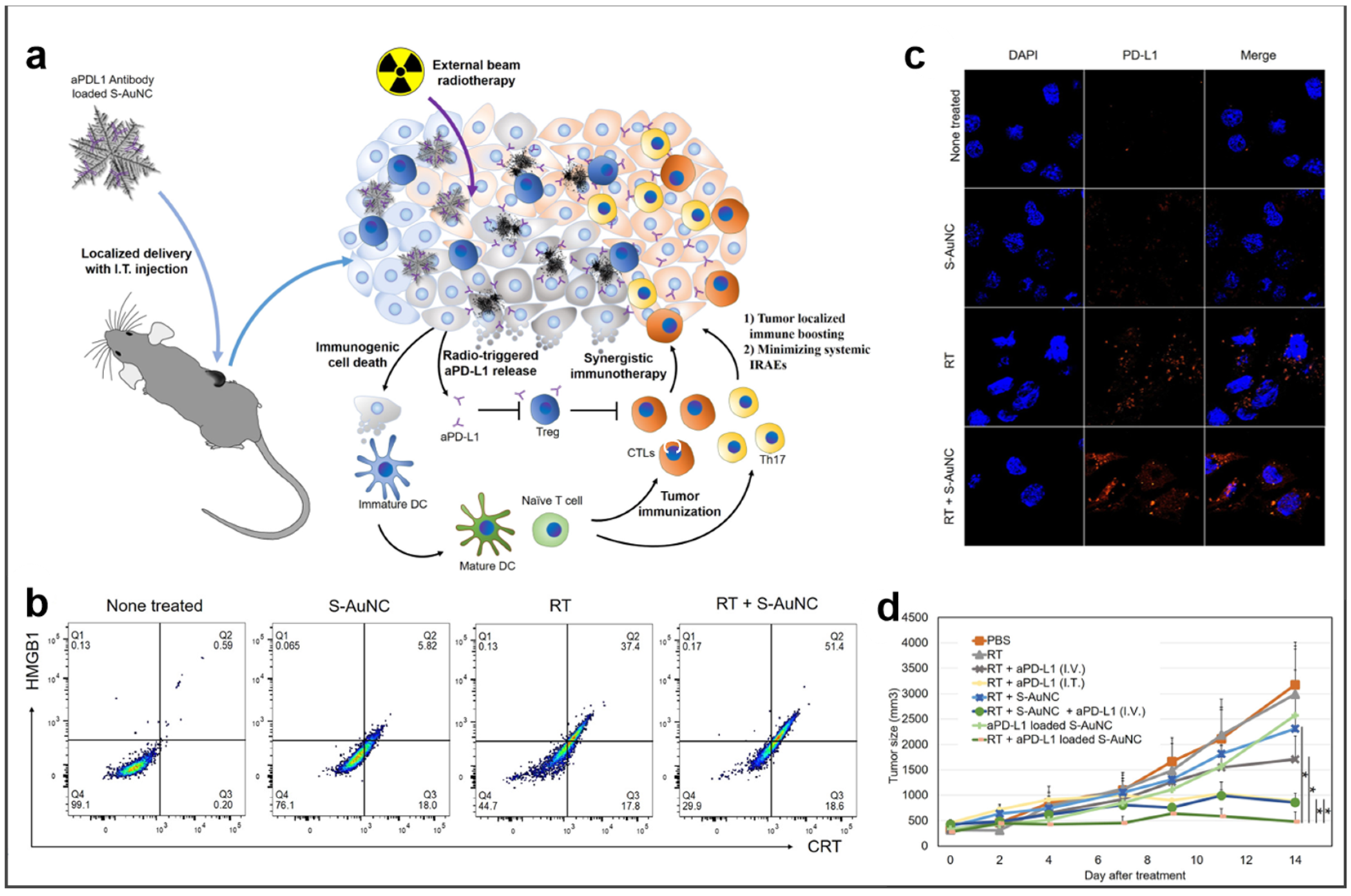
- Choi, B.; Choi, H.; Yu, B.; Kim, D.H. Synergistic local combination of radiation and anti-programmed death ligand 1 immunotherapy using radiation-responsive splintery metallic nanocarriers. ACS Nano 2020, 14, 13115–13126. [Google Scholar] [CrossRef]
- Moding, E.J.; Kastan, M.B.; Kirsch, D.G. Strategies for optimizing the response of cancer and normal tissues to radiation. Nat. Rev. Drug Discov. 2013, 12, 526–542. [Google Scholar] [CrossRef]
- Yang, G.; Gong, H.; Qian, X.; Tan, P.; Li, Z.; Liu, T.; Liu, J.; Li, Y.; Liu, Z. Mesoporous silica nanorods intrinsically doped with photosensitizers as a multifunctional drug carrier for combination therapy of cancer. Nano Res. 2015, 8, 751–764. [Google Scholar] [CrossRef]
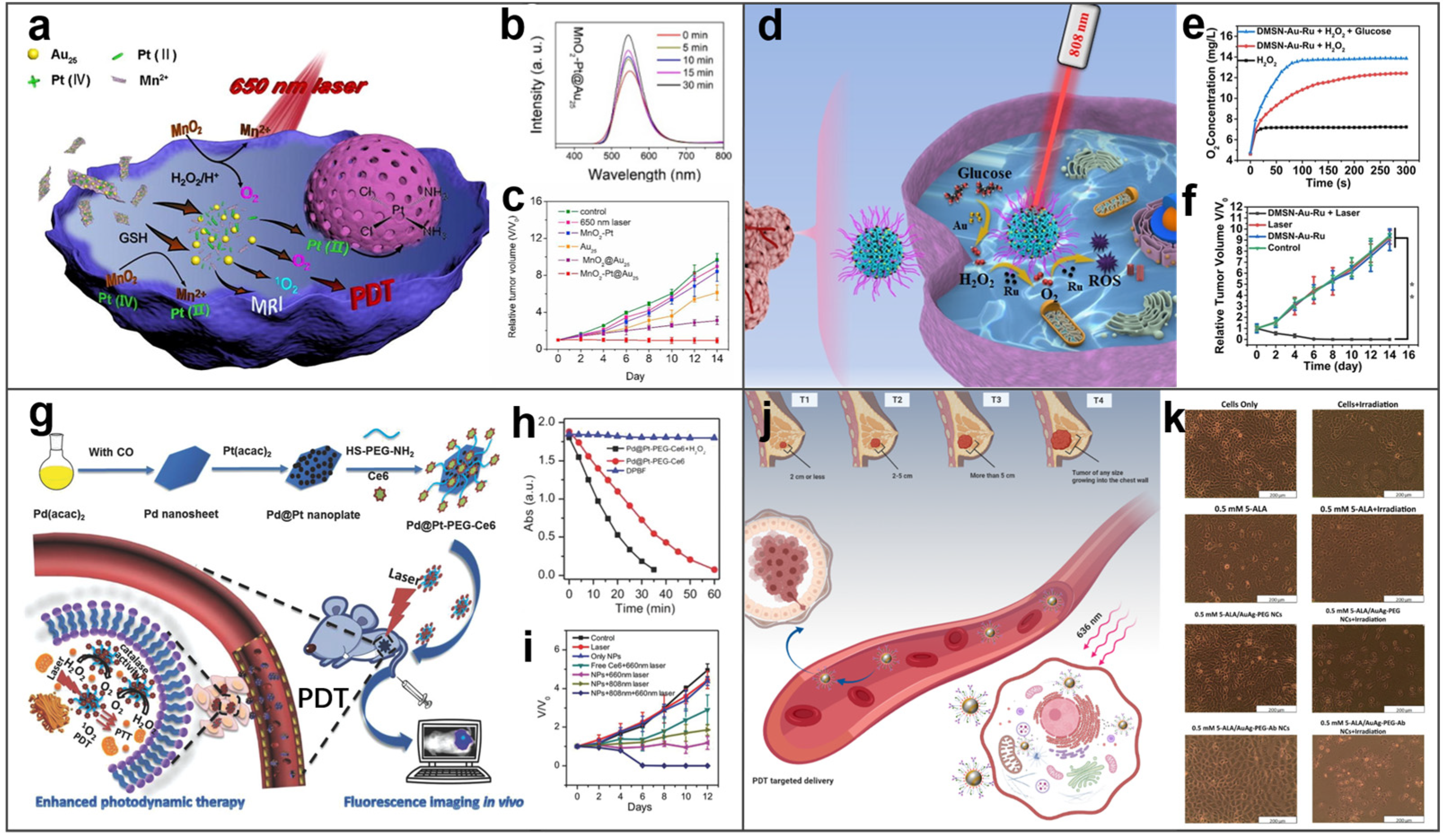
- Bi, H.T.; Dai, Y.L.; Yang, P.P.; Xu, J.T.; Yang, D.; Gai, S.L.; He, F.H.; An, G.H.; Zhong, C.N.; Lin, J. Glutathione and H2O2 consumption promoted photodynamic and chemotherapy based on biodegradable MnO2-Pt@Au-25 nanosheets. Chem. Eng. J. 2019, 356, 543–553. [Google Scholar] [CrossRef]
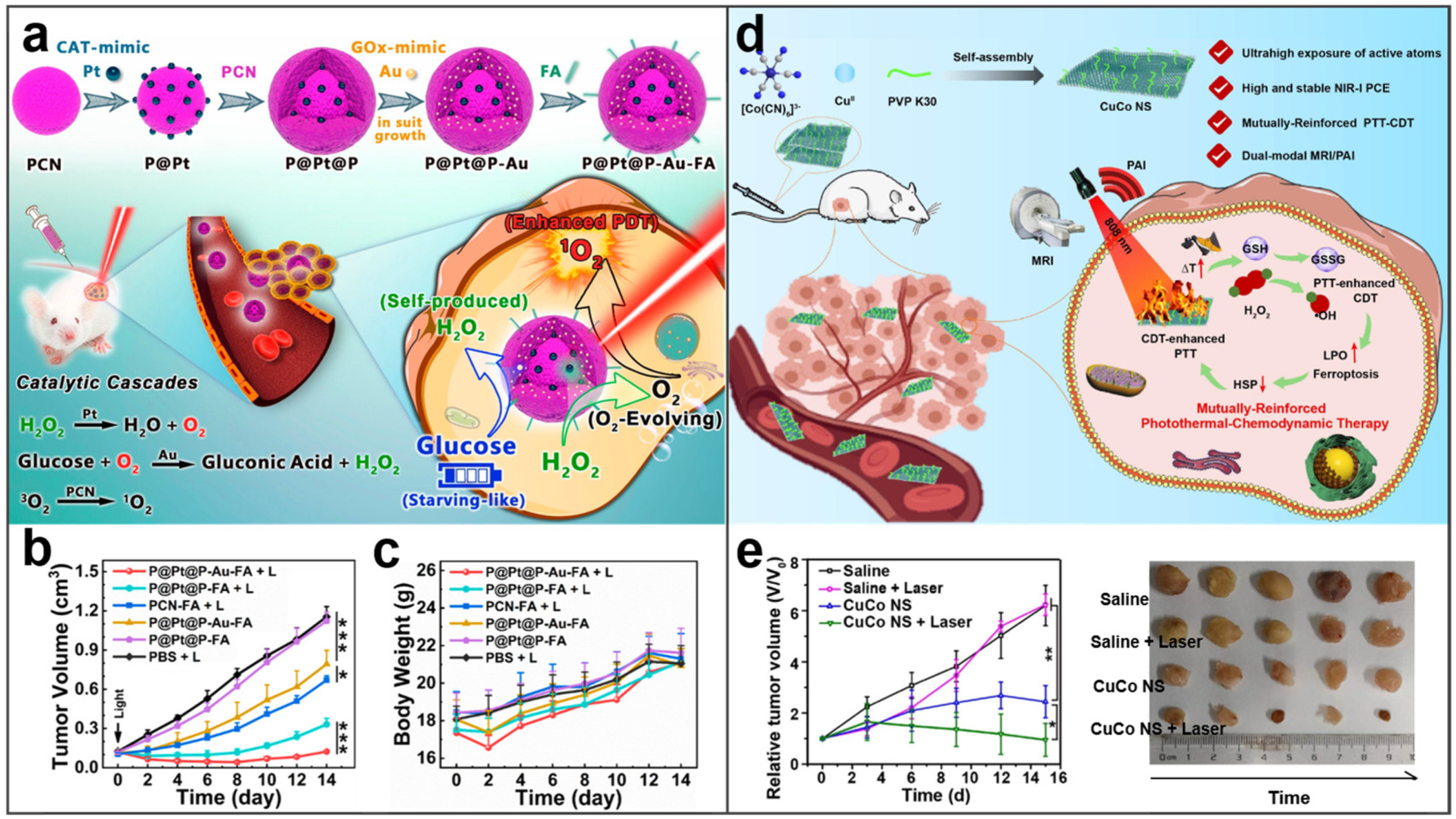
- Li, X.Q.; Cao, Y.; Xu, B.; Zhao, Y.; Zhang, T.Q.; Wang, Y.H.; Wang, D.G.; Liu, J.H.; Song, S.Y.; Zhang, H.J. A bimetallic nanozyme with cascade effect for synergistic therapy of cancer. ChemMedChem 2022, 17, 663. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.P.; Li, J.C.; Sun, D.; Li, Q.; Ma, J.Y.; Chen, X.L.; Zhu, X.; Zheng, N.F. A novel theranostic nanoplatform based on Pd@Pt-PEG-Ce6 for enhanced photodynamic therapy by modulating tumor hypoxia microenvironment. Adv. Funct. Mater. 2018, 28, 1706310. [Google Scholar] [CrossRef]
- Montaseri, H.; Kruger, C.A.; Abrahamse, H. Targeted photodynamic therapy using alloyed nanoparticle-conjugated 5-aminolevulinic acid for breast cancer. Pharmaceutics 2021, 13, 1375. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, W.J.; Zhao, L.; Rong, L.; Lan, S.J.; Sun, H.C.; Zhang, H.; Yang, B. Hydroquinone-assisted synthesis of branched Au-Ag nanoparticles with polydopamine coating as highly efficient photothermal agents. ACS Appl. Mater. Interfaces 2015, 7, 11613–11623. [Google Scholar] [CrossRef]
- Tang, J.L.; Jiang, X.M.; Wang, L.M.; Zhang, H.; Hu, Z.J.; Liu, Y.; Wu, X.C.; Chen, C.Y. Au@Pt nanostructures: A novel photothermal conversion agent for cancer therapy. Nanoscale 2014, 6, 3670–3678. [Google Scholar] [CrossRef]
- Liu, Z.; Cheng, L.; Zhang, L.; Yang, Z.B.; Liu, Z.; Fang, J.X. Sub-100 nm hollow au-ag alloy urchin-shaped nanostructure with ultrahigh density of nanotips for photothermal cancer therapy. Biomaterials 2014, 35, 4099–4107. [Google Scholar] [CrossRef]
- Tang, M.; Zhang, Z.; Ding, C.; Li, J.; Shi, Y.; Sun, T.; Chen, C. Two birds with one stone: Innovative ceria-loaded gold@platinum nanospheres for photothermal-catalytic therapy of tumors. J. Colloid Interface Sci. 2022, 627, 299–307. [Google Scholar] [CrossRef]
- Wang, X.W.; Zhong, X.Y.; Liu, Z.; Cheng, L. Recent progress of chemodynamic therapy-induced combination cancer therapy. Nano Today 2020, 35, 100946. [Google Scholar] [CrossRef]
- Zuo, W.B.; Liu, N.; Chang, Z.Y.; Liu, J.X.; Jin, Q.Y.; Chen, L.P.; Zhu, X. Single-site bimetallic nanosheet for imaging guided mutually-reinforced photothermal-chemodynamic therapy. Chem. Eng. J. 2022, 442, 136125. [Google Scholar] [CrossRef]
- Peng, C.; Liang, Y.; Su, N.; Chen, S.W.; Yuan, Z.; Chen, Y.Q.; Wu, D.; Wu, B.; Zhang, Y.; Xu, Z.T.; et al. Dual nanoenzymes loaded hollow mesoporous organotantalum nanospheres for chemo-radio sensitization. J. Control. Release 2022, 347, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.Y.; Wang, J.P.; Hu, W.; Wang, Y.H.; Zhang, Q.; Hu, X.T.; Chou, T.M.; Zhang, B.L.; Gallaro, C.; Halloran, M.; et al. A porous bimetallic Au@Pt core-shell oxygen generator to enhance hypoxia-dampened tumor chemotherapy synergized with NIR-II photothermal therapy. ACS Nano 2022, 16, 10711–10728. [Google Scholar] [CrossRef] [PubMed]
- Ran, B.; Chen, C.; Liu, B.; Lan, M.; Chen, H.; Zhu, Y. A Ti3C2Tx/Pt-Pd based amperometric biosensor for sensitive cancer biomarker detection. Electrophoresis 2022, 43, 2033–2043. [Google Scholar] [CrossRef]
- Ge, X.; Feng, Y.; Cen, S.; Wang, A.; Mei, L.; Luo, X.; Feng, J. A label-free electrochemical immnunosensor based on signal magnification of oxygen reduction reaction catalyzed by uniform PtCo nanodendrites for highly sensitive detection of carbohydrate antigen 15-3. Anal. Chim. Acta 2021, 1176, 338750. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liang, X.; Chen, F.; Ding, S. Ultrasensitive amperometric cytosensor for drug evaluation with monitoring early cell apoptosis based on Cu2O@PtPd nanocomposite as signal amplified label. Sens. Actuators B Chem. 2019, 300, 127046. [Google Scholar] [CrossRef]
- Yang, Q.; Peng, J.R.; Xiao, Y.; Li, W.T.; Tan, L.W.; Xu, X.H.; Qian, Z.Y. Porous Au@Pt nanoparticles: Therapeutic platform for tumor chemo-photothermal co-therapy and alleviating doxorubicin-induced oxidative damage. ACS Appl. Mater. Interfaces 2018, 10, 150–164. [Google Scholar] [CrossRef]
- He, J.C.; Dong, J.W.; Hu, Y.F.; Li, G.K.; Hu, Y.L. Design of raman tag-bridged core-shell Au@Cu3(BTC)(2) nanoparticles for raman imaging and synergistic chemo-photothermal therapy. Nanoscale 2019, 11, 6089–6100. [Google Scholar] [CrossRef]


| Types of BMNS | Products | Lasers | Ref. |
|---|---|---|---|
| Pt−modified Au NPs | H2 | 460 < λ < 820 nm | [77] |
| Ag–Cu alloy NPs | •OH radical, •O2− radical | λ = 980 nm | [78] |
| Au–Pt alloys | H2 | λ = 325 nm | [79] |
| Pt@MIL−125/Au | H2 | 380 < λ < 800 nm | [80] |
| Au–Cu/CaIn2S4 composites | H2 | 420 nm ≤ λ ≤ 750 nm | [81] |
| Au/Pd/TiO2 nanoparticles | O•− radical | 310 < λ < 380 nm | [82] |
| Au–Pd/TiO2/NB nanostructures | O•− radical | λ = 450 nm | [83] |
| Au@Pd@MOF−74 | CO | λ = 707 nm | [84] |
| Pt/Au@Pd@MOF−74 | CH4 (H• radicals) | λ = 523 nm | [84] |
| Au@Ag/TiO2 NP | •OH radical, •O2− radical | λ = 664 nm | [85] |
| Cu–Au NPs | •O2− radical | 620 nm ≤ λ ≤ 670 nm | [86] |
| Ag/Au/TiO2 NPs | •O2− radical | λ = 254 nm | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niu, G.; Gao, F.; Wang, Y.; Zhang, J.; Zhao, L.; Jiang, Y. Bimetallic Nanomaterials: A Promising Nanoplatform for Multimodal Cancer Therapy. Molecules 2022, 27, 8712. https://doi.org/10.3390/molecules27248712
Niu G, Gao F, Wang Y, Zhang J, Zhao L, Jiang Y. Bimetallic Nanomaterials: A Promising Nanoplatform for Multimodal Cancer Therapy. Molecules. 2022; 27(24):8712. https://doi.org/10.3390/molecules27248712
Chicago/Turabian StyleNiu, Guiming, Fucheng Gao, Yandong Wang, Jie Zhang, Li Zhao, and Yanyan Jiang. 2022. "Bimetallic Nanomaterials: A Promising Nanoplatform for Multimodal Cancer Therapy" Molecules 27, no. 24: 8712. https://doi.org/10.3390/molecules27248712
APA StyleNiu, G., Gao, F., Wang, Y., Zhang, J., Zhao, L., & Jiang, Y. (2022). Bimetallic Nanomaterials: A Promising Nanoplatform for Multimodal Cancer Therapy. Molecules, 27(24), 8712. https://doi.org/10.3390/molecules27248712