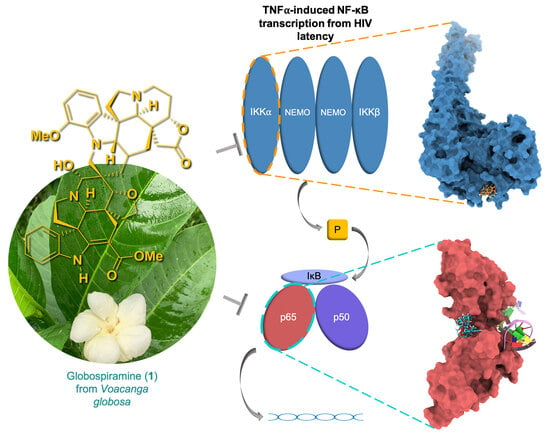
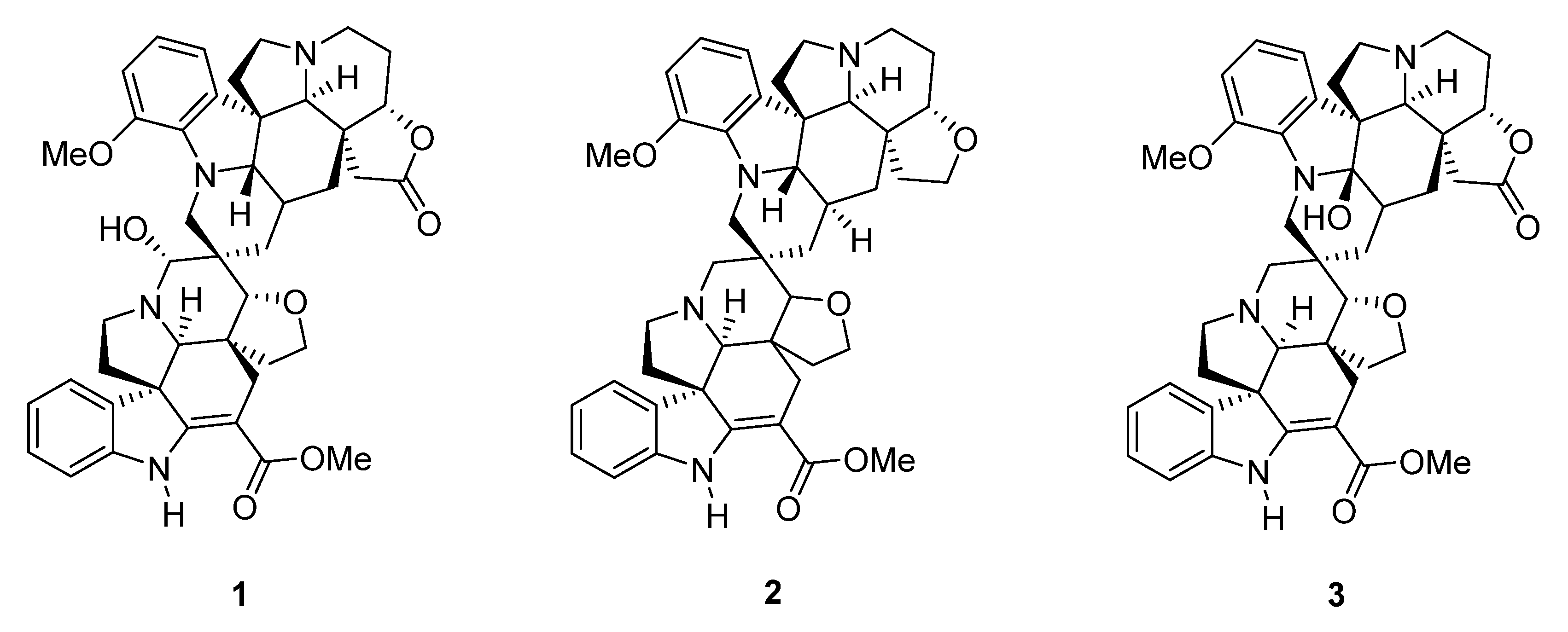
Voacanga globosa Spirobisindole Alkaloids Exert Antiviral Activity in HIV Latently Infected Cell Lines by Targeting the NF-κB Cascade: In Vitro and In Silico Investigations
Abstract
1. Introduction
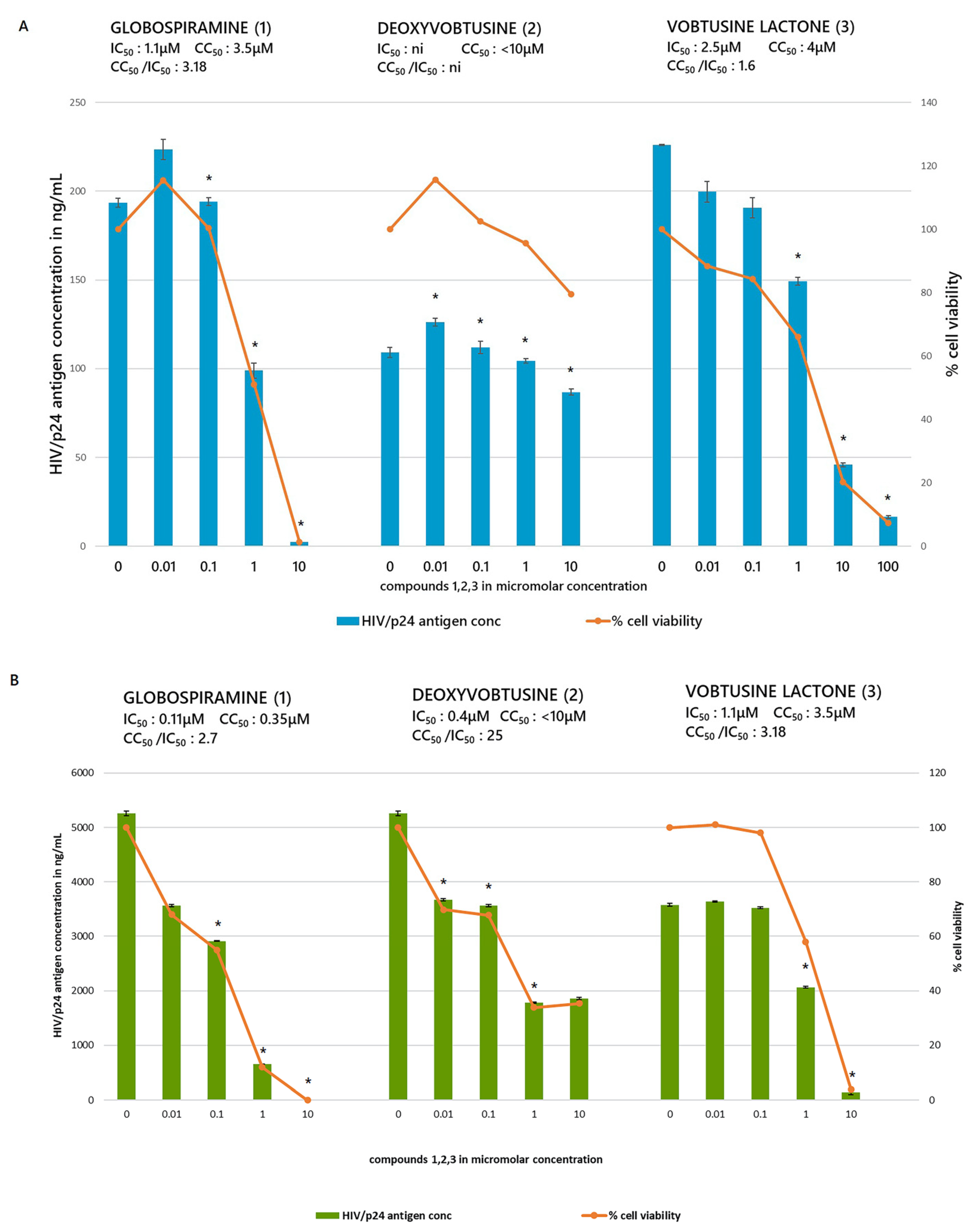
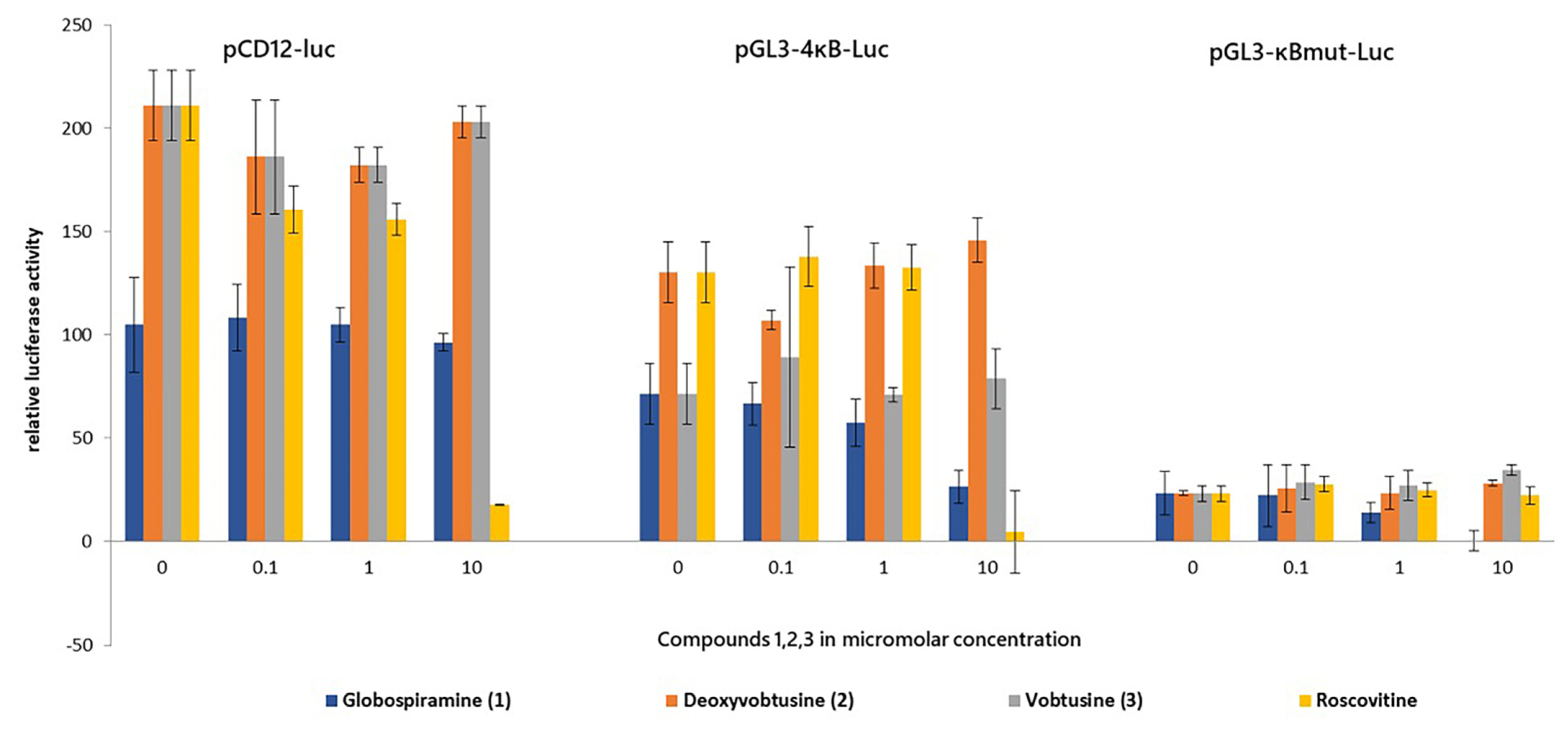
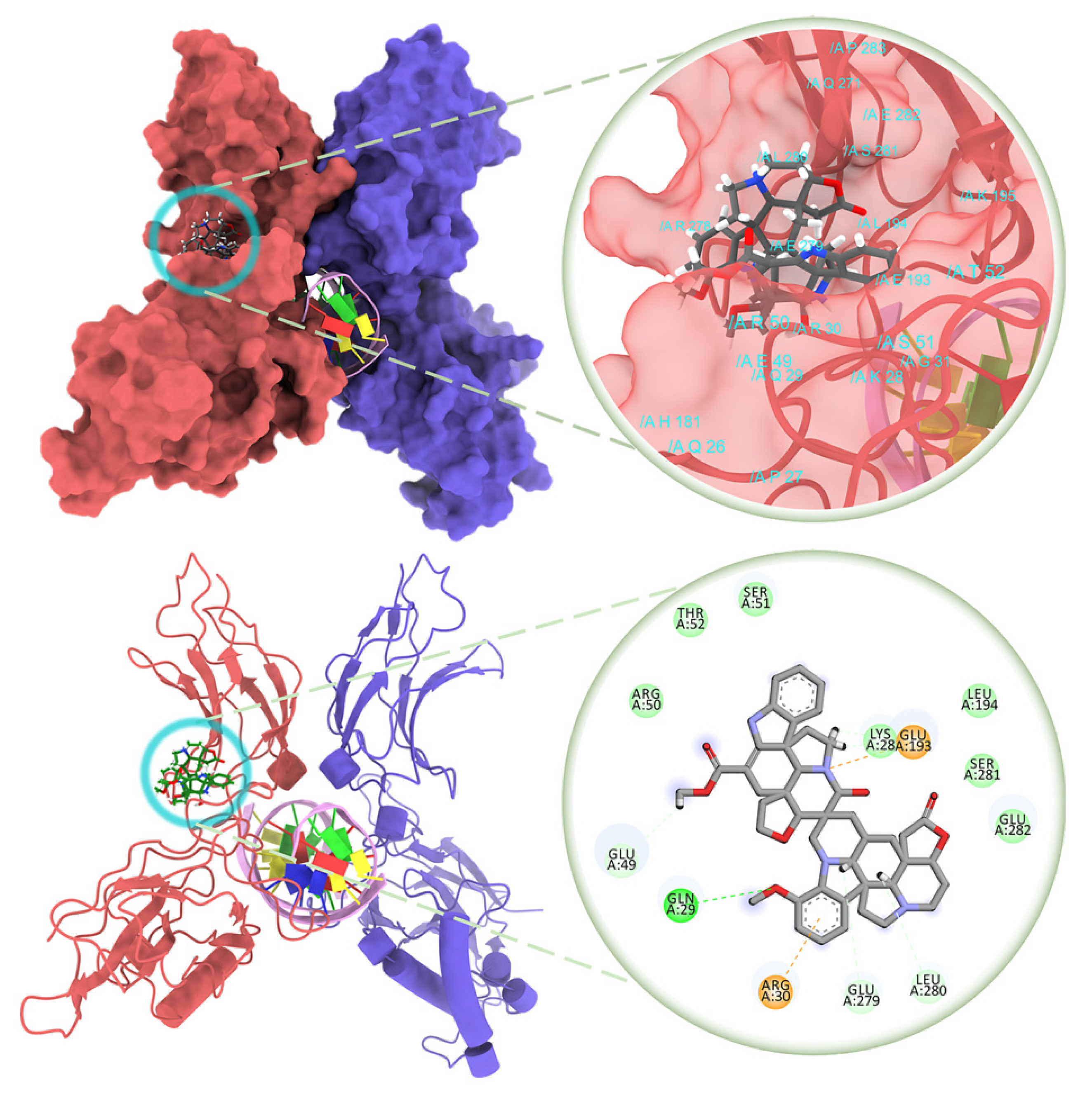
2. Results and Discussion
3. Materials and Methods
3.1. Test Compounds
3.2. Cell Lines and Culture Conditions
3.3. Cytotoxicity Determination
3.4. Anti-HIV Assay
3.5. Transient Transfection and Luciferase Assay
3.6. Data Analysis
3.7. Molecular Docking Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Correction Statement
Sample Availability
References
- UNAIDS. Fact Sheet 2021 Global HIV Statistics; UNADIS: Geneva, Switzerland, 2021. [Google Scholar]
- Epidemiology Bureau. HIV/AIDS & ART Registry of the Philippines; Department of Health: Manila, Philippines, 2021.
- Arts, E.J.; Hazuda, D.J. HIV-1 antiretroviral drug therapy. Cold Spring Harb. Perspect. Med. 2012, 2, a007161. [Google Scholar] [CrossRef]
- De Clercq, E.; Li, G. Approved antiviral drugs over the past 50 years. Clin. Microbiol. Rev. 2016, 29, 695–747. [Google Scholar] [CrossRef] [PubMed]
- Deeks, S.G.; Overbaugh, J.; Phillips, A.; Buchbinder, S. HIV infection. Nat. Rev. Dis. Prim. 2015, 1, 115035. [Google Scholar] [CrossRef] [PubMed]
- Pham, M.D.; Romero, L.; Parnell, B.; Anderson, D.A.; Crowe, S.M.; Luchters, S. Feasibility of antiretroviral treatment monitoring in the era of decentralized HIV care: A systematic review. AIDS Res. Ther. 2017, 14, 3. [Google Scholar] [CrossRef] [PubMed]
- Smyth, R.P.; Schlub, T.E.; Grimm, A.J.; Waugh, C.; Ellenberg, P.; Chopra, A.; Mallal, S.; Cromer, D.; Mak, J.; Davenport, M.P.; et al. Identifying recombination hot spots in the HIV-1 genome. J. Virol. 2014, 88, 2891–2902. [Google Scholar] [CrossRef] [PubMed]
- Spivak, A.M.; Planelles, V. HIV-1 Eradication: Early Trials (and Tribulations). Trends Mol. Med. 2016, 22, 10–27. [Google Scholar] [CrossRef] [PubMed]
- Cary, D.C.; Fujinaga, K.; Peterlin, B.M. Molecular mechanisms of HIV latency. J. Clin. Investig. 2016, 126, 448–454. [Google Scholar] [CrossRef]
- Eisele, E.; Siliciano, R.F. Redefining the viral reservoirs that prevent HIV-1 eradication. Immunity 2012, 37, 377–388. [Google Scholar] [CrossRef]
- Williams, S.A.; Kwon, H.; Chen, L.-F.; Greene, W.C. Sustained induction of NF-κB Is required for efficient expression of latent human immunodeficiency virus type 1. J. Virol. 2007, 81, 6043–6056. [Google Scholar] [CrossRef]
- Kilareski, E.M.; Shah, S.; Nonnemacher, M.R.; Wigdahl, B. Regulation of HIV-1 transcription in cells of the monocyte-macrophage lineage. Retrovirology 2009, 6, 118. [Google Scholar] [CrossRef]
- Sengupta, S.; Siliciano, R.F. Targeting the latent reservoir for HIV-1. Immunity 2018, 48, 872–895. [Google Scholar] [CrossRef]
- Le Douce, V.; Ait-Amar, A.; Far, F.F.; Fahmi, F.; Quiel, J.; El Mekdad, H.; Daouad, F.; Marban, C.; Rohr, O.; Schwartz, C. Improving combination antiretroviral therapy by targeting HIV-1 gene transcription. Expert Opin. Ther. Targets 2016, 20, 1311–1324. [Google Scholar] [CrossRef]
- Wong, L.M.; Jiang, G. NF-κB sub-pathways and HIV cure: A revisit. EBioMedicine 2021, 63, 103159. [Google Scholar] [CrossRef]
- Takada, Y.; Kobayashi, Y.; Aggarwal, B.B. Evodiamine abolishes constitutive and inducible NF-κB activation by inhibiting IκBα kinase activation, thereby suppressing NF-κB-regulated antiapoptotic and metastatic gene expression, up-regulating apoptosis, and inhibiting invasion. J. Biol. Chem. 2005, 280, 17203–17212. [Google Scholar] [CrossRef]
- Ho, L.J.; Juan, T.Y.; Chao, P.; Wu, W.L.; Chang, D.M.; Chang, S.Y.; Lai, J.H. Plant alkaloid tetrandrine downregulates IκBα kinases-IκBα-NF-κB signaling pathway in human peripheral blood T cell. Br. J. Pharmacol. 2004, 143, 919–927. [Google Scholar] [CrossRef]
- Cary, D.C.; Peterlin, B.M.; Matija Peterlin, B. Natural products and HIV/AIDS. AIDS Res. Hum. Retrovir. 2017, 34, 31–38. [Google Scholar] [CrossRef]
- Ravanelli, N.; Santos, K.P.; Motta, L.B.; Lago, J.H.G.; Furlan, C.M. Alkaloids from Croton echinocarpus Baill.: Anti-HIV potential. S. Afr. J. Bot. 2016, 102, 153–156. [Google Scholar] [CrossRef]
- Chaves Valadão, A.L.; Abreu, C.M.; Dias, J.Z.; Arantes, P.; Verli, H.; Tanuri, A.; de Aguiar, R.S. Natural plant alkaloid (emetine) inhibits HIV-1 replication by interfering with reverse transcriptase activity. Molecules 2015, 20, 11474–11489. [Google Scholar] [CrossRef]
- Kurapati, K.R.V.; Atluri, V.S.; Samikkannu, T.; Garcia, G.; Nair, M.P.N. Natural products as anti-HIV agents and role in HIV-associated neurocognitive disorders (HAND): A brief overview. Front. Microbiol. 2016, 6, 1444. [Google Scholar] [CrossRef]
- Dang, Z.; Zhu, L.; Lai, W.; Bogerd, H.; Lee, K.H.; Huang, L.; Chen, C.H. Aloperine and its derivatives as a new class of HIV-1 entry inhibitors. ACS Med. Chem. Lett. 2016, 7, 240–244. [Google Scholar] [CrossRef]
- Shikalepo, R.; Mukakalisa, C.; Kandawa-Schulz, M.; Chingwaru, W.; Kapewangolo, P. In vitro anti-HIV and antioxidant potential of Bulbine frutescens (Asphodelaceae). J. Herb. Med. 2018, 12, 73–78. [Google Scholar] [CrossRef]
- Mousseau, G.; Kessing, C.F.; Fromentin, R.; Trautmann, L.; Chomont, N.; Valente, S.T. The tat inhibitor didehydro-cortistatin a prevents HIV-1 reactivation from latency. MBio 2015, 6, e00465-15. [Google Scholar] [CrossRef] [PubMed]
- Asamitsu, K.; Fujinaga, K.; Okamoto, T. HIV Tat/P-TEFb interaction: A potential target for novel Anti-HIV therapies. Molecules 2018, 23, 933. [Google Scholar] [CrossRef] [PubMed]
- Macabeo, A.P.G.; Krohn, K.; Gehle, D.; Read, R.W.; Brophy, J.J.; Franzblau, S.G.; Aguinaldo, M.A.M. Activity of the extracts and indole alkaloids from Alstonia scholaris against Mycobacterium tuberculosis H37Rv. Philipp. Agric. Sci. 2008, 91, 348–351. [Google Scholar]
- Singh, T.P.; Singh, O.M. Recent Progress in biological activities of indole and indole alkaloids. Mini Rev. Med. Chem. 2017, 18, 8–25. [Google Scholar] [CrossRef]
- Macabeo, A.P.G.; Alejandro, G.J.D.; Hallare, A.V.; Vidar, W.S.; Villaflores, O.B. Phytochemical survey and pharmacological activities of the indole alkaloids in the genus Voacanga Thouars (Apocynaceae)—An update. Pharmacogn. Rev. 2009, 3, 143–153. [Google Scholar]
- Zhang, M.Z.; Chen, Q.; Yang, G.F. A review on recent developments of indole-containing antiviral agents. Eur. J. Med. Chem. 2015, 89, 421–441. [Google Scholar] [CrossRef]
- Hashimoto, K.; Baba, M.; Gohnai, K.; Sato, M.; Shigeta, S. Heat shock induces HIV-1 replication in chronically infected promyelocyte cell line OM10.1. Arch. Virol. 1996, 141, 439–447. [Google Scholar] [CrossRef]
- Jordan, A.; Bisgrove, D.; Verdin, E. HIV reproducibly establishes a latent infection after acute infection of T cells in vitro. EMBO J. 2003, 22, 1868–1877. [Google Scholar] [CrossRef]
- De Paz-Silava, S.L.M.; Victoriano-Belvis, A.F.B.; Gloriani, N.G.; Hibi, Y.; Asamitsu, K.; Okamoto, T. In vitro antiviral activity of Mentha cordifolia plant extract in HIV-1 latently infected cells using an established human cell line. AIDS Res. Hum. Retrovir. 2021, 38, 64–72. [Google Scholar] [CrossRef]
- MacAbeo, A.P.G.; Vidar, W.S.; Chen, X.; Decker, M.; Heilmann, J.; Wan, B.; Franzblau, S.G.; Galvez, E.V.; Aguinaldo, M.A.M.; Cordell, G.A. Mycobacterium tuberculosis and cholinesterase inhibitors from Voacanga globosa. Eur. J. Med. Chem. 2011, 46, 3118–3123. [Google Scholar] [CrossRef]
- Wan, F.; Lenardo, M.J. Specification of DNA binding activity of NF-kappaB proteins. Cold Spring Harb. Perspect. Biol. 2009, 1, a000067. [Google Scholar] [CrossRef]
- Liu, F.; Xia, Y.; Parker, A.S.; Verma, I.M. IKK Biology. Immunol. Rev. 2012, 246, 239–253. [Google Scholar] [CrossRef]
- Schwartz, C.; Bouchat, S.; Marban, C.; Gautier, V.; Van Lint, C.; Rohr, O.; Le Douce, V. On the way to find a cure: Purging latent HIV-1 reservoirs. Biochem. Pharmacol. 2017, 146, 10–22. [Google Scholar] [CrossRef]
- Fujinaga, K.; Cary, D.C. Experimental systems for measuring hiv latency and reactivation. Viruses 2020, 12, 1279. [Google Scholar] [CrossRef]
- Hamasaki, T.; Okamoto, M.; Baba, M. Identification of novel inhibitors of human immunodeficiency virus type 1 replication by in silico screening targeting cyclin T1/tat interaction. Antimicrob. Agents Chemother. 2013, 57, 1323–1331. [Google Scholar] [CrossRef]
- Baba, M. Recent status of HIV-1 gene expression inhibitors. Antiviral Res. 2006, 71, 301–306. [Google Scholar] [CrossRef]
- Dey, A.; Wong, E.T.; Cheok, C.F.; Tergaonkar, V.; Lane, D.P. R-Roscovitine simultaneously targets both the p53 and NF-κB pathways and causes potentiation of apoptosis: Implications in cancer therapy. Cell Death Differ. 2008, 15, 263–273. [Google Scholar] [CrossRef]
- Qi, X.; Koya, Y.; Saitoh, T.; Saitoh, Y.; Shimizu, S.; Ohba, K.; Yamamoto, N.; Yamaoka, S.; Yamamoto, N. Efficient induction of HIV-1 replication in latently infected cells through contact with CD4+ T cells: Involvement of NF-κB activation. Virology 2007, 361, 325–334. [Google Scholar] [CrossRef]
- Agbottah, E.; De La Fuente, C.; Nekhai, S.; Barnett, A.; Gianella-Borradori, A.; Pumfery, A.; Kashanchi, F. Antiviral activity of CYC202 in HIV-1-infected cells. J. Biol. Chem. 2005, 280, 3029–3042. [Google Scholar] [CrossRef]
- Chen, Q.; Wu, C.; Zhu, J.; Li, E.; Xu, Z. Therapeutic potential of indole derivatives as anti-HIV agents: A mini-review. Curr. Top. Med. Chem. 2021, 21. [Google Scholar] [CrossRef]
- Silva, E.M.; Cirne-Santos, C.C.; Frugulhetti, I.C.P.P.; Galvão-Castro, B.; Saraiva, E.M.B.; Kuehnes, M.E.; Bou-Habib, D.C. Anti-HIV-1 activity of the Iboga alkaloid congener 18-methoxycoronaridine. Planta Med. 2004, 70, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.J.; Di, Y.T.; Wang, Y.H.; Zhang, Y.; Si, Y.K.; Zhang, Q.; Gao, S.; Hu, X.J.; Fang, X.; Li, S.F.; et al. Three new indole alkaloids from Trigonostemon lii. Org. Lett. 2010, 12, 2370–2373. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Kalia, R.; Raj, T.; Gupta, V.K.; Suri, N.; Saxena, A.K.; Sharma, D.; Bhella, S.S.; Singh, G.; Ishar, M.P.S. Synthesis and cytotoxic evaluation of substituted 3-(3′-indolyl-/3′-pyridyl)-isoxazolidines and bis-indoles. Acta Pharm. Sin. B 2012, 2, 32–41. [Google Scholar] [CrossRef]
- Okamoto, T. Regulation of HIV replication by a host transcription factor NF-κB. Jpn. J. Med. Sci. Biol. 1994, 26795. [Google Scholar]
- Chan, J.K.; Greene, W.C. Dynamic roles for NF-κB in HTLV-I and HIV-1 retroviral pathogenesis. Immunol. Rev. 2012, 246, 286–310. [Google Scholar] [CrossRef]
- Victoriano, A.F.B.; Okamoto, T. Transcriptional control of HIV replication by multiple modulators and their implication for a novel antiviral therapy. AIDS Res. Hum. Retrovir. 2012, 28, 125–138. [Google Scholar] [CrossRef]
- Cie, M.; Bekiranov, S. Genome-wide predictors of NF-κB recruitment and transcriptional activity. BioData Min. 2015, 8, 37. [Google Scholar] [CrossRef][Green Version]
- Chan, J.K.L.; Greene, W.C. NF-κB/Rel: Agonist and antagonist roles in HIV-1 latency. Curr. Opin. HIV AIDS 2011, 6, 12–18. [Google Scholar] [CrossRef]
- Jiang, G.; Dandekar, S. Targeting NF-κB signaling with protein kinase C agonists as an emerging strategy for combating HIV latency. AIDS Res. Hum. Retrovir. 2015, 31, 4–12. [Google Scholar] [CrossRef]
- Mingyan, Y.; Xinyong, L.; De Clercq, E. NF-κB: The inducible factors of HIV-1 transcription and their inhibitors. Mini Rev. Med. Chem. 2009, 9, 60–69. [Google Scholar] [CrossRef]
- Miller, S.C.; Huang, R.; Sakamuru, S.; Shukla, S.J.; Attene-Ramos, M.S.; Shinn, P.; Van Leer, D.; Leister, W.; Austin, C.P.; Xia, M. Identification of known drugs that act as inhibitors of NF-κB signaling and their mechanism of action. Biochem. Pharmacol. 2010, 79, 1272–1280. [Google Scholar] [CrossRef]
- Schmitz, M.L.; Baeuerle, P.A. The p65 subunit is responsible for the strong transcription activating potential of NF-κB. EMBO J. 1991, 10, 3805–3817. [Google Scholar] [CrossRef]
- Hochrainer, K.; Racchumi, G.; Anrather, J. Site-specific phosphorylation of the p65 protein subunit mediates selective gene expression by differential NF-κB and RNA polymerase II promoter recruitment. J. Biol. Chem. 2013, 288, 285–293. [Google Scholar] [CrossRef]
- Marienfeld, R.B.; Palkowitsch, L.; Ghosh, S. Dimerization of the IκB kinase-binding domain of NEMO is required for tumor necrosis factor alpha-induced NF-κB activity. Mol. Cell. Biol. 2006, 26, 9209–9219. [Google Scholar] [CrossRef][Green Version]
- Polley, S.; Passos, D.O.; Huang, D.-B.; Mulero, M.C.; Mazumder, A.; Biswas, T.; Verma, I.M.; Lyumkis, D.; Ghosh, G. Structural Basis for the activation of IKK1/α. Cell Rep. 2016, 17, 1907–1914. [Google Scholar] [CrossRef]
- Israël, A. The IKK complex, a central regulator of NF-kappaB activation. Cold Spring Harb. Perspect. Biol. 2010, 2, a000158. [Google Scholar] [CrossRef]
- Butera, S.T.; Victor, L.; Folks, T.M.; Perez, V.L.; Wu, B.Y.; Nabel, G.J.; Folks, T.M. Oscillation of the human immunodeficiency virus surface receptor is regulated by the state of viral activation in a CD4+ cell model of chronic infection. J. Virol. 1991, 65, 4645–4653. [Google Scholar] [CrossRef]
- Victoriano, A.F.B.; Asamitsu, K.; Hibi, Y.; Imai, K.; Barzaga, N.G.; Okamoto, T. Inhibition of human immunodeficiency virus type 1 replication in latently infected cells by a novel IκB kinase inhibitor. Antimicrob. Agents Chemother. 2006, 50, 547–555. [Google Scholar] [CrossRef]
- Takada, N.; Sanda, T.; Okamoto, H.; Yang, J.-P.; Asamitsu, K.; Sarol, L.; Kimura, G.; Uranishi, H.; Tetsuka, T.; Okamoto, T. RelA-associated inhibitor blocks transcription of human immunodeficiency virus type 1 by inhibiting NF-κB and Sp1 actions. J. Virol. 2002, 76, 8019–8030. [Google Scholar] [CrossRef]
- Sato, T.; Asamitsu, K.; Yang, J.P.; Takahashi, N.; Tetsuka, T.; Yoneyama, A.; Kanagawa, A.; Okamoto, T. Inhibition of human immunodeficiency virus type 1 replication by a bioavailable serine/threonine kinase inhibitor, fasudil hydrochloride. AIDS Res. Hum. Retrovir. 1998, 14, 293–298. [Google Scholar] [CrossRef]
- Chen, F.E.; Huang, D.B.; Chen, Y.Q.; Ghosh, G. Crystal structure of p50/p65 heterodimer of transcription factor NF-κb bound to DNA. Nature 1998, 391, 410–412. [Google Scholar] [CrossRef]
- Rushe, M.; Silvian, L.; Bixler, S.; Chen, L.L.; Cheung, A.; Bowes, S.; Cuervo, H.; Berkowitz, S.; Zheng, T.; Guckian, K.; et al. Structure of a NEMO/IKK-associating domain reveals architecture of the interaction site. Structure 2008, 16, 798–808. [Google Scholar] [CrossRef]
- Grover, A.; Shandilya, A.; Punetha, A.; Bisaria, V.S.; Sundar, D. Inhibition of the NEMO/IKKβ association complex formation, a novel mechanism associated with the NF-κB activation suppression by Withania somnifera’s key metabolite withaferin A. BMC Genomics 2010, 11, S25. [Google Scholar] [CrossRef]
- Polley, S.; Huang, D.B.; Hauenstein, A.V.; Fusco, A.J.; Zhong, X.; Vu, D.; Schröfelbauer, B.; Kim, Y.; Hoffmann, A.; Verma, I.M.; et al. A structural basis for IκB kinase 2 activation via oligomerization-dependent trans auto-phosphorylation. PLoS Biol. 2013, 11, e1001581. [Google Scholar] [CrossRef]
- Kadioglu, O.; Nass, J.; Saeed, M.E.M.; Schuler, B.; Efferth, T. Kaempferol is an anti-inflammatory compound with activity towards NF-ℵB pathway proteins. Anticancer Res. 2015, 35, 2645–2650. [Google Scholar]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Demange, L.; Abdellah, F.N.; Lozach, O.; Ferandin, Y.; Gresh, N.; Meijer, L.; Galons, H. Potent inhibitors of CDK5 derived from roscovitine: Synthesis, biological evaluation and molecular modelling. Bioorganic Med. Chem. Lett. 2013, 23, 125–131. [Google Scholar] [CrossRef]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef]
- Wang, J.; Wang, W.; Kollman, P.A.; Case, D.A. Automatic atom type and bond type perception in molecular mechanical calculations. J. Mol. Graph. Model. 2006, 25, 247–260. [Google Scholar] [CrossRef]
- De Leon, V.N.O.; Manzano, J.A.H.; Pilapil, D.Y.H.; Fernandez, R.A.T.; Ching, J.K.A.R.; Quimque, M.T.J.; Agbay, J.C.M.; Notarte, K.I.R.; Macabeo, A.P.G. Anti-HIV reverse transcriptase plant polyphenolic natural products with in silico inhibitory properties on seven non-structural proteins vital in SARS-CoV-2 pathogenesis. J. Genet. Eng. Biotechnol. 2021, 19, 104. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, R.A.; Quimque, M.T.; Notarte, K.I.; Manzano, J.A.; Pilapil, D.Y.; de Leon, V.N.; San Jose, J.J.; Villalobos, O.; Muralidharan, N.H.; Gromiha, M.M.; et al. Myxobacterial depsipeptide chondramides interrupt SARS-CoV-2 entry by targeting its broad, cell tropic spike protein. J. Biomol. Struct. Dyn. 2021, 1–12. [Google Scholar] [CrossRef] [PubMed]

| Alkaloid | HL-60 Cells (µM) | Jurkat Cells (µM) | OM10.1 Cells (µM) | J-Lat Cells (µM) |
|---|---|---|---|---|
| Globospiramine (1) | 0.75 | 0.50 | 0.70 | 0.350 |
| Deoxyvobtusine (2) | >56.940 | >56.940 | >10.0 | >10.0 |
| Vobtusine lactone (3) | 8.0 | 11.0 | 0.250 | 0.100 |
| OM10.1 Cells | J-Lat Cells | |||||
|---|---|---|---|---|---|---|
| Alkaloid | CC50 (µM) | IC50 (µM) | CC50/IC50 | CC50 (µM) | IC50(µM) | CC50/IC50 |
| Globospiramine (1) | 3.5 | 1.1 | 3.18 | 0.35 | 0.11 | 2.7 |
| Deoxyvobtusine (2) | >10 | ni | ni | >10 | 0.4 | 25 |
| Vobtusine lactone (3) | 4 | 2.5 | 1.6 | 3.5 | 1.1 | 3.18 |
| Roscovitine | 8 | 2.75 | 2.90 | 0.45 | 0.25 | 1.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Jesus, M.S.M.; Macabeo, A.P.G.; Ramos, J.D.A.; de Leon, V.N.O.; Asamitsu, K.; Okamoto, T. Voacanga globosa Spirobisindole Alkaloids Exert Antiviral Activity in HIV Latently Infected Cell Lines by Targeting the NF-κB Cascade: In Vitro and In Silico Investigations. Molecules 2022, 27, 1078. https://doi.org/10.3390/molecules27031078
de Jesus MSM, Macabeo APG, Ramos JDA, de Leon VNO, Asamitsu K, Okamoto T. Voacanga globosa Spirobisindole Alkaloids Exert Antiviral Activity in HIV Latently Infected Cell Lines by Targeting the NF-κB Cascade: In Vitro and In Silico Investigations. Molecules. 2022; 27(3):1078. https://doi.org/10.3390/molecules27031078
Chicago/Turabian Stylede Jesus, Ma. Sheila M., Allan Patrick G. Macabeo, John Donnie A. Ramos, Von Novi O. de Leon, Kaori Asamitsu, and Takashi Okamoto. 2022. "Voacanga globosa Spirobisindole Alkaloids Exert Antiviral Activity in HIV Latently Infected Cell Lines by Targeting the NF-κB Cascade: In Vitro and In Silico Investigations" Molecules 27, no. 3: 1078. https://doi.org/10.3390/molecules27031078
APA Stylede Jesus, M. S. M., Macabeo, A. P. G., Ramos, J. D. A., de Leon, V. N. O., Asamitsu, K., & Okamoto, T. (2022). Voacanga globosa Spirobisindole Alkaloids Exert Antiviral Activity in HIV Latently Infected Cell Lines by Targeting the NF-κB Cascade: In Vitro and In Silico Investigations. Molecules, 27(3), 1078. https://doi.org/10.3390/molecules27031078