LC-ESI-MS/MS Identification of Biologically Active Phenolics in Different Extracts of Alchemilla acutiloba Opiz
Abstract
:1. Introduction
2. Results and Discussion
2.1. Phytochemical Analysis
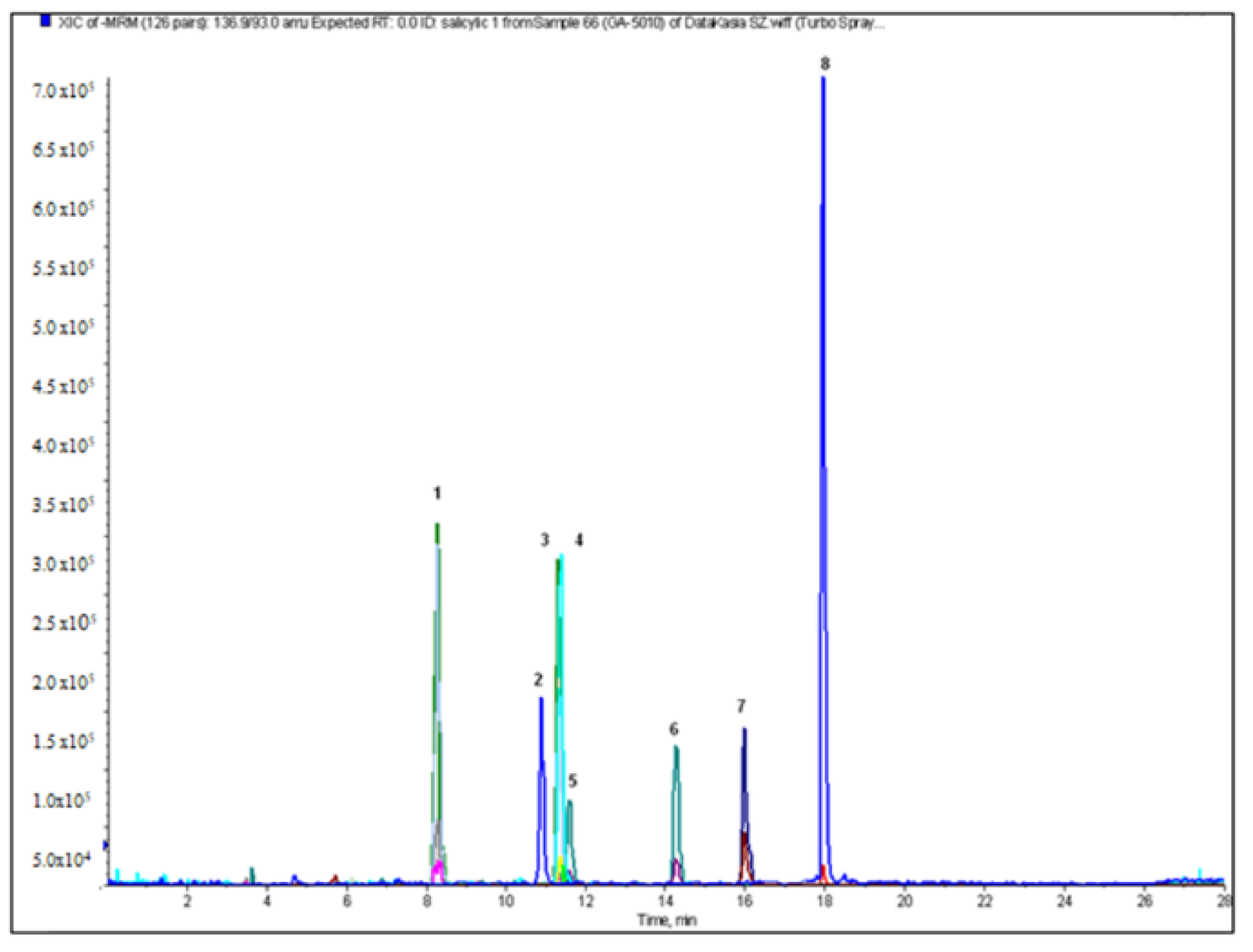
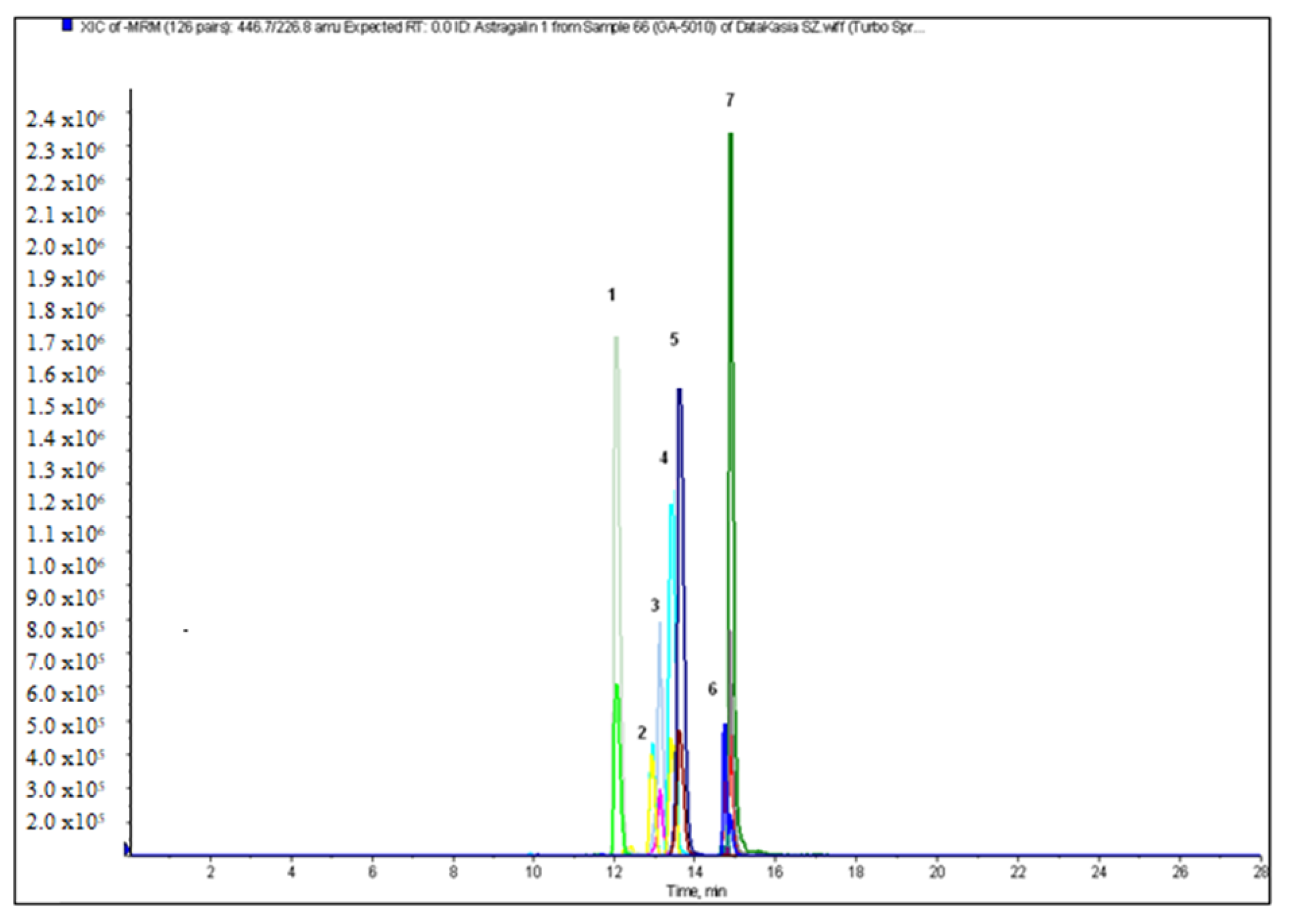
2.2. Qualitative and Quantitative Analysis
2.3. Antioxidant Activities
2.4. Anti-Inflammatory Activity
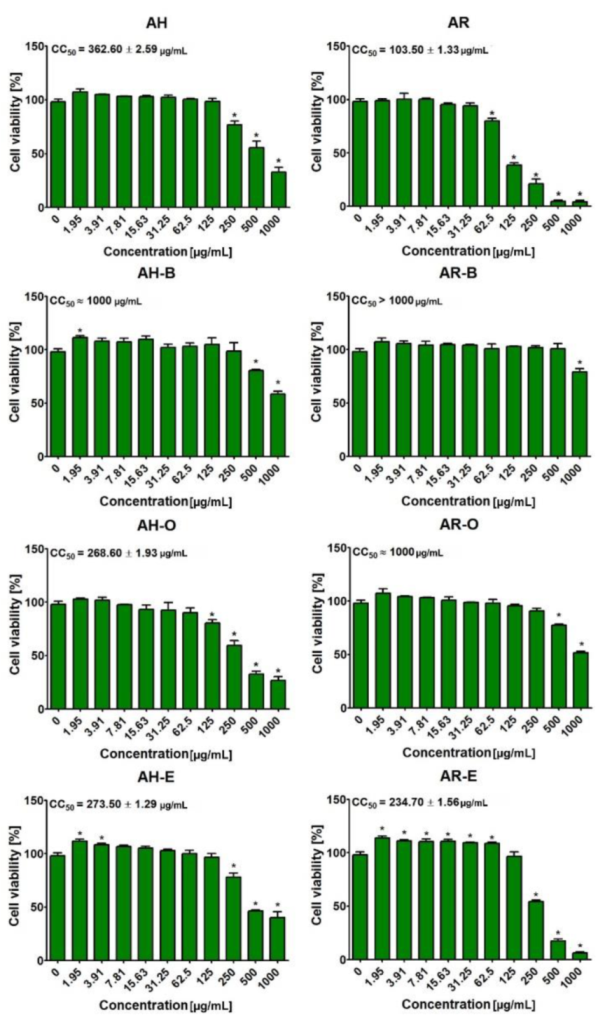
2.5. Evaluation of Cytotoxicity
3. Materials and Methods
3.1. Plant Material
3.2. Chemicals and Reagents
3.3. Extraction Procedure
3.4. Total Flavonoid, Phenolic and Phenolic Acids Content
3.5. LC-ESI-MS/MS Analysis
3.6. Antioxidant Activity
3.7. Cyclooxygenase-1 (COX-1) and Cyclooxygenase-2 (COX-2) Inhibitory Activity
3.8. Evaluation of Cytotoxicity
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Jarić, S.; Mačukanović-Jocić, M.; Djurdjević, L.; Mitrović, M.; Kostić, O.; Karadžić, B.; Pavlović, P. An ethnobotanical survey of traditionally used plants on Suva planina mountain (south-eastern Serbia). J. Ethnopharmacol. 2015, 175, 93–108. [Google Scholar] [CrossRef]
- Ilić-Stojanović, S.; Nikolić, V.; Kundaković, T.; Savić, I.; Savić-Gajić, I.; Jocić, E.; Nikolić, L. Thermosensitive hydrogels for modified release of ellagic acid obtained from Alchemilla vulgaris L. extract. Int. J. Polym. Mater. 2018, 67, 553–563. [Google Scholar] [CrossRef] [Green Version]
- Ożarowski, A. Ziołolecznictwo. Poradnik dla Lekarzy, 3rd ed.; PZWL: Warsaw, Poland, 1982; pp. 53–54. [Google Scholar]
- Ożarowski, A.; Jaroniewski, W. Rośliny Lecznicze i ich Praktyczne Zastosowanie, 1st ed.; IWZZ: Warsaw, Poland, 1987; pp. 318–319. [Google Scholar]
- Shrivastava, R.; Cucuat, N.; John, G.W. Effects of Alchemilla vulgaris and glycerine on epithelial and myofibroblast cell growth and cutaneous lesion healing in rats. Phytother. Res. 2007, 21, 369–373. [Google Scholar] [CrossRef]
- Vitkova, A.; Nikolova, M.; Delcheva, M.; Tashev, A.; Gavrilova, A.; Aneva, I.; Dimitrov, D. Influence of species composition on total phenolic content and antioxidant properties of Herba Alchemillae. Bulg. J. Agric. Sci. 2015, 21, 990–997. [Google Scholar]
- Ertürk, S.; Karatoprak, G.; Koşar, M. Antioxidant properties and phenolic composition of Alchemilla mollis from Turkey. Planta Med. 2011, 77, 1383–1384. [Google Scholar]
- Kaya, B.; Menemen, Y.; Saltan, Z.F. Flavonoids in the endemic species of Alchemilla L. (Section Alchemilla L. Subsection Calycanthum Rothm. Ser. Elatae Rothm.) from Northeast Black sea region in Turkey. Pal. J. Bot. 2012, 44, 595–597. [Google Scholar]
- Duckstein, S.; Lotter, E.; Mayer, U.; Lindequist, U.; Stintzing, F. Phenolic constituents from Alchemilla vulgaris L. and Alchemilla mollis (Buser) Rothm. at different dates of harvest. Z. Naturforsch. C. 2013, 68, 529–540. [Google Scholar]
- Falchero, L.; Coppa, M.; Esposti, S.; Tava, A. Essential oil composition of Alchemilla alpina L. em. Buser from Western Alpine Pastures. J. Essent. Oil Res. 2008, 20, 542–545. [Google Scholar] [CrossRef]
- Ayaz, F.A.; Ayirlioglu-Ayaz, S.; Beyazoglu, O. Fatty acid composition of leaf lipids of some Alchemilla L (Rosaceae) species from Northeast Anatolia (Turkey). Grasas Aceites. 1999, 50, 341–344. [Google Scholar] [CrossRef] [Green Version]
- Matthaus, B.; Özcan, M.M. Fatty acid, tocopherol and squalene contents of Rosaceae seed oils. Bot. Stud. 2014, 55, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngawhirunpat, T.; Opanasopi, P.; Sukma, M.; Sittisombut, C.; Kat, A.; Adachi, I. Antioxidant, free radical-scavenging activity and cytotoxicity of different solvent extracts and their phenolic constituents from the fruit hull of mangosteen (Garcinia mangostana). Pharm. Biol. 2010, 48, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Neagu, E.; Paun, G.; Albu, C.; Radu, G.-L. Assessment of acetylcholinesterase and tyrosinase inhibitory and antioxidant activity of Alchemilla vulgaris and Filipendula ulmaria extracts. J. Taiwan Inst. Chem. Eng. 2015, 52, 1–6. [Google Scholar] [CrossRef]
- Boroja, T.; Mihailović, V.; Katanić, J.; Pan, S.-P.; Nikles, S.; Imbimbo, P.; Monti, D.M.; Stanković, N.; Stanković, M.S.; Bauer, R. The biological activities of roots and aerial parts of Alchemilla vulgaris L. S. Afr. J. Bot. 2018, 116, 175–184. [Google Scholar] [CrossRef]
- Karatoprak, G.Ş.; Ilgün, S.; Koşar, M. Phenolic composition, anti-inflammatory, antioxidant, and antimicrobial activities of Alchemilla mollis (BUSER) ROTHM. Chem. Biodivers. 2017, 14, e1700150. [Google Scholar] [CrossRef]
- Szewczyk, K.; Bogucka-Kocka, A.; Vorobets, N.; Grzywa-Celińska, A.; Granica, S. Phenolic composition of the leaves of Pyrola rotundifolia L. and their antioxidant and cytotoxic activity. Molecules 2020, 25, 1749. [Google Scholar] [CrossRef] [Green Version]
- Nikolova, M.; Dincheva, I.; Vitkova, A.; Badjakov, I. Phenolic acids and free radical scavenging activity of Alchemilla jumrukczalica Pawl. Int. J. Pharm. Sci. Res. 2012, 3, 802–804. [Google Scholar]
- Eldeen, I.M.S.; Van Staden, J. Cyclooxygenase inhibition and antimycobacterial effects of extracts from Sudanese medicinal plants. S. Afr. J. Bot. 2008, 74, 225–229. [Google Scholar] [CrossRef] [Green Version]
- Trouillas, P.; Calliste, C.; Allais, D.; Simon, A.; Marfak, A.; Delage, C.; Duroux, J. Antioxidant, anti-inflammatory and antiproliferative properties of sixteen water plant extracts used in the Limousin countryside as herbal teas. Food Chem. 2003, 80, 399–407. [Google Scholar] [CrossRef]
- Polish Pharmacopoeia IX, PTFarm; Polish Pharmaceutical Society: Warsaw, Poland, 2011; p. 150.
- Klimek, K.; Strubińska, J.; Czernel, G.; Ginalska, G.; Gagoś, M. In vitro evaluation of antifungal and cytotoxic activities as also the therapeutic safety of the oxidized form of amphotericin B. Chem. Biol. Interact. 2016, 256, 47–54. [Google Scholar] [CrossRef]
- Gallol, L.C.; Bucalá, V.; Rigo, M.V.R.; Piña, J. Herbal Medicine: Dry extracts production and applications. In Plant Extracts: Role in Agriculture, Health Effects and Medical Applications; Giordano, A., Costs, A., Eds.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2013; pp. 171–198. [Google Scholar]
- Kicel, A.; Kolodziejczyk-Czepas, J.; Owczarek, A.; Marchelak, A.; Sopinska, M.; Ciszewski, P.; Nowak, P.; Olszewska, M.A. Polyphenol-rich extracts from Cotoneaster leaves inhibit pro-inflammatory enzymes and protect human plasma components against oxidative stress in vitro. Molecules 2018, 23, 2472. [Google Scholar] [CrossRef] [Green Version]
- Pietrzak, W.; Nowak, R.; Olech, M. Effect of extraction method on phenolic content and antioxidant activity of mistletoe extracts from Viscum album subsp. abietis. Chem. Pap. 2014, 68, 976–982. [Google Scholar] [CrossRef]
- Olech, M.; Nowak, R. Influence of different extraction procedures on the antiradical activity and phenolic profile of Rosa rugosa petals. Acta Pol. Pharm. 2012, 69, 501–507. [Google Scholar] [PubMed]
| Sample | Total Flavonoid Content [mg QE/g DE] | Total Phenolic Content [mg GAE/g DE] | Total Phenolic Acids [mg CAE/g DE] |
|---|---|---|---|
| AH | 113.79 ± 1.09 | 279.82 ± 1.52 | 39.15 ± 0.18 |
| AH-B | 57.09 ± 0.31 | 215.61 ± 2.10 | 72.19 ± 0.39 |
| AH-O | 189.25 ± 0.95 | 154.29 ± 1.09 | 11.09 ± 0.01 |
| AH-E | 1.57 ± 0.02 | 95.72 ± 0.89 | nd |
| AR | 19.85 ± 0.19 | 192.54 ± 0.49 | 6.52 ± 0.02 |
| AR-B | 4.63 ± 0.10 | 175.19 ± 1.15 | 9.32 ± 0.03 |
| AR-O | 23.18 ± 0.50 | 93.15 ± 0.95 | 2.15 ± 0.10 |
| AR-E | 1.92 ± 0.01 | 128.37 ± 1.10 | 5.89 ± 0.05 |
| Compound | AH | AR | AH-B | AR-B | AH-O | AR-O | AH-E | AR-E |
|---|---|---|---|---|---|---|---|---|
| gallic acid | 5.89 ± 0.05 | 2.45 ± 0.10 | <LOQ | <LOQ | <LOQ | nd | 0.00 | 0.00 |
| protocatechuic acid | 22.13 ± 0.18 | 0.66 ± 0.02 | 32.88 ± 0.18 | 5.71 ± 0.01 | 19.42 ± 0.12 | 0.80 ± 0.00 | 0.87 ± 0.01 | 2.41 ± 0.03 |
| caffeic acid | 23.50 ± 0.18 | 0.00 | 78.88 ± 0.18 | 9.71 ± 0.10 | 9.82 ± 0.02 | <LOQ | 0.28 ± 0.00 | 1.26 ± 0.01 |
| syringic acid | 4.98 ± 0.11 | 0.00 | 13.01 ± 0.02 | 1.81 ± 0.13 | 4.40 ± 1.25 | 0.00 | 0.00 | <LOQ |
| 4-hydroxybenzoic acid | 12.81 ± 0.09 | <LOQ | 70.36 ± 0.18 | 13.04 ± 0.05 | 5.58 ± 0.28 | < LOQ | 1.43 ± 0.02 | 2.16 ± 0.04 |
| vanilic acid | 12.56 ± 0.70 | 0.00 | 76.75 ± 0.35 | 9.32 ± 0.05 | <LOQ | 0.00 | 0.00 | 0.00 |
| gentisic acid | 0.00 | <LOQ | <LOQ | <LOQ | 3.10 ± 0.05 | 0.24 ± 0.01 | 0.00 | 0.00 |
| sinapic acid | 0.00 | 0.00 | <LOQ | <LOQ | 0.00 | 0.00 | 0.00 | 0.00 |
| p-coumaric acid | 20.88 ± 0.18 | <LOQ | 187.63 ± 0.18 | 24.79 ± 0.20 | 4.26 ± 0.01 | <LOQ | 1.84 ± 0.01 | 4.47 ± 0.05 |
| ferulic acid | 0.00 | 0.00 | 0.00 | 15.93 ± 0.10 | 0.00 | 0.00 | <LOQ | <LOQ |
| rosmarinic acid | 7.56 ± 0.52 | 0.14 ± 0.00 | 0.84 ± 0.01 | 0.02 ± 0.00 | 5.87 ± 0.02 | 0.31 ± 0.01 | 0.04 ± 0.00 | 0.05 ± 0.00 |
| salicylic acid | 11.59 ± 0.05 | <LOQ | 55.50 ± 0.35 | 6.01 ± 0.02 | 7.52 ± 0.12 | <LOQ | 0.17 ± 0.01 | 0.63 ± 0.00 |
| Total of Phenolic Acids | 121.90 | 3.25 | 515.85 | 86.34 | 59.97 | 1.35 | 4.63 | 10.98 |
| astragalin | 23.75 ± 0.18 | <LOQ | 7.48 ± 0.11 | 0.33 ± 0.04 | 22.42 ± 0.35 | <LOQ | 0.03 ± 0.00 | 2.25 ± 0.02 |
| quercitrin | 294.38 ± 4.12 | 1.19 ± 0.02 | 395.00 ± 3.54 | 57.36 ± 0.10 | 335.83 ± 1.18 | 4.02 ± 0.19 | 10.24 ± 0.12 | 18.58 ± 0.02 |
| isoquercitrin | 139.38 ± 0.88 | 1.21 ± 0.03 | 22.59 ± 0.16 | 0.66 ± 0.02 | 147.75 ± 1.06 | 5.86 ± 0.03 | 1.67 ± 0.02 | 4.85 ± 0.02 |
| isorhamnetin-3-glucoside | 4.36 ± 0.10 | <LOQ | <LOQ | <LOQ | 2.31±0.01 | <LOQ | <LOQ | <LOQ |
| kaempferol-3-rutinoside | 191.88 ± 0.88 | 37.74 ± 0.07 | 7.41 ± 0.16 | 0.63 ± 0.10 | 244.17 ± 3.54 | 53.91 ± 0.04 | 8.35 ± 0.00 | 12.82 ± 0.16 |
| rutin | 235.00 ± 3.54 | 83.16 ± 0.00 | 5.05 ± 0.60 | < LOQ | 332.50 ± 1.18 | 97.94 ± 0.42 | 15.99 ± 0.08 | 24.97 ± 0.14 |
| narcissoside | 145.00 ± 3.54 | 47.71 ± 0.04 | 5.21 ± 0.12 | 0.58 ± 0.01 | 206.67 ± 4.71 | 61.76 ± 0.00 | 8.05 ± 0.02 | 13.45 ± 0.07 |
| hyperoside | 0.00 | 0.00 | 2.47 ± 0.08 | 0.66 ± 0.02 | 0.00 | 0.00 | 0.00 | 1.52 ± 0.01 |
| tiliroside | 0.00 | 0.00 | <LOQ | <LOQ | 0.00 | 0.00 | 0.00 | 0.00 |
| naringenin-7-glucoside | <LOQ | 0.00 | 0.00 | 0.00 | <LOQ | 0.00 | 0.00 | 0.00 |
| Total of Flavonoid Glycosides | 1033.75 | 171.01 | 445.21 | 60.22 | 1291.65 | 223.49 | 44.33 | 78.44 |
| luteolin | 0.05 ± 0.00 | 0.00 | 0.51 ± 0.00 | 0.02 ± 0.00 | <LOQ | 0.00 | <LOQ | <LOQ |
| kaempferol | 0.00 | 0.00 | 1.23 ± 0.00 | <LOQ | 0.00 | 0.00 | 0.00 | 0.04 ± 0.00 |
| 3-O-methylquercetin | 0.00 | 0.00 | <LOQ | 0.00 | 0.00 | <LOQ | 0.00 | <LOQ |
| naringenin | <LOQ | 0.00 | <LOQ | 0.00 | <LOQ | 0.00 | 0.00 | 0.00 |
| eriodictyol | <LOQ | 0.00 | 0.17 ± 0.00 | <LOQ | <LOQ | 0.00 | 0.00 | 0.00 |
| quercetin | 0.15 ± 0.00 | <LOQ | 22.38 ± 0.39 | 1.04 ± 0.01 | 0.14 ± 0.00 | <LOQ | 0.00 | 0.00 |
| isorhamnetin | <LOQ | 0.00 | 1.89 ± 0.00 | 0.04 ± 0.00 | <LOQ | 0.00 | 0.00 | 0.00 |
| rhamnetin | 0.00 | 0.00 | <LOQ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Total of Flavonoid Aglycones | 0.20 | 0.00 | 26.18 | 1.10 | 0.14 | 0.00 | 0.00 | 0.04 |
| AH | AR | AH-B | AR-B | AH-O | AR-O | AH-E | AR-E | AA | Trolox | Na2EDTA*2H2O | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| DPPH | 18.69 ± 0.04 | 29.87 ± 0.15 | 8.96 ± 0.10 | 12.08 ± 0.18 | 8.83 ± 0.37 | 15.37 ± 0.19 | 41.46 ± 0.32 | 51.42 ± 0.18 | 4.90 ± 0.09 | - | - |
| ABTS | 6.17 ± 0.24 | 14.29 ± 0.06 | 1.42 ± 0.18 | 8.78 ± 0.01 | 6.54 ± 0.03 | 10.39 ± 0.15 | 16.28 ± 0.15 | 24.82 ± 0.20 | - | 3.07 ± 0.01 | - |
| CHEL | 21.60 ± 0.39 | 25.76 ± 0.03 | 11.43 ± 0.18 | 12.33 ± 0.33 | 18.89 ± 0.94 | 19.30 ± 0.22 | 25.51 ± 0.89 | 44.12 ± 0.24 | - | - | 9.45 ± 0.03 |
| COX-1 Inibition [%] ±SD | COX-2 Inhibition [%] ±SD | |||
|---|---|---|---|---|
| Extract | 50 µg/mL | 100 µg/mL | 50 µg/mL | 100 µg/mL |
| AH | 50.17 ± 0.85 | 71.32 ± 2.73 | 43.65 ± 1.36 | 78.52 ± 2.18 |
| AR | 31.27 ± 0.76 | 47.80 ± 1.12 | 48.59 ± 2.19 | 54.25 ± 1.23 |
| AH-B | 76.82 ± 0.69 | 83.14 ± 1.08 | 79.75 ± 1.29 | 95.10 ± 1.81 |
| AR-B | 63.17 ± 1.28 | 78.29 ± 2.25 | 72.64 ± 1.93 | 90.93 ± 2.65 |
| AH-O | 52.65 ± 2.19 | 74.50 ± 1.29 | 60.23 ± 0.67 | 80.12 ± 1.73 |
| AR-O | 45.73 ± 1.45 | 64.49 ± 2.74 | 58.08 ± 1.79 | 79.15 ± 3.10 |
| AH-E | nd | 10.43 ± 0.19 | nd | 28.09 ± 0.13 |
| AR-E | nd | 28.71 ± 0.35 | nd | 41.96 ± 1.30 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dos Santos Szewczyk, K.; Pietrzak, W.; Klimek, K.; Grzywa-Celińska, A.; Celiński, R.; Gogacz, M. LC-ESI-MS/MS Identification of Biologically Active Phenolics in Different Extracts of Alchemilla acutiloba Opiz. Molecules 2022, 27, 621. https://doi.org/10.3390/molecules27030621
Dos Santos Szewczyk K, Pietrzak W, Klimek K, Grzywa-Celińska A, Celiński R, Gogacz M. LC-ESI-MS/MS Identification of Biologically Active Phenolics in Different Extracts of Alchemilla acutiloba Opiz. Molecules. 2022; 27(3):621. https://doi.org/10.3390/molecules27030621
Chicago/Turabian StyleDos Santos Szewczyk, Katarzyna, Wioleta Pietrzak, Katarzyna Klimek, Anna Grzywa-Celińska, Rafał Celiński, and Marek Gogacz. 2022. "LC-ESI-MS/MS Identification of Biologically Active Phenolics in Different Extracts of Alchemilla acutiloba Opiz" Molecules 27, no. 3: 621. https://doi.org/10.3390/molecules27030621







