ICP-MS Multi-Elemental Analysis of the Human Meninges Collected from Sudden Death Victims in South-Eastern Poland
Abstract
:1. Introduction
2. Results
2.1. Correlations of Average Trace Element Concentrations in the Meninges for the Whole Population of Subjects (n = 40)
2.2. Statistically Significant Differences between Meninges
2.3. Statistically Significant Differences between Elemental Composition of Meninges for Controls and Cases
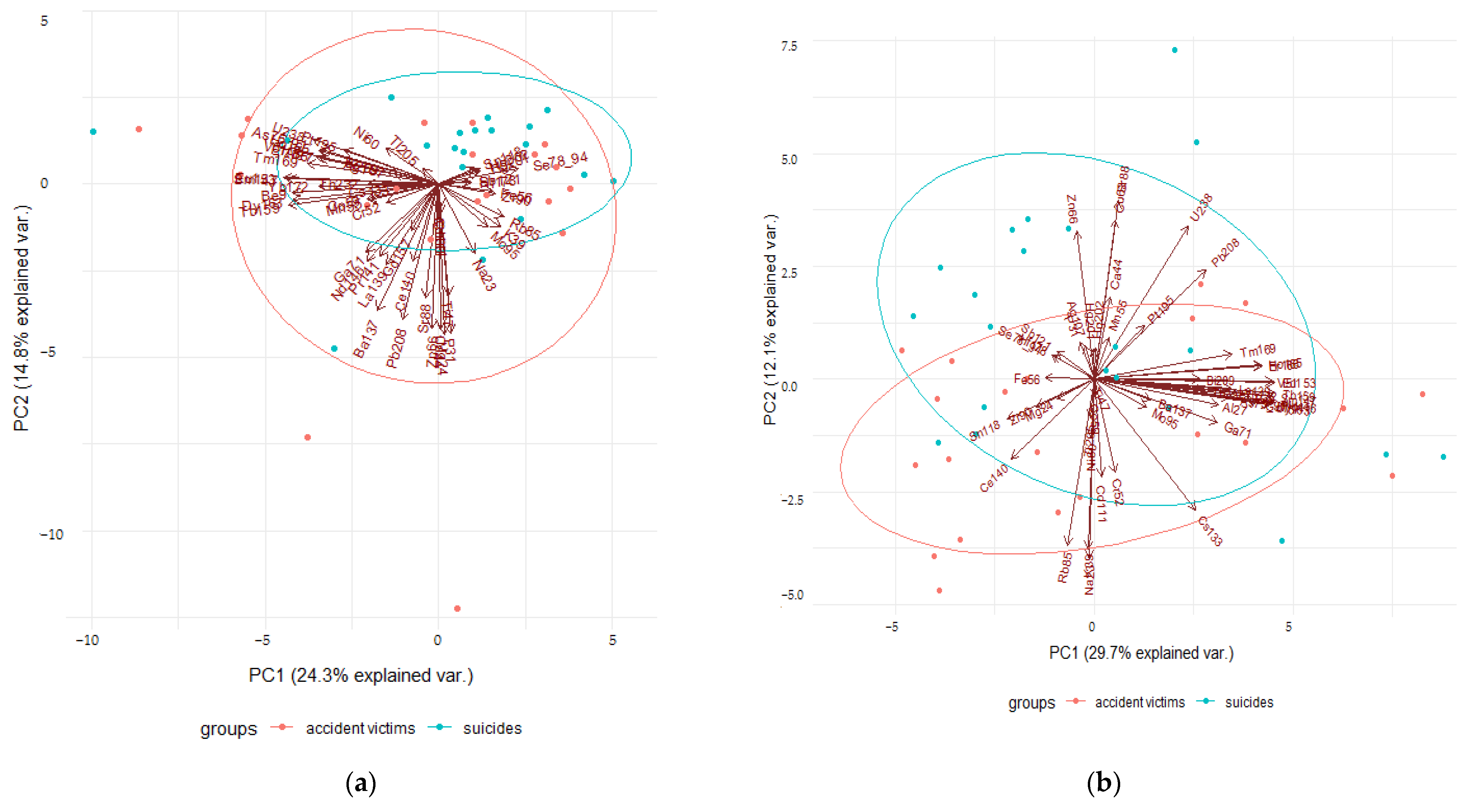
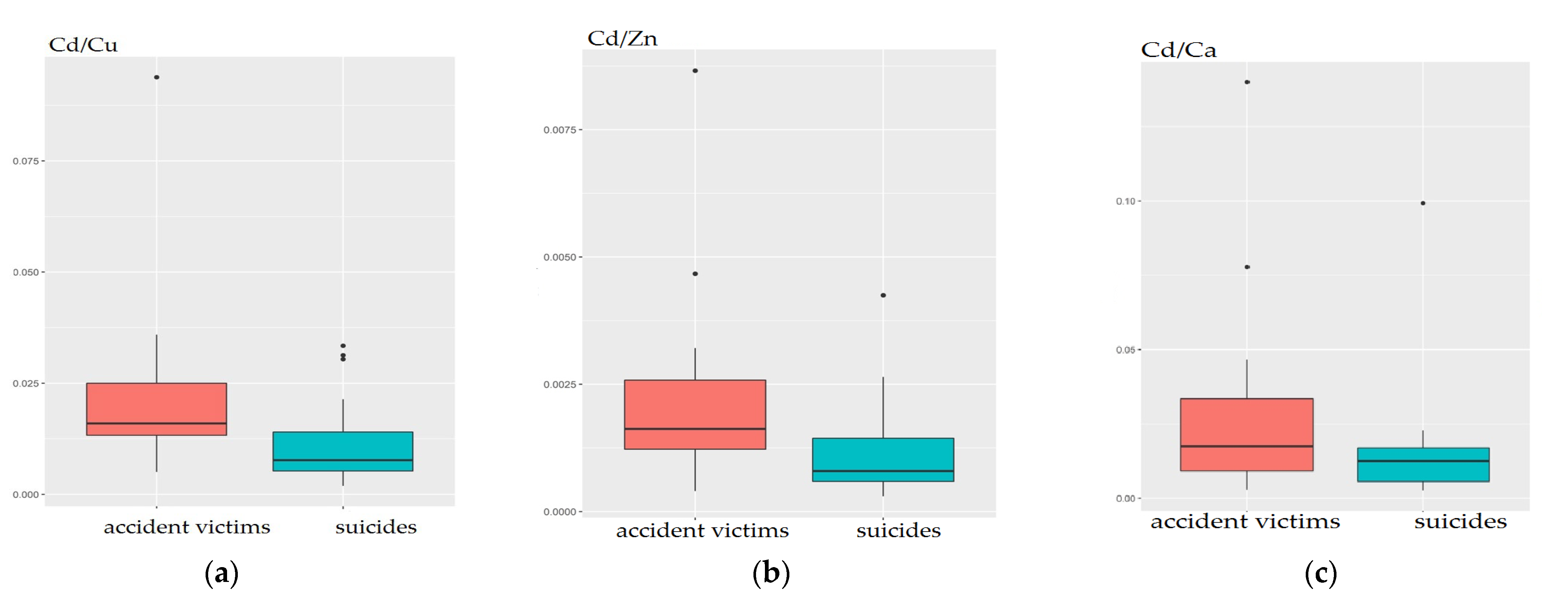
2.4. Statistically Significant Differences between Elements’ Ratios for Suicide Cases in Comparison to Sudden Death from Accidents
3. Discussion
4. Materials and Methods
4.1. Population
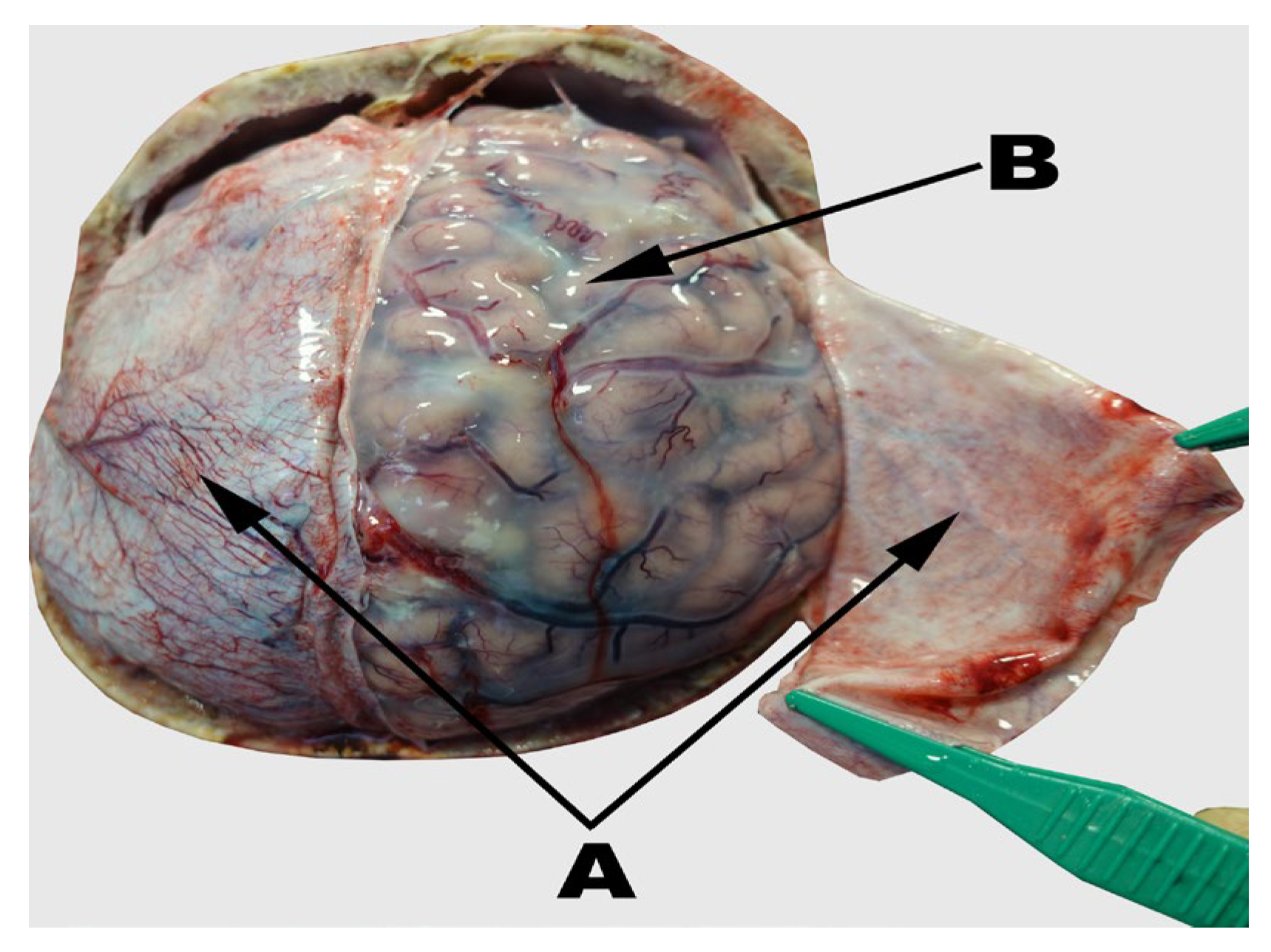
4.2. Sample Collection
4.3. Sample Preparation
4.4. ICP-MS Measurements
4.5. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Andreini, C.; Bertin, I.; Cavallaro, G.; Holliday, G.L.; Thornton, J.M. Metal ions in biological catalysis: From enzyme databases to general principles. J. Biol. Inorg. Chem. 2008, 13, 1205–1218. [Google Scholar] [CrossRef] [PubMed]
- Griffith, G.C.; Butt, E.M.; Walker, J. The inorganic element content of certain human tissues. Ann. Intern. Med. 1954, 41, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Perry, H.M., Jr.; Tipton, I.H.; Schroeder, H.A.; Cook, M.J. Variability in the metal content of human organs. J. Lab. Clin. Med. 1962, 60, 245–253. [Google Scholar] [PubMed]
- Tipton, I.H.; Schroeder, H.A.; Perry, H.M., Jr.; Cook, M.J. Trace Elements in Human Tissue: III. Subjects from Africa, the near and Far East and Europe. Health Phys. 1965, 11, 403–451. [Google Scholar] [CrossRef]
- Duce, J.A.; Bush, A.I. Biological metals and Alzheimer’s disease: Implications for therapeutics and diagnostics. Prog. Neurobiol. 2010, 92, 1–18. [Google Scholar] [CrossRef]
- Verma, P.K.; Sharma, A.; Shankar, H.; Sharma, A.; Rao, D.N. Role of Trace Elements, Oxidative Stress and Immune System: A Triad in Premature Ovarian Failure. Biol. Trace Elem. Res. 2017, 184, 325–333. [Google Scholar] [CrossRef]
- Cicero, C.E.; Mostile, G.; Vasta, R.; Rapisarda, V.; Signorelli, S.S.; Ferrante, M.; Zappia, M.; Nicoletti, A. Metals and neurodegenerative diseases. A systematic review. Environ. Res. 2017, 159, 82–94. [Google Scholar] [CrossRef]
- Grochowski, C.; Radzikowska, E.; Maciejewski, R. Neural stem cell therapy—Brief review. Clin. Neurol. Neurosurg. 2018, 173, 8–14. [Google Scholar] [CrossRef]
- Vilella, A.; Belletti, D.; Sauer, A.K.; Hagmeyer, S.; Sarowar, T.; Masoni, M.; Stasiak, N.; Mulvihill, J.J.; Ruozi, B.; Forni, F.; et al. Reduced plaque size and inflammation in the APP23 mouse model for Alzheimer’s disease after chronic application of polymeric nanoparticles for CNS targeted zinc delivery. J. Trace Elem. Med. Biol. 2018, 49, 210–221. [Google Scholar] [CrossRef]
- Corona, C.; Masciopinto, F.; Silvestri, E.; Del Viscovo, A.; Lattanzio, R.; La Sorda, R.; Ciavardelli, D.; Goglia, F.; Piantelli, M.; Canzoniero, L.M.T.; et al. Dietary zinc supplementation of 3xTg-AD mice increases BDNF levels and prevents cognitive deficits as well as mitochondrial dysfunction. Cell Death Dis. 2010, 1, e91. [Google Scholar] [CrossRef]
- Fourie, C.; Vyas, Y.; Lee, K.; Jung, Y.; Garner, C.C.; Montgomery, J.M. Dietary Zinc Supplementation Prevents Autism Related Behaviors and Striatal Synaptic Dysfunction in Shank3 Exon 13–16 Mutant Mice. Front. Cell. Neurosci. 2018, 12, 374. [Google Scholar] [CrossRef] [Green Version]
- Cezar, L.C.; Kirsten, T.B.; da Fonseca, C.C.N.; de Lima, A.P.N.; Bernardi, M.M.; Felicio, L.F. Zinc as a therapy in a rat model of autism prenatally induced by valproic acid. Prog. Neuro. Psychopharmacol. Biol. Psychiatry 2018, 84, 173–180. [Google Scholar] [CrossRef]
- Kirsten, T.B.; Queiroz-Hazarbassanov, N.; Bernardi, M.M.; Felicio, L.F. Prenatal zinc prevents communication impairments and BDNF disturbance in a rat model of autism induced by prenatal lipopolysaccharide exposure. Life Sci. 2015, 130, 12–17. [Google Scholar] [CrossRef] [Green Version]
- Laur, N.; Kinscherf, R.; Pomytkin, K.; Kaiser, L.; Knes, O.; Deigner, H.-P. ICP-MS trace element analysis in serum and whole blood. PLoS ONE 2020, 15, e0233357. [Google Scholar] [CrossRef]
- Liu, T.; Lu, Q.-B.; Yan, L.; Guo, J.; Feng, F.; Qiu, J.; Wang, J. Comparative Study on Serum Levels of 10 Trace Elements in Schizophrenia. PLoS ONE 2015, 10, e0133622. [Google Scholar] [CrossRef]
- Cai, L.; Chen, T.; Yang, J.; Zhou, K.; Yan, X.; Chen, W.; Sun, L.; Li, L.; Qin, S.; Wang, P.; et al. Serum trace element differences between Schizophrenia patients and controls in the Han Chinese population. Sci. Rep. 2015, 5, 15013. [Google Scholar] [CrossRef] [Green Version]
- Nawi, A.M.; Chin, S.-F.; Jamal, R. Simultaneous analysis of 25 trace elements in micro volume of human serum by inductively coupled plasma mass spectrometry (ICP-MS). Pract. Lab. Med. 2020, 18, e00142. [Google Scholar] [CrossRef]
- Lee, Y.H.; Bang, E.-S.; Lee, J.H.; Lee, J.D.; Kang, D.R.; Hong, J.; Lee, J.M. Serum Concentrations of Trace Elements Zinc, Copper, Selenium, and Manganese in Critically Ill Patients. Biol. Trace Elem. Res. 2019, 188, 316–325. [Google Scholar] [CrossRef] [Green Version]
- Höck, A.; Demmel, U.; Schicha, H.; Kasperek, K.; Feinendegen, L.E. Trace element concentration in human brain. Activation analysis of cobalt, iron, rubidium, selenium, zinc, chromium, silver, cesium, antimony and scandium. Brain 1975, 98, 49–64. [Google Scholar] [CrossRef]
- Grochowski, C.; Blicharska, E.; Krukow, P.; Jonak, K.; Maciejewski, M.; Szczepanek, D.; Jonak, K.; Flieger, J.; Maciejewski, R. Analysis of Trace Elements in Human Brain: Its Aim, Methods, and Concentration Levels. Front. Chem. 2019, 5, 115. [Google Scholar] [CrossRef] [Green Version]
- Grochowski, C.; Szukała, M.; Litak, J.; Budny, A.; Proch, J.; Majerek, D.; Blicharska, E.; Niedzielski, P. Correlations Between Trace Elements in Selected Locations of the Human Brain in Individuals with Alcohol Use Disorder. Molecules 2020, 25, 359. [Google Scholar] [CrossRef] [Green Version]
- Adani, G.; Filippini, T.; Michalke, B.; Vinceti, M. Selenium and Other Trace Elements in the Etiology of Parkinson’s Disease: A Systematic Review and Meta-Analysis of Case-Control Studies. Neuroepidemiology 2020, 54, 1–23. [Google Scholar] [CrossRef]
- De Benedictis, C.A.; Vilella, A.; Grabrucker, A.M. The Role of Trace Metals in Alzheimer’s Disease. In Alzheimer’s Disease; Wisniewski, T., Ed.; Codon Publications: Brisbane, Australia, 2018. [Google Scholar]
- Cilliers, K.; Muller, C.J.F.; Page, B.J. Trace Element Concentration Changes in Brain Tumors: A Review. Anat. Rec. 2020, 303, 1293–1299. [Google Scholar] [CrossRef]
- Grochowski, C.; Blicharska, E.; Bogucki, J.; Proch, J.; Mierzwińska, A.; Baj, J.; Litak, J.; Podkowiński, A.; Flieger, J.; Teresiński, G.; et al. Increased Aluminum Content in Certain Brain Structures is Correlated with Higher Silicon Concentration in Alcoholic Use Disorder. Molecules 2019, 24, 1721. [Google Scholar] [CrossRef] [Green Version]
- Fukada, T.; Yamasaki, S.; Nishida, K.; Murakami, M.; Hirano, T. Zinc homeostasis and signaling in health and diseases. JBIC J. Biol. Inorg. Chem. 2011, 16, 1123–1134. [Google Scholar] [CrossRef] [Green Version]
- Clardy, S.L.; Wang, X.; Zhao, W.; Liu, W.; Chase, G.A.; Beard, J.L.; Felt, B.T.; Connor, J.R. Acute and chronic effects of developmental iron deficiency on mRNA expression patterns in the brain. In Oxidative Stress and Neuroprotection; Parvez, H., Riederer, P., Eds.; Springer: Vienna, Austria, 2006. [Google Scholar]
- Georgieff, M.K. The role of iron in neurodevelopment: Fetal iron deficiency and the developing hippocampus. Biochem. Soc. Trans. 2008, 36, 1267–1271. [Google Scholar] [CrossRef] [Green Version]
- Yasuda, H.; Yoshida, K.; Yasuda, Y.; Tsutsui, T. Infantile zinc deficiency: Association with autism spectrum disorders. Sci. Rep. 2011, 1, 129. [Google Scholar] [CrossRef]
- Grabrucker, A.M. Environmental factors in autism. Front. Psychiatry 2013, 3, 118. [Google Scholar] [CrossRef] [Green Version]
- Bjørklund, G. The role of zinc and copper in autism spectrum disorders. Acta Neurobiol. Exp. 2013, 73, 225–236. [Google Scholar] [PubMed]
- Kaler, S.G. Diagnosis and therapy of Menkes syndrome, a genetic form of copper deficiency. Am. J. Clin. Nutr. 1998, 67, 1029S–1034S. [Google Scholar] [CrossRef] [Green Version]
- Kodama, H.; Fujisawa, C. Copper metabolism and inherited copper transport disorders: Molecular mechanisms, screening, and treatment. Metallomics 2009, 1, 42–52. [Google Scholar] [CrossRef]
- Stewart, T.J. Across the spectrum: Integrating multidimensional metal analytics for in situ metallomic imaging. Metallomics 2019, 11, 29–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blicharska, E.; Flieger, J.; Huber, M.; Kocjan, R. Examination of the Elemental Composition of Hair in Cholelithiasis, Kidney Stone, Hypertension and Diabetes by Scanning Electron Microscopy and Energy Dispersive Spectrometry SEM/EDS. J. Anal. Bioanal. Tech. 2014, 5, 207. [Google Scholar] [CrossRef] [Green Version]
- Baj, J.; Forma, A.; Sitarz, E.; Karakuła, K.; Flieger, W.; Sitarz, M.; Grochowski, C.; Maciejewski, R.; Karakula-Juchnowicz, H. Beyond the Mind-Serum Trace Element Levels in Schizophrenic Patients: A Systematic Review. Int. J. Mol. Sci. 2020, 21, 9566. [Google Scholar] [CrossRef] [PubMed]
- Dolar-Szczasny, J.; Święch, A.; Flieger, J.; Tatarczak-Michalewska, M.; Niedzielski, P.; Proch, J.; Majerek, D.; Kawka, J.; Mackiewicz, J. Levels of Trace Elements in the Aqueous Humor of Cataract Patients Measured by the Inductively Coupled Plasma Optical Emission Spectrometry. Molecules 2019, 24, 4127. [Google Scholar] [CrossRef] [Green Version]
- Konz, I.; Fernandez, B.; Fernández, M.L.; Pereiro, R.; González-Iglesias, H.; Coca-Prados, M.; Sanz-Medel, A. Quantitative bioimaging of trace elements in the human lens by LA-ICP-MS. Anal. Bioanal. Chem. 2014, 406, 2343–2348. [Google Scholar] [CrossRef] [PubMed]
- Baj, J.; Flieger, W.; Teresiński, G.; Buszewicz, G.; Sitarz, E.; Forma, A.; Karakuła, K.; Maciejewski, R. Magnesium, Calcium, Potassium, Sodium, Phosphorus, Selenium, Zinc, and Chromium Levels in Alcohol Use Disorder: A Review. J. Clin. Med. 2020, 9, 1901. [Google Scholar] [CrossRef] [PubMed]
- Krebs, N.; Langkammer, C.; Goessler, W.; Ropele, S.; Fazekas, F.; Yen, K.; Scheurer, E. Assessment of trace elements in human brain using inductively coupled plasma mass spectrometry. J. Trace Elem. Med. Biol. 2014, 28, 1–7. [Google Scholar] [CrossRef]
- Becker, J.S.; Zoriy, M.V.; Pickhardt, C.; Palomero-Gallagher, N.; Zilles, K. Imaging of copper, zinc, and other elements in thin section of human brain samples (hippocampus) by laser ablation inductively coupled plasma mass spectrometry. Anal. Chem. 2005, 77, 3208–3216. [Google Scholar] [CrossRef] [Green Version]
- Turecki, G. The molecular bases of the suicidal brain. Nat. Rev. Neurosci. 2014, 15, 802–816. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, M.; Matsugi, E.; Miyasaki, K. PIXE measurement applied to trace elemental analysis of human tissues. Nucl. Instrum. Meth. B. 1987, 22, 152–155. [Google Scholar] [CrossRef]
- Ishii, K. PIXE and Its Applications to Elemental Analysis. Quantum Beam Sci. 2019, 3, 12. [Google Scholar] [CrossRef] [Green Version]
- Rahil-Khazen, R.; Bolann, B.J.; Myking, A.; Ulvik, R.J. Multi-element analysis of trace element levels in human autopsy tissues by using inductively coupled atomic emission spectrometry technique (ICP-AES). J. Trace Elem. Med. Biol. 1987, 16, 15–25. [Google Scholar] [CrossRef]
- Peters, J.L.; Kubzansky, L.D.; Ikeda, A.; Spiro, A., 3rd; Wright, R.O.; Weisskopf, M.G.; Kim, D.; Sparrow, D.; Nie, L.H.; Hu, H.; et al. Childhood and adult socioeconomic position, cumulative lead levels, and pessimism in later life: The VA Normative Aging Study. Am. J. Epidemiol. 2011, 174, 1345–1353. [Google Scholar] [CrossRef]
- Weiss, B.; Cory-Slechta, D.; Gilbert, S.G.; Mergler, D.; Miller, E.; Miller, C.; Newland, M.C.; Rice, D.; Schettler, T. The new tapestry of risk assessment. Neurotoxicology 2008, 29, 883–890. [Google Scholar] [CrossRef] [Green Version]
- Peen, J.; Schoevers, R.A.; Beekman, A.T.; Dekker, J. The current status of urban-rural differences in psychiatric disorders. Acta Psychiatr. Scand. 2010, 121, 84–93. [Google Scholar] [CrossRef]
- Dekker, J.; Peen, J.; Koelen, J.; Smit, F.; Schoevers, R. Psychiatric disorders and urbanization in Germany. BMC Public Health 2008, 8, 17. [Google Scholar] [CrossRef]
- Miura, S.; Takahashi, K.; Imagawa, T.; Uchida, K.; Saito, S.; Tominaga, M.; Ohta, T. Involvement of TRPA1 activation in acute pain induced by cadmium in mice. Mol. Pain. 2013, 9, 7. [Google Scholar] [CrossRef] [Green Version]
- Marchetti, C. Interaction of metal ions with neurotransmitter receptors and potential role in neurodiseases. Biometals 2014, 27, 1097–1113. [Google Scholar] [CrossRef]
- Matović, V.; Buha, A.; Bulat, Z.; Dukić-Ćosić, D. Cadmium toxicity revisited: Focus on oxidative stress induction and interactions with zinc and magnesium. Arh. Hig. Rada Toksikol. 2011, 62, 65–76. [Google Scholar] [CrossRef] [Green Version]
- Nemmiche, S. Oxidative Signaling Response to Cadmium Exposure. Toxicol. Sci. 2017, 156, 4–10. [Google Scholar] [CrossRef] [Green Version]
- Branca, J.J.V.; Morucci, G.; Maresca, M.; Tenci, B.; Cascella, R.; Paternostro, F.; Ghelardini, C.; Gulisano, M.; Di Cesare Mannelli, L.; Pacini, A. Selenium and zinc: Two key players against cadmium-induced neuronal toxicity. Toxicol. Vitr. 2018, 48, 48159–48169. [Google Scholar] [CrossRef] [Green Version]
- Usai, C.; Barberis, A.; Moccagatta, L.; Marchetti, C. Pathways of cadmium influx in mammalian neurons. J. Neurochem. 1999, 72, 2154–2161. [Google Scholar] [CrossRef] [Green Version]
- Choong, G.; Liu, Y.; Templeton, D.M. Interplay of calcium and cadmium in mediating cadmium toxicity. Chem. Bio. Interact. 2014, 211, 54–65. [Google Scholar] [CrossRef]
- Zhang, Y.; Gu, A.Z.; Cen, T.; Li, X.; He, M.; Li, D.; Chen, J. Sub-Inhibitory Concentrations of Heavy Metals Facilitate the Hori- 542 zontal Transfer of Plasmid-Mediated Antibiotic Resistance Genes In Water Environment. Environ. Pollut. 2018, 237, 74–82. [Google Scholar] [CrossRef]
- Lu, J.; Wang, Y.; Jin, M.; Yuan, Z.; Bond, P.; Guo, J. Both Silver Ions and Silver Nanoparticles Facilitate The Horizontal Transfer Of Plasmid-Mediated Antibiotic Resistance Genes. Water Res. 2020, 169, 115229. [Google Scholar] [CrossRef]
- Bussan, D.D.; Snaychuk, L.; Bartzas, G.; Douvris, C. Quantification of trace elements in surgical and KN95 face masks widely used during the SARS-COVID-19 pandemic. Sci. Total Environ. 2022, 814, 151924. [Google Scholar] [CrossRef]
- van der Schot, A.C.; Vonk, R.; Brans, R.G.; van Haren, N.E.; Koolschijn, P.C.; Nuboer, V.; Schnack, H.G.; van Baal, G.C.; Boomsma, D.I.; Nolen, W.A.; et al. Influence of genes and environment on brain volumes in twin pairs concordant and discordant for bipolar disorder. Arch. Gen. Psychiatry 2009, 66, 142–151. [Google Scholar] [CrossRef] [Green Version]
- Orisakwe, O.E. The role of lead and cadmium in psychiatry. N. Am. J. Med. Sci. 2014, 6, 370–376. [Google Scholar] [CrossRef] [Green Version]
- González-Estecha, M.; Trasobares, E.M.; Tajima, K.; Cano, S.; Fernández, C.; López, J.L.; Unzeta, B.; Arroyo, M.; Fuentenebro, F. Trace elements in bipolar disorder. J. Trace Elem. Med. Biol. 2011, 25, S78–S83. [Google Scholar] [CrossRef]
- Schafer, J.H.; Glass, T.A.; Bressler, J.; Todd, A.C.; Schwartz, B.S. Blood lead is a predictor of homocysteine levels in a population-based study of older adults. Environ. Health Perspect. 2005, 113, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Guallar, E.; Silbergeld, E.K.; Navas-Acien, A.; Malhotra, S.; Astor, B.C.; Sharrett, A.R.; Schwartz, B.S. Confounding of the relation between homocysteine and peripheral arterial disease by lead, cadmium, and renal function. Am. J. Epidemiol. 2006, 163, 700–708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López, E.; Figueroa, S.; Oset-Gasque, M.J.; González, M.P. Apoptosis and necrosis: Two distinct events induced by cadmium in cortical neurons in culture. Br. J. Pharmacol. 2003, 138, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Pacini, S.; Punzi, T.; Morucci, G.; Gulisano, M.; Ruggiero, M. A paradox of cadmium: A carcinogen that impairs the capability of human breast cancer cells to induce angiogenesis. J. Environ. Pathol. Toxicol. Oncol. 2009, 28, 85–88. [Google Scholar] [CrossRef]
- Scinicariello, F.; Buser, M. Blood cadmium and depressive symptoms in young adults (aged 20–39 years). Psychol. Med. 2015, 45, 807–815. [Google Scholar] [CrossRef] [Green Version]
- Fevrier-Paul, A.; Soyibo, A.K.; Mitchell, S.; Voutchkov, M. Role of Toxic Elements in Chronic Kidney Disease. J. Health Pollut. 2018, 8, 181202. [Google Scholar] [CrossRef]
- van Os, J.; Hanssen, M.; Bijl, R.V.; Vollebergh, W. Prevalence of psychotic disorder and community level of psychotic symptoms: An urban-rural comparison. Arch. Gen. Psychiatry 2001, 58, 663–668. [Google Scholar] [CrossRef] [Green Version]
- Stanley, P.C.; Wakwe, V.C. Toxic trace metals in the mentally ill patients. Niger Postgrad. Med. J. 2002, 9, 199–204. [Google Scholar]
- Karim, P.; Hossain, M.I.; Nazmus Sadat, A.F.; Nahar, Z.; Md Khalid Hossain, M.K.; Hasnat, A. Serum levels of cadmium, calcium, lead and iron in schizophrenic patients. Dhaka Univ. J. Pharm. Sci. 2006, 5, 9–13. [Google Scholar] [CrossRef]
- Arinola, G.; Idonije, B.; Akinlade, K.; Ihenyen, O. Essential trace metals and heavy metals in newly diagnosed schizophrenic patients and those on anti-psychotic medication. J. Res. Med. Sci. 2010, 15, 245–249. [Google Scholar]
- Baker, E.L.; Feldman, R.G.; White, R.F.; Harley, J.P. The role of occupational lead exposure in the genesis of psychiatric and behavioral disturbances. Acta Psychiatr. Scand. Suppl. 1983, 303, 38–48. [Google Scholar] [CrossRef]
- Maizlish, N.A.; Parra, G.; Feo, O. Neurobehavioural evaluation of Venezuelan workers exposed to inorganic lead. Occup. Environ. Med. 1995, 52, 408–414. [Google Scholar] [CrossRef] [Green Version]
- Rhodes, D.; Spiro, A., 3rd; Aro, A.; Hu, H. Relationship of bone and blood lead levels to psychiatric symptoms: The normative aging study. J. Occup. Environ. Med. 2003, 45, 1144–1151. [Google Scholar] [CrossRef]
- Schwartz, B.S.; Lee, B.K.; Bandeen-Roche, K.; Stewart, W.; Bolla, K.; Links, J.; Weaver, V.; Todd, A. Occupational lead exposure and longitudinal decline in neurobehavioral test scores. Epidemiology 2005, 16, 106–113. [Google Scholar] [CrossRef]
- Opler, M.G.; Buka, S.L.; Groeger, J.; McKeague, I.; Wei, C.; Factor-Litvak, P.; Bresnahan, M.; Graziano, J.; Goldstein, J.M.; Seidman, L.J.; et al. Prenatal exposure to lead, delta-aminolevulinic acid, and schizophrenia: Further evidence. Environ. Health Perspect. 2008, 116, 1586–1590. [Google Scholar] [CrossRef] [Green Version]
- Bouchard, M.F.; Bellinger, D.C.; Weuve, J.; Matthews-Bellinger, J.; Gilman, S.E.; Wright, R.O.; Schwartz, J.; Weisskopf, M.G. Blood lead levels and major depressive disorder, panic disorder, and generalized anxiety disorder in US young adults. Arch. Gen. Psychiatry 2009, 66, 1313–1319. [Google Scholar] [CrossRef]
- Eum, K.D.; Korrick, S.A.; Weuve, J.; Okereke, O.; Kubzansky, L.D.; Hu, H.; Weisskopf, M.G. Relation of cumulative low-level lead exposure to depressive and phobic anxiety symptom scores in middle-age and elderly women. Environ. Health Perspect. 2012, 120, 817–823. [Google Scholar] [CrossRef] [Green Version]
- McFarlane, A.C.; Searle, A.K.; Van Hooff, M.; Baghurst, P.A.; Sawyer, M.G.; Galletly, C.; Sim, M.R.; Clark, L.S. Prospective associations between childhood low-level lead exposure and adult mental health problems: The Port Pirie cohort study. Neurotoxicology 2013, 39, 11–17. [Google Scholar] [CrossRef]
- Cobley, J.N.; Fiorello, M.L.; Bailey, D.M. 13 reasons why the brain is susceptible to oxidative stress. Redox Biol. 2018, 15, 490–503. [Google Scholar] [CrossRef]
- Mahaffey, K.R.; Gartside, P.S.; Glueck, C.J. Blood lead levels and dietary calcium intake in 1- to 11-year-old children: The Second National Health and Nutrition Examination Survey, 1976 to 1980. Pediatrics 1986, 78, 257–262. [Google Scholar] [CrossRef]
- Isani, G.; Carpenč, E. Metallothioneins, unconventional proteins from unconventional animals: A long journey from nematodes to mammals. Biomolecules 2014, 4, 435–457. [Google Scholar] [CrossRef] [Green Version]
- Foulquier, F.; Legrand, D. Biometals and glycosylation in humans: Congenital disorders of glycosylation shed lights into the crucial role of Golgi manganese homeostasis. Biochim. Biophys. Acta Gen. Subj. 2020, 1864, 129674. [Google Scholar] [CrossRef]
- Baj, J.; Flieger, W.; Flieger, M.; Forma, A.; Sitarz, E.; Skórzyńska, K.; Dziduszko, J.; Grochowski, C.; Maciejewski, R.; Karakuła-Juchnowicz, H. Autism spectrum disorder: Trace elements imbalances and the pathogenesis and severity of autistic symptoms. Neurosci. Biobehav. Rev. 2021, 129, 117–132. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Y.; Hao, X.; Zhang, Q.; Yang, K.; Li, L.; Ma, L.; Wang, S.; Li, X. Recent progress in therapeutic and diagnostic applications of lanthanides. Mini Rev. Med. Chem. 2011, 11, 678–694. [Google Scholar] [CrossRef]
- Morrow, J.R.; Tóth, É. Next-generation magnetic resonance imaging contrast agents. Inorg. Chem. 2017, 56, 6029–6034. [Google Scholar] [CrossRef] [Green Version]
- Boros, E.; Gale, E.M.; Caravan, P. MR imaging probes: Design and applications. Dalton Trans. 2015, 44, 4804–4818. [Google Scholar] [CrossRef]
- Kostova, I. Lanthanides as anticancer agents. Curr. Med. Chem. Anticancer Agents 2005, 5, 591–602. [Google Scholar] [CrossRef]
- Teo, R.D.; Termini, J.; Gray, H.B. Lanthanides: Applications in Cancer Diagnosis and Therapy. J. Med. Chem. 2016, 59, 6012–6024. [Google Scholar] [CrossRef] [Green Version]
- Flieger, J.; Dolar-Szczasny, J.; Rejdak, R.; Majerek, D.; Tatarczak-Michalewska, M.; Proch, J.; Blicharska, E.; Flieger, W.; Baj, J.; Niedzielski, P. The Multi-Elemental Composition of the Aqueous Humor of Patients Undergoing Cataract Surgery, Suffering from Coexisting Diabetes, Hypertension, or Diabetic Retinopathy. Int. J. Mol. Sci. 2021, 22, 9413. [Google Scholar] [CrossRef]
- Dolar-Szczasny, J.; Flieger, J.; Kowalska, B.; Majerek, D.; Tatarczak-Michalewska, M.; Zakrocka, I.; Załuska, W.; Rejdak, R. Hemodialysis effect on the composition of the eye fluid of cataract patients. J. Clin. Med. 2021, 10, 5485. [Google Scholar] [CrossRef]
- El-Fakahany, E.; Richelson, E. Effects of lanthanides on muscarinic acetylcholine receptor function. Mol. Pharmacol. 1981, 19, 282–290. [Google Scholar] [PubMed]
- Chantler, P.D. Lanthanides do not function as calcium analogues in scallop myosin. J. Biol. Chem. 1983, 258, 4702–4705. [Google Scholar] [CrossRef]
- Wang, K.; Cheng, Y.; Yang, X.; Li, R. Cell responses to lanthanides and potential pharmacological actions of lanthanides. Met. Ions Biol. Syst. 2003, 40, 707–751. [Google Scholar] [PubMed]
- Coffey, J.C.; Walsh, D.; Byrnes, K.G.; Hohenberger, W.; Heald, R.J. Mesentery—A ’New‘ organ. Emerg. Top Life Sci. 2020, 4, 191–206. [Google Scholar] [PubMed]
- Schmeling, M.; Gaynes, B.; Tidow-Kebritchi, S. Heavy metal analysis in lens and aqueous humor of cataract patients by total reflection X-ray fluorescence spectrometry. Powder Diffr. 2014, 29, 155–158. [Google Scholar] [CrossRef] [Green Version]
- Tabatadze, T.; Zhorzholiani, L.; Kherkheulidze, M.; Kandelaki, E.; Ivanashvili, T. Hair heavy metal and essential trace element concentration in children with autism spectrum disorder. Georgian Med. News 2015, 248, 77–82. [Google Scholar]
- Heinemann, G.; Fichtl, B.; Vogt, W. Pharmacokinetics of vanadium in humans after intravenous administration of a vanadium containing albumin solution. Br. J. Clin. Pharmacol. 2003, 55, 241–245. [Google Scholar] [CrossRef] [Green Version]


| Element | Tissue | Mean Value for Cases | Mean Value for Controls | ANSp | WILCOXp | BMp |
|---|---|---|---|---|---|---|
| Na 1 | T | 661 (252) | 846 (224) | 0.984 | 0.044 | 0.034 |
| PL | 369 (235) | 710 (310) | 0.980 | 0.000 | 0.000 | |
| PP | 415 (269) | 659 (254) | 0.795 | 0.006 | 0.003 | |
| K 1 | T | 581 (252) | 741 (226) | 0.766 | 0.047 | 0.040 |
| PL | 507 (302) | 739 (231) | 0.784 | 0.005 | 0.003 | |
| PP | 541 (315) | 710 (194) | 0.283 | 0.034 | 0.029 | |
| Rb 1 | PL | 414 (236) | 571 (179) | 0.746 | 0.011 | 0.007 |
| Cs 2 | T | 1.35 (0.68) | 1.73 (0.56) | 0.682 | 0.047 | 0.039 |
| PL | 1.27 (0.72) | 1.74 (0.76) | 0.901 | 0.044 | 0.037 | |
| Co 2 | T | 5.12 (1.83) | 8.70 (4.61) | 0.449 | 0.003 | 0.001 |
| PL | 5.94 (2.48) | 10.46 (12.39) | 0.469 | 0.055 | 0.047 | |
| Cd 2 | PL | 7.54 (4.19) | 11.09 (4.72) | 0.566 | 0.016 | 0.010 |
| PP | 7.55 (5.76) | 11.26 (6.49) | 0.469 | 0.043 | 0.046 | |
| Sb 2 | PP | 0.55 (2.40) | 0.55 (1.24) | 0.008 | 0.036 | 0.035 |
| Pr 2 | PP | 0.84 (0.95) | 0.45 (0.78) | 0.769 | 0.054 | 0.044 |
| T | 1.15 (1.98) | 4.53 (13.22) | 0.666 | 0.037 | 0.033 | |
| Sm 2 | PP | 0.67 (0.71) | 0.35 (0.69) | 0.792 | 0.056 | 0.049 |
| Er 2 | PP | 0.81 (0.63) | 0.50 (0.42) | 0.770 | 0.054 | 0.044 |
| Dy 2 | T | 0.27 (0.52) | 0.51 (0.53) | 0.562 | 0.038 | 0.033 |
| Tb 2 | T | 0.05 (0.09) | 0.1 (0.1) | 0.475 | 0.050 | 0.049 |
| Element | Sudden Death Due to Suicide | Sudden Death from Accidents | ||||||
|---|---|---|---|---|---|---|---|---|
| Friedp | PR/PL | T/PL | T/PR | Friedp | PR/PL | T/PL | T/PR | |
| Na | 0.002 | 0.686 | 0.008 | 0.008 | 0.143 | 0.996 | 0.219 | 0.054 |
| Mg | 0.066 | 1.000 | 0.062 | 0.071 | 0.003 | 1.000 | 0.000 | 0.001 |
| Ca | 0.009 | 0.325 | 0.027 | 0.296 | 0.018 | 1.000 | 0.042 | 0.077 |
| Mn | 0.024 | 1.000 | 0.036 | 0.042 | 0.229 | 1.000 | 0.032 | 0.148 |
| Fe | 0.000 | 1.000 | 0.001 | 0.000 | 0.000 | 1.000 | 0.004 | 0.009 |
| Cu | 0.015 | 1.000 | 0.081 | 0.002 | 0.368 | 1.000 | 1.000 | 1.000 |
| Mo | 0.000 | 0.589 | 0.000 | 0.000 | 0.000 | 1.000 | 0.000 | 0.000 |
| Cd | 0.000 | 1.000 | 0.000 | 0.000 | 0.000 | 1.000 | 0.000 | 0.000 |
| Sn | 0.206 | 0.317 | 0.424 | 1.000 | 0.126 | 1.000 | 0.043 | 0.205 |
| Ba | 0.946 | 1.000 | 1.000 | 1.000 | 0.076 | 1.000 | 0.506 | 0.010 |
| La | 0.211 | 1.000 | 0.130 | 0.686 | 0.003 | 1.000 | 0.048 | 0.019 |
| Pr | 0.607 | 1.000 | 0.502 | 1.000 | 0.007 | 1.000 | 0.148 | 0.014 |
| Nd | 0.946 | 1.000 | 1.000 | 1.000 | 0.015 | 1.000 | 0.490 | 0.039 |
| Sm | 0.607 | 1.000 | 1.000 | 1.000 | 0.006 | 1.000 | 0.950 | 0.035 |
| Eu | 0.796 | 1.000 | 0.544 | 1.000 | 0.037 | 1.000 | 1.000 | 0.061 |
| Gd | 0.958 | 1.000 | 1.000 | 1.000 | 0.024 | 0.773 | 0.829 | 0.035 |
| Tb | 0.573 | 1.000 | 1.000 | 1.000 | 0.054 | 0.826 | 0.888 | 0.028 |
| Dy | 0.678 | 1.000 | 1.000 | 1.000 | 0.015 | 1.000 | 0.620 | 0.079 |
| Hg | 0.047 | 1.000 | 0.047 | 0.088 | 0.150 | 1.000 | 0.025 | 0.312 |
| Tl | 0.092 | 0.389 | 0.071 | 1.000 | 0.065 | 0.219 | 0.028 | 1.000 |
| Bi | 0.009 | 0.244 | 0.027 | 0.851 | 0.241 | 0.164 | 0.370 | 1.000 |
| U | 0.000 | 0.851 | 0.002 | 0.003 | 0.003 | 1.000 | 0.001 | 0.002 |
| Element Ratio | Wilcox x | Wilcox p | Median Cases | IQR Cases | Median Controls | IQR Controls |
|---|---|---|---|---|---|---|
| Cd/Zn | 93 | 0.017 | 0.001 | 0.001 | 0.002 | 0.001 |
| Cd/Se | 49 | 0.043 | 5.345 | 11.419 | 12.346 | 35.369 |
| Cd/Cu | 92 | 0.016 | 0.008 | 0.009 | 0.016 | 0.012 |
| Cd/Ca | 125 | 0.169 | 0.013 | 0.011 | 0.018 | 0.024 |
| Cd/P | 124 | 0.159 | 0.011 | 0.011 | 0.018 | 0.013 |
| Cd/Fe | 116 | 0.098 | 0.000 | 0.000 | 0.000 | 0.000 |
| Group | Gender | n | % | Min–Max Age | Median Age | Mean Age ± SD |
|---|---|---|---|---|---|---|
| cases (n = 20) | Female | 2 | 10 | 13–17 | 15 | 15 ± 2.83 |
| Male | 18 | 90 | 20–62 | 49 | 44.67 ± 13.58 | |
| controls (n = 20) | Female | 3 | 15 | 29–83 | 58 | 56.67 ± 27.02 |
| Male | 17 | 85 | 18–93 | 57 | 55.46 ± 16.67 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baj, J.; Teresiński, G.; Kowalska, B.; Krajka, T.; Buszewicz, G.; Forma, A.; Flieger, W.; Karakuła, K.H.; Kędzierawski, P.; Cywka, T.; et al. ICP-MS Multi-Elemental Analysis of the Human Meninges Collected from Sudden Death Victims in South-Eastern Poland. Molecules 2022, 27, 1911. https://doi.org/10.3390/molecules27061911
Baj J, Teresiński G, Kowalska B, Krajka T, Buszewicz G, Forma A, Flieger W, Karakuła KH, Kędzierawski P, Cywka T, et al. ICP-MS Multi-Elemental Analysis of the Human Meninges Collected from Sudden Death Victims in South-Eastern Poland. Molecules. 2022; 27(6):1911. https://doi.org/10.3390/molecules27061911
Chicago/Turabian StyleBaj, Jacek, Grzegorz Teresiński, Beata Kowalska, Tomasz Krajka, Grzegorz Buszewicz, Alicja Forma, Wojciech Flieger, Kaja Hanna Karakuła, Paweł Kędzierawski, Tomasz Cywka, and et al. 2022. "ICP-MS Multi-Elemental Analysis of the Human Meninges Collected from Sudden Death Victims in South-Eastern Poland" Molecules 27, no. 6: 1911. https://doi.org/10.3390/molecules27061911







