Abstract
The genus Bidens a member of family Compositae, is widely documented as an ethno-medicinally important genus of plants. In the present study, anticancer potential of three ethno-medicinally important species i.e., B. bipinnata, B. biternata and B. pilosa were tested. For in-vitro evaluation, an MTT (Thiazolyl blue tetrazolium bromide) assay was performed against cervical cancer cells (HeLa), hepatocellular carcinoma (HepG), and adenocarcinoma human alveolar basal epithelial cells (A549). For in vivo evaluation, Artemia salina, Danio rerio, and Caenorhabditis elegans were used. Among all the tested extracts, the ethanol extract of B. biternata appeared to have highest anticancer activity, and the compounds responsible for this activity were identified to be Tris (2,4-di-tert-butylphenyl), 4-hydroxy-2,4′-dimethoxychalcone, and 2,4-di-tert-butylphenol. This is the first report of the isolation of Tris (2,4-di-tert-butylphenyl) phosphate from the genus Bidens and the first report of 4-hydroxy-2,4′-dimethoxychalcone and 2,4-di-tert-butylphenol from B. biternata. Among the isolated compounds, 4-hydroxy-2,4′-dimethoxychalcone showed the highest anticancer activity with an LD50 value of 236.7 µg/mL. Therefore, this compound carries promising potential for being established as a pharmaceutical for chemoprevention and chemotherapy.
1. Introduction
Overproduction of several reactive oxygen species, i.e., oxygen radicals and non-free radical species is reflected to be the chief provider to oxidative stress, which has been associated to numerous ailments like cancer, tissue damage in rheumatoid arthritis and atherosclerosis [1]. Plants are a representative source of drugs and edible plant are chief sources of antioxidants that have the capability to defend the body from injury produced by free radicals prompted oxidative stress [2].
The protective mechanisms of phytochemicals on tumor advancement vary from the inhibition of genotoxic effects to inhibition of proteases and cell proliferation, better antioxidant activity, signal transduction pathways and defense of intracellular infrastructures to control apoptosis [3].
Bidens is a widespread genus consisting of 247 species that are cosmopolitan in distribution [4]. Many of these species have been reported to have sesquiterpene, acetylenes, lactones and flavonoids [5]. However, the most widespread are aromatic derivatives, thiophenes, carotene, coumarins (umbelliferon, scopoletin and aesculetin), vitamin C and C17-, C14-, C13-polyacetylenes [6]. B. pilosa is reported to cure various ailments i.e., infectious diseases, immunological disorders, metabolic syndrome and etc. [7]. This herb is taken in the form of decoction, infusion or juice. However, in the case of snakebite and bleeding wounds it can be applied externally (Table 1). It can be used alone or with other medicinal plants i.e., Cissus sicyoides, Aloe vera, Valeriana officinalis and Plectranthus mollis [8].
B. bipinnata is emmenagogue, stimulant, antispasmodic, and have expectorant effect. Traditionally it is used to treat laryngeal, asthma and respiratory disorders (Table 1). In vivo studies of B. bipinnata extract has shown antimalarial effect. Its ethanol extract shows 70% inhibition of plasmodium growth [9]. The studies have reported that butanol extract of B. biternata have showed a very high antiradical potential whereas its n-hexane extract showed very low antiradical potential [10]. The main constituents of B. biternata are saponins, steroids, terpenoids, coumarins, glycosides, athraquinones, iridoids, alkaloids, tannins, phlobatannins and flavonoids [10]. In the present study three species of genus Bidens has been investigated for their anticancer potential using multiple in-vivo and in-vitro assays and the compounds responsible for these activities were isolated and analyzed for their subsequent activity.

Table 1.
Ethno-medical evidence about B. pilosa, B. biternata, and B. bipinnata.
Table 1.
Ethno-medical evidence about B. pilosa, B. biternata, and B. bipinnata.
| Disorder | Plant Part | Dosage Form | Region/Country | References |
|---|---|---|---|---|
| B. pilosa | ||||
| Stomach ache | LE | Not stated | Africa | [11] |
| Colic | WP | Decoction | China, Africa | [11] |
| Catarrh | WP | Juice | Cuba | [12] |
| Diarrhea | LE, WP | Decoction | Uganda, Africa | [13] |
| Constipation | WP | Decoction | India | [14] |
| Dysentery | WP | Infusion | Africa | |
| Choleretic | WP | Decoction | America | [15] |
| Antirheumatic | RT, WP | Infusion | Hong Kong | [16] |
| Appendicitis | WP | Not stated | Hong Kong | [16] |
| Enteritis | WP | Decoction | China | [17] |
| Otitis | WP | Decoction | China, Africa | [18] |
| Gastritis | WP | Juice | Cuba | [19] |
| Diabetes | WP | Decoction | Taiwan, Cuba | [11] |
| Headache | WP | Decoction | Bafia, Cameroon | [11] |
| Diuretic | WP | Decoction | Central America | [20] |
| Hypotensive | WP | Juice | Cameroon | [21] |
| Fever | WP | Decoction | Not stated | [19] |
| Yellow Fever | LE, WP | Decoction | America | [13] |
| Acute hepatitis | WP | Decoction | Hong Kong | [22] |
| Intestinal worms | LE | Decoction | Africa | [14] |
| Malaria | WP | Juice | China | [18] |
| Eye diseases | LE | Juice | Uganda | [13] |
| B. biternata | ||||
| Leprosy | LE | Not stated | India | [23] |
| Cuts and wounds | LE | Decoction | India | [23] |
| Nose bleeds | WP | Decoction | China | [23] |
| Gastric ulcers | LE | Maceration taken orally | Central America | [10] |
| Skin problems | WP | Topical application | Africa | [24] |
| Wounds | WP | Crushed herb | China | [24] |
| Snake bites | WP | Crushed herb | China | [23] |
| B. bipinnata L. | ||||
| Asthma | WP | Decoction | China | [25] |
| Colds | LE | Decoction | China | [25] |
| Fever | WP | Decoction | Not stated | [9] |
| Antimicrobial | AP | Decoction | Trinidad | [26] |
| Eye Diseases | LE | Juice | Uganda | [13] |
| Colds | LE, WP | Decoction | Uganda, China | [21] |
LE: leaves; WP: whole plant; AP: aerial parts; RT: root.
2. Material and Method
2.1. Formation of Crude Methanolic Plant Extract
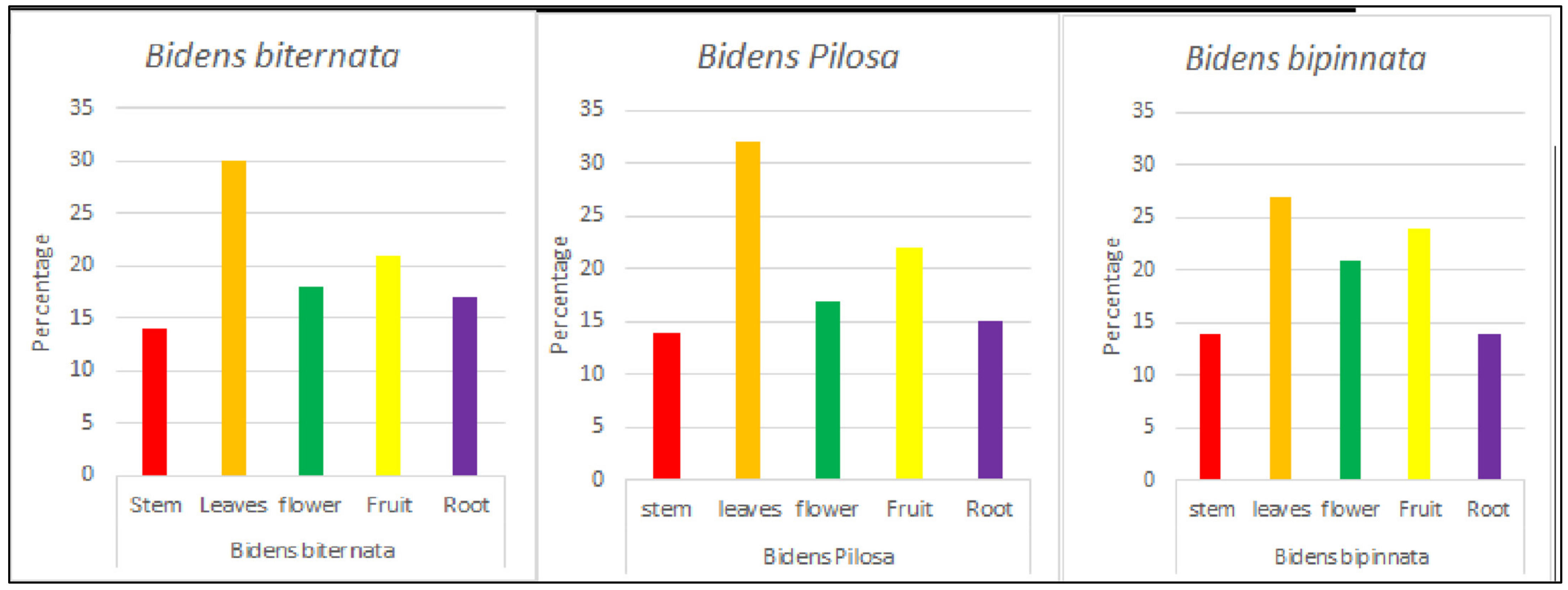
The plants parts were collected during October 2016 to October 2017. After collection the collected parts were thoroughly washed, fully desiccated and ground into fine powder. Powdered plant material (150 g) is measured, and using cold maceration technique crude methanol extract is prepared (Figure 1).
Figure 1.
The percentage yield of plants obtained.
The crude extracts obtained were then subjected to liquid-liquid partition. All the solvents used are (HPLC)-grade purity from Sigma-Aldrich Co. (St. Louis, MI, USA). In 250 mL water, extract will be suspended separately and partitioned with n-hexane in a separating funnel. The hexane layer and aqueous layers were collected and concentrated in rotary evaporator. In the concentrated aqueous layer acetone was added and placed in sonicator bath for one hour. The acetone soluble supernatant was separated and dried as acetone extract whereas the precipitates were again treated with ethanol and placed in sonicator bath. The supernatant was concentrated as ethanol extract and precipitates were taken as aqueous extract.
2.2. In-Vitro Cytotoxicity Assay
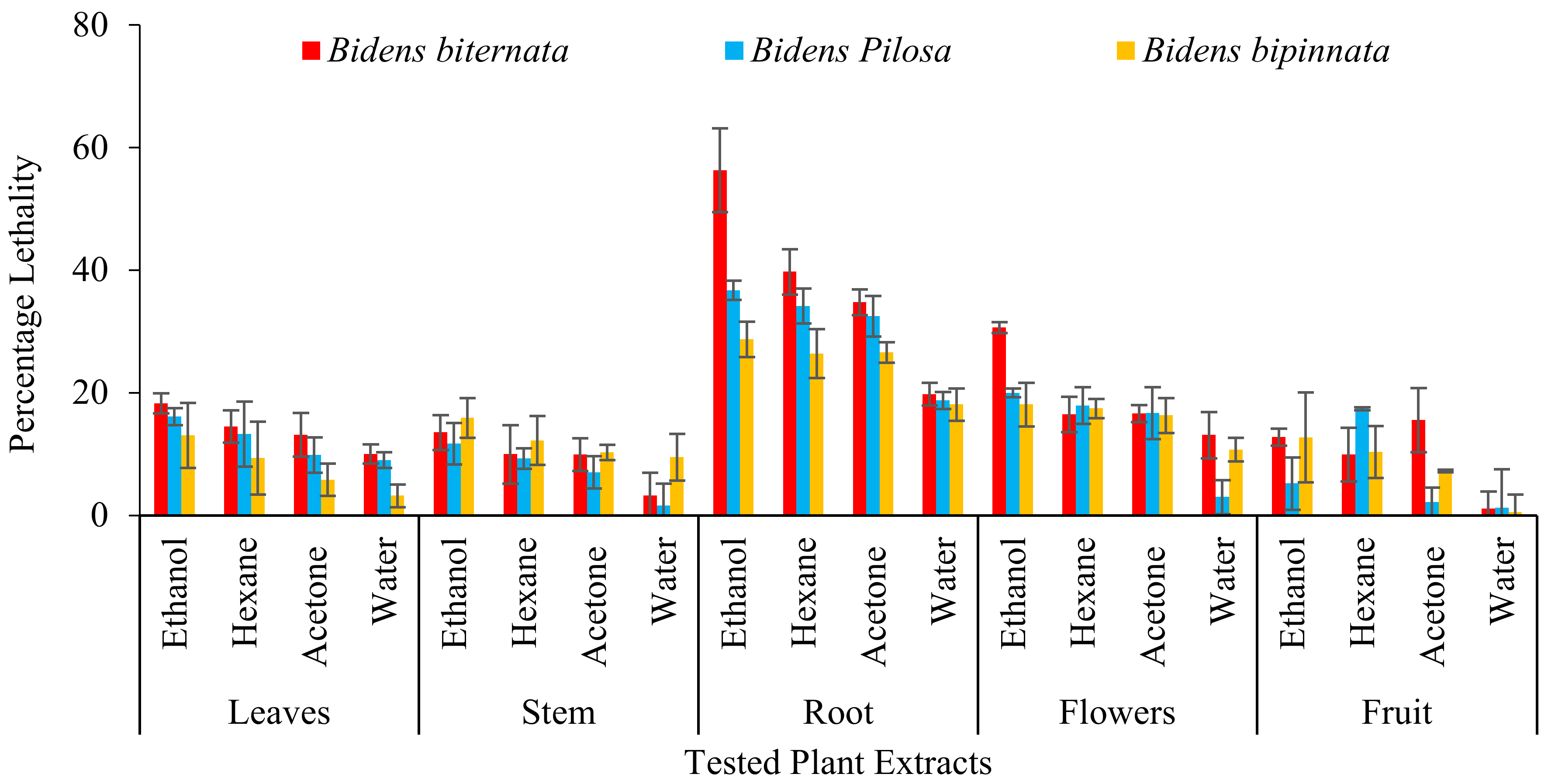
During evaluation of cytotoxicity plant parts i.e., stem, root, leaves, flowers and achenes were separately tested in parallel with four different solvents i.e., ethanol, hexane, acetone, water using MTT assay. In in-vitro conditions three different cell lines i.e., cervical cancer cells (Hela), hepatocellular carcinoma (HEPG) and adenocarcinomic human alveolar basal epithelial cells (A549) were used. This assay is an inexpensive, standard method, to measure cell death. It is established on the reduction of to formazan crystals (purple) due to metabolic active cells [27].
Thiazolyl Blue Tetrazolium Bromide (MTT)
Product Number M 2128 (Lab M Ltd., Lancashire, UK); Storage Temperature 2–8 °C.
Procedure
From a cultured plate, cells were dislocated using trypsin (T4799 Sigma-Aldrich (St. Louis, MO 63118, USA). In a flask 5 mL of complete media (D5796 Sigma-Aldrich (Hamburg, Germany)) is added to trypsin zed cells. The trypsinized cells were centrifuged in a 15 mL falcon tube (500 rpm for 5 min). Cell culture media is removed and cells were suspended to 1.0 mL culture media. Suspended cells were counted and by using complete media the cell suspension is diluted (75,000 cells/mL). In a 96 well plate 100 µL of suspended cells were added into each well. The plate is placed in CO2 incubator. Next day tested extracts were added and final volume is kept to 100 µL per well. To each well 20 µL of MTT is mixed. As a control wells with no MTT are used. These plates are placed for 3.5 h at 37 °C in CO2 incubator. The Absorbance is obtained at 590 nm using a reference filter of 620 nm.
2.3. In-Vivo Cytotoxicity Assay Caenorhabditis elegans
The C. elegans (N2 wild-type) were used. At L4 stage the worms are subjected for synchronization. The synchronized populations were acquired using alkaline bleaching method [28]. In a 96-well microplate (10 μL, ∼40–45 L4 synchronized larvae were added with 189 μL of E. coli OP50 culture (OD = 0.5 at 620 nm and, 1 μL of plant extract. As a solvent control DMSO (1 μL) was used and Levamisole (final concentration 50 μM) Levamisole (Sigma-Aldrich (St. Louis, MO 63118, USA)). The microplate was incubated for 16 h at 20 °C into a WMicroTracker. The movement was recorded every 30 min by the WMicroTracker.
2.4. In Vivo Cytotoxicity Assay on Zebrafish
The in-vivo toxicity test is performed on zebrafish larva using permitted protocol of Institutional Animal Ethics and Biosafety Committee of KU Leuven, Belgium [29]. The toxicity assay was performed in 96-well plate. In each well 199 μL of E3 medium with 3 zebra fish larva is with 1 μL of plant extract transferred. DMSO (1 μL) was used as solvent control and gossypol (1 μL) was used as drug control. The microplate was placed into a WMicroTracker for 48 h at 28 °C. The movement of zebrafishes is measured by the WMicroTracker after every 30 min. The plate was also analyzed with microscope to see the number of deaths.
2.5. In Vivo Cytotoxicity Assay on Brine Shrimp
The prepared extracts and fractions were tested against brine shrimp lethality test (BSLT) as described [30]. Brine shrimp (Artemia salina) eggs (JBL Artemiopur, Germany) were placed in well aerated artificial sea water. A two chambered container was used with one chambered covered whereas other chamber open. In the middle of the two chambers small openings were present. Sea water was prepared (38 g of sea salt/one liter of distilled water). To feed hatched larvae a pinch of yeast was added. After 24 h, the shrimp’s larvae were ready to be used.
The tested extracts were dissolved in 100% DMSO (stock). From stock different concentrations of solution were prepared using artificial sea water. As a positive control Nicotine N3876 Sigma-Aldrich was utilized. The phototropic nauplii were collected and 10 nauplii were added in each container. After 24 h incubation at room temperature dead nauplii were counted and percentage lethality was measured according to the following formula:
Percentage of Death = (Total nauplii − Alive nauplii) × 100/Total nauplii
2.6. Bioassay-Guided Purification
On silica gel (70–230 mesh) dried plant extract was adsorbed and loaded on silica column (600 mm height × 55 mm diameter). Elution were obtained with an increased polarity gradient of hexane–dichloromethane i.e., 9.5:0.5, 9:1, 8.5:0.5, 8:2, 7:3, 6:4, 5:5, 4:6, 3:7, 2:8, 1:9 and 0:10. After that elutions were collected with 100% dichloromethane, and 100% ethyl acetate, ethyl acetate and methanol (9.5:0.5, 9:1, 8:2, 7:3, 6:4, 5:5, 4:6, 3:7, 2:8, and 1:9) and 100% methanol.
The entire process was supervised at 280 nm and 254 by a Dual λ absorbance detector (Waters Milford, MA 01757, USA). 5 µL Aliquots of 225 fractions were marked on large TLC glass plates (20 cm × 20 cm) and placed in glass jars (20 cm × 10 cm × 20 cm), with mobile phase at room temperature. The plates were monitored using ultra-violet (UV) light at 254 and 360 nm.
Using Shimadzu, LC-20AT system equipped with LC-20AT quaternary pump, a on-line degasser, a photodiode array detector HPLC was done. The mobile phase of 30:70 H2O and acetonitrile was used.
2.7. Identification of Isolated Compound
Collected peaks were exposed to a gas chromatograph along with a mass spectrometer. A Restek RXi-5sil MS 20 m column was utilized. Helium is injected with a rate of 0.9 mL/min. The temperature was gradually adjusted at 20 °C, 120 °C, 200 °C, 250 °C and to end with to 350 °C for 4 min.
3. Results and Discussion
Plants have played a vital role in human healthcare management [31]. Medicinal plants are defined as plants that have healing properties or provide valuable pharmacological effects on the body [32].
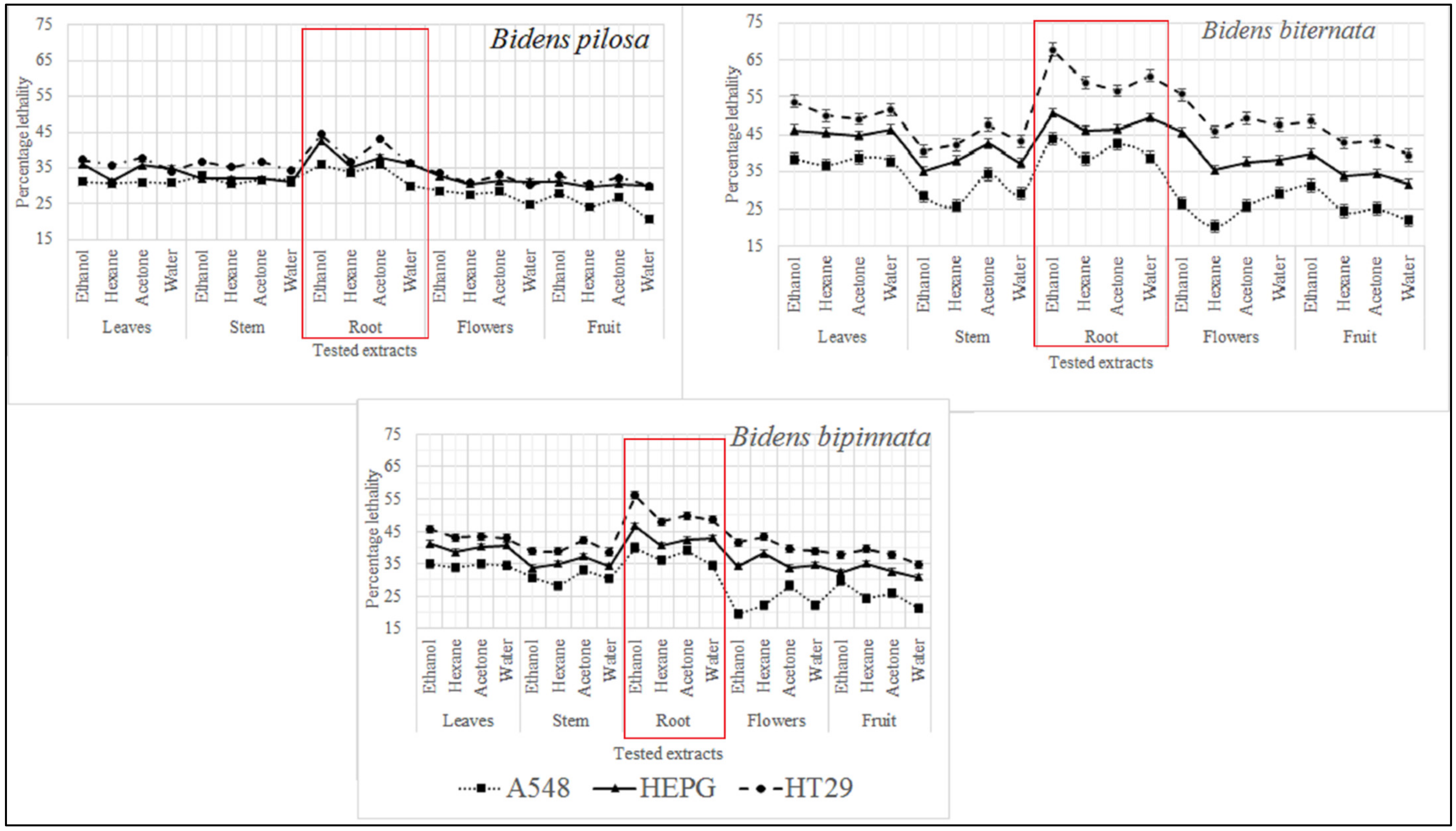
In the present study four different solvents i.e., hexane, acetone, ethanol and water were used and different plant parts of selected plants of genus Bidens i.e., root, stem, leaves, flowers and achenes were tested. Three different cell lines were tested i.e., Hela (cervical cancer), HEPG (liver hepatocellular carcinoma) and A549 (adenocarcinoma human alveolar basal epithelial cells). Our results indicate that the ethanol extracts of all tested plants and their parts were appeared to have cytotoxic activity. Studies conducted by Karagöz [33] and Nemati [34] also indicate that ethanol extracts of plants contain some cytotoxic compounds. It was also observed that root extracts of all tested plants have significant activity against all tested cell lines.
As described in Figure 2 it was observed that the most active extract is ethanol extract of B. biternata with percentage inhibition of 67.75% against HT29, 50.82% against HEPG and 43.8% against A549 cell lines. This is also worth mentioning that roots extract of all the tested plants are appeared to significant activity against all the tested cell lines (Figure 2).
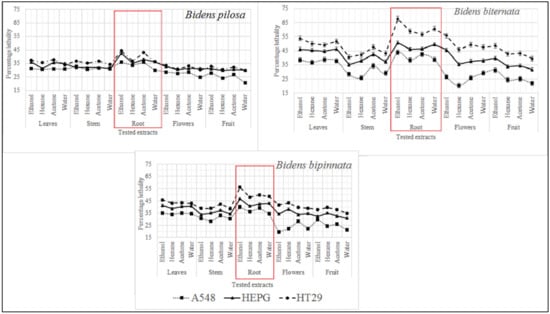
Figure 2.
Cytotoxic activity of selected plants of the genus Bidens against cervical cancer cells (HeLa), hepatocellular carcinoma (HepG), and adenocarcinoma human alveolar basal epithelial cells (A549).
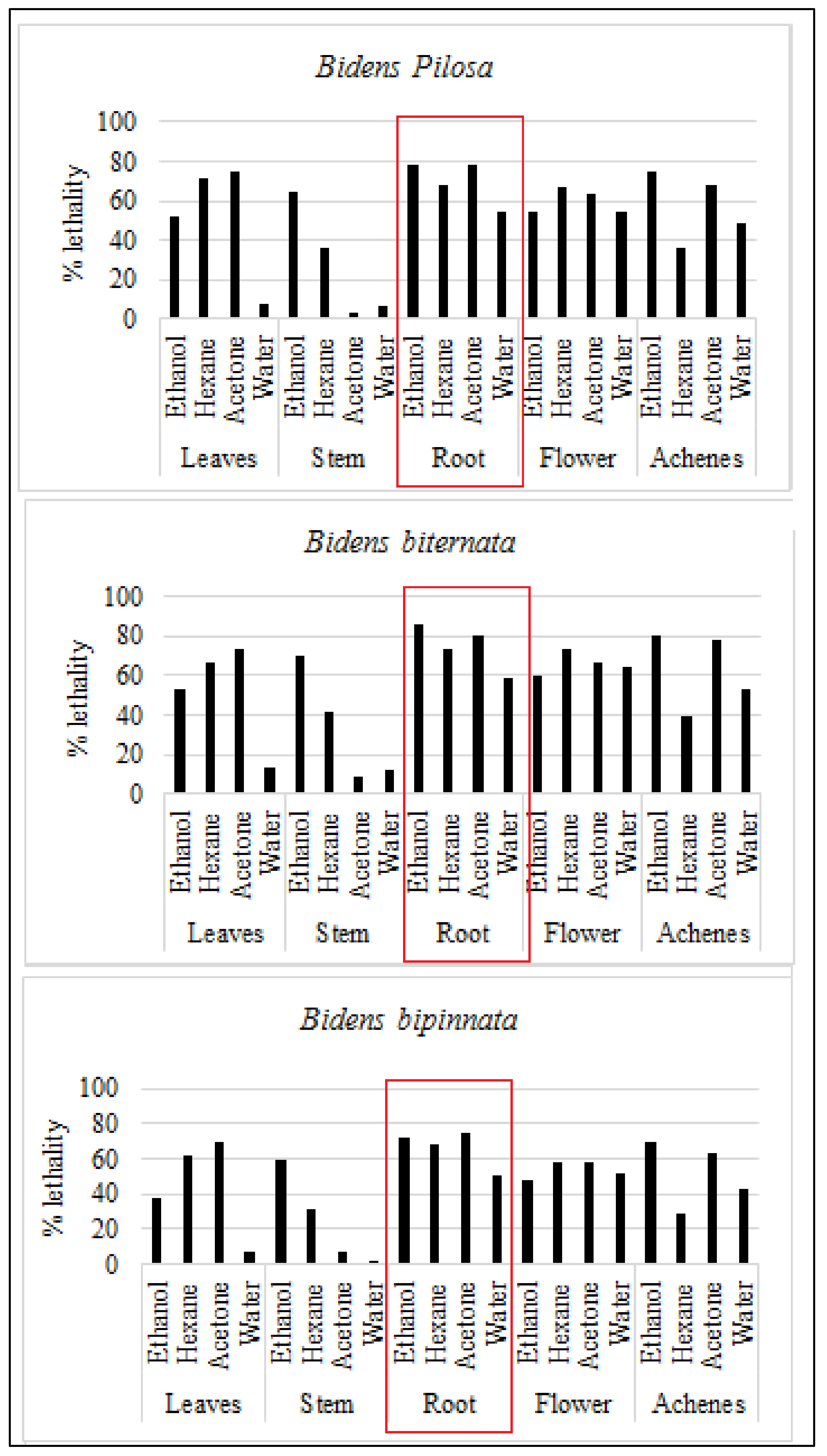
For initial toxicity screening brine shrimp lethality bioassay is performed. Brine shrimp assay is appeared to be interrelated with human nasopharyngeal carcinoma cytotoxicity. Our results are in line with previous in-vitro assessment as roots of all the tested plants are appeared to have toxic effect. The ethanol extract of B. biternata roots showed highest percentage lethality i.e., 86.67% (Figure 3).
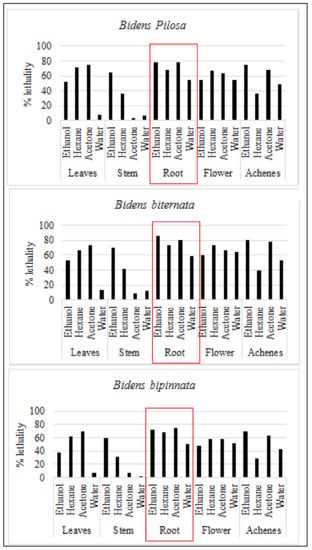
Figure 3.
Mean percentage death (lethality) of selected plants against brine shrimp.
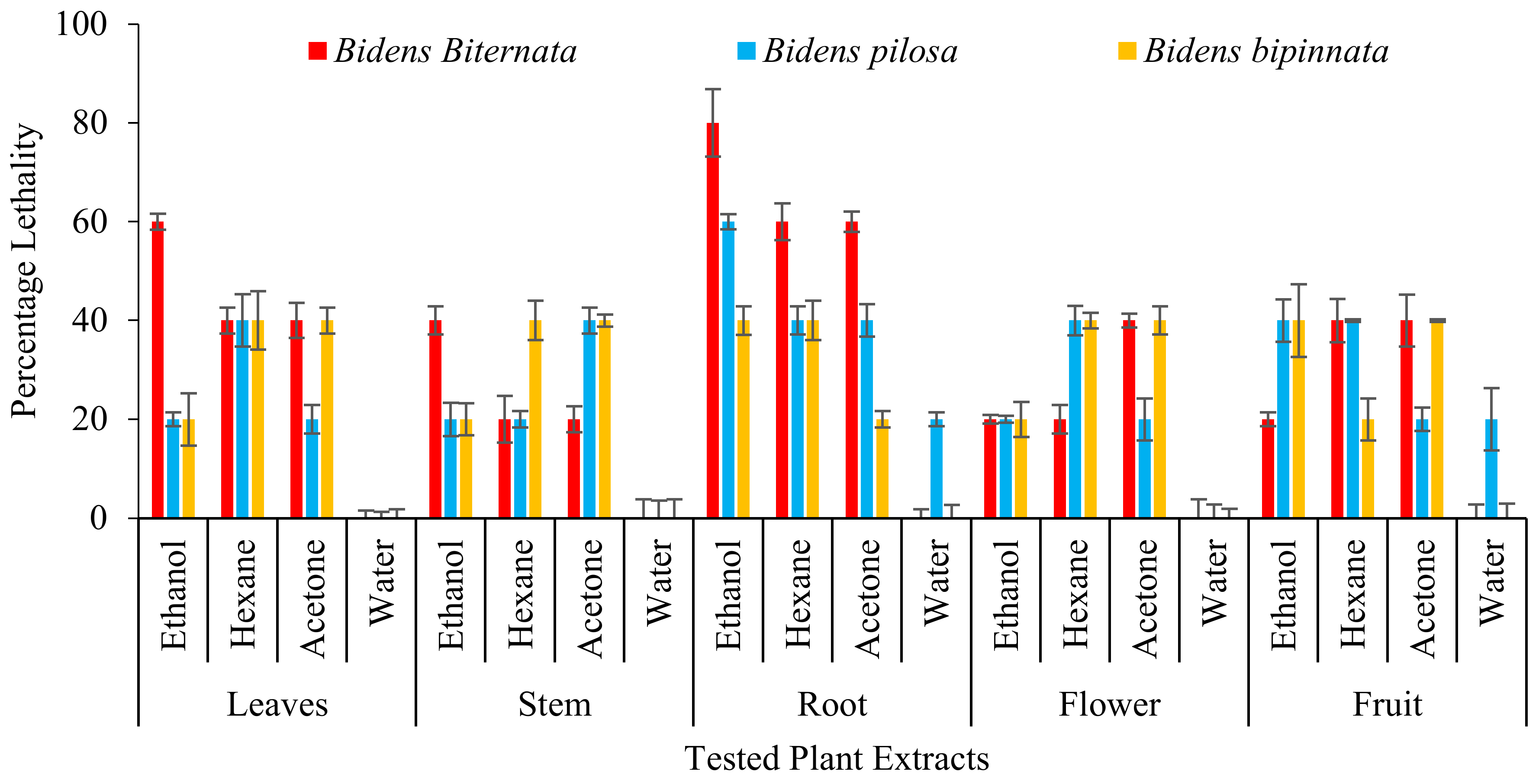
Zebra fish is been used as an ideal vertebrate organism used in diverse research zones as ecotoxicology, genetics and developmental biology [35]. Studies have showed that humans show great genetic resemblances of genomic sequences and brain patterning with zebra fish. Therefore, this makes zebra fishes a beneficial assay in exploring many toxicology studies yielding a rapid outcome. On zebra fishes all the extracts of B. biternata roots except water extract show toxic effect i.e., 80% (ethanol), 60% (hexane) and 60% acetone (Figure 4). Root ethanol extract of B. pilosa is also appeared to have toxic effect with percentage lethality of 60%. It is also worth noting that water extract of all tested plant species have less or no toxic effect on zebra fishes.
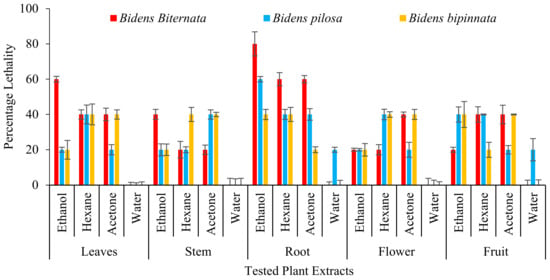
Figure 4.
Percentage death of Zebra fishes after treatment of plant extracts.
The Caenorhabditis elegans a nematode, is remarkably well deliberated animal model and several investigators have utilized it for evaluation of toxicity. They offer a channel among in-vitro tests and mammalian toxicity analysis by merging conventional in-vitro management practices and oral toxicity experiment records from a complete organism [36]. During present analysis on C. elegans toxicity it is observed by B. biternata root ethanol extract with percentage inhibition of 53.32% as compared to control (Levamisole) i.e., 91.93% (Figure 5).
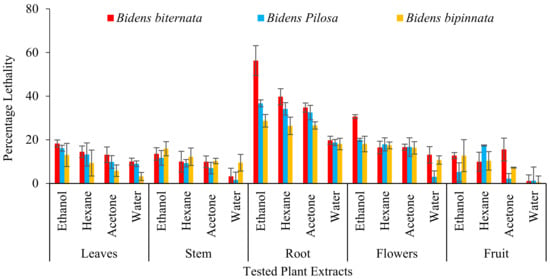
Figure 5.
Toxicity assessment on Caenorhabditis elegans.
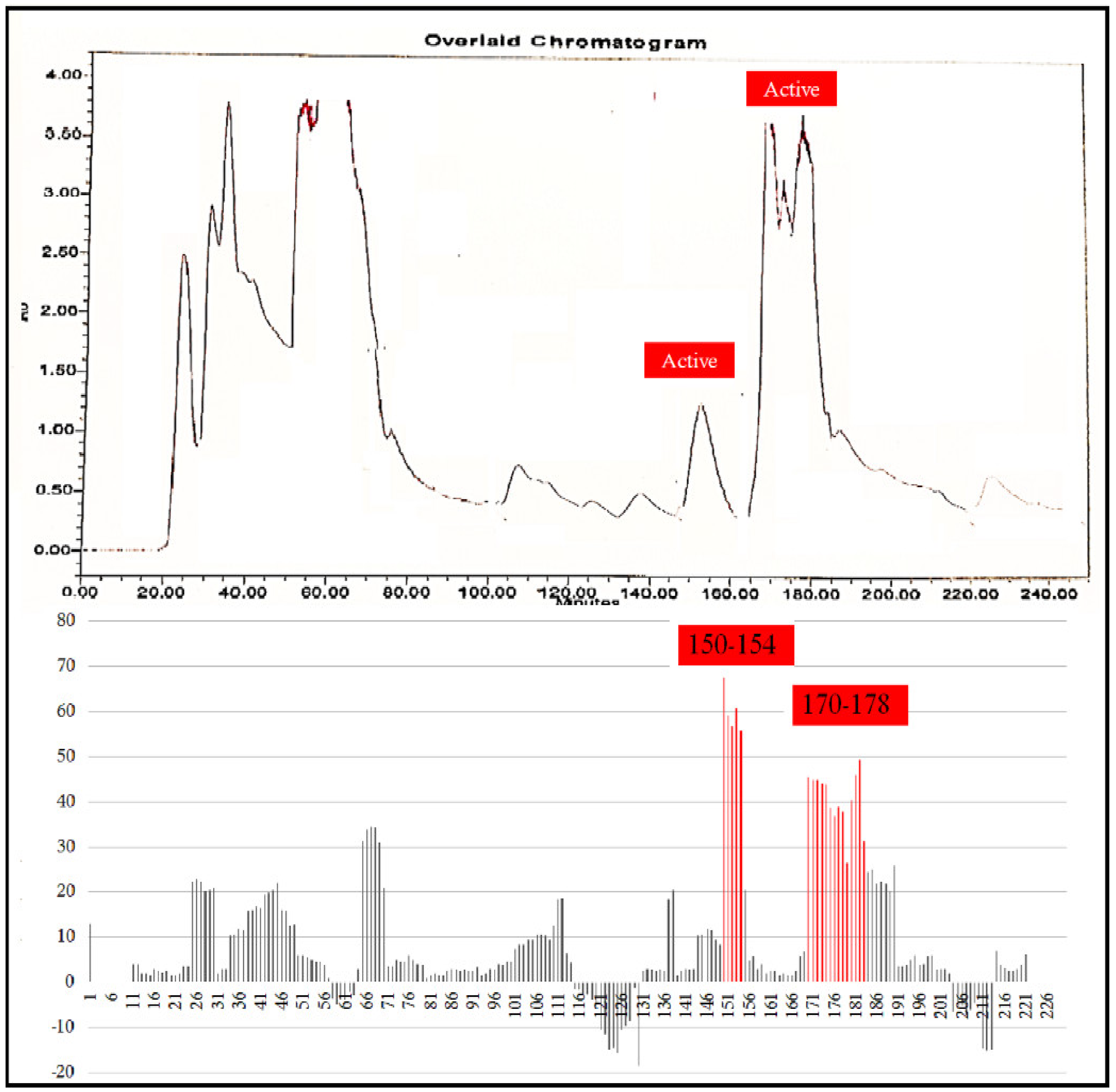
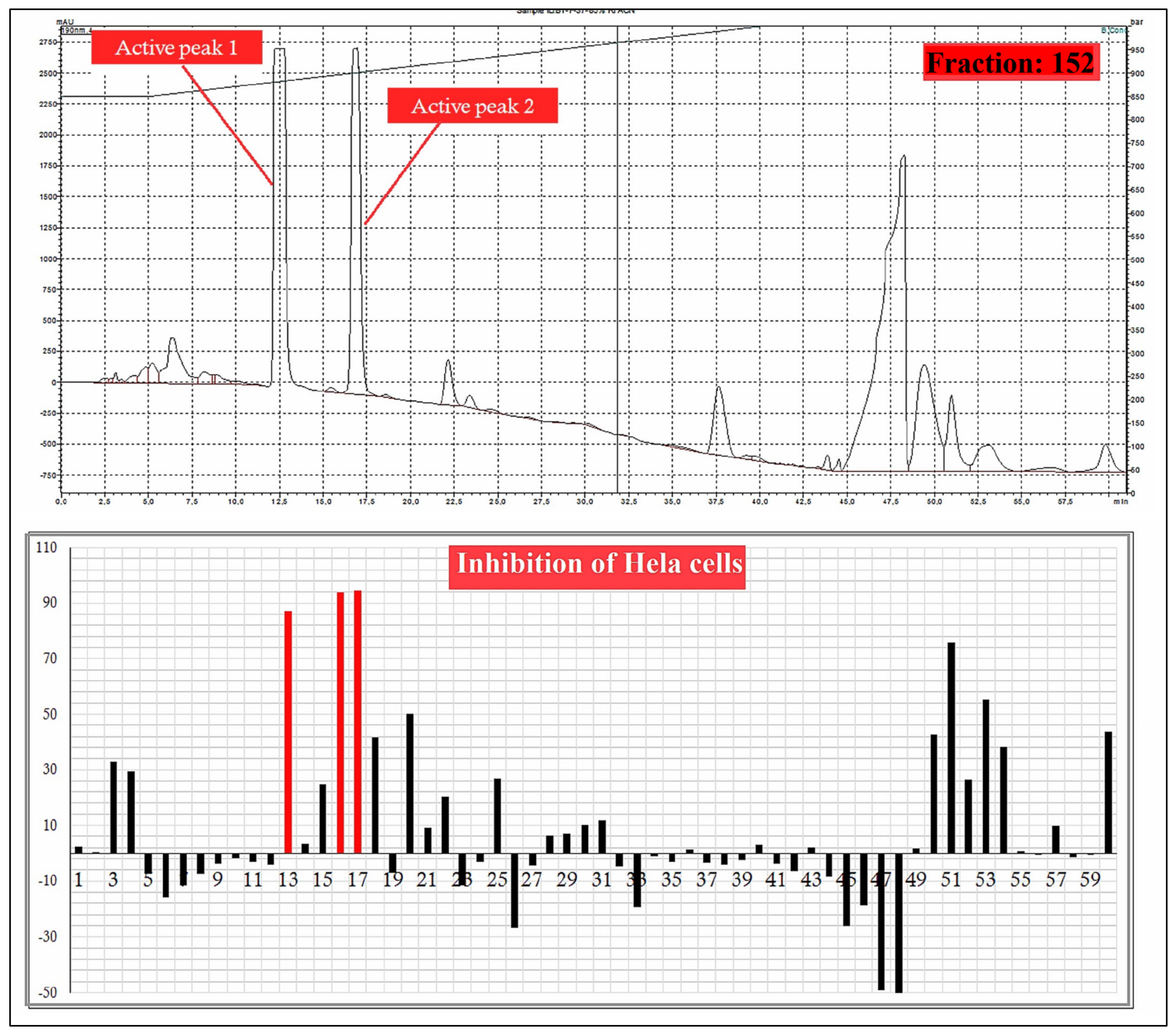
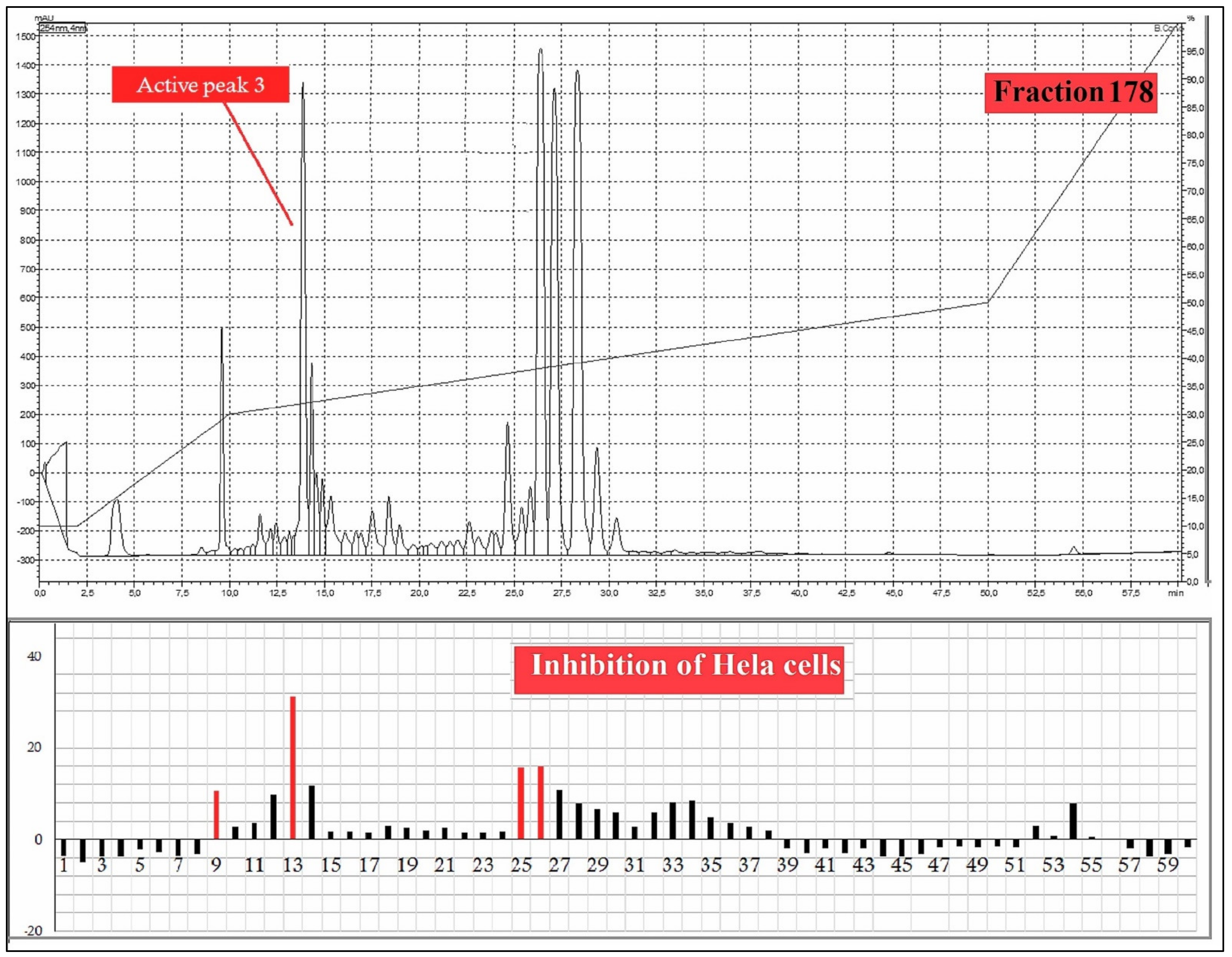
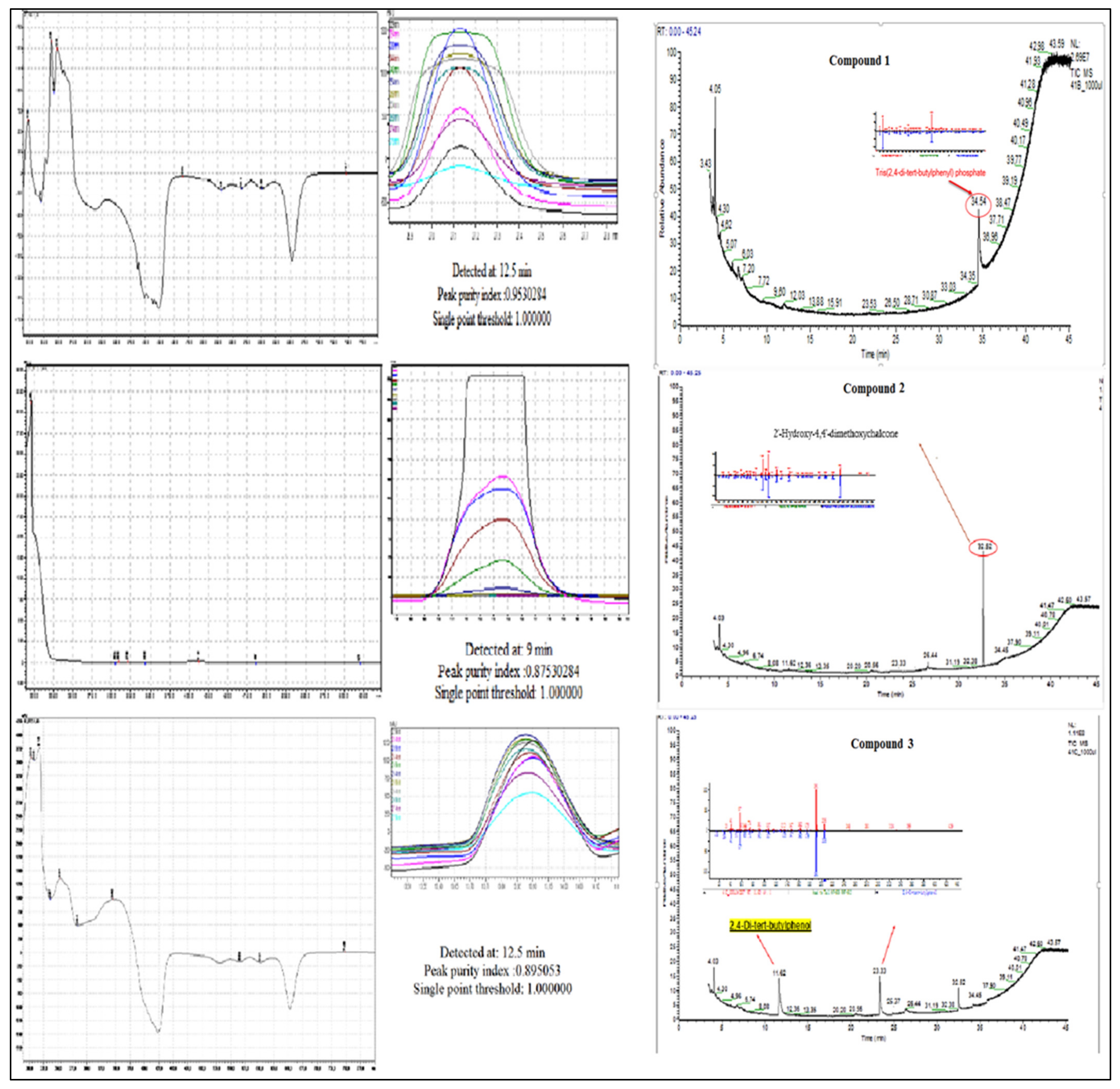
A total 225 fractions were collected from silica column chromatography fraction 152 and 178 is appeared to be active against HeLa cell line (Figure 6). These fractions were again using HPLC-DAD analysis (Figure 7 and Figure 8) and active fractions were collected. Before further analysis the purity of collected peaks were tested. Using thin layer chromatography their purity is tested using hexane: ethyl acetate mobile phases. A total of three pure active compounds were identified and were again subjected to gas chromatography mass spectrometry.
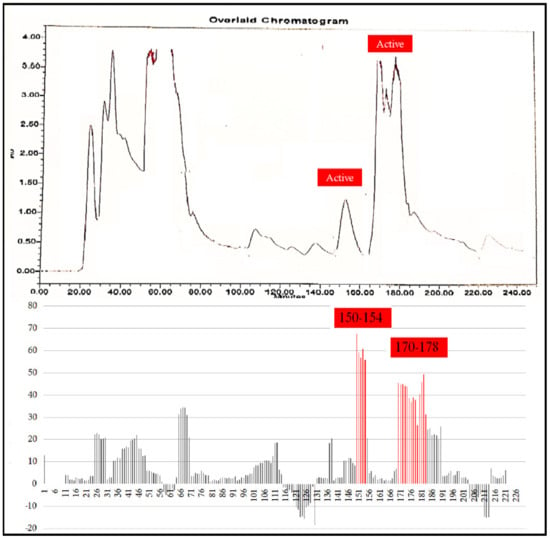
Figure 6.
Top panel: Overlaid chromatogram of Ethanol extract of B. biternata leaves; percentage inhibition of obtained fractions against cervical cancer cells (Hela) (bottom panel).
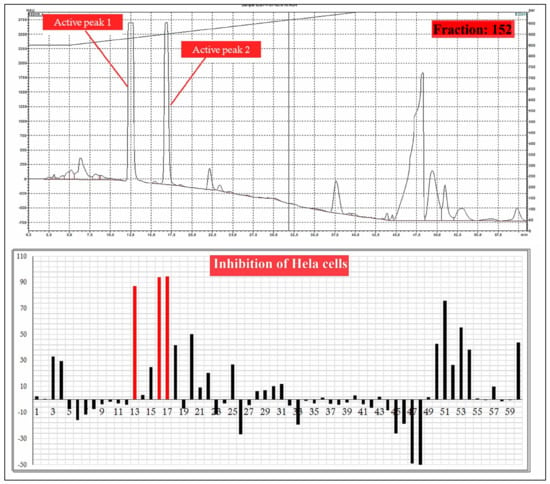
Figure 7.
HPLC chromatogram of fraction 152 of silica gel column; percentage inhibition of obtained fractions against cervical cancer cells (Hela) (bottom panel).
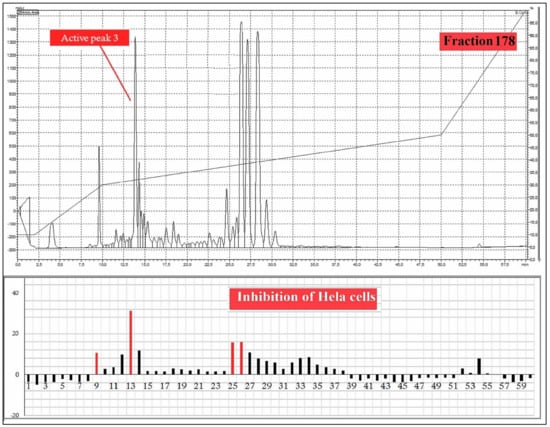
Figure 8.
HPLC chromatogram of fraction 178 of silica gel column; percentage inhibition of obtained fractions against cervical cancer cells (Hela) (bottom panel).
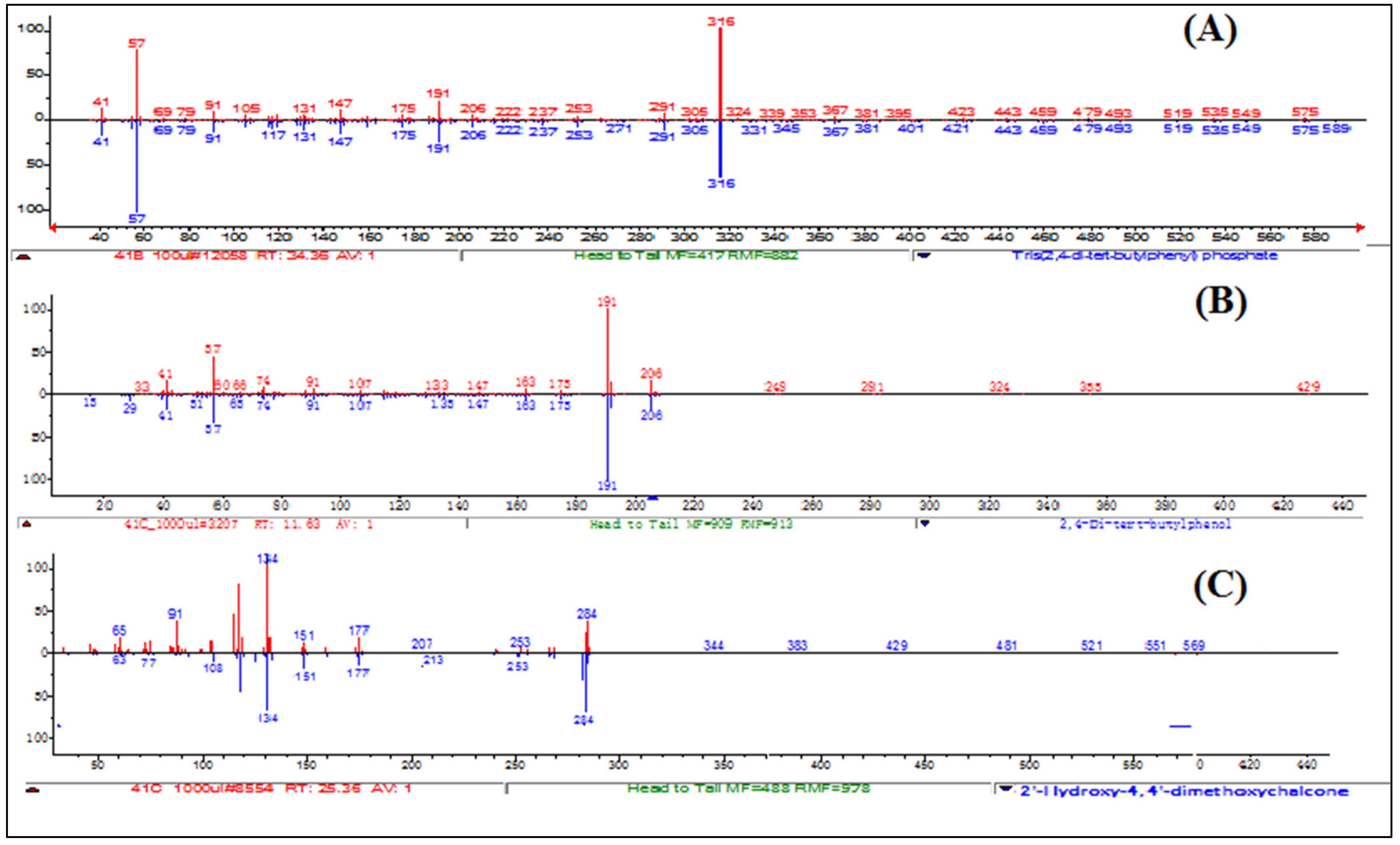
Compound 1 is a white-coloured solid and identified to be Tris (2,4-di-tert-butylphenyl) phosphate, C42H63O4P (Figure 8 and Figure 9). Tris (2,4-di-tert-butylphenyl) phosphate is an organophosphorus compound which is a phosphate ester derived from di-tart-butylphenol. It has also been identified from the flowers of Camellia sasanqua Thunb. [37], Aquilaria sinensis (Lour.) Gilg [38] and the leaves of Chimonanthus spp. [39]. 3,5-DTBP is reported in the flowers of Aquilaria sinensis (Lour.) Gilg [40] and the seeds of Plukenetia volubilis L. [41]. From genus Bidens, this compound has previously been identified from B. Pilosa [42]. However, this compound is not reported in B. biternata.
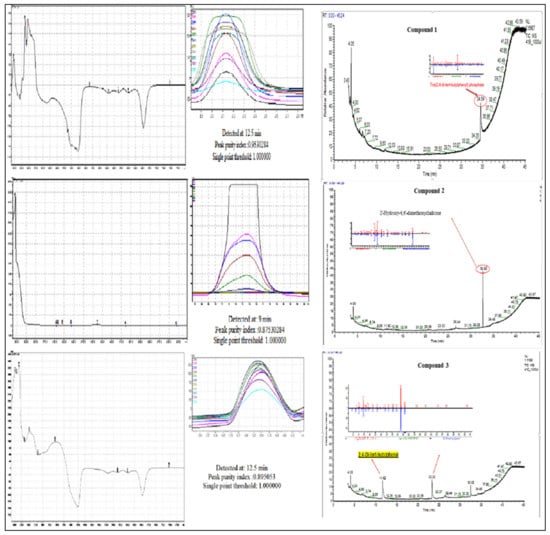
Figure 9.
UV–vis apex absorption spectra and mass spectra of Compound 1 Tris (2,4-di-tert-butylphenyl), 2 (4-Hydroxy-2,4′-dimethoxychalcone), and 3 (2,4-di-tert-butylphenol).
Compound 2 is identified to be 4-Hydroxy-2,4′-dimethoxychalcone (Figure 8, Figure 9 and Figure 10). The compound is 4-Hydroxy-2,4′-dimethoxychalcone (C17H16O4) belongs to the class of organic compounds known as chalcones. They are one of the leading classes of flavonoids throughout the entire kingdom of plants. Chalcones are reported to have clinical applications in humans. Previously Licochalcones isolated from licorice has been listed to have an array of biological activities [43].
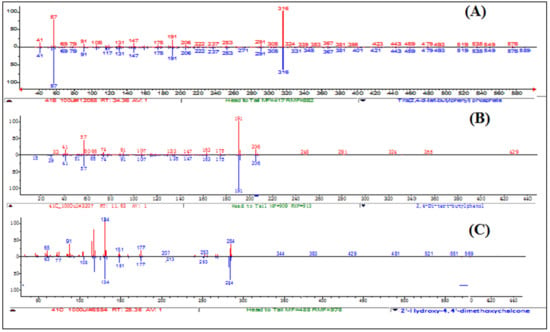
Figure 10.
Mass spectra of Compound (A) Tris (2,4-di-tert-butylphenyl), (B) (2,4-di-tert-butylphenol), and (C) (4-Hydroxy-2,4′-dimethoxychalcone).
Compound 3 is a yellow powder identified to be 2,4-di-tert-butylphenol (Figure 8, Figure 9 and Figure 10) with molecular formula C14H22O. This compound is a member of the class of phenols with two tert-butyl substituents at positions 2 and 4. This compound is previously been reported from variety of plants i.e., from chloroform and methanol extracts of Cuscuta reflexa [44], methanolic extract of Cordia dicodoma, Malvastrum coromandelianum (L.) Garcke leaves [45]. However, there is no report of this compound “Tris (2,4-di-tert-butylphenyl) phosphate” from Asteraceae members.
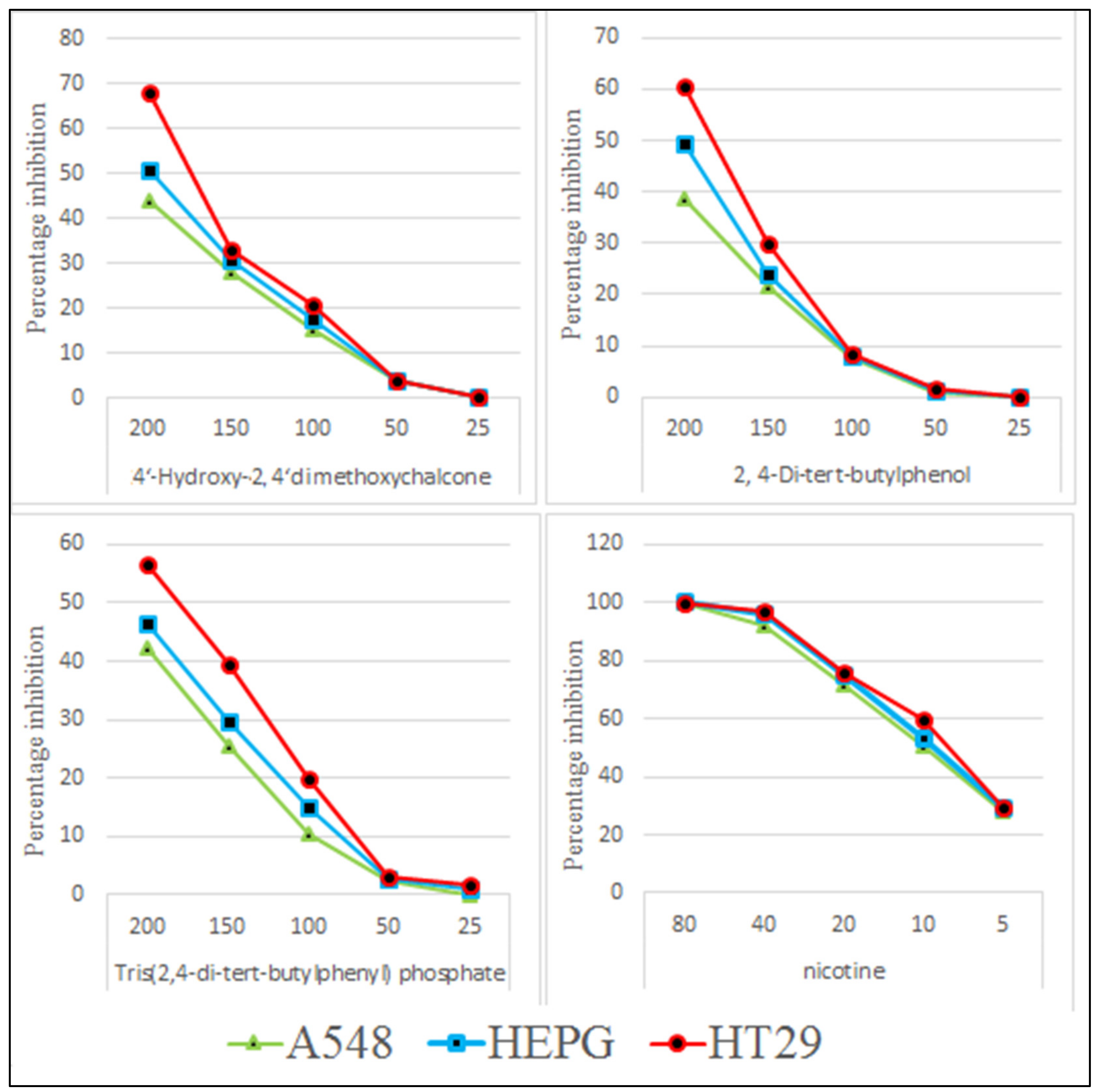
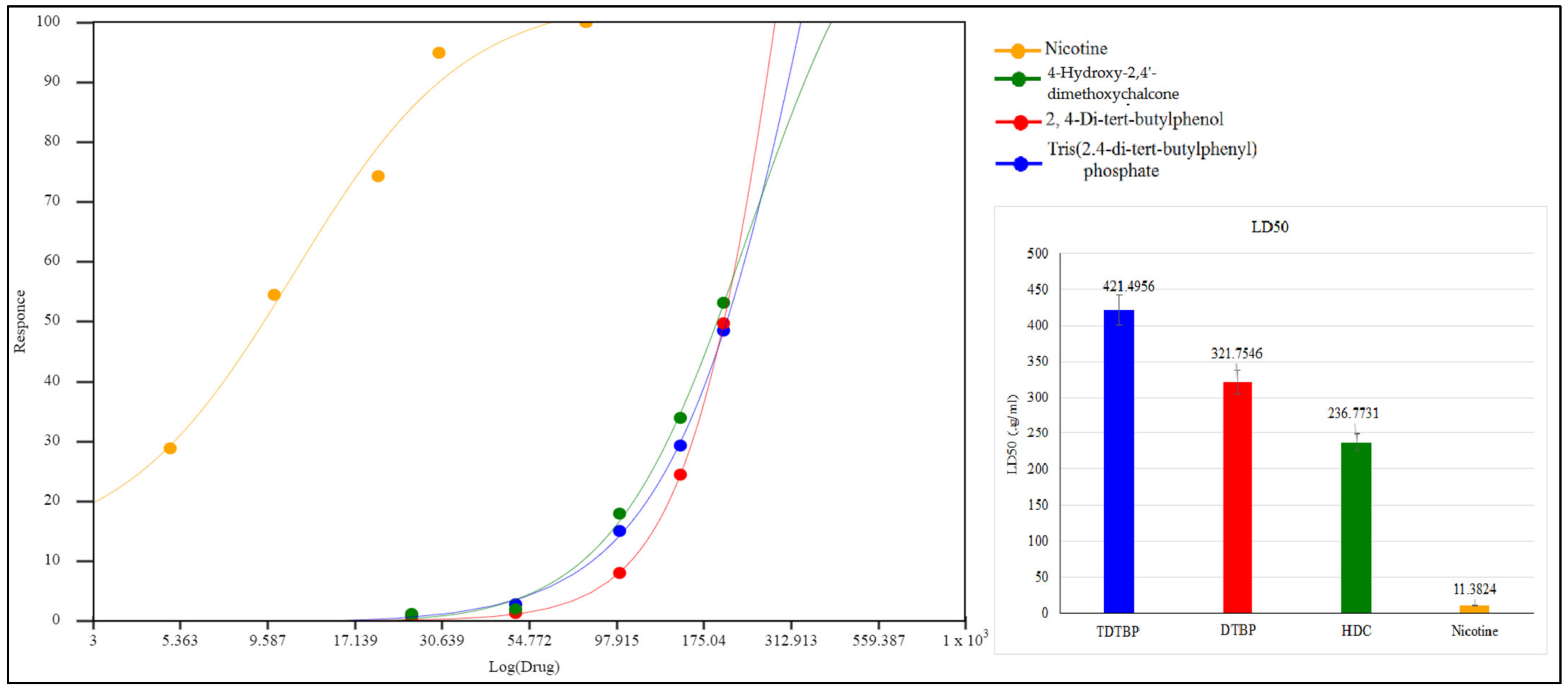
The isolated pure compounds show a moderate cytotoxicity against tested cell lines (Figure 11). Highest activity is observed by 4-Hydroxy-2,4′-dimethoxychalcone with LD50 of 236.7 µg/mL. Previously different reports on cytotoxic activity of chalcones been reported i.e., IC50 values of 45.39 µg/mL and 41.73 µg/mL against MCF-7 and SK-Hep-1 cell lines [46]. 2,4-di-tert-butylphenol show a moderate cytotoxicity with LD50 of 321.7 µg/mL (Figure 12). It is proposed that that cytotoxic properties of 2,4-di-tert-butylphenol is because it displayed greater results in the initiation of apoptotic [47].
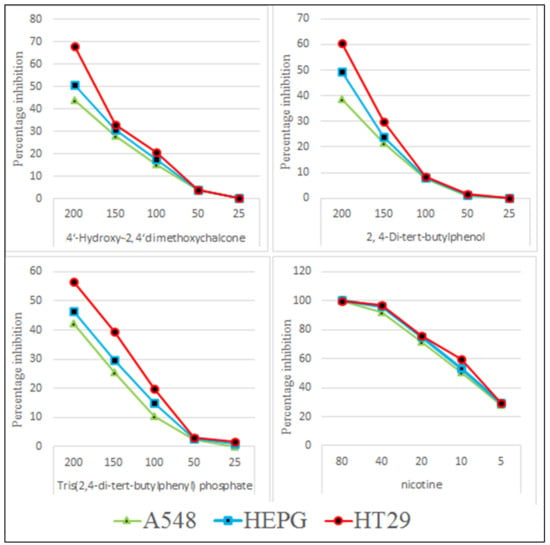
Figure 11.
Cytotoxic potential of TDTBP: Tris (2,4-di-tert-butylphenyl), HDC: 2 (4-Hydroxy-2,4′-dimethoxychalcone), and DTBP 3 (2,4-di-tert-butylphenol).
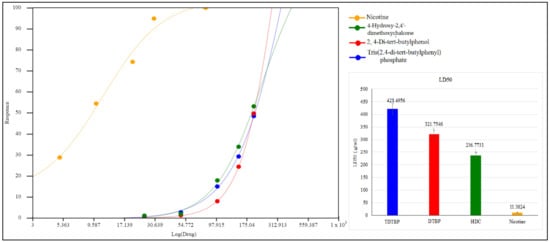
Figure 12.
LD50 µg/mL of test compounds TDTBP: Tris (2,4-di-tert-butylphenyl), HDC: (4-Hydroxy-2,4′-dimethoxychalcone), and DTBP 3 (2,4-di-tert-butylphenol) and the standard (nicotine).
4. Conclusions
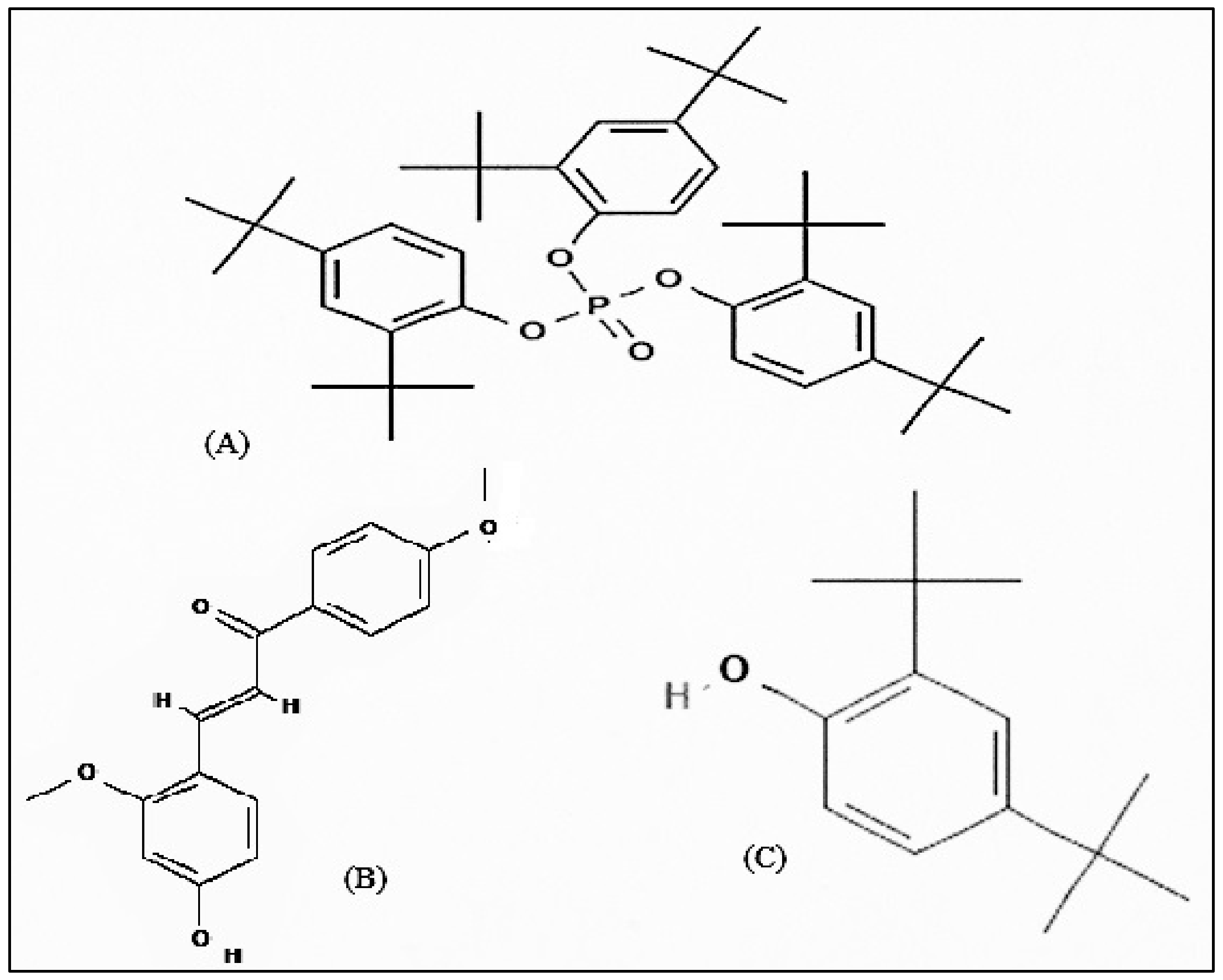
The members of genus Bidens are widely documented to be used for treating infectious diseases, immunological disorders, metabolic syndrome, wounds, and many others. In the current study we can conclude that B. biternata has anti-cancer constituents active against the Hela, A549 and HEPG cells. The roots ethanol extract of B. biternata is appeared to have highest anticancer potential. The compound responsible for anticancer activity are tris (2,4-di-tert-butylphenyl), (4-hydroxy-2,4′-dimethoxychalcone) and (2,4-di-tert-butylphenol) (Figure 13). These isolated compounds from this extract show a notable anticancer activity especially 4-hydroxy-2,4′-dimethoxychalcone show a promising potential to be chemically standardized for chemoprevention and for treating certain types of cancer in association with conventional treatments.
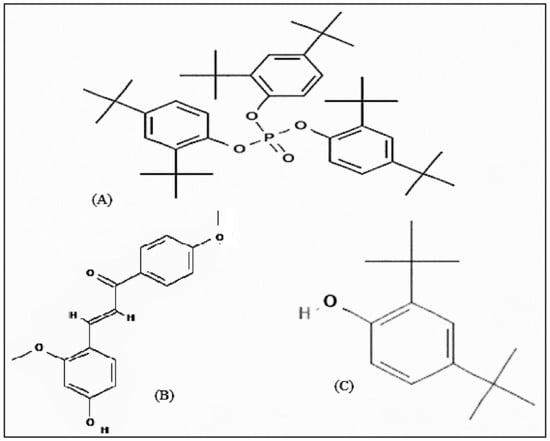
Figure 13.
Chemical structure of isolated compounds: (A) Tris (2,4-di-tert-butylphenyl), (B) (4-Hydroxy-2,4′-dimethoxychalcone), and (C) (2,4-di-tert-butylphenol).
Author Contributions
Y.B. and S.N. conceived of the idea; K.Z. conducted the experiment. A.S., N.A., S.K., A.Q. and W.A. conducted the literature review; S.N. provided technical expertise; S.M. helped with statistical analysis; Y.B. and S.N. proofread and provided intellectual guidance; R.S., A.A.M.A.-M. and A.H.A. contributed to obtaining funding. All authors read the first draft, helped in revision, and approved the article. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported byTaif University Researchers Supporting Project Number (TURSP-2020/140), Taif University, Taif, Saudi Arabia. Princess Nourah bint Abdulrahman University Researchers Supporting Project Number (PNURSP2022R249), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. Also, the authors thank Prince Sattam Bin Abdulaziz University, Al-Kharj, Saudi Arabia for their scientific contributions.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Available upon request from the corresponding author.
Acknowledgments
The authors acknowledge the support of Taif University Researchers Supporting Project Number (TURSP-2020/140), Taif University, Taif, Saudi Arabia. Princess Nourah bint Abdulrahman University Researchers Supporting Project Number (PNURSP2022R249), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. Also, the authors thank Prince Sattam Bin Abdulaziz University, Al-Kharj, Saudi Arabia for their scientific contributions.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Not applicable.
References
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly) phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxidants 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Oesch, F. Repurposing of plant alkaloids for cancer therapy: Pharmacology and toxicology. In Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 143–163. [Google Scholar]
- Zahara, K.; Bibi, Y.; Arshad, M.; Kaukab, G.; Ayoubi, S.A.; Qayyum, A. In-vitro examination and isolation of antidiarrheal compounds using five bacterial strains from invasive species Bidens bipinnata L. Saudi J. Biol. Sci. 2022, 29, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Maarfia, S.; Zellagui, A. Study of Essential Oils and Phenolic Compounds Their Changes and Anticancer Activity in Some Species Belonging to Asteraceae and Lamiaceae Families. 2019. Available online: http://bib.univ-oeb.dz:8080/jspui/handle/123456789/9129 (accessed on 6 March 2022).
- Matos, M.J.; Santana, L.; Uriarte, E.; Abreu, O.A.; Molina, E.; Yordi, E.G. Coumarins—An important class of phytochemicals. Phytochem.-Isol. Charact. Role Hum. Health 2015, 25, 113–140. [Google Scholar]
- Yang, M.-T.; Lin, Y.-X.; Yang, G.; Kuo, T.-F.; Liang, Y.-C.; Lee, T.-H.; Chang, C.L.-T.; Yang, W.-C. Functional and Mechanistic Studies of Two Anti-coccidial Herbs, Bidens pilosa and Artemisia indica. Planta Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Deba, F.; Xuan, T.D.; Yasuda, M.; Tawata, S. Chemical composition and antioxidant, antibacterial and antifungal activities of the essential oils from Bidens pilosa Linn. var. Radiata. Food Control 2008, 19, 346–352. [Google Scholar] [CrossRef]
- Yuan, L.p.; Chen, F.h.; Ling, L.; Bo, H.; Chen, Z.w.; Li, F.; Zhong, M.m.; Xia, L.j. Protective effects of total flavonoids of Bidens bipinnata L. against carbon tetrachloride-induced liver fibrosis in rats. J. Pharm. Pharmacol. 2008, 60, 1393–1402. [Google Scholar] [CrossRef]
- Sukumaran, P.; Nair, A.G.; Chinmayee, D.M.; Mini, I.; Sukumaran, S.T. Phytochemical Investigation of Bidens biternata (Lour.) Merr. and Sheriff.—A Nutrient-Rich Leafy Vegetable from Western Ghats of India. Appl. Biochem. Biotechnol. 2012, 167, 1795–1801. [Google Scholar] [CrossRef]
- Bartolome, A.P.; Villaseñor, I.M.; Yang, W.-C. Bidens pilosa L. (Asteraceae): Botanical properties, traditional uses, phytochemistry, and pharmacology. Evid.-Based Complement. Altern. Med. 2013, 2013, 340215. [Google Scholar] [CrossRef] [Green Version]
- Pérez, M.; Boffill, M.A.; Morón, F.J.; Sueiro, M.L.; Marrero, E.; Betancourt, E. Ethnopharmacological and preclinical study of diuretic activity inmedicinal and food plants used by cuban population. Emir. J. Food Agric. 2011, 23, 214–221. [Google Scholar]
- Orwa, J.A.; Jondiko, I.; Minja, R.J.; Bekunda, M. The use of Toddalia asiatica (L) Lam. (Rutaceae) in traditional medicine practice in East Africa. J. Ethnopharmacol. 2008, 115, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Grierson, D.; Afolayan, A. Antibacterial activity of some indigenous plants used for the treatment of wounds in the Eastern Cape, South Africa. J. Ethnopharmacol. 1999, 66, 103–106. [Google Scholar] [CrossRef]
- Pachter, L.M.; Cloutier, M.M.; Bernstein, B.A. Ethnomedical (folk) remedies for childhood asthma in a mainland Puerto Rican community. Arch. Pediatr. Adolesc. Med. 1995, 149, 982–988. [Google Scholar] [CrossRef]
- Zahara, K.; Bibi, Y.; Masood, S.; Nisa, S.; Qayyum, A.; Ishaque, M.; Shahzad, K.; Ahmed, W.; Shah, Z.H.; Alsamadany, H.; et al. Using HPLC–DAD and GC–MS analysis isolation and identification of anticandida compounds from Gui Zhen Cao Herbs (Genus Bidens): An important Chinese medicinal formulation. Molecules 2021, 26, 5820. [Google Scholar] [CrossRef]
- Anyinam, C. Ecology and ethnomedicine: Exploring links between current environmental crisis and indigenous medical practices. Soc. Sci. Med. 1995, 40, 321–329. [Google Scholar] [CrossRef]
- Romero-Benavides, J.C.; Ruano, A.L.; Silva-Rivas, R.; Castillo-Veintimilla, P.; Vivanco-Jaramillo, S.; Bailon-Moscoso, N. Medicinal plants used as anthelmintics: Ethnomedical, pharmacological, and phytochemical studies. Eur. J. Med. Chem. 2017, 129, 209–217. [Google Scholar] [CrossRef]
- Fornet, A.R. Ethnobotany in HolguÃn, Cuba: Cultural and natural heritage to be preserved. Rev. Cuba. Plantas Med. 2018, 22. [Google Scholar]
- Svetaz, L.; Zuljan, F.; Derita, M.; Petenatti, E.; Tamayo, G.; Cáceres, A.; Cechinel Filho, V.; Giménez, A.; Pinzón, R.; Zacchino, S.A. Value of the ethnomedical information for the discovery of plants with antifungal properties. A survey among seven Latin American countries. J. Ethnopharmacol. 2010, 127, 137–158. [Google Scholar] [CrossRef] [PubMed]
- Nole, T.; Lionel, T.; Cedrix, T.; Gabriel, A. Ethnomedical and ethnopharmacological study of plants used for potential treatments of diabetes and arterial hypertension by indigenous people in three phytogeographic regions of Cameroon. Diabetes Case Rep. 2016, 1, 2. [Google Scholar] [CrossRef] [Green Version]
- Fabricant, D.S.; Farnsworth, N.R. The value of plants used in traditional medicine for drug discovery. Environ. Health Perspect. 2001, 109, 69–75. [Google Scholar]
- Zahara, K.; Bibi, Y.; Tabassum, S.; Mudrikah; Bashir, T.; Haider, S.; Araa, A.; Ajmal, M. A review on pharmacological properties of Bidens biternata: A potential nutraceutical. Asian Pac. J. Trop. Dis. 2015, 5, 595–599. [Google Scholar] [CrossRef]
- Sharma, A.; Bargali, K.; Pande, N. The allelopathic potential of bryophyte extract on seed germination and seedling growth of Bidens biternata. Nat. Sci. 2009, 7, 30–38. [Google Scholar]
- Wang, R.; Yan, W.; Quan, G.; Liu, S.; Zhang, J. Effects of light intensity on morphology and physiology of exotic invasive Bidens pilosa L. and non-invasive congener Bidens bipinnata L. Allelopath. J. 2017, 42, 157–168. [Google Scholar] [CrossRef]
- Lans, C. Comparison of plants used for skin and stomach problems in Trinidad and Tobago with Asian ethnomedicine. J. Ethnobiol. Ethnomed. 2007, 3, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ismail, N.; Ab Ghani, N.; Rusli, S.; Abdullah, N.; Awang, R. Cytotoxicity assessment of zirconia-reinforced experimental nanohybrid dental composite using MTT assay. Ann. Rom. Soc. Cell Biol. 2021, 2021, 14878–14886. [Google Scholar]
- Gagman, H.A.; Him, N.A.I.I.N.; Ahmad, H.; Sulaiman, S.F.; Zakaria, R.; Termizi, F.H.M. In Vitro Efficacy of Aqueous and Methanol Extract of Cassia siamea against the Motility of Caenorhabditis elegans. Trop. Life Sci. Res. 2020, 31, 145–159. [Google Scholar] [CrossRef]
- Van Dyck, A.; Bollaerts, I.; Beckers, A.; Vanhunsel, S.; Glorian, N.; van Houcke, J.; van Ham, T.J.; De Groef, L.; Andries, L.; Moons, L. Müller glia–myeloid cell crosstalk accelerates optic nerve regeneration in the adult zebrafish. Glia 2021, 69, 1444–1463. [Google Scholar] [CrossRef]
- Sasidharan, R.; Gerstein, M. Protein fossils live on as RNA. Nature 2008, 453, 729–731. [Google Scholar] [CrossRef]
- Van Wyk, B.-E.; Wink, M. Medicinal Plants of the World; CABI: Wallingford, UK, 2018. [Google Scholar]
- Pengelly, A. The Constituents of Medicinal Plants; CABI: Wallingford, UK, 2021. [Google Scholar]
- Karagöz, A.; Turgut-Kara, N.; Çakır, Ö.; Demirgan, R.; Arı, Ş. Cytotoxic activity of crude extracts from Astragalus chrysochlorus (Leguminosae). Biotechnol. Biotechnol. Equip. 2007, 21, 220–222. [Google Scholar] [CrossRef]
- Nemati, F.; Dehpouri, A.A.; Eslami, B.; Mahdavi, V.; Mirzanejad, S. Cytotoxic properties of some medicinal plant extracts from Mazandaran, Iran. Iran. Red Crescent Med. J. 2013, 15, e8871. [Google Scholar] [CrossRef] [Green Version]
- Di Paolo, C.; Seiler, T.-B.; Keiter, S.; Hu, M.; Muz, M.; Brack, W.; Hollert, H. The value of zebrafish as an integrative model in effect-directed analysis—A review. Environ. Sci. Eur. 2015, 27, 8. [Google Scholar] [CrossRef] [Green Version]
- Ganguly, P.; Breen, A.; Pillai, S.C. Toxicity of nanomaterials: Exposure, pathways, assessment, and recent advances. ACS Biomater. Sci. Eng. 2018, 4, 2237–2275. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-Y.; Chen, G.-R.; Pan, C.-X.; Deng, Z.-Y.; Ge, J.-F.; Li, N.; Chen, F.-H. Polyacetylenes from Bidens bipinnata L. and their biological activities. Phytochem. Lett. 2014, 7, 198–201. [Google Scholar] [CrossRef]
- Mei, W.-L.; Zeng, Y.-B.; Liu, J.; Dai, H.-F. GC-MS analysis of volatile constituents from five different kinds of Chinese eaglewood. J. Chin. Med. Mater. 2007, 30, 551–555. [Google Scholar]
- Liu, H. Study on the Chemical Composition Analysis and ISSR Molecular Marker of Chimonanthus. Master’s Thesis, Zhejiang Forestry University, Hangzhou, China, 2013. [Google Scholar]
- Gao, Y.; Guo, L.; Meng, X.; Zhang, L.; Yang, G. The optimal GC-MS analysis of essential oil (fresh, dried and bud) and aroma enhanced by β-glucosidase on Aesculus chinensis flowers. Prod. Specif. Importing China 2018, 5, 1–4. [Google Scholar]
- Chen, H.; Peng, Y.; Liu, G.; Li, H.; Gao, L.; Zhan, N.; Xie, Y. Dynamic changes of volatile components from developing seeds of Plukenetia volubilis. Sci. Silvae Sin. 2018, 54, 157–168. [Google Scholar]
- Ndiege, M.L.; Kengara, F.; Maiyoh, G.K. Characterization of Phenolic Compounds from Leaf Extract of Bidens pilosa Linn. Var. Radiata. South Asian Res. J. Nat. Prod. 2021, 4, 44–58. [Google Scholar]
- Freitas, S.; Costa, S.; Azevedo, C.; Carvalho, G.; Freire, S.; Barbosa, P.; Velozo, E.; Schaer, R.; Tardy, M.; Meyer, R. Flavonoids inhibit angiogenic cytokine production by human glioma cells. Phytother. Res. 2011, 25, 916–921. [Google Scholar] [CrossRef]
- Afrin, N.S.; Hossain, M.A.; Saha, K. Phytochemical screening of plant extracts and GC-MS analysis of n-Hexane soluble part of crude chloroform extract of Cuscuta reflexa (Roxb.). J. Pharmacogn. Phytochem. 2019, 8, 560–564. [Google Scholar]
- Saxena, S.; Rao, P. GC-MS screening of bioactive constituents and antioxidant profiling in an invasive weed, Malvastrum coromandelianum (L.) Garcke. Pharma Innov. 2018, 7, 738–746. [Google Scholar]
- Ding, Y.; Nguyen, H.T.; Kim, S.I.; Kim, H.W.; Kim, Y.H. The regulation of inflammatory cytokine secretion in macrophage cell line by the chemical constituents of Rhus sylvestris. Bioorgan. Med. Chem. Lett. 2009, 19, 3607–3610. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.V.; Jayasree, D.V.; Biju, P.G.; Baby, S. Anti-inflammatory and anticancer activities of erythrodiol-3-acetate and 2,4-di-tert-butylphenol isolated from Humboldtia unijuga. Nat. Prod. Res. 2020, 34, 2319–2322. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).