Analysis of the Composition of Deinagkistrodon acutus Snake Venom Based on Proteomics, and Its Antithrombotic Activity and Toxicity Studies
Abstract
:1. Introduction
2. Material and Methods
2.1. Materials Preparation
2.2. Venom Proteome Analysis
2.2.1. Nano-Liquid Chromatography–Tandem Mass Spectrometry Analysis
2.2.2. Protein Identification
2.2.3. Protein Function and Interaction Analyses
2.3. Lethal Activity of Deinagkistrodon acutus Venom
2.4. In Vitro Platelet Aggregation Assay
2.5. Anti-Coagulant Activity
Blood Clotting Assays
2.6. Collagen-Adrenalin-Induced Acute Pulmonary Thrombosis in Mice
2.7. Carrageenan-Induced Tail Thrombosis in Mice
2.8. Flow Restriction of the Inferior Vena Cava (IVC) in Mouse Induced Thrombosis
2.9. Statistical Analysis
3. Results
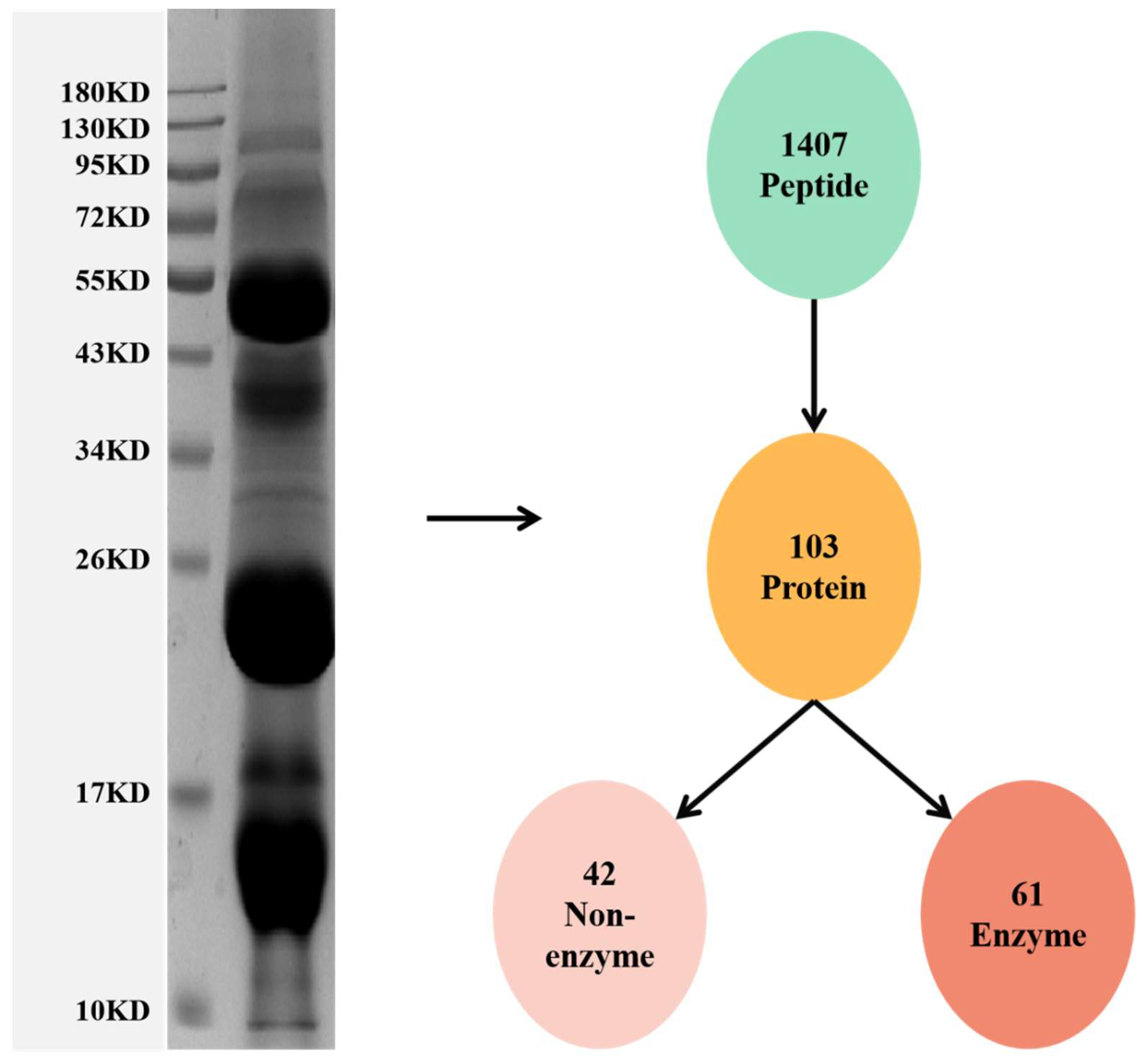
3.1. Proteomic Analysis of the Venom of Deinagkistrodon Acutus
3.2. Acute Toxicity Test of Deinagkistrodon acutus Venom
3.3. Coagulant Activity In Vivo
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| ADP | Adenosine diphosphate |
| PLT | Platelet |
| PRP | Platelet Rich Plasma |
| PPP | Platelet-Poor Plasma |
| SPF | Specific pathogen Free |
| IVC | Inferior Vena Cava |
| LC-MS/MS | Liquid chromatography-tandem mass spectrometer |
| GO | Gene ontology Terms |
| iBAQ | Intensity based absolute quantification |
References
- Saviola, A.J.; Peichoto, M.E.; Mackessy, S.P. Rear-fanged snake venoms: An untapped source of novel compounds and potential drug leads. Toxin Rev. 2014, 33, 185–201. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Calvete, J.J.; Habib, A.G.; Harrison, R.A.; Williams, D.J.; Warrell, D.A. Snakebite envenoming. Nat. Rev. Dis. Primers 2017, 3, 17063. [Google Scholar] [CrossRef] [PubMed]
- Scheib, H.; Tyndall, J.D.; King, G.F.; Nevalainen, T.J.; Norman, J.A. The Toxicogenomic Multiverse: Convergent Recruitment of Proteins into Animal Venoms. Annu. Rev. Genom. Hum. Genet. 2009, 10, 483–511. [Google Scholar]
- Fox, J.W.; Serrano, S.M. Exploring snake venom proteomes: Multifaceted analyses for complex toxin mixtures. Proteomics 2008, 8, 909–920. [Google Scholar] [CrossRef]
- Gutierrez, J.M.; Lomonte, B.; Leon, G.; Alape-Giron, A.; Flores-Diaz, M.; Sanz, L.; Angulo, Y.; Calvete, J.J. Snake venomics and antivenomics: Proteomic tools in the design and control of antivenoms for the treatment of snakebite envenoming. J. Proteom. 2009, 72, 165–182. [Google Scholar] [CrossRef]
- Cheng, C.-L.; Mao, Y.-C.; Liu, P.-Y.; Chiang, L.-C.; Liao, S.-C.; Yang, C.-C. Deinagkistrodon acutus envenomation: A report of three cases. J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 20–28. [Google Scholar] [CrossRef] [Green Version]
- Chen, P.C.; Huang, M.N.; Chang, J.F.; Liu, C.C.; Chen, C.K.; Hsieh, C.H. Snake venom proteome and immuno-profiling of the hundred-pace viper, Deinagkistrodon acutus, in Taiwan. Acta Trop. 2018, 189, 137–144. [Google Scholar] [CrossRef]
- Shan, L.; Gao, J.; Zhang, Y.; Shen, S.; He, Y.; Wang, J.; Ma, X.; Ji, X. Proteomic characterization and comparison of venoms from two elapid snakes (Bungarus multicinctus and Naja atra) from China. J. Proteom. 2016, 138, 83–94. [Google Scholar] [CrossRef]
- Wong, K.Y.; Tan, C.H.; Tan, N.H. Venom and Purified Toxins of the Spectacled Cobra (Naja naja) from Pakistan: Insights into Toxicity and Antivenom Neutralization. Am. J. Trop. Med. Hyg. 2016, 94, 1392–1399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei-Hsin, H.; Jie, S.; Wen-Yee, C.; Lu-Ting, C.; Hei-Tung, Y.; Cheng-Chung, W.J. Risk of stroke with antivenom usage after venomous snakebite in Taiwan: A population-based cohort study. QJM Int. J. Med. 2021, 10, hcab259. [Google Scholar]
- Li, Q.B.; Yu, Q.S.; Huang, G.W.; Tokeshi, Y.; Nakamura, M.; Kinjoh, K.; Kosugi, T. Hemostatic disturbances observed in patients with snakebite in south China. Toxicon 2000, 38, 1355–1366. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, Q.; Yin, W.; Zhang, X.; Huang, Y.; Luo, Y.; Qiu, P.; Su, X.; Yu, J.; Hu, S.; et al. Transcriptome analysis of Deinagkistrodon acutus venomous gland focusing on cellular structure and functional aspects using expressed sequence tags. BMC Genom. 2006, 7, 152. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.X.; Shi, Y.Y.; Shan, C.M.; Wang, T.; Wang, Z.X.; Wang, S.S.; Wu, J.W. Transcriptome analysis of venom glands of Qishen and mining of snake venom protein functional genes. China J. Tradit. Chin. Med. 2019, 44, 10. [Google Scholar]
- Yin, W.; Wang, Z.J.; Li, Q.Y.; Lian, J.M.; Zhou, Y.; Lu, B.Z.; Jin, L.J.; Qiu, P.X.; Zhang, P.; Zhu, W.B.; et al. Evolutionary trajectories of snake genes and genomes revealed by comparative analyses of five-pacer viper. Nat. Commun. 2016, 7, 13107. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; He, Q.; Zhou, B.; Huang, D.; Chen, J.; Chen, Q.; Yang, S.; Yu, X. Deinagkistrodon acutusExploring the five-paced viper venom proteome by integrating a combinatorial peptide ligand library approach with shotgun LC-MS/MS. J. Venom. Anim. Toxins Incl. Trop. Dis. 2021, 27, e20200196. [Google Scholar] [CrossRef]
- Chen, M.; Ye, X.; Ming, X.; Chen, Y.; Wang, Y.; Su, X.; Su, W.; Kong, Y. A novel direct factor Xa inhibitory peptide with anti-platelet aggregation activity from Deinagkistrodon acutus venom hydrolysates. Sci. Rep. 2015, 5, 10846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, B.; Xu, Z.; Qian, C.; Jiang, F.; Ding, X.; Ruan, Y.; Ding, Z.; Fan, Y. Antiplatelet Aggregation and Antithrombosis Efficiency of Peptides in the Snake Venom of Deinagkistrodon acutus: Isolation, Identification, and Evaluation. Evid. Based Complement. Altern. Med. 2015, 2015, 412841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J.; Fan, H.; Yin, X.J.; Huang, F. Isolation of a novel metalloproteinase from Agkistrodon venom and its antithrombotic activity analysis. Int. J. Mol. Sci. 2019, 20, 4088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J.; Song, W.; Hua, H.; Yin, X.; Huang, F.; Alolga, R.N. Antithrombotic and anticoagulant effects of a novel protein isolated from the venom of the Deinagkistrodon acutus snake—ScienceDirect. Biomed. Pharmacother. 2021, 138, 111527. [Google Scholar] [CrossRef] [PubMed]
- Herrera, M.; Fernández, J.; Vargas, M.; Villalta, M.; Segura, Á.; León, G.; Angulo, Y.; Paiva, O.; Matainaho, T.; Jensen, S.D.; et al. Comparative proteomic analysis of the venom of the taipan snake, Oxyuranus scutellatus, from Papua New Guinea and Australia: Role of neurotoxic and procoagulant effects in venom toxicity. J. Proteom. 2012, 75, 2128–2140. [Google Scholar] [CrossRef]
- Xie, C.; Albulescu, L.O.; Still, K.; Slagboom, J.; Zhao, Y.; Jiang, Z.; Somsen, G.W.; Vonk, F.J.; Casewell, N.R.; Kool, J. Varespladib Inhibits the Phospholipase A2 and Coagulopathic Activities of Venom Components from Hemotoxic Snakes. Biomedicines 2020, 8, 165. [Google Scholar] [CrossRef] [PubMed]
- Katayama, H.; Nagasu, T.; Oda, Y. Improvement of in-gel digestion protocol for peptide mass fingerprinting by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2010, 15, 1416–1421. [Google Scholar] [CrossRef] [PubMed]
- Ogata, H.; Goto, S.; Sato, K.; Fujibuchi, W.; Bono, H.; Kanehisa, M. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 1999, 27, 29–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szklarczyk, D.; Franceschini, A.; Wyder, S.; Forslund, K.; Heller, D.; Huerta-Cepas, J.; Simonovic, M.; Roth, A.; Santos, A.; Tsafou, K.P.; et al. STRING v10: Protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 2015, 43, D447–D452. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hao, J.; Gao, W.; Liu, Z.; Wu, S.; Jing, S. Study on hemostatic activities of the rhizome of Paris bashanensis. Pharm. Biol. 2013, 51, 1321–1325. [Google Scholar] [CrossRef]
- Li, H.; Huang, W.; Wen, Y.; Gong, G.; Zhao, Q.; Yu, G. Anti-thrombotic activity and chemical characterization of steroidal saponins from Dioscorea zingiberensis c.h. wright. Fitoterapia 2010, 81, 1147–1156. [Google Scholar]
- Li-Ming, L.; Lin, K.H.; Huang, L.T.; Tseng, M.F.; Chiu, H.C.; Chen, R.J.; Lu, W.J. Licochalcone A Prevents Platelet Activation and Thrombus Formation through the Inhibition of PLCγ2-PKC, Akt, and MAPK Pathways. Int. J. Mol. Sci. 2017, 18, 1500. [Google Scholar]
- Martinod, K.; Witsch, T.; Farley, K.; Gallant, M.; Remold-O’Donnell, E.; Wagner, D.D. Neutrophil elastase-deficient mice form neutrophil extracellular traps in an experimental model of deep vein thrombosis. J. Thromb. Haemost. 2016, 14, 551–558. [Google Scholar] [CrossRef] [Green Version]
- Tadayuki, Y.; Liu, Z.; Ahamed, J.; McEver, R.P. Cooperative PSGL-1 and CXCR2 signaling in neutrophils promotes deep vein thrombosis in mice. Blood 2018, 132, 1426–1437. [Google Scholar]
- Fox, J.W.; Serrano, S. Timeline of key events in snake venom metalloproteinase research. J. Proteom. 2009, 72, 200–209. [Google Scholar] [CrossRef]
- Lalloo, D.G.; Trevett, A.J.; Owens, D.; Minei, J.; Naraqi, S.; Saweri, A.; Hutton, R.A.; Theakston, R.D.; Warrell, D.A. Coagulopathy following bites by the Papuan taipan (Oxyuranus scutellatus canni). Blood Coagul. Fibrinolysis 1995, 6, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Souza, D.; Gomes, M.; Ferreira, F.B.; Rodrigues, R.S.; Achê, D.C.; Richardson, M.; Borges, M.H.; Rodrigues, V.M. Biochemical and enzymatic characterization of BpMP-I, a fibrinogenolytic metalloproteinase isolated from Bothropoides pauloensis snake venom. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2012, 161, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Rucavado, A.; Escalante, T.; Díaz, C. Hemorrhage induced by snake venom metalloproteinases: Biochemical and biophysical mechanisms involved in microvessel damage. Toxicon 2005, 45, 1007–1011. [Google Scholar] [CrossRef] [PubMed]
- Undas, A.; Ariëns, R.A.S. Fibrin Clot Structure and Function a Role in the Pathophysiology of Arterial and Venous Thromboembolic Diseases. Arterioscler. Thromb. Vasc. Biol. 2010, 31, 88–99. [Google Scholar]
- Bennett, J.S. Platelet-Fibrinogen Interactions. Ann. N. Y. Acad. Sci. 2001, 936, 340–354. [Google Scholar] [CrossRef]
- Lord, S.T. Molecular mechanisms affecting fibrin structure and stability. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 494–499. [Google Scholar] [CrossRef] [Green Version]
- Harris, J.B.; Grubb, B.D.; Maltin, C.A.; Dixon, R. The Neurotoxicity of the Venom Phospholipases A2, Notexin and Taipoxin. Exp. Neurol. 2000, 161, 520–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamiya, N.; Yagi, T. Studies on sea snake venom. Proc. Jpn. Acad. 2011, 87, 41–52. [Google Scholar] [CrossRef] [Green Version]
- Kordi, D.F.; Gubenšek, F. Ammodytoxin C gene helps to elucidate the irregular structure of Crotalinae group II phospholipase A2 genes. Eur. J. Biochem. 2015, 240, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Takeda, S.; Takeya, H.; Iwanaga, S. Snake venom metalloproteinases: Structure, function and relevance to the mammalian ADAM/ADAMTS family proteins. Biochim. Biophys. Acta 2012, 1824, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Devaraja, S.; Girish, K.S.; Devaraj, V.R.; Kemparaju, K. Factor Xa-like and fibrin(ogen)olytic activities of a serine protease from Hippasa agelenoides spider venom gland extract. J. Thromb. Thrombolysis 2010, 29, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Ye, F.P.; Wang, J.; Liao, G.Y.; Zhang, Y.; Fan, Q.S.; Lee, W.H. Purification, characterization and gene cloning of Da-36, a novel serine protease from Deinagkistrodon acutus venom. Toxicon 2013, 67, 1–11. [Google Scholar] [CrossRef]
- Mackessy, S.P. Thrombin-Like Enzymes in Snake Venoms. Vopr. Meditsinskoi Khimii 2010, 39, 2–6. [Google Scholar]
- Liu, Y.P.; Kong, Y.; Li, Q. Research Progress in Snake Venom Thrombin-Like Enzymes. Pharm. Biotechnol. 2017, 24, 353–358. [Google Scholar]
- Castro, H.C.; Zingali, R.B.; Albuquerque, M.G.; Pujol-Luz, M.; Rodrigues, C.R. Snake venom thrombin-like enzymes: From reptilase to now. Cell. Mol. Life Sci. Cmls 2004, 61, 843. [Google Scholar] [CrossRef] [PubMed]
- Bell, W.R., Jr. Defibrinogenating Enzymes. Drugs 1997, 54, 18–31. [Google Scholar] [CrossRef] [PubMed]









| Protein ID | Description | Coverage | Protein Mass | Peptide | Unique Peptide | Uniq Spectrum | snak_iBAQ | % | Function |
|---|---|---|---|---|---|---|---|---|---|
| CAJ85790.1 | Phospholipase A2, acidic precursor, partial [Deinagkistrodon acutus] | 0.9756 | 14,031.6 | 25 | 25 | 1191 | 6,623,486.35 | 13.80 | Binding catalytic_activity, Inhibits platelet aggregation. |
| sp|Q9DEA2.1|SLCA_DEIAC | Snaclec agkicetin-C subunit alpha; | 0.7935 | 17,798.4 | 30 | 9 | 259 | 6,026,939.81 | 12.56 | Binding, Inhibits platelet aggregation, Inhibits platelet adhesion. |
| AAM22784.1 | Antithrombin A B-chain [Deinagkistrodon acutus] | 0.7671 | 16,688 | 22 | 22 | 678 | 5,987,824.836 | 12.48 | Inhibits platelet aggregation, Inhibits platelet adhesion. |
| AAL36975.1 | Lys-49 phospholipase A2 precursor [Deinagkistrodon acutus] | 0.6739 | 15,803.4 | 35 | 35 | 550 | 5,154,993.67 | 10.74 | Binding catalytic_activity, Indirect hemolytic activity and anticoagulant activity |
| AAF76377.1 | Thrombin-like protein acutobin precursor [Deinagkistrodon acutus] | 0.6923 | 28,814.8 | 29 | 25 | 917 | 3,435,511.45 | 7.16 | Catalytic_activity, Coagulates human fibrinogen by hydrolysis of the alpha chains. |
| SPQ1ZY03.1|PA2B_DEIAC | Basic phospholipase A2 DAV-N6; Short = svPLA2; AltName: Full = Phosphatidylcholine 2-acylhydrolase; Flags: Precursor | 0.7174 | 15,853.5 | 16 | 16 | 310 | 2,603,255.33 | 5.43 | Binding catalytic_activity, Blocking acetylcholine release from the nerve termini. |
| AAL66390.1 | Antithrombin 1 B chain [Deinagkistrodon acutus] | 0.8591 | 17,249.8 | 41 | 15 | 274 | 2,102,366.73 | 4.38 | Inhibits platelet aggregation, Inhibits platelet adhesion. |
| SP|Q9W7S2.1|VM11_DEIAC | Snake venom metalloproteinase aculysin-1; Short = SVMP; Flags: Precursor | 0.4604 | 47,304.2 | 105 | 58 | 1689 | 1,918,894.51 | 4.00 | Binding catalytic_activity, Hemorrhagic activity |
| SP|Q9DEF9.1|SLAA_DEIAC | Snaclec anticoagulant protein subunit A; Short = AaACP-A; Flags: Precursor | 0.7829 | 17,124.2 | 27 | 19 | 1019 | 1,865,840.86 | 3.89 | Binding, Inhibits platelet aggregation, Antithrombotic and thrombolytic activities, Hydrolyzes the fibrinogen. |
| SP|Q8JIV8.1|SL_DEIAC | Snaclec clone 2100755; AltName: Full = C-type lectin clone 2100755; Flags: Precursor | 0.671 | 17,944.1 | 25 | 25 | 464 | 1,649,481.55 | 3.44 | Binding, Modulation of platelet aggregation, or coagulation cascade. |
| AAK26430.1 | Agkisasin-b [Deinagkistrodon acutus] | 0.7823 | 14,701.3 | 25 | 3 | 29 | 1,590,986.26 | 3.32 | Binding, Anticoagulant, Inhibits platelet aggregation, Antithrombotic and thrombolytic activities, Hydrolyzes the Aalpha-chain of fibrinogen. |
| AFR11355.1 | Snake venom serine protease Da-36 [Deinagkistrodon acutus] | 0.5654 | 29,057.1 | 17 | 12 | 110 | 874,312.48 | 1.82 | Catalytic_activity, Blood coagulation factor activity, Serine elastase activity. |
| AAN23124.1 | Agglucetin-alpha 1 subunit precursor [Deinagkistrodon acutus] | 0.8052 | 17,317.4 | 33 | 3 | 405 | 771,737.66 | 1.61 | Binding, Blood coagulation factor activity, Serine elastase activity. |
| AAM22785.1 | Agkisacutacin B-chain [Deinagkistrodon acutus] | 0.8288 | 16,739.8 | 37 | 7 | 488 | 746,482.39 | 1.56 | Binding, Anticoagulant, Inhibits platelet aggregation, Antithrombotic and thrombolytic activities, Hydrolyzes the Aalpha-chain of fibrinogen. |
| BAO23490.1 | Metalloproteinase [Deinagkistrodon acutus] | 0.4745 | 67,764.7 | 48 | 41 | 702 | 740,656.11 | 1.54 | Binding catalytic, Enzyme_regulator_activity, Metalloendoproteinase activity. |
| SP|Q9IAX6.1|VM2M2_DEIAC | Zinc metalloproteinase/disintegrin | 0.6438 | 52,517.3 | 117 | 25 | 762 | 740,180.03 | 1.54 | Binding, enzyme_regulator_activity, inhibits Platelet aggregation, Cleaves fibrinogen, Cleaves collagen and VWF. |
| PDB|3HDB|A | Chain A, Crystal Structure of Aahiv, A Metalloproteinase From Venom of Deinagkistrodon acutus | 0.8345 | 46,681.5 | 95 | 12 | 590 | 639,917.60 | 1.33 | Binding, Anticoagulant, Inhibits platelet aggregation, Antithrombotic and thrombolytic activities, Hydrolyzes the Aalpha-chain of fibrinogen. |
| SP|Q9IAM1.2|SLUA_DEIAC | Snaclec agkisacutacin subunit A; Short = Agk-A; Flags: Precursor | 0.7895 | 17,109.1 | 21 | 3 | 287 | 502,397.74 | 1.05 | Binding, Anticoagulant, Inhibits platelet aggregation, Antithrombotic and thrombolytic activities, Hydrolyzes the Aalpha-chain of fibrinogen. |
| ABB79955.1 | Agkihagin [Deinagkistrodon acutus] | 0.5789 | 67,572.2 | 50 | 48 | 969 | 500,625.222 | 1.045 | Binding, Enzyme_regulator_activity, Inhibits platelet aggregation. Hydrolyzes fibrinogen, Induces apoptosis and inhibits proliferation of endothelial cells. |
| AAF76378.1 | Thrombin-like protein DAV-PA precursor [Deinagkistrodon acutus] | 0.6008 | 28,031.8 | 21 | 19 | 133 | 435,941.55 | 0.91 | Catalytic_activity, Fibrinogenolytic activities, Esterolysis and amidolytic activities. |
| AEJ31983.1 | Phospholipase B [Crotalus adamanteus] | 0.5642 | 64,048.6 | 40 | 20 | 356 | 375,155.62 | 0.78 | Catalytic_activity, Hemolysis. |
| AAL66391.1 | Antithrombin 1 A chain [Deinagkistrodon acutus] | 0.8608 | 17,950.1 | 51 | 19 | 445 | 332,370.033 | 0.69 | Platelet aggregation, Binds to human platelets, Promotes angiogenesis. |
| JAG44660.1 | Glutaminyl-peptide cyclotransferase [Crotalus horridus] | 0.5815 | 42,369 | 16 | 16 | 116 | 320,694.98 | 0.67 | Binding |
| AAM22789.1 | ACF 1/2 B-chain [Deinagkistrodon acutus] | 0.6712 | 16,924.9 | 26 | 5 | 229 | 265,179.49 | 0.55 | Binding, Anticoagulant, Inhibits platelet aggregation, Antithrombotic and thrombolytic activities, Hydrolyzes the Aalpha-chain of fibrinogen. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, J.; Zhao, M.; Xue, C.; Liang, J.; Huang, F. Analysis of the Composition of Deinagkistrodon acutus Snake Venom Based on Proteomics, and Its Antithrombotic Activity and Toxicity Studies. Molecules 2022, 27, 2229. https://doi.org/10.3390/molecules27072229
Huang J, Zhao M, Xue C, Liang J, Huang F. Analysis of the Composition of Deinagkistrodon acutus Snake Venom Based on Proteomics, and Its Antithrombotic Activity and Toxicity Studies. Molecules. 2022; 27(7):2229. https://doi.org/10.3390/molecules27072229
Chicago/Turabian StyleHuang, Jin, Minrui Zhao, Chu Xue, Jiqiang Liang, and Fang Huang. 2022. "Analysis of the Composition of Deinagkistrodon acutus Snake Venom Based on Proteomics, and Its Antithrombotic Activity and Toxicity Studies" Molecules 27, no. 7: 2229. https://doi.org/10.3390/molecules27072229





