Synthesis of Cyclic Peptides in SPPS with Npb-OH Photolabile Protecting Group
Abstract
:1. Introduction
2. Results and Discussion
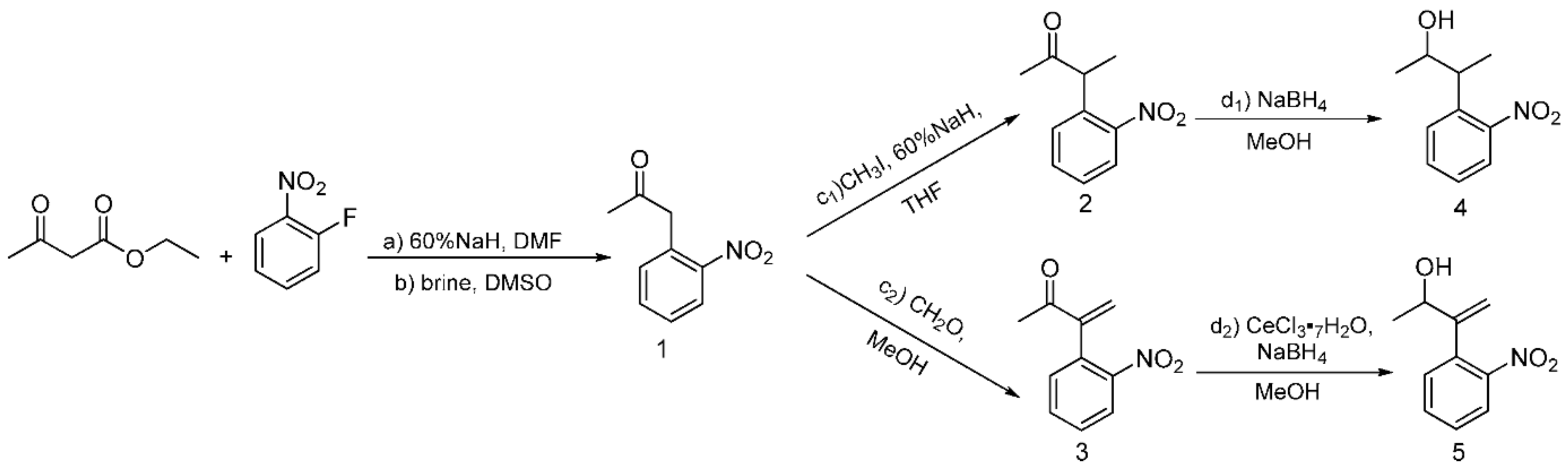
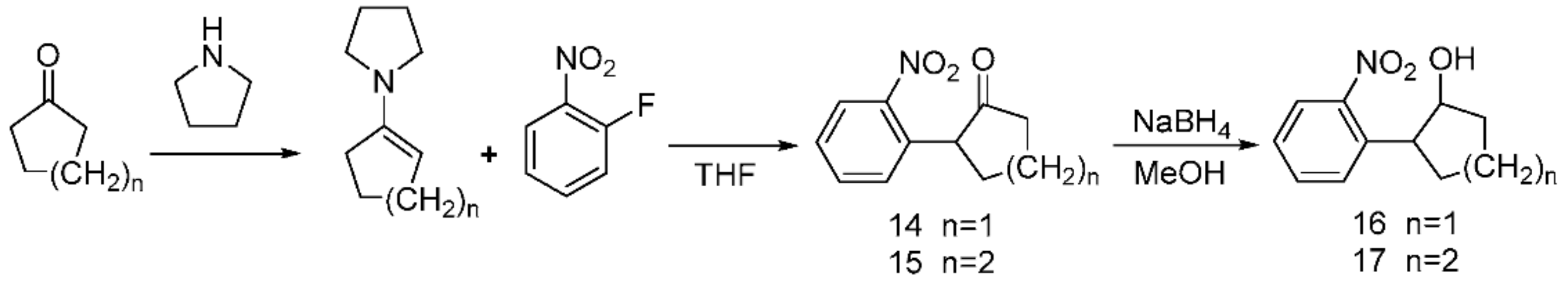
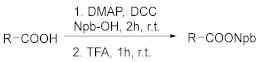
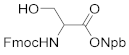
2.1. Synthesis of the Photolabile Protecting Groups (PPGs)
2.2. Characterization of PPGs
2.2.1. Acid and Alkali Stability of the PPGs
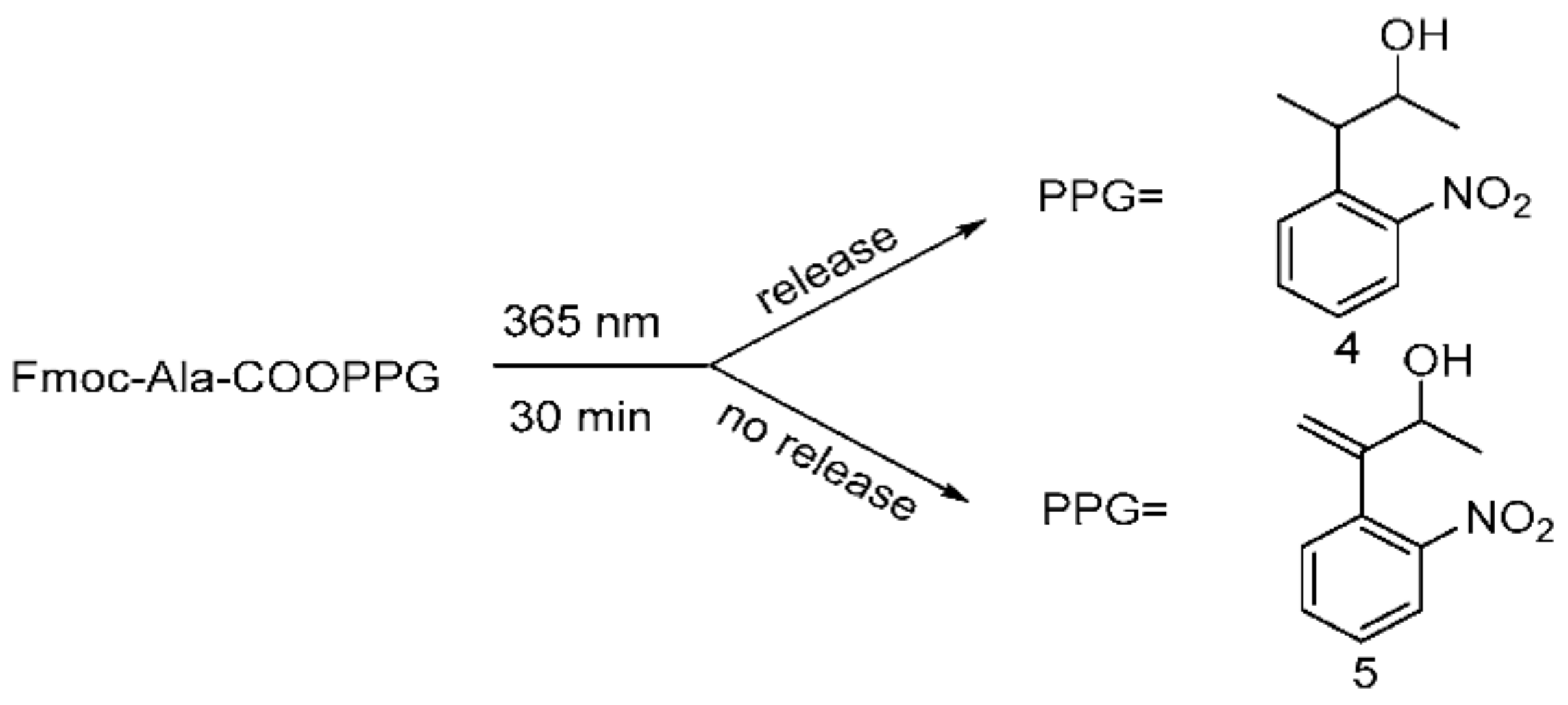
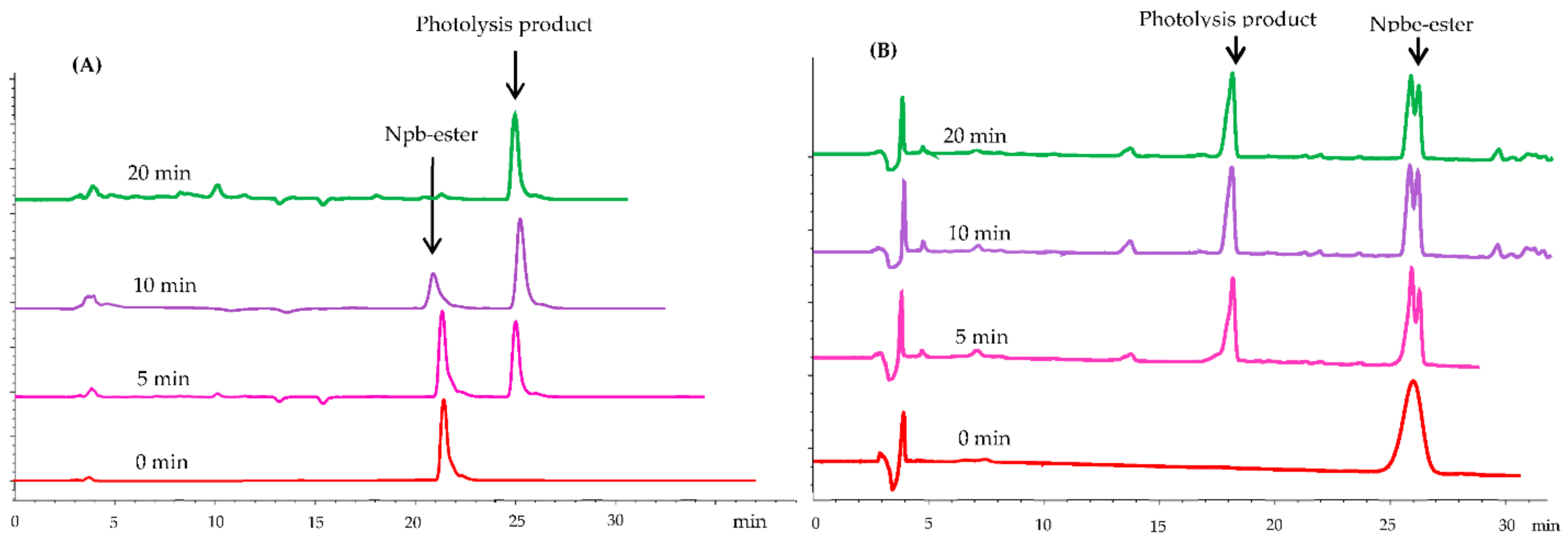
2.2.2. Photolysis of Esterified Compounds (4) and (5)
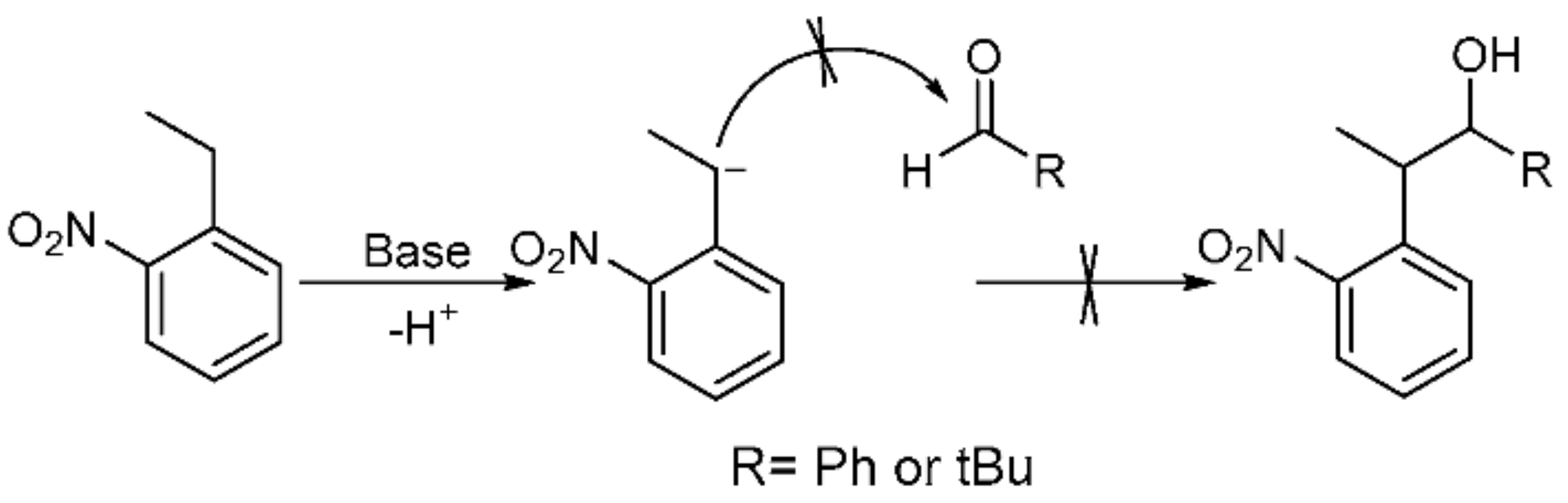
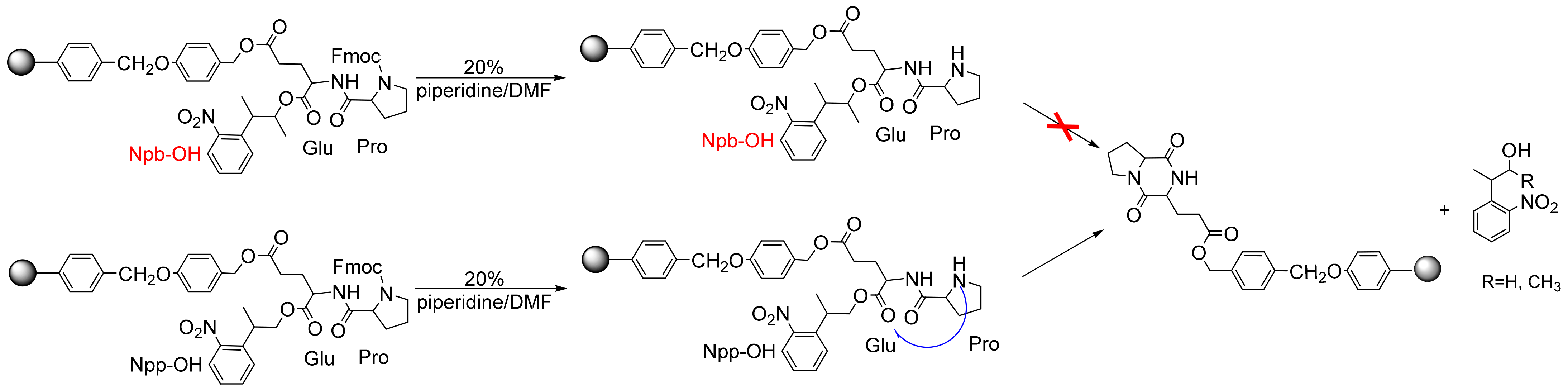
2.2.3. DKP Side Reactions
2.2.4. Applicability of PPG 4
2.3. Application of PPGs in Cyclic Peptide Synthesis
3. Materials and Methods
3.1. Synthesis of the Photolabile Protecting Groups (PPGs)
General Reduction Procedure
3.2. Photolysis of Esterified Compounds (4) and (5)
3.3. Applicability of PPG 4
3.3.1. General Esterification Procedure
3.3.2. General TFA Deprotection Procedure
3.4. Application of PPGs in Cyclic Peptide Synthesis
3.4.1. General Procedure for Solid-Phase Reactions
3.4.2. Removal of the Npb Group via Photolysis Experiment
3.4.3. Cyclization of Peptides I, II, and III
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nielsen, D.S.; Shepherd, N.E.; Xu, W.; Lucke, A.J.; Stoermer, M.J.; Fairlie, D.P. Orally Absorbed Cyclic Peptides. Chem. Rev. 2017, 117, 8094–8128. [Google Scholar] [CrossRef]
- Zhang, R.Y.; Thapa, P.; Espiritu, M.J.; Menon, V.; Bingham, J.P. From Nature to Creation: Going around in Circles, the Art of Peptide Cyclization. Bioorg. Med. Chem. 2018, 26, 1135–1150. [Google Scholar] [CrossRef] [PubMed]
- Yap, K.; Du, J.; Looi, F.Y.; Tang, S.R.; De Veer, S.J.; Bony, A.R.; Rehm, F.B.H.; Xie, J.; Chan, L.Y.; Wang, C.K.; et al. An Environmentally Sustainable Biomimetic Production of Cyclic Disulfide-Rich Peptides. Green Chem. 2020, 22, 5002–5016. [Google Scholar] [CrossRef]
- Al Musaimi, O.; De La Torre, B.G.; Albericio, F. Greening Fmoc/: T Bu Solid-Phase Peptide Synthesis. Green Chem. 2020, 22, 996–1018. [Google Scholar] [CrossRef]
- Wang, G.; Peng, T.; Zhang, S.; Wang, J.; Wen, X.; Yan, H.; Hu, L.; Wang, L. 2-(2-Nitrophenyl) Propyl: A Rapidly Released Photolabile COOH-Protecting Group for Solid-Phase Peptide Synthesis. RSC Adv. 2015, 5, 28344–28348. [Google Scholar] [CrossRef]
- Kaplan, J.H.; Forbush, B.; Hoffman, J.F. Rapid Photolytic Release of Adenosine 5′-Triphosphate from a Protected Analogue: Utilization by the Na:K Pump of Human Red Blood Cell Ghosts. Biochemistry 1978, 17, 1929–1935. [Google Scholar] [CrossRef]
- Engels, J.; Schlaeger, E.J. Synthesis, Structure, and Reactivity of Adenosine Cyclic 3′,5′-Phosphate-Benzyl Triesters. J. Med. Chem. 1977, 20, 907–911. [Google Scholar] [CrossRef]
- Klán, P.; Šolomek, T.; Bochet, C.G.; Blanc, A.; Givens, R.; Rubina, M.; Popik, V.; Kostikov, A.; Wirz, J. Photoremovable Protecting Groups in Chemistry and Biology: Reaction Mechanisms and Efficacy. Chem. Rev. 2013, 113, 119–191. [Google Scholar] [CrossRef]
- Bochet, C.G. Photolabile Protecting Groups and Linkers. J. Chem. Soc. Perkin Trans. 1 2002, 1, 125–142. [Google Scholar] [CrossRef]
- Ohtsuka, E.; Tanaka, S.; Ikehara, M. Studies on Transfer Ribonucleic Acids and Related Compounds. IX(1) Ribo-Oligonucleotide Synthesis Using a Photosensitive O-Nitrobenzyl Protection at the 2′-Hydroxyl Group. Nucleic Acids Res. 1974, 1, 1351–1358. [Google Scholar] [CrossRef]
- Zehavi, U.; Amit, B.; Patchornik, A. Light-Sensitive Glycosides. I. 6-Nitroveratryl β-D-Glucopyranoside and 2-Nitrobenzyl β-D-Glucopyranoside. J. Org. Chem. 1972, 37, 2281–2285. [Google Scholar] [CrossRef]
- Barltrop, J.A.; Schofield, P. Photosensitive Protecting Groups. Tetrahedron Lett. 1962, 3, 697–699. [Google Scholar] [CrossRef]
- Rich, D.H.; Gurwara, S.K. Preparation of a New O-Nitrobenzyl Resin for Solid-Phase Synthesis of Tert-Butyloxycarbonyl-Protected Peptide Acids. J. Am. Chem. Soc. 1975, 97, 1575–1579. [Google Scholar] [CrossRef]
- Lloyd-Williams, P.; Albericio, F.; Giralt, E. Convergent Solid-Phase Peptide Synthesis. Tetrahedron 1993, 49, 11065. [Google Scholar] [CrossRef]
- HAMMER, R.P.; ALBERICIO, F.; GERA, L.; BARANY, G. Practical Approach to Solid-phase Synthesis of C-terminal Peptide Amides under Mild Conditions Based on a Photolysable Anchoring Linkage. Int. J. Pept. Protein Res. 1990, 36, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Wöll, D.; Smirnova, J.; Galetskaya, M.; Prykota, T.; Bühler, J.; Stengele, K.P.; Pfleiderer, W.; Steiner, U.E. Intramolecular Sensitization of Photocleavage of the Photolabile 2-(2-Nitrophenyl)Propoxycarbonyl (NPPOC) Protecting Group: Photoproducts and Photokinetics of the Release of Nucleosides. Chem. A Eur. J. 2008, 14, 6490–6497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, P.; Lu, W.; Devalankar, D.A.; Ding, Z. Structurally Simple Benzyl-Type Photolabile Protecting Groups for Direct Release of Alcohols and Carboxylic Acids. Org. Lett. 2015, 17, 2114–2117. [Google Scholar] [CrossRef]
- Kretschy, N.; Holik, A.K.; Somoza, V.; Stengele, K.P.; Somoza, M.M. Next-Generation o-Nitrobenzyl Photolabile Groups for Light-Directed Chemistry and Microarray Synthesis. Angew. Chem.-Int. Ed. 2015, 54, 8555–8559. [Google Scholar] [CrossRef] [Green Version]
- Bühler, S.; Lagoja, I.; Giegrich, H.; Stengele, K.P.; Pfleiderer, W. New Types of Very Efficient Photolabile Protecting Groups Based upon the [2-(2-Nitrophenyl)Propoxy]Carbonyl (NPPOC) Moiety. Helv. Chim. Acta 2004, 87, 620–659. [Google Scholar] [CrossRef]
- Hoffmann, J.; Kazmaier, U. Development of a New NPPOC-Derived Photolabile Protecting Group Suitable for Cyclizations via Ring Closing Metathesis. Curr. Org. Synth. 2015, 12, 475–483. [Google Scholar] [CrossRef]
- Hasan, A.; Stengele, K.P.; Giegrich, H.; Cornwell, P.; Isham, K.R.; Sachleben, R.A.; Pfleiderer, W.; Foote, R.S. Photolabile Protecting Groups for Nucleosides: Synthesis and Photodeprotection Rates. Tetrahedron 1997, 53, 4247–4264. [Google Scholar] [CrossRef] [Green Version]
- Goodman, B.M.; Stueben, K.C. Peptide Synthesis via Amino Acid Active Esters. II.1 Some Abnormal Reactions during Peptide Synthesis. J. Am. Chem. Soc. 1962, 84, 1279–1283. [Google Scholar] [CrossRef]
- Yang, H.; Peng, T.; Wen, X.; Chen, T.; Sun, Y.; Liu, S.; Wang, G.; Zhang, S.; Wang, L. A Photolabile Carboxyl Protecting Group for Solid Phase Peptide Synthesis. ChemistryOpen 2021, 10, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Okuro, K.; Gurnham, J.; Alper, H. Ionic Diamine Rhodium Complex Catalyzed Reductive N-Heterocyclization of 2-Nitrovinylarenes. J. Org. Chem. 2011, 76, 4715–4720. [Google Scholar] [CrossRef] [PubMed]
- Straub, A.; Greul, J.N.; Lui, N. Method for Producing Alkenylnitrobenzene Derivatives Unbranched in the 1′-Positon 2010, A1. Available online: https://patentscope2.wipo.int/search/en/detail.jsf?docId=WO2008006576 (accessed on 14 January 2022).
- Gall1, C. Evidence for a Non-Chain SRN1 Reaction Occurring on a Nitroarylhalide. Tetrahedron 1988, 44, 5205–5208. [Google Scholar] [CrossRef]
- Iwama, T.; Birman, V.B.; Kozmin, S.A.; Rawal, V.H. Regiocontrolled Synthesis of Carbocycle-Fused Indoles via Arylation of Siyl Enol Ethers with o-Nitrophenylphenyliodonium Fluoride. Org. Lett. 1999, 1, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.S.; Gu, B.Q.; Zhu, M.Y.; Yu, X.; Deng, W.P. Nonenzymatic Kinetic Resolution of α-Aryl Substituted Allylic Alcohols Catalyzed by Acyl Transfer Catalyst Np-PIQ. Tetrahedron 2015, 71, 1187–1191. [Google Scholar] [CrossRef]
- Gisin, B.F.; Merrifield, R.B. Carboxyl-Catalyzed Intramolecular Aminolysis. A Side Reaction in Solid-Phase Peptide Synthesis. J. Am. Chem. Soc. 1972, 94, 3102–3106. [Google Scholar] [CrossRef]
- Barany, G.; Albericio, F. Three-Dimensional Orthogonal Protection Scheme for Solid-Phase Peptide Synthesis under Mild Conditions. J. Am. Chem. Soc. 1985, 107, 4936–4942. [Google Scholar] [CrossRef]
- Pedroso, E.; Grandas, A.; de las Heras, X.; Eritja, R.; Giralt, E. Diketopiperazine Formation in Solid Phase Peptide Synthesis Using P-Alkoxybenzyl Ester Resins and Fmoc-Amino Acids. Tetrahedron Lett. 1986, 27, 743–746. [Google Scholar] [CrossRef]
- Chiva, C.; Vilaseca, M.; Giralt, E.; Albericio, F. An HPLC-ESMS Study on the Solid-Phase Assembly of C-Terminal Proline Peptides. J. Pept. Sci. 1999, 5, 131–140. [Google Scholar] [CrossRef]
- Moronta, J.; Smaldini, P.L.; Docena, G.H.; Añón, M.C. Peptides of Amaranth Were Targeted as Containing Sequences with Potential Anti-Inflammatory Properties. J. Funct. Foods 2016, 21, 463–473. [Google Scholar] [CrossRef]
- Del Carmen Millán-Linares, M.; Millán, F.; Pedroche, J.; del Mar Yust, M. GPETAFLR: A New Anti-Inflammatory Peptide from Lupinus Angustifolius L. Protein Hydrolysate. J. Funct. Foods 2015, 18, 358–367. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.; Nishizawa, M.; Shimizu, Y.; Saeki, H. Anti-Inflammatory Effects of Dulse (Palmaria palmata) Resulting from the Simultaneous Water-Extraction of Phycobiliproteins and Chlorophyll a. Food Res. Int. 2017, 100, 514–521. [Google Scholar] [CrossRef]
- Ruhland, B.; Leclerc, G. Synthesis of 1-Hydroxy-2H,5H-Dihydroisoxazolo[5,4-c]Quinoline. A Novel Heterocyclic Ring System. J. Heterocycl. Chem 1989, 26, 469–471. [Google Scholar] [CrossRef]
- Nykaza, T.v.; Li, G.; Yang, J.; Luzung, M.R.; Radosevich, A.T. P III /P V =O Catalyzed Cascade Synthesis of N-Functionalized Azaheterocycles. Angew. Chem. 2020, 132, 4535–4540. [Google Scholar] [CrossRef]
- Gao, D.M.; Ma, W.L.; Li, T.R.; Huang, L.Z.; Du, Z.T. An Improved Synthesis of 1,2-Diarylethanols under Conventional Heating and Ultrasound Irradiation. Molecules 2012, 17, 10708–10715. [Google Scholar] [CrossRef] [Green Version]
- Coffman, K.C.; Palazzo, T.A.; Hartley, T.P.; Fettinger, J.C.; Tantillo, D.J.; Kurth, M.J. Heterocycle-Heterocycle Strategies: (2-Nitrophenyl)Isoxazole Precursors to 4-Aminoquinolines, 1 H-Indoles, and Quinolin-4(1 H)-Ones. Org. Lett. 2013, 15, 2062–2065. [Google Scholar] [CrossRef] [Green Version]





| Compound | TFA | 20% Piperidine/DMF |
|---|---|---|
| 4a | + a | + |
| 12a | − b | + |
| 13a | − | + |
| 16a | − | + |
| 17a | − | + |
| 5a | + | + |
 | |||
 |  |  | |
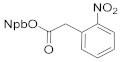
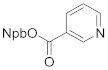
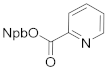
| 4b 83% | 4ca 72% | 4da 91% | |
 |  |  |  |
| 4e 80% | 4f 94% | 4g 90% | 4h 93% |
| Peptide | Sequences | Observed Mass (Da) [M+H]+ | Theoretical Mass (Da) [M+H]+ |
|---|---|---|---|
| I | c (SSEDIKE) | 789.361 | 788.836 |
| II | c (GPE) TAFLR | 872.496 | 872.134 |
| III | c (LRDGE) IILRY | 1228.726 | 1228.693 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, T.; Wang, G.; Tang, L.; Yang, H.; Xu, J.; Wen, X.; Sun, Y.; Liu, S.; Peng, T.; Zhang, S.; et al. Synthesis of Cyclic Peptides in SPPS with Npb-OH Photolabile Protecting Group. Molecules 2022, 27, 2231. https://doi.org/10.3390/molecules27072231
Chen T, Wang G, Tang L, Yang H, Xu J, Wen X, Sun Y, Liu S, Peng T, Zhang S, et al. Synthesis of Cyclic Peptides in SPPS with Npb-OH Photolabile Protecting Group. Molecules. 2022; 27(7):2231. https://doi.org/10.3390/molecules27072231
Chicago/Turabian StyleChen, Tingting, Gang Wang, Lin Tang, Hongpeng Yang, Jing Xu, Xiaoxue Wen, Yunbo Sun, Shuchen Liu, Tao Peng, Shouguo Zhang, and et al. 2022. "Synthesis of Cyclic Peptides in SPPS with Npb-OH Photolabile Protecting Group" Molecules 27, no. 7: 2231. https://doi.org/10.3390/molecules27072231






