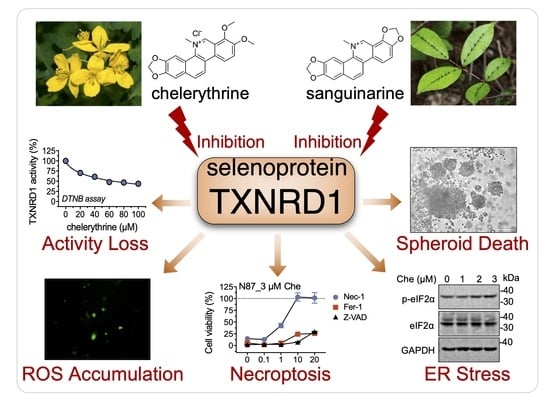
Benzophenanthridine Alkaloid Chelerythrine Elicits Necroptosis of Gastric Cancer Cells via Selective Conjugation at the Redox Hyperreactive C-Terminal Sec498 Residue of Cytosolic Selenoprotein Thioredoxin Reductase
Abstract
:1. Introduction
2. Results
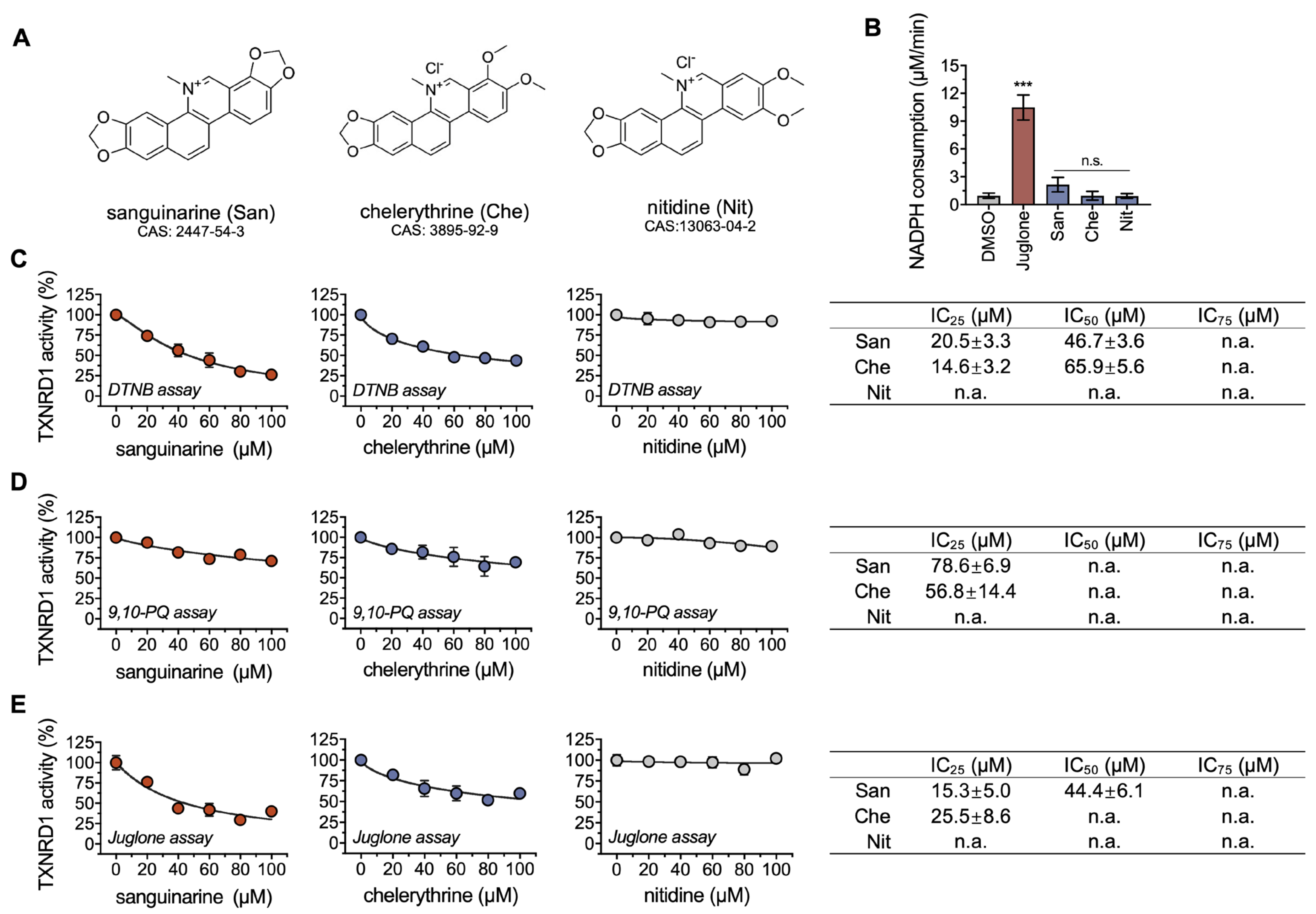
2.1. Chelerythrine and Sanguinarine Are Inhibitors of TXNRD1
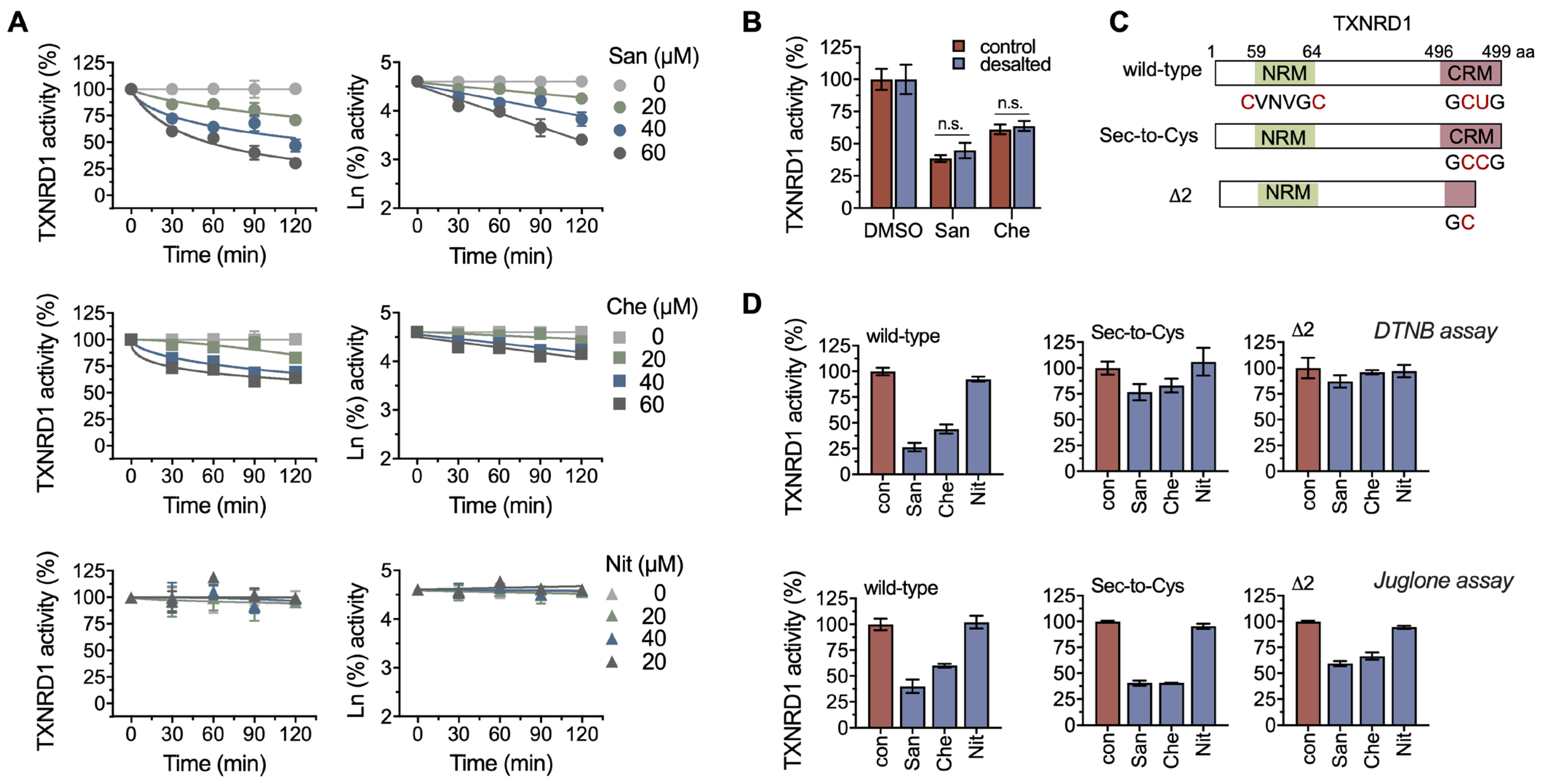
2.2. Chelerythrine and Sanguinarine Primarily Target the Hyperreactive Selenocysteine Residues
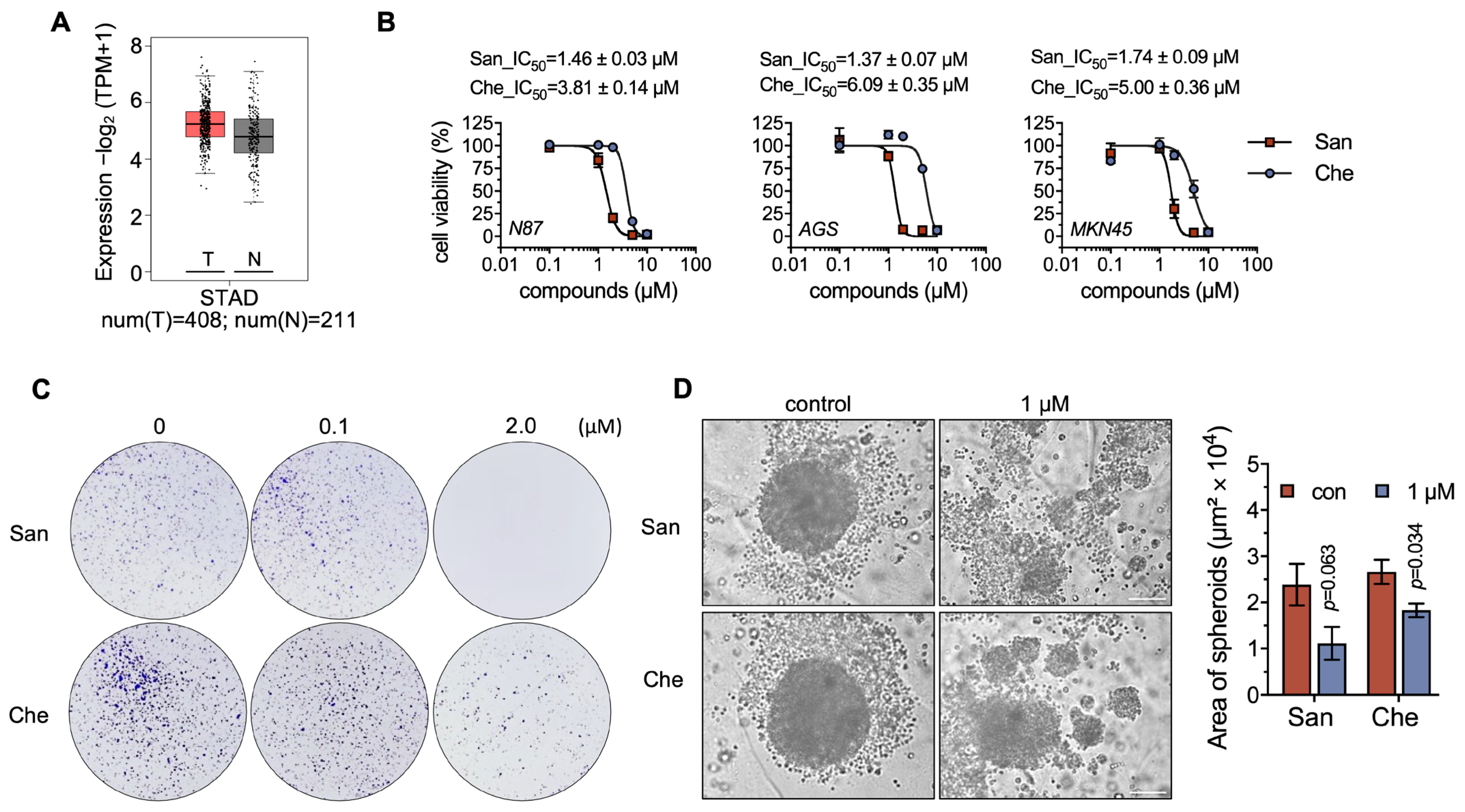
2.3. Chelerythrine and Sanguinarine Suppress the Proliferation of Gastric Cancer Cells
2.4. Chelerythrine and Sanguinarine Induce Oxidative Stress in Gastric Cancer Cells
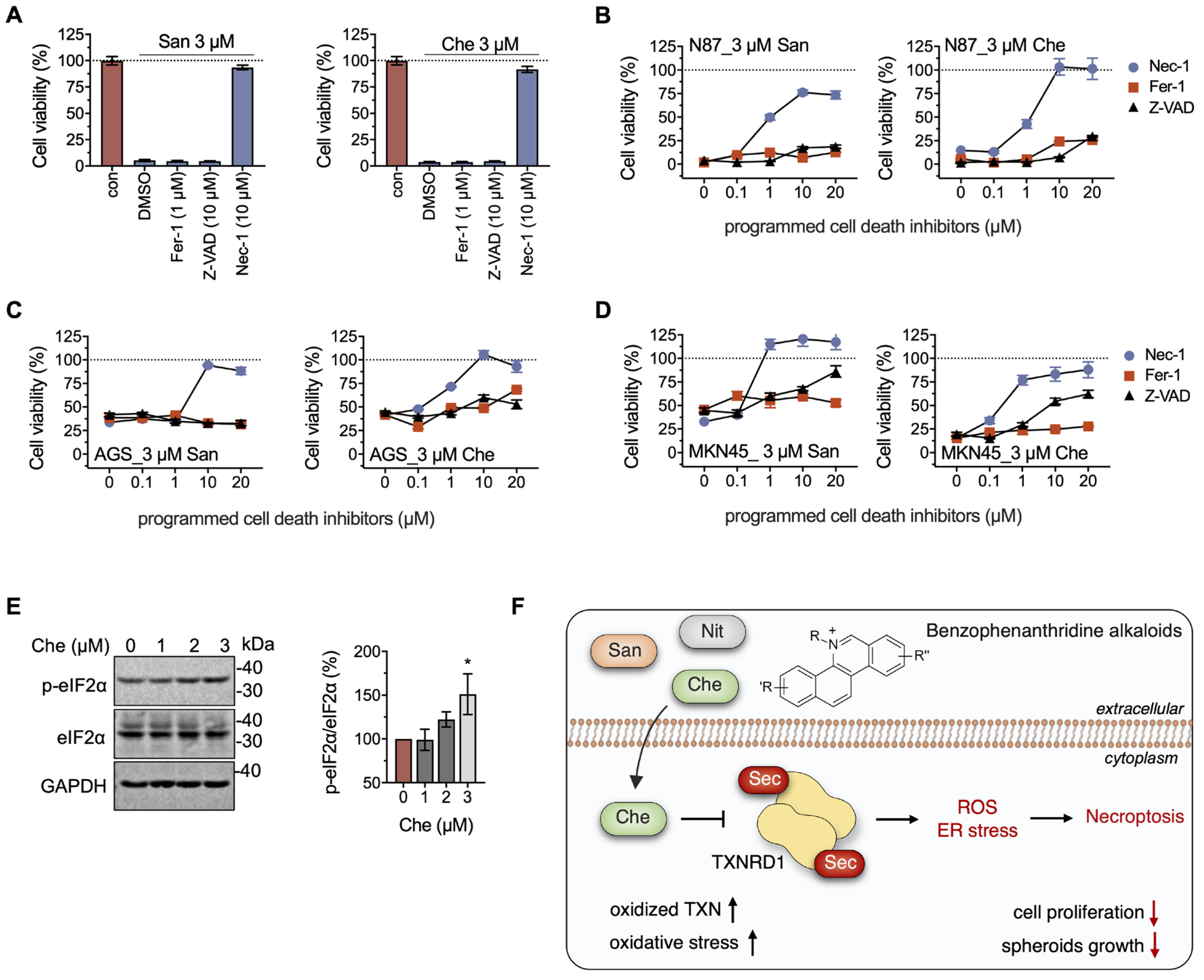
2.5. Chelerythrine and Sanguinarine Promote Necroptosis in Gastric Cancer Cells
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Cancer Cell Culture
4.3. Cell Viability
4.4. Cell Proliferation
4.5. Spheroid Formation
4.6. Western Blotting
4.7. Cellular TXNRD Activity
4.8. Inhibition of Compounds on Recombinant TXNRD1
4.9. ROS Detection
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Bjorklund, G.; Zou, L.; Wang, J.; Chasapis, C.T.; Peana, M. Thioredoxin Reductase as a Pharmacological Target. Pharmacol. Res. 2021, 174, 105854. [Google Scholar] [CrossRef] [PubMed]
- Arnér, E.S.J. Focus on mammalian thioredoxin reductases--important selenoproteins with versatile functions. Biochim. Biophys. Acta 2009, 1790, 495–526. [Google Scholar] [CrossRef] [PubMed]
- Delgobo, M.; Goncalves, R.M.; Delazeri, M.A.; Falchetti, M.; Zandona, A.; Nascimento das Neves, R.; Almeida, K.; Fagundes, A.C.; Gelain, D.P.; Fracasso, J.I.; et al. Thioredoxin reductase-1 levels are associated with NRF2 pathway activation and tumor recurrence in non-small cell lung cancer. Free Radic. Biol. Med. 2021, 177, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Dagnell, M.; Schmidt, E.E.; Arnér, E.S.J. The A to Z of modulated cell patterning by mammalian thioredoxin reductases. Free Radic. Biol. Med. 2018, 115, 484–496. [Google Scholar] [CrossRef]
- Prigge, J.R.; Coppo, L.; Martin, S.S.; Ogata, F.; Miller, C.G.; Bruschwein, M.D.; Orlicky, D.J.; Shearn, C.T.; Kundert, J.A.; Lytchier, J.; et al. Hepatocyte Hyperproliferation upon Liver-Specific Co-disruption of Thioredoxin-1, Thioredoxin Reductase-1, and Glutathione Reductase. Cell Rep. 2017, 19, 2771–2781. [Google Scholar] [CrossRef] [PubMed]
- McLoughlin, M.R.; Orlicky, D.J.; Prigge, J.R.; Krishna, P.; Talago, E.A.; Cavigli, I.R.; Eriksson, S.; Miller, C.G.; Kundert, J.A.; Sayin, V.I.; et al. TrxR1, Gsr, and oxidative stress determine hepatocellular carcinoma malignancy. Proc. Natl. Acad. Sci. USA 2019, 116, 11408–11417. [Google Scholar] [CrossRef] [PubMed]
- Harris, I.S.; Treloar, A.E.; Inoue, S.; Sasaki, M.; Gorrini, C.; Lee, K.C.; Yung, K.Y.; Brenner, D.; Knobbe-Thomsen, C.B.; Cox, M.A.; et al. Glutathione and thioredoxin antioxidant pathways synergize to drive cancer initiation and progression. Cancer Cell 2015, 27, 211–222. [Google Scholar] [CrossRef]
- Bansal, A.; Simon, M.C. Glutathione metabolism in cancer progression and treatment resistance. J. Cell Biol. 2018, 217, 2291–2298. [Google Scholar] [CrossRef]
- Lu, J.; Holmgren, A. The thioredoxin antioxidant system. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef]
- Lennicke, C.; Cocheme, H.M. Redox metabolism: ROS as specific molecular regulators of cell signaling and function. Mol. Cell 2021, 81, 3691–3707. [Google Scholar] [CrossRef]
- Cheung, E.C.; Vousden, K.H. The role of ROS in tumour development and progression. Nat. Rev. Cancer 2022, 22, 280–297. [Google Scholar] [CrossRef]
- Torrente, L.; DeNicola, G.M. Targeting NRF2 and Its Downstream Processes: Opportunities and Challenges. Annu. Rev. Pharmacol. Toxicol. 2022, 62, 279–300. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X.; Zhao, Z.; Cai, W.; Fang, J. Thioredoxin signaling pathways in cancer. Antioxid. Redox Signal. 2022, 38, 403–424. [Google Scholar] [CrossRef] [PubMed]
- Karunanithi, S.; Liu, R.; Hou, Y.; Gonzalez, G.; Oldford, N.; Roe, A.J.; Idipilly, N.; Gupta, K.; Amara, C.S.; Putluri, S.; et al. Thioredoxin reductase is a major regulator of metabolism in leukemia cells. Oncogene 2021, 40, 5236–5246. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Duan, D.; Osama, A.; Fang, J. Natural Molecules Targeting Thioredoxin System and Their Therapeutic Potential. Antioxid. Redox Signal. 2021, 34, 1083–1107. [Google Scholar] [CrossRef]
- Pickering, I.J.; Cheng, Q.; Rengifo, E.M.; Nehzati, S.; Dolgova, N.V.; Kroll, T.; Sokaras, D.; George, G.N.; Arner, E.S.J. Direct Observation of Methylmercury and Auranofin Binding to Selenocysteine in Thioredoxin Reductase. Inorg. Chem. 2020, 59, 2711–2718. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Zhong, L.; Zhao, R.; Holmgren, A. Ebselen: A thioredoxin reductase-dependent catalyst for alpha-tocopherol quinone reduction. Toxicol. Appl. Pharmacol. 2005, 207, 103–109. [Google Scholar] [CrossRef]
- Yang, R.; Sun, S.; Guo, Y.; Meng, Y.; Liu, H.; Shi, M.; Guan, S.; Xu, J. Anti-Inflammatory Effect of Dimethyl Fumarate Associates with the Inhibition of Thioredoxin Reductase 1 in RAW 264.7 Cells. Molecules 2022, 28, 107. [Google Scholar] [CrossRef]
- Stafford, W.C.; Peng, X.; Olofsson, M.H.; Zhang, X.; Luci, D.K.; Lu, L.; Cheng, Q.; Tresaugues, L.; Dexheimer, T.S.; Coussens, N.P.; et al. Irreversible inhibition of cytosolic thioredoxin reductase 1 as a mechanistic basis for anticancer therapy. Sci. Transl. Med. 2018, 10, eaaf7444. [Google Scholar] [CrossRef]
- Duan, D.; Zhang, J.; Yao, J.; Liu, Y.; Fang, J. Targeting Thioredoxin Reductase by Parthenolide Contributes to Inducing Apoptosis of HeLa Cells. J. Biol. Chem. 2016, 291, 10021–10031. [Google Scholar] [CrossRef]
- Yang, Y.; Sun, S.; Xu, W.; Zhang, Y.; Yang, R.; Ma, K.; Zhang, J.; Xu, J. Piperlongumine Inhibits Thioredoxin Reductase 1 by Targeting Selenocysteine Residues and Sensitizes Cancer Cells to Erastin. Antioxidants 2022, 11, 710. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, S.; Xu, W.; Yang, R.; Yang, Y.; Guo, J.; Ma, K.; Xu, J. Thioredoxin reductase 1 inhibitor shikonin promotes cell necroptosis via SecTRAPs generation and oxygen-coupled redox cycling. Free Radic. Biol. Med. 2022, 180, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Hu, Y.; Zhu, X.; Zhang, J.; Yuwen, D.; Wei, X.; Tang, C.; Zhang, W. Plasma thioredoxin reductase: A potential diagnostic biomarker for gastric cancer. Carcinogenesis 2022, 43, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.; Wang, H.; Wu, X.; He, L.; Zhou, Q.; Wang, F.; Chen, S.; Huang, L.; Chen, J.; Wang, H.; et al. ROS-mediated inactivation of the PI3K/AKT pathway is involved in the antigastric cancer effects of thioredoxin reductase-1 inhibitor chaetocin. Cell Death Dis. 2019, 10, 809. [Google Scholar] [CrossRef]
- Zou, P.; Xia, Y.; Ji, J.; Chen, W.; Zhang, J.; Chen, X.; Rajamanickam, V.; Chen, G.; Wang, Z.; Chen, L.; et al. Piperlongumine as a direct TrxR1 inhibitor with suppressive activity against gastric cancer. Cancer Lett. 2016, 375, 114–126. [Google Scholar] [CrossRef]
- Yao, J.; Duan, D.; Song, Z.L.; Zhang, J.; Fang, J. Sanguinarine as a new chemical entity of thioredoxin reductase inhibitor to elicit oxidative stress and promote tumor cell apoptosis. Free Radic. Biol. Med. 2020, 152, 659–667. [Google Scholar] [CrossRef]
- Zhu, M.; Gong, Z.; Wu, Q.; Shi, X.; Su, Q.; Zhang, Y. Sanguinarine suppresses migration and metastasis in colorectal carcinoma associated with the inversion of EMT through the Wnt/beta-catenin signaling. Clin. Transl. Med. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Su, Q.; Fan, M.; Wang, J.; Ullah, A.; Ghauri, M.A.; Dai, B.; Zhan, Y.; Zhang, D.; Zhang, Y. Sanguinarine inhibits epithelial-mesenchymal transition via targeting HIF-1alpha/TGF-beta feed-forward loop in hepatocellular carcinoma. Cell Death Dis. 2019, 10, 939. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, X.; Deng, J.; Zheng, H.; Liu, W.; Chen, S.; Tian, J.; Wang, F. p53-dependent upregulation of miR-16-2 by sanguinarine induces cell cycle arrest and apoptosis in hepatocellular carcinoma. Cancer Lett. 2019, 459, 50–58. [Google Scholar] [CrossRef]
- Cho, O.; Lee, J.W.; Kim, H.S.; Jeong, Y.J.; Heo, T.H. Chelerythrine, a novel small molecule targeting IL-2, inhibits melanoma progression by blocking the interaction between IL-2 and its receptor. Life Sci. 2023, 320, 121559. [Google Scholar] [CrossRef]
- Hong, Y.; Xu, W.Q.; Feng, J.; Lou, H.; Liu, H.; Wang, L.; Cui, H.; Jiang, L.T.; Xu, R.C.; Xu, H.H.; et al. Nitidine chloride induces cardiac hypertrophy in mice by targeting autophagy-related 4B cysteine peptidase. Acta Pharmacol. Sin. 2023, 44, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Plazas, E.; Avila, M.M.; Munoz, D.R.; Cuca, S.L. Natural isoquinoline alkaloids: Pharmacological features and multi-target potential for complex diseases. Pharmacol. Res. 2022, 177, 106126. [Google Scholar] [CrossRef]
- Sun, S.; Zhang, Y.; Xu, W.; Yang, R.; Yang, Y.; Guo, J.; Ma, Q.; Ma, K.; Zhang, J.; Xu, J. Plumbagin reduction by thioredoxin reductase 1 possesses synergy effects with GLUT1 inhibitor on KEAP1-mutant NSCLC cells. Biomed. Pharmacother. 2022, 146, 112546. [Google Scholar] [CrossRef] [PubMed]
- Walterova, D.; Ulrichova; Preininger, V.; Simanek, V.; Lenfeld, J.; Lasovsky, J. Inhibition of liver alanine aminotransferase activity by some benzophenanthridine alkaloids. J. Med. Chem. 1981, 24, 1100–1103. [Google Scholar] [CrossRef] [PubMed]
- Belyaeva, T.; Leontieva, E.; Shpakov, A.; Mozhenok, T.; Faddejeva, M. Sensitivity of lysosomal enzymes to the plant alkaloid sanguinarine: Comparison with other SH-specific agents. Cell Biol. Int. 2003, 27, 887–895. [Google Scholar] [CrossRef]
- Liu, S.Y.; Stadtman, T.C. Heparin-binding properties of selenium-containing thioredoxin reductase from HeLa cells and human lung adenocarcinoma cells. Proc. Natl. Acad. Sci. USA 1997, 94, 6138–6141. [Google Scholar] [CrossRef]
- Lothrop, A.P.; Snider, G.W.; Ruggles, E.L.; Patel, A.S.; Lees, W.J.; Hondal, R.J. Selenium as an electron acceptor during the catalytic mechanism of thioredoxin reductase. Biochemistry 2014, 53, 654–663. [Google Scholar] [CrossRef]
- Fritz-Wolf, K.; Kehr, S.; Stumpf, M.; Rahlfs, S.; Becker, K. Crystal structure of the human thioredoxin reductase-thioredoxin complex. Nat. Commun. 2011, 2, 383. [Google Scholar] [CrossRef]
- Xu, J.; Cheng, Q.; Arner, E.S. Details in the catalytic mechanism of mammalian thioredoxin reductase 1 revealed using point mutations and juglone-coupled enzyme activities. Free Radic. Biol. Med. 2016, 94, 110–120. [Google Scholar] [CrossRef]
- Sun, S.; Xu, W.; Zhang, Y.; Yang, Y.; Ma, Q.; Xu, J. Menadione inhibits thioredoxin reductase 1 via arylation at the Sec498 residue and enhances both NADPH oxidation and superoxide production in Sec498 to Cys498 substitution. Free Radic. Biol. Med. 2021, 172, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Gencheva, R.; Arnér, E.S.J. Thioredoxin Reductase Inhibition for Cancer Therapy. Annu. Rev. Pharmacol. Toxicol. 2022, 62, 4.1–4.20. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Zhang, H.; Lu, J.; Holmgren, A. Glutathione and glutaredoxin act as a backup of human thioredoxin reductase 1 to reduce thioredoxin 1 preventing cell death by aurothioglucose. J. Biol. Chem. 2012, 287, 38210–38219. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Stockwell, B.R. The Hallmarks of Ferroptosis. Annu. Rev. Cancer Biol. 2019, 3, 35–54. [Google Scholar] [CrossRef]
- Stockwell, B.R. Ferroptosis turns 10: Emerging mechanisms, physiological functions, and therapeutic applications. Cell 2022, 185, 2401–2421. [Google Scholar] [CrossRef]
- Mishima, E.; Conrad, M. Nutritional and Metabolic Control of Ferroptosis. Annu. Rev. Nutr. 2022, 42, 275–309. [Google Scholar] [CrossRef]
- Saveljeva, S.; Mc Laughlin, S.L.; Vandenabeele, P.; Samali, A.; Bertrand, M.J. Endoplasmic reticulum stress induces ligand-independent TNFR1-mediated necroptosis in L929 cells. Cell Death Dis. 2015, 6, e1587. [Google Scholar] [CrossRef]
- Zhu, P.; Hu, S.; Jin, Q.; Li, D.; Tian, F.; Toan, S.; Li, Y.; Zhou, H.; Chen, Y. Ripk3 promotes ER stress-induced necroptosis in cardiac IR injury: A mechanism involving calcium overload/XO/ROS/mPTP pathway. Redox Biol. 2018, 16, 157–168. [Google Scholar] [CrossRef]
- Rellmann, Y.; Eidhof, E.; Dreier, R. Review: ER stress-induced cell death in osteoarthritic cartilage. Cell Signal 2021, 78, 109880. [Google Scholar] [CrossRef]
- Dixon, S.J.; Pratt, D.A. Ferroptosis: A flexible constellation of related biochemical mechanisms. Mol. Cell 2023, 83, 1030–1042. [Google Scholar] [CrossRef]
- Falchetti, M.; Delgobo, M.; Zancanaro, H.; Almeida, K.; das Neves, R.N.; dos Santos, B.; Stefanes, N.M.; Bishop, A.; Santos-Silva, M.C.; Zanotto-Filho, A. Omics-based identification of an NRF2-related auranofin resistance signature in cancer: Insights into drug repurposing. Comput. Biol. Med. 2022, 152, 106347. [Google Scholar] [CrossRef]
- Singh, A.; Daemen, A.; Nickles, D.; Jeon, S.M.; Foreman, O.; Sudini, K.; Gnad, F.; Lajoie, S.; Gour, N.; Mitzner, W.; et al. NRF2 Activation Promotes Aggressive Lung Cancer and Associates with Poor Clinical Outcomes. Clin. Cancer Res. 2021, 27, 877–888. [Google Scholar] [CrossRef] [PubMed]
- Binkley, M.S.; Jeon, Y.J.; Nesselbush, M.; Moding, E.J.; Nabet, B.Y.; Almanza, D.; Kunder, C.; Stehr, H.; Yoo, C.H.; Rhee, S.; et al. KEAP1/NFE2L2 Mutations Predict Lung Cancer Radiation Resistance That Can Be Targeted by Glutaminase Inhibition. Cancer Discov. 2020, 10, 1826–1841. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative Stress in Cancer. Cancer Cell 2020, 38, 167–197. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wang, H.; Yang, X.; Wu, Q.; An, P.; Jin, X.; Liu, W.; Huang, X.; Li, Y.; Yan, S.; et al. Auranofin mitigates systemic iron overload and induces ferroptosis via distinct mechanisms. Signal Transduct. Target. Ther. 2020, 5, 138. [Google Scholar] [CrossRef]
- Kelleher, Z.T.; Sha, Y.; Foster, M.W.; Foster, W.M.; Forrester, M.T.; Marshall, H.E. Thioredoxin-mediated denitrosylation regulates cytokine-induced nuclear factor kappaB (NF-kappaB) activation. J. Biol. Chem. 2014, 289, 3066–3072. [Google Scholar] [CrossRef]
- Zahedi Avval, F.; Holmgren, A. Molecular mechanisms of thioredoxin and glutaredoxin as hydrogen donors for Mammalian s phase ribonucleotide reductase. J. Biol. Chem. 2009, 284, 8233–8240. [Google Scholar] [CrossRef]
- Arnér, E.S.J. Effects of Mammalian Thioredoxin Reductase Inhibitors. Handb. Exp. Pharmacol. 2021, 264, 289–309. [Google Scholar] [CrossRef]
- Xu, J.; Eriksson, S.E.; Cebula, M.; Sandalova, T.; Hedstrom, E.; Pader, I.; Cheng, Q.; Myers, C.R.; Antholine, W.E.; Nagy, P.; et al. The conserved Trp114 residue of thioredoxin reductase 1 has a redox sensor-like function triggering oligomerization and crosslinking upon oxidative stress related to cell death. Cell Death Dis. 2015, 6, e1616. [Google Scholar] [CrossRef]
- Zhong, L.; Arner, E.S.; Holmgren, A. Structure and mechanism of mammalian thioredoxin reductase: The active site is a redox-active selenolthiol/selenenylsulfide formed from the conserved cysteine-selenocysteine sequence. Proc. Natl. Acad. Sci. USA 2000, 97, 5854–5859. [Google Scholar] [CrossRef]
- Duan, D.; Zhang, B.; Yao, J.; Liu, Y.; Sun, J.; Ge, C.; Peng, S.; Fang, J. Gambogic acid induces apoptosis in hepatocellular carcinoma SMMC-7721 cells by targeting cytosolic thioredoxin reductase. Free Radic. Biol. Med. 2014, 69, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Duan, D.; Zhang, B.; Yao, J.; Liu, Y.; Fang, J. Shikonin targets cytosolic thioredoxin reductase to induce ROS-mediated apoptosis in human promyelocytic leukemia HL-60 cells. Free Radic. Biol. Med. 2014, 70, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Duan, D.; Song, Z.L.; Liu, T.; Hou, Y.; Fang, J. Small molecules regulating reactive oxygen species homeostasis for cancer therapy. Med. Res. Rev. 2021, 41, 342–394. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Cao, D.; Cui, W.; Ji, M.; Qian, X.; Zhong, L. Molecular bases of thioredoxin and thioredoxin reductase-mediated prooxidant actions of (-)-epigallocatechin-3-gallate. Free Radic. Biol. Med. 2010, 49, 2010–2018. [Google Scholar] [CrossRef]
- Anestal, K.; Prast-Nielsen, S.; Cenas, N.; Arner, E.S. Cell death by SecTRAPs: Thioredoxin reductase as a prooxidant killer of cells. PLoS ONE 2008, 3, e1846. [Google Scholar] [CrossRef]
- Wang, P.; Zheng, S.Y.; Jiang, R.L.; Wu, H.D.; Li, Y.A.; Lu, J.L.; Xiong, Y.; Han, B.; Lin, L. Necroptosis signaling and mitochondrial dysfunction cross-talking facilitate cell death mediated by chelerythrine in glioma. Free Radic. Biol. Med. 2023, 202, 76–96. [Google Scholar] [CrossRef]
- Fennema, E.; Rivron, N.; Rouwkema, J.; van Blitterswijk, C.; de Boer, J. Spheroid culture as a tool for creating 3D complex tissues. Trends Biotechnol. 2013, 31, 108–115. [Google Scholar] [CrossRef]
- Ge, Q.; Shi, Z.; Zou, K.A.; Ying, J.; Chen, J.; Yuan, W.; Wang, W.; Xiao, L.; Lin, X.; Chen, D.; et al. Protein phosphatase PPM1A inhibition attenuates osteoarthritis via regulating TGF-beta/Smad2 signaling in chondrocytes. JCI Insight 2023, 8, e166688. [Google Scholar] [CrossRef]
- Wang, Z.; Lu, J.; Yuan, Z.; Pi, W.; Huang, X.; Lin, X.; Zhang, Y.; Lei, H.; Wang, P. Natural Carrier-Free Binary Small Molecule Self-Assembled Hydrogel Synergize Antibacterial Effects and Promote Wound Healing by Inhibiting Virulence Factors and Alleviating the Inflammatory Response. Small 2023, 19, e2205528. [Google Scholar] [CrossRef]
- Arnér, E.S.J.; Sarioglu, H.; Lottspeich, F.; Holmgren, A.; Bock, A. High-level expression in Escherichia coli of selenocysteine-containing rat thioredoxin reductase utilizing gene fusions with engineered bacterial-type SECIS elements and co-expression with the selA, selB and selC genes. J. Mol. Biol. 1999, 292, 1003–1016. [Google Scholar] [CrossRef]
- Arner, E.S.; Holmgren, A. Measurement of thioredoxin and thioredoxin reductase. Curr. Protoc. Toxicol. 2001, 24, 7.4.1–7.4.14. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, M.; Sun, S.; Meng, Y.; Wang, L.; Liu, H.; Shi, W.; Zhang, Q.; Xu, W.; Sun, B.; Xu, J. Benzophenanthridine Alkaloid Chelerythrine Elicits Necroptosis of Gastric Cancer Cells via Selective Conjugation at the Redox Hyperreactive C-Terminal Sec498 Residue of Cytosolic Selenoprotein Thioredoxin Reductase. Molecules 2023, 28, 6842. https://doi.org/10.3390/molecules28196842
Liu M, Sun S, Meng Y, Wang L, Liu H, Shi W, Zhang Q, Xu W, Sun B, Xu J. Benzophenanthridine Alkaloid Chelerythrine Elicits Necroptosis of Gastric Cancer Cells via Selective Conjugation at the Redox Hyperreactive C-Terminal Sec498 Residue of Cytosolic Selenoprotein Thioredoxin Reductase. Molecules. 2023; 28(19):6842. https://doi.org/10.3390/molecules28196842
Chicago/Turabian StyleLiu, Minghui, Shibo Sun, Yao Meng, Ling Wang, Haowen Liu, Wuyang Shi, Qiuyu Zhang, Weiping Xu, Bingbing Sun, and Jianqiang Xu. 2023. "Benzophenanthridine Alkaloid Chelerythrine Elicits Necroptosis of Gastric Cancer Cells via Selective Conjugation at the Redox Hyperreactive C-Terminal Sec498 Residue of Cytosolic Selenoprotein Thioredoxin Reductase" Molecules 28, no. 19: 6842. https://doi.org/10.3390/molecules28196842
APA StyleLiu, M., Sun, S., Meng, Y., Wang, L., Liu, H., Shi, W., Zhang, Q., Xu, W., Sun, B., & Xu, J. (2023). Benzophenanthridine Alkaloid Chelerythrine Elicits Necroptosis of Gastric Cancer Cells via Selective Conjugation at the Redox Hyperreactive C-Terminal Sec498 Residue of Cytosolic Selenoprotein Thioredoxin Reductase. Molecules, 28(19), 6842. https://doi.org/10.3390/molecules28196842