1. Introduction
A great number of biologically active molecules contain at least one or more amino functional groups. Biogenic amines are found in various organisms, cells, foods, and beverages and can be classified as monoamines, oligoamines, and polyamines depending on the number of amine functions they contain. They are typically synthesized by living organisms through enzymatic decarboxylation of amino acids [
1,
2,
3]. Biogenic aliphatic oligoamines such as spermine (
4), spermidine (
3), putrescine (
1), and cadaverine (
2) (
Figure 1) are among the most important ones, which can be found in almost all living organisms. They exist within cells in protonated forms and play important roles in diverse biological processes, such as cell growth, gene regulation, nucleic acid stabilization, and cell proliferation [
4,
5,
6].
In vertebrates, two oligoamines, spermidine (
3) and spermine (
4), are present along with their precursor, the diamine putrescine (
1). The absence of a normal oligoamine content can have a significant impact on cell functions including differentiation, apoptosis, motility, and resistance to oxidative and other stresses [
7,
8]. The importance of these oligoamines is shown not only in the effects of their absence but also in their extensive spectrum of applications. It has been shown that elevated spermine (
4) levels can be used to indicate the presence of cancer cells, making it possible to use spermine (
4) as a cancer biomarker [
9]. Furthermore, the high concentration of spermine (
4) and spermidine (
3) can also be connected to psoriasis, where patients have about 5.5 times the amount of these oligoamines than healthy people [
10]. On the other hand, concentrations below the normal amount can be linked to aging related illnesses, such as
Parkinson’s disease [
11]. Studies have also shown that spermidine (
3) holds promise for both diagnosis and treatment of ocular diseases [
12].
The relationship between oligoamines and both cancer and essential cellular processes has stimulated the development of methods for tracking oligoamine-mediated biological processes. Given that most tumors exhibit elevated activity in the oligoamine transport [
13], chemical probes designed to exploit this system could be used to target cancer cells for both imaging and chemotherapy [
14]. Typically, studying oligoamine transport in cells relies on the use of radiolabeled (
3H or
14C) or fluorescently labeled oligoamines. Recently, fluorescent oligoamine conjugates incorporating a symmetrical BODIPY fluorophore have been reported and used to monitor oligoamine uptake by cancer cells [
15]. The simple synthesis of these clickable oligoamine live-cell probes facilitates their commercial viability, making the tracking of oligoamine-mediated processes more accessible. The biological role of spermine (
4) and spermidine (
3) is still a dynamically researched area, with new discoveries being made even in recent years. It has been shown that spermidine (
3) plays a role in both aging and aging related illnesses [
16]. Spermidine (
3) level decreases with age, but supplementation can improve or postpone various age-related conditions, including those impacting the immune system, through directly affecting T cell function and ATP production. Due to these properties, it shows promising antitumor effects [
17]. In the case of spermine (
4), a study in 2024 suggested that the elevated levels of spermine (
4) in the brains of
Alzheimer’s disease patients indicate its involvement in the disease and highlight its potential as a therapeutic target [
18]. Along with their importance in human health, these oligoamines can also be used in the agricultural sector to enhance seed germination and seedling performance [
19].
Today, mostly traditional chromatographic methods are used for the identification and quantification of spermine (
4) and spermidine (
3) [
2]; among them, the most used techniques are capillary electrophoresis (CE) and high-performance liquid chromatography (HPLC) for their small sample requirement and relatively low detection limit [
20,
21,
22]. However, chromatographic methods require extensive sample preparation including solid-phase extraction and matrix solid-phase dispersion of the biogenic amines [
23]. Instrumental-based analytics also require the use of complex and expensive devices, making them less accessible and useful in some routine diagnostics. Chemosensors provide a fast and straightforward alternative for the detection of the biogenic amines, eliminating the need of specified instrumentation. They can be used to monitor biological samples, such as blood, saliva, and urine reducing the need for invasive procedures. Although detecting substances present in bio-fluids can be crucial for investigating various diseases and illnesses (such as predicting prostate cancer based on spermine (
4) levels in urine [
24]), this task is complicated by the presence of hundreds of molecules, often with similar properties, appearing in both trace and micromolar amounts [
25]. Investigating bio-fluids with such complex molecular compositions using classical methods poses a challenge, but chemosensors’ versatility enables selective examination of specific substances of interest without any need of pretreatment procedure. Furthermore, chemosensors provide real-time analysis enabling continuous monitoring of biological processes. They can also be used for bioimaging [
26], capturing cellular-level photographs of biological indicators, organs, tissues, etc.
These sensors are based on molecular recognition and generate a signal in the presence of an analyte [
25]. The molecule used as a chemosensor must have chromogenic or fluorogenic properties, which change upon the complexation of the analyte. This change in the optical properties enables a straightforward detection [
27,
28]. Most chemosensors use the “binding site-signaling site” approach, where separate units provide these properties. In our method, these properties are in the same place by incorporating the fluorophore unit as a part of the binding cavity. This fluorophore can alter its emission through a direct internal charge transfer (ICT) process upon complexation, offering numerous advantages over other methods, such as fast response, high sensitivity, and easy regulation [
29].
The research and development of chromogenic and fluorogenic chemosensors for detecting biologically active molecules have become an increasingly prominent area in recent years, especially in the cases of those lacking useful UV–vis spectral signatures. Several articles report on new chemosensors for detecting biogenic amines by utilizing the interactions of small molecular chromophores and fluorophores [
30,
31,
32,
33,
34], organic polymers [
35], coordination compounds [
36], and nanoparticles [
37]. Selective detection of aliphatic biogenic amines is complicated by their high structural similarity. In the case of protonated oligoamines, they contain several positive centers, which can serve as potential binding sites. Their aliphatic nature results in high conformational flexibility, making the increase in entropy difficult during coordination. Due to these difficulties, many chemosensors exhibit limited ability to distinguish between spermine (
4), spermidine (
3), cadaverine (
2), and putrescine (
1) [
33,
38]. To our knowledge, there is only one small fluorescent chemosensor molecule, which can selectively detect spermine (
4) with little to no response from other analogues [
34]. The use of small chemosensor molecules is advantageous, because their selectivity for specific analytes can easily be adjusted [
39]. They are generally inexpensive to produce, tolerant to varying conditions, while interactions in biological environments are minimal. Moreover, their small molecular size facilitates easy integration into samples.
Compounds developed in recent years and capable of chemosensor-based oligoamine analysis often include carboxyl groups, which interact with amines through hydrogen bonding or electrostatic interactions [
40]. Previously used compounds include carboxylic acid-functionalized poly(thiophenes), which employ a colorimetric method for the detection and identification of amines [
41]. Additionally, tetraphenylethenes with carboxyl groups have been used; these are aggregation-induced emission active molecules, which remain non-emissive in solution but exhibit intense fluorescence when aggregated or in the solid state. This is due to the restriction of intramolecular rotations, which prevents nonradiative deactivation and fluorescence quenching [
32,
33]. A dicarboxylated ethynylarene was also used for the detection of oligoamines by its carboxylic acid subunits, demonstrating a turn-on fluorescence signal [
39]. Other methods include the use of small-molecule amphiphiles and function by aggregation, such as fluorescent dyes like trisodium-8-hydroxypyrene-1,3,6-trisulfonate, which can be converted into a frustrated amphiphile upon alkylation [
34]. It was found by
Köstereli et al. that a turn-off response is demonstrated by this amphiphile in the presence of spermine (
4), even at nanomolar concentrations, making it an effective spermine-selective chemosensor. A similar method was developed by
Singh and coworkers using a perylenediimid derivative as the fluorescent dye, achieving a turn-on response [
42].
Macrocycles with an 18-crown-6 ether-type cavity are able to complex both protonated and neutral primary amines (with the complex stability being weaker in the case of neutral ones) by forming hydrogen bonds with the nucleophilic heteroatoms of the host [
43,
44]. Attributed to these properties, crown ethers have been found suitable for several protonated amine-selective applications, for example the separation of ammonium salts from byproducts [
45], the extraction of biogenic amines [
46], and the separation of both chiral and non-chiral amino acids [
47]. The achievements of crown ethers, in the selective separation of amine derivatives based on molecular recognition, suggest that these macrocycles could serve as effective chemosensors for the detection and selective separation of biogenic amines [
48]. The difference in the number of amine functions, and chain lengths of the oligoamines can be utilized, if crown ethers containing two macrocycle units are used, making a multiple-point interaction to take place. In such cases, aside from the hydrogen bonds formed by the two macrocycle rings, other interactions (for example cation
-π interaction) can also take place with the heterocyclic subunits, thereby enhancing the overall stability of the complex.
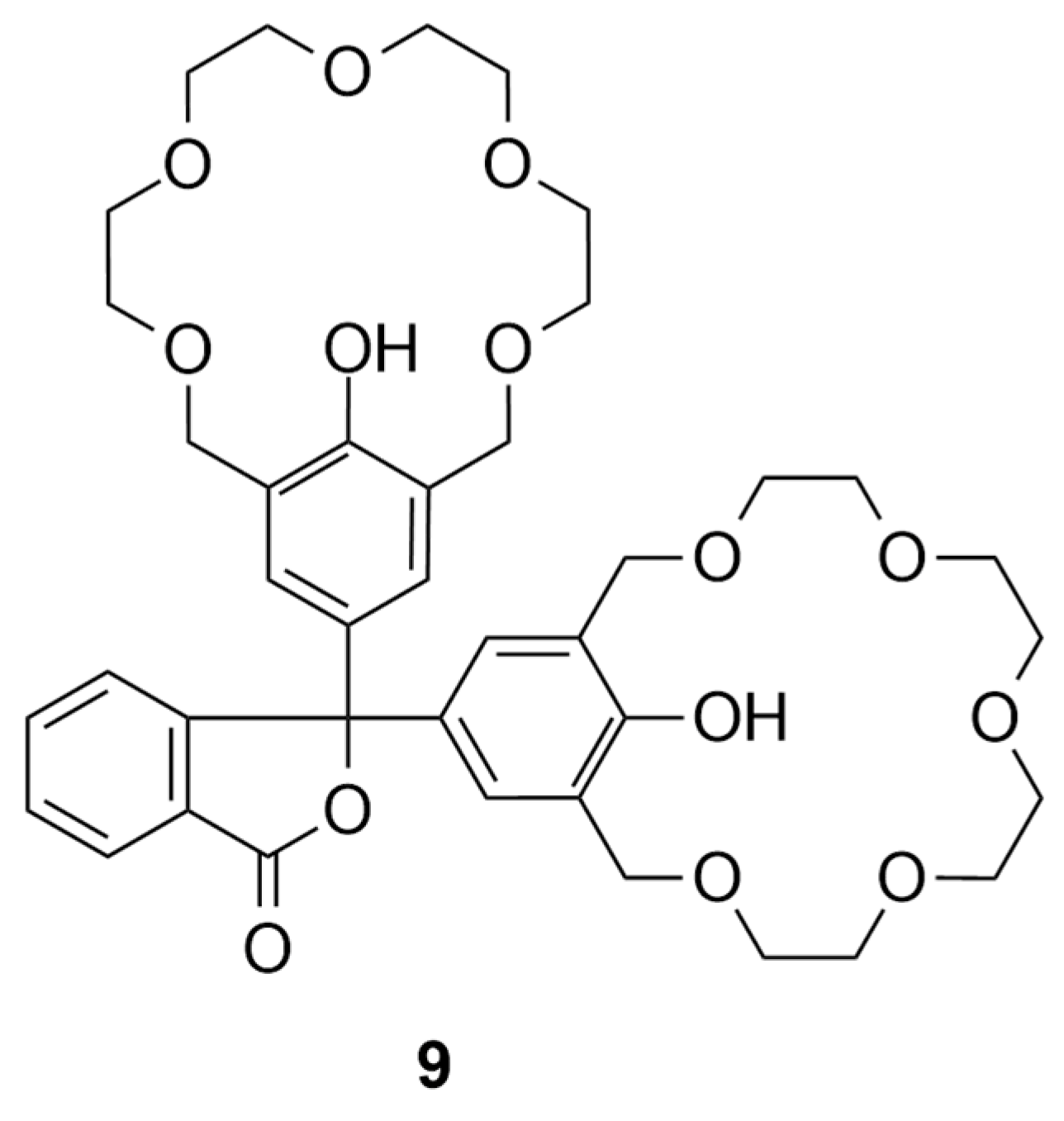
Such crown ether has been used before as a chemosensor for the detection of spermine analogues, based on colorimetry reported by
Tanima and coworkers [
30]. This chemosensor (
9, see
Figure 2) is the closest one, which we report here. To our knowledge, no other method utilizes molecules containing two crown ether units for this purpose.
A phenolphthalein derivative with two crown ether units (9) was used by Tanima and coworkers, which was able to bind spermidine (3) and spermine (4) by a three-podal interaction. With structural modifications of host molecule 9, the colorimetric response was also transformed into a fluorescent one. The host molecules demonstrated a turn-off response, which is less suitable for distinguishing between response quenching and background interference. In contrast, turn-on fluorescence response provides a clearer and more definitive signal. Great selectivity for the “naked eye” recognition of spermine (4) and spermidine (3) was achieved over the other biogenic amines, while the host remained inactive against tryptamine (10 eq.) and cadaverine (2) (10 eq.), although showing the color response after spermine (4) was added (<1 eq.) to these interfering amines. However, a clear distinction between spermine (4) and spermidine (3) was not achieved, as the binding constants (logK) were close. For spermidine (3), the logK values ranged from 2.9 to 3.2, and for spermine (4), they ranged from 3.4 to 3.6, depending on the host used.
2. Results and Discussion
2.1. Molecular Design
We designed bis(acridino)-crown ether
10 (
Figure 3), which functions as a sensitive chemosensor for protonated aliphatic biogenic oligoamines and can selectively differentiate between short- and long-chained ones.
Host molecule
10 has two 18-crown-6 ether-type cavities with sizes, which match those of the protonated primary amino groups (
r = 1.4 Å and
r = 1.42 Å, respectively) [
43,
44]. This allows stable coordination by hydrogen bonding between the electrophilic ammonium ion and nucleophilic heteroatoms of the host [
49]. The molecule exhibits fluorescence due to its acridine units. Due to electron conjugation, filling even one cavity induces a fluorescent response in these units. It shows a turn-on fluorescent response in contrast to the host molecules used by
Tanima and coworkers [
30]. Although both have their advantages, turn-on fluorescence responses are typically preferred to turn-off ones. They amplify the fluorescence signal significantly upon binding the analyte, thereby increasing the sensitivity to detect it. This amplification not only reduces the detectable lowest concentration of the analyte, but also improves the background suppression by maintaining minimal baseline fluorescence during interaction with the analyte, minimizing interference from other substances in the sample [
50].
Besides being a good fluorophore, the acridine unit provides structural rigidity due to its planar structure. With the two macrorings being connected through the acridine units, not only is the distance between the two macrocyclic rings optimal for the length of the spermine (
4) chain without causing significant structural distortion, but they also fixate both macrorings due to their planarity. In this arrangement, if the guest molecule contains a protonated interchain amine function, the acridine units within the host molecule can further enhance the formation of the host–guest complex by an additional aromatic
π-cation interaction. Furthermore, the nitrogen atom of the acridine unit shows a stronger affinity for hydrogen bonding compared to oxygen atoms, thereby enhancing selectivity toward protonated primary amines [
49]. These combined features offer great sensitivity, selectivity, and overall detection capabilities in analytical and diagnostic applications, making host molecule
10 suitable for use as a chemosensor.
2.2. Synthesis of the New Host Molecule
Herein, we introduce two different procedures for the synthesis of host molecule
10 and demonstrate its use as a chemosensor by both analytical studies and computational chemistry simulations using several biogenic and non-biogenic mono- and oligoamines, including spermine (
4) and spermidine (
3). The synthesis of macrocycle
10 started with the commercially available and relatively cheap 3-methoxybenzoic acid, which gave 4,5-dimethoxyacridone (
11) by a multistep procedure as reported [
51]. 4,5-dimethoxyacridone (
11) was converted to the appropriate chloro-acridine derivative (
12) using phosphorus pentachloride in phosphoryl chloride according to a previously described method [
52], but with an improved workup, in which most of the phosphoryl chloride was evaporated from the reaction mixture under reduced pressure prior to pouring it into the trimethylamine solution. This adjustment eliminated the need for the excessive amounts of trimethylamine and dichloromethane used in the original method and resulted in a purer product.
The chloro-acridine derivative (
12) was then subjected to a
Kharasch type cross-coupling reaction with dilithium tetrachlorocuprate and palladium acetate as catalysts, in the presence of phenylmagnesium bromide, where the intended product was 9-phenyl derivative
13 (
Scheme 1).
Although the expected product could not be obtained, byproduct 4,4′,5,5′-tetramethoxy-9,9′-biacridine (
14) was isolated with a good yield (71%). After identifying this byproduct, we determined that it would be used for the synthesis of macrocycle
10 (
Scheme 2). The methyl groups of
14 were cleaved with pyridinium chloride to obtain tetrahydroxy-biacridine
15. Macrocyclization of
15 with tetraethylene glycol ditosylate was carried out in DMF using potassium carbonate as a base to produce
10. Bismacrocycle
10 was isolated with a low yield (6%), which is typical for these types of reactions, especially in this case where the macrocycle rings can be closed in several ways. The primary byproducts include compounds where the ditosylate reacted with hydroxyl groups on two different acridine subunits of the biacridine, as well as partially reacted compounds where only one ring has closed. Other minor byproducts include compounds where the ditosylate reacted with only one hydroxyl group, as well as those where the ditosylate reacted with hydroxyl groups on two different biacridine molecules, leading to no macrocyclic ring formation.
Another method for the synthesis of macrocycle
10 is outlined in
Scheme 3, where the starting material was a reported acridono-crown ether (
16) [
53]. Crown ether
16 reacted with phosphorus pentachloride in phosphoryl chloride resulting in chloroacridino-crown ether
17, which is reported as an unstable compound and has not been isolated or characterized before [
53]. Chloroacridino-crown ether
17 was converted to macrocycle
10 by a coupling reaction using zinc powder in
tert-butylbenzene giving a 7% yield.
The probable explanation for the low yield of this reaction is that chloroacridino-crown ether
17 can form a complex with about four moles of water (according to the NMR spectrum of the crude product). Coordinating water within the cavity is a common phenomenon among analogous macrocycles [
54]. In this case, decomplexation of water takes place during the reaction, which leads to the hydrolysis of
17 resulting in
16 as a byproduct. Attempts were made to decomplex water by passing
17 through a column filled with aluminum oxide, but one mole of water always remained in complex form even after multiple attempts, which decreased the yield of
10.
2.3. In Silico Structure Optimization of Crown Ether—Oligoamine Complexes
Molecular modeling was used to gain insight into the nature of interactions between the host molecule and investigated oligoamines. The optimal structures of 10 and its complexes formed with spermine (4), spermidine (3), norspermidine (5) and N,N′-bis-(2-aminoethyl)propane-1,3-diamine (19) were determined by density functional theory (DFT) calculations.
In its minimal energy state, host molecule
10 assumes a fully extended conformation (
Figure 4). The planarity of the acridine units is undisrupted and the centers of the crown ether macrorings stay in the proximity of the aromatic planes. The bond connecting the acridine units falls into both aromatic planes; therefore, bending cannot be observed between the two rings. The equilibrium conformation determined in the modeling of host molecule
10 closely aligns with the observed conformation of biacridines [
55], with an angle of approximately 80 degrees between the planes of the two acridine units.
The optimized structure for bis(crown ether)
10 complex with spermine (
4) (
Figure 5) suggests a four-podal interaction. The crown ether macrorings coordinate the protonated primary amino groups of spermine (
4), forming hydrogen bonds with the oxygens at the 7th and 13th positions and the nitrogen of the acridine units. The protonated secondary amine parts are in proximity to the acridine units, and both parts form cation–
π interactions with the acridine units. According to the model, spermine (
4) is ideal in terms of chain length, since
10 retains its extended conformation, where both crown ether rings lie in the aromatic planes of the acridine units. Torsion in the acridine units can be observed upon the coordination of spermine (
4).
A distance of 12.2 Å can be measured between the terminal amines of spermine in complex with compound
10 (
Figure 6a). It is apparent that the geometry of bound spermine is very close to, and highly overlaps with, a minimal energy state conformation of the molecule, i.e., only a minuscule strain occurs in the structure of spermine upon binding to the host molecule
10 (
Figure 6b).
In the complexes with optimal coordination determined for spermidine (
3) and norspermidine (
5) (
Figure 7), the protonated secondary amine part forms cation–
π interactions with both acridine units, while the protonated primary amino groups coordinate with the heteroatoms of the crown ether rings. Due to the shorter chain lengths, at least one of the two crown ether rings has to rotate out of the plane of the acridine unit in order to coordinate with the protonated primary amino groups. Both protonated primary amino groups of spermine (
4) are ideally positioned for coordination with the crown ether rings; however, in our model, norspermidine (
5) lacks one hydrogen bond with the heteroatoms of the crown ether rings, due to its shorter chain than the ideal distance for coordination. The acridine units show some degree of distortion and bending compared to the optimized structure of the uncomplexed host molecule.
In the coordination complex determined for
N,
N′-bis-(2-aminoethyl)propane-1,3-diamine (
19), the two protonated primary amino groups occupy the interiors of the crown ether rings, but only one cation–
π interaction is present with one of the acridine units, although
19 has two protonated secondary amine parts (
Figure 8).
The distance of the protonated amine parts is not ideal to form two cation–π interactions at the same time. Torsion of the acridine units can be observed, as well as considerable bending between the aromatic planes compared to those of uncomplexed biscrown ether 10 can also be seen.
2.4. Spectroscopic Studies on Molecular Recognition with Oligoamines
The molecular recognition of host molecule 10 with protonated oligoamines was studied by fluorescence spectrophotometric titrations. One of the aims was validating the complexation properties predicted by computational modeling. On the other hand, experimental results may reveal important differences and effects, which play crucial roles in practice, but cannot be predicted by computational chemistry. Moreover, the comparison of theoretical and experimental methods enables the extrapolation of established relationships to the range of potential guest molecules, which have not been investigated yet.
The titrations were carried out both in nonaqueous and semi-aqueous media, using acetonitrile and distilled water as solvents. (Fully aqueous medium could not be used due to the poor solubility of the host molecule in water.) The analyte solutions were added with a Hamilton-syringe to the 10−7 M acetonitrile solutions of the ligand. The amine guest molecules were in fully protonated forms in water. In the cases of titrations in semi-aqueous media, the HCl salts of the amines were uniformly used, while the acetonitrile solutions of the corresponding neutral amines were added to the solutions of the host compound during the nonaqueous titrations.
Molecular recognition studies were carried out on selected commercially available mono- and oligoamine derivatives (
1,
3,
4,
5,
18,
19) including the biologically relevant ones. The investigated compounds are shown in
Figure 9.
Spectrofluorimetric titrations were carried out in a wide range of guest compound concentrations. To determine the structural preference of the host in amine recognition and to calculate the stability constants, global nonlinear regression was applied on the titration datasets. Both the complex stoichiometry and complex stabilities were determined according to the most recent and well-established methods [
56,
57,
58]. The adequacy of the fitted models was checked by statistical
F-probe. Detailed information regarding the regression methods can be found in the Experimental Section. The results can be seen in
Figure 10,
Figure 11 and
Figure 12.
The results for complex stoichiometry and stability constant calculations based on the regression analyses on titration datasets are summarized in
Table 1. The complex stoichiometries are not shown separately in the table, since in all cases the formation of 1:1 and then in a consecutive step 1:2 (host:guest) complexes were observed. This is not surprising as the host molecule contains two macrocycle rings as the main binding sites besides other possibilities to form additional intermolecular non-covalent binding forces. Accordingly, it is obvious that the cooperativity in guest-binding has a considerable effect on the complexation equilibrium.
According to the calculations, the following stability order can be concluded: spermidine~spermine (4) > norspermidine (5) > N,N′-bis(2-aminoethyl)-1,3-propanediamine (19) > putrescine (1) > n-butylamine (18). In most cases, cooperativity effects did not take place since there is a homotropic system of the host molecule with two identical binding sites. As the two macrocycle rings of the host molecule are separated from each other, the occupation of the first one practically does not affect the coordination ability of the second one; thus, at most two amine guests can be bonded simultaneously and independently. As the first complexed amine guest can be fixed with additional different non-covalent binding forces within the complex in comparison to the second one, the corresponding logK1 values are consequently higher. The coordination of the second amine guest starts after the host molecule forms a 1:1 host–guest complex. Thus, it is a stepwise binding process. This complexation behavior is also supported by the observed spectral changes as the 1:2 complexes at the second coordination step result in an opposite-direction in the spectral change. This phenomenon is probably attributed to the termination of the cation-π interaction(s) established between the first coordinated ammonium part and the appearance of the more distant acridine unit upon the occupation of the second macrocyclic cavity. However, as the cation-π interaction is significantly weaker than the three-podal hydrogen bonding within the macrocycle cavity, the complexation of the first amine guest does not hinder significantly the acceptance of the second one.
The only exception to the binding cooperativity was the case of the spermine-coordination, when regression analysis revealed a negative cooperativity in coordinating the second protonated amine part corresponding to the 1:2 (host:guest) complex stoichiometry. This is attributed to the increased chain length of spermine (
4) compared to the other oligoamine analogues, which allows the opposite-side terminal protonated primary amino group to be complexed by the second macrocycle ring without a significant structural distortion (see
Figure 5). Although the complexation of the first ammonium group hinders the second one to dock into the cavity of the other crown ether ring, it seems to be a slight effect, since the competing ammonium group can push off the previously complexed one from one of the binding sites.
The latter phenomenon cannot take place regarding the shorter-chain amines, because the double-inclusion complexation in the case of the initial 1:1 stoichiometry causes a conformational distortion of the structure of the host molecule moving the crown ether rings slightly toward each other. This results in a reduced kinetic stability of the complex, which is also supported by the modeling (
Figure 13).
Finally, one of the terminal primary ammonium groups departs followed by the conformational relaxation of the host molecule. As revealed by the modeling, the other terminal primary ammonium group remained in the cavity after this partial dissociation of the complex. Thus, the short-lived complexation of both chain ends causes the lack of cooperation effect when forming 1:2 (host:guest) complex with amines of a shorter chain than spermine (4).
Formation of complexes with other stoichiometries were not observed. It is not surprising, as more than two protonated amine parts can only be coordinated by cation-
π interactions with the empty heteroaromatic regions. As the cavity of the 18-crown-6 ether-type ring exactly fits to the size of the primary ammonium ion [
43,
44], only weaker interactions than the three-podal hydrogen bonding could take place when approaching the guest molecule to the acridine units.
The stability constants for the second-step complexations proved to be smaller than those for the 1:1 complex in accordance with the expectations for a stepwise binding model. In the cases when logK values did not reached 2.0, the uncertainty of the fitting process on the titration datasets significantly increased, thus accurate values could not be determined. However, the interactions for these very unstable complexes are too weak to be exploited in any applications.
Some stability constants indicate a very stable complexation among reversible complexing host molecules. Although the high degree of conformational flexibility of aliphatic oligoamines is not favored for complexation from the entropic point of view, the several protonated amine parts and electron-rich acridine units stabilize the complex effectively by increasing the number of coordinative interactions.
The first investigated amine was n-butylamine (
18), which is the simplest structural subpart of the oligoamines in focus. In this case, only complexation of the primary ammonium group of the guest with the macrocycle ring of the host can take place. According to literature analogues [
49], there is a three-podal hydrogen bonding within the complex involving the ammonium protons of the guest and the nucleophilic acridine-
N atom and two alternating ethereal
O atoms of the host. It is obvious that only one macroring can take part in the complexation due to the rigid structure of the host. Accordingly, a significantly smaller complex stability constant is expected than those for oligoamines. As we increase the chain length of the oligoamine host, the possibility of further interactions opens up. The protonated secondary amine parts, outside the complexed primary ammonium groups, can establish cation-
π interactions with one or two acridine units, depending on the chain length of the oligoamine. In parallel, the corresponding complex stabilities are continuously increasing (see
Table 1).
Additionally, we also determined the stability constants by titrating with unprotonated amines. It is well known that the lack of the positive charge results in a smaller complex stability constant even if the coordination is possible [
43,
44], while the additional coordinative interactions with the delocalized
π-electron system of the acridine units are also missing. The results showed an orders of magnitude decrease in stability constants (see
Supplementary Materials).