The Chemistry of Selenosilanes: A Topic Overview
Abstract
:1. Introduction
2. Synthesis of Selenosilanes
3. Selenosilanes in Chemical Synthesis
3.1. Silyl Selenides in the Nucleophilic Substitutions on Organic Substrates
3.1.1. Reaction with Halogens or Halogenated Compounds
3.1.2. Reaction with Benzyl and Allylic Alcohols
3.1.3. Reaction with Acetates and Ethers
3.1.4. Reaction of Selenosilanes with Sulfurated Organic Substrates
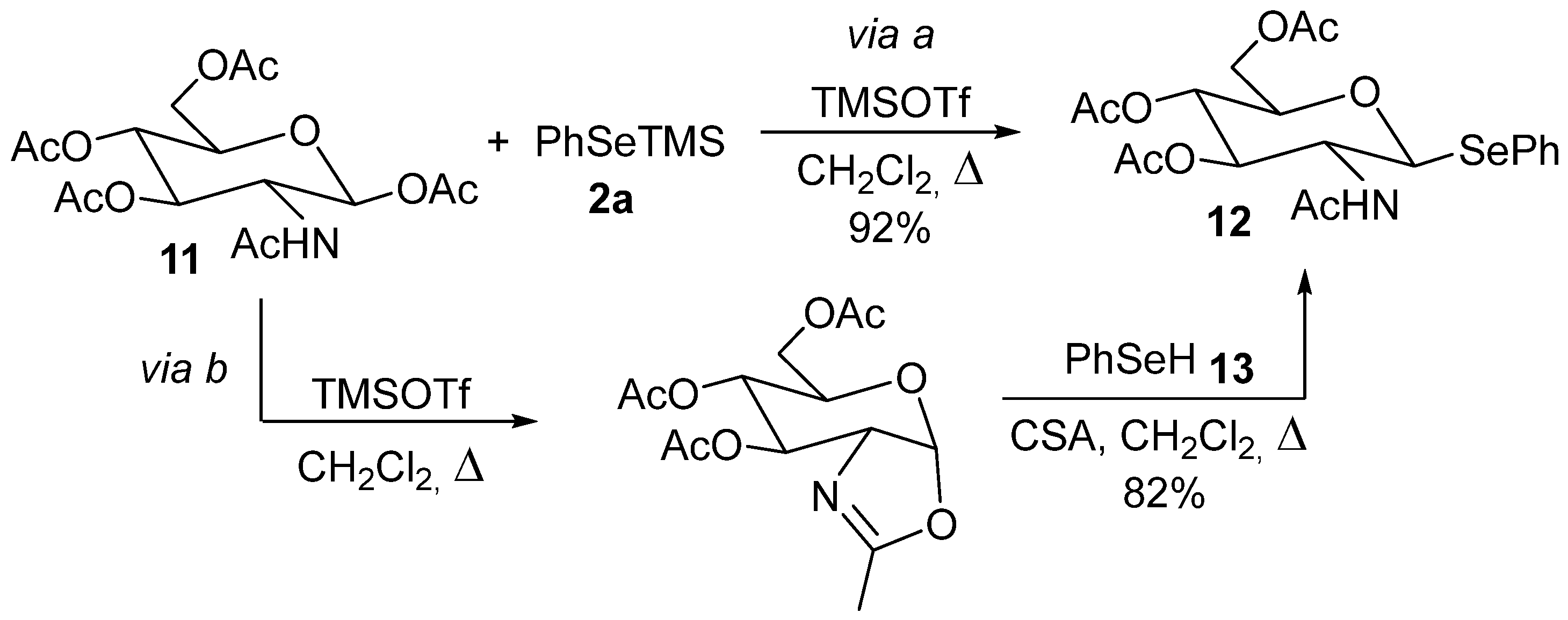
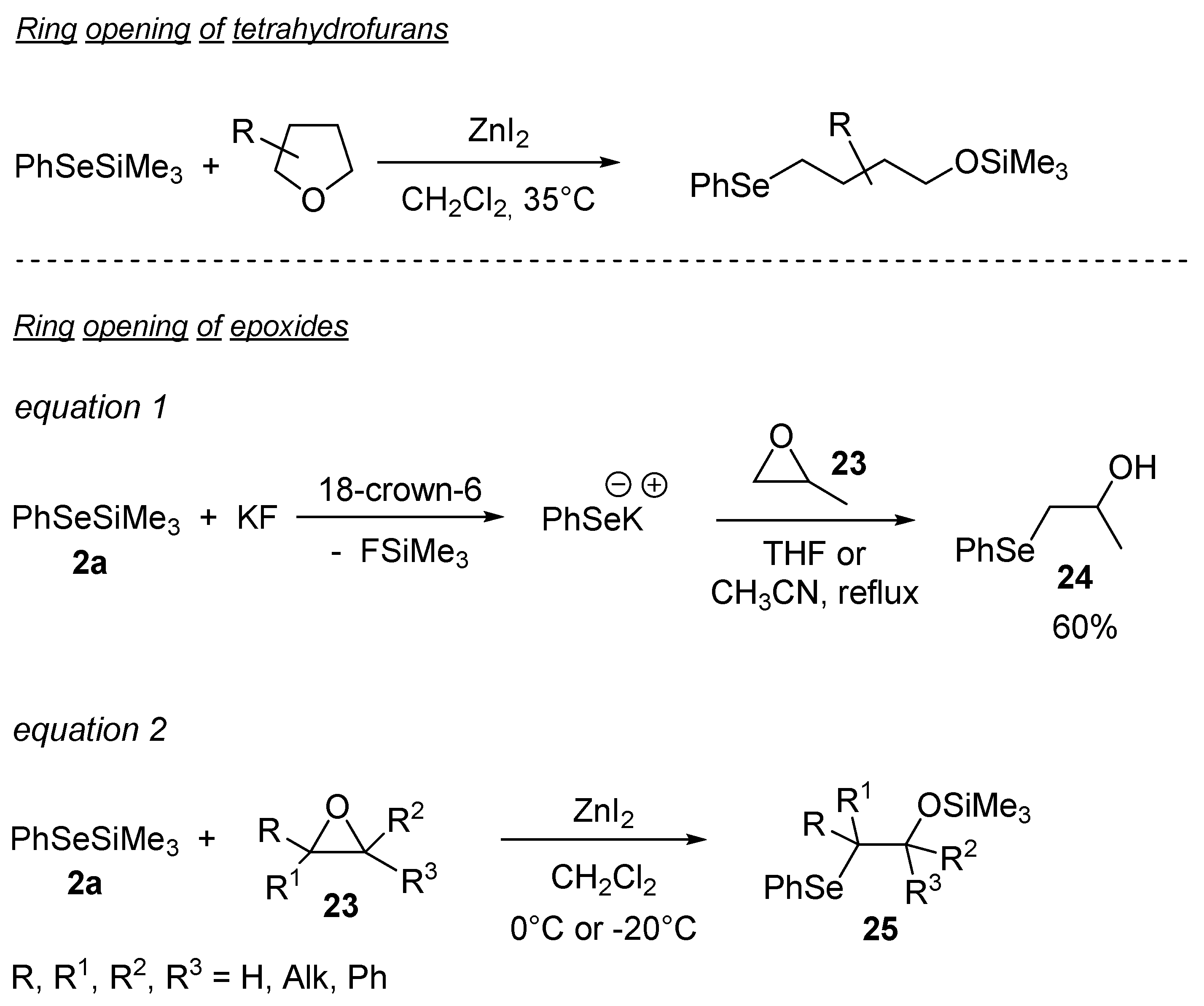
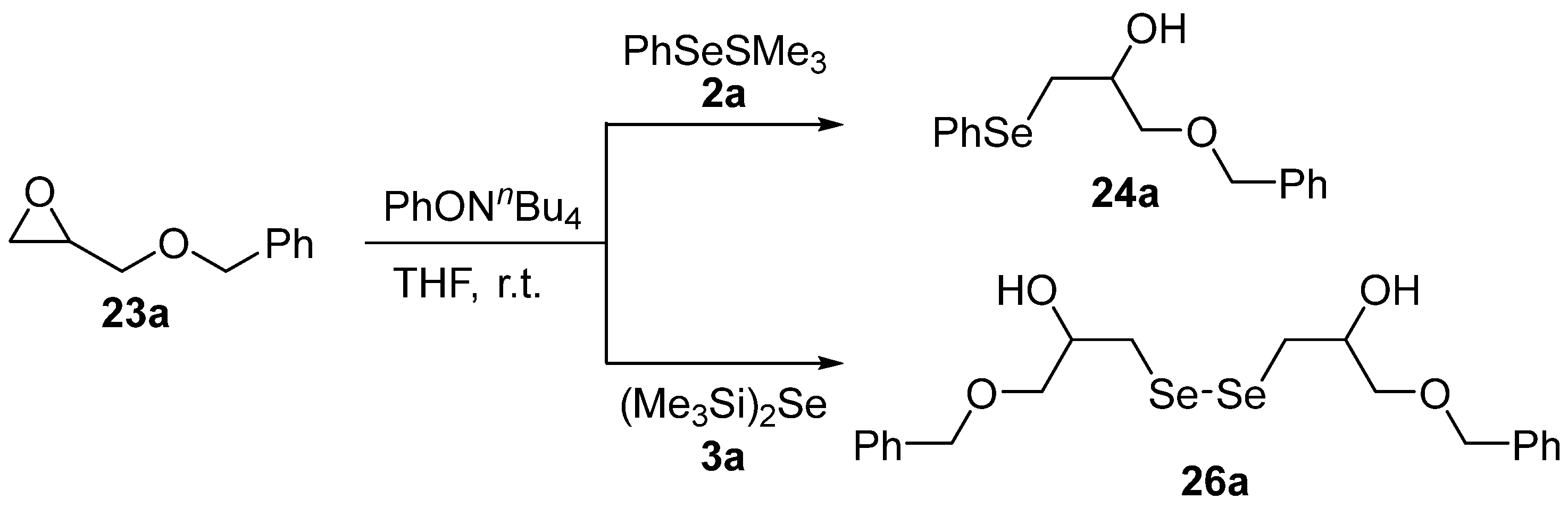
3.1.5. Ring Opening of Heterocyclic Rings (O, S, N) by Selenosilanes
3.2. Reaction with C=O Containing Compounds
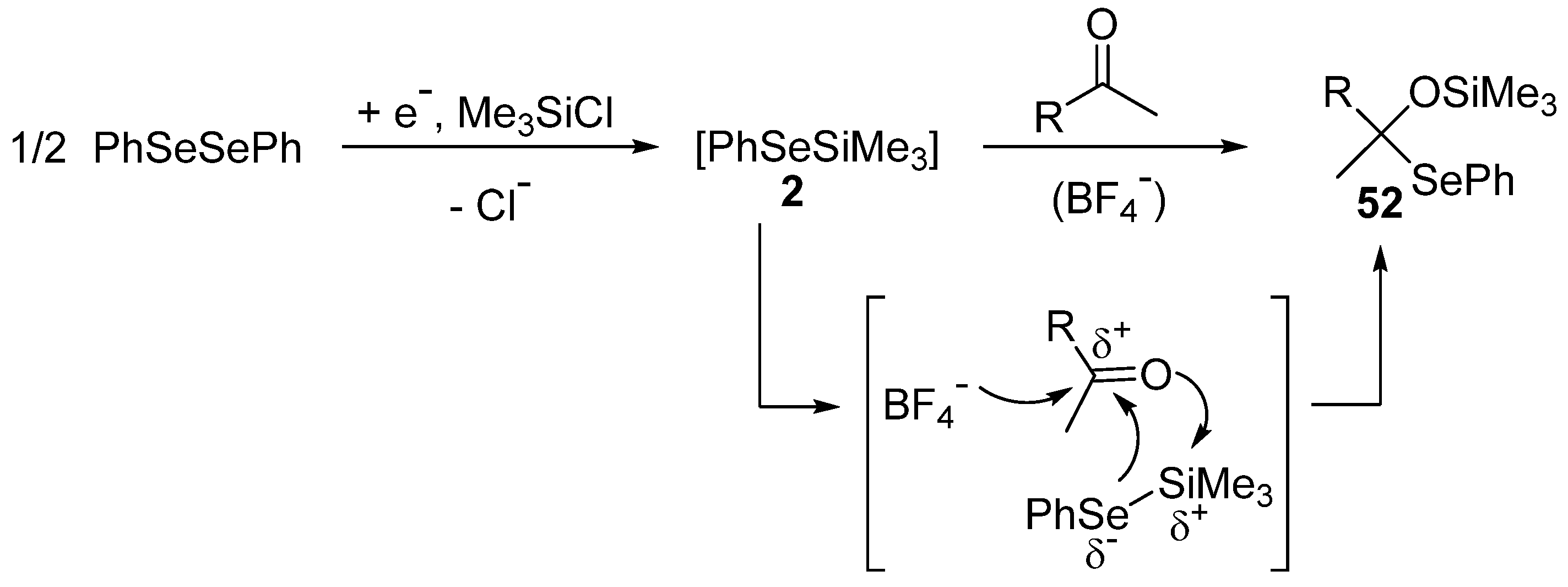
3.2.1. Reaction with Aldehydes and Ketones
3.2.2. Reaction with Acyl Chlorides
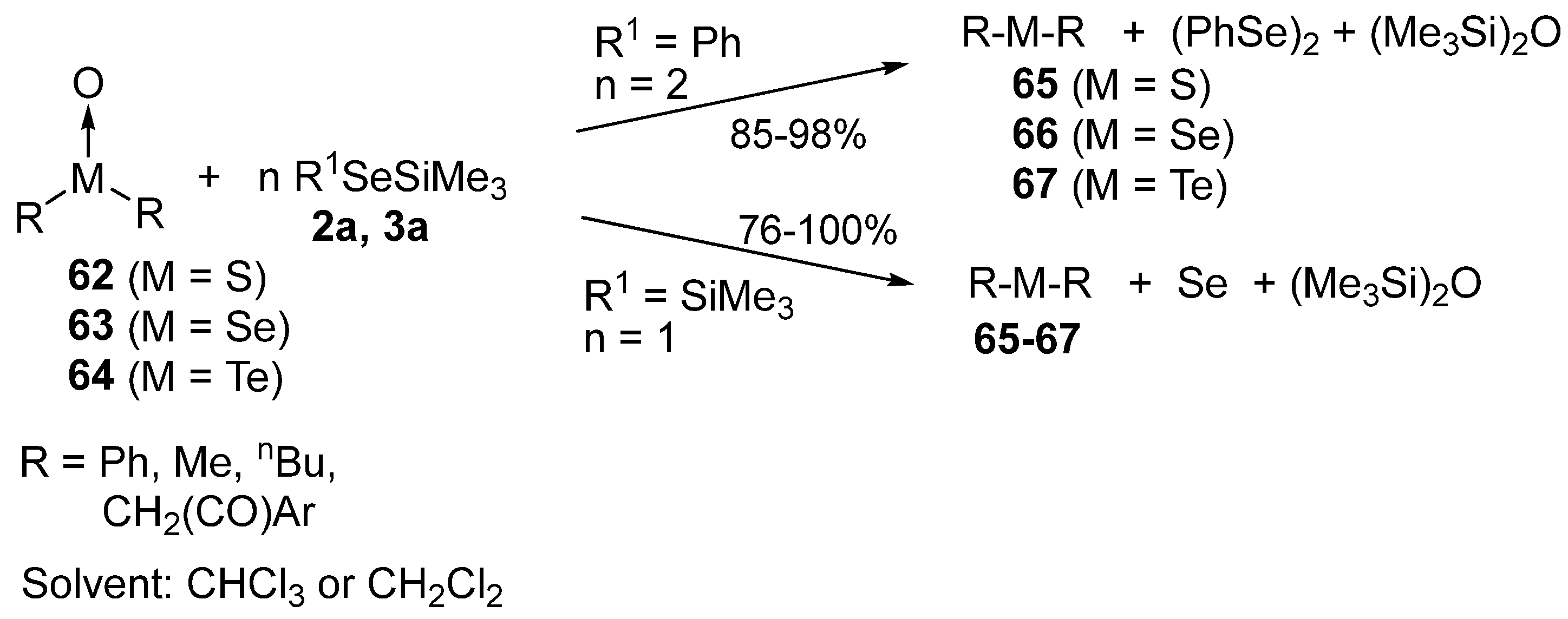
3.3. Reduction of Oxides of the Group 16 Elements (S, Se, Te)
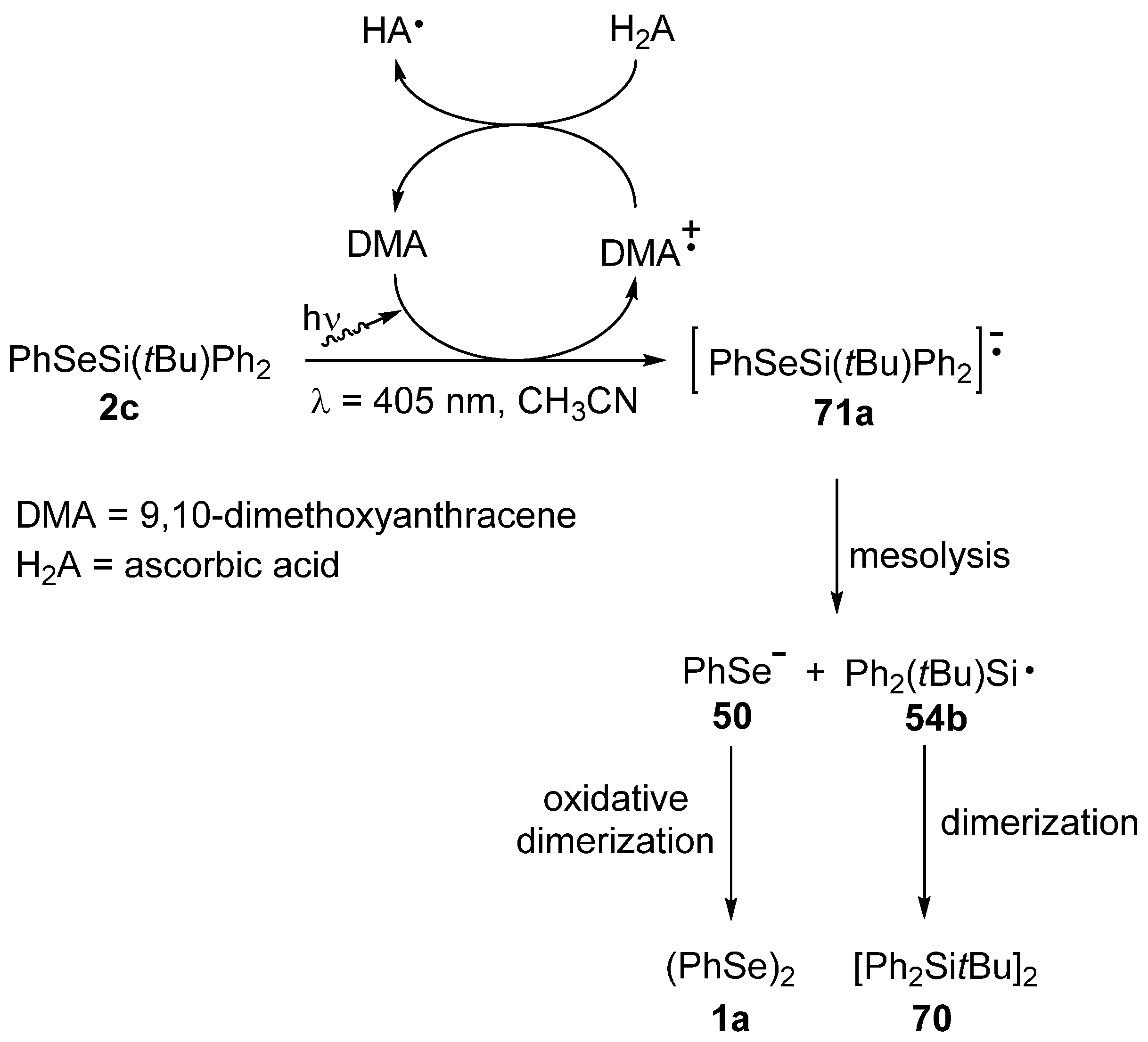
3.4. Reactivity of Selenosilanes under Radical Conditions
3.5. Metal-Selenium Cluster Compounds
3.6. Silyl Selenides as Se-Precursors for Atomic Layer Deposition (ALD)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lenardão, E.J.; Santi, C.; Sancineto, L. New Frontiers in Organoselenium Compounds; Springer: New York, NY, USA, 2018. [Google Scholar] [CrossRef]
- Wirth, T. (Ed.) Organoselenium Chemistry: Synthesis and Reactions; Wiley-VCH: Weinheim, Germany, 2012; ISBN 978-3-527-32944-1. [Google Scholar]
- Armitage, D.A. The Chemistry of Organic Silicon Compounds; Rappoport, Z., Apeloig, Y., Eds.; John Wiley & Sons: Chichester, UK, 1998; Volume 2, Chapter 31; pp. 1869–1894. ISBN 978-0-471-96757-6. [Google Scholar]
- Tanini, D.; Capperucci, A.; Menichetti, S. Nucleophilic Chalcogen-containing Reagents. In Chalcogen Chemistry; Lippolis, V., Santi, C., Lenardão, E.J., Braga, A.L., Eds.; Royal Society of Chemistry: London, UK, 2023; Chapter 12; pp. 307–321. [Google Scholar]
- Baker, A.; Wirth, T. Silyl Sulfides and Selenides. Sci. Synth. Updates 2017, 1, 189–202. [Google Scholar] [CrossRef]
- Miyoshi, N.; Ishii, H.; Kondo, K.; Murai, S.; Sonoda, N. Convenient Syntheses of Phenyl Trimethylsilyl Selenide and Benzeneselenol. Synthesis 1979, 1979, 300–301. [Google Scholar] [CrossRef]
- Detty, M.R. Trimethylsilyl Iodide. Preparation from and Catalytic Behavior with Phenylselenotrimethylsilane. Tetrahedron Lett. 1978, 43, 4189–4192. [Google Scholar] [CrossRef]
- Abe, H.; Yamasaki, A.; Harayama, T. Direct Conversion of a Benzylic Hydroxy Group into a Selenenyl Group Using the Phenyl Trimethylsilyl Selenide-Aluminum Bromide Combination. Chem. Pharm. Bull. 1998, 46, 1311–1313. [Google Scholar] [CrossRef]
- Pandey, G.; Poleshwar Rao, K.S.S. A New Dimension in Radical Chain Group Transfer Reaction by Photosensitized Electron Transfer (PET) Reductive Activation of PhSeSiR3. Angew. Chem. Int. Ed. Engl. 1996, 34, 2669–2671. [Google Scholar] [CrossRef]
- Herzog, U. Synthesis and NMR Investigation of Selenobutyl Substituted Silanes and Oligosilanes. J. Prakt. Chem. 2000, 342, 379–388. [Google Scholar] [CrossRef]
- Poleschner, H.; Heydenreich, M.; Schilde, U. Reactions of RSe-EMe3 (E = Si, Ge, Sn, Pb) with XeF2 -RSe–F Equivalents in the Fluoroselenenylation of Acetylenes. Eur. J. Inorg. Chem. 2000, 2000, 1307–1313. [Google Scholar] [CrossRef]
- Clarembeau, M.; Cravador, A.; Dumont, W.; Hevesi, L.; Krief, A.; Lucchetti, J.; Van Ende, D. Synthesis of selenoacetals. Tetrahedron 1985, 41, 4793–4812. [Google Scholar] [CrossRef]
- Kuciński, K.; Gruszczyński, M.; Hreczycho, G. Ru-catalyzed Formation of Thiosilanes and Selenosilanes using Dichalcogenides as a User-Friendly Alternative to Thiols and Selenols. ChemCatChem 2022, 14, e202200961. [Google Scholar] [CrossRef]
- Jouikov, V.; Grigorieva, L. Competitive electrochemical thio- and selenenylation of chlorosilanes. Electrochim. Acta 1996, 41, 2489–2491. [Google Scholar] [CrossRef]
- Syper, L.; Mlochowski, J. Lithium diselenide in aprotic medium—A convenient reagent for synthesis of organic diselenides. Tetrahedron 1988, 44, 6119–6130. [Google Scholar] [CrossRef]
- Drake, J.E.; Glavineevski, B.M.; Hemmings, R.T.; Henderson, H.E. Silyl and Germyl Selenides and Tellurides. In Inorganic Syntheses; Bush, D.H., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 1980; Volume XX, Chapter 6; pp. 171–176. ISBN 0-471-07715-1. [Google Scholar]
- Detty, M.R.; Seidler, M.D. Bis(trialkylsilyl) Chalcogenides. 1. Preparation and Reduction of Group 6A Oxides. J. Org. Chem. 1982, 47, 1354–1356. [Google Scholar] [CrossRef]
- Hatanpää, T.; Pore, V.; Ritala, M.; Leskelä, M. Alkylsilyl Compounds of Selenium and Tellurium: New Precursors for ALD. ECS Trans. 2009, 25, 609–616. [Google Scholar] [CrossRef]
- Detty, M.R.; Seidler, M.D. Silyl Halides from (Phenylseleno)silanes. Reaction with Oxiranes and Alcohols to Give Hydrolytically Stable Silyl Ethers. J. Org. Chem. 1981, 46, 1283–1292. [Google Scholar] [CrossRef]
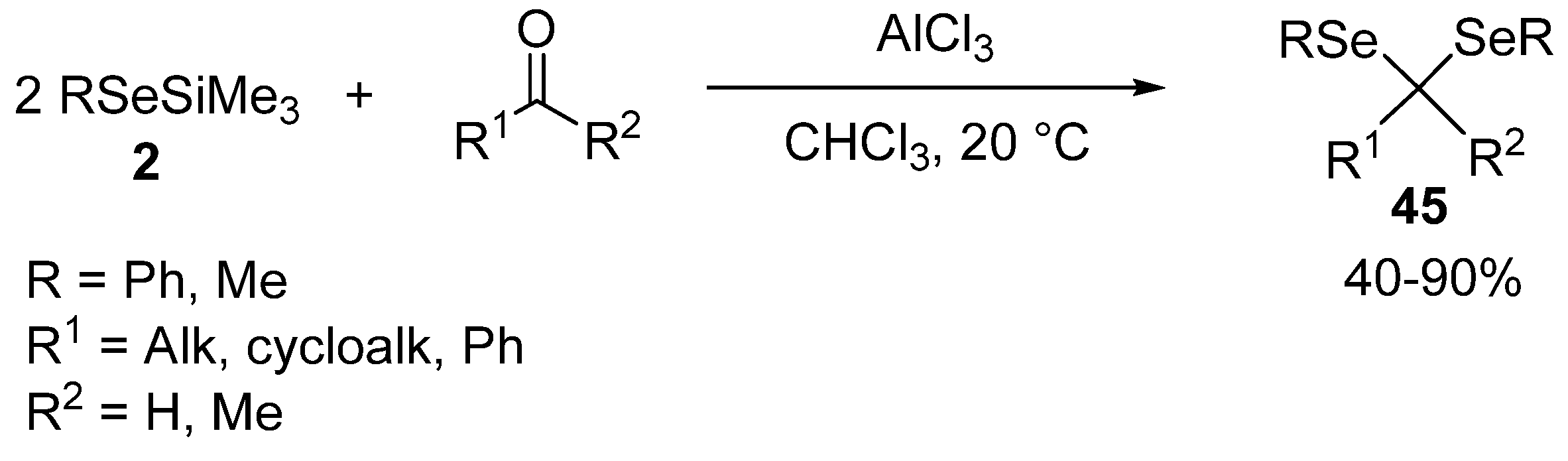
- Segi, M.; Kato, M.; Nakajima, T.; Suga, S.; Sonoda, N. A Convenient One-pot Synthesis of Unsymmetrical Selenium Compounds Using Bis(trimethylsilyl) Selenide. Chem. Lett. 1989, 1989, 1009–1012. [Google Scholar] [CrossRef]
- Taher, D.; Wallbank, A.I.; Turner, E.A.; Cuthbert, H.L.; Corrigan, J.F. Alk-2-ynyl Trimethylsilyl Chalcogenoethers by Nucleophilic Substitution of Propargyl Bromides. Eur. J. Inorg. Chem. 2006, 2006, 4616–4620. [Google Scholar] [CrossRef]
- Wrackmeyer, B.; García Hernández, Z.; Herberhold, M. 1-Cyclohepta-2,4,6-trienyl-selanes—a 77Se NMR study: Indirect nuclear 77Se-13C spin-spin coupling constants and application of density functional theory (DFT) calculations. Magn. Reson. Chem. 2007, 45, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Yamasaki, A.; Koshiba, N.; Takeuchi, Y.; Harayama, T. One-Pot Conversion of Allyl Alcohols into Selenochroman Derivatives. Chem. Pharm. Bull. 2001, 49, 1223–1225. [Google Scholar] [CrossRef]
- Grant, L.; Liu, Y.; Walsh, K.E.; Walter, D.S.; Gallagher, T. Galacto, Gluco, Manno, and Disaccharide-Based C-Glycosides of 2-Amino-2-deoxy Sugars. Org. Lett. 2002, 4, 4623–4625. [Google Scholar] [CrossRef]
- Pfrengle, F.; Reissig, H.-U. Internally Protected Amino Sugar Equivalents from Enantiopure 1,2-Oxazines: Synthesis of Variably Configured Carbohydrates with C-Branched Amino Sugar Units. Chem. Eur. J. 2010, 16, 11915–11925. [Google Scholar] [CrossRef]
- Crimmins, M.T.; Hauser, E.B. Synthesis of Crossed [2 + 2] Photocycloadducts: A Novel Approach to the Synthesis of Bridged Bicyclic Alkenes. Org. Lett. 2000, 2, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Fujii, H.; Yamasaki, A.; Kinome, Y.; Takeuchi, Y.; Harayama, T. Preparation of β-Seleno-α,β-Unsaturated Nitroalkenes Via A Sulfur-Selenium Exchange Reaction. Synth. Commun. 2000, 30, 543–549. [Google Scholar] [CrossRef]
- Viglianisi, C.; Bonardi, C.; Ermini, E.; Capperucci, A.; Menichetti, S.; Tanini, D. Selenosilane-Promoted Selective Mild Transformation of N-Thiophthalimides into Symmetric Disulfides. Synthesis 2019, 51, 1819–1824. [Google Scholar] [CrossRef]
- Miyoshi, N.; Hatayama, Y.; Ryu, I.; Kambe, N.; Murai, T.; Murai, S.; Sonoda, N. Ring Opening of Tetrahydrofurans with Phenyl Trimethylsilyl Selenide Catalyzed by Moist Zinc Iodide. Synthesis 1988, 1988, 175–177. [Google Scholar] [CrossRef]
- Dapkekar, A.B.; Satyanarayana, G. Electrochemical selenofunctionalization of unactivated alkenes: Access to β-hydroxyselenides. Org. Biomol. Chem. 2024, 22, 1775–1781. [Google Scholar] [CrossRef]
- Detty, M.R. Phenylselenotrimethylsilane. A novel source of phenylselenide anion. Tetrahedron Lett. 1978, 51, 5087–5090. [Google Scholar] [CrossRef]
- Miyoshi, N.; Kondo, K.; Murai, S.; Sonoda, N. Synthesis of β-siloxyalkyl phenyl selenides by the reaction of phenyl trimethylsilyl selenide with epoxides. Chem. Lett. 1979, 8, 909–912. [Google Scholar] [CrossRef]
- Wang, P.-A. Organocatalyzed enantioselective desymmetrization of aziridines and epoxides. Beilstein J. Org. Chem. 2013, 9, 1677–1695. [Google Scholar] [CrossRef]
- Tiecco, M.; Testaferri, L.; Marini, F.; Sternativo, S.; Del Verme, F.; Santi, C.; Bagnoli, L.; Temperini, A. Synthesis of enantiomerically enriched β-hydroxy selenides by catalytic asymmetric ring opening of meso-epoxides with (phenylseleno)silanes. Tetrahedron 2008, 64, 3337–3342. [Google Scholar] [CrossRef]
- Capperucci, A.; Tiberi, C.; Pollicino, S.; Degl’Innocenti, A. Tetrabutylammonium phenoxide induced reaction of silyl nucleophiles. Tetrahedron Lett. 2009, 50, 2808–2810. [Google Scholar] [CrossRef]
- Tanini, D.; Tiberi, C.; Gellini, C.; Salvi, P.R.; Capperucci, A. A Straightforward Access to Stable β-Functionalized Alkyl Selenols. Adv. Synth. Catal. 2018, 360, 3367–3375. [Google Scholar] [CrossRef]
- Capperucci, A.; Salles, C.; Scarpelli, S.; Tanini, D. Selective access to sulfurated and selenated heterocycles by intramolecular cyclization of β-substituted sulfides and selenides. Phosphorus Sulfur Silicon Relat. Elem. 2017, 192, 172–174. [Google Scholar] [CrossRef]
- Capperucci, A.; Tanini, D.; Borgogni, C.; DegľInnocenti, A. Thiosilane- and Organoselenosilane-Mediated Novel Access to 3,7-Disubstituted-1,2,5-trithiepanes and -1,2,5-dithiaselenepanes. Heteroat. Chem. 2014, 25, 678–683. [Google Scholar] [CrossRef]
- Tanini, D.; D’Esopo, V.; Tatini, D.; Ambrosi, M.; Lo Nostro, P.; Capperucci, A. Selenated and Sulfurated Analogues of Triacyl Glycerols: Selective Synthesis and Structural Characterization. Chem. Eur. J. 2020, 26, 2719–2725. [Google Scholar] [CrossRef]
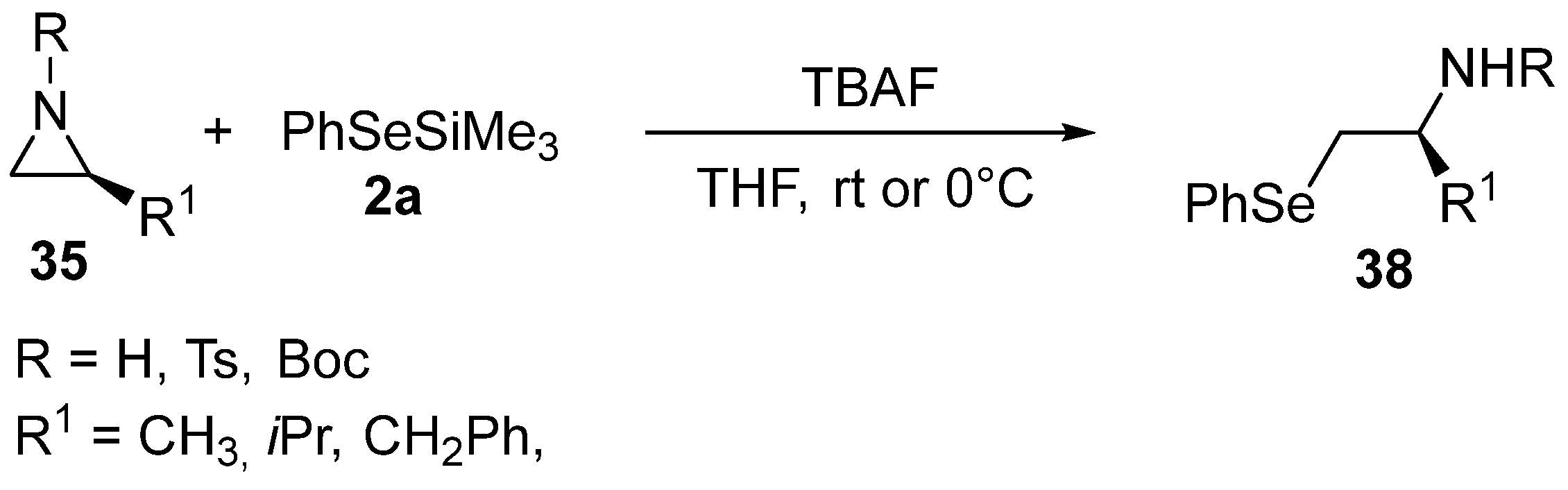
- Tanini, D.; Barchielli, G.; Benelli, F.; Degl’Innocenti, A.; Capperucci, A. Aziridines Ring Opening by Silyl Chalcogenides: A Stereoselective Access to Polyfunctionalized Molecules as Precursor of Sulfurated and Selenated Heterocycles. Phosphorus Sulfur Silicon Relat. Elem. 2015, 190, 1265–1270. [Google Scholar] [CrossRef]
- Tanini, D.; Borgogni, C.; Capperucci, A. Mild and selective silicon-mediated access to enantioenriched 1,2-mercaptoamines and β-amino arylchalcogenides. New J. Chem. 2019, 43, 6388–6393. [Google Scholar] [CrossRef]
- Senatore, M.; Lattanzi, A.; Santoro, S.; Santi, C.; Della Sala, G. A general phosphoric acid-catalyzed desymmetrization of meso-aziridines with silylated selenium nucleophiles. Org. Biomol. Chem. 2011, 9, 6205–6207. [Google Scholar] [CrossRef]
- Della Sala, G. Studies on the true catalyst in the phosphate-promoted desymmetrization of meso-aziridines with silylated nucleophiles. Tetrahedron 2013, 69, 50–56. [Google Scholar] [CrossRef]
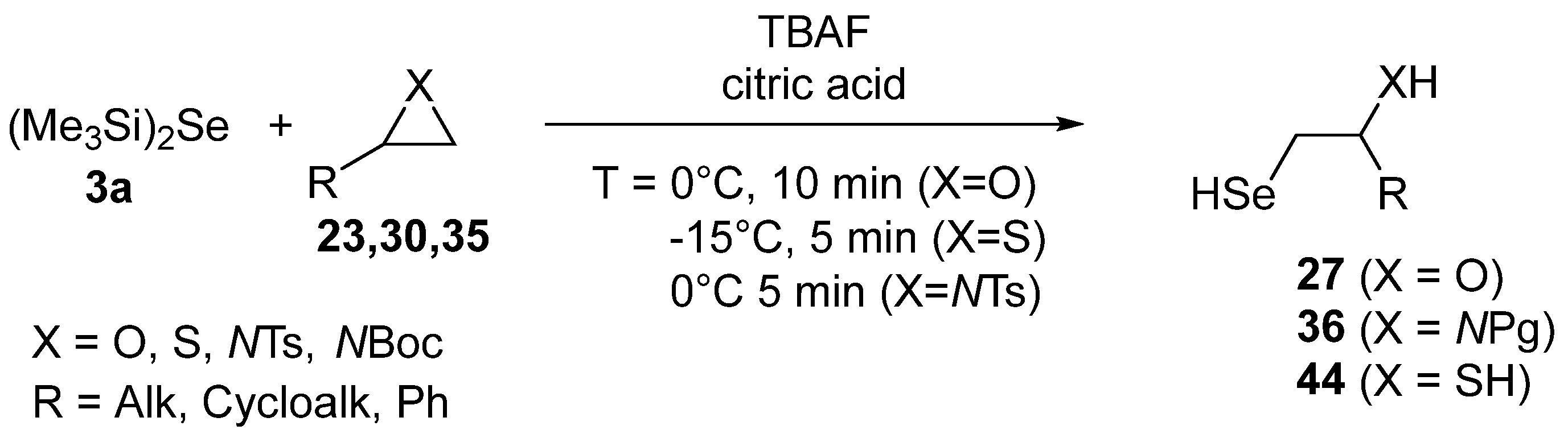
- Tanini, D.; Degl’Innocenti, A.; Capperucci, A. Bis(trimethylsilyl)selenide in the Selective Synthesis of β-Hydroxy, β-Mercapto, and β-Amino Diorganyl Diselenides and Selenides Through Ring Opening of Strained Heterocycles. Eur. J. Org. Chem. 2015, 2015, 357–369. [Google Scholar] [CrossRef]
- Capperucci, A.; Coronnello, M.; Salvini, F.; Tanini, D.; Dei, S.; Teodori, E.; Giovannelli, L. Synthesis of functionalised organochalcogenides and in vitro evaluation of their antioxidant activity. Bioorg. Chem. 2021, 110, 104812. [Google Scholar] [CrossRef] [PubMed]
- Tanini, D.; Pecchi, T.; Ignat’ev, N.V.; Capperucci, A. Ionic Liquids-Assisted Ring Opening of Three-Membered Heterocycles with Thio- and Seleno-Silanes. Catalysts 2022, 12, 1259. [Google Scholar] [CrossRef]
- Dumont, W.; Krief, A. Synthesis of Diselenoacetals and O-(Trimethylsilyl)monoselenoacetals. Angew. Chem. Int. Ed. Engl. 1977, 16, 540–541. [Google Scholar] [CrossRef]
- Degl’Innocenti, A.; Capperucci, A.; Acciai, M.; Tiberi, C. Silicon-Mediated Synthesis of Selenoaldehydes and Selenoacylsilanes and Their Hetero Diels-Alder Reactions. Phosphorus Sulfur Silicon Relat. Elem. 2009, 184, 1621–1626. [Google Scholar] [CrossRef]
- Torii, S.; Inokuchi, T.; Hasegawa, N. A direct arylselenenylation of enones with diaryl diselenides by a electroreductive procedure. Chem. Lett. 1980, 1980, 639–640. [Google Scholar] [CrossRef]
- Nishiyama, Y.; Kajimoto, H.; Kotani, K.; Nishida, T.; Sonoda, N. Reaction of Carbonyl Compounds with Trialkylsilyl Phenylselenide and Tributylstannyl Hydride under Radical Conditions. J. Org. Chem. 2002, 67, 5696–5700. [Google Scholar] [CrossRef] [PubMed]
- Capperucci, A.; Degl’Innocenti, A.; Tiberi, C. Organoselenosilane-Mediated Selective Mild Access to Selenolesters, Selenoanhydrides and Diacyl Diselenides. Synlett 2011, 2011, 2248–2252. [Google Scholar] [CrossRef]
- Taher, D.; Corrigan, J.F. Aryl(trimethylsilyl)selenides as Reagents for the Synthesisof Mono- and Diselenoesters. Organometallics 2011, 30, 5943–5952. [Google Scholar] [CrossRef]
- Detty, M.R. Mild Red uctions of Oxides of the Group 6a Elements Sulfur, Selenium, and Tellurium with (Phenylseleno)trimethylsilane. J. Org. Chem. 1979, 44, 4528–4531. [Google Scholar] [CrossRef]
- Pandey, G.; Poleswara Rao, K.S.S.; Palit, D.K.; Mittal, J.P. Generation and Mesolysis of PhSeSiR3]•-: Mechanistic Studies by Laser Flash Photolysis and Application for Bimolecular Group Transfer Radical Reactions. J. Org. Chem. 1998, 63, 3968–3978. [Google Scholar] [CrossRef]
- Pandey, G.; Gadre, S.R. Generation and Mesolytic Dynamics of Organoselenane and Selenosilane Radical Ions: Development of Mechanistically Interesting and Synthetically Useful Chemistry. Acc. Chem. Res. 2004, 37, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Olkowska-Oetzel, J.; Sevillano, P.; Eichhöfer, A.; Fenske, D. Binary and Ternary Cluster Complexes Containing Gold-Selenium, Gold-Indium-Selenium and Gold-Gallium-Tellurium—Synthesis and Structures of [Au5Se2(PPh3)4]Cl, [(Au3Se)2{Ph2P(CH2)6PPh2}3]Cl2, [Au10Se4{Ph2P(CH2)5PPh2}4]InCl5, [Au4(SeInCl3)2{Ph2P(CH2)5PPh2}2], [Au2(TeGaCl3){Ph2P(CH2)6PPh2}]2 and [Au8Se4In{Ph2P(CH2)2PPh2}4](InCl4)3. Eur. J. Inorg. Chem. 2004, 2004, 1100–1106. [Google Scholar] [CrossRef]
- Cave, D.; Corrigan, J.F.; Eichhöfer, A.; Fenske, D.; Kowalchuk, C.M.; Rösner, H.; Scheer, P. Investigation of the Thermal Properties of a Series of Copper Selenide Cluster Molecules. J. Clust. Sci. 2007, 18, 157–172. [Google Scholar] [CrossRef]
- Schaub, T.; Backes, M.; Radius, U. Square-Planar (Pentafluorophenyl)nickel(II) Complexes by Derivatization of a C–F Activation Product. Eur. J. Inorg. Chem. 2008, 2008, 2680–2690. [Google Scholar] [CrossRef]
- Pore, V.; Hatanpää, T.; Ritala, M.; Leskelä, M. Atomic Layer Deposition of Metal Tellurides and Selenides Using Alkylsilyl Compounds of Tellurium and Selenium. J. Am. Chem. Soc. 2009, 131, 3478–3480. [Google Scholar] [CrossRef] [PubMed]
- George, S.M. Atomic Layer Deposition: An Overview. Chem. Rev. 2010, 110, 111–131. [Google Scholar] [CrossRef]
- Charvot, J.; Zazpe, R.; Macak, J.M.; Bureš, F. Organoselenium Precursors for Atomic Layer Deposition. ACS Omega 2021, 6, 6554–6558. [Google Scholar] [CrossRef]
- Charvot, J.; Pokorný, D.; Zazpe, R.; Krumpolec, R.; Pavliňák, D.; Hromádko, L.; Přikryl, J.; Rodriguez-Pereira, J.; Klikar, M.; Jelínková, V.; et al. Cyclic Silylselenides: Convenient Selenium Precursors for Atomic Layer Deposition. ChemPlusChem 2020, 85, 576–579. [Google Scholar] [CrossRef] [PubMed]

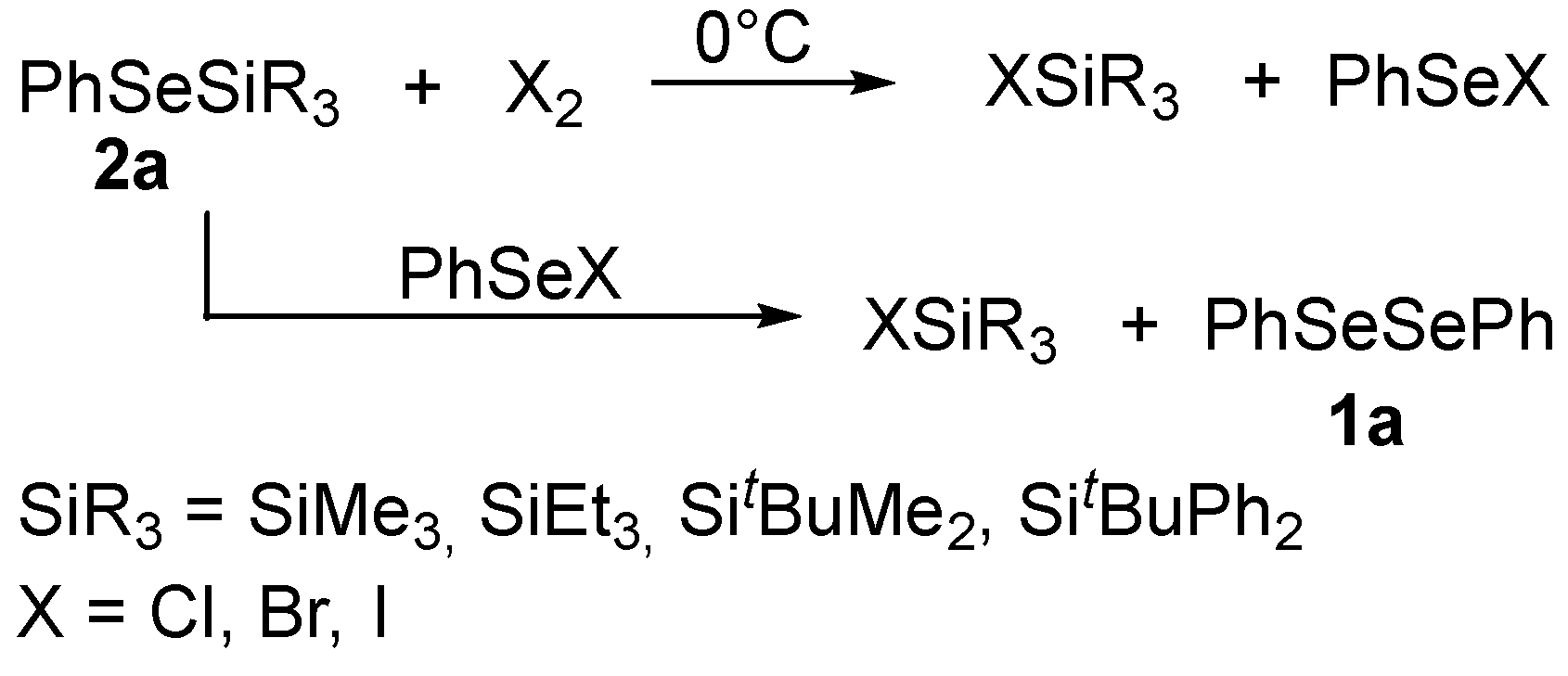
















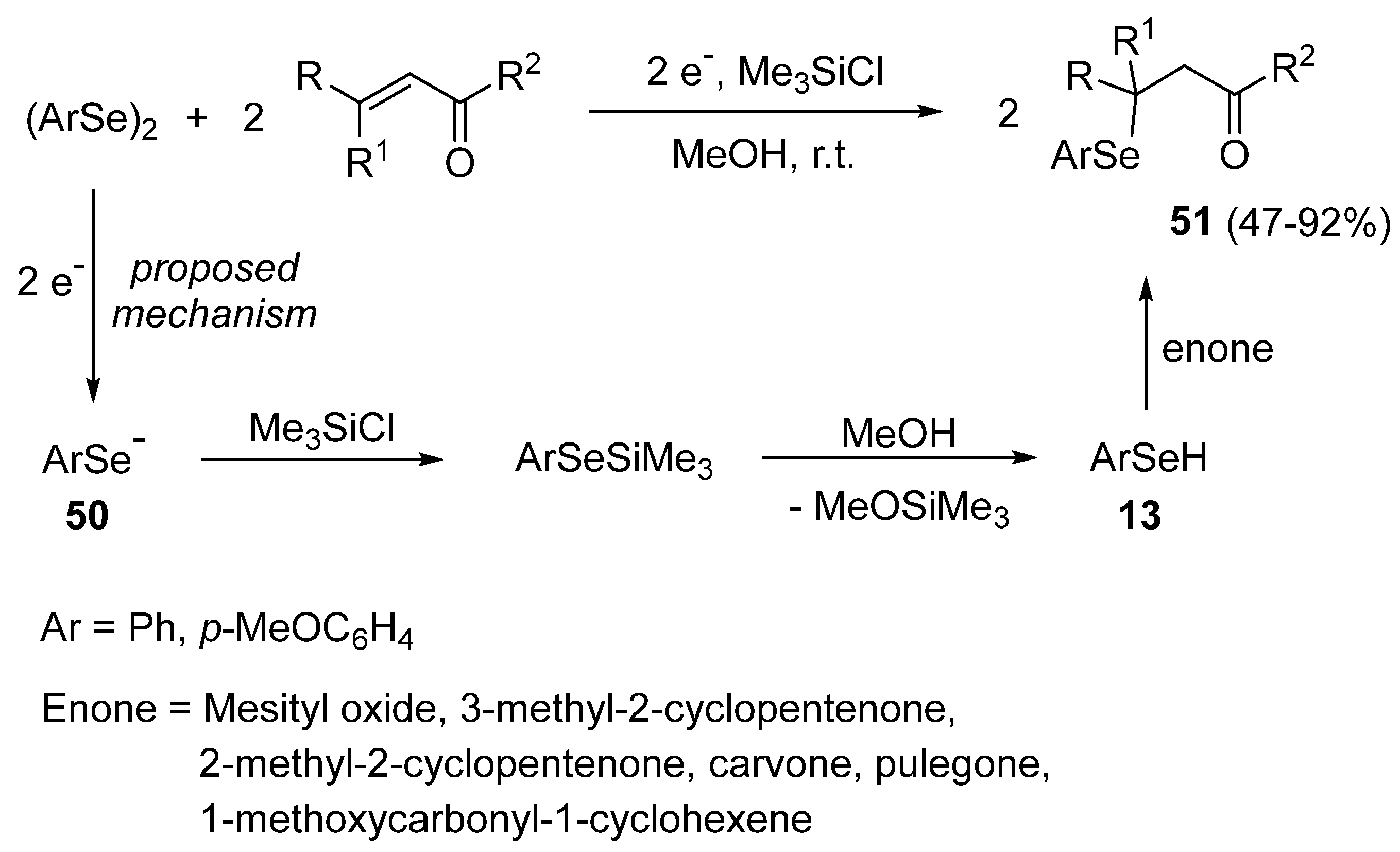






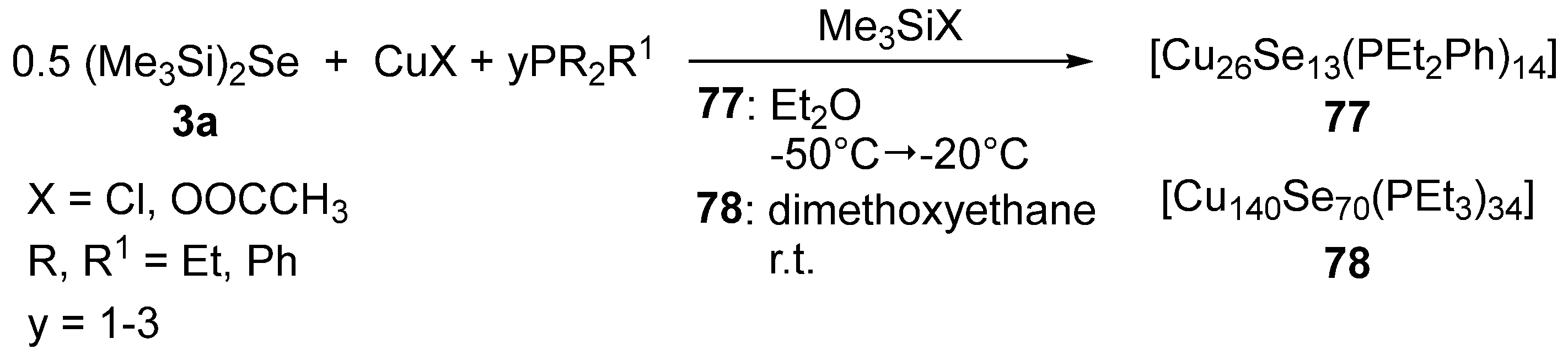



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanini, D.; Capperucci, A. The Chemistry of Selenosilanes: A Topic Overview. Molecules 2024, 29, 4595. https://doi.org/10.3390/molecules29194595
Tanini D, Capperucci A. The Chemistry of Selenosilanes: A Topic Overview. Molecules. 2024; 29(19):4595. https://doi.org/10.3390/molecules29194595
Chicago/Turabian StyleTanini, Damiano, and Antonella Capperucci. 2024. "The Chemistry of Selenosilanes: A Topic Overview" Molecules 29, no. 19: 4595. https://doi.org/10.3390/molecules29194595








