Silver Nanoparticles as a Potent Nanopesticide: Toxic Effects and Action Mechanisms on Pest Insects of Agricultural Importance—A Review
Abstract
1. Introduction
2. Silver Nanoparticles (AgNPs)
3. Role of AgNPs in Insect Control
3.1. AgNPs on Lepidoptera
3.2. AgNPs on Diptera
3.3. AgNPs on Coleoptera
3.4. AgNPs on Hemiptera
| Synthesis Method (Size and Concentration) | Especie (Order: Family) | Main Results | Mechanism Action | References |
|---|---|---|---|---|
| Commercial AgNPs. 209.5 nm. 500, 1000, 2000, and 4000 mg/L | Achaea janata (Lepidoptera: Erebidae), Spodoptera litura (Lepidoptera: Noctuidae) | Decrease in body weight | CarE, Glu, GST, SOD, CAT, and POD enzyme activity interference | [42] |
| Green synthesis: Entomopathogenic bacterium Bacillus thuringiensis kurstaki. 85 nm. 0.63, 1.25, 2.5, 5, 10, 20 mg/mL | Agrotis ípsilon (Lepidoptera: Noctuidae), Trichoplusia ni (Lepidoptera: Noctuidae) | LC50 of 0.46–0.81 mg/mL for T. ni. LC50 of 1.95–5.20 mg/mL for A. ipsilon | ND | [55] |
| Commercial AgNPs. 30 nm. 100, 200, 400, 800, 1600, 3200 mg/L | Bombyx mori (Lepidoptera: Bombycidae) | Cocoon growth and weight increase (100 and 200 mg/L). Increase in insect growth and mortality (1600 and 3200 mg/L) | Downregulation of expression of Calexcitin-2, cytosolic non-specific dipeptidase, S-formylglutathione hydrolase gene, s-formylglutathione hydrolase gene. Upregulation of Glutathione S-transferase s1 (GSTs1) genes, AK protein, Juvenile hormone binding protein (JHBP), and Isocitrate dehydrogenase. | [48] |
| Commercial AgNPs. 30 nm. 100, 200 and 400 mg/L. | Bombyx mori (Lepidoptera: Bombycidae) | Chaotic crawling, shrinking, disorders in the growth cycle, and diarrhea (100–400 mg/L). Destruction of the basal lamina, expansion of the calciform cells, and deformation of the columnar cells (100–400 mg/L). Reduction in enzyme expression (100 mg/L). Damage at the intestinal level and accumulation of reactive oxygen species (ROS). (400 mg/L) | Upregulation of genes related to location, activity and transmembrane transport (genes DEG-1, DEG-2, DEG-3, DEG-4, DEG-5, and DEG-6). Upregulation of HSP1, Cu/Zn Superoxide Dismutase (SOD), and EF-1. Downregulation of MIOX and ERP57. | [49] |
| Chemical method with stearic acid. 35–55 nm. 25, 50, 75, 100 and 200 mg/L. | Earias vittela (Lepidoptera: Noctuidae) | LC50 y LC90 of 45.46 y 172.98 mg/L, respectively. Mortality of 93.77% (200 mg/L). LC50 and LC90 of 25.12 y 160.36 mg/L respectively | Oxidative stress and interference with free thiol groups | [56] |
| Green synthesis: Leonotis nepetifolia leaves extract. 25–53 nm. 30, 60, 90, 120 and 150 mg/L. | Helicoverpa armigera (Lepidoptera: Noctuidae), Spodoptera litura (Lepidoptera: Noctuidae) | Antifeedant effect of 78.77% and 82.16%. LC50 values at 74.09 mg/L on S. litura and 84.58 mg/L on H. armigera. Pupicidal activity on S. litura (84.66%) and (77.44%) at 150 mg/L. Deformation of ecdysis, pupal malformation, and shrunken pupa. Increase in the larval developmental period (13.65 and 14.83 days, respectively) and pupal duration (15.17 and 17.58 days, respectively) | ND | [41] |
| Commercial AgNPs. 7–9, 45 nm. 3.5, 35, 100, and 180 mg/L. | Heliothis virescens (Lepidoptera: Noctuidae), Podisus maculiventris (Hemiptera: Pentatomidae), Trichoplusia ni (Lepidoptera: Noctuidae) | It increased adult pupation and hatching time at 180 mg/L on H. virescens. Delayed pupation, delayed hatched adults, anormal weigt and sooner oviposition at a concentration of 100 mg/L on P. maculiventris. | Increase expression of heat shock protein 70. Increase activities of caspase-3 and caspase-9, markers of apoptosis. Alteration of lipid peroxidation, malondialdehyde, and increased activities of antioxidant enzymes, Glutathione, Superoxide Dismutase, and Catalase. Increase in expression of p53 and p38 proteins. Reduction in tyrosinase activity. | [46] |
| Suaeda maritima aqueous leaf extract. 20–60 nm. 5, 10, 15, 20, 25 mg/L. | Spodoptera litura (Lepidoptera: Noctuidae) | LC50 of 20.937 (larval instar I) and 46.896 mg/L (pupa). Reduction in egg incubation (100%) to a concentration of 20 mg/L | Neurosecretory cell inhibition, shrinkage of internal cuticle | [57] |
| Green synthesis: Punica granatum peel extract. 14–28 nm. 3.95, 7.8, 15.6, 31.2, 62.5, 125, 250 μg/mL for cellular lines(SF-21) 10, 25, 50, 75 and 100 μg/mL against larvae. | Spodoptera litura (Lepidoptera: Noctuidae) | LC50 of 31.2 μg/mL. 100% mortality in all larval stages at 100 μg/mL concentration. Reduction in gut microflora (complete inhibition of Klebsiella pneumoniae, Bacillus licheniformis, Bacillus cereus, and Citrobacter freundi) | Reduction in the activity of intestinal enzymes such as amylase, protease, lipase, and invertase. Interacting silver ions with the functional groups of nitrogen bases and phosphate groups in DNA and intracellular proteins. Generation of reactive oxygen species, oxidative stress, membrane disruption, protein unfolding, and inflammation. | [54] |
| Commercial AgNPs. 50–60 nm. 10 mg/mL. | Spodoptera littoralis (Lepidoptera: Noctuidae) | Reduction in larval weight gain and pupal weight. Increase in the number of circulating hemocytes | Increase in Glutathione S-Transferase (GST) enzyme activity. Generation of free radicals and ROS, induction of RNA, and DNA damage. Interference with Juvenile hormone (JH) and ecdysone. | [44] |
| Gamma irradiation with polyvinyl pyrrolidone (PVP). 30 nm. 200 to 600 mg/L. | Spodoptera littoralis (Lepidoptera: Noctuidae) | Low toxicity. The maximum mortalities (11.7 and 11.5%) were recorded at 500 and 600 mg/L of Ag/PVP. | Denaturation of proteins containing sulfur or DNA causes the denaturation of insect enzymes. Alteration of gene expression in the midgut, reducing the gut microflora and also amylase, protease, lipase, and invertase activities was reduced and loss of melanin cuticular pigments. | [45] |
| Green synthesis: Beauveria bassiana, Metarhizium anisopliae, and Isaria fumosorosea. 86.26–257.07 nm. 0.1; 0.3; 0.7; 0.9 and 1.2 mg/mL. | Plutella xylostella (Lepidoptera: Plutellidae) | LC30 = 0.144 mg/mL, LC50 = 0.691 mg/mL, and LC90 = 2.011 mg/mL. | ND | [53] |
| Chemical synthesis (modified Tollens process). 4–24 nm. 10, 20, 40, 60, 80 and 100 mg/L. | Drosophila melanogaster (Diptera: Drosophilidae) | LC100 at 100 mg/L. Slightly reduction in pigmentation of adult flies (10 mg/L) A 50% decrease in the number of hatched individuals, and all the hatched adult flies had highly reduced body pigmentation (20 mg/L). Reduction in larvae development (60, 80, and 100 mg/L) 97% of larvae were dead, and no pupae were formed (100 mg/L) The long-term exposure to AgNPs influenced the fertility of Drosophila during the first three filial generations | Heat shock stress, generation of free oxygen radicals, and apoptosis. Disturbance of metabolic synthesis pathways of biogenic amines and several reproduction hormones (dopamine (DA), octopamine (OA), juvenile hormone (JH), and ecdysteroids (20-hydroxyecdisone (20HE)). Oocyte maturation delays, degradation of early vitellogenic egg chambers, inhibition of yolk protein gene expression in follicle cells, and accumulation of mature oocytes. | [61] |
| Commercial AgNPs. 20 nm. 50 µg/mL. | Drosophila melanogaster (Diptera: Drosophilidae) | Acute and chronic toxicity | Toxicity in HSP70 protein. Interference with Dopamine (DA), Octopamine (OA). | [80] |
| Green synthesis: Olea europaea, Ficus carica, Eriobotrya japonica, Citrus limon, Pistacia vera, Morus nigra leaves extract. 5, 8, 10, 18, 22 nm. 10, 50, 100 and 200 mg/L. | Drosophila melanogaster (Diptera: Drosophilidae) | Reduction in the number of larvae hatched. Mortality on larvae, pupae, and adults. Reduction in larvae longevity | ND | [2] |
| Chemical method with sodium citrate. 1–50 nm. 50 mg/L. | Drosophila melanogaster (Diptera: Drosophilidae) | Loss of melanin production. | Interruption of the activity of copper-dependent cellular enzymes tyrosinase and copper/zinc Superoxide Dismutase (Cu/ZnSOD) | [81] |
| Commercial AgNPs. <60 nm. 0.1, 1, 5, 10 mM. | Drosophila melanogaster (Diptera: Drosophilidae) | Induction of genotoxic activity | Increased expression of p53 protein and Rad51 affecting critical proteins in the repair of double chain breaks inducing mitochondrial dysfunction and oxidative stress | [62] |
| Green synthesis: Manilkara zapota leaf extract 70–140 nm. 2, 4, 6, 8, 10 mg/mL. | Musca domestica (Diptera: Muscidae) | LC50 of 3.64 mg/mL and LC90 of 7.74 mg/mL. 100% of mortality (10 mg/mL) | ND | [63] |
| Solvothermal synthesis. 20–60 nm. 1, 1.5, 2, 2.5 g/kg. | Callosobruchus maculatus (Coleoptera: Chrysomelidae) | LC50 2.06 for adults. LC50 1.00 for larvae. | Generation of reactive oxygen species and oxidative stress | [68] |
| Green synthesis: Polyunsaturated fatty acids (linoleic acid), monounsaturated fatty acids (oleic acid), and saturated fatty acids (stearic acid and palmitic acid). 3–13 nm. ND (AgBox). | Hylotrupes bajulus (Coleoptera: Cerambycidae) | Agsbox-CL exercised mortality of 20–25%. Agsbox-T exercised mortality of 10–15% | ND | [70] |
| Green synthesis: Metarhizium anisopliae, Beauveria bassiana, Verticillium lecanii and Bacillus thuringiensis, Bacillus subtilis culture supernatant. ND. 103, 104, and 105 UFC/mL. | Rhynchophorus ferrugineus (Coleoptera: Curculionidae) | 90% mortality in eggs, 95% in larvae, and 77% in adults | ND | [71] |
| Chemical method. 15–31 nm. 25, 50, 75 y 100 kGy | Sitophilus granarius (Coleoptera: Curculionidae) | 100% mortality for biofilm loaded with 75 kGy of AgNp | ND | [72] |
| Green synthesis: Avicennia marina extract. 15–25 nm. 50, 100, 150, 200 y 250 mg/kg. | Sitophilus oryzae (Coleoptera: Curculionidae) | The sum of all the treatments allowed to obtain a mortality of 100% after 4 d | ND | [73] |
| Green synthesis: Peganum harmala L. seeds alkaloids. 22.5–66.2 nm. 3.6, 7.19, 14.37, 28.74, 57.48, 115.0 µg/cm2. | Trogoderma granarium (Coleoptera: Dermestidae) | LC50 between 23.1 and 32.6 µg/cm2 against larvae and LC50 between 19.6 and 28.4 µg/cm2 against the adult. Malformed larvae and pupae. | Disruption of the endocrine system and hormone balance | [47] |
| Chemical method. ND. 10, 100 y 1000 mg/L. | Tribolium castaneum (Coleoptera: Tenebrionidae), Callosobruchus chinensis (Coleoptera: Chrysomelidae) | LC50 of 15.917 mg/L for T. castaneum and LC50 of 37.6365 C. chinensis. Mortality of 67% for T. castaneum and 73% for C. chinensis. | ND | [74] |
| Solvothermal method. 41–46 nm. 300, 371, 458, 566, and 700 mg/mL | Aphis nerii (Hemiptera: Aphididae) | LC50 of 424.67 mg/mL | ND | [76] |
| Green synthesis: Ziziphus sp. aqueous leaf extract. 44.67 nm. 1000, 2000 and 3000 mg/L | Bemisia tabaci (Hemiptera: Aleyrodoidea) | 100% mortality at 3000 mg/L | Plasma membrane penetration. Biomolecule breakage. Coagulation of proteins and plasma membrane. | [77] |
| Green synthesis: Solanum melongena leaf extract. 75.14 nm. 2.5, 10, 15, 20, 25, 50 and 100 μg/mL | Bemisia tabaci (Hemiptera: Aleyrodoidea) | High mortality at 100 μg/mL | ND | [78] |
| Green synthesis: Beauveria bassiana culture supernatant. 3–25 nm. ND. | Lipaphis erysimi (Hemiptera: Aphididae) | Mortality of 90% | ND | [79] |
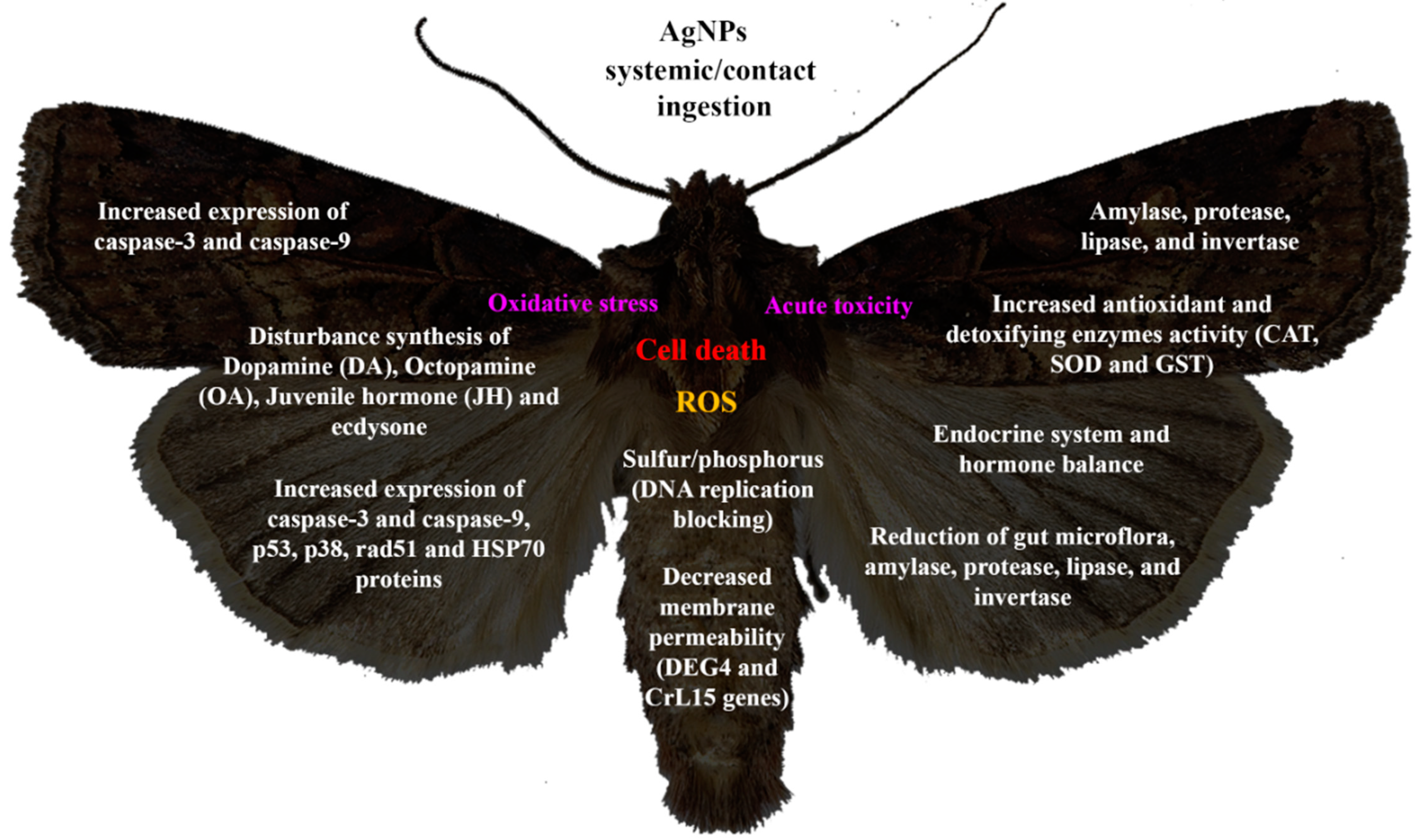
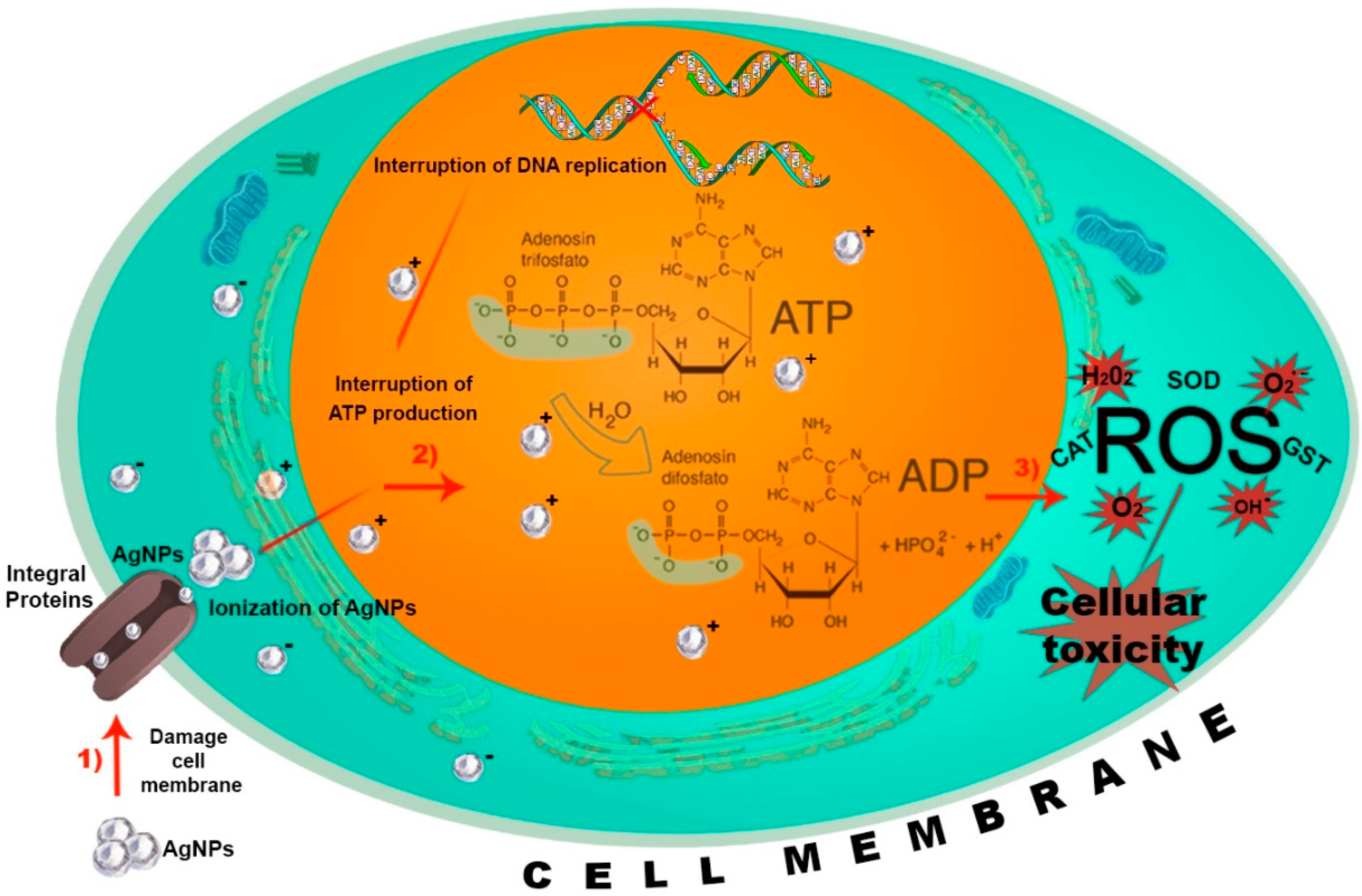
4. AgNP Action Mechanisms on Insects
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jiménez, O.R.; Bornemann, A.C.; Medina, Y.E.; Romero, K.; Bravo, J.R. Prospects of biological inputs as a measure for reducing crop losses caused by climate change effects. J. Agric. Food. Res. 2023, 14, 100689. [Google Scholar] [CrossRef]
- Araj, S.E.; Salem, N.M.; Ghabeish, I.H.; Awwad, A.M. Toxicity of Nanoparticles against Drosophila melanogaster (Diptera: Drosophilidae). J. Nanomater. 2015, 2015, 758132. [Google Scholar] [CrossRef]
- Borah, P.; Biswas, R. Impactful analytical schemes for assessing pesticides in tea: A comprehensive review. Measurement 2023, 221, 113505. [Google Scholar] [CrossRef]
- Vurro, M.; Miguel-Rojas, C.; Pérez, A. Safe nanotechnologies for increasing the effectiveness of environmentally friendly natural agrochemicals. Pest. Manag. Sci. 2019, 75, 2403–2412. [Google Scholar] [CrossRef]
- Iqbal, H.; Jahan, N.; Ali, S.; Shahzad, A.; Iqbal, R. Formulation of Moringa oleifera nanobiopesticides and their evaluation against Tribolium castaneum and Rhyzopertha dominica. JPDP 2024, 131, 133–142. [Google Scholar] [CrossRef]
- Ullah, I.; Toor, M.D.; Basit, A.; Mohamed, H.I.; Gamal, M.; Tanveer, N.A.; Shah, S.T. Nanotechnology: An integrated approach towards agriculture production and environmental stress tolerance in plants. Water Air Soil Pollut. 2023, 234, 666. [Google Scholar] [CrossRef]
- Hosseinzadeh-Bandbafha, H.; Panahi, H.K.S.; Dehhaghi, M.; Orooji, Y.; Shahbeik, H.; Mahian, O.; Karimi-Maleh, H.; Sulaiman, A.; Mei, C.; Kiehbadroudinezhad, M.; et al. Nanomaterials and their role in advancing biodiesel feedstock production: A comprehensive review. Biofuel Res. J. 2023, 10, 1901–1932. [Google Scholar] [CrossRef]
- Servin, A.; Pagano, L.; Castillo-Michel, H.; De la Torre-Roche, R.; Hawthorne, J.; Hernandez-Viezcas, J.A.; Loredo-Portales, R.; Majumdar, S.; Gardea-Torresday, J.; Dhankher, O.P.; et al. Weathering in soil increases nanoparticle CuO bioaccumulation within a terrestrial food chain. Nanotoxicology 2016, 11, 98–111. [Google Scholar] [CrossRef]
- Keller, A.A.; McFerran, S.; Lazareva, A.; Suh, S. Global life cycle releases of engineered nanomaterials. J. Nanopart. Res. 2013, 15, 1692. [Google Scholar] [CrossRef]
- Rizwan, M.; Ali, S.; Qayyum, M.F.; Ok, Y.S.; Adrees, M.; Ibrahim, M.; Abbas, F. Effect of metal and metal oxide nanoparticles on growth and physiology of globally important food crops: A critical review. J. Hazard. Mater. 2017, 322, 2–16. [Google Scholar] [CrossRef]
- Tripathi, D.K.; Singh, S.; Singh, S.; Srivastava, P.K.; Singh, V.P.; Singh, S.; Chauhan, D.K. Nitric oxide alleviates silver nanoparticles (AgNPs)-induced phytotoxicity in Pisum sativum seedlings. Plant Physiol. Biochem. 2017, 110, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Chang, J.; Wang, Z.; Zhang, H.; Wang, T. Advances in transport and toxicity of nanoparticles in plants. J. Nanobiotechnol. 2023, 21, 75. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Kumarasamy, S.; Narayanan, M.; Ranganathan, M.; Rathinavel, T.; Chinnathambi, A.; Alahmadi, T.A.; Karuppusamy, I.; Pugazhendhi, A.; Whangchai, K. Spectral and structure characterization of Ferula assafoetida fabricated silver nanoparticles and evaluation of its cytotoxic, and photocatalytic competence. Environ. Res. 2022, 204, 111987. [Google Scholar] [CrossRef]
- Shrivastava, S.K.; Kulshreshtha, A.; Gangwar, P.K.; Srivastava, S.; Tiwari, A.P.; Singh, S.; Kumar, S.; Parveen, S.; Banerjee, M.; Chaudhary, D.K. Phyto-synthesis of silver nanoparticles from Plumeria pudica leaf extract and its application in anti-cancerous activity. J. Exp. Nanosci. 2023, 18, 2267183. [Google Scholar] [CrossRef]
- Rozina; Ahmad, M.; Asif, S.; Klemes, J.J.; Mubashir, M.; Bokhari, A.; Sultana, S.; Mukhtar, A.; Zafar, M.; Bazmi, A.A.; et al. Conversion of the toxic and hazardous Zanthoxylum armatum seed oil into methyl ester using green and recyclable silver oxide nanoparticles. Fuel 2022, 310, 122296. [Google Scholar] [CrossRef]
- Das, R.K.; Pachapur, V.L.; Lonappan, L.; Naghdi, M.; Pulicharla, R.; Maiti, S.; Cledon, M.; Larios-Martinez, A.D.; Sarma, S.J.; Brar, S.K. Biological synthesis of metallic nanoparticles: Plants, animals and microbial aspects. Nanotechnol. Environ. Eng. 2017, 2, 18. [Google Scholar] [CrossRef]
- Zhao, L.; Lu, L.; Wang, A.; Zhang, H.; Huang, M.; Wu, H.; Xing, B.; Wang, Z.; Ji, R. Nano-Biotechnology in Agriculture: Use of Nanomaterials to Promote Plant Growth and Stress Tolerance. J. Agric. Food. Chem. 2020, 68, 1935–1947. [Google Scholar] [CrossRef]
- Athanassiou, C.G.; Kavallieratos, N.G.; Benelli, G.; Losic, D. Nanoparticles for pest control: Current status and future perspectives. J. Pest Sci. 2018, 91, 1–15. [Google Scholar] [CrossRef]
- Jafir, M.; Irfan, M.; Zia-ur-Rehman, M.; Hafeez, F.; Ahmad, J.N.; Sabir, M.A.; Zulfiqar, U.; Iqbal, R.; Zulfiqar, F.; Moosa, A. The global trend of nanomaterial usage to control the important agricultural arthropod pests: A comprehensive review. Plant Stress 2023, 10, 100208. [Google Scholar] [CrossRef]
- Sadigh-Eteghad, S.; Shahi, S.; Mahmoudi, J.; Farjami, A.; Bazmani, A.; Naghili, B.; Dizaj, S.M.; Salatin, S. Application of Nano-based drug loading systems in the treatment of neurological infections: An updated Review. Curr. Pharm. Des. 2022, 28, 2330–2342. [Google Scholar] [CrossRef]
- Dhanislas, M.; Sampath, S.; Shamya, M.; Joseph, J.; Yasasve, M.; Ahmed, M.Z.; Alqahtani, A.S.; Kazmi, S.; Asaithambi, P.; Suresh, A. Green synthesis of biofabricated silver nanoparticles from Syzygium aromaticum seeds: Spectral characterization and of its anti-mycobacterial activity, cytotoxicity assessment on zebrafish embryo and Artemia salina. Mater. Technol. 2023, 38, 2269358. [Google Scholar] [CrossRef]
- Naderi-Samani, E.; Razavi, R.S.; Nekouee, K.; Naderi-Samani, H. Synthesis of silver nanoparticles for use in conductive inks by chemical reduction method. Heliyon 2023, 9, e20548. [Google Scholar] [CrossRef]
- Rawtani, D.; Khatri, N.; Tyagi, S.; Pandey, G. Nanotechnology-based recent approaches for sensing and remediation of pesticides. J. Environ. Manag. 2018, 206, 749–762. [Google Scholar] [CrossRef]
- Durán, N.; Mercato, P.D.; Durán, M.; Yadav, A.; Gade, A.; Rai, M. Mechanistic aspects in the biogenic synthesis of extracellular metal nanoparticles by peptides, bacteria, fungi, and plants. Appl. Microbiol. Biotechnol. 2011, 90, 1609–1624. [Google Scholar] [CrossRef] [PubMed]
- Bahrulolum, H.; Nooraei, S.; Javanshir, N.; Tarrahimofrad, H.; Mirbagheri, V.; Easton, A.J.; Ahmadian, G. Green synthesis of metal nanoparticles using microorganisms and their application in the agrifood sector. J. Nanobiotechnol. 2021, 19, 86. [Google Scholar] [CrossRef] [PubMed]
- Zor, M.; Baran, M.F.; Sayin, D.N. Rapid synthesis of silver nanoparticles with Rheum ribes L. fruit peels: Anticancer and antimicrobial effects with biocompatible structures. J. Agric. Sci. 2024, 30, 386–399. [Google Scholar] [CrossRef]
- Huang, P.; Fang, W.Y.; Yang, L.; Sun, Y.X.; Yang, H.Y.; Chen, X.Z.; Zeng, H.B. Ultralong hydroxyapatite nanowires-based flow-through reactor with high loading of silver nanoparticles for fast continuous catalytic reduction of organic dyes and disinfection of wastewater. J. Chem. Eng. 2023, 475, 146305. [Google Scholar] [CrossRef]
- Tudi, M.; Daniel-Ruan, H.; Wang, L.; Lyu, J.; Sadler, R.; Connell, D.; Chu, C.; Phung, D.T. Agriculture Development, Pesticide Application and Its Impact on the Environment. Int. J. Environ. Res. Public Health 2021, 18, 1112. [Google Scholar] [CrossRef]
- Tiryaki, O.; Temur, C. The fate of Pesticide in the Environment. J. Biol. Environ. Sci. 2010, 4, 29–38. [Google Scholar]
- Durán, N.; Fávaro, W.J.; Alborés, S.; Da Costa, T.S.; Tasic, L. Biogenic silver nanoparticles capped with proteins: Timed knowledge and perspectives. J. Braz. Chem. Soc. 2023, 34, 897–905. [Google Scholar] [CrossRef]
- Bretado-Aragón, L.; Jiménez-Mejía, R.; López-Meza, J.; Loeza-Lara, P. Compósitos de nanopartículas de plata-quitosano: Una fuente potencial de nuevas terapias antimicrobianas. Rev. Mex. Cienc. Farm. 2017, 47, 7–25. [Google Scholar]
- Pita, W.; Yufisatari, A.; Adi, W. Synthesis of Silver nanoparticles from silver nitrate solution using green tea extract (Camelia sinensis) as bioreductor. JBAT 2017, 6, 32–38. [Google Scholar] [CrossRef]
- Juárez-Maldonado, A.; Ortega-Ortíz, H.; Morales-Díaz, A.; González-Morales, S.; Morelos-Moreno, A.; Cabrera-De la Fuente, M.; Sandoval-Rangel, A.; Cadenas-Pliego, G.; Benavidez-Mendoza, A. Nanoparticles and Nanomaterials as Plant Bioestimulants. Int. J. Mol. Sci. 2019, 20, 162. [Google Scholar] [CrossRef]
- Domínguez-Vera, J.M.; Gálvez, N.; Sánchez-Purificación, A.J.; Trasobares, S.; Hernández, J.C.; Calvino, J.J. Size-Controlled Water-Soluble Ag Nanoparticles. Eur. J. Inorg. Chem. 2007, 2007, 4823–4826. [Google Scholar] [CrossRef]
- Puthukulangara, J.; Kadanthottu, J. Photocatalytic and antioxidant potential of silver nanoparticles biosynthesized using Artemia stelleriana leaf extracts. Water Pract. Technol. 2023, 18, 2664–2674. [Google Scholar] [CrossRef]
- Mishra, V.K.; Husen, A.; Rahman, Q.I.; Iqbal, M.; Sohrab, S.S.; Yassin, M.O. Plant-based fabrication of silver nanoparticles and their application. Nano Plant Pot. 2019, 5, 135–175. [Google Scholar] [CrossRef]
- Thakkar, K.N.; Mhatre, S.S.; Parikh, R.Y. Biological synthesis of metallic nanoparticles. Nanomed. Nanotechnol. 2010, 6, 257–262. [Google Scholar] [CrossRef]
- Kurian, S.; Rajan, A. Characterization and antibacterial response of biosynthesized silver nanoparticles using Eclipta alba (L.) Hassk. leaf extract. Ann. Phytomed. 2023, 12, 453–459. [Google Scholar] [CrossRef]
- Achari, G.; Kowshik, M. Recent Developments on Nanotechnology in Agriculture: Plant Mineral Nutrition, Health, and Interactions with Soil Microflora. J. Agric. Food Chem. 2018, 66, 8647–8661. [Google Scholar] [CrossRef]
- Ahamed, M.; Posgai, R.; Gorey, T.J.; Nielsen, M.; Hussain, S.M.; Rowe, J.J. Silver nanoparticles induced heat shock protein 70, oxidative stress and apoptosis in Drosophila melanogaster. Toxicol. Appl. Pharmacol. 2010, 242, 263–269. [Google Scholar] [CrossRef]
- Manimegalai, T.; Raguvaran, K.; Kalpana, M.; Maheswaran, R. Green synthesis of silver nanoparticle using Leonotis nepetifolia and their toxicity against vector mosquitoes of Aedes aegypti and Culex quinquefasciatus and agricultural pests of Spodoptera litura and Helicoverpa armigera. Environ. Sci. Pollut. Res. 2020, 27, 43103–43116. [Google Scholar] [CrossRef] [PubMed]
- Yasur, J.; Rani, P.U. Lepidopteran insect susceptibility to silver nanoparticles and measurement of changes in their growth, development and physiology. Chemosphere 2015, 124, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Kafel, A.; Nowak, A.; Bembenek, J.; Szczygiel, J.; Nakonieczny, M.; Swiergosz-Kowalewska, R. The localisation of HSP70 and oxidative stress indices in heads Spodoptera exigua larvae in a cadmium-exposed population. Ecotoxicol. Environ. Saf. 2012, 78, 22–27. [Google Scholar] [CrossRef]
- Ahmed, M.; Ali, A.M. Silver and zinc oxide nanoparticles induce developmental and physiological changes in the larval and pupal stages of Spodoptera littoralis (Lepidoptera: Noctuidae). J. Asia-Pac. Entomol. 2018, 21, 1373–1378. [Google Scholar] [CrossRef]
- Ghobashy, M.M.; Sayed, W.A.; El-Helaly, A. Impact of Silver Nanoparticles Synthesized by Irradiated Polyvinylpyrrolidone on Spodoptera littoralis Nucleopolyhedrosis Virus Activity. J. Polym. Environ. 2021, 29, 3364–3374. [Google Scholar] [CrossRef]
- Afrasiabi, Z.; Popham, H.; Stanley, D.; Suresh, D.; Finley, K.; Campbell, J.; Kannan, R.; Upendran, A. Dietary Silver nanoparticles reduce fitness in a beneficial, but not pest, insect species. Arch. Insect Biochem. 2016, 93, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Almadiy, A.A.; Nenaah, G.E.; Shawer, D.M. Facile synthesis of silver nanoparticles using harmala alkaloids and their insecticidal and growth inhibitory activities against the khapra beetle. J. Pest Sci. 2017, 91, 727–737. [Google Scholar] [CrossRef]
- Meng, X.; Abdlli, N.; Wang, N.; Lü, P.; Nie, Z.; Dong, X.; Lu, S.; Chen, K. Effects of Ag Nanoparticles on Growth and Fat Body Proteins in Silkworms (Bombyx mori). Biol. Trace Elem. Res. 2017, 180, 327–337. [Google Scholar] [CrossRef]
- Chen, L.; Meng, X.; Gu, J.; Fan, W.; Abdlli, N.; Peprah, F.; Wang, N.; Zhu, F.; Lü, P.; Ma, S.; et al. Silver nanoparticle toxicity in silkworms: Omics technologies for a mechanistic understanding. Ecotoxicol. Environ. Saf. 2019, 172, 388–395. [Google Scholar] [CrossRef]
- Jin, P.; Chen, J.; Zhan, H.R.; Huang, S.; Wang, J.; Shu, Y. Accumulation and excretion of zinc and their effects on growth and food utilization of Spodoptera litura (Lepidoptera: Noctuidae). Ecotoxicol. Environ. Saf. 2020, 202, 110883. [Google Scholar] [CrossRef]
- Plata-Rueda, A.; de Menezes, C.H.; Cunha, W.D.; Alvarenga, T.M.; Barbosa, B.F.; Zanuncio, J.C.; Martínez, L.C.; Serrao, J.E. Side-effects caused by chlorpyrifos in the velvetbean caterpillar Anticarsia gemmatalis (Lepidoptera: Noctuidae). Chemosphere 2020, 259, 127530. [Google Scholar] [CrossRef]
- Pigino, G.; Migliorini, M.; Paccagnini, E.; Leonzio, C. Fine structure of the midgut and Malpighian papillae in Campodea quilisi (Hexapoda, Diplura) with special reference to the metal composition and physiological significance of midgut intracellular electron-dense granules. Tissue Cell 2005, 37, 223–232. [Google Scholar] [CrossRef]
- Santos, T.S.; de Souza, C.; Sanchez-Lopez, E.; Jain, S.A.; Souto, E.B.; Severino, P.; Mendonça, M. Entomopathogenic Fungi-Mediated AgNPs: Synthesis and Insecticidal Effect against Plutella xylostella (Lepidoptera: Plutellidae). Materials 2022, 15, 7596. [Google Scholar] [CrossRef]
- Arvind, R.S.; Raja, S.K. Biogenic silver nanoparticles mediated stress on developmental period and gut physiology of major lepidopteran pest Spodoptera litura (Fab.) (Lepidoptera: Noctuidae)—An eco-friendly approach of insect pest control. J. Environ. Chem. Eng. 2017, 5, 453–467. [Google Scholar] [CrossRef]
- Sayed, A.M.; Kim, S.; Behle, R.W. Characterization of silver nanoparticles synthesized by Bacillus thuringiensis as a nanobiopesticide for insect pest control. Biocontrol. Sci. Technol. 2017, 27, 1308. [Google Scholar] [CrossRef]
- Pavunraj, M.; Baskar, K.; Duraipandiyan, V.; Al-Dhabi, N.A.; Rajendran, V.; Benelli, G. Toxicity of Ag Nanoparticles Synthesized Using Stearic Acid from Catharanthus roseus Leaf Extract Against Earias vittela and Mosquito Vectors (Culex quinquefasciatus and Aedes aegypti). J. Clust. Sci. 2017, 28, 2477–2492. [Google Scholar] [CrossRef]
- Suresh, U.; Murugan, K.; Panneerselvam, C. Suaeda maritima-based herbal coils and green nanoparticles as potential biopesticides against the dengue vector Aedes aegypti and the tobacco cutworm Spodoptera litura. Physiol. Mol. Plant Pathol. 2017, 101, 225–235. [Google Scholar] [CrossRef]
- Kong, F.; Song, Y.; Zhang, Q.; Wang, Z.; Liu, Y. Sublethal effects of Chlorantraniliprole on Spodoptera litura (Lepidoptera: Noctuidae) Moth: Implication for Attract-and-Kill Strategy. Toxics 2021, 9, 20. [Google Scholar] [CrossRef]
- Ali, S.; Zhang, C.; Wang, L.; Wang, M. Toxicity of chlorantraniliprole and emamectin benzoate against Trichoplusia ni and other Lepidoptera species. J. Econ. Entomol. 2018, 111, 2298–2304. [Google Scholar] [CrossRef]
- Zhao, S.; Yang, F.; Yang, S.; Wu, Y. Susceptibility of Plutella xylostella to spinosad and indoxacarb: Dose-response and effects on detoxification enzyme activities. Pestic. Biochem. Physiol. 2020, 167, 104581. [Google Scholar] [CrossRef]
- Panacek, A.; Prucek, R.; Safarova, D.; Dittrich, M.; Richtrova, J.; Benickova, K.; Zboril, R.; Kvitek, L. Acute and chronic toxicity effects of silver nanoparticles (NPs) on Drosophila melanogaster. Environ. Sci. Technol. 2011, 45, 4974–4979. [Google Scholar] [CrossRef] [PubMed]
- Demir, E.R.; Valdes, G.; Kaya, B.; Creus, A.; Marcos, R. Genotoxic analysis of silver nanoparticles in Drosophila. Nanotoxicology 2011, 5, 417–424. [Google Scholar] [CrossRef]
- Kamaraj, C.; Rajakumar, G.; Rahuman, A.A. Feeding deterrent activity of synthesized silver nanoparticles using Manilkara zapota leaf extract against the house fly, Musca domestica (Diptera: Muscidae). Parasitol. Res. 2012, 111, 2439–2448. [Google Scholar] [CrossRef]
- Ahmed, M.; Vogel, C.F. Hazardous effects of octopamine receptor agonists on altering metabolism-related genes and behavior of Drosophila melanogaster. Chemosphere 2020, 253, 126629. [Google Scholar] [CrossRef]
- Martelli, F.; Zhongyuan, Z.; Wang, J. Low doses of the neonicotinoid insecticide imidacloprid induce ROS triggering neurological and metabolic impairments in Drosophila. Proc. Natl. Acad. Sci. USA 2020, 117, 25840–25850. [Google Scholar] [CrossRef]
- Saini, S.; Rani, L.; Shukla, N.; Banerjee, M.; Chowdhuri, D.K.; Gautam, N.K. Development of a Drosophila melanogaster based model for the assessment of cadmium and mercury mediated renal tubular toxicity. Ecotoxicol. Environ. Saf. 2020, 201, 110811. [Google Scholar] [CrossRef] [PubMed]
- Nas, B.; Colak, D.A. Genotoxic análisis of nickel-iron oxide in Drosophila. Toxicol. Ind. Health 2020, 36, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Rouhani, M.; Samih, M.A.; Kalantari, S. Insecticidal effect of silica and silver nanoparticles on the cowpea seed beetle, Callosobruchus maculatus F. (Col.: Bruchidae). J. Entomol. Res. 2012, 4, 297–305. [Google Scholar]
- Ojo, D.O.; Omotoso, O.T.; Obembe, O.M.; Odeteye, A.A. Effects of Cleistopholis patens (Benth) extracts on cowpea seed beetle, Callosobruchus maculatus (Fab.) (Coleoptera: Chrysomelidae) infesting cowpea seeds in storage. Int. J. Trop. Insect Sci. 2020, 41, 209–216. [Google Scholar] [CrossRef]
- Can, A.; Palanti, S.; Sivrikaya, H.; Hazer, B.; Stefani, F. Physical, biological and chemical characterisation of Wood treated with silver nanoparticles. Cellulose 2018, 26, 5075–5084. [Google Scholar] [CrossRef]
- Abdel-Raheem, M.A.; Reyad, N.F.; Alghamdi, H.A. Virulence of Nanoparticle preparation of Entomopathogenic fungi and Entomopathogenic Bacteria against red palm weevil Rhynchophorus ferrugineus (Olivier) (Coleoptera: Curculionidae). Rom. Biotechnol. Lett. 2019, 25, 1151–1159. [Google Scholar] [CrossRef]
- Eyssa, H.M.; Sawires, S.G.; Senna, M. Gamma irratiation of polyethylene nanocomposites for food packaging applications against stored-product insect pests. J. Vinyl. Addit. Technol. 2018, 25, E120–E129. [Google Scholar] [CrossRef]
- Sankar, M.V.; Abideen, S. Pesticidal effect of Green synthesized silver and lead nanoparticles using Avicennia marina against grain storage pest Sitophilus oryzae. J. Nanomater. Biostruct. 2015, 5, 32–39. [Google Scholar]
- Ahmad, S.; Rashid, H.; Jalil, Q.; Munir, S.; Barkatullah; Khan, S.; Ullah, R.; Shahat, A.A.; Mahmood, H.M.; Abdullah A-Mishari, A.A.N. Polymers encapsulated aspirin loaded silver oxide nanoparticles: Synthesis, Characterization and its Bio-Applications. Sains Malays. 2019, 48, 1887–1897. [Google Scholar] [CrossRef]
- Mantzoukas, S.; Korbou, G.; Magita, A.; Eliopoulos, P.A.; Poulas, K. Leguminous seeds powder diet reduces the survival and development of the Khapra beetle, Trogoderma granarium Everts (Coleoptera: Dermestidae). Biology 2020, 9, 204. [Google Scholar] [CrossRef]
- Rouhani, M.; Samih, M.A.; Kalantari, S. Insecticide effect of silver and zinc nanoparticles against Aphis nerii Boye de fonscolombe (Hemiptera: Aphididae). Chil. J. Agric. Res. 2012, 72, 590–594. [Google Scholar] [CrossRef]
- Al Shammari, H.I.; Al-Khazraji, H.I.; Falih, S.K. The effectivity of silver nanoparticles prepared by Jujube ziziphus sp. Extract against whitefly Bemisia tabaci Nymphs. Res. J. Pharm. Biol. Chem. Sci. 2018, 9, 551. [Google Scholar]
- Pushparaj, K.; Balasubramanian, B.; Kandasamy, Y.; Arumugam, V.A.; Kaliannan, D.; Arumugam, M.; Alodaini, H.A.; Hatamleh, A.A.; Pappuswamy, M.; Meyyazhagan, A. Green synthesis, characterization of silver nanoparticles using aqueous leaf extracts of Solanum melongena and in vitro evaluation of antibacterial, pesticidal and anticancer activity in human MDA-MB-231 breast cancer cell lines. J. King Saud Univ. Sci. 2023, 35, 102663. [Google Scholar] [CrossRef]
- Kamil, D.; PramelaDevi, S.; Ganesh, P. Green synthesis of silver nanoparticles by entomopathogenic fungus Beauveria bassiana and their bioefficacy against mustard aphid (Lipaphis erysimi Kalt.). Indian. J. Exp. Biol. 2017, 55, 555–561. [Google Scholar]
- Posgai, R.; Ahamed, M.; Hussain, S.M.; Rowe, J.J.; Nielsen, M.G. Inhalation method for delivery of nanoparticles to the Drosophila respiratory system for toxicity testing. Sci. Total Environ. 2009, 408, 439–443. [Google Scholar] [CrossRef]
- Armstrong, N.; Ramamoorthy, M.; Lyon, D.; Jones, K.; Duttaroy, A. Mechanism of Silver Nanoparticles Action on Insect Pigmentation Reveals Intervention of Copper Homeostasis. PLoS ONE 2013, 8, e53186. [Google Scholar] [CrossRef] [PubMed]
- Bihal, R.; Al-Khayri, J.M.; Kudesia, N.; Ahmed, F.K.; Sarkar, R.; Arora, A.; Abd-Elsalam, K.A. Entomopathogenic Fungi: An Eco-Friendly synthesis of sustainable nanoparticles and their nanopesticide properties. Microorganisms 2023, 11, 1617. [Google Scholar] [CrossRef] [PubMed]
- Benelli, G.; Maggi, F.; Romano, D.; Stefanini, C.; Vaseeharan, B.; Kumar, S.; Higuchi, A.; Alarfaj, A.A.; Mehlorn, H.; Canale, A. Nanoparticles as effective acaricides against ticks—A review. Ticks. Tick. Borne Dis. 2017, 8, 821–826. [Google Scholar] [CrossRef]
- Danyue, N.; Jiaqiao, L.; Qinghua, X.; Lele, A.; Changqiang, Z.; Yifan, W.; Qiyuan, G.; Lingling, Z.; Weilong, T. Nanoparticles: A potential and effective method to control insect-borne diseases. Bioinorg. Chem. 2023, 2023, 5898160. [Google Scholar] [CrossRef]
- Benelli, G. Mode of action of nanoparticles against insects. Environ. Sci. Pollut. Res. 2018, 5, 12329–12341. [Google Scholar] [CrossRef]
- Nosál, E.; Reinprecht, L. Preparation and application of silver and zinc oxide nanoparticles in wood industry: The review. Acta Fac. Xylologiae 2018, 60, 5–23. [Google Scholar] [CrossRef]
- Chandrasekaran, R.; Seetharaman, P.; Krishnan, M.; Gnanasekar, S.; Sivaperumal, S. Carica papaya (Papaya) latex: A new paradigm to combat against dengue and filariasis vectors Aedes aegypti and Culex quinquefasciatus (Diptera: Culicidae). 3 Biotech. 2018, 8, 83. [Google Scholar] [CrossRef]
- Nair, P.M.G.; Park, S.Y.; Lee, S.W.; Choi, J. Differential expression of ribosomal protein gene, gonadotrophin releasing hormone gene and Balbiani ring protein gene in silver nanoparticles exposed Chironomus riparius. Aquat. Toxicol. 2011, 101, 31–37. [Google Scholar] [CrossRef]
- Nair, P.M.G.; Choi, J. Identification, characterization and expression profiles of Chironomus riparius glutathione S-transferase (GST) genes in response to cadmium and silver nanoparticles exposure. Aquat. Toxicol. 2011, 101, 550–560. [Google Scholar] [CrossRef]
- Durán, N.; Durán, M.; Souza, C.E.D. Silver and silver chloride nanoparticles and their anti-tick activity: A mini review. J. Braz. Chem. Soc. 2017, 28, 927–935. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant. Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Avalos, A.; Haza, A.I.; Drosopoulou, E.; Mavragani-Tsipidou, P.; Morales, P. In vivo genotoxicity assessment of silver nanoparticles of different sizes by the Somatic Mutation and Recombination Test (SMART) on Drosophila. Food Chem. Toxicol. 2015, 85, 114–119. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Cisterna, D.; Rubilar, O.; Tortella, G.; Chen, L.; Chacón-Fuentes, M.; Lizama, M.; Parra, P.; Bardehle, L. Silver Nanoparticles as a Potent Nanopesticide: Toxic Effects and Action Mechanisms on Pest Insects of Agricultural Importance—A Review. Molecules 2024, 29, 5520. https://doi.org/10.3390/molecules29235520
Martínez-Cisterna D, Rubilar O, Tortella G, Chen L, Chacón-Fuentes M, Lizama M, Parra P, Bardehle L. Silver Nanoparticles as a Potent Nanopesticide: Toxic Effects and Action Mechanisms on Pest Insects of Agricultural Importance—A Review. Molecules. 2024; 29(23):5520. https://doi.org/10.3390/molecules29235520
Chicago/Turabian StyleMartínez-Cisterna, Daniel, Olga Rubilar, Gonzalo Tortella, Lingyun Chen, Manuel Chacón-Fuentes, Marcelo Lizama, Pablo Parra, and Leonardo Bardehle. 2024. "Silver Nanoparticles as a Potent Nanopesticide: Toxic Effects and Action Mechanisms on Pest Insects of Agricultural Importance—A Review" Molecules 29, no. 23: 5520. https://doi.org/10.3390/molecules29235520
APA StyleMartínez-Cisterna, D., Rubilar, O., Tortella, G., Chen, L., Chacón-Fuentes, M., Lizama, M., Parra, P., & Bardehle, L. (2024). Silver Nanoparticles as a Potent Nanopesticide: Toxic Effects and Action Mechanisms on Pest Insects of Agricultural Importance—A Review. Molecules, 29(23), 5520. https://doi.org/10.3390/molecules29235520










