1. Introduction
While they are not experimental observables, molecular orbitals are a conceptually useful and elegant tool for elucidating molecular properties [
1]. They have long been a significant tool in the arsenal of chemists [
2,
3,
4,
5,
6], tracing back to the early work of Hückel, Mulliken, and others. One prominent example is the principle of conservation of orbital symmetry [
7,
8,
9], which subsumes the Woodward–Hoffmann rules. Helical frontier molecular orbitals, first introduced by Hendon et al. [
10] in 2013, have seeded a surge of interest. Helical frontier molecular orbitals appear in disubstituted allenes and even-
n cumulenes. Later, many more types of molecules possessing this interesting property were reported [
11,
12,
13,
14,
15,
16,
17,
18,
19,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29,
30], and it was discovered that molecules with helical orbitals have interesting physicochemical properties. For example, oligoyne-bridged bifluorenes can induce spin–orbit coupling [
30].
Boron forms clusters with unique bonding, aromaticity, and reactivity properties [
31,
32,
33]. Very recently [
34], for the first time, we observed helical spin densities of anionic boron clusters. In this work, we report that
[
35],
[
36],
[
37], and
[
38] (see
Scheme 1) not only have helical molecular orbitals, but also helical spin densities. This is interesting because, unlike molecular orbitals, spin densities are experimental observables, and this allows the edifice of (spin)-resolved (conceptual) density-functional theory [
39,
40,
41,
42,
43,
44,
45,
46,
47,
48,
49,
50] to be directly applied to these compounds.
To pin down the origin of spin-density helicity, we forced the quasi-planar boron atoms to be exactly in a plane (where we simply set a column of Cartesian coordinates to zero): helical spin densities are no longer observed. Thus, it is chiral Jahn–Teller distortion that governs the formation of helical spin densities. This seems to be the first observation of this intriguing phenomenon in inorganic boron clusters. We also show that the elongation or enlargement of helical molecular orbitals can be achieved by simply adding more structural motifs via a linker. Moreover, we have exhibited that helical-shape molecules have a large propensity to assume helical molecular orbitals as shown in inorganic species
[
51], Be
6 [
52], and
[
53] (vide infra).
2. Results
To characterize the planarity of anionic boron clusters (in both the ground and excited state), we used a few parameters [
54], including the molecular planarity parameter (MPP), span of deviation from the plane (SDP), and maximum positive/negative deviation (MPD/MND) to the fitted plane, as listed in
Table 1. The definitions of MPP, SDP, MPD, and MND are given in
Section 4. The fitted parameters of a plane are listed in
Table S1. Of note, in this work, the planarity of a molecule is a geometric concept. Exact planarity means that all the atoms lie in a plane, simply like a benzene molecule. Quasi-planarity indicates that one or more than one atom lie slightly above/below a plane. One can easily discover that all the systems studied in this work are quasi-planar. Based upon the optimized structures in the ground state, we have observed helical spin densities as exhibited in
Figure 1. No similar results are discerned for the excited-state structures (see
Figure S1).
To elucidate the origin of helical spin densities, we force the ground-state quasi-planar boron structures to be exactly planar followed by single-point calculations at the PBE0/6-311+G(d) [
56,
57] level. The spin densities’ helicity then vanishes (see
Figure S2). Accordingly, a chiral Jahn–Teller distortion plays a key role where the right- and left-handed deformations are (quasi)equal in energy, and the planar structure deforms slightly to break symmetry, thus lowering in energy. More intriguingly, these chiral structures [in terms of vibrational circular dichroism (VCD) spectra; see
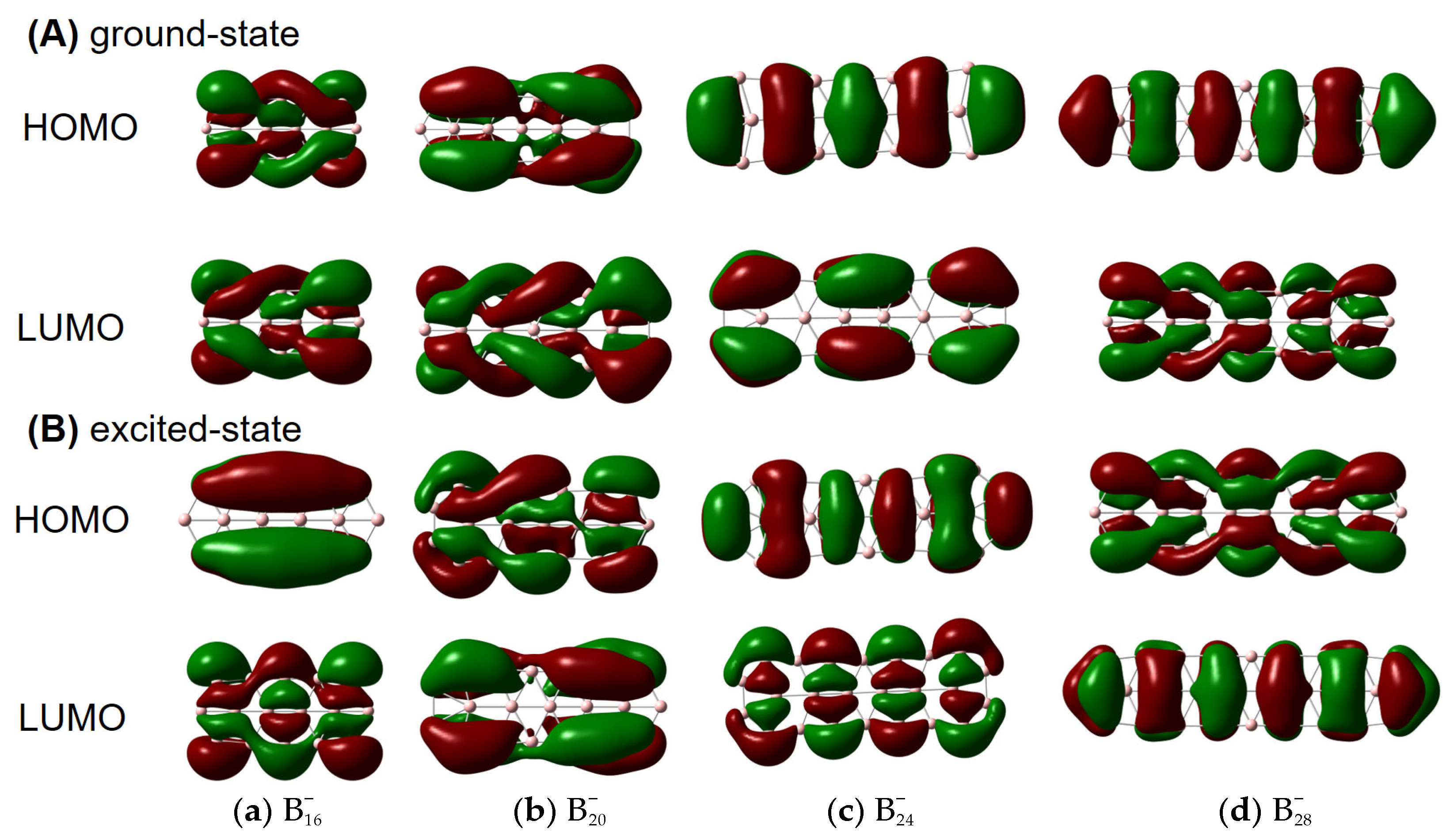
Figure 2 for details] can also have helical frontier molecular orbitals as shown in
Figure 3. Of note, excited-state VCD spectra are also observed as shown in
Figure S3. In the ground state,
,
and
(but not
) have helical
β–LUMOs (with lower orbital energies than their
α counterparts) and
and
also have helical
β–HOMOs in
Figure 3; in the excited state, only
and
have helical
β–HOMOs. Then, how to understand such a phenomenon?
Our results seem to be specific to π-electron deficient (Be, B) and abundant (P, As) structures, where buckling evidently occurs to accommodate a slight contamination with chirality-supporting sp
3 hybridization. Presumably the difference in these different clusters comes from a delicate balance between a preference for Hückel-esque orbitals and chiral orbitals, which in turn probably reflects electron correlation. We note that the boron cluster with achiral HOMO and LUMO (
) is the least strongly correlated, suggesting that near-degeneracy of the valence orbitals is important for induced chirality. More specifically, the chirality seems to be induced by the type of strong electron correlation that can be modelled with spin-symmetry breaking, which is reflected in the fact that the
α and
β HOMO and HOMO-1 energies differ quite significantly in
and
, but much less in
. In addition,
has a remarkably large gap between the low-energy
β-LUMO and the higher-energy
α-LUMO, which perhaps explains its exceptional behavior in
Figure 3A.
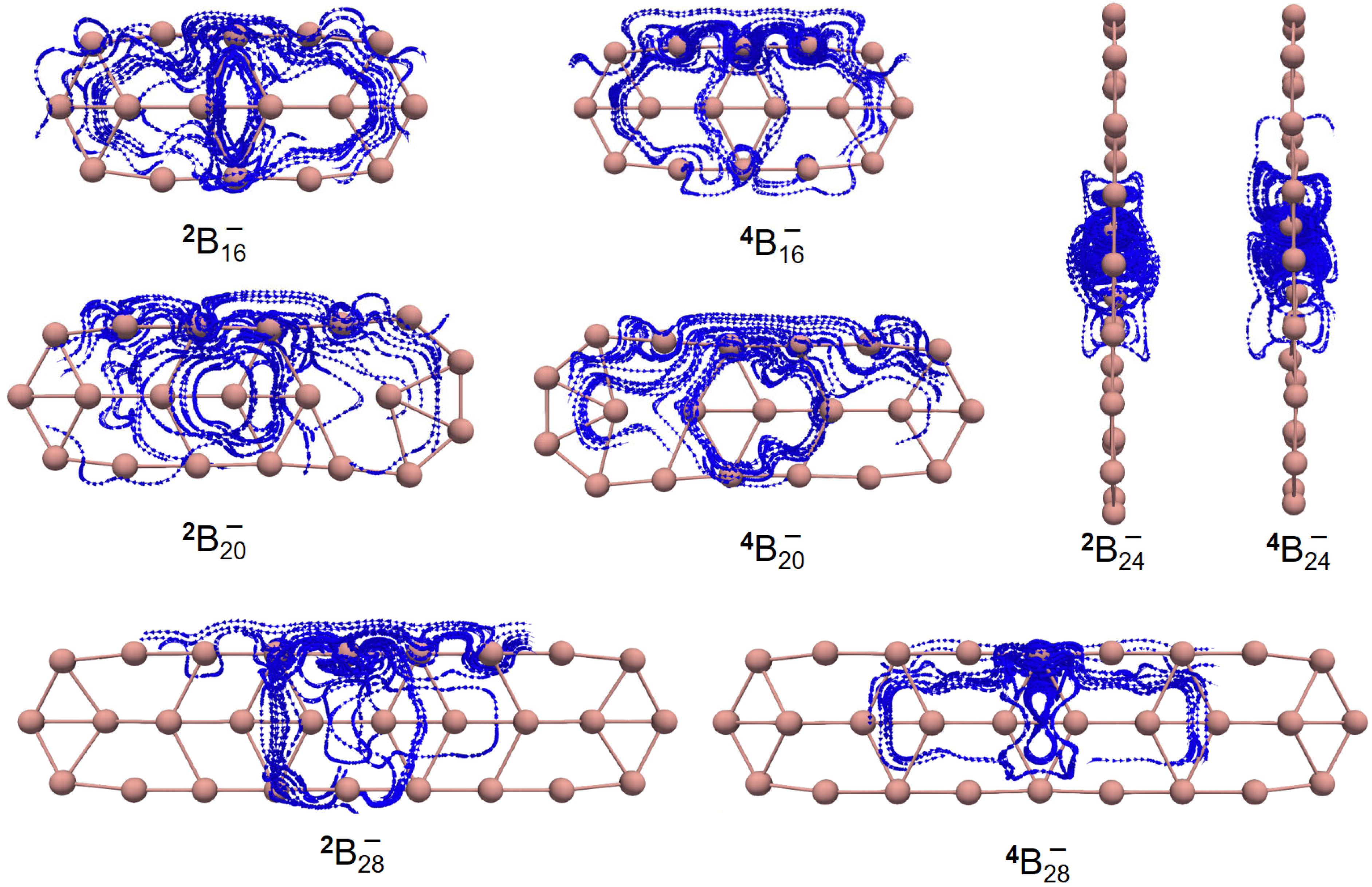
Figure 4 shows the GIMIC (gauge-including magnetically induced current) [
59,
60] distributions of both ground- and excited-state
,
,
and
. For
2 and
4, the induced electric currents are running counter-clockwise, which is indicative of aromaticity, as evidenced by the negative NICS (nucleus-independent chemical shift) [
61] values as shown in
Table 2. Similar results are observed for
and
. However, this is not the case for
. The overall effect indicates that
is antiaromatic while the
Z-component of the induced electric current also runs in a counter-clockwise manner as showcased by the NICS
ZZ values in
Table 2. Yet, the dominant contributions of the induced electric current lie in the
x–
y-plane, which is the source of antiaromaticity. To go a step further,
Figure 5 showcases the 3D isotropic shielding surface (ICSS) [
62] calculations for both ground- and excited-state boron clusters and it is clearly revealed that there exists a strongly shielded chemical environment along the direction perpendicular to the quasi-planar boron clusters.
Hyperfine coupling constants [
64,
65] provide a direct experimental measure of the distribution of unpaired spin density in paramagnetic molecules. The interactions of unpaired electrons with external magnetic fields arise from the Zeeman effect and from the hyperfine coupling with nuclei having nonzero spins. The latter contribution is related to the chemical environment. For each nucleus
of a molecule located at
, the isotropic component of the hyperfine interaction tensor,
, is related to the local spin density through [
66]
where
,
, and
are the electronic and nuclear magnetons and the nuclear magnetogiric ratio, the indices
and
run over the basis functions,
is the difference between the density matrices of spin
and spin
electrons, and
is the Dirac delta function. Therefore, once the density matrices for different spins have been determined, the calculation of
for each nucleus is achieved in a straightforward way. The (isotropic) hyperfine coupling tensor,
, consists of the Fermi contact term (
) and a spin orbit correction, the pseudocontact term (
).
Shown in
Table 3 are the isotropic NMR shielding (α
iso) constants and hyperfine coupling (
Aiso) constants for both ground- and excited-state
at the PBE0/pcJ-2 [
67,
68] level. It is clearly shown that the 16 boron atoms can be roughly grouped into 5 different atoms in different chemical environments as evidenced by both the α
iso and
Aiso data. Among all the boron atoms, one can easily see that atoms 5 and 6 (as shown in
Scheme 1), lying at the two ends of the middle line composed of atoms 1–6, are the most unique. For example, they have the least positive α
iso values and the most negative
Aiso data. In addition, they undergo the largest changes when going from the ground state to the excited state. Specifically,
Aiso changes by ~18 MHz while the largest change of the other atoms is ~8 MHz. Similar trends can be observed for
,
, and
as shown in
Table 4,
Table 5 and
Table 6.
3. Discussion
Helical frontier molecular orbitals were reported first for hydrocarbon systems, then also for boron-containing molecules [
10]. In this work, we have also observed similar results for quasi-planar boron clusters. In addition, as shown in
Figure 6, for some other helical (inorganic) motifs,
, Be
6 (a B
11 helical structure plus a distorted prism of Be
6), and
, helical frontier molecular orbitals are also observed. Is this helicity a ubiquitous phenomenon or a special feature of some elements in special molecular topologies? This seems to be an open question, and will be a topic for future research.
While a systematic rule for designing molecular templates with helical spin densities is unknown to us, we can show how to elongate the helical frontier molecular orbitals from a given template structure. For example, starting from one conjugated hydrocarbon molecule
1 with helical frontier molecular orbitals, combining two monomers of
1 and a linker, such as CH
2(
1)
2, [NH
2(
1)
2]
+, and [OH(
1)
2]
+, leads to elongated or enlarged helical frontier molecular orbitals as shown in
Figure 7. Yet, when three or four monomers of
1 are grouped together, such as CH(
1)
3 or C(
1)
4, the helical frontier molecular orbitals are no longer elongated (results not shown). Is it possible to generate an infinite chain of (
1)
∞? We do not know; possibly other linkers would work better. For the anionic boron clusters, we failed to even generate a dimer of
; this is presumably because the repulsion between the anionic monomers prevents electron delocalization between them.
Finally, we have to point out that in a broader sense, the dissection of chiral boron clusters and the electron spin should be beneficial to its applications to chiral spintronics and materials [
69,
70,
71].
4. Materials and Methods
To obtain the MPP or SDP values for a molecule, the least square method is used to generate a fitted plane by all the atoms considered. First, one can have a coordinate matrix whose dimension is 3 × Natom. After subtracting out the geometry center, one can easily perform a singular value decomposition for this matrix. To obtain the coefficients (A, B, and C) and constant (D) for a fitting plane Ax + By + Cz + D = 0, one can set A = u1, B = u2, C = u3 to be the left singular vectors corresponding to the least singular values. The constant D is determined to be −(u1xc + u2yc + u3zc) if the fitting plane passes through the geometry center (xc, yc, zc) of a molecule. The molecular planarity parameter (MPP) can be readily calculated as the root-mean-squared deviation (RMSD) of the atoms from the fitting plane, , where is the distance between an atom i and the fitting plane, and it can be easily evaluated as . The signed distance of an atom i to the fitting plane is defined as ; then, the span of deviation from the plane (SDP) can be calculated as , where / denote the most positive/negative values of among all considered atoms, respectively.
For all the molecular systems, structure optimization was performed at the density functional theory (DFT) [
72,
73] PBE0/6-311+G(d) level. Stability of molecular wavefunctions was confirmed via keywords of “guess = mix” and “stable = opt” in Gaussian 16 [
74]. Vibrational frequency calculations were ensued to make sure that all the structures were true local minima on the potential energy surface. The optimized atomic Cartesian coordinates are supplied in the
Supplementary Materials. Multireference (MR) characteristics of all boron clusters were checked via the T1 diagnostics [
75] regarding the coupled cluster theory with single and double substitutions [CCSD/6-311+G(d)] and the frozen core formalism was used for CCSD calculations. The reported values for
,
,
, and
are 0.0365, 0.0495, 0.0344, and 0.0411 for the ground state, and 0.0414, 0.0408, 0.0398, and 0.0367 for the excited state, indicative of non-negligible multi-reference characteristics (because their T1 > 0.02). For the ground state, we also employed a larger basis set cc-pVTZ [
76] and very close results were obtained for T1, 0.0360, 0.0488, 0.0344, and 0.0415, respectively. However, for radicals, somewhat larger T1 values (~0.03) are acceptable, and the usual thresholds for T1 are based on accurate quantitative recovery of the dynamic correlation energy, rather than qualitative considerations like those we consider here. The T1 values we report are consistent with the coefficient of the leading determinant being greater than 0.90, implying that single-determinant methods (DFT) and single-reference methods (UCCSD) are good enough to elucidate qualitative features of the boron clusters.
In addition, to force all the atoms to be exactly in a plane, we simply set, for example, the z-components of all the boron atoms to be zero if the original quasi-planar boron cluster is lying in a xy-plane.
To further analyze the aromaticity properties of boron clusters, we employed PBE0/pcJ-2 to calculate the global NICS (nucleus-independent chemical shift) values and GIMIC (gauge-including magnetically induced current) distributions. NMR chemical shielding constants and isotropic hyperfine coupling parameters were obtained at the PBE0/pcJ-2 level with default gauge-including atomic orbitals (GIAOs) [
77,
78,
79,
80,
81]. The global NICS value is obtained at the geometric center, denoted as NICS(0), and its
z-axis component as NICS(0)
ZZ. Moreover, another two points (1 Å away from a global center) are also considered and they are signified as NICS(1) and NICS(−1), with their
z-axis components denoted as NICS(1)
ZZ and NICS(−1)
ZZ.
All DFT calculations were performed by using the Gaussian 16 package with tight self-consistent field (SCF) convergence criteria and ultrafine integration grids to ensure good accuracy. Multiwfn 3.8 [
82] software was used to analyze the planarity of boron clusters and prepare the ICSS input files.