Preclinical Evaluation of 2-Aminobenzothiazole Derivatives: In Silico, In Vitro, and Preliminary In Vivo Studies as Diabetic Treatments and Their Complications
Abstract
1. Introduction
2. Results and Discussion
2.1. In Silico
2.1.1. Physicochemical Properties, Pharmacokinetic and Toxicologic Profile
| Physicochemical Properties | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Lipinski’s Rule of Five | Aqueous Solubility | |||||||||
| MW | cLogP | nON | nOHNH | nrot | TPSA | logS | [mg/mL] | Class | MR | |
| 5d | 371.49 | 2.37 | 5 | 2 | 7 | 74.6 | −5.09 | 3.05 × 10−3 | III | 104.71 |
| 8c | 371.49 | 4.23 | 5 | 1 | 7 | 63.6 | −5.11 | 2.92 × 10−3 | III | 104.22 |
| 8d | 385.51 | 4.44 | 5 | 1 | 8 | 63.6 | −5.29 | 1.97 × 10−3 | III | 109.03 |
| 12d | 384.53 | 3.69 | 5 | 2 | 7 | 66.4 | −4.93 | 4.55 × 10−3 | III | 110.75 |
| Zop | 419.38 | 3.54 | 6 | 1 | 5 | 85.1 | −4.83 | 6.27 × 10−3 | III | 101.48 |
| Epa | 319.41 | 2.44 | 4 | 1 | 4 | 59.30 | −4.19 | 2.08 × 10−2 | II | 91.76 |
| PGZ | 356.44 | 3.09 | 7 | 4 | 1 | 93.59 | −4.31 | 1.76 × 10−2 | III | 102.59 |
| Pharmacokinetic | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| BBB | P-gp | CYP450 Inhibitor | %ABS | HIA | |||||
| 1A2 | 2C19 | 2C9 | 2D6 | 3A4 | |||||
| 5d | √ | × | √ | √ | √ | × | × | 83.27 | 98.24 |
| 8c | √ | × | √ | √ | √ | × | × | 87.06 | 98.17 |
| 8d | √ | × | √ | √ | √ | × | × | 87.06 | 98.06 |
| 12d | × | × | × | × | × | × | × | 86.10 | 95.75 |
| Epa | × | × | √ | √ | √ | √ | √ | 79.64 | 97.53 |
| Zop | × | × | √ | √ | √ | √ | √ | 88.54 | 99.52 |
| PGZ | × | × | √ | √ | √ | √ | √ | 76.71 | 97.35 |
2.1.2. Molecular Docking
| ALR2 (4JIR *) | PPAR-γ (2FVJ **) | |||||||
|---|---|---|---|---|---|---|---|---|
| ΔG (Kcal/mol) | Ki (µM) | #IT | #I-ABP | ΔG (Kcal/mol) | Ki (µM) | #IT | #I-LBD | |
| 5d | −7.98 | 0.603 | 11 | 1 | −8.45 | 0.338 | 18 | 6 |
| 8b | −7.29 | 1.770 | 8 | 0 | −6.48 | 15.230 | 11 | 4 |
| 8c | −8.14 | 0.379 | 9 | 2 | −8.80 | 0.122 | 19 | 0 |
| 8d | −8.39 | 0.262 | 13 | 1 | −7.77 | 0.550 | 16 | 3 |
| 11d | −8.75 | 0.162 | 17 | 5 | −7.65 | 1.140 | 18 | 8 |
| 12d | −8.62 | 0.171 | 16 | 3 | −7.97 | 0.541 | 17 | 4 |
| Epa | −8.50 | 0.359 | 23 | 7 | −7.79 | 1.490 | 15 | 5 |
| Zop | −8.40 | 0.328 | 19 | 9 | −8.08 | 0.511 | 23 | 5 |
| PGZ | −9.38 | 0.067 | 8 | 6 | −9.48 | 0.099 | 11 | 4 |
2.2. Chemistry
2.3. In Vitro
2.3.1. Inhibitory Activity Against ALR2
2.3.2. Antioxidant Capacity
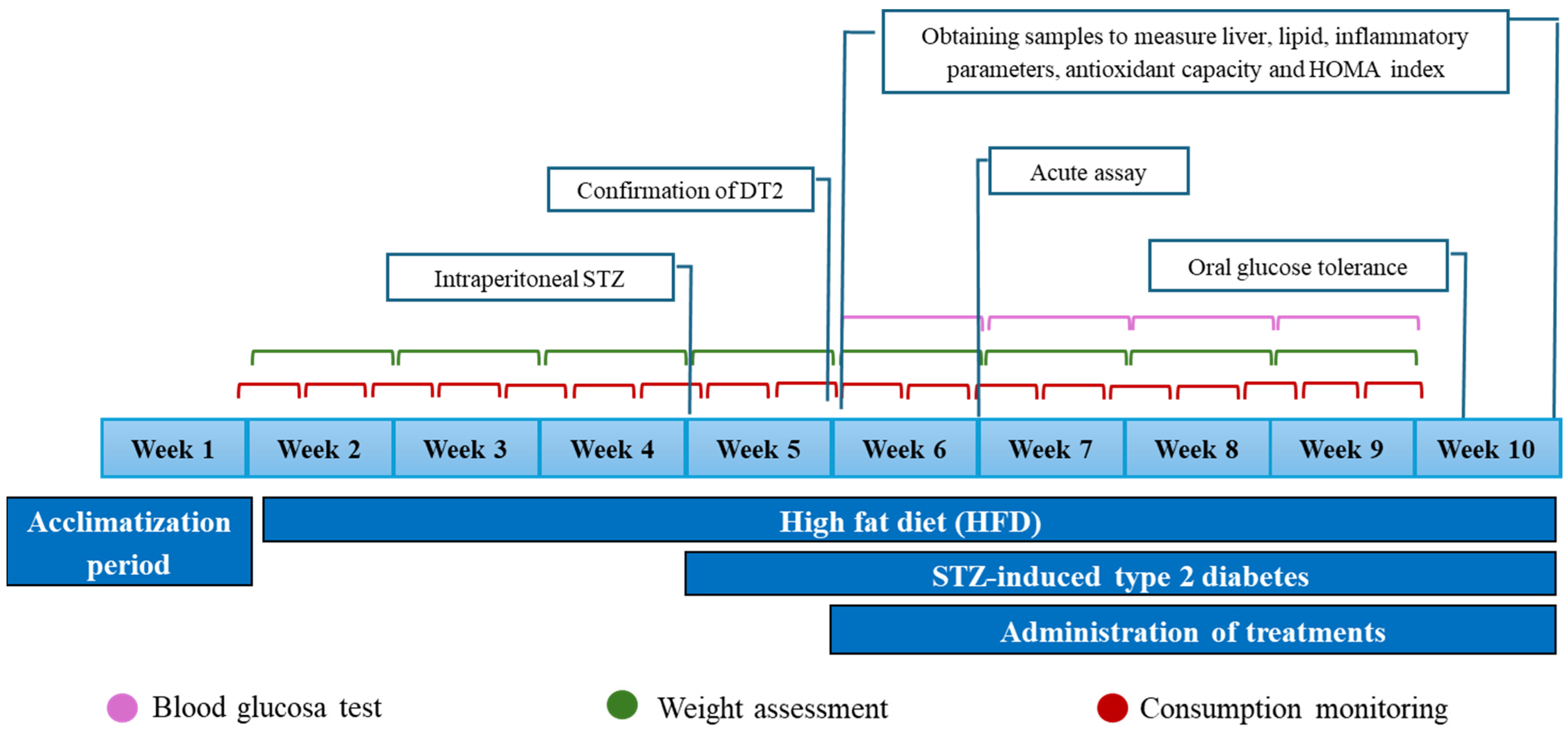
2.4. In Vivo
2.4.1. Acute Oral Toxicity (AOT)
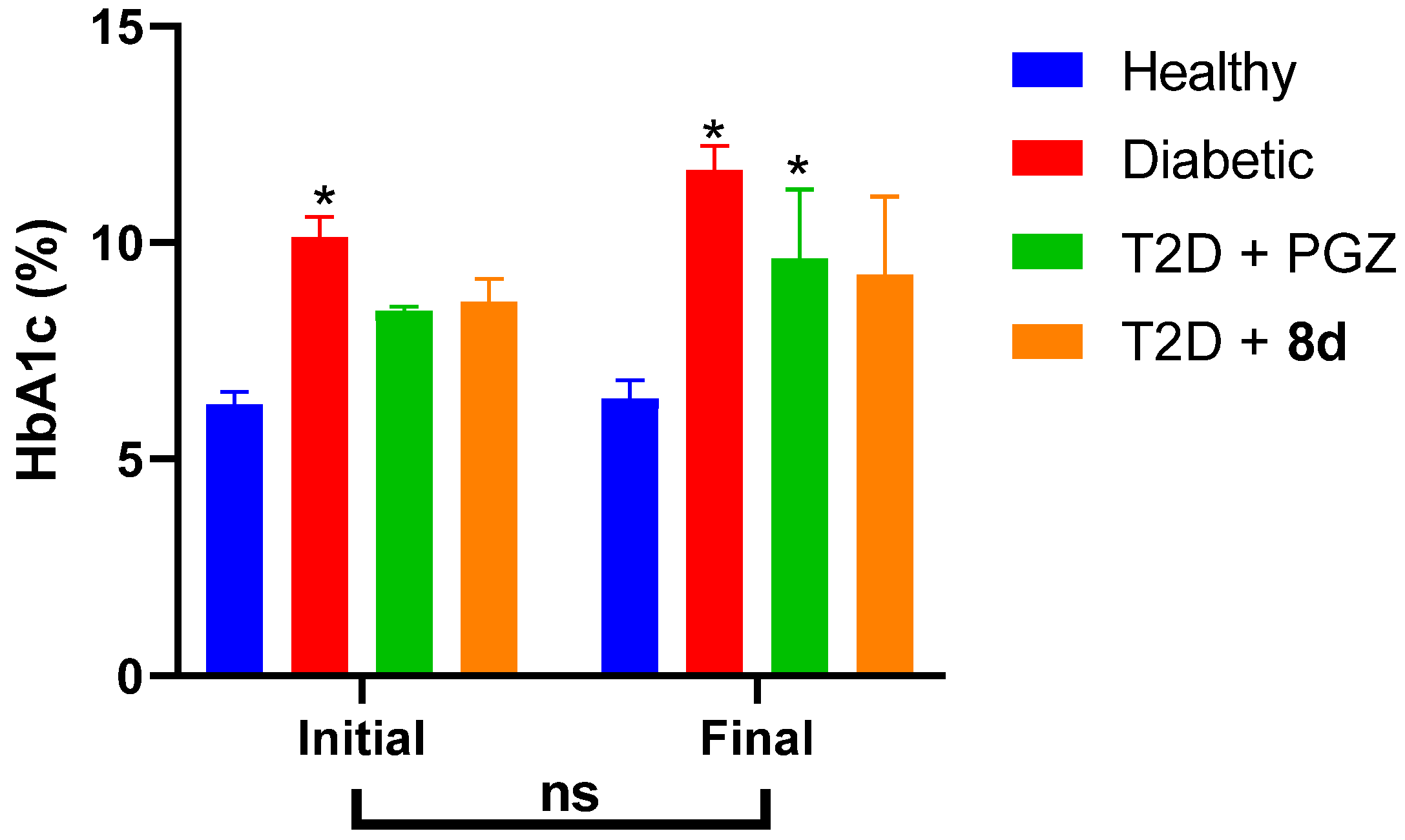
2.4.2. Antidiabetic Activity
Acute Evaluation
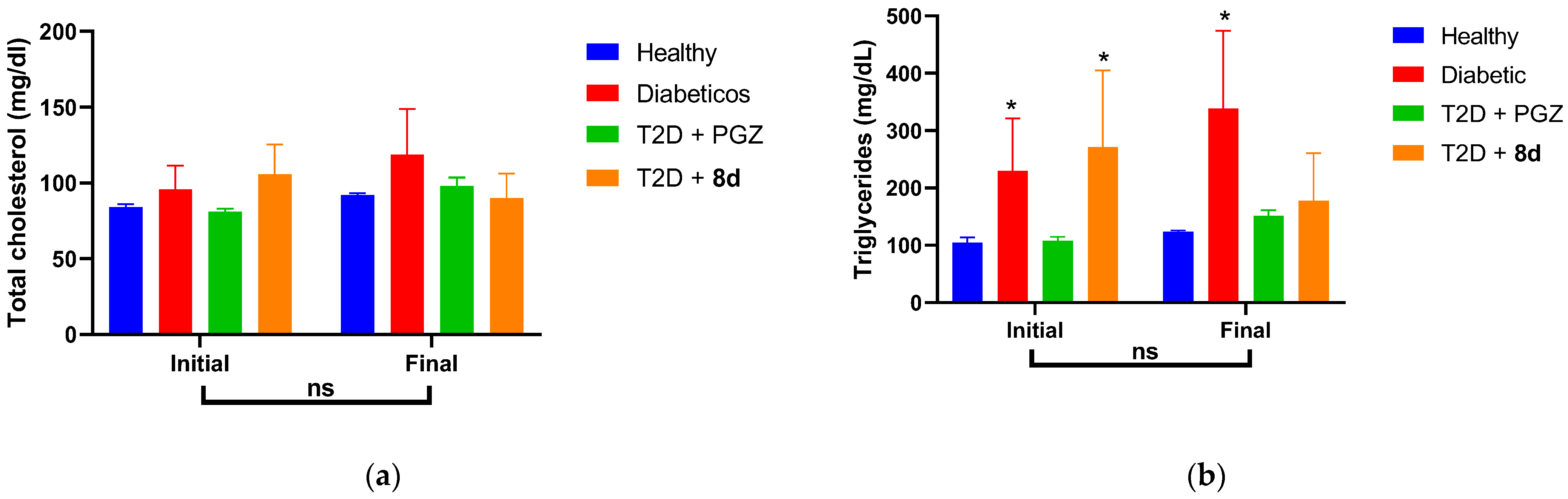
Subchronic Evaluation
2.5. Ex Vivo
2.5.1. Determination of the Antioxidant Effect
2.5.2. Quantification of Inflammation Markers
3. Materials and Methods
3.1. In Silico
3.1.1. Prediction of Physicochemical Properties, Pharmacokinetic and Toxicological Profile
3.1.2. Molecular Docking
3.2. Chemistry
3.2.1. Synthesis of the Dimethyl Benzo[d]thiazol-2-ylcarbonimidodithioate Intermediate [Compound 2]
3.2.2. Synthesis of (E)-((benzo[d]thiazol-2-ylimino)(methylthio)methyl)-L-phenylalanine [5d]
3.2.3. Synthesis of Methyl (E)-((benzo[d]thiazol-2-ylimino)(methylthio)methyl)-L-phenylalaninate [8d]
3.2.4. Synthesis of Methyl (RS, E)-N’-(benzo[d]thiazol-2-yl)-N-(1-(methylamino)-1-oxo-3-phenylpropan-2-yl)carbamimidothioate [12d]
3.2.5. Synthesis of Methyl (S,E)-2-(((benzo[d]thiazol-2-ylimino)(methylthio)methyl)amino)-2-phenylacetate [8c]
3.3. In Vitro
3.3.1. Inhibitory Activity Against ALR2
3.3.2. Evaluation of Antioxidant Capacity
3.4. In Vivo
3.4.1. Acute Oral Toxicity
3.4.2. Antidiabetic Activity
Acute Evaluation
Subchronic Evaluation
3.5. Ex Vivo
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALR2 | Aldose reductase |
| PPAR-γ | Peroxisome proliferator-activated receptor gamma |
| HFD | High-fat diet |
| STZ | Streptozotocin |
| LD50 | Median lethal dose |
| DM | Diabetes mellitus |
| IDF | International Diabetes Federation |
| PGZ | Pioglitazone |
| T2D | Type 2 diabetes |
| BBB | Blood–brain barrier |
| MW | Molecular weight |
| cLogP | Octanol/water partition coefficient calculated |
| nON | Number of hydrogen bond donors |
| nOHNH | Number of hydrogen bond acceptors |
| Nrot | Number of rotal link donors |
| TPSA | Topological polar surface area |
| LogS | Logarithm of the aqueous solubility |
| MR | Molar refractivity |
| Zop | Zopolrestat |
| Epa | Epalrestat |
| Caco-2 | Permeability through cells derived from human colon adenocarcinoma |
| MDCK | Through Madin–Darby canine kidney cells |
| NADP+/NADPH | Oxidized/reduced nicotinamide adenine dinucleotide phosphate |
| P-gp | P-glycoprotein |
| OATP1B1 | Organic anion transporting polypeptide 1B1 |
| BSEP | Bile salt export pump |
| BCRP | Breast cancer resistance |
| CYP450 | Cytochrome P-450 |
| %ABS | Oral absorption percentage |
| HIA | Human intestinal absorption |
| GHS | Globally Harmonized System |
| ABP | Anion-binding pockets |
| ΔG | Gibbs free energy |
| Ki | Inhibition constant |
| #IT | Number of total interactions |
| # I-ABP | Number of interactions with anion-binding pocket residues |
| LBD | Ligand-binding domain |
| TBAH | Tetrabutylammonium hydroxide |
| FRAP | Ferric reducing antioxidant power |
| AOT | Acute oral toxicity |
| OECD | Organization for Economic Co-operation and Development |
| SEM | Standard error of the mean |
| HbA1c | Glycated hemoglobin |
| HOMA-IR | Homeostatic model assessment for insulin resistance |
| T-chol | Total cholesterol |
| TG | Triglyceride |
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| GGT | Gamma-glutamyltransferase |
| ALP | Alkaline phosphatase |
| BUN | Blood urea nitrogen |
| CK | Creatine kinase |
| TAC | Total antioxidant capacity |
| IL-6 | Interleukin-6 |
| TNF-α | Tumor necrosis factor alpha |
| R2 | Coefficient of determination |
| MMFF94 | Merck molecular force field |
| TLC | Thin-layer chromatography |
| 5-ASA | 5-aminosalicylic acid |
| ANOVA | A two-way analysis of variance |
| PAINS | Pan-assay interference compounds |
| AUC | Area under the curve |
| TP | Total protein |
| ALB | Albumin |
| GLO | Globulin |
| TBIL | Total bilirubin |
| TBA | Total bile acids |
| AMY | Amylase |
| CRE | Creatinine |
| OGTT | Oral glucose tolerance test |
| Mp | Melting point |
| tr | Retention time |
References
- Cabrera-Pérez, L.C.; Padilla-Martínez, I.I.; Cruz, A.; Mendieta-Wejebe, J.E.; Tamay-Cach, F.; Rosales-Hernández, M.C. Evaluation of a new benzothiazole derivative with antioxidant activity in the initial phase of acetaminophen toxicity. Arabian J. Chem. 2019, 12, 3871–3882. [Google Scholar] [CrossRef]
- International Diabetes Federation. IDF Diabetes Atlas, 11th ed.; International Diabetes Federation: Brussels, Belgium, 2025. [Google Scholar]
- Pan American Health Organization. Topic on Diabetes Mellitus. Available online: https://www.paho.org/es/temas/diabetes (accessed on 3 January 2024).
- Lima, J.E.B.F.; Moreira, N.C.S.; Sakamoto-Hojo, E.T. Mechanisms underlying the pathophysiology of type 2 diabetes: From risk factors to oxidative stress, metabolic dysfunction, and hyperglycemia. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2022, 874–875, 503437. [Google Scholar] [CrossRef]
- Preguiça, I.; Alves, A.; Nunes, S.; Gomes, P.; Fernandes, R.; Viana, S.D.; Reis, F. Diet-Induced rodent models of diabetic peripheral neuropathy, retinopathy and nephropathy. Nutrients 2022, 12, 250. [Google Scholar] [CrossRef]
- Montoya, A.; Gallardo-Rincón, H.; Silva-Tinoco, R.; García-Cerde, R.; Razo, C.; Ong, L.; Stafford, L.; Lenox, H.; Tapia-Conyer, R. Type 2 diabetes epidemic in Mexico. Burden of disease 1990-2021 analysis and implications for public policies. Gac. Med. Mex. 2023, 159, 474–486. [Google Scholar] [CrossRef]
- Álvarez-Almazán, S.; Solís-Domínguez, L.C.; Duperou-Luna, P.; Fuerte-Gómez, T.; González-Andrade, M.; Aranda-Barradas, M.E.; Palacios-Espinosa, J.F.; Pérez-Villanueva, J.; Matadamas-Martínez, F.; Miranda-Castro, S.P.; et al. Anti-diabetic activity of glycyrrhetinic acid derivatives FC-114 and FC-122: Scale-up, in silico, in vitro, and in vivo studies. Int. J. Mol. Sci. 2023, 24, 12812. [Google Scholar] [CrossRef] [PubMed]
- Almeida, C.; Monteiro, C.; Silvestre, S. Inhibitors of 11β-hydroxysteroid dehydrogenase type 1 as potential drugs for type 2 diabetes mellitus—A systematic review of clinical and in vivo preclinical studies. Sci. Pharm. 2021, 89, 5. [Google Scholar] [CrossRef]
- Padhi, S.; Nayak, A.K.; Behera, A. Type II diabetes mellitus: A review on recent drug-based therapeutics. Biomed. Pharmacother. 2020, 131, 110708. [Google Scholar] [CrossRef]
- Rosales-Hernández, M.C.; Cruz, A.; Mendieta-Wejebe, J.E.; Tamay-Cach, F. 2-Guanidinobenzazoles as building blocks to afford biologically active derivatives. Curr. Org. Chem. 2023, 27, 38–54. [Google Scholar] [CrossRef]
- Rosales-Hernández, M.C.; Mendieta-Wejebe, J.E.; Tamay-Cach, F.; Cruz, A. Synthetic procedures to access 2-guanidinobenzazoles of biological interest. Curr. Org. Synth. 2023, 20, 504–522. [Google Scholar] [CrossRef]
- Yadav, R.K.; Kumar, R.; Singh, H.; Mazumdar, A.; Salahuddin; Chauhan, B.; Abdullah, M.M. Recent insights on synthetic methods and pharmacological potential in relation with structure of benzothiazoles. Med. Chem. 2023, 19, 325–360. [Google Scholar] [CrossRef]
- Kumar, A.; Mishra, A.K. Advancement in pharmacological activities of benzothiazole and its derivatives: An up to date review. Mini Rev. Med. Chem. 2021, 21, 314–335. [Google Scholar] [CrossRef]
- Keri, R.S.; Patil, M.R.; Patil, S.A.; Budagumpi, S. A comprehensive review in current developments of benzothiazole-based molecules in medicinal chemistry. Eur. J. Med. Chem. 2015, 89, 207–251. [Google Scholar] [CrossRef]
- Alvarado-Salazar, J.A.; Valdes, M.; Cruz, A.; Moreno de Jesús, B.; Patiño-González, D.; Olivares-Corichi, I.M.; Tamay-Cach, F.; Mendieta-Wejebe, J.E. In Silico and In vivo evaluation of novel 2-aminobenzothiazole derivative compounds as antidiabetic agents. Int. J. Mol. Sci. 2025, 26, 909. [Google Scholar] [CrossRef] [PubMed]
- Hamdi, A.; Yaseen, M.; Ewes, W.A.; Bhat, M.A.; Ziedan, N.I.; El-Shafey, H.W.; Mohamed, A.A.B.; Elnagar, M.R.; Haikal, A.; Othman, D.I.A.; et al. Development of new thiazolidine-2,4-dione hybrids as aldose reductase inhibitors endowed with antihyperglycaemic activity: Design, synthesis, biological investigations, and in silico insights. J. Enzyme Inhib. Med. Chem. 2023, 38, 2231170. [Google Scholar] [CrossRef] [PubMed]
- Kousaxidis, A.; Petrou, A.; Lavrentaki, V.; Fesatidou, M.; Nicolaou, I.; Geronikaki, A. Aldose reductase and protein tyrosine phosphatase 1B inhibitors as a promising therapeutic approach for diabetes mellitus. Eur. J. Med. Chem. 2025, 207, 112742. [Google Scholar] [CrossRef]
- Gupta, S.K.; Tripathi, P.K. CADD studies in the discovery of potential ARI (Aldose Reductase inhibitors) agents for the treatment of diabetic complications. Curr. Diabetes Rev. 2023, 19, e180822207672. [Google Scholar] [CrossRef]
- Sharma, S.R.; Sharma, N. Epalrestat, an aldose reductase inhibitor, in diabetic neuropathy: An Indian perspective. Ann. Indian Acad. Neurol. 2008, 11, 231–235. [Google Scholar] [CrossRef]
- Qiu, L.; Lin, J.; Xu, F.; Gao, Y.; Zhang, C.; Liu, Y.; Luo, Y.; Yang, J.Y. Inhibition of aldose reductase activates hepatic peroxisome proliferator-activated receptor-α and ameliorates hepatosteatosis in diabetic db/db mice. J. Diabetes Res. 2012, 2012, 789730. [Google Scholar] [CrossRef]
- Mal, S.; Dwivedi, A.R.; Kumar, V.; Kumar, N.; Kumar, B.; Kumar, V. Role of peroxisome proliferator-activated receptor gamma (PPARγ) in different disease states: Recent updates. Curr. Med. Chem. 2021, 28, 3193–3215. [Google Scholar] [CrossRef]
- Miyachi, H. Structural biology inspired development of a series of human peroxisome proliferator-activated receptor gamma (PPARγ) ligands: From agonist to antagonist. Int. J. Mol. Sci. 2023, 24, 3940. [Google Scholar] [CrossRef]
- Farhadi, T.; Hashemian, S.M. Computer-aided design of amino acid-based therapeutics: A review. Drug Des. Dev. Ther. 2018, 12, 1239–1254. [Google Scholar] [CrossRef]
- Padilla-Martínez, I.I.; González-Encarnación, J.M.; García-Báez, E.V.; Cruz, A.; Ramos-Organillo, Á.A. Isothioureas, ureas, and their N-Methyl amides from 2-aminobenzothiazole and chiral amino acids. Molecules 2019, 24, 3391. [Google Scholar] [CrossRef] [PubMed]
- DIA-DB. Structural Bioinformatics and High Performance Computing (BIO-HPC) Research Group. Universidad Católica de Murcia (UCAM), Spain. Available online: https://bio-hpc.ucam.edu/dia-db/index.php (accessed on 28 December 2023).
- Jia, C.Y.; Li, J.Y.; Hao, G.F.; Yang, G.F. A drug-likeness toolbox facilitates ADMET study in drug discovery. Drug Discov. Today 2017, 25, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed]
- Molinspiration Cheminformatics. SK-900 26 Slovensky Grob, Slovak Republic. Available online: https://www.molinspiration.com (accessed on 2 January 2024).
- ADMETLab 3.0. Available online: https://admetlab3.scbdd.com/server/evaluationCal (accessed on 2 January 2024).
- PreADMET. Available online: https://preadmet.webservice.bmdrc.org/ (accessed on 2 January 2024).
- Moser, P.; Zelli, R.; Dos Santos, L.J.; Henry, M.; Sanchez-Garcia, K.; Caspar, Y.; Marro, F.C.; Chovelon, B.; Saez Cabodevilla, J.; de Choudens, S.O.; et al. Fast release of carboxylic acid inside cells. ChemMedChem 2025, 20, e202500056. [Google Scholar] [CrossRef]
- Kovacikova, L.; Prnova, M.S.; Majekova, M.; Bohac, A.; Karasu, C.; Stefek, M. Development of novel indole-based bifunctional aldose reductase inhibitors/antioxidants as promising drugs for the treatment of diabetic complications. Molecules 2021, 26, 2867. [Google Scholar] [CrossRef]
- Amin, M.L. P-glycoprotein inhibition for optimal drug delivery. Drug Target Insights 2013, 7, 27–34. [Google Scholar] [CrossRef]
- Herrera Ruiz, D.; Hernández Baltazar, E.; Espinosa Lara, J.C.; Luz Martínez, I.d.l.; Beltrán Torres, A.A.; Martínez Alejo, J.M. Técnicas de complejidad variable para evaluar la absorción de fármacos. Rev. Mex. Cienc. Farm. 2012, 43, 18–32. Available online: https://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1870-01952012000100003 (accessed on 22 May 2025).
- SwissADME. SwissDrugDesign. Available online: http://www.swissadme.ch/ (accessed on 3 January 2024).
- Kazmi, S.R.; Jun, R.; Yu, M.S.; Jung, C.; Na, D. In silico approaches and tools for the prediction of drug metabolism and fate: A review. Comput. Biol. Med. 2019, 106, 54–64. [Google Scholar] [CrossRef]
- Guengerich, F.P. Cytochrome p450 and chemical toxicology. Chem. Res. Toxicol. 2008, 21, 70–83. [Google Scholar] [CrossRef]
- United Nations Economic Commission for Europe. Sustainable Development Goals. About the GHS. Palais des Nations, CH-1211 Geneva 10, Switzerland. Available online: https://unece.org/about-ghs (accessed on 8 October 2024).
- ProTox 3.0. Prediction of Toxicity of Chemicals. Tox-Prediction. Charite University of Medicine, Institute for Physiology, Structural Bioinformatics Group, Philippstrasse 12, 10115 Berlin, Germany. Available online: https://tox.charite.de/protox3/ (accessed on 6 January 2024).
- Lin, P.; Niu, Y. Inhibitory selectivity to the AKR1B10 and aldose reductase (AR): Insight from molecular dynamics simulations and free energy calculations. RSC Adv. 2023, 13, 26709–26718. [Google Scholar] [CrossRef] [PubMed]
- Protein Data Bank (RCSB PDB). Available online: https://www.rcsb.org/structure/4JIR (accessed on 10 January 2024).
- Protein Data Bank (RCSB PDB). Available online: https://www.rcsb.org/structure/2FVJ (accessed on 10 January 2024).
- Carvalho, V.F.; Barreto, E.O.; Serra, M.F.; Cordeiro, R.S.; Martins, M.A.; Fortes, Z.B.; e Silva, P.M. Aldose reductase inhibitor zopolrestat restores allergic hyporesponsiveness in alloxan-diabetic rats. Eur. J. Pharmacol. 2006, 549, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Zentrum für Bioinformatik: Universität Hamburg. Proteins Plus Server. Proteins. Plus. Available online: https://proteins.plus/ (accessed on 12 January 2024).
- Burgermeister, E.; Schnoebelen, A.; Flament, A.; Benz, J.; Stihle, M.; Gsell, B.; Meyer, M. A novel partial agonist of peroxisome proliferator-activated receptor-gamma (PPARgamma) recruits PPARgamma-coactivator-1alpha, prevents triglyceride accumulation, and potentiates insulin signaling in vitro. Mol. Endocrinol. 2006, 20, 809–830. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Mosure, S.A.; Zheng, J.; Brust, R.; Bass, J.; Nichols, A.; Kojetin, D.J. A molecular switch regulating transcriptional repression and activation of PPARγ. Nat. Commun. 2020, 11, 956. [Google Scholar] [CrossRef]
- Álvarez-Almazán, S.; Bello, M.; Tamay-Cach, F.; Martínez-Archundia, M.; Alemán-González-Duhart, D.; Correa-Basurto, J.; Mendieta-Wejebe, J.E. Study of new interactions of glitazone’s stereoisomers and the endogenous ligand 15d-PGJ2 on six different PPAR gamma proteins. Biochem. Pharmacol. 2017, 142, 168–193. [Google Scholar] [CrossRef]
- Téllez, F.; Cruz, A.; López-Sandoval, H.; Ramos-García, I.; Gayosso, M.; Castillo-Sierra, R.N.; Paz-Michel, B.; Nöth, H.; Flores-Parra, A.; Contreras, R. Dithiocarbamates, thiocarbamic esters, dithiocarboimidates, guanidines, thioureas, isothioureas, and tetraazathiapentalene derived from 2-aminobenzothiazole. Eur. J. Org. Chem. 2004, 20, 4203–4214. [Google Scholar] [CrossRef]
- Merchán, F.; Garín, J.; Meléndez, E. A Facile Synthesis of Dimethyl N-(2-Benzothiazolyl)-dithiocarbonimidates and Methyl N-(2-Benzothiazolyl)-dithiocarbamates. Synthesis 1982, 07, 590–591. [Google Scholar] [CrossRef]
- Rosales-Hernández, M.C.; Mendieta-Wejebe, J.E.; Padilla-Martínez, I.I.; García-Báez, E.V.; Cruz, A. Synthesis and biological importance of 2-(thio)ureabenzothiazoles. Molecules 2022, 27, 6104. [Google Scholar] [CrossRef]
- Rumpf, J.; Burger, R.; Schulze, M. Statistical evaluation of DPPH, ABTS, FRAP, and Folin-Ciocalteu assays to assess the antioxidant capacity of lignins. Int. J. Biol. Macromol. 2023, 233, 123470. [Google Scholar] [CrossRef]
- Organization for Economic Co-operation and Development (OECD). Acute Oral Toxicity: Up-and-Down-Procedure (UDP); Test Guideline No. 425; OECD: Paris, France, 2008; pp. 1–28. [Google Scholar] [CrossRef]
- AlTamimi, L.; Zakaraya, Z.Z.; Hailat, M.; Ahmad, M.N.; Qinna, N.A.; Hamad, M.F.; Dayyih, W.A. Test of insulin resistance in nondiabetic and streptozotocin induced diabetic rats using glycosylated hemoglobin test and other interventions. J. Adv. Pharm. Technol. Res. 2024, 15, 1–7. [Google Scholar] [CrossRef]
- Pandhare, R.B.; Sangameswaran, B.; Mohite, P.B.; Khanage, S.G. Antihyperglycaemic and lipid lowering potential of Adenanthera pavonina Linn. In streptozotocin induced diabetic rats. Orient. Pharm. Exp. Med. 2012, 12, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, D.A.; Kume, W.T.; Correia, F.S.; Queiroz, T.S.; Allebrandt Neto, E.W.; Santos, M.P.D.; Kawashita, N.H.; França, S.A. High-fat diet and streptozotocin in the induction of type 2 diabetes mellitus: A new proposal. An. Acad. Bras. Cienc. 2019, 91, e20180314. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; Cao, Y.; Chen, P.; Li, J.; Li, W.; Huang, G.; Zheng, H.; Zhu, X.; Zhang, H.; Chen, Y.; et al. A reliable strategy for establishment of an animal model of diabetic cardiomyopathy: Induction by a high-fat diet combined with single or multiple injections of low-dose streptozotocin. Life Sci. 2024, 358, 123161. [Google Scholar] [CrossRef]
- Percie du Sert, N.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; Emerson, M.; et al. Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 2.0. PLoS Biol. 2020, 18, e3000411. [Google Scholar] [CrossRef]
- Maheshwari, R.A.; Parmar, G.R.; Hinsu, D.; Seth, A.K.; Balaraman, R. Novel therapeutic intervention of coenzyme Q10 and its combination with pioglitazone on the mRNA expression level of adipocytokines in diabetic rats. Life Sci. 2020, 258, 118155. [Google Scholar] [CrossRef]
- Miyazaki, Y.; Mahankali, A.; Matsuda, M.; Mahankali, S.; Hardies, J.; Cusi, K.; Mandarino, L.J.; DeFronzo, R.A. Effect of pioglitazone on abdominal fat distribution and insulin sensitivity in type 2 diabetic patients. J. Clin. Endocrinol. Metab. 2002, 87, 2784–2791. [Google Scholar] [CrossRef]
- Li, W.; Wang, Y.; Liu, C.; Yu, Y.; Xu, L.; Dong, B. Comparing efficacy of chiglitazar, pioglitazone, and semaglutide in type 2 diabetes: A retrospective study. Diabetes Ther. 2025, 16, 993–1017. [Google Scholar] [CrossRef]
- Dowarah, J.; Singh, V.P. Anti-diabetic drugs recent approaches and advancements. Bioorg. Med. Chem. 2020, 28, 115263. [Google Scholar] [CrossRef]
- Quintana-Pérez, J.C.; García-Dolores, F.; Valdez-Guerrero, A.S.; Alemán-González-Duhart, D.; Arellano-Mendoza, M.G.; Rojas Hernández, S.; Olivares-Corichi, I.M.; García Sánchez, J.R.; Trujillo Ferrara, J.G.; Tamay-Cach, F. Modeling type 2 diabetes in rats by administering tacrolimus. Islets 2022, 14, 114–127. [Google Scholar] [CrossRef]
- Morones, J.; Pérez, M.; Muñoz, M.; Sánchez, E.; Ávila, M.; Topete, J.; Ventura, J.; Martínez, S. Evaluation of the effect of an α-adrenergic blocker, a PPAR-γ receptor agonist, and a glycemic regulator on chronic kidney disease in diabetic rats. Int. J. Mol. Sci. 2024, 25, 11372. [Google Scholar] [CrossRef]
- Al-Muzafar, H.M.; Alshehri, F.S.; Amin, K.A. The role of pioglitazone in antioxidant, anti-inflammatory, and insulin sensitivity in a high fat-carbohydrate diet-induced rat model of insulin resistance. Braz. J. Med. Biol. Res. 2021, 54, e10782. [Google Scholar] [CrossRef]
- Alharbi, K.S.; Afzal, M.; Al-Abbasi, F.A.; Moglad, E.; Al-Qahtani, S.D.; Almalki, N.A.R.; Imam, F.; Sayyed, N.; Kazmi, I. In vivo and in silico study of europinidin against streptozotocin-isoproterenol-induced myocardial damage via alteration of hs-CRP/CPK-MB/Caspase-3/Bcl-2 pathways. Sci. Rep. 2025, 15, 3076. [Google Scholar] [CrossRef]
- Xu, J.; Shi, X.; Pan, Y. The association of aspartate aminotransferase/alanine aminotransferase ratio with diabetic nephropathy in patients with type 2 diabetes. Diabetes Metab. Syndr. Obes. Targets Ther. 2021, 14, 3831–3837. [Google Scholar] [CrossRef] [PubMed]
- Rafaqat, S.; Sattar, A.; Khalid, A.; Rafaqat, S. Role of liver parameters in diabetes mellitus—a narrative review. Endocr. Regul. 2023, 57, 200–220. [Google Scholar] [CrossRef] [PubMed]
- Faramarzi, E.; Mehrtabar, S.; Molani-Gol, R.; Dastgiri, S. The relationship between hepatic enzymes, prediabetes, and diabetes in the Azar cohort population. BMC Endocr. Disord. 2025, 25, 41. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Dawuti, A.; Gong, D.; Wang, R.; Yuan, T.; Wang, S.; Xing, C.; Lu, Y.; Du, G.; Fang, L. Puerarin-V improve mitochondrial respiration and cardiac function in a rat model of diabetic cardiomyopathy via inhibiting pyroptosis pathway through P2X7 receptors. Int. J. Mol. Sci. 2022, 23, 13015. [Google Scholar] [CrossRef]
- Usatiuc, L.O.; Pârvu, M.; Pop, R.M.; Uifălean, A.; Vălean, D.; Szabo, C.E.; Țicolea, M.; Cătoi, F.A.; Ranga, F.; Pârvu, A.E. Phytochemical profile and antidiabetic, antioxidant, and anti-inflammatory activities of Gypsophila paniculata ethanol extract in rat streptozotocin-induced diabetes mellitus. Antioxidants 2024, 13, 1029. [Google Scholar] [CrossRef]
- Park, I.R.; Chung, Y.G.; Won, K.C. Overcoming β-cell dysfunction in type 2 diabetes mellitus: CD36 inhibition and antioxidant system. Diabetes Metab. J. 2025, 49, 1–12. [Google Scholar] [CrossRef]
- Cruz-Rodríguez, J.; López-Solís, R. El estrés oxidativo en la fisiopatología del síndrome metabólico. Rev. Med. Investig. Univ. Autónoma Estado México 2021, 9, 88–94. [Google Scholar]
- Pawar, H.D.; Patil, Y.; Patil, A.; Nakhate, K.T.; Agrawal, Y.O.; Suchal, K.; Ojha, S.; Goyal, S.N. Cardioprotective effect of CB1 receptor antagonist AM251 against β receptor-stimulated myocardial infarction via modulation of NF-kB signaling pathway in diabetic mice. Heliyon 2024, 10, e35138. [Google Scholar] [CrossRef]
- Civelek, E.; Karaman, E.F.; Özden, S.; Büyükpınarbaşılı, N.; Uydeş Doğan, B.; Kaleli Durman, D. Evaluation of the effects of pioglitazone on perivascular adipose tissue function, properties, and structure in a rat model of type-2 diabetes. Can. J. Physiol. Pharmacol. 2025, 103, 12–28. [Google Scholar] [CrossRef] [PubMed]
- Palavicini, J.P.; Chavez-Velazquez, A.; Fourcaudot, M.; Tripathy, D.; Pan, M.; Norton, L.; DeFronzo, R.A.; Shannon, C.E. The Insulin-Sensitizer Pioglitazone Remodels Adipose Tissue Phospholipids in Humans. Front. Physiol. 2021, 12, 784391. [Google Scholar] [CrossRef] [PubMed]
- Drug Discovery. Version 20.1.0.19295; BIOVIA Discovery Studio Visualizer; Dassault Systems: Waltham, MA, USA, 2022; Available online: https://www.3ds.com/products/biovia/discovery-studio (accessed on 16 January 2024).
- Cruz, A.; Padilla-Martínez, I.I.; García-Báez, E.V. A synthetic method to access symmetric and non-symmetric 2-(N,N’-disubstituted)guanidinebenzothiazoles. Molecules 2012, 17, 10178–10191. [Google Scholar] [CrossRef] [PubMed]
- Abcam.cn. ab273276 Aldose Reductase Activity Assay Kit (Colorimetric). Available online: https://www.abcam.cn/ps/products/273/ab273276/documents/Aldose-Reductase-Activity-assay-protocol-book-v1-ab273276%20(website).pdf (accessed on 10 June 2025).
- Pharmaceutical and Biotechnological Innovation-Services, Pruebas Antioxidants. Available online: https://www.pharbiois.com/reactivos-kits (accessed on 30 September 2024).
- Ferdowsian, H.R.; Beck, N. Ethical and scientific considerations regarding animal testing and research. PLoS ONE 2011, 6, e24059. [Google Scholar] [CrossRef]
- Morton, D.B. Humane endpoints in animal experimentation for biomedical research: Ethical, legal and practical aspects. In Humane Endpoints in Animal Experiments for Biomedical Research; Morton, D.B., Hendriksen, C.F.M., Eds.; Royal Society of Medicine Press: London, UK, 1998; pp. 5–12. [Google Scholar]
- Gheibi, S.; Kashfi, K.; Ghasemi, A. A practical guide for induction of type-2 diabetes in rat: Incorporating a high-fat diet and streptozotocin. Biomed. Pharmacother. 2017, 95, 605–613. [Google Scholar] [CrossRef]
- Abo-elmatty, D.M.; Essawy, S.S.; Badr, J.M.; Sterner, O. Antioxidant and anti-inflammatory effects of Urtica pilulifera extracts in type 2 diabetic rats. J. Ethnopharmacol. 2013, 145, 269–277. [Google Scholar] [CrossRef]
- Shiming, Z.; Mak, K.K.; Balijepalli, M.K.; Chakravarthi, S.; Pichika, M.R. Swietenine potentiates the antihyperglycemic and antioxidant activity of Metformin in Streptozotocin induced diabetic rats. Biomed. Pharmacother. 2021, 139, 111576. [Google Scholar] [CrossRef]
- Ahn, T.; Bae, C.S.; Yun, C.H. Selenium supplementation restores the decreased albumin level of peripheral blood mononuclear cells in streptozotocininduced diabetic mice. J. Vet. Med. Sci. 2016, 78, 669–674. [Google Scholar] [CrossRef]
- SIGMA-ALDRICH. Antioxidant Assay Kit MAK334 Technical Bulletin. Available online: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/114/194/mak334bul.pdf?srsltid=AfmBOorsNzcAYYxqwvurAL-ynmJzy1nydGNuIz2pkYqFcTrYxplY9b7q (accessed on 10 June 2025).
- SIGMA-ALDRICH. User Guide Rat TNF-alpha ELISA Kit. RAB0479. Available online: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/904/631/rab0479ugrev0824-mk.pdf (accessed on 10 June 2025).
- SIGMA-ALDRICH. User Guide Rat IL-6 ELISA Kit. RAB0311. Available online: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/379/232/rab0311ugrev0824-mk.pdf (accessed on 10 June 2025).
- SIGMA-ALDRICH. RAB0904-1KT Certificate of Analysis/Protocol. Available online: https://www.sigmaaldrich.com/certificates/Graphics/COfAInfo/SigmaABNO/RAB0904_1027J0743.pdf (accessed on 6 May 2025).


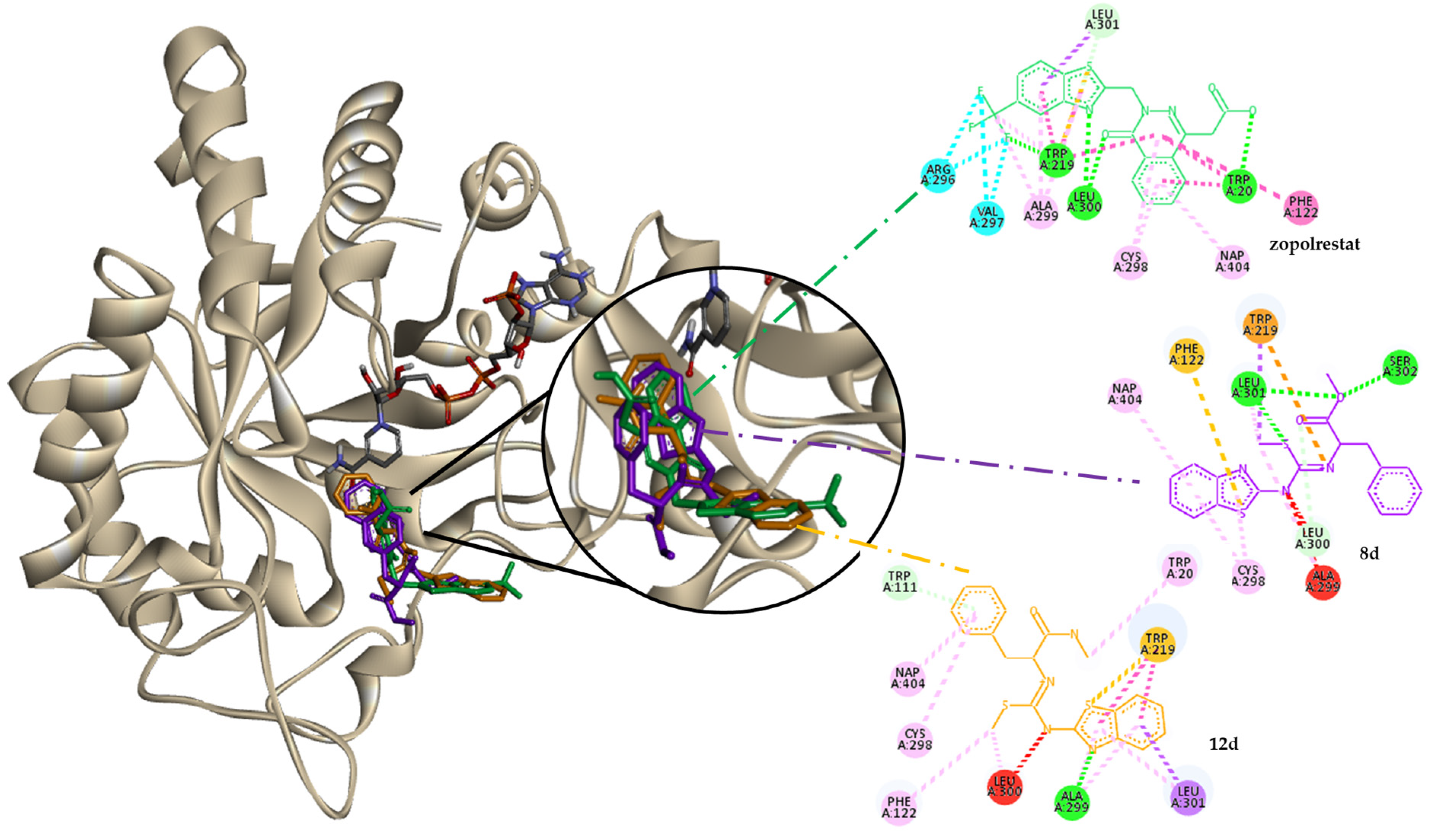
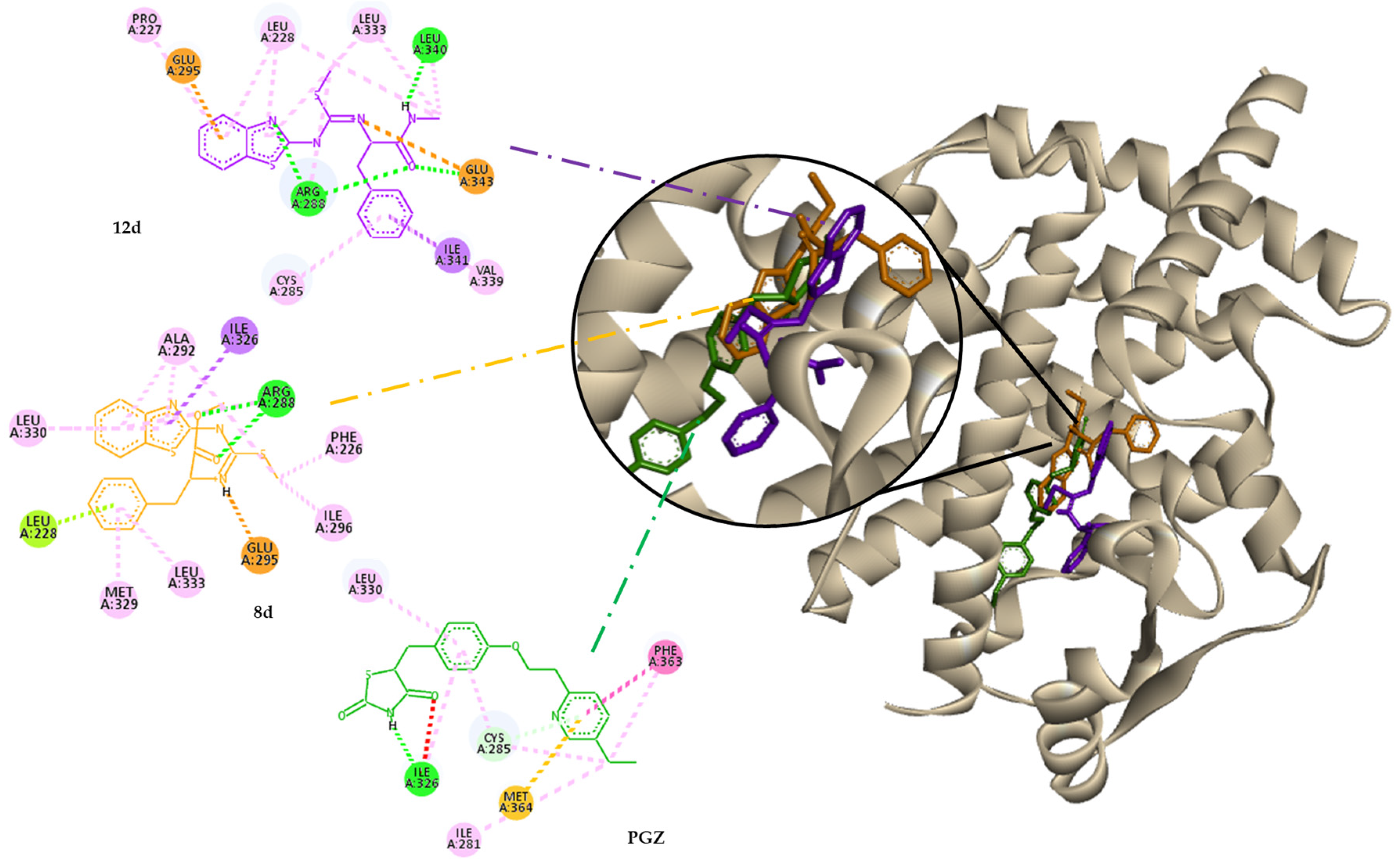


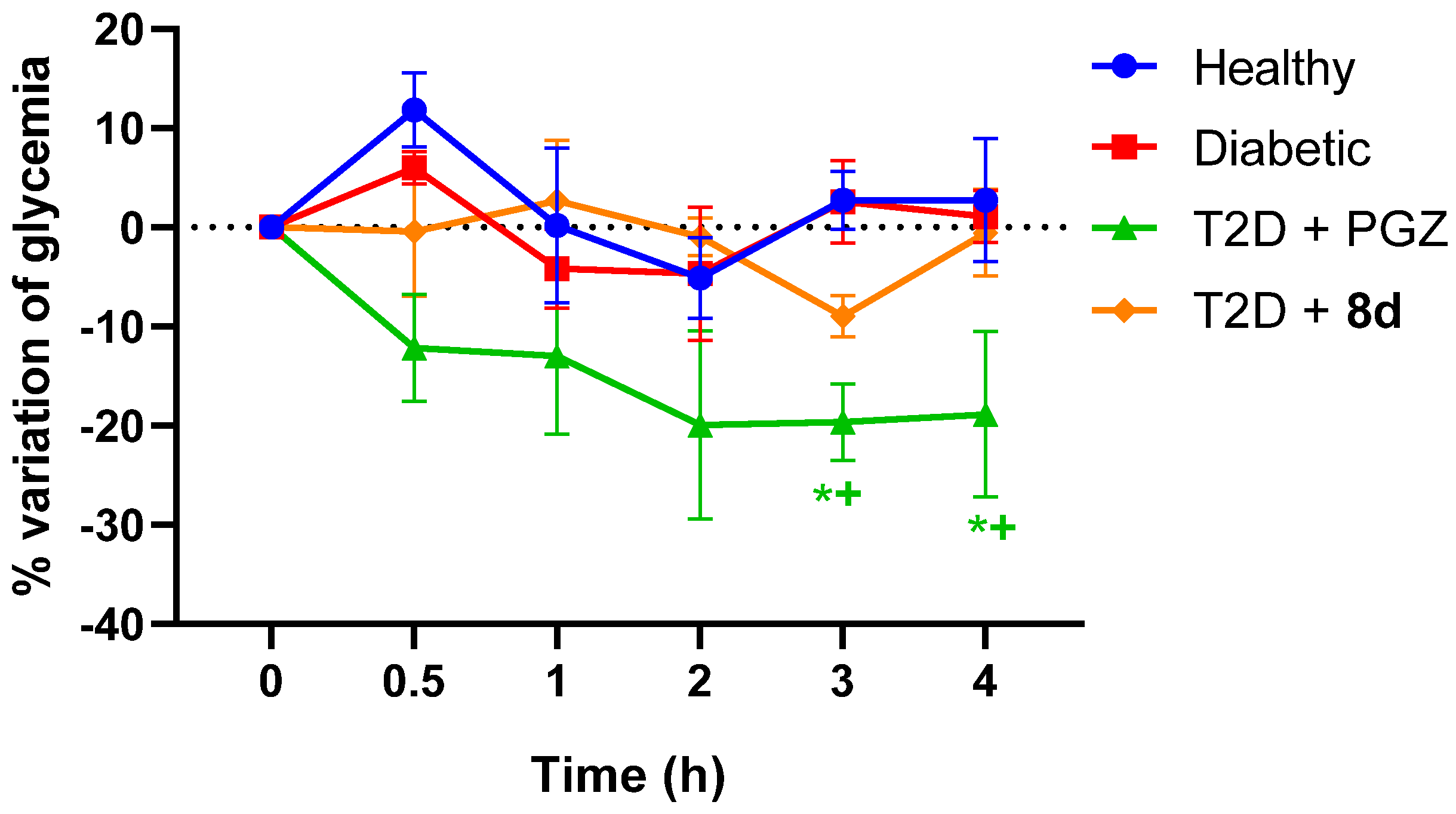
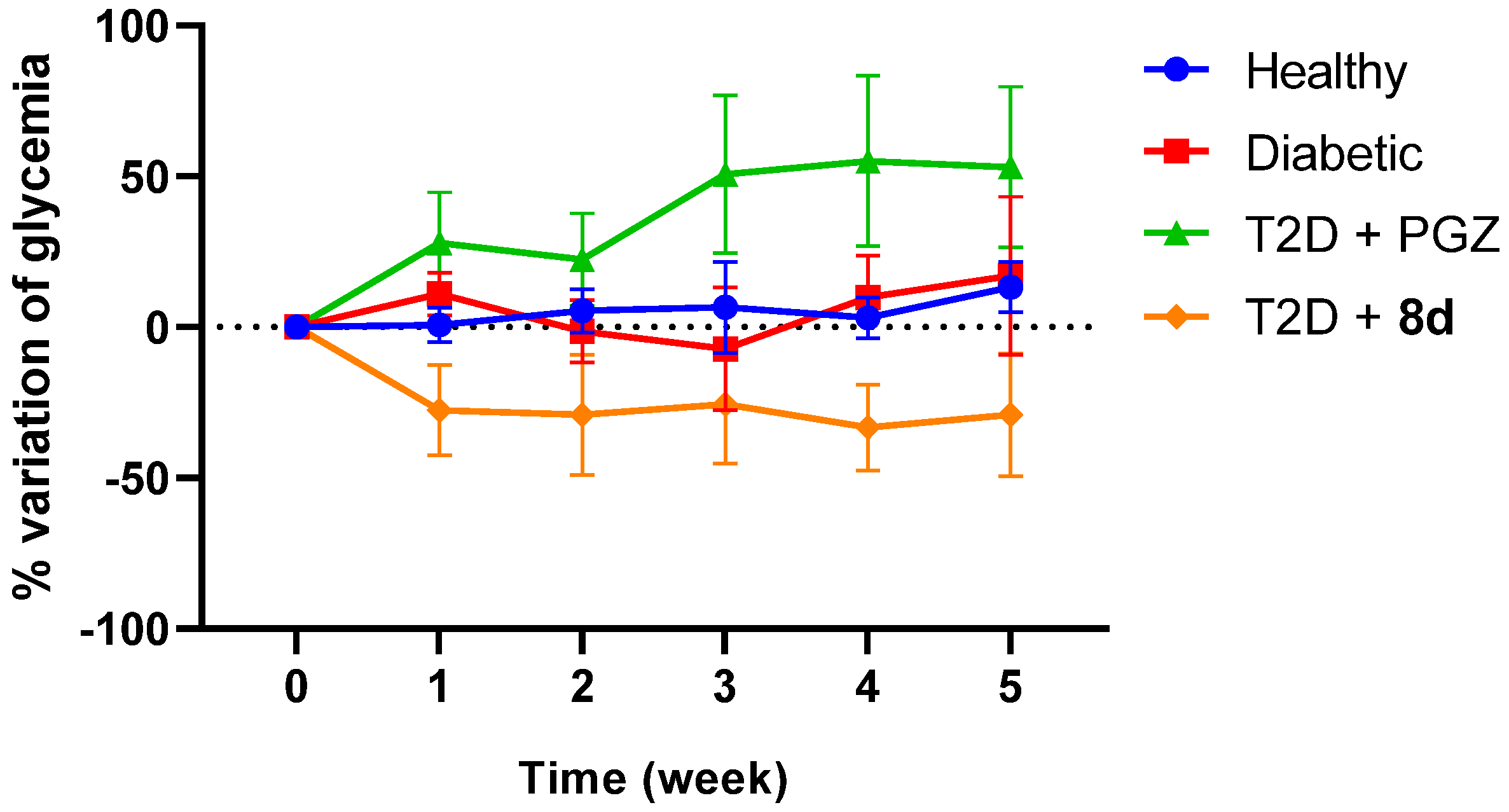



| Toxicity prediction | ||||||
|---|---|---|---|---|---|---|
| Mutagenicity | Cytotoxicity | Reproductive Effect | Irritant Effect | LD50 | Class | |
| 5d | × | × | × | × | 1000 | IV |
| 8c | × | × | × | × | 1000 | IV |
| 8d | × | × | × | × | 1000 | IV |
| 12d | × | × | × | × | 1000 | IV |
| Epa | × | × | × | × | 1365 | IV |
| Zop | × | × | × | × | 5 | II |
| PGZ | × | × | × | × | 1000 | IV |
| Vehicle* | 8c | 8d | 12d | |
|---|---|---|---|---|
| IWA | 238.00 | 306.20 ± 9.70 | 201.0 ± 7.17 | 208.47 ± 6.47 |
| FWA | 331.50 | 204.37 ± 1.47 | 310.57 ± 11.6 | 309.67 ± 4.67 |
| Spleen | 0.80 | 0.73 ± 0.03 | 0.67 ± 0.07 | 0.83 ± 0.03 |
| Liver | 19.00 | 18.87 ± 1.47 | 17.57 ± 0.50 | 19.33 ± 0.03 |
| Intestine | 24.70 | 30.20 ± 1.50 | 29.97 ± 0.72 | 29.10 ± 0.50 |
| Stomach | 2.00 | 1.57 ± 0.07 | 1.70 ± 0.10 | 1.53 ± 0.07 |
| Kidney | 3.20 | 2.57 ± 0.27 | 2.97 ± 0.03 | 2.87 ± 0.067 |
| Heart | 1.60 | 1.07 ± 0.17 | 1.30 ± 0.03 | 1.23 ±0.13 |
| Healthy | Diabetic | T2D + PGZ | T2D + 8d | ||
|---|---|---|---|---|---|
| HOMA-IR | Initial | 1.97 ± 0.44 | 2.68 ± 0.57 | 2.59 ± 0.34 | 4.22 ± 1.58 |
| Final | 1.41± 0.40 | 5.42 ± 1.85 | 2.32 ± 0.41 | 1.66 ± 0.61 | |
| Group | TAC Equivalent to Trolox [μM] |
|---|---|
| Healthy control | 113.29 ± 8.39 |
| Diabetic control | 87.49 ± 22.28 |
| T2D + PGZ | 97.75 ± 1.29 |
| T2D + 8d | 79.26 ± 17.28 |
| Group | IL-6 [pg/mL] |
|---|---|
| Healthy control | 373.75 ± 130.19 |
| Diabetic control | 536.82 ± 136.49 |
| T2D + PGZ | 234.25 ± 52.05 |
| T2D + 8d | 887.53 ± 243.91 |
| Group | TNF-α [pg/mL] |
|---|---|
| Healthy control | 1641.41 ± 266.06 |
| Diabetic control | 4452.61 ± 294.68 |
| T2D + PGZ | 3000.53 ± 519.24 |
| T2D + 8d | 5417.72 ± 2104.2 |
| Group | Food | Treatment |
|---|---|---|
| Healthy | LabDiet 5001 | None |
| Diabetic | HFD | None |
| T2D + PGZ | HFD | PGZ (30 mg/Kg) * |
| T2D + 8d | HFD | Compound 8d (32 mg/Kg) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Vallejo, N.; Valdes, M.; Reyes-Ramírez, A.; Alvarado-Salazar, J.A.; Cruz, A.; Andrade-Jorge, E.; Mendieta-Wejebe, J.E. Preclinical Evaluation of 2-Aminobenzothiazole Derivatives: In Silico, In Vitro, and Preliminary In Vivo Studies as Diabetic Treatments and Their Complications. Molecules 2025, 30, 3427. https://doi.org/10.3390/molecules30163427
Reyes-Vallejo N, Valdes M, Reyes-Ramírez A, Alvarado-Salazar JA, Cruz A, Andrade-Jorge E, Mendieta-Wejebe JE. Preclinical Evaluation of 2-Aminobenzothiazole Derivatives: In Silico, In Vitro, and Preliminary In Vivo Studies as Diabetic Treatments and Their Complications. Molecules. 2025; 30(16):3427. https://doi.org/10.3390/molecules30163427
Chicago/Turabian StyleReyes-Vallejo, Natalia, Miguel Valdes, Adelfo Reyes-Ramírez, Juan Andres Alvarado-Salazar, Alejandro Cruz, Erik Andrade-Jorge, and Jessica Elena Mendieta-Wejebe. 2025. "Preclinical Evaluation of 2-Aminobenzothiazole Derivatives: In Silico, In Vitro, and Preliminary In Vivo Studies as Diabetic Treatments and Their Complications" Molecules 30, no. 16: 3427. https://doi.org/10.3390/molecules30163427
APA StyleReyes-Vallejo, N., Valdes, M., Reyes-Ramírez, A., Alvarado-Salazar, J. A., Cruz, A., Andrade-Jorge, E., & Mendieta-Wejebe, J. E. (2025). Preclinical Evaluation of 2-Aminobenzothiazole Derivatives: In Silico, In Vitro, and Preliminary In Vivo Studies as Diabetic Treatments and Their Complications. Molecules, 30(16), 3427. https://doi.org/10.3390/molecules30163427








