Synthesis and Characterization of the Complexes of Pentane-2,4-dione with Nickel(II) and Cobalt(III): [Ni(acac)2]·0.5CH3OH and [Co(acac)2NO3]·2H2O (acac = Pentane-2,4-dione)
Abstract
:Introduction
Results and Discussion
| Complexes | 1 | 2 |
|---|---|---|
| Formula | C11H18NiO5 | C10CoH18O9 |
| FW | 288.96 | 355.18 |
| Crystal shape/color | Yellow | brown-red |
| Crystal size/ mm | 0.30 x 0.24 x 0.20 mm | 0.42 x 0.37 x 0.31 mm |
| T/K | 293K | 298 K |
| Crystal system | Monoclinic | Triclinic |
| Space group | P2(1) | P(-1) |
| a/Å | 9.254(4) | 8.153(9) |
| b/Å | 11.450(5) | 9.925(11) |
| c/Å | 12.974(6) | 10.355(12) |
| α/o | 90.00 | 70.530(16) |
| β/o | 92.854(7) | 71.154(15) |
| γ/o | 90.00 | 80.698(16) |
| U/Å3 | 1379.1(11) | 746.3(15) |
| μ/mm-1(Mo-Kα) | 1.413 | 1.192 |
| Dx/g cm-3 | 1.392 | 1.581 |
| Reflections/parameters | 2434/173 | 2546/204 |
| Independent reflections (Rint) | 1203 (0.0478) | 2024 (0.0163) |
| F(000) | 608 | 368 |
| Goodness of fit on F2 | 0.957 | 0.984 |
| R1, wR2 [I ≥ 2σ(I)]a) | 0.0554, 0.1469 | 0.0520, 0.1384 |
| R1, wR2 (all data)a) | 0.1135, 0.1670 | 0.0645, 0.1480 |
| 1 | 2 | ||
|---|---|---|---|
| Ni(1)-O(1) | 1.865(5) | Co(1)-O(1) | 1.888(4) |
| Ni(1)-O(2) | 1.858(4) | Co(1)-O(2) | 1.876(4) |
| Ni(2)-O(3) | 1.861(5) | Co(1)-O(3) | 1.894(4) |
| Ni(1)-O(4) | 1.855(5) | Co(1)-O(4) | 1.908(4) |
| O(2)-Ni(1)-O(2A) | 180.000(1) | Co(1)-O(5) | 1.928(3) |
| O(2)-Ni(1)-O(1) | 90.5(2) | Co(1)-O(6) | 1.958(3) |
| O(2A)-Ni(1)-O(1) | 89.5(2) | O(2)-Co(1)-O(1) | 89.57(17) |
| O(2)-Ni(1)-O(1A) | 89.5(2) | O(2)-Co(1)-O(3) | 92.62(16) |
| O(2A)-Ni(1)-O(1A) | 90.5(2) | O(1)-Co(1)-O(3) | 89.21(16) |
| O(1)-Ni(1)-O(1A) | 180.000(1) | O(2)-Co(1)-O(4) | 89.99(17) |
| O(4A)-Ni(2)-O(4) | 180.000(1) | O(1)-Co(1)-O(4) | 178.30(14) |
| O(4A)-Ni(2)-O(3A) | 90.2(2) | O(3)-Co(1)-O(4) | 89.17(16) |
| O(4)-Ni(2)-O(3) | 89.8(2) | O(2)-Co(1)-O(5) | 97.61(4) |
| O(4A)-Ni(2)-O(3) | 89.8(2) | O(1)-Co(1)-O(5) | 91.51(15) |
| O(4)-Ni(2)-O(3) | 90.2(2) | O(3)-Co(1)-O(5) | 169.76(13) |
| O(3A)-Ni(2)-O(3) | 180.000(1) | O(4)-Co(1)-O(5) | 90.17(15) |
| O(2)-Ni(1)-O(2A) | 180.000(1) | O(2)-Co(1)-O(6) | 165.34(14) |
| O(2)-Ni(1)-O(1) | 90.5(2) | O(1)-Co(1)-O(6) | 89.54(16) |
| O(2A)-Ni(1)-O(1) | 89.5(2) | O(3)-Co(1)-O(6) | 102.00(14) |
| O(2)-Ni(1)-O(1A) | 89.5(2) | O(4)-Co(1)-O(6) | 91.30(15) |
| O(2A)-Ni(1)-O(1A) | 90.5(2) | O(5)-Co(1)-O(6) | 67.80(12) |
| O(2)-Co(1)-N(1) | 131.70(15) | ||
| O(1)-Co(1)-N(1) | 90.30(16) | ||
| O(3)-Co(1)-N(1) | 135.68(15) | ||
| O(4)-Co(1)-N(1) | 91.21(16) | ||
| O(5)-Co(1)-N(1) | 34.12(13) | ||
| Complexes | D-H···A | D-H | H···A | D···A | D-H···A |
|---|---|---|---|---|---|
| 1 | O5-H1···O1 | 0.878 | 2.23 | 3.072 | 161 |
| 2 | O8-H12··· O6 | 0.882 | 2.47 | 3.189 | 139 |
| 2 | O9-H13···O7 | 0.884 | 1.94 | 2.794 | 161 |
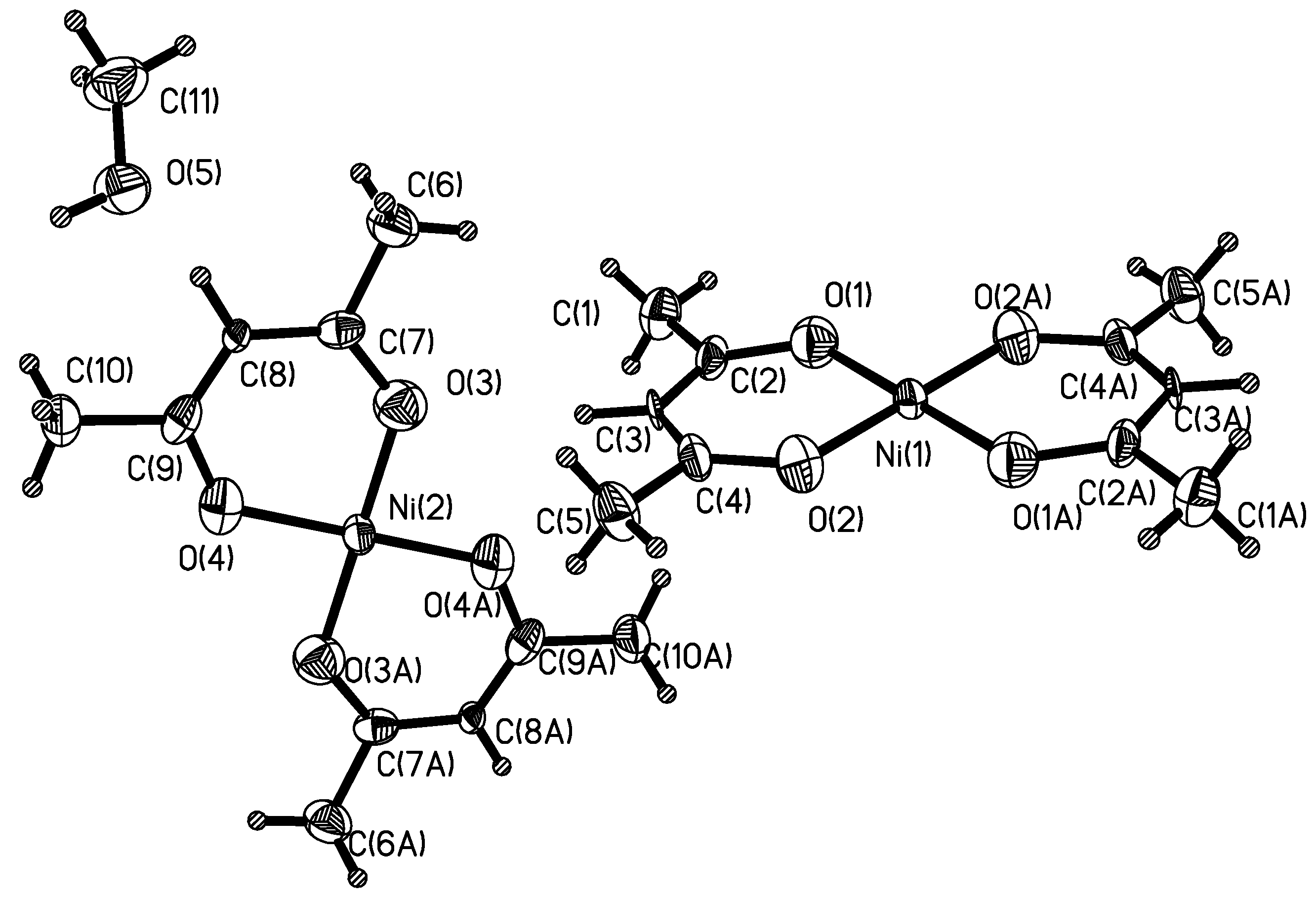
Structural Descriptions of Ni(acac)]2·0.5CH3OH (1) and [Co(acac)2NO3]·2H2O (2).



Experimental
General
Synthesis of [Ni(acac)2]·0.5 CH3OH (1)
Synthesis of [Co(acac)2NO3]·2H2O (2)
Data collection, Crystal Structure Determination and Refinement
References
- Smith, M. E.; Andersen, R. A. Me5C5Ni(acac): A Monomeric, Paramagnetic, 18-Electron, Spin-Equilibrium Molecule. J. Am. Chem. Soc. 1996, 118, 11119–11128. [Google Scholar]
- Silvana, C. N.; Kulbinder, K. B.; Paul, J. T.; John, T. W. Synthesis and physical and structural characterization of Ag(I) complexes supported by non-fluorinated β-diketonate and related ancillary ligands. Polyhedron 2002, 21, 1289–1297. [Google Scholar]
- Zhu, W.G.; Qing, J.; Lu, Z.Y.; Wei, X.Q.; Xie, M.G. Novel binuclear uropium β-diketone chelate used as red emitter in organic electroluminescent device. Thin Solid Films 2000, 363, 167–169. [Google Scholar]
- Song, D.T.; Liu, S.F.; Wang, R.Y.; Wang, S.N. Syntheses and structures of new blue luminescent B(III) and Al(III) complexes: BPh2(acac-azain) and Al(CH3)(acac-azain)2, acac-azain=1-N-7-azaindolyl-1,3-butanedionato. J. Organomet. Chem. 2001, 631, 175–180. [Google Scholar]
- Mehrotra, R.C.; Bohra, R.; Gaur, D.P. Metal-Diketonates and Allied Derivatives; Academic Press: New York, 1978. [Google Scholar]
- Sievers, R.E.; Eisentraut, K.J.; Springer, C.S., Jr. Lanthanide/Actinide Chemistry; Gould, R.F., Ed.; American Chemical Society: Washington, DC, 1967; Chapter 11. [Google Scholar]
- Alexander, D. G.; Alexander, P. S.; Marina, I. S. Common and less-common coordination modes of the typical chelating and heteroaromatic ligands. Coord. Chem. Rev. 1998, 17331–77. [Google Scholar]
- Burgess, J.; Fawcett, J.; Russell, D. R.; Gilani, S. R. Monomeric bis(2,4-pentanedionato)cobalt(II). Acta Cryst. 2000, C56, 649–650. [Google Scholar]
- Zhou, X. F.; Han, A. J.; Chu, D.B.; Huang, Z. X. Electrochemical synthesis and a redetermination of Ni(acac)2(H2O)2·H2O. Acta Cryst. 2001, E57, 506–508. [Google Scholar]
- Sample Availability: Available from the authors.
© 2004 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Qu, Y.; Zhu, H.-L.; You, Z.-L.; Tan, M.-Y. Synthesis and Characterization of the Complexes of Pentane-2,4-dione with Nickel(II) and Cobalt(III): [Ni(acac)2]·0.5CH3OH and [Co(acac)2NO3]·2H2O (acac = Pentane-2,4-dione). Molecules 2004, 9, 949-956. https://doi.org/10.3390/91100949
Qu Y, Zhu H-L, You Z-L, Tan M-Y. Synthesis and Characterization of the Complexes of Pentane-2,4-dione with Nickel(II) and Cobalt(III): [Ni(acac)2]·0.5CH3OH and [Co(acac)2NO3]·2H2O (acac = Pentane-2,4-dione). Molecules. 2004; 9(11):949-956. https://doi.org/10.3390/91100949
Chicago/Turabian StyleQu, Yang, Hai-Liang Zhu, Zhong-Lu You, and Min-Yu Tan. 2004. "Synthesis and Characterization of the Complexes of Pentane-2,4-dione with Nickel(II) and Cobalt(III): [Ni(acac)2]·0.5CH3OH and [Co(acac)2NO3]·2H2O (acac = Pentane-2,4-dione)" Molecules 9, no. 11: 949-956. https://doi.org/10.3390/91100949
APA StyleQu, Y., Zhu, H.-L., You, Z.-L., & Tan, M.-Y. (2004). Synthesis and Characterization of the Complexes of Pentane-2,4-dione with Nickel(II) and Cobalt(III): [Ni(acac)2]·0.5CH3OH and [Co(acac)2NO3]·2H2O (acac = Pentane-2,4-dione). Molecules, 9(11), 949-956. https://doi.org/10.3390/91100949




