Accelerating in Situ Endothelialisation of Cardiovascular Bypass Grafts
Abstract
:1. Introduction

2. Materials for Graft Construction
3. Potential Recruits for in Situ Endothelialisation
4. Endothelial Progenitor Cells: Mobilization
| Factor | Application | Model | Outcome/Effects |
|---|---|---|---|
| EPO [38] | Myocardial infarction was induced in wild-type mice and EPCs with or without EPO were introduced into myocardium around the infarct. | Murine | Enhanced transplanted EPC survival and improved EPC mobilization. |
| SDF-1 [39] | SDF-1α was fixed onto heparin, which was conjugated onto microfibrous vascular grafts. | Murine | Increased recruitment of EPCs. Also recruited SMPCs. |
| G-CSF [40] | Heparin-immobilized, decellularized grafts were implanted and subcutaneous injections introduced to subjects. | Murine | EPCs increased and endothelialisation enhanced. Significantly smaller hyperplastic neointima area. |
| HMG-CoA reductase inhibitors (e.g., Atorvastatin) [41] | Subjects orally administered atorvastatin. | Murine | Circulating EPCs increased, angiogenesis induced and functional recovery improved. |
| Angiotensin II antagonists [33] | Subjects treated with irbesartan. | Hypertensive-hypercholestrolaemic hamster | EPC mobilization increased. |
| NGF [42] | Human mononuclear cells isolated and cultured with NGF. CD133+ progenitor cells were incubated with NGF and injected into mice with carotid artery injury. NGF treated TEBV implanted into injured mice. | In vitro & murine | In vitro, human EPCs form more colonies, are stimulated to differentiate into ECs and show improved migration. In vivo, mice EPC show improved mobilization and homing and TEBV endothelialisation enhanced. |
| BDNF [32] | In vitro, BDNF introduced to early and late outgrowth EPCs. BDNF-modified TEBV introduced to murine model. | In vitro & murine | In vitro, BDNF shows ability to enhance single clone formation and paracrine functions of EPCs. BDNF also helps late EPCs proliferate, migrate and differentiate. In vivo, TEBV shows greater endothelialisation than control. |
| VEGF | Covalent immobilization of VEGF onto surfaces of PLLA and PCL. | In vitro | Functionalization process created. VEGF known to increase number of EPCs [25]. |
| PPAR-γ agonist [43] | Endothelial progenitor cells from rat bone marrow were cultured with pioglitazone, a PPAR-γ agonist. | In vitro | Apoptosis of EPCs reduced. |
5. EPC and EC Adhesion and Proliferation
5.1. Biofunctionalization of Graft Surfaces to Enhance EPC and EC Adhesion and Proliferation
5.1.1. Techniques to Biofunctionalize Graft Surfaces
Surface Adsorption of Molecules
Chemical Immobilisation

5.1.2. Targets that Can Be Utilized to Biofunctionalize Graft Surfaces
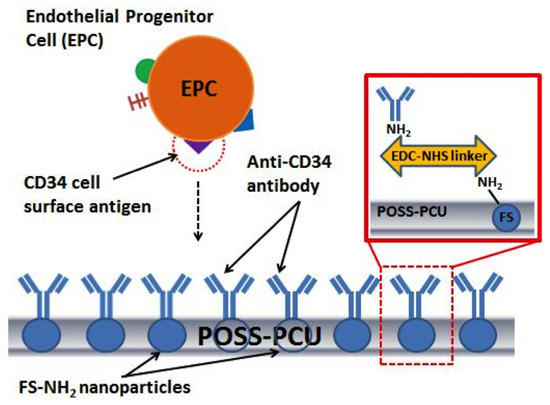
Immobilization of Antibodies onto Graft Surfaces
| Target | Application | Model | Outcome |
|---|---|---|---|
| CD133+ EPCs [60] | An ePTFE graft with an anti-CD133 antibody multilayer functionalized by heparin/collagen was developed. After being tested for surface modification stability, blood compatibility, haemolysis rate, cellular proliferation and adhesion, in vivo testing was carried out as a carotid artery transplant in a porcine model. | Porcine | Endothelialisation onset and rate improved. |
| CD34+ EPCs [63] | ePTFE grafts coated with anti-CD34 antibodies were implanted in 11 pigs between the carotid artery and internal jugular vein. | Porcine | Endothelialisation rate in 72 h increased but IH increased 4 weeks later. |
| KDR+ EPCs and Ecs [57] | Coating of glass coverslips with monoclonal mouse anti-human KDR IgG1 and then incubated with recombinant human KDR/Fc chimera before flow study. Orientation of antibody altered using adsorbed protein G. | In vitro | VEGFR-2+ HUVECs successfully captured from flow onto solid surface at sub-arterial shear rate. However, when orientation of antibody was altered, 2.5-fold greater capture efficiency observed. |
Immobilization of Proteins and Peptide Sequences onto Graft Surfaces
| Type of Ligand | Protein/Peptide | Method | Model | Outcome/Results |
|---|---|---|---|---|
| Peptides | cRGD [76] | Transplantation of aortic cRGD-coated self-expanding nitinol stent into rabbit model. | Rabbit | The cRGD peptide was shown to have improved EC adhesion and proliferation. |
| Nap-FFGRGD [72] | RGD containing molecule coated onto electrospun biodegradable PCL grafts and the grafts implanted into rabbit carotid arteries. | Rabbit | Increased endothelial coverage, decreased platelet accumulation, and increased smooth muscle remodelling. | |
| CAG [77] | Electrospun vascular graft constructed containing PCL and CAG and implanted into Sprague-Dawley rats. | Murine | Endothelialisation improved, increased expression of endothelial nitric oxide synthase, lower α-smooth muscle actin. | |
| REDV [78] | Zwitterionic carboxybetaine methacrylate and butyl methacrylate were copolymerized as coating materials, spin-coated onto substrates, and immobilized with REDV. | In vitro | Increased growth of ECs. Decreased accumulation of platelets, limited smooth muscle growth. | |
| YIGSR [79] | Poly(ethylene glycol) and a diazeniumdiolate NO donor incorporated into polyurethane together with YIGSR peptide sequence. | In vitro | Increased EC growth, decreased platelet adhesion. | |
| TPS [80] | Zwitterionic carboxybetaine methacrylate and TPS incorporated onto electrospun PCL mats. | In vitro | Improved hydrophilicity, specifically captures EPCs, decreased platelet adhesion and increased growth of vascular cells. | |
| PDAM [81] | PDAM coated on 316L stainless steel stents and tested in vitro. | In vitro | Increased HUVEC adhesion, proliferation, and migration, release of NO, and secretion of prostaglandin I(2). PDAM-modified surface shows ability to decrease the adhesion and proliferation of human umbilical artery smooth muscle cells. | |
| Collagen and MAP-RGD [82] | PCL scaffolds were first coated in collagen. The collagen-coated scaffolds were then immersed in MAP-RGD solution to immobilize the MAP-RGD. DNA quantification was used to evaluate EC proliferation. | In vitro | Highest expression level shown in the PCL/collagen/MAP-RGD group, indicative of improved endothelium sheet formation. | |
| Proteins | Fibronectin [83] | Decellularized rat aortic conduits coated with Alexa488-labelled fibronectin and implanted into Wistar rats for 8 weeks. | Murine | Accelerated endothelialisation but IH occurs after 8 weeks. |
| Laminin type-1 [51] | Laminin type-1 is covalently bound to ePTFE grafts and implanted into rats. | Murine | Increased endothelialisation and neovascularization. | |
| Collagen type-1 with fibronectin [84] | Polystyrene surfaces coated with single and double layers of collagen, fibronectin and collagen + fibronectin. | In vitro | Double coating of collagen + fibronectin shows better EC growth. | |
| ELP4 [85] | ELP4 cross-linked onto polyurethane surface, subjected to reconstituted human blood. | In vitro | Enhanced EC adhesion, EC showed organized actin cytoskeleton and enhanced endothelial nitric oxide synthase expression. Decreased platelet adhesion and activation. |

Immobilization of Nucleic Acid Sequences onto Graft Surfaces
Oligosaccharides and Phospholipids
5.2. Magnetic Homing of EPCs and ECs to Site of Endothelialisation
5.3. Micropatterning, Nanopatterning, and the Surface Physical Characteristics of Grafts

5.3.1. Electrospun Fibre Diameter and Alignment
5.3.2. Surface Roughness
5.3.3. Porosity of Graft Surfaces
5.3.4. Peptide Patterning
5.4. Shear Stress Preconditioning
6. Regulating Differentiation: From Progenitor Cells to Mature Endothelium
6.1. Materials and Topography of Grafts
6.2. Utilization of Signalling Biomolecules
6.3. Shear Stress Acting on Graft Surfaces
7. Preventing Thrombogenesis, IH Formation and Inflammation during Endothelialisation
8. Conclusions and Future Directions
Conflicts of Interest
References
- World Health Organization. World Health Statistics 2012; WHO press: Geneva, Switzerland, 2012. [Google Scholar]
- Michaels, A.D.; Chatterjee, K. Cardiology patient pages. Angioplasty versus bypass surgery for coronary artery disease. Circulation 2002, 106, e187–e190. [Google Scholar] [CrossRef]
- Sarkar, S.; Sales, K.; Hamilton, G.; Seifalian, A. Addressing thrombogenicity in vascular graft construction. J. Biomed. Mater. Res. B 2007, 82, 100–108. [Google Scholar] [CrossRef]
- Zilla, P.; Bezuidenhout, D.; Human, P. Prosthetic vascular grafts: Wrong models, wrong questions and no healing. Biomaterials 2007, 28, 5009–5027. [Google Scholar] [CrossRef] [PubMed]
- Abbott, W.M.; Callow, A.; Moore, W.; Rutherford, R.; Veith, F.; Weinberg, S. Evaluation and performance standards for arterial prostheses. J. Vasc. Surg. 1993, 17, 746–756. [Google Scholar] [CrossRef] [PubMed]
- Bunting, S.; Moncada, S.; Vane, J.R. Antithrombotic properties of vascular endothelium. Lancet 1977, 310, 1075–1076. [Google Scholar] [CrossRef]
- Patel, S.D.; Waltham, M.; Wadoodi, A.; Burnand, K.G.; Smith, A. The role of endothelial cells and their progenitors in intimal hyperplasia. Ther. Adv. Cardiovasc. Dis. 2010, 4, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Thébaud, N.B.; Pierron, D.; Bareille, R.; le Visage, C.; Letourneur, D.; Bordenave, L. Human endothelial progenitor cell attachment to polysaccharide-based hydrogels: A pre-requisite for vascular tissue engineering. J. Mater. Sci. Mater. Med. 2007, 18, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Seifalian, A.M.; Tiwari, A.; Rashid, S.T.; Salacinski, H.; Hamilton, G. Comment on: Clinical long-term results of vascular prosthesis sealed with fragmented autologous adipose tissue. Artif. Organs 2002, 26, 209–210. [Google Scholar] [CrossRef] [PubMed]
- Prasad, C.K.; Krishnan, L.K. Regulation of endothelial cell phenotype by biomimetic matrix coated on biomaterials for cardiovascular tissue engineering. Acta Biomater. 2008, 4, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Abbott, W.M.; Green, R.M.; Matsumoto, T.; Wheeler, J.R.; Miller, N.; Veith, F.J.; Suggs, W.D.; Hollier, L.; Money, S.; Garrett, H.E. Prosthetic above-knee femoropopliteal bypass grafting: Results of a multicenter randomized prospective trial. Above-Knee Femoropopliteal Study Group. J. Vasc. Surg. 1997, 25, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Seifalian, A.M.; Salacinski, H.J.; Tiwari, A.; Edwards, A.; Bowald, S.; Hamilton, G. In vivo biostability of a poly(carbonate-urea)urethane graft. Biomaterials 2003, 24, 2549–2557. [Google Scholar] [CrossRef] [PubMed]
- Ravi, S.; Chaikof, E.L. Biomaterials for vascular tissue engineering. Regen. Med. 2010, 5. [Google Scholar] [CrossRef]
- Clowes, A.W.; Kirkman, T.R.; Reidy, M.A. Mechanisms of arterial graft healing. Rapid transmural capillary ingrowth provides a source of intimal endothelium and smooth muscle in porous PTFE prostheses. Am. J. Pathol. 1986, 123, 220–230. [Google Scholar] [PubMed]
- Hibino, N.; Villalona, G.; Pietris, N.; Duncan, D.R.; Schoffner, A.; Roh, J.D.; Yi, T.; Dobrucki, L.W.; Mejias, D.; Sawh-Martinez, R.; et al. Tissue-engineered vascular grafts form neovessels that arise from regeneration of the adjacent blood vessel. FASEB J. 2011, 25, 2731–2739. [Google Scholar] [CrossRef]
- Lyman, D.J.; Stewart, S.F.; Murray-Wijelath, J.; Wijelath, E. Role of fluid dynamics on the healing of an in vivo tissue engineered vascular graft. J. Biomed. Mater. Res. B 2006, 77, 389–400. [Google Scholar] [CrossRef]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Basile, D.P.; Yoder, M.C. Circulating and tissue resident endothelial progenitor cells. J. Cell. Physiol. 2014, 229, 10–16. [Google Scholar] [PubMed]
- Yoder, M.C.; Ingram, D.A. The definition of EPCs and other bone marrow cells contributing to neoangiogenesis and tumor growth: Is there common ground for understanding the roles of numerous marrow-derived cells in the neoangiogenic process? Biochim. Biophys. Acta Rev. Cancer 2009, 1796, 50–54. [Google Scholar] [CrossRef]
- Fadini, G.P.; Losordo, D.; Dimmeler, S. Critical reevaluation of endothelial progenitor cell phenotypes for therapeutic and diagnostic use. Circ. Res. 2012, 110, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Hirschi, K.K.; Ingram, D.A.; Yoder, M.C. Assessing identity, phenotype, and fate of endothelial progenitor cells. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1584–1595. [Google Scholar] [CrossRef] [PubMed]
- Reed, D.M.; Foldes, G.; Harding, S.E.; Mitchell, J.A. Stem cell-derived endothelial cells for cardiovascular disease: A therapeutic perspective. Br. J. Clin. Pharmacol. 2013, 75, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Kiechl, S.; Patel, S.; Oberhollenzer, F.; Weger, S.; Mayr, A.; Metzler, B.; Reindl, M.; Hu, Y.; Willeit, J.; et al. Endothelial progenitor cells, cardiovascular risk factors, cytokine levels and atherosclerosis—Results from a large population-based study. PLoS One 2007, 2, e975. [Google Scholar] [CrossRef]
- Aicher, A.; Zeiher, A.M.; Dimmeler, S. Mobilizing endothelial progenitor cells. Hypertension 2005, 45, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Hattori, K.; Dias, S.; Heissig, B.; Hackett, N.R.; Lyden, D.; Tateno, M.; Hicklin, D.J.; Zhu, Z.; Witte, L.; Crystal, R.G.; et al. Vascular endothelial growth factor and angiopoietin-1 stimulate postnatal hematopoiesis by recruitment of vasculogenic and hematopoietic stem cells. J. Exp. Med. 2001, 193, 1005–1014. [Google Scholar] [CrossRef]
- Sobrino, T.; Perez-Mato, M.; Brea, D.; Rodriguez-Yanez, M.; Blanco, M.; Castillo, J. Temporal profile of molecular signatures associated with circulating endothelial progenitor cells in human ischemic stroke. J. Neurosci. Res. 2012, 90, 1788–1793. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.F.; Du, Z.D.; Chen, Z.; Han, Z.C.; He, Z.X. Granulocyte colony-stimulating factor attenuates monocrotaline-induced pulmonary hypertension by upregulating endothelial progenitor cells via the nitric oxide system. Exp. Ther. Med. 2013, 6, 1402–1408. [Google Scholar] [PubMed]
- Hibbert, B.; Simard, T.; Ramirez, F.D.; Pourdjabbar, A.; Raizman, J.E.; Maze, R.; Wilson, K.R.; Hawken, S.; O’Brien, E.R. The effect of statins on circulating endothelial progenitor cells in humans: A systematic review. J. Cardiovasc. Pharmacol. 2013, 62, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Gensch, C.; Clever, Y.P.; Werner, C.; Hanhoun, M.; Bohm, M.; Laufs, U. The PPAR-γ agonist pioglitazone increases neoangiogenesis and prevents apoptosis of endothelial progenitor cells. Atherosclerosis 2007, 192, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Heeschen, C. Erythropoietin is a potent physiologic stimulus for endothelial progenitor cell mobilization. Blood 2003, 102, 1340–1346. [Google Scholar] [CrossRef] [PubMed]
- Jadhao, C.S.; Bhatwadekar, A.D.; Jiang, Y.; Boulton, M.E.; Steinle, J.J.; Grant, M.B. Nerve growth factor promotes endothelial progenitor cell-mediated angiogenic responses. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2030–2037. [Google Scholar] [CrossRef]
- Zeng, W.; Wen, C.; Wu, Y.; Li, L.; Zhou, Z.; Mi, J.; Chen, W.; Yang, M.; Hou, C.; Sun, J.; et al. The use of BDNF to enhance the patency rate of small-diameter tissue-engineered blood vessels through stem cell homing mechanisms. Biomaterials 2012, 33, 473–484. [Google Scholar] [CrossRef]
- Georgescu, A.; Alexandru, N.; Nemecz, M.; Titorencu, I.; Popov, D. Irbesartan administration therapeutically influences circulating endothelial progenitor cell and microparticle mobilization by involvement of pro-inflammatory cytokines. Eur. J. Pharmacol. 2013, 711, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Rankin, S.M. Chemokines and adult bone marrow stem cells. Immunol. Lett. 2012, 145, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.C.; Chang, S.J.; Chueh, Y.N.; Huang, T.S.; Huang, P.H.; Cheng, S.M.; Tsai, T.N.; Chen, J.W.; Wang, H.W. Distinct angiogenesis roles and surface markers of early and late endothelial progenitor cells revealed by functional group analyses. BMC Genomics 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Walpoth, B.H.; Zammaretti, P.; Cikirikcioglu, M.; Khabiri, E.; Djebaili, M.K.; Pache, J.C.; Tille, J.C.; Aggoun, Y.; Morel, D.; Kalangos, A.; et al. Enhanced intimal thickening of expanded polytetrafluoroethylene grafts coated with fibrin or fibrin-releasing vascular endothelial growth factor in the pig carotid artery interposition model. J. Thorac. Cardiovasc. Surg. 2007, 133, 1163–1170. [Google Scholar] [CrossRef]
- Sheng, J.; Cai, W.W.; Fang, N.Y.; Wang, S.Q.; Wu, J.J. Role of stromal-derived factor-1α/CXCR4 in neo-intimal repair. Cardiovasc. J. Afr. 2011, 22, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Hu, R.; Lv, L.; Ling, L.; Jiang, S. Erythropoietin improves the efficiency of endothelial progenitor cell therapy after myocardial infarction in mice: Effects on transplanted cell survival and autologous endothelial progenitor cell mobilization. J. Surg. Res. 2012, 176, e47–e55. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Wang, A.; Tang, Z.; Henry, J.; Lee, B.; Zhu, Y.; Yuan, F.; Huang, F.; Li, S. The effect of stromal cell-derived factor-1alpha/heparin coating of biodegradable vascular grafts on the recruitment of both endothelial and smooth muscle progenitor cells for accelerated regeneration. Biomaterials 2012, 33, 8062–8074. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Liu, Z.; Li, K.; Qiao, W.; Jiang, X.; Ran, F.; Qiao, T.; Liu, C. Beneficial effects of granulocyte-colony stimulating factor on small-diameter heparin immobilized decellularized vascular graft. J. Biomed. Mater. Res. A 2010, 95, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Sun, L.; Tian, Y.; Li, Z.; Wei, H.; Wang, D.; Yang, Z.; Chen, J.; Zhang, J.; Jiang, R. Effects of atorvastatin in the regulation of circulating EPCs and angiogenesis in traumatic brain injury in rats. J. Neurol. Sci. 2012, 319, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Yuan, W.; Li, L.; Mi, J.; Xu, S.; Wen, C.; Zhou, Z.; Xiong, J.; Sun, J.; Ying, D.; et al. The promotion of endothelial progenitor cells recruitment by nerve growth factors in tissue-engineered blood vessels. Biomaterials 2010, 31, 1636–1645. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.F.; Wang, L.; Yuan, H.J.; Ma, Y.H.; Wang, Y.F.; Hu, Z.Y.; Su, Y.; Zhao, Z.G. PPAR-γ agonist pioglitazone prevents apoptosis of endothelial progenitor cells from rat bone marrow. Cell Biol. Int. 2013, 37, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Bu, X.; Yan, Y.; Zhang, Z.; Gu, X.; Wang, M.; Gong, A.; Sun, X.; Cui, Y.; Zeng, Y. Properties of extracellular matrix-like scaffolds for the growth and differentiation of endothelial progenitor cells. J. Surg. Res. 2010, 164, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Pike, D.B.; Cai, S.; Pomraning, K.R.; Firpo, M.A.; Fisher, R.J.; Shu, X.Z.; Prestwich, G.D.; Peattie, R.A. Heparin-regulated release of growth factors in vitro and angiogenic response in vivo to implanted hyaluronan hydrogels containing VEGF and bFGF. Biomaterials 2006, 27, 5242–5251. [Google Scholar] [CrossRef] [PubMed]
- De Visscher, G.; Mesure, L.; Meuris, B.; Ivanova, A.; Flameng, W. Improved endothelialization and reduced thrombosis by coating a synthetic vascular graft with fibronectin and stem cell homing factor SDF-1α. Acta Biomater. 2012, 8, 1330–1338. [Google Scholar]
- Krijgsman, B.; Seifalian, A.M.; Salacinski, H.J.; Tai, N.R.; Punshon, G.; Fuller, B.J.; Hamilton, G. An assessment of covalent grafting of RGD peptides to the surface of a compliant poly(carbonate-urea)urethane vascular conduit versus conventional biological coatings: Its role in enhancing cellular retention. Tissue Eng. 2002, 8, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Nguyen, K.T.; Brilakis, E.S.; Yang, J.; Fuh, E.; Banerjee, S. Enhanced endothelialization of a new stent polymer through surface enhancement and incorporation of growth factor-delivering microparticles. J. Cardiovasc. Transl. Res. 2012, 5, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.; Farhatnia, Y.; Goh, D.; G, N.; de Mel, A.; Lim, J.; Teoh, S.H.; Malkovskiy, A.V.; Chawla, R.; Rajadas, J.; et al. Surface modification of a polyhedral oligomeric silsesquioxane poly(carbonate-urea) urethane (POSS-PCU) nanocomposite polymer as a stent coating for enhanced capture of endothelial progenitor cells. Biointerphases 2013, 8. [Google Scholar] [CrossRef]
- Tan, A.; Goh, D.; Farhatnia, Y.; G, N.; Lim, J.; Teoh, S.H.; Rajadas, J.; Alavijeh, M.S.; Seifalian, A.M. An anti-CD34 antibody-functionalized clinical-grade POSS-PCU nanocomposite polymer for cardiovascular stent coating applications: A preliminary assessment of endothelial progenitor cell capture and hemocompatibility. PLoS One 2013, 8, e77112. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.K.; Kleinert, L.B.; Patula-Steinbrenner, V. Accelerated neovascularization and endothelialization of vascular grafts promoted by covalently bound laminin type 1. J. Biomed. Mater. Res. A 2011, 99, 67–73. [Google Scholar] [CrossRef] [PubMed]
- NA. Covalent Coupling. 2002. Available online: http://www.bangslabs.com/sites/default/files/bangs/docs/pdf/205.pdf (accessed on 9 December 2013).
- Xu, H.; Deshmukh, R.; Timmons, R.; Nguyen, K.T. Enhanced endothelialization on surface modified poly(l-lactic acid) substrates. Tissue Eng. A 2011, 17, 865–876. [Google Scholar] [CrossRef]
- Wulf, K.; Teske, M.; Lobler, M.; Luderer, F.; Schmitz, K.P.; Sternberg, K. Surface functionalization of poly(epsilon-caprolactone) improves its biocompatibility as scaffold material for bioartificial vessel prostheses. J. Biomed. Mater. Res. B 2011, 98, 89–100. [Google Scholar] [CrossRef]
- Avci-Adali, M.; Ziemer, G.; Wendel, H.P. Induction of EPC homing on biofunctionalized vascular grafts for rapid in vivo self-endothelialization—A review of current strategies. Biotechnol. Adv. 2010, 28, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, Z.; Wang, Z.; Feng, S.; Xu, H.; Zhao, Q.; Wang, S.; Fang, J.; Qiao, M.; Kong, D. Immobilization of anti-CD31 antibody on electrospun poly(varepsilon-caprolactone) scaffolds through hydrophobins for specific adhesion of endothelial cells. Colloids Surf. B 2011, 85, 32–39. [Google Scholar] [CrossRef]
- Markway, B.D.; McCarty, O.J.; Marzec, U.M.; Courtman, D.W.; Hanson, S.R.; Hinds, M.T. Capture of flowing endothelial cells using surface-immobilized anti-kinase insert domain receptor antibody. Tissue Eng. C 2008, 14, 97–105. [Google Scholar] [CrossRef]
- Jackson, D.E. The unfolding tale of PECAM-1. FEBS Lett. 2003, 540, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Hristov, M.; Erl, W.; Weber, P.C. Endothelial progenitor cells: Mobilization, differentiation, and homing. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1185–1189. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Zhang, P.; Sun, X.; Gong, F.; Yang, S.; Shen, L.; Huang, Z.; Wang, C. Synthetic ePTFE grafts coated with an anti-CD133 antibody-functionalized heparin/collagen multilayer with rapid in vivo endothelialization properties. ACS Appl. Mater. Interfaces 2013, 5, 7360–7369. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liu, T.; Chen, J.; Maitz, M.; Chen, C.; Huang, N. Influence of a layer-by-layer-assembled multilayer of anti-CD34 antibody, vascular endothelial growth factor, and heparin on the endothelialization and anticoagulation of titanium surface. J. Biomed. Mater. Res. A 2013, 101, 1144–1157. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Zhu, H.; Lu, S.; Yang, X. Accelerated endothelialization with a polymer-free sirolimus-eluting antibody-coated stent. J. Mater. Sci. Mater. Med. 2013, 24, 2601–2609. [Google Scholar] [CrossRef] [PubMed]
- Rotmans, J.I.; Heyligers, J.M.; Verhagen, H.J.; Velema, E.; Nagtegaal, M.M.; de Kleijn, D.P.; de Groot, F.G.; Stroes, E.S.; Pasterkamp, G. In vivo cell seeding with anti-CD34 antibodies successfully accelerates endothelialization but stimulates intimal hyperplasia in porcine arteriovenous expanded polytetrafluoroethylene grafts. Circulation 2005, 112, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Peichev, M.; Naiyer, A.J.; Pereira, D.; Zhu, Z.; Lane, W.J.; Williams, M.; Oz, M.C.; Hicklin, D.J.; Witte, L.; Moore, M.A.S.; et al. Expression of VEGFR-2 and AC133 by circulating human CD34+ cells identifies a population of functional endothelial precursors. Blood 2000, 95, 952–958. [Google Scholar] [PubMed]
- Yeh, E.T.H.; Zhang, S.; Wu, H.D.; Korbling, M.; Willerson, J.T.; Estrov, Z. Transdifferentiation of human peripheral blood CD34+-enriched cell population into cardiomyocytes, endothelial cells, and smooth muscle cells in vivo. Circulation 2003, 108, 2070–2073. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Feng, S.C.; Pang, X.J.; Li, W.X.; Bi, Y.H.; Zhao, Q.; Zhang, S.X.; Wang, Y.; Feng, B. Combination coating of chitosan and anti-CD34 antibody applied on sirolimus-eluting stents can promote endothelialization while reducing neointimal formation. BMC Cardiovasc. Disord. 2012, 12. [Google Scholar] [CrossRef]
- Lee, J.M.; Choe, W.; Kim, B.K.; Seo, W.W.; Lim, W.H.; Kang, C.K.; Kyeong, S.; Eom, K.D.; Cho, H.J.; Kim, Y.C.; et al. Comparison of endothelialization and neointimal formation with stents coated with antibodies against CD34 and vascular endothelial-cadherin. Biomaterials 2012, 33, 8917–8927. [Google Scholar] [CrossRef] [PubMed]
- Yoon, M.; Hwang, H.J.; Kim, J.H. Immobilization of antibodies on the self-assembled monolayer by antigen-binding site protection and immobilization kinetic control. J. Biomed. Sci. Eng. 2011, 4. [Google Scholar] [CrossRef]
- Caiado, F.; Dias, S. Endothelial progenitor cells and integrins: Adhesive needs. Fibrogenes. Tissue Repair 2012, 5, 4:1–4:13. [Google Scholar]
- Xiao, Y.; Truskey, G.A. Effect of receptor-ligand affinity on the strength of endothelial cell adhesion. Biophys. J. 1996, 71, 2869–2884. [Google Scholar] [CrossRef] [PubMed]
- Walter, D.H.; Rittig, K.; Bahlmann, F.H.; Kirchmair, R.; Silver, M.; Murayama, T.; Nishimura, H.; Losordo, D.W.; Asahara, T.; Isner, J.M. Statin therapy accelerates reendothelialization: A novel effect involving mobilization and incorporation of bone marrow-derived endothelial progenitor cells. Circulation 2002, 105, 3017–3024. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wang, Z.; Song, L.; Zhao, Q.; Zhang, J.; Li, D.; Wang, S.; Han, J.; Zheng, X.L.; Yang, Z.; et al. Endothelialization and patency of RGD-functionalized vascular grafts in a rabbit carotid artery model. Biomaterials 2012, 33, 2880–2891. [Google Scholar] [CrossRef] [PubMed]
- Hersel, U.; Dahmen, C.; Kessler, H. RGD modified polymers: Biomaterials for stimulated cell adhesion and beyond. Biomaterials 2003, 24, 4385–4415. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Cortes, J.; Mrksich, M. The platelet integrin alphaIIbbeta3 binds to the RGD and AGD motifs in fibrinogen. Chem. Biol. 2009, 16, 990–1000. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.H.; Chu, W.P.; Lin, Y.S.; Chiang, Y.L.; Chen, D.C.; Tsai, C.L. The effect of an RGD-containing fusion protein CBD-RGD in promoting cellular adhesion. J. Biotechnol. 2004, 111, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Joner, M.; Cheng, Q.; Schonhofer-Merl, S.; Lopez, M.; Neubauer, S.; Mas-Moruno, C.; Laufer, B.; Kolodgie, F.D.; Kessler, H.; Virmani, R. Polymer-free immobilization of a cyclic RGD peptide on a nitinol stent promotes integrin-dependent endothelial coverage of strut surfaces. J. Biomed. Mater. Res. B 2012, 100, 637–645. [Google Scholar] [CrossRef]
- Kuwabara, F.; Narita, Y.; Yamawaki-Ogata, A.; Kanie, K.; Kato, R.; Satake, M.; Kaneko, H.; Oshima, H.; Usui, A.; Ueda, Y. Novel small-caliber vascular grafts with trimeric Peptide for acceleration of endothelialization. Ann. Thorac. Surg. 2012, 93, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Wei, Y.; Liu, X.; Wang, J.; Ren, K.; Ji, J. Zwitterionic polycarboxybetaine coating functionalized with REDV peptide to improve selectivity for endothelial cells. J. Biomed. Mater. Res. A 2012, 100, 1387–1397. [Google Scholar] [CrossRef] [PubMed]
- Taite, L.J.; Yang, P.; Jun, H.W.; West, J.L. Nitric oxide-releasing polyurethane-PEG copolymer containing the YIGSR peptide promotes endothelialization with decreased platelet adhesion. J. Biomed. Mater. Res. B 2008, 84, 108–116. [Google Scholar] [CrossRef]
- Li, Q.; Wang, Z.; Zhang, S.; Zheng, W.; Zhao, Q.; Zhang, J.; Wang, L.; Wang, S.; Kong, D. Functionalization of the surface of electrospun poly(epsilon-caprolactone) mats using zwitterionic poly(carboxybetaine methacrylate) and cell-specific peptide for endothelial progenitor cells capture. Mater. Sci. Eng. C 2013, 33, 1646–1653. [Google Scholar] [CrossRef]
- Yang, Z.; Tu, Q.; Zhu, Y.; Luo, R.; Li, X.; Xie, Y.; Maitz, M.F.; Wang, J.; Huang, N. Mussel-inspired coating of polydopamine directs endothelial and smooth muscle cell fate for re-endothelialization of vascular devices. Adv. Healthc. Mater. 2012, 1, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Kang, T.-Y.; Hong, J.M.; Kim, B.J.; Cha, H.J.; Cho, D.-W. Enhanced endothelialization for developing artificial vascular networks with a natural vessel mimicking the luminal surface in scaffolds. Acta Biomater. 2013, 9, 4716–4725. [Google Scholar] [CrossRef] [PubMed]
- Assmann, A.; Delfs, C.; Munakata, H.; Schiffer, F.; Horstkotter, K.; Huynh, K.; Barth, M.; Stoldt, V.R.; Kamiya, H.; Boeken, U.; et al. Acceleration of autologous in vivo recellularization of decellularized aortic conduits by fibronectin surface coating. Biomaterials 2013, 34, 6015–6026. [Google Scholar] [CrossRef] [PubMed]
- Sgarioto, M.; Vigneron, P.; Patterson, J.; Malherbe, F.; Nagel, M.D.; Egles, C. Collagen type I together with fibronectin provide a better support for endothelialization. C. R. Biol. 2012, 335, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Blit, P.H.; McClung, W.G.; Brash, J.L.; Woodhouse, K.A.; Santerre, J.P. Platelet inhibition and endothelial cell adhesion on elastin-like polypeptide surface modified materials. Biomaterials 2011, 32, 5790–5800. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.P. Filamentous fusion phage: Novel expression vectors that display cloned antigens on the virion surface. Science 1985, 228, 1315–1317. [Google Scholar] [CrossRef] [PubMed]
- Filova, E.; Brynda, E.; Riedel, T.; Chlupac, J.; Vandrovcova, M.; Svindrych, Z.; Lisa, V.; Houska, M.; Pirk, J.; Bacakova, L. Improved adhesion and differentiation of endothelial cells on surface-attached fibrin structures containing extracellular matrix proteins. J. Biomed. Mater. Res. A 2013, 102, 698–712. [Google Scholar] [CrossRef] [PubMed]
- Heilshorn, S.C.; DiZio, K.A.; Welsh, E.R.; Tirrell, D.A. Endothelial cell adhesion to the fibronectin CS5 domain in artificial extracellular matrix proteins. Biomaterials 2003, 24, 4245–4252. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, C.; Lu, X.; Yin, M. Co-polypeptides of 3,4-dihydroxyphenylalanine and l-lysine to mimic marine adhesive protein. Biomaterials 2007, 28, 3456–3468. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.M.; Lee, Y.B.; Kim, S.J.; Kang, J.K.; Park, J.C.; Jang, W.; Shin, H. Mussel-inspired immobilization of vascular endothelial growth factor (VEGF) for enhanced endothelialization of vascular grafts. Biomacromolecules 2012, 13, 2020–2028. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Liu, D.; Yang, Z. A brief review of methods for terminal functionalization of DNA. Methods 2013, 67, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, T.; Ohno, J.; Hayakawa, T.; Kawaguchi, M.; Inoue, Y.; Takeda, S.; Toyoda, M.; Okahata, Y. Mold fabrication and biological assessment of porous DNA-chitosan complexes. J. Biomed. Mater. Res. B 2009, 91, 746–754. [Google Scholar] [CrossRef]
- Schleicher, M.; Hansmann, J.; Elkin, B.; Kluger, P.J.; Liebscher, S.; Huber, A.J.; Fritze, O.; Schille, C.; Muller, M.; Schenke-Layland, K.; et al. Oligonucleotide and parylene surface coating of polystyrene and ePTFE for improved endothelial cell attachment and hemocompatibility. Int. J. Biomater. 2012, 2012. [Google Scholar] [CrossRef]
- Tuerk, C.; Gold, L. Systemiatic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DN polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.; Wendel, H.P.; Scheideler, L.; Ziemer, G.; Scheule, A.M. Aptamer-based capture molecules as a novel coating strategy to promote cell adhesion. J. Cell. Mol. Med. 2005, 9, 731–736. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhang, Z.; Soontornworajit, B.; Zhou, J.; Wang, Y. Cell adhesion on an artificial extracellular matrix using aptamer-functionalized PEG hydrogels. Biomaterials 2012, 33, 1353–1362. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.; Paul, A.; Harwardt, M.; Groll, J.; Reeswinkel, T.; Klee, D.; Moeller, M.; Fischer, H.; Walker, T.; Greiner, T.; et al. Immobilized DNA aptamers used as potent attractors for porcine endothelial precursor cells. J. Biomed. Mater. Res. A 2008, 84, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Strahm, Y.; Flueckiger, A.; Billinger, M.; Meier, P.; Mettler, D.; Weisser, S.; Schaffner, T.; Hess, O. Endothelial-cell-binding aptamer for coating of intracoronary stents. J. Invasive Cardiol. 2010, 22, 481–487. [Google Scholar] [PubMed]
- Fukushima, T.; Inoue, Y.; Hayakawa, T.; Taniguchi, K.; Miyazaki, K.; Okahata, Y. Preparation of and tissue response to DNA-lipid films. J. Dent. Res. 2001, 80, 1772–1776. [Google Scholar] [CrossRef] [PubMed]
- Soontornworajit, B.; Wang, Y. Nucleic acid aptamers for clinical diagnosis: Cell detection and molecular imaging. Anal. Bioanal. Chem. 2011, 399, 1591–1599. [Google Scholar] [CrossRef] [PubMed]
- Biancone, L.; Cantaluppi, V.; Duo, D.; Deregibus, M.C.; Torre, C.; Camussi, G. Role of l-selectin in the vascular homing of peripheral blood-derived endothelial progenitor cells. J. Immunol. 2004, 173, 5268–5274. [Google Scholar] [CrossRef] [PubMed]
- Suuronen, E.J.; Zhang, P.; Kuraitis, D.; Cao, X.; Melhuish, A.; McKee, D.; Li, F.; Mesana, T.G.; Veinot, J.P.; Ruel, M. An acellular matrix-bound ligand enhances the mobilization, recruitment and therapeutic effects of circulating progenitor cells in a hindlimb ischemia model. FASEB J. 2009, 23, 1447–1458. [Google Scholar] [CrossRef] [PubMed]
- Camci-Unal, G.; Aubin, H.; Ahari, A.F.; Bae, H.; Nichol, J.W.; Khademhosseini, A. Surface-modified hyaluronic acid hydrogels to capture endothelial progenitor cells. Soft Matter. 2010, 6, 5120–5126. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.; Flanagan, C.E.; Masters, K.S. Differential support of cell adhesion and growth by copolymers of polyurethane with hyaluronic acid. J. Biomed. Mater. Res. A 2013, 101, 2870–2882. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.W.; Masters, K.S. Regulation of polyurethane hemocompatibility and endothelialization by tethered hyaluronic acid oligosaccharides. Biomaterials 2009, 30, 5341–5351. [Google Scholar] [CrossRef] [PubMed]
- Soletti, L.; Nieponice, A.; Hong, Y.; Ye, S.H.; Stankus, J.J.; Wagner, W.R.; Vorp, D.A. In vivo performance of a phospholipid-coated bioerodable elastomeric graft for small-diameter vascular applications. J. Biomed. Mater. Res. A 2011, 96, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Tardif, K.; Cloutier, I.; Miao, Z.; Lemieux, C.; St-Denis, C.; Winnik, F.M.; Tanguay, J.F. A phosphorylcholine-modified chitosan polymer as an endothelial progenitor cell supporting matrix. Biomaterials 2011, 32, 5046–5055. [Google Scholar] [CrossRef] [PubMed]
- De Mel, A.; Punshon, G.; Ramesh, B.; Sarkar, S.; Darbyshire, A.; Hamilton, G.; Seifalian, A.M. In situ endothelialization potential of a biofunctionalised nanocomposite biomaterial-based small diameter bypass graft. Biomed. Mater. Eng. 2009, 19, 317–331. [Google Scholar] [PubMed]
- Chen, J.; Huang, N.; Ma, B.; Maitz, M.F.; Wang, J.; Li, J.; Li, Q.; Zhao, Y.; Xiong, K.; Liu, X. Guidance of stem cells to a target destination in vivo by magnetic nanoparticles in a magnetic field. ACS Appl. Mater. Interfaces 2013, 5, 5976–5985. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.J.; Kim, J.Y.; Lee, H.J.; Kim, K.H.; Kim, T.Y.; Lee, C.S.; Lee, H.C.; Park, T.H.; Kim, H.S.; Park, Y.B. Magnetic bionanoparticle enhances homing of endothelial progenitor cells in mouse hindlimb ischemia. Korean Circ. J. 2012, 42, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Fayol, D.; le Visage, C.; Ino, J.; Gazeau, F.; Letourneur, D.; Wilhelm, C. Design of biomimetic vascular grafts with magnetic endothelial patterning. Cell Transplant. 2013, 22, 2105–2118. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, A.; Punshon, G.; Kidane, A.; Hamilton, G.; Seifalian, A.M. Magnetic beads (Dynabead) toxicity to endothelial cells at high bead concentration: Implication for tissue engineering of vascular prosthesis. Cell Biol. Toxicol. 2003, 19, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Nazneen, F.; Herzog, G.; Arrigan, D.W.M.; Caplice, N.; Benvenuto, P.; Galvin, P.; Thompson, M. Surface chemical and physical modification in stent technology for the treatment of coronary artery disease. J. Biomed. Mater. Res. B 2012, 100B, 1989–2014. [Google Scholar] [CrossRef]
- Oh, S.; Brammer, K.S.; Li, Y.S.; Teng, D.; Engler, A.J.; Chien, S.; Jin, S. Stem cell fate dictated solely by altered nanotube dimension. Proc. Natl. Acad. Sci. USA 2009, 106, 2130–2135. [Google Scholar] [CrossRef] [PubMed]
- Simon, C.; Palmaz, J.C.; Sprague, E.A. Influence of topography on endothelialization of stents: Clues for new designs. J. Long-Term Eff. Med. Implants 2000, 10, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.S.; Gadegaard, N.; Dalby, M.J.; Riehle, M.O.; Wilkinson, C.D.; Aitchison, G. Cells react to nanoscale order and symmetry in their surroundings. IEEE Trans. Nanobiosci. 2004, 3, 61–65. [Google Scholar] [CrossRef]
- Anderson, D.E.; Hinds, M.T. Endothelial cell micropatterning: Methods, effects, and applications. Ann. Biomed. Eng. 2011, 39, 2329–2345. [Google Scholar] [CrossRef] [PubMed]
- Chong, D.S.; Lindsey, B.; Dalby, M.J.; Gadegaard, N.; Seifalian, A.M.; Hamilton, G. Luminal surface engineering, “micro and nanopatterning”: Potential for self endothelialising vascular grafts? Eur. J. Vasc. Endovasc. Surg. 2014, 47, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Du, F.; Zhao, W.; Zhang, M.; Mao, H.; Kong, D.; Yang, J. The synergistic effect of aligned nanofibers and hyaluronic acid modification on endothelial cell behavior for vascular tissue engineering. J. Nanosci. Nanotechnol. 2011, 11, 6718–6725. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Ahn, G.; Kim, J.Y.; Cho, D.W. Evaluating cell proliferation based on internal pore size and 3D scaffold architecture fabricated using solid freeform fabrication technology. J. Mater. Sci. Mater. Med. 2010, 21, 3195–3205. [Google Scholar] [CrossRef] [PubMed]
- Olbrich, M.; Punshon, G.; Frischauf, I.; Salacinski, H.J.; Rebollar, E.; Romanin, C.; Seifalian, A.M.; Heitz, J. UV surface modification of a new nanocomposite polymer to improve cytocompatibility. J. Biomater. Sci. Polym. Ed. 2007, 18, 453–468. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Bayer, I.S.; Biris, A.S.; Wang, T.; Dervishi, E.; Faupel, F. Advances in top-down and bottom-up surface nanofabrication: Techniques, applications & future prospects. Adv. Colloid Interface Sci. 2012, 170, 2–27. [Google Scholar] [CrossRef] [PubMed]
- Ingavle, G.C.; Leach, J.K. Advancements in electrospinning of polymeric nanofibrous scaffolds for tissue engineering. Tissue Eng. B 2013, 20, 277–293. [Google Scholar] [CrossRef]
- Inoguchi, H.; Tanaka, T.; Maehara, Y.; Matsuda, T. The effect of gradually graded shear stress on the morphological integrity of a huvec-seeded compliant small-diameter vascular graft. Biomaterials 2007, 28, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Vaz, C.M.; van Tuijl, S.; Bouten, C.V.; Baaijens, F.P. Design of scaffolds for blood vessel tissue engineering using a multi-layering electrospinning technique. Acta Biomater. 2005, 1, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Whited, B.M.; Rylander, M.N. The influence of electrospun scaffold topography on endothelial cell morphology, alignment, and adhesion in response to fluid flow. Biotechnol. Bioeng. 2014, 111, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Heath, D.E.; Lannutti, J.J.; Cooper, S.L. Electrospun scaffold topography affects endothelial cell proliferation, metabolic activity, and morphology. J. Biomed. Mater. Res. A 2010, 94, 1195–1204. [Google Scholar] [PubMed]
- Kurpinski, K.T.; Stephenson, J.T.; Janairo, R.R.; Lee, H.; Li, S. The effect of fiber alignment and heparin coating on cell infiltration into nanofibrous PLLA scaffolds. Biomaterials 2010, 31, 3536–3542. [Google Scholar] [CrossRef] [PubMed]
- Hadjizadeh, A.; Ajji, A.; Jolicoeur, M.; Liberelle, B.; de Crescenzo, G. Effects of electrospun nanostructure versus microstructure on human aortic endothelial cell behavior. J. Biomed. Nanotechnol. 2013, 9, 1195–1209. [Google Scholar] [CrossRef] [PubMed]
- Sundararaghavan, H.G.; Saunders, R.L.; Hammer, D.A.; Burdick, J.A. Fiber alignment directs cell motility over chemotactic gradients. Biotechnol. Bioeng. 2013, 110, 1249–1254. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Yao, C.; Yang, L.; Webster, T.J. Decreased platelet adhesion and enhanced endothelial cell functions on nano and submicron-rough titanium stents. Tissue Eng. A 2012, 18, 1389–1398. [Google Scholar] [CrossRef]
- Dickinson, L.E.; Rand, D.R.; Tsao, J.; Eberle, W.; Gerecht, S. Endothelial cell responses to micropillar substrates of varying dimensions and stiffness. J. Biomed. Mater. Res. A 2012, 100, 1457–1466. [Google Scholar] [CrossRef] [PubMed]
- Salem, A.K.; Stevens, R.; Pearson, R.G.; Davies, M.C.; Tendler, S.J.; Roberts, C.J.; Williams, P.M.; Shakesheff, K.M. Interactions of 3T3 fibroblasts and endothelial cells with defined pore features. J. Biomed. Mater. Res. 2002, 61, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, Z.; Liu, S.; Kodama, M. Pore size, tissue ingrowth, and endothelialization of small-diameter microporous polyurethane vascular prostheses. Biomaterials 2004, 25, 177–187. [Google Scholar] [CrossRef] [PubMed]
- De Valence, S.; Tille, J.C.; Giliberto, J.P.; Mrowczynski, W.; Gurny, R.; Walpoth, B.H.; Moller, M. Advantages of bilayered vascular grafts for surgical applicability and tissue regeneration. Acta Biomater. 2012, 8, 3914–3920. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Grater, S.V.; Corbellini, F.; Rinck, S.; Bock, E.; Kemkemer, R.; Kessler, H.; Ding, J.; Spatz, J.P. Impact of order and disorder in RGD nanopatterns on cell adhesion. Nano Lett. 2009, 9, 1111–1116. [Google Scholar] [CrossRef] [PubMed]
- Le Saux, G.; Magenau, A.; Bocking, T.; Gaus, K.; Gooding, J.J. The relative importance of topography and RGD ligand density for endothelial cell adhesion. PLoS One 2011, 6, e21869. [Google Scholar] [CrossRef] [PubMed]
- Akimoto, S.; Mitsumata, M.; Sasaguri, T.; Yoshida, Y. Laminar shear stress inhibits vascular endothelial cell proliferation by inducing cyclin-dependent kinase inhibitor p21(Sdi1/Cip1/Waf1). Circ. Res. 2000, 86, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Simper, D.; Stalboerger, P.G.; Panetta, C.J.; Wang, S.; Caplice, N.M. Smooth muscle progenitor cells in human blood. Circulation 2002, 106, 1199–1204. [Google Scholar] [CrossRef] [PubMed]
- Weng, Y.; Chen, J.; Tu, Q.; Li, Q.; Maitz, M.F.; Huang, N. Biomimetic modification of metallic cardiovascular biomaterials: From function mimicking to endothelialization in vivo. Interface Focus 2012, 2, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Riha, G.M.; Yang, H.; Li, M.; Yao, Q.; Chen, C. Differentiation and proliferation of endothelial progenitor cells from canine peripheral blood mononuclear cells. J. Surg. Res. 2005, 126, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Ziebart, T.; Schnell, A.; Walter, C.; Kammerer, P.W.; Pabst, A.; Lehmann, K.M.; Ziebart, J.; Klein, M.O.; Al-Nawas, B. Interactions between endothelial progenitor cells (EPC) and titanium implant surfaces. Clin. Oral Investig. 2013, 17, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Neoh, K.G.; Kang, E.T.; Poh, C.K.; Wang, W. Enhanced endothelial differentiation of adipose-derived stem cells by substrate nanotopography. J. Tissue Eng. Regen. Med. 2012, 8, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Fong, G.H.; Rossant, J.; Gertsenstein, M.; Breitman, M.L. Role of the Flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature 1995, 376, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Motwani, M.S.; Rafiei, Y.; Tzifa, A.; Seifalian, A.M. In situ endothelialization of intravascular stents from progenitor stem cells coated with nanocomposite and functionalized biomolecules. Biotechnol. Appl. Biochem. 2011, 58, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Gao, Y.; Qin, J.; Kuang, C.Y.; Song, M.B.; Yu, S.Y.; Cui, B.; Chen, J.F.; Huang, L. CCN1 promotes the differentiation of endothelial progenitor cells and reendothelialization in the early phase after vascular injury. Basic Res. Cardiol. 2010, 105, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Dubey, S.; Varney, M.L.; Dave, B.J.; Singh, R.K. IL-8 directly enhanced endothelial cell survival, proliferation, and matrix metalloproteinases production and regulated angiogenesis. J. Immunol. 2003, 170, 3369–3376. [Google Scholar] [CrossRef] [PubMed]
- Obi, S.; Yamamoto, K.; Shimizu, N.; Kumagaya, S.; Masumura, T.; Sokabe, T.; Asahara, T.; Ando, J. Fluid shear stress induces arterial differentiation of endothelial progenitor cells. J. Appl. Physiol. 2009, 106, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Zhang, X.; Guan, X.; Li, H.; Li, X.; Lu, H.; Cheng, M. Shear stress augments the endothelial cell differentiation marker expression in late EPCs by upregulating integrins. Biochem. Biophys. Res. Commun. 2012, 425, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Guan, X.; Li, H.; Cui, X.; Zhang, X.; Li, X.; Jing, X.; Wu, H.; Avsar, E. Shear stress regulates late EPC differentiation via mechanosensitive molecule-mediated cytoskeletal rearrangement. PLoS One 2013, 8, e67675. [Google Scholar] [CrossRef] [PubMed]
- Ye, C.; Bai, L.; Yan, Z.Q.; Wang, Y.H.; Jiang, Z.L. Shear stress and vascular smooth muscle cells promote endothelial differentiation of endothelial progenitor cells via activation of Akt. Clin. Biomech. 2008, 23 (Suppl. 1), S118–S124. [Google Scholar] [CrossRef]
- Zeng, L.; Xiao, Q.; Margariti, A.; Zhang, Z.; Zampetaki, A.; Patel, S.; Capogrossi, M.C.; Hu, Y.; Xu, Q. HDAC3 is crucial in shear- and VEGF-induced stem cell differentiation toward endothelial cells. J. Cell Biol. 2006, 174, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.B.; Yan, Z.Q.; Yao, Q.P.; Shen, B.R.; Wang, J.Y.; Gao, L.Z.; Li, Y.Q.; Yuan, H.T.; Qi, Y.X.; Jiang, Z.L. Association of SIRT1 expression with shear stress induced endothelial progenitor cell differentiation. J. Cell. Biochem. 2012, 113, 3663–3671. [Google Scholar] [CrossRef] [PubMed]
- Morgan, J.T.; Wood, J.A.; Shah, N.M.; Hughbanks, M.L.; Russell, P.; Barakat, A.I.; Murphy, C.J. Integration of basal topographic cues and apical shear stress in vascular endothelial cells. Biomaterials 2012, 33, 4126–4135. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Tu, Q.; Wang, J.; Huang, N. The role of heparin binding surfaces in the direction of endothelial and smooth muscle cell fate and re-endothelialization. Biomaterials 2012, 33, 6615–6625. [Google Scholar] [CrossRef] [PubMed]
- Hoshi, R.A.; van Lith, R.; Jen, M.C.; Allen, J.B.; Lapidos, K.A.; Ameer, G. The blood and vascular cell compatibility of heparin-modified ePTFE vascular grafts. Biomaterials 2013, 34, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Stamler, J.S. S-Nitrosothiols in the blood: Roles, amounts, and methods of analysis. Circ. Res. 2004, 94, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Riccio, D.A.; Dobmeier, K.P.; Hetrick, E.M.; Privett, B.J.; Paul, H.S.; Schoenfisch, M.H. Nitric oxide-releasing S-nitrosothiol-modified xerogels. Biomaterials 2009, 30, 4494–4502. [Google Scholar] [CrossRef] [PubMed]
- Fleser, P.S.; Nuthakki, V.K.; Malinzak, L.E.; Callahan, R.E.; Seymour, M.L.; Reynolds, M.M.; Merz, S.I.; Meyerhoff, M.E.; Bendick, P.J.; Zelenock, G.B.; et al. Nitric oxide-releasing biopolymers inhibit thrombus formation in a sheep model of arteriovenous bridge grafts. J. Vasc. Surg. 2004, 40, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.M.; Hrabie, J.A.; Oh, B.K.; Politis, J.K.; Citro, M.L.; Keefer, L.K.; Meyerhoff, M.E. Nitric oxide releasing polyurethanes with covalently linked diazeniumdiolated secondary amines. Biomacromolecules 2006, 7, 987–994. [Google Scholar] [CrossRef] [PubMed]
- De Mel, A.; Naghavi, N.; Cousins, B.G.; Clatworthy, I.; Hamilton, G.; Darbyshire, A.; Seifalian, A.M. Nitric oxide-eluting nanocomposite for cardiovascular implants. J. Mater. Sci. Mater. Med. 2014, 25, 917–929. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goh, E.T.; Wong, E.; Farhatnia, Y.; Tan, A.; Seifalian, A.M. Accelerating in Situ Endothelialisation of Cardiovascular Bypass Grafts. Int. J. Mol. Sci. 2015, 16, 597-627. https://doi.org/10.3390/ijms16010597
Goh ET, Wong E, Farhatnia Y, Tan A, Seifalian AM. Accelerating in Situ Endothelialisation of Cardiovascular Bypass Grafts. International Journal of Molecular Sciences. 2015; 16(1):597-627. https://doi.org/10.3390/ijms16010597
Chicago/Turabian StyleGoh, Ee Teng, Eleanor Wong, Yasmin Farhatnia, Aaron Tan, and Alexander M. Seifalian. 2015. "Accelerating in Situ Endothelialisation of Cardiovascular Bypass Grafts" International Journal of Molecular Sciences 16, no. 1: 597-627. https://doi.org/10.3390/ijms16010597