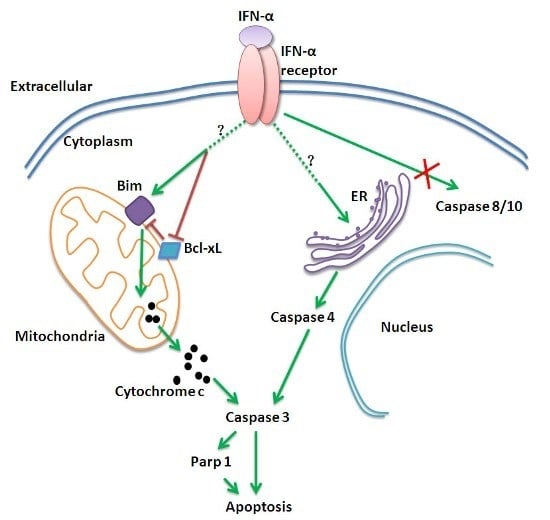
Interferon α Induces the Apoptosis of Cervical Cancer HeLa Cells by Activating both the Intrinsic Mitochondrial Pathway and Endoplasmic Reticulum Stress-Induced Pathway
Abstract
:1. Introduction
2. Results
2.1. Interferon α (IFN-α) Inhibits the Proliferation of HeLa Cells
2.2. IFN-α Induces the Apoptosis of HeLa Cells
2.3. The Mitochondrial Apoptotic Pathway Is Activated by IFN-α Treatment
2.4. IFN-α Activates the ER Stress-Induced Apoptotic Pathway but Not the Extrinsic Apoptotic Pathway in HeLa Cells
2.5. The ER Stress-Induced Apoptotic Pathway and the Mitochondrial Apoptotic Pathway Are Independently Induced by IFN-α
3. Discussion
4. Materials and Methods
4.1. Cell Lines, Cell Culture, Chemicals, and Reagents
4.2. Glucose/Lactate Assay
4.3. Cell Proliferation Assay
4.4. Flow Cytometric Analysis
4.5. Mitochondria Isolation
4.6. Real-Time Quantatitive Reverse Transcription PCR (qRT-PCR)
4.7. Western Blot Analysis
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McNab, F.; Mayer-Barber, K.; Sher, A.; Wack, A.; O’Garra, A. Type I interferons in infectious disease. Nat. Rev. Immunol. 2015, 15, 87–103. [Google Scholar] [CrossRef] [PubMed]
- Zitvogel, L.; Galluzzi, L.; Kepp, O.; Smyth, M.J.; Kroemer, G. Type I interferons in anticancer immunity. Nat. Rev. Immunol. 2015, 15, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Caraglia, M.; Dicitore, A.; Marra, M.; Castiglioni, S.; Persani, L.; Sperlongano, P.; Tagliaferri, P.; Abbruzzese, A.; Vitale, G. Type I interferons: Ancient peptides with still under-discovered anti-cancer properties. Protein Pept. Lett. 2013, 20, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Bake, V.; Roesler, S.; Eckhardt, I.; Belz, K.; Fulda, S. Synergistic interaction of smac mimetic and IFN α to trigger apoptosis in acute myeloid leukemia cells. Cancer Lett. 2014, 355, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Manna, S.K.; Mukhopadhyay, A.; Aggarwal, B.B. IFN-α suppresses activation of nuclear transcription factors NF-κB and activator protein 1 and potentiates TNF-induced apoptosis. J. Immunol. 2000, 165, 4927–4934. [Google Scholar] [CrossRef] [PubMed]
- Omori, R.; Eguchi, J.; Hiroishi, K.; Ishii, S.; Hiraide, A.; Sakaki, M.; Doi, H.; Kajiwara, A.; Ito, T.; Kogo, M.; et al. Effects of interferon-α-transduced tumor cell vaccines and blockade of programmed cell death-1 on the growth of established tumors. Cancer Gene Ther. 2012, 19, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Shigeno, M.; Nakao, K.; Ichikawa, T.; Suzuki, K.; Kawakami, A.; Abiru, S.; Miyazoe, S.; Nakagawa, Y.; Ishikawa, H.; Hamasaki, K.; et al. Interferon-α sensitizes human hepatoma cells to TRAIL-induced apoptosis through DR5 upregulation and NF-κB inactivation. Oncogene 2003, 22, 1653–1662. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Lu, Y.; Zhang, A.; Zhou, A.; Zhang, L.; Gao, L.; Zang, Y.; Tang, X.; Sun, L. Doxycycline induces apoptosis and inhibits proliferation and invasion of human cervical carcinoma stem cells. PLoS ONE 2015, 10, e0129138. [Google Scholar]
- Zuo, C.; Qiu, X.; Liu, N.; Yang, D.; Xia, M.; Liu, J.; Wang, X.; Zhu, H.; Xie, H.; Dan, H.; et al. Interferon-α and cyclooxygenase-2 inhibitor cooperatively mediates TRAIL-induced apoptosis in hepatocellular carcinoma. Exp. Cell Res. 2015, 333, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Benito, M.; Balsas, P.; Carvajal-Vergara, X.; Pandiella, A.; Anel, A.; Marzo, I.; Naval, J. Mechanism of apoptosis induced by IFN-α in human myeloma cells: Role of Jak1 and Bim and potentiation by rapamycin. Cell Signal. 2007, 19, 844–854. [Google Scholar] [CrossRef] [PubMed]
- Herzer, K.; Hofmann, T.G.; Teufel, A.; Schimanski, C.C.; Moehler, M.; Kanzler, S.; Schulze-Bergkamen, H.; Galle, P.R. IFN-α-induced apoptosis in hepatocellular carcinoma involves promyelocytic leukemia protein and TRAIL independently of p53. Cancer Res. 2009, 69, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Krasagakis, K.; Kruger-Krasagakis, S.; Tzanakakis, G.N.; Darivianaki, K.; Stathopoulos, E.N.; Tosca, A.D. Interferon-α inhibits proliferation and induces apoptosis of merkel cell carcinoma in vitro. Cancer Investig. 2008, 26, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Lesinski, G.B.; Raig, E.T.; Guenterberg, K.; Brown, L.; Go, M.R.; Shah, N.N.; Lewis, A.; Quimper, M.; Hade, E.; Young, G.; et al. IFN-α and bortezomib overcome Bcl-2 and Mcl-1 overexpression in melanoma cells by stimulating the extrinsic pathway of apoptosis. Cancer Res. 2008, 68, 8351–8360. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Dong, Z.R.; Guo, Z.Y.; Wang, C.H.; Tang, Z.Y.; Qu, S.F.; Chen, Z.T.; Li, X.W.; Zhi, X.T. Aspirin enhances IFN-α-induced growth inhibition and apoptosis of hepatocellular carcinoma via JAK1/STAT1 pathway. Cancer Gene Ther. 2013, 20, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Trimble, C.L.; Frazer, I.H. Development of therapeutic HPV vaccines. Lancet Oncol. 2009, 10, 975–980. [Google Scholar] [CrossRef]
- Lee, S.J.; Yang, A.; Wu, T.C.; Hung, C.F. Immunotherapy for human papillomavirus-associated disease and cervical cancer: Review of clinical and translational research. J. Gynecol. Oncol. 2016, 27, e51. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; El-Deiry, W.S. Overview of cell death signaling pathways. Cancer Biol. Ther. 2005, 4, 139–163. [Google Scholar] [CrossRef] [PubMed]
- Klener, P., Jr.; Andera, L.; Klener, P.; Necas, E.; Zivny, J. Cell death signalling pathways in the pathogenesis and therapy of haematologic malignancies: Overview of therapeutic approaches. Folia Biol. 2006, 52, 119–136. [Google Scholar]
- Plati, J.; Bucur, O.; Khosravi-Far, R. Apoptotic cell signaling in cancer progression and therapy. Integr. Biol. 2011, 3, 279–296. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhang, X.Q.; Dinney, C.N.; Benedict, W.F. Direct cytotoxicity produced by adenoviral-mediated interferon α gene transfer in interferon-resistant cancer cells involves ER stress and caspase 4 activation. Cancer Gene Ther. 2011, 18, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.G.; White, M.C.; Grimaldi, M. Stressed to death: Targeting endoplasmic reticulum stress response induced apoptosis in gliomas. Curr. Pharm. Des. 2011, 17, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Xiong, S.; Mu, T.; Wang, G.; Jiang, X. Mitochondria-mediated apoptosis in mammals. Protein Cell 2014, 5, 737–749. [Google Scholar] [CrossRef] [PubMed]
- Ashkenazi, A. Targeting the extrinsic apoptosis pathway in cancer. Cytokine Growth Factor Rev. 2008, 19, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Krelin, Y.; Zhang, L.; Kang, T.B.; Appel, E.; Kovalenko, A.; Wallach, D. Caspase-8 deficiency facilitates cellular transformation in vitro. Cell Death Differ. 2008, 15, 1350–1355. [Google Scholar] [CrossRef] [PubMed]
- Sayers, T.J. Targeting the extrinsic apoptosis signaling pathway for cancer therapy. Cancer Immunol. Immunother. 2011, 60, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Wallach, D.; Kang, T.B.; Kovalenko, A. The extrinsic cell death pathway and the elan mortel. Cell Death Differ. 2008, 15, 1533–1541. [Google Scholar] [CrossRef] [PubMed]
- Bekisz, J.; Baron, S.; Balinsky, C.; Morrow, A.; Zoon, K.C. Antiproliferative properties of type I and type II interferon. Pharmaceuticals 2010, 3, 994–1015. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.H.; Reynolds, C.P. Bcl-2 inhibitors: Targeting mitochondrial apoptotic pathways in cancer therapy. Clin. Cancer Res. 2009, 15, 1126–1132. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, K.; Garcia-Saez, A.J. Mitochondrial alterations in apoptosis. Chem. Phys. Lipids 2014, 181, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Hitomi, J.; Katayama, T.; Eguchi, Y.; Kudo, T.; Taniguchi, M.; Koyama, Y.; Manabe, T.; Yamagishi, S.; Bando, Y.; Imaizumi, K.; et al. Involvement of caspase-4 in endoplasmic reticulum stress-induced apoptosis and Aβ-induced cell death. J. Cell Biol. 2004, 165, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Obeng, E.A.; Boise, L.H. Caspase-12 and caspase-4 are not required for caspase-dependent endoplasmic reticulum stress-induced apoptosis. J. Biol. Chem. 2005, 280, 29578–29587. [Google Scholar] [CrossRef] [PubMed]
- Hampson, L.; Martin-Hirsch, P.; Hampson, I.N. An overview of early investigational drugs for the treatment of human papilloma virus infection and associated dysplasia. Expert Opin. Investig. Drugs 2015, 24, 1529–1537. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, B.H.; Ramos, M.D.; Michelin, M.A.; Murta, E.F.C. Progress in the use of immunotherapy to treat uterine cervical cancer. Tumori 2009, 95, 1–7. [Google Scholar] [PubMed]
- Van de Wall, S.; Nijman, H.W.; Daemen, T. HPV-specific immunotherapy: Key role for immunomodulators. Anticancer Agents Med. Chem. 2014, 14, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Kurabayashi, A.; Inoue, K.; Fukuhara, H.; Karashima, T.; Fukata, S.; Kawada, C.; Shuin, T.; Furihata, M. Combination with third-generation bisphosphonate (YM529) and interferon-α can inhibit the progression of established bone renal cell carcinoma. Cancer Sci. 2015, 106, 1092–1099. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Zhao, M.; Teng, Y.; Zhang, Y.; Hou, K.; Jiang, Y.; Yang, X.; Shang, H.; Qu, X.; Liu, Y. Interferon-α sensitizes human gastric cancer cells to TRAIL-induced apoptosis via activation of the c-CBL-dependent MAPK/ERK pathway. Cancer Biol. Ther. 2011, 12, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Nihal, M.; Wu, J.; Wood, G.S. Methotrexate inhibits the viability of human melanoma cell lines and enhances Fas/Fas-ligand expression, apoptosis and response to interferon-α: Rationale for its use in combination therapy. Arch. Biochem. Biophys. 2014, 563, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Alao, H.; Jake Liang, T. Alternative interferons and immunomodulators in the treatment of hepatitis C. Liver Int. 2014, 34 (Suppl. S1), 133–138. [Google Scholar] [CrossRef] [PubMed]
- Caraglia, M.; Marra, M.; Pelaia, G.; Maselli, R.; Caputi, M.; Marsico, S.A.; Abbruzzese, A. α-interferon and its effects on signal transduction pathways. J. Cell. Physiol. 2005, 202, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Nakao, Y.; Iwasaka, T.; Pater, A.; Sugimori, H. Retinoic acid and interferon-α effects on cell growth and differentiation in cervical carcinoma cell lines. Obstet. Gynecol. 2001, 98, 332–340. [Google Scholar] [CrossRef] [PubMed]








| Gene Name | GenBank ID | Forward Primer (5′→3′) | Reverse Primer (5′→3′) | Size of Product (bp) |
|---|---|---|---|---|
| β-Actin | BC009275 | TCCATCATGAAGTGTGACGT | CTCAGGAGGAGCAATGATCT | 161 |
| Caspase 4 | EF636667 | TTGCTTTCTGCTCTTCAACG | GTGTGATGAAGATAGAGCCCATT | 72 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, W.-Y.; Cao, C.; Liu, L. Interferon α Induces the Apoptosis of Cervical Cancer HeLa Cells by Activating both the Intrinsic Mitochondrial Pathway and Endoplasmic Reticulum Stress-Induced Pathway. Int. J. Mol. Sci. 2016, 17, 1832. https://doi.org/10.3390/ijms17111832
Shi W-Y, Cao C, Liu L. Interferon α Induces the Apoptosis of Cervical Cancer HeLa Cells by Activating both the Intrinsic Mitochondrial Pathway and Endoplasmic Reticulum Stress-Induced Pathway. International Journal of Molecular Sciences. 2016; 17(11):1832. https://doi.org/10.3390/ijms17111832
Chicago/Turabian StyleShi, Wei-Ye, Cheng Cao, and Li Liu. 2016. "Interferon α Induces the Apoptosis of Cervical Cancer HeLa Cells by Activating both the Intrinsic Mitochondrial Pathway and Endoplasmic Reticulum Stress-Induced Pathway" International Journal of Molecular Sciences 17, no. 11: 1832. https://doi.org/10.3390/ijms17111832