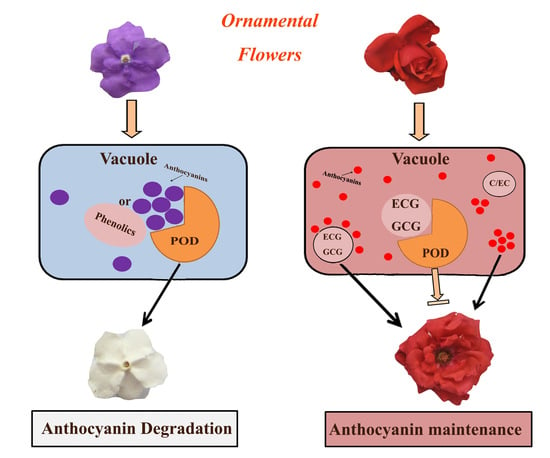
Characterization of Active Anthocyanin Degradation in the Petals of Rosa chinensis and Brunfelsia calycina Reveals the Effect of Gallated Catechins on Pigment Maintenance
Abstract
:1. Introduction
2. Results
2.1. Anthocyanin Degradation Was Found in B. calycina but Not in R. chinensis during Flower Development
2.2. High H2O2-Dependent Anthocyanin Degradation Enzyme Activity Was Found in the Petals of B. calycina but Not in R. chinensis during Flower Development
2.3. Peroxidase and Polyphenol Oxidase Activities during the Flower Development
2.4. Low pH and High Levels of Tannins Were Found for the Anthocyanin Accumulating Cells of R. chinensis but Not B. calycina
2.5. Tannin Contents in the Flowers Was Found to be Related to the Preservation of Anthocyanins during Flower Development
2.6. ECG and GCG, but not Catechin (C) and EC, Showed High Efficacy of Inhibition on POD Activities and Increased the Colour Intensity of the Anthocyanins In Vitro
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Measurement of Anthocyanin Content
4.3. Measurement of Total Phenolic and Tannin Content
4.4. Tissue Cell Sap pH Measurement and In Vivo Vacuolar Acidity Prediction by Neutral Red Staining and Microscopy
4.5. Vanillin Staining of Tannins in the Petals
4.6. Quantification of Tannin Constituents by HPLC
4.7. Extraction of Crude Enzyme, SDS-PAGE and Immunodetection
4.8. Peroxidase Activity Assay
4.9. Polyphenol Oxidase Activity Assay
4.10. Anthocyanin Degradation Enzyme (ADE) Substrate Preparation and Activity Assay
4.11. POD Purification from B. calycina Flower Petals
4.12. Measurement of IC50 and Km Values of Tannin Constituents on B. calycina POD Activity
4.13. Determination of the Effect of Tannin Constituents on Colour Intensities of Anthocyanins
4.14. Statistical Analyses
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Passeri, V.; Koes, R.; Quattrocchio, M.F. New Challenges for the Design of High Value Plant Products: Stabilization of Anthocyanins in Plant Vacuoles. Front. Plant. Sci. 2016, 7, 153. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Tulipani, S.; Casoli, T.; Di Stefano, G.; González-Paramás, A.M.; Santos-Buelga, C.; Busco, F.; Quiles, J.L.; Cordero, M.D.; et al. One-month strawberry-rich anthocyanin supplementation ameliorates cardiovascular risk, oxidative stress markers and platelet activation in humans. J. Nutr. Biochem. 2014, 25, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Song, X.; Zhang, D.; Zhou, F.; Wang, D.; Wei, Y.; Gao, F.; Xie, L.; Jia, G.; Wu, W.; et al. Blueberry anthocyanins: Protection against ageing and light-induced damage in retinal pigment epithelial cells. Brit. J. Nutr. 2012, 108, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Petroni, K.; Tonelli, C. Recent advances on the regulation of anthocyanin synthesis in reproductive organs. Plant. Sci. 2011, 181, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Oren-Shamir, M. Does anthocyanin degradation play a significant role in determining pigment concentration in plants? Plant. Sci. 2009, 177, 310–316. [Google Scholar] [CrossRef]
- Zhang, Z.; Pang, X.; Ji, Z.; Jiang, Y. Role of anthocyanin degradation in litchi pericarp browning. Food Chem. 2001, 75, 217–221. [Google Scholar] [CrossRef]
- Zhang, Z.; Pang, X.; Duan, X.; Ji, Z.; Jiang, Y. Role of peroxidase in anthocyanin degradation in litchi fruit pericarp. Food Chem. 2005, 90, 47–52. [Google Scholar] [CrossRef]
- Vaknin, H.; Bar-Akiva, A.; Ovadia, R.; Nissim-Levi, A.; Forer, I.; Weiss, D.; Oren-Shamir, M. Active anthocyanin degradation in Brunfelsia calycina (yesterday-today-tomorrow) flowers. Planta 2005, 222, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Cheynier, V.; Souquet, J.M.; Kontek, A.; Moutounet, M. Anthocyanin degradation in oxidising grape musts. J. Sci. Food Agric. 1994, 66, 283–288. [Google Scholar] [CrossRef]
- Kader, F.; Haluk, J.; Nicolas, J.; Metche, M. Degradation of Cyanidin 3-Glucoside by Blueberry Polyphenol Oxidase: Kinetic Studies and Mechanisms. J. Agric. Food Chem. 1998, 46, 3060–3065. [Google Scholar] [CrossRef]
- Jiang, Y. Role of anthocyanins, polyphenol oxidase and phenols in lychee pericarp browning. J. Sci. Food Agric. 2000, 80, 305–310. [Google Scholar] [CrossRef]
- Sarni, P.; Fulcrand, H.; Souillol, V.; Souquet, J.; Cheynier, V. Mechanisms of anthocyanin degradation in grape must-like model solutions. J. Sci. Food Agric. 1995, 69, 385–391. [Google Scholar] [CrossRef]
- Kader, F.; Rovel, B.; Girardin, M.; Metche, M. Mechanism of browning in fresh highbush blueberry fruit (Vaccinium corymbosum L). Partial purification and characterisation of blueberry polyphenol oxidase. J. Sci. Food Agric. 1997, 73, 513–516. [Google Scholar] [CrossRef]
- Pang, X.; Huang, X.; Yang, X.; Ji, Z.; Zhang, Z. Role of polyphenol oxidase in anthocyanin degradation of lychee pericarp. Sci. Agric. Sin. 2008, 41, 540–545. [Google Scholar]
- Fang, Z.; Zhang, M.; Sun, Y.; Sun, J. Polyphenol oxidase from bayberry (Myrica rubra Sieb. et Zucc.) and its role in anthocyanin degradation. Food Chem. 2007, 103, 268–273. [Google Scholar] [CrossRef]
- Chisari, M.; Barbagallo, R.N.; Spagna, G. Characterization of polyphenol oxidase and peroxidase and influence on browning of cold stored strawberry fruit. J. Agric. Food Chem. 2007, 55, 3469–3476. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Zhang, X.; Luo, H.; Zhou, J.; Gong, Y.; Li, W.; Shi, Z.; He, Q.; Wu, Q.; Li, L.; et al. An intracellular laccase is responsible for epicatechin-mediated anthocyanin degradation in litchi fruit pericarp. Plant. Physiol. 2015, 169, 2391–2408. [Google Scholar] [CrossRef] [PubMed]
- Bar-Akiva, A.; Ovadia, R.; Rogachev, I.; Bar-Or, C.; Bar, E.; Freiman, Z.; Nissim-Levi, A.; Gollop, N.; Lewinsohn, E.; Aharoni, A.; et al. Metabolic networking in Brunfelsia calycina petals after flower opening. J. Exp. Bot. 2010, 61, 1393–1403. [Google Scholar] [CrossRef] [PubMed]
- Zipor, G.; Duarte, P.; Carqueijeiro, I.; Shahar, L.; Ovadia, R.; Teper Bamnolker, P.; Eshel, D.; Levin, Y.; Doron Faigenboim, A.; Sottomayor, M.; et al. In planta anthocyanin degradation by a vacuolar class III peroxidase in Brunfelsia calycina flowers. New Phytol. 2015, 205, 653–665. [Google Scholar] [CrossRef] [PubMed]
- Schmitzer, V.; Veberic, R.; Osterc, G.; Stampar, F. Changes in the phenolic concentration during flower development of rose ‘KORcrisett’. J. Am. Soc. Hortic. Sci. 2009, 134, 491–496. [Google Scholar]
- Ferrante, A.; Vernieri, P.; Tognoni, F.; Serra, G. Changes in abscisic acid and flower pigments during floral senescence of petunia. Biol. Plant. 2006, 50, 581. [Google Scholar] [CrossRef]
- Mazza, G.; Miniati, E. Anthocyanins in Fruits, Vegetables, and Grains; Taylor & Francis Group: University of Michigan, Ann Arbor, MI, USA, 1993; p. 362. [Google Scholar]
- Darias-Martían, J.; Martían-Luis, B.; Carrillo-López, M.; Lamuela-Raventós, R.; Diaz-Romero, C.; Boulton, R. Effect of caffeic acid on the color of red wine. J. Agric. Food Chem. 2002, 50, 2062–2067. [Google Scholar] [CrossRef]
- Talcott, S.T.; Brenes, C.H.; Pires, D.M.; Del Pozo-Insfran, D. Phytochemical stability and color retention of copigmented and processed muscadine grape juice. J. Agric. Food Chem. 2003, 51, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Sasaki, N.; Ohmiya, A. Biosynthesis of plant pigments: Anthocyanins, betalains and carotenoids. Plant. J. 2008, 54, 733–749. [Google Scholar] [CrossRef] [PubMed]
- Février, H.; Le Quéré, J.M.; Le Bail, G.; Guyot, S. Polyphenol profile, PPO activity and pH variation in relation to colour changes in a series of red-fleshed apple juices. LWT Food Sci. Technol. 2016. [Google Scholar] [CrossRef]
- Zipor, G.; Oren-Shamir, M. Do vacuolar peroxidases act as plant caretakers? Plant. Sci. 2013, 199, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D.; Sang, S.; Yang, C.S. Biotransformation of green tea polyphenols and the biological activities of those metabolites. Mol. Pharm. 2007, 4, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, L.; Kim, S.; Hagerman, A.E.; Lü, J. Anti-cancer, anti-diabetic and other pharmacologic and biological activities of penta-galloyl-glucose. Pharm. Res.-Dordr. 2009, 26, 2066–2080. [Google Scholar] [CrossRef] [PubMed]
- Sadik, C.D.; Sies, H.; Schewe, T. Inhibition of 15-lipoxygenases by flavonoids: Structure-activity relations and mode of action. Biochem. Pharmacol. 2003, 65, 773–781. [Google Scholar] [CrossRef]
- Wang, X.; Song, K.; Guo, Q.; Tian, W. The galloyl moiety of green tea catechins is the critical structural feature to inhibit fatty-acid synthase. Biochem. Pharmacol. 2003, 66, 2039–2047. [Google Scholar] [CrossRef]
- Ishii, T.; Mori, T.; Tanaka, T.; Mizuno, D.; Yamaji, R.; Kumazawa, S.; Nakayama, T.; Akagawa, M. Covalent modification of proteins by green tea polyphenol (−)-epigallocatechin-3-gallate through autoxidation. Free Radical. Biol. Med. 2008, 45, 1384–1394. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Ishikawa, M.; Miyoshi, N.; Yasunaga, M.; Akagawa, M.; Uchida, K.; Nakamura, Y. Catechol type polyphenol is a potential modifier of protein sulfhydryls: Development and application of a new probe for understanding the dietary polyphenol actions. Chem. Res. Toxicol. 2009, 22, 1689–1698. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, X.; Lu, G.; Picinich, S.C. Cancer prevention by tea: Animal studies, molecular mechanisms and human relevance. Nat. Rev. Cancer 2009, 9, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Kiss, A.; Bécsi, B.; Kolozsvári, B.; Komáromi, I.; Kövér, K.E.; Erdődi, F. Epigallocatechin-3-gallate and penta-O-galloyl-β-d-glucose inhibit protein phosphatase-1. FEBS J. 2013, 280, 612–626. [Google Scholar] [CrossRef] [PubMed]
- Kamiyama, O.; Sanae, F.; Ikeda, K.; Higashi, Y.; Minami, Y.; Asano, N.; Adachi, I.; Kato, A. In vitro inhibition of α-glucosidases and glycogen phosphorylase by catechin gallates in green tea. Food Chem. 2010, 122, 1061–1066. [Google Scholar] [CrossRef]
- Yilmazer-Musa, M.; Griffith, A.M.; Michels, A.J.; Schneider, E.; Frei, B. Grape seed and tea extracts and catechin 3-gallates are potent inhibitors of α-amylase and α-glucosidase activity. J. Agric. Food Chem. 2012, 60, 8924–8929. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Dey, S.K.; Saha, C. Inhibition of catalase by tea catechins in free and cellular state: A biophysical approach. PLoS ONE 2014, 9, e102460. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.A.; Xie, D.Y.; Sharma, S.B. Proanthocyanidins-a final frontier in flavonoid research? New Phytol. 2005, 165, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.A.; Liu, C.; Jun, J.H. Metabolic engineering of anthocyanins and condensed tannins in plants. Curr. Opin. Biotechnol. 2013, 24, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Wrolstad, R.E.; Culbertson, J.D.; Cornwell, C.J.; Mattick, L.R. Detection of adulteration in blackberry juice concentrates and wines. J. Assoc. Off. Anal. Chem. 1982, 65, 1417–1423. [Google Scholar] [PubMed]
- Makkar, H.P.S.; Blümmel, M.; Borowy, N.K.; Becker, K. Gravimetric determination of tannins and their correlations with chemical and protein precipitation methods. J. Sci. Food Agric. 1993, 61, 161–165. [Google Scholar] [CrossRef]
- Hagerman, A.; Harvey-Mueller, I.; Makkar, H.P.S. Quantification of Tannins in Tree Foliage-A Laboratory Manual; FAO/IAEA: Vienna, Austria, 2000; pp. 4–7. [Google Scholar]
- Poustka, F.; Irani, N.G.; Feller, A.; Lu, Y.; Pourcel, L.; Frame, K.; Grotewold, E. A trafficking pathway for anthocyanins overlaps with the endoplasmic reticulum-to-vacuole protein-sorting route in Arabidopsis and contributes to the formation of vacuolar inclusions. Plant. Physiol. 2007, 145, 1323–1335. [Google Scholar] [CrossRef] [PubMed]
- Aastrup, S.; Outtrup, H.; Erdal, K. Location of the proanthocyanidins in the barley grain. Carlsberg Res. Commun. 1984, 49, 105–109. [Google Scholar] [CrossRef]
- Deshpande, S.S.; Cheryan, M.; Salunkhe, D.K.; Luh, B.S. Tannin analysis of food products. Crit. Rev. Food Sci. 1986, 24, 401–449. [Google Scholar] [CrossRef] [PubMed]
- Rzeppa, S.; Von Bargen, C.; Bittner, K.; Humpf, H. Analysis of flavan-3-ols and procyanidins in food samples by reversed phase high-performance liquid chromatography coupled to electrospray ionization tandem mass spectrometry (RP-HPLC-ESI-MS/MS). J. Agric. Food Chem. 2011, 59, 10594–10603. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Halaly, T.; Crane, O.; Keilin, T.; Keren-Keiserman, A.; Ogrodovitch, A.; Galbraith, D.; Or, E. Involvement of calcium signalling in dormancy release of grape buds. J. Exp. Bot 2007, 58, 3249–3262. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: New York, NY, USA, 1989. [Google Scholar]
- Chakraborty, T.K.; Das, N.; Sengupta, S.; Mukherjee, M. Accumulation of a natural substrate of laccase in gills of Pleurotus florida during sporulation. Curr. Microbiol. 2000, 41, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y. Purification and some properties of polyphenol oxidase of longan fruit. Food Chem. 1999, 66, 75–79. [Google Scholar] [CrossRef]
- Eichlerová, I.; Šnajdr, J.; Baldrian, P. Laccase activity in soils: Considerations for the measurement of enzyme activity. Chemosphere 2012, 88, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.S.; Siqueira, F.P.; Oliveira, A.E.; Fernandes, K.V. A wounding-induced PPO from cowpea (Vigna unguiculata) seedlings. Phytochemistry 2008, 69, 2297–2302. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Xuequn, P.; Yang, C.; Ji, Z.; Jiang, Y. Purification and structural analysis of anthocyanins from litchi pericarp. Food Chem. 2004, 84, 601–604. [Google Scholar] [CrossRef]







© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, H.; Deng, S.; Fu, W.; Zhang, X.; Zhang, X.; Zhang, Z.; Pang, X. Characterization of Active Anthocyanin Degradation in the Petals of Rosa chinensis and Brunfelsia calycina Reveals the Effect of Gallated Catechins on Pigment Maintenance. Int. J. Mol. Sci. 2017, 18, 699. https://doi.org/10.3390/ijms18040699
Luo H, Deng S, Fu W, Zhang X, Zhang X, Zhang Z, Pang X. Characterization of Active Anthocyanin Degradation in the Petals of Rosa chinensis and Brunfelsia calycina Reveals the Effect of Gallated Catechins on Pigment Maintenance. International Journal of Molecular Sciences. 2017; 18(4):699. https://doi.org/10.3390/ijms18040699
Chicago/Turabian StyleLuo, Honghui, Shuangfan Deng, Wei Fu, Xin Zhang, Xuelian Zhang, Zhaoqi Zhang, and Xuequn Pang. 2017. "Characterization of Active Anthocyanin Degradation in the Petals of Rosa chinensis and Brunfelsia calycina Reveals the Effect of Gallated Catechins on Pigment Maintenance" International Journal of Molecular Sciences 18, no. 4: 699. https://doi.org/10.3390/ijms18040699