Human Miro Proteins Act as NTP Hydrolases through a Novel, Non-Canonical Catalytic Mechanism
Abstract
:1. Introduction
2. Results and Discussion
2.1. Miro1 and Miro2 N-Terminal GTPase Domains Are Catalytically Active
2.2. C-Terminal GTPase Domains of Miro1 and Miro2 Exhibit Hydrolytic Activity against GTP
2.3. EF Hands do not Influence C-Terminal Domain Catalytic Activity
2.4. The Miro1 and Miro2 C-Terminal GTPase Domains Function as an NTPase, with Propensities towards GTP and ATP as Substrates
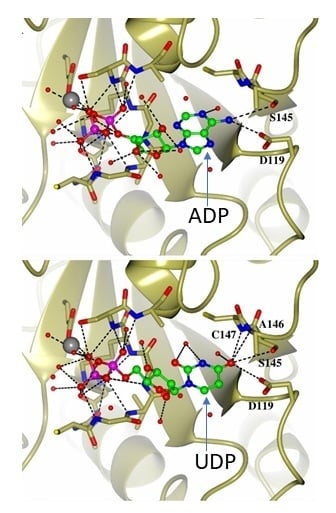
2.5. Rationale for Miro NTP Binding through Structural Modelling
2.6. Structural Comparison with Ras GTPases Suggests that Miro Proteins Utilise a Different Catalytic Mechanism
2.7. The Miro Proteins’ Catalytic Mechanism May Involve an “Internal Arginine Finger”
3. Materials and Methods
3.1. Cloning of Miro1 and Miro2 Mutants
3.2. Expression and Purification of Miro Constructs
3.3. PiColourLock™ Assessment of Phosphate Generation
3.4. Structural Modelling and Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Attwell, D.; Laughlin, S.B. An energy budget for signaling in the grey matter of the brain. J. Cereb. Blood Flow. Metab. 2001, 21, 1133–1145. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, D.G.; Budd, S.L. Mitochondria and neuronal survival. Physiol. Rev. 2000, 80, 315–360. [Google Scholar] [CrossRef] [PubMed]
- Emptage, N.J.; Reid, C.A.; Fine, A. Calcium Stores in Hippocampal Synaptic Boutons Mediate Short-Term Plasticity, Store-Operated Ca2+ Entry, and Spontaneous Transmitter Release. Neuron 2001, 29, 197–208. [Google Scholar] [CrossRef]
- Mattson, M.P.; Gleichmann, M.; Cheng, A. Mitochondria in neuroplasticity and neurological disorders. Neuron 2008, 60, 748–766. [Google Scholar] [CrossRef] [PubMed]
- Mochida, S.; Few, A.P.; Scheuer, T.; Catterall, W.A. Regulation of Presynaptic CaV2.1 Channels by Ca2+ Sensor Proteins Mediates Short-Term Synaptic Plasticity. Neuron 2008, 57, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Fransson, Å.; Ruusala, A.; Aspenström, P. Atypical Rho GTPases have roles in mitochondrial homeostasis and apoptosis. J. Biol. Chem. 2003, 278, 6495–6502. [Google Scholar] [CrossRef] [PubMed]
- Fransson, Å.; Ruusala, A.; Aspenström, P. The atypical Rho GTPases Miro-1 and Miro-2 have essential roles in mitochondrial trafficking. Biochem. Biophys. Res. Commun. 2006, 344, 500–510. [Google Scholar] [CrossRef]
- Boureux, A.; Vignal, E.; Faure, S.; Fort, P. Evolution of the Rho family of ras-like GTPases in eukaryotes. Mol. Biol. Evol. 2007, 24, 203–216. [Google Scholar] [CrossRef]
- López-Doménech, G.; Covill-Cooke, C.; Ivankovic, D.; Halff, E.F.; Sheehan, D.F.; Norkett, R.; Birsa, N.; Kittler, J.T. Miro proteins coordinate microtubule- and actin-dependent mitochondrial transport and distribution. EMBO J. 2018. [Google Scholar] [CrossRef]
- MacAskill, A.F.; Brickley, K.; Stephenson, F.A.; Kittler, J.T. GTPase dependent recruitment of Grif-1 by Miro1 regulates mitochondrial trafficking in hippocampal neurons. Mol. Cell. Neurosci. 2009, 40, 301–312. [Google Scholar] [CrossRef]
- Schwarz, T.L. Mitochondrial trafficking in neurons. Cold Spring Harb. Perspect. Biol. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Macleod, G.T.; Wellington, A.; Hu, F.; Panchumarthi, S.; Schoenfield, M.; Marin, L.; Charlton, M.P.; Atwood, H.L.; Zinsmaier, K.E. The GTPase dMiro is required for axonal transport of mitochondria to Drosophila synapses. Neuron 2005, 47, 379–393. [Google Scholar] [CrossRef] [PubMed]
- Reis, K.; Fransson, A.; Aspenstrom, P. The Miro GTPases: At the heart of the mitochondrial transport machinery. FEBS Lett. 2009, 583, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Tada, H.; Taira, Y.; Morichika, K.; Kinoshita, T. Mitochondrial trafficking through Rhot1 is involved in the aggregation of germinal granule components during primordial germ cell formation in Xenopus embryos. Dev. Growth Differ. 2016, 58, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Birsa, N.; Norkett, R.; Wauer, T.; Mevissen, T.E.; Wu, H.-C.; Foltynie, T.; Bhatia, K.; Hirst, W.D.; Komander, D.; Plun-Favreau, H. Lysine 27 ubiquitination of the mitochondrial transport protein Miro is dependent on serine 65 of the Parkin ubiquitin ligase. J. Biol. Chem. 2014, 289, 14569–14582. [Google Scholar] [CrossRef] [PubMed]
- Shlevkov, E.; Kramer, T.; Schapansky, J.; LaVoie, M.J.; Schwarz, T.L. Miro phosphorylation sites regulate Parkin recruitment and mitochondrial motility. Proc. Natl. Acad. Sci. 2016, 113, E6097–E6106. [Google Scholar] [CrossRef] [PubMed]
- Beal, M.F. Mitochondria and neurodegeneration. Novartis Found. Symp. 2007, 287, 183–192. [Google Scholar]
- Itoh, K.; Nakamura, K.; Iijima, M.; Sesaki, H. Mitochondrial dynamics in neurodegeneration. Trends Cell. Biol. 2013, 23, 64–71. [Google Scholar] [CrossRef] [Green Version]
- Babic, M.; Russo, G.J.; Wellington, A.J.; Sangston, R.M.; Gonzalez, M.; Zinsmaier, K.E. Miro’s N-Terminal GTPase Domain Is Required for Transport of Mitochondria into Axons and Dendrites. J. Neurosci. 2015, 35, 5754–5771. [Google Scholar] [CrossRef]
- Cai, Q.; Sheng, Z.-H. Mitochondrial Transport and Docking in Axons. Exp. Neurol. 2009, 218, 257–267. [Google Scholar] [CrossRef]
- Kornmann, B.; Osman, C.; Walter, P. The conserved GTPase Gem1 regulates endoplasmic reticulum-mitochondria connections. Proc. Natl. Acad. Sci. USA 2011, 108, 14151–14156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, B.E.; Bar-Sagi, D.; Nassar, N. The structural basis for the transition from Ras-GTP to Ras-GDP. Proc. Natl. Acad. Sci. USA 2002, 99, 12138–12142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Díaz, J.F.; Escalona, M.M.; Kuppens, S.; Engelborghs, Y. Role of the switch II region in the conformational transition of activation of Ha-ras-p21. Protein Sci. 2000, 9, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Caruso, M.E.; Jenna, S.; Beaulne, S.; Lee, E.H.; Bergeron, A.; Chauve, C.; Roby, P.; Rual, J.F.; Hill, D.E.; Vidal, M.; et al. Biochemical clustering of monomeric GTPases of the Ras superfamily. Mol. Cell. Proteomics 2005, 4, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Bourne, H.R.; Sanders, D.A.; McCormick, F. The GTPase superfamily: Conserved structure and molecular mechanism. Nature 1991, 349, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Babich, A.; Burkhardt, J.K. Coordinate control of cytoskeletal remodeling and calcium mobilization during T-cell activation. Immunol. Rev. 2013, 256, 80–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vlahou, G.; Elias, M.; von Kleist-Retzow, J.C.; Wiesner, R.J.; Rivero, F. The Ras related GTPase Miro is not required for mitochondrial transport in Dictyostelium discoideum. Eur. J. Cell Biol. 2011, 90, 342–355. [Google Scholar] [CrossRef]
- Walker, S.J.; Brown, H.A. Specificity of Rho insert-mediated activation of phospholipase D1. J. Biol. Chem. 2002, 277, 26260–26267. [Google Scholar] [CrossRef]
- Bustelo, X.R.; Sauzeau, V.; Berenjeno, I.M. GTP-binding proteins of the Rho/Rac family: Regulation, effectors and functions in vivo. BioEssays 2007, 29, 356–370. [Google Scholar] [CrossRef]
- Zheng, Y.; Quilliam, L.A. Activation of the Ras superfamily of small GTPases: Workshop on Exchange Factors. EMBO Rep. 2003, 4, 463–468. [Google Scholar] [CrossRef] [Green Version]
- Klosowiak, J.L.; Focia, P.J.; Chakravarthy, S.; Landahl, E.C.; Freymann, D.M.; Rice, S.E. Structural coupling of the EF hand and C-terminal GTPase domains in the mitochondrial protein Miro. EMBO Rep. 2013, 14, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Klosowiak, J.L.; Park, S.; Smith, K.P.; French, M.E.; Focia, P.J.; Freymann, D.M.; Rice, S.E. Structural insights into Parkin substrate lysine targeting from minimal Miro substrates. Sci. Rep. 2016, 6, 33019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, G.; MacDonald, M.J.; Khosravi-Far, R.; Hisaka, M.M.; Der, C.J. Alternate mechanisms of ras activation are complementary and favor and formation of ras-GTP. Oncogene 1992, 7, 283–288. [Google Scholar] [PubMed]
- Zhong, J.M.; Chen-Hwang, M.C.; Hwang, Y.W. Switching nucleotide specificity of Ha-Ras p21 by a single amino acid substitution at aspartate 119. J. Biol. Chem. 1995, 270, 10002–10007. [Google Scholar] [CrossRef] [PubMed]
- Carr-Schmid, A.; Durko, N.; Cavallius, J.; Merrick, W.C.; Kinzy, T.G. Mutations in a GTP-binding motif of eukaryotic elongation factor 1A reduce both translational fidelity and the requirement for nucleotide exchange. J. Biol. Chem. 1999, 274, 30297–30302. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.; Litt, R.J.; Richardson, C.J.; Segev, N. Requirement of nucleotide exchange factor for Ypt1 GTPase mediated protein transport. J. Cell Biol. 1995, 130, 1051–1061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartels, C.; Urlaub, H.; Luhrmann, R.; Fabrizio, P. Mutagenesis suggests several roles of Snu114p in pre-mRNA splicing. J. Biol. Chem. 2003, 278, 28324–28334. [Google Scholar] [CrossRef]
- Guilfoyle, A.; Maher, M.J.; Rapp, M.; Clarke, R.; Harrop, S.; Jormakka, M. Structural basis of GDP release and gating in G protein coupled Fe(2+) transport. EMBO J. 2009, 28, 2677–2685. [Google Scholar] [CrossRef]
- Guilfoyle, A.P.; Deshpande, C.N.; Schenk, G.; Maher, M.J.; Jormakka, M. Exploring the correlation between the sequence composition of the nucleotide binding G5 loop of the FeoB GTPase domain (NFeoB) and intrinsic rate of GDP release. Biosci. Rep. 2014, 34, e00158. [Google Scholar] [CrossRef]
- Grosdidier, A.; Zoete, V.; Michielin, O. Fast docking using the CHARMM force field with EADock DSS. J. Comput. Chem. 2011, 32, 2149–2159. [Google Scholar] [CrossRef]
- Grosdidier, A.; Zoete, V.; Michielin, O. SwissDock, a protein-small molecule docking web service based on EADock DSS. Nucleic Acids Res. 2011, 39, W270–W277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Traut, T.W. Physiological concentrations of purines and pyrimidines. Mol. Cell. Biochem. 1994, 140, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Bros, H.; Hauser, A.; Paul, F.; Niesner, R.; Infante-Duarte, C. Assessing Mitochondrial Movement Within Neurons: Manual Versus Automated Tracking Methods. Traffic 2015, 16, 906–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niescier, R.F.; Kwak, S.K.; Joo, S.H.; Chang, K.T.; Min, K.-T. Dynamics of Mitochondrial Transport in Axons. Front. Cell. Neurosci. 2016, 10, 123. [Google Scholar] [CrossRef] [PubMed]
- Fidyk, N.J.; Cerione, R.A. Understanding the catalytic mechanism of GTPase-activating proteins: Demonstration of the importance of switch domain stabilization in the stimulation of GTP hydrolysis. Biochemistry 2002, 41, 15644–15653. [Google Scholar] [CrossRef] [PubMed]
- Spoerner, M.; Herrmann, C.; Vetter, I.R.; Kalbitzer, H.R.; Wittinghofer, A. Dynamic properties of the Ras switch I region and its importance for binding to effectors. Proc. Natl. Acad. USA 2001, 98, 4944–4949. [Google Scholar] [CrossRef] [PubMed]
- Buhrman, G.; Holzapfel, G.; Fetics, S.; Mattos, C. Allosteric modulation of Ras positions Q61 for a direct role in catalysis. Proc. Natl. Acad. Sci. 2010, 107, 4931. [Google Scholar] [CrossRef] [PubMed]
- Bos, J.L.; Rehmann, H.; Wittinghofer, A. GEFs and GAPs: Critical Elements in the Control of Small G Proteins. Cell 2007, 129, 865–877. [Google Scholar] [CrossRef]
- Kötting, C.; Kallenbach, A.; Suveyzdis, Y.; Wittinghofer, A.; Gerwert, K. The GAP arginine finger movement into the catalytic site of Ras increases the activation entropy. Proc. Natl. Acad. Sci. 2008, 105, 6260. [Google Scholar] [CrossRef]
- Buhrman, G.; Wink, G.; Mattos, C. Transformation efficiency of Q61 Ras mutants linked to structural features of the switch regions. Structure 2007, 15, 1618–1629. [Google Scholar] [CrossRef]
- Kelley, L.A.; Mezulis, S.; Yates, C.M.; Wass, M.N.; Sternberg, M.J.E. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 2015, 10, 845. [Google Scholar] [CrossRef] [PubMed]
- Bunda, S.; Heir, P.; Srikumar, T.; Cook, J.D.; Burrell, K.; Kano, Y.; Lee, J.E.; Zadeh, G.; Raught, B.; Ohh, M. Src promotes GTPase activity of Ras via tyrosine 32 phosphorylation. Proc. Natl. Acad. Sci. 2014, 111, E3785–E3794. [Google Scholar] [CrossRef] [PubMed]
- Fetics, S.K.; Guterres, H.; Kearney, B.M.; Buhrman, G.; Ma, B.; Nussinov, R.; Mattos, C. Allosteric effects of the oncogenic RasQ61L mutant on Raf-RBD. Structure 2015, 23, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Muraoka, S.; Shima, F.; Araki, M.; Inoue, T.; Yoshimoto, A.; Ijiri, Y.; Seki, N.; Tamura, A.; Kumasaka, T.; Yamamoto, M.; et al. Crystal structures of the state 1 conformations of the GTP-bound H-Ras protein and its oncogenic G12V and Q61L mutants. FEBS Lett. 2012, 586, 1715–1718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daumke, O.; Weyand, M.; Chakrabarti, P.P.; Vetter, I.R.; Wittinghofer, A. The GTPase-activating protein Rap1GAP uses a catalytic asparagine. Nature 2004, 429, 197. [Google Scholar] [CrossRef] [PubMed]
- McNicholas, S.; Potterton, E.; Wilson, K.S.; Noble, M.E.M. Presenting your structures: The CCP4mg molecular-graphics software. Acta Crystallogr. D 2011, 67, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- McWilliam, H.; Li, W.; Uludag, M.; Squizzato, S.; Park, Y.M.; Buso, N.; Cowley, A.P.; Lopez, R. Analysis Tool Web Services from the EMBL-EBI. Nucleic Acids Res. 2013, 41, W597–W600. [Google Scholar] [CrossRef]
- Robert, X.; Gouet, P. Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res. 2014, 42, W320–W324. [Google Scholar] [CrossRef] [Green Version]







| Clone | Forward Primers | Reverse Primers |
|---|---|---|
| Miro1 NT-GTPase (K3-P169) | 5′-TGCCATAGCATATGAAGAAAGACGTGCGGAT-3′ | 5′-TGCCATAGCTCGAGTCAAGGATGAAGAACAGCTTTCTGTG-3′ |
| Miro1 CT-GTPase (Q415-P580) | 5′-TGCCATAGCATATGCAAAGAAATGTGTTCAGATGTAATG-3′ | 5′-TGCCATAGCTCGAGTCACGGATACATGGCCATTGTT-3′ |
| Miro1 EF-CT-GTPase (E177-R590) | 5′-TGCCATAGCATATGGAGGAGAAGGAGATGAAACCA-3′ | 5′-TGCCATAGCTCGAGTCAAGCTCTTGGGGTCAGCTTGT-3′ |
| Miro1 NT-GTPase (R3-P169) | 5′-TGCCATAGCATATGCGGGACGTGCGCATCCTGTTA-3′ | 5′-TGCCATAGCTCGAGTCATGGGATGCAGGACGGCCTT-3′ |
| Miro1 CT-GTPase (Q413-F589) | 5′-TGCCATAGCATATGCAGCGGAGCGTCCTCCTGT-3′ | 5′-TGCCATAGCTCGAGTCAGAAGGAAGAGGGATGCAGCTCT-3′ |
| Miro2 EF-CT-GTPase (E177-P586) | 5′-TGCCATAGCATATGGAGGCCAAGCAGTTGAGG-3′ | 5′-TGCCATAGCTCGAGTCAGGGATGCAGCTCTGCGTG-3′ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peters, D.T.; Kay, L.; Eswaran, J.; Lakey, J.H.; Soundararajan, M. Human Miro Proteins Act as NTP Hydrolases through a Novel, Non-Canonical Catalytic Mechanism. Int. J. Mol. Sci. 2018, 19, 3839. https://doi.org/10.3390/ijms19123839
Peters DT, Kay L, Eswaran J, Lakey JH, Soundararajan M. Human Miro Proteins Act as NTP Hydrolases through a Novel, Non-Canonical Catalytic Mechanism. International Journal of Molecular Sciences. 2018; 19(12):3839. https://doi.org/10.3390/ijms19123839
Chicago/Turabian StylePeters, Daniel T., Laura Kay, Jeyanthy Eswaran, Jeremy H. Lakey, and Meera Soundararajan. 2018. "Human Miro Proteins Act as NTP Hydrolases through a Novel, Non-Canonical Catalytic Mechanism" International Journal of Molecular Sciences 19, no. 12: 3839. https://doi.org/10.3390/ijms19123839