Delivery of Proapoptotic Agents in Glioma Cell Lines by TSPO Ligand–Dextran Nanogels
Abstract
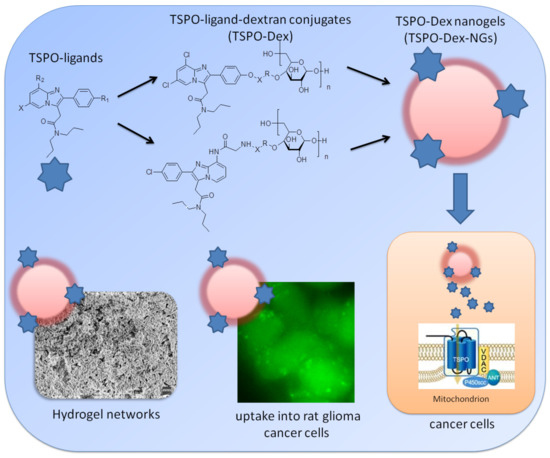
:1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization of the TSPO Ligand–Dextran Conjugates
2.2. Molecular Weight by SEC Analysis
2.3. Determination of Carboxylic Group Substitution Degree of the Carboxylated Dextran Derivatives and TSPO Ligands Content in TSPO Ligand–Dextran Conjugates
2.4. Differential Scanning Calorimetry (DSC) Studies
2.5. In Vitro Stability Studies on TSPO LigandDextran Conjugates
2.6. Preparation and Characterization of the TSPO Ligand–Dextran NGs
2.7. In Vitro Release Studies on TSPO Ligand–Dextran NGs
2.8. In Vitro Swelling Studies
2.9. Rheological Study of Viscoelastic Properties
2.10. Cytotoxicity of the TSPO Ligand–Dextran NGs
2.11. Uptake Studies of the TSPO Ligand–Dextran NGs
3. Experimental Section
3.1. Materials and Methods
3.2. Synthesis of TSPO Ligand–Dextran Conjugates
3.2.1. Synthesis of the Carboxylated Dextran Derivatives 6 and 9
3.2.2. Synthesis of the TSPO Ligand–Dextran Conjugates 7 and 10
3.2.3. Synthesis of the TSPO Ligand–Dextran Conjugates 8 and 11
3.3. Size Exclusion Chromatography Analysis
3.4. Determination of the Substitution Degree of Carboxylic Groups in the Carboxylated Dextran Derivatives
3.5. Determination of TSPO Ligand Contents on TSPO Ligand–Dextran Conjugates
3.6. Differential Scanning Calorimetry (DSC) Studies
3.7. Stability Studies of the TSPO Ligand–Dextran Conjugates in Phosphate Buffer Solution
3.8. Stability Studies of the TSPO Ligand–Dextran Conjugates in Human Serum Solution
3.9. Preparation of the TSPO Ligand–Dextran NGs
3.10. Physicochemical Characterization of TSPO Ligand–Dextran NGs
3.10.1. Average Hydrodynamic Diameters, Polydispersity and Zeta Potential
3.10.2. Transmission Electron Microscopy Investigation
3.10.3. Field Emission Scanning Electron Microscopy Investigation
3.11. Release Studies in Human Serum Solution of the TSPO-Ligand-Dextran Nanogels
3.12. Swelling Studies
3.13. Rheological Study of Viscoelastic Properties
3.14. Cell Cultures and Cell Viability Analysis by MTT Assay
3.15. Fluorescence Microscopy
3.16. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CBR | Central-type benzodiazepine receptor |
| C-Dex | Carboxylated dextran derivatives |
| CDI | 1,1′-carbonyldiimidazole |
| DSC | Differential scanning calorimetry |
| EDC | N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride |
| HOBt | 1-hydroxybenzotriazole hydrate |
| MPTP | Mitochondrial permeability transitional pore |
| NGs | Nanogels |
| NPs | Nanoparticles |
| PBR | Peripheral-type benzodiazepine receptor |
| PBS | Phosphate buffered saline |
| PLGA | poly(d,l-lactic-co-glycolic acid) |
| SEM | Scanning electron microscopy |
| TEA | Triethylamine |
| TEER | Transendothelial electrical resistance |
| TEM | Transmission electron microscopy |
| TSPO | Translocator protein 18 kDa |
| TSPO-Dex | TSPO ligand–dextran conjugates |
References
- Varshosaz, J. Dextran conjugates in drug delivery. Expert Opin. Drug Deliv. 2012, 9, 509–523. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, V.; Baraldi, M.; Guilarte, T.R.; Knudsen, T.B.; Lacapère, J.J.; Limdermann, P.; Norenberg, M.D.; Nutt, D.; Weizman, A.; Zhang, M.R.; et al. Translocator protein (18 kDa): New nomenclature for the peripheral-type benzodiazepine receptor based on its structure and molecular function. Trends Pharmacol. Sci. 2006, 27, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Rupprecht, R.; Papadopoulos, V.; Rammes, G.; Baghai, T.C.; Fan, J.; Akula, N.; Groyer, G.; Adams, D.; Schumacher, M. Translocator protein (18 kDa) (TSPO) as a therapeutic target for neurological and psychiatric disorders. Nat. Rev. Drug Discov. 2010, 9, 971–988. [Google Scholar] [CrossRef] [PubMed]
- Austin, C.J.; Kahlert, J.; Kassiou, M.; Rendina, L.M. The translocator protein (TSPO): A novel target for cancer chemotherapy. Int. J. Biochem. Cell Biol. 2013, 45, 1212–1216. [Google Scholar] [CrossRef] [PubMed]
- Werry, E.L.; Barron, M.L.; Kassiou, M. TSPO as a target for glioblastoma therapeutics. Biochem. Soc. Trans. 2015, 43, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Rechichi, M.; Salvetti, A.; Chelli, B.; Costa, B.; Da Pozzo, E.; Spinetti, F.; Lena, A.; Evangelista, M.; Rainaldi, G.; Martini, C.; et al. TSPO over-expression increases motility, transmigration and proliferation properties of C6 rat glioma cells. Biochim. Biophys. Acta 2008, 2, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Perrone, M.; Moon, B.S.; Park, H.S.; Laquintana, V.; Jung, J.H.; Cutrignelli, A.; Lopedota, A.; Franco, M.; Kim, S.E.; Lee, B.C.; et al. A Novel PET Imaging Probe for the Detection and Monitoring of Translocator Protein 18 kDa Expression in Pathological Disorders. Sci. Rep. 2016, 6, 20422. [Google Scholar] [CrossRef] [PubMed]
- Denora, N.; Laquintana, V.; Trapani, A.; Suzuki, H.; Sawada, M.; Trapani, G. New fluorescent probes targeting the mitochondrial-located translocator protein 18 kDa (TSPO) as activated microglia imaging agents. Pharm. Res. 2011, 28, 2820–2832. [Google Scholar] [CrossRef] [PubMed]
- Iacobazzi, R.M.; Lopalco, A.; Cutrignelli, A.; Laquintana, V.; Lopedota, A.; Franco, M.; Denora, N. Bridging Pharmaceutical Chemistry with Drug and Nanoparticle Targeting to Investigate the Role of the 18-kDa Translocator Protein TSPO. Chem. Med. Chem. 2017, 12, 1261–1274. [Google Scholar] [CrossRef] [PubMed]
- Iacobazzi, R.M.; Porcelli, L.; Lopedota, A.A.; Laquintana, V.; Lopalco, A.; Cutrignelli, A.; Altamura, E.; Di Fonte, R.; Azzariti, A.; Franco, M.; et al. Targeting human liver cancer cells with lactobionic acid-G(4)-PAMAM-FITC sorafenib loaded dendrimers. Int. J. Pharm. 2017, 528, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Sekimata, K.; Hatano, K.; Ogawa, M.; Abe, J.; Magata, Y.; Biggio, G.; Serra, M.; Laquintana, V.; Denora, N.; Latrofa, A.; et al. Radiosynthesis and in vivo evaluation of N-[11C]methylated imidazopyridineacetamides as PET tracers for peripheral benzodiazepine receptors. Nuclear Med. Biol. 2008, 35, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Veenman, L.; Levin, E.; Weisinger, G.; Leschiner, S.; Spanier, I.; Snyder, S.H.; Weizman, A.; Gavish, M. Peripheral-type benzodiazepine receptor density and in vitro tumorigenicity of glioma cell lines. Biochem. Pharmacol. 2004, 68, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Valente, G.; Depalo, N.; de Paola, I.; Iacobazzi, R.M.; Denora, N.; Laquintana, V.; Comparelli, R.; Altamura, E.; Latronico, T.; Altomare, M.; et al. Integrin-targeting with peptide-bioconjugated semiconductor-magnetic nanocrystalline heterostructures. Nano Res. 2016, 9, 644–662. [Google Scholar] [CrossRef]
- Trapani, G.; Denora, N.; Trapani, A.; Laquintana, V. Recent advances in ligand targeted therapy. J. Drug Target. 2012, 1, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Denora, N.; Laquintana, V.; Trapani, A.; Lopedota, A.; Latrofa, A.; Gallo, J.M.; Trapani, G. Translocator protein (TSPO) ligand-Ara-C (cytarabine) conjugates as a strategy to deliver antineoplastic drugs and to enhance drug clinical potential. Mol. Pharm. 2010, 7, 2255–2269. [Google Scholar] [CrossRef] [PubMed]
- Midzak, A.; Denora, N.; Laquintana, V.; Cutrignelli, A.; Lopedota, A.; Franco, M.; Altomare, C.D.; Papadopoulos, V. 2-Phenylimidazo[1,2-a]pyridine-containing ligands of the 18-kDa translocator protein (TSPO) behave as agonists and antagonists of steroidogenesis in a mouse leydig tumor cell line. Eur. J. Pharm. Sci. 2015, 76, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Musacchio, T.; Laquintana, V.; Latrofa, A.; Trapani, G.; Torchilin, V.P. PEG-PE Micelles Loaded with Paclitaxel and Surface-Modified by a PBR-Ligand: Synergistic Anticancer Effect. Mol. Pharm. 2009, 2, 468–479. [Google Scholar] [CrossRef]
- Denora, N.; Laquintana, V.; Lopalco, A.; Iacobazzi, R.M.; Lopedota, A.; Cutrignelli, A.; Iacobellis, G.; Annese, C.; Cascione, M.; Leporatti, S.; et al. In vitro targeting and imaging the translocator protein TSPO 18-kDa through G(4)-PAMAM-FITC labeled dendrimer. J. Control. Release 2013, 172, 1111–1125. [Google Scholar] [CrossRef] [PubMed]
- Laquintana, V.; Denora, N.; Musacchio, T.; Lasorsa, M.; Latrofa, A.; Trapani, G. Peripheral benzodiazepine receptor ligand-PLGA polymer conjugates potentially useful as delivery systems of apoptotic agents. J. Control. Release 2009, 137, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Laquintana, V.; Denora, N.; Lopalco, A.; Lopedota, A.; Cutrignelli, A.; Lasorsa, F.M.; Agostino, G.; Franco, M. Translocator protein ligand-PLGA conjugated nanoparticles for 5-fluorouracil delivery to glioma cancer cells. Mol. Pharm. 2014, 11, 859–871. [Google Scholar] [CrossRef] [PubMed]
- Laquintana, V.; Denora, N.; Cutrignelli, A.; Perrone, M.; Iacobazzi, R.M.; Annese, C.; Lopalco, A.; Lopedota, A.A.; Franco, M. TSPO Ligand-Methotrexate Prodrug Conjugates: Design, Synthesis, and Biological Evaluation. Int. J. Mol. Sci. 2016, 17, 967. [Google Scholar] [CrossRef] [PubMed]
- Chacko, R.T.; Ventura, J.; Zhuang, J.; Thayumanavan, S. Polymer nanogels: A versatile nanoscopic drug delivery platform. Adv. Drug Deliv. Rev. 2012, 64, 836–851. [Google Scholar] [CrossRef] [PubMed]
- Soto-Otero, R.; Méndez-Álvarez, E.; Sánchez-Iglesias, S.; Labandeira-García, J.L.; Rodríguez-Pallares, J.; Zubkov, F.I.; Zaytsev, V.P.; Voskressensky, L.G.; Varlamov, A.V.; de Candia, M.; et al. 2-Benzazepine nitrones protect dopaminergic neurons against 6-hydroxydopamine-induced oxidative toxicity. Arch. Pharm. 2012, 345, 598–609. [Google Scholar] [CrossRef] [PubMed]
- Cassano, T.; Lopalco, A.; de Candia, M.; Laquintana, V.; Lopedota, A.; Cutrignelli, A.; Perrone, M.; Iacobazzi, R.M.; Bedse, G.; Franco, M.; et al. Oxazepam-Dopamine Conjugates Increase Dopamine Delivery into Striatum of Intact Rats. Mol. Pharm. 2017, 14, 3178–3187. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Xu, D.; Jiang, M.; Yuan, W. Preparation of bioactive interferon alpha-loaded polysaccharide nanoparticles using a new approach of temperature-induced water phase/water-phase emulsion. Int. J. Nanomed. 2012, 7, 4841–4848. [Google Scholar]
- Denora, N.; Lopedota, A.; Perrone, M.; Laquintana, V.; Iacobazzi, R.M.; Milella, A.; Fanizza, E.; Depalo, N.; Cutrignelli, A.; Lopalco, A.; et al. Spray-dried mucoadhesives for intravesical drug delivery using N-acetylcysteine- and glutathione-glycol chitosan conjugates. Acta Biomater. 2016, 33, 2195–2208. [Google Scholar] [CrossRef] [PubMed]
- Lopedota, A.; Cutrignelli, A.; Laquintana, V.; Denora, N.; Iacobazzi, R.M.; Perrone, M.; Fanizza, E.; Mastrodonato, M.; Mentino, D.; Lopalco, A.; et al. Spray dried chitosan microparticles for intravesical delivery of celecoxib: Preparation and characterization. Pharm. Res. 2016, 33, 2195–2208. [Google Scholar] [CrossRef] [PubMed]
- Beesu, M.; Caruso, G.; Salyer, A.C.; Shukla, N.M.; Khetani, K.K.; Smith, L.J.; Fox, L.M.; Tanji, H.; Ohto, U.; Shimizu, T.; et al. Identification of a Human Toll-Like Receptor (TLR) 8-Specific Agonist and a Functional Pan-TLR Inhibitor in 2-Aminoimidazoles. J. Med. Chem. 2016, 59, 3311–3330. [Google Scholar] [CrossRef] [PubMed]
- De Candia, M.; Zaetta, G.; Denora, N.; Tricarico, D.; Majellaro, M.; Cellamare, S.; Altomare, C.D. New azepino[4,3-b]indole derivatives as nanomolar selective inhibitors of human butyrylcholinesterase showing protective effects against NMDA-induced neurotoxicity. Eur. J. Med. Chem. 2017, 125, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Lopalco, A.; Douglas, J.; Denora, N.; Stella, V.J. Determination of pKa and Hydration Constants for a Series of α-Keto-Carboxylic Acids Using Nuclear Magnetic Resonance Spectrometry. J. Pharm. Sci. 2016, 105, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Lopalco, A.; Stella, V.J. Effect of molecular structure on the relative hydrogen peroxide scavenging ability of some α-keto carboxylic acids. J. Pharm. Sci. 2016, 105, 2879–2885. [Google Scholar] [CrossRef] [PubMed]
- Cutrignelli, A.; Denora, N.; Lopedota, A.; Trapani, A.; Laquintana, V.; Latrofa, A.; Trapani, G.; Liso, G. Comparative effects of some hydrophilic excipients on the rate of gabapentin and baclofen lactamization in lyophilized formulations. Int. J. Pharm. 2007, 332, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Cutrignelli, A.; Lopedota, A.; Trapani, A.; Boghetich, G.; Franco, M.; Denora, N.; Laquintana, V.; Trapani, G. Relationship between dissolution efficiency of Oxazepam/carrier blends and drug and carrier molecular descriptors using multivariate regression analysis. Int. J. Pharm. 2008, 358, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Corricelli, M.; Altamura, D.; Curri, M.L.; Sibillano, T.; Siliqi, D.; Mazzone, A.; Depalo, N.; Fanizza, E.; Zanchet, D.; Giannini, C.; et al. GISAXS and GIWAXS study on self-assembling processes of nanoparticle based superlattices. CrystEngComm 2014, 16, 9482–9492. [Google Scholar] [CrossRef]
- Perrone, M.; Lopedota, A.; Liberati, E.; Russo, V.; Cutrignelli, A.; Laquintana, V.; de Sousa, I.P.; Franco, M.; Tongiani, S.; Denora, N.; et al. Natural dendrimers: Synthesis and in vitro characterization of glycogen-cysteamine conjugates. Eur. J. Pharm. Biopharm. 2017, 115, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Perrone, M.; Lopalco, A.; Lopedota, A.; Cutrignelli, A.; Laquintana, V.; Douglas, J.; Franco, M.; Liberati, E.; Russo, V.; Tongiani, S.; et al. Preactivated thiolated glycogen as mucoadhesive polymer for drug delivery. Eur. J. Pharm. Biopharm. 2017, 119, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Lasorsa, F.M.; Pinton, P.; Palmieri, L.; Scarcia, P.; Rottensteiner, H.; Rizzuto, R.; Palmieri, F. Peroxisomes as novel players in cell calcium homeostasis. J. Biol. Chem. 2008, 283, 15300–15308. [Google Scholar] [CrossRef] [PubMed]












| Compound | Mna | Mwb | Mpc | PDI | Elemental Analysis | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Calculated d | Found | |||||||||
| (kDa ± SD) | (Mw/Mn) | %C | %H | %N | %C | %H | %N | |||
| 5 | 25.1 ± 0.5 | 42.9 ± 0.5 | 27.7 ± 0.5 | 1.71 | 44.44 | 6.17 | - | 44.19 | 6.60 | - |
| 6 | 56.1 ± 0.5 | 105.5 ± 2.5 | 64.4 ± 0.5 | 1.88 | 46.22 | 5.94 | - | 44.27 | 5.99 | - |
| 7 | 73.3 ± 2.5 | 128.4 ± 1.8 | 82.2 ± 0.9 | 1.75 | 48.69 | 5.86 | 3.83 | 48.08 | 5.90 | 3.59 |
| 8 | 79.9 ± 0.4 | 155.8 ± 2.2 | 87.7 ± 1.5 | 1.95 | 49.27 | 5.44 | 3.68 | 48.52 | 5.56 | 3.10 |
| 9 | 62.3 ± 0.5 | 117.7 ± 2.0 | 77.2 ± 0.7 | 1.89 | 44.87 | 5.91 | - | 44.84 | 5.93 | - |
| 10 | 85.4 ± 1.2 | 161.4 ± 5.5 | 90.8 ± 2.0 | 1.89 | 51.38 | 5.64 | 3.77 | 50.83 | 5.68 | 3.47 |
| 11 | 80.7 ± 0.5 | 162.2 ± 8.2 | 89.0 ± 1.0 | 2.01 | 52.66 | 4.95 | 5.91 | 52.04 | 5.04 | 5.47 |
| Compound | Yield % | pH Analysis | UV Analysis | 1H-NMR Analysis | Elemental Analysis | SEC Analysis | |||
|---|---|---|---|---|---|---|---|---|---|
| n | E1% c | λmax (nm) | TSPO Ligand Conjugation Degree | Carboxylic Group Substitution Degree | Residual Free Carboxylic Groups | TSPO Ligand Conjugation Degree | TSPO Ligand Conjugation Degree | ||
| (w/w) a | (mg/g) b | (g/g) e | (mol/mol) f | (g/g) e | (mol/mol) g | ||||
| 1 | 62.8 d | 253 | |||||||
| 4 | 76.5 d | 254 | |||||||
| 6 | 82 | 55.7 | 0.51 | ||||||
| 7 | 68 | 23.2 | 250 | 0.37 | 0.23 | 0.31 | 0.28 | ||
| 8 | 51 | 25.2 | 254 | 0.33 | 0.26 | 0.28 | 0.25 | ||
| 9 | 80 | 54.2 | 0.55 | ||||||
| 10 | 71 | 36.4 | 251 | 0.58 | 0.18 | 0.55 | 0.37 | ||
| 11 | 54 | 43.6 | 254 | 0.57 | 0.20 | 0.52 | 0.35 | ||
| NGs | Size dmean (nm) | PDI c | ζ (mV) d | |
|---|---|---|---|---|
| z-Average a | Number PSD b | |||
| 7 | 320.8 ± 14.13 | 180.9 ± 1.8 | 0.24 ± 0.11 | −28.1 ± 2.1 |
| 8 | 280.8 ± 12.45 | 165.3 ± 2.8 | 0.22 ± 0.10 | −32.0 ± 5.2 |
| 10 | 391.2 ± 16.99 | 220.5 ± 5.7 | 0.27 ± 0.06 | −22.1 ± 2.4 |
| 11 | 420.4 ± 34.11 | 294.4 ± 6.3 | 0.28 ± 0.09 | −25.5 ± 3.3 |
| Tablet | Swelling PBS pH 7.4 | ||||
|---|---|---|---|---|---|
| Polymer | Nanogels | Polymer | Nanogels | ||
| t, min | (w/w, %) | t, min | (w/w, %) | ||
| TSPO-Dex 7 | TSPO-Dex NGs 7 | 120 | 18.1 ± 2.0 | 45 | 22.6 ± 2.5 |
| TSPO-Dex 8 | TSPO-Dex NGs 8 | 120 | 15.2 ± 2.4 | 45 | 19.0 ± 1.9 |
| TSPO-Dex 10 | TSPO-Dex NGs 10 | 90 | 6.5 ± 2.1 | 60 | 7.8 ± 0.7 |
| TSPO-Dex 11 | TSPO-Dex NGs 11 | 90 | 9.2 ± 3.0 | 60 | 11.1 ± 1.0 |
| C-Dex 6 | - | 20 | 31.5 ± 4.6 | - | - |
| C-Dex 9 | - | 20 | 27.4 ± 2.5 | - | - |
| Nanogels | G′ (Pa) | G″ (Pa) | η (Pa s) |
|---|---|---|---|
| 6 | 1320 ± 56.5 | 220 ± 16.0 | 125.1 ± 2.5 |
| 9 | 1191 ± 48.3 | 182 ± 15.5 | 117.2 ± 9.4 |
| TSPO-Dex NGs 7 | 870 ± 66.5 | 85 ± 6.6 | 75.1 ± 2.5 |
| TSPO-Dex NGs 8 | 801 ± 28.8 | 82 ± 8.5 | 70.2 ± 6.5 |
| TSPO-Dex NGs 10 | 720 ± 33.5 | 74 ± 6.7 | 63.5 ± 1.9 |
| TSPO-Dex NGs 11 | 683 ± 37.2 | 68 ± 5.5 | 57.4 ± 2.6 |
| Sample | IC50 (µM) a |
|---|---|
| 1 | 17.5 ± 0.7 |
| 4 | 19.2 ± 0.6 |
| TSPO-Dex NGs 7 | 7.32 ± 0.15 |
| TSPO-Dex NGs 8 | 2.24 ± 0.06 |
| TSPO-Dex NGs 10 | 1.85 ± 0.03 |
| TSPO-Dex NGs 11 | 1.20 ± 0.07 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopalco, A.; Cutrignelli, A.; Denora, N.; Perrone, M.; Iacobazzi, R.M.; Fanizza, E.; Lopedota, A.; Depalo, N.; De Candia, M.; Franco, M.; et al. Delivery of Proapoptotic Agents in Glioma Cell Lines by TSPO Ligand–Dextran Nanogels. Int. J. Mol. Sci. 2018, 19, 1155. https://doi.org/10.3390/ijms19041155
Lopalco A, Cutrignelli A, Denora N, Perrone M, Iacobazzi RM, Fanizza E, Lopedota A, Depalo N, De Candia M, Franco M, et al. Delivery of Proapoptotic Agents in Glioma Cell Lines by TSPO Ligand–Dextran Nanogels. International Journal of Molecular Sciences. 2018; 19(4):1155. https://doi.org/10.3390/ijms19041155
Chicago/Turabian StyleLopalco, Antonio, Annalisa Cutrignelli, Nunzio Denora, Mara Perrone, Rosa Maria Iacobazzi, Elisabetta Fanizza, Angela Lopedota, Nicoletta Depalo, Modesto De Candia, Massimo Franco, and et al. 2018. "Delivery of Proapoptotic Agents in Glioma Cell Lines by TSPO Ligand–Dextran Nanogels" International Journal of Molecular Sciences 19, no. 4: 1155. https://doi.org/10.3390/ijms19041155