Syntheses, Crystal Structures, and Antitumor Activities of Copper(II) and Nickel(II) Complexes with 2-((2-(Pyridin-2-yl)hydrazono)methyl)quinolin-8-ol
Abstract
:1. Introduction
2. Results
2.1. Synthesis
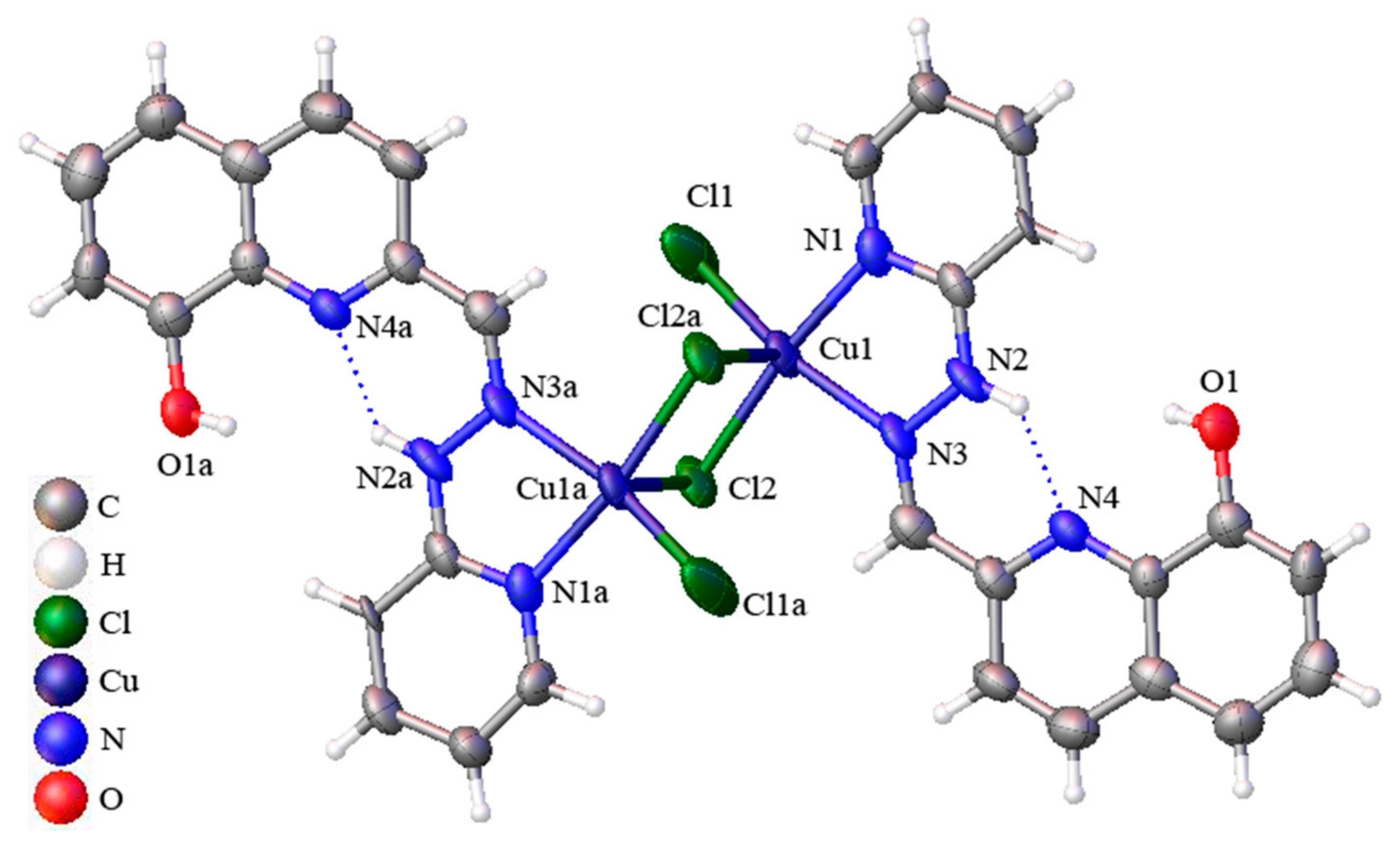
2.2. Crystal Structures of Complexes 1 and 2
2.3. Stability in Solution
2.4. In Vitro Cytotoxicity
2.5. Cell Cycle Analysis and Expressions of the Related Proteins
2.6. Apoptosis Assay
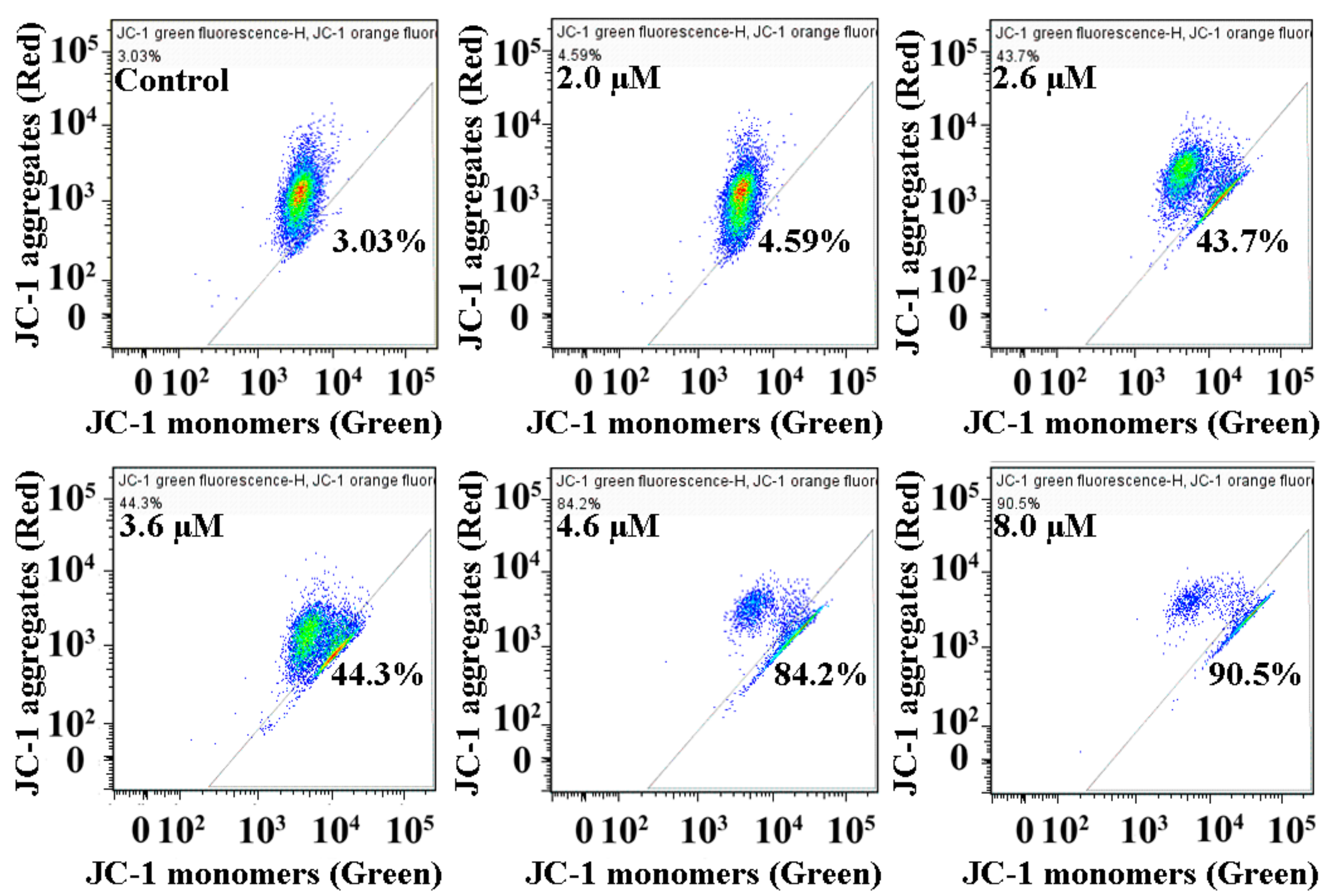
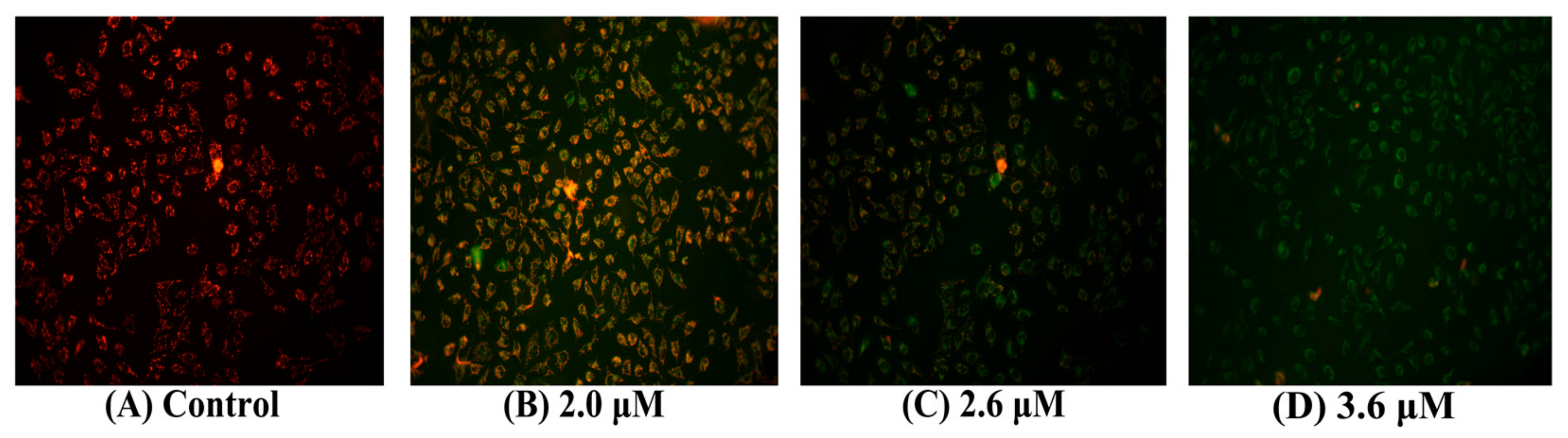
2.7. Loss of Mitochondrial Membrane Potential in MGC80-3 Cells
2.8. Intracellular Ca2+
2.9. Reactive Oxygen Species (ROS) Level
2.10. Western Blot Assay
2.11. Assessment of Caspase-3/9/8 Activation
3. Materials and Methods
3.1. Materials
3.2. Instrumentation
3.3. Synthesis
3.3.1. Synthesis of L
3.3.2. Synthesis of Complex 1
3.3.3. Synthesis of Complex 2
3.4. X-ray Crystallography
3.5. In Vitro Cytotoxicity
3.6. Cell Cycle Arrest
3.7. Other Experimental Methods
3.8. Statistics
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chaudhuri, A.R.; Callen, E.; Ding, X.; Gogola, E.; Duarte, A.A.; Lee, J.E.; Wong, N.; Lafarga, V.; Calvo, J.A.; Panzarino, N.J.; et al. Replication fork stability confers chemoresistance in BRCA-deficient cells. Nature 2016, 535, 382–387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheff, D.M.; Hall, M.D. A Drug of Such Damned Nature.1 Challenges and Opportunities in Translational Platinum Drug Research. J. Med. Chem. 2017, 60, 4517–4532. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, A.; Wu, J.; He, Y.; Yu, H.; Chai, R.; Li, H. MiR-182-5p protects inner ear hair cells from cisplatin-induced apoptosis by inhibiting FOXO3a. Cell Death Dis. 2016, 7, e2362. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Fang, C.; Zong, Z.; Cui, L.; Bi, C.; Fan, Y. Synthesis, characterization and anticancer activity of two ternary copper(ІІ) Schiff base complexes. Inorg. Chim. Acta 2015, 432, 198–207. [Google Scholar] [CrossRef]
- Saleem, K.; Wani, W.A.; Haque, A.; Lone, M.N.; Hsieh, M.F.; Jairajpuri, M.A.; Ali, I. Synthesis, DNA binding, hemolysis assays and anticancer studies of copper(II), nickel(II) and iron(III) complexes of a pyrazoline-based ligand. Future Med. Chem. 2013, 5, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Muralisankar, M.; Haribabu, J.; Bhuvanesh, N.S.P.; Karvembu, R.; Sreekanth, A. Synthesis, X-ray crystal structure, DNA/protein binding, DNA cleavage and cytotoxicity studies of N(4) substituted thiosemicarbazone based copper(II)/nickel(II) complexes. Inorg. Chim. Acta 2016, 449, 82–95. [Google Scholar] [CrossRef]
- Wani, W.A.; Al-Othman, Z.; Ali, I.; Saleem, K.; Hsieh, M.F. Copper(II), nickel(II), and ruthenium(III) complexes of an oxopyrrolidine-based heterocyclic ligand as anticancer agents. J. Coord. Chem. 2014, 67, 2110–2130. [Google Scholar] [CrossRef]
- Haleel, A.; Arthi, P.; Reddy, N.D.; Veenac, V.; Sakthivelc, N.; Arund, Y.; Perumald, P.T.; Rahiman, K. DNA binding, molecular docking and apoptotic inducing activity of nickel(II), copper(II) and zinc(II) complexes of pyridine-based tetrazolo[1,5-a] pyrimidine ligands. RSC Adv. 2014, 4, 60816–60830. [Google Scholar] [CrossRef]
- Jin, Q.M.; Lu, Y.; Jin, J.L.; Guo, H.; Lin, G.W.; Wang, Y.; Lu, T. Synthesis, characterization, DNA binding ability and cytotoxicity of the novel platinum(II); copper(II), cobalt(II) and nickel(II) complexes with 3-(1H-benzo[d]imidazol-2-yl)-β-carboline. Inorg. Chim. Acta 2014, 421, 91–99. [Google Scholar] [CrossRef]
- Rajalakshmi, S.; Kiran, M.S.; Nair, B.U. DNA condensation by copper(II) complexes and their anti-proliferative effect on cancerous and normal fibroblast cells. Eur. J. Med. Chem. 2014, 80, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Totta, X.; Papadopoulou, A.A.; Hatzidimitriou, A.G.; Papadopoulosb, A.; Psomas, G. Synthesis, structure and biological activity of nickel(II) complexes with mefenamato and nitrogen-donor ligands. J. Inorg. Biochem. 2015, 145, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.W.; Kuo, C.F.; Chuang, S.M.; Hou, M.H. Elucidation of the DNA-interacting properties and anticancer activity of a Ni(II)-coordinated mithramycin dimer complex. Biometals 2013, 26, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Tabrizi, L.; McArdle, P.; Erxleben, A.; Chiniforoshan, H. Nickel(II) and cobalt(II) complexes of lidocaine: Synthesis, structure and comparative invitro evaluations of biological perspectives. Eur. J. Med. Chem. 2015, 103, 516–529. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.F.; Wang, Y.; Ding, W.J.; Xu, J.; Chen, R.H.; Xie, J.; Zhu, W.J.; Jia, L.; Ma, T.L. Anticancer Activity and DNA-Binding Investigations of the Cu(II) and Ni(II) Complexes with Coumarin Derivative. Chem. Biol. Drug Des. 2015, 85, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Alomar, K.; Landreau, A.; Allain, M.; Boueta, G.; Larcher, G. Synthesis, structure and antifungal activity of thiophene-2,3-dicarboxaldehyde bis(thiosemicarbazone) and nickel(II), copper(II) and cadmium(II) complexes: Unsymmetrical coordination mode of nickel complex. J. Inorg. Biochem. 2013, 126, 76–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramírez-Macías, I.; Maldonado, C.R.; Marín, C.; Olmoa, F.; Gutiérrezsánchezc, R.; Rosalesa, M.J.; Quirósb, M.; Salasb, J.M. In vitro anti-leishmania evaluation of nickel complexes with a triazolopyrimidine derivative against Leishmania infantum and Leishmania braziliensis. J. Inorg. Biochem. 2012, 112, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Betanzos-Lara, S.; Gómez-Ruiz, C.; Barrón-Sosa, L.R.; Gracia-Mora, I.; Flores-Álamo, M.; Barba-Behrens, N. Cytotoxic copper(II), cobalt(II), zinc(II), and nickel(II) coordination compounds of clotrimazole. J. Inorg. Biochem. 2012, 114, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Turnaturi, R.; Oliveri, V.; Vecchio, G. Biotin-8-hydroxyquinoline conjugates and their metal complexes: Exploring the chemical properties and the antioxidant activity. Polyhedron 2016, 110, 254–260. [Google Scholar] [CrossRef]
- Xu, H.; Chen, W.; Zhan, P.; Liu, X. 8-Hydroxyquinoline: A privileged structure with a broad-ranging pharmacological potential. MedChemComm 2015, 6, 61–74. [Google Scholar] [CrossRef]
- Prachayasittikul, V.; Prachayasittikul, S.; Ruchirawat, S.; Prachayasittikul, V. 8-Hydroxyquinolines: A review of their metal chelating properties and medicinal applications. Drug Des. Dev. Ther. 2013, 7, 1157–1178. [Google Scholar] [CrossRef] [PubMed]
- Solomon, R.; Lee, H. Quinoline as a Privileged Scaffold in Cancer Drug Discovery. Curr. Med. Chem. 2011, 18, 1488–1508. [Google Scholar] [CrossRef] [PubMed]
- Schimmer, A.D. Clioquinol—A Novel Copper-Dependent and Independent Proteasome Inhibitor. Cancer Drug Targets 2011, 11, 325–331. [Google Scholar] [CrossRef]
- Matlack, K.E.; Tardiff, D.F.; Narayan, P.; Hamamichi, S.; Caldwell, K.A.; Caldwell, G.A.; Lindquist, S. Clioquinol promotes the degradation of metal-dependent amyloid-β (Aβ) oligomers to restore endocytosis and ameliorate Aβ toxicity. Proc. Natl. Acad. Sci. USA 2014, 111, 4013–4018. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Esteban, G.; Ojima, M.; Bautista-Aguilera, O.M.; Inokuchi, T.; Moraleda, I.; Iriepa, I.; Samadi, A.; Youdim, M.B.; Romero, A.; et al. Donepezil + propargylamine + 8-hydroxyquinoline hybrids as new multifunctional metal-chelators; ChE and MAO inhibitors for the potential treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2014, 80, 543–561. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.Y.; Esteban, G.; Brogi, S.; Shionoya, M.; Wang, L.; Campiani, G.; Unzeta, M.; Inokuchi, T.; Butini, S.; Marco-Contelles, J. Donepezil-like multifunctional agents: Design, synthesis, molecular modeling and biological evaluation. Eur. J. Med. Chem. 2016, 121, 864–879. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Taggart, J.E.; Zhang, X.; Benbrook, D.M.; Lind, S.E.; Ding, W.Q. Nitroxoline (8-hydroxy-5-nitroquinoline) is more a potent anti-cancer agent than clioquinol (5-chloro-7-iodo-8-quinoline). Cancer Lett. 2011, 312, 11–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sosić, I.; Mirković, B.; Arenz, K.; Stefane, B.; Kos, J.; Gobec, S. Development of New Cathepsin B Inhibitors: Combining Bioisosteric Replacements and Structure-Based Design To Explore the Structure–Activity Relationships of Nitroxoline Derivatives. J. Med. Chem. 2013, 56, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Knez, D.; Brus, B.; Coquelle, N.; Sosič, I.; Šink, R.; Brazzolotto, X.; Mravljak, J.; Colletier, J.P.; Gobec, S. Structure-based development of nitroxoline derivatives as potential multifunctional anti-Alzheimer agents. Bioorg. Med. Chem. 2015, 23, 4442–4452. [Google Scholar] [CrossRef] [PubMed]
- Rotili, D.; Tomassi, S.; Conte, M.; Benedetti, R.; Tortorici, M.; Ciossani, G.; Valente, S.; Marrocco, B.; Labella, D.; Novellino, E.; et al. Pan-Histone Demethylase Inhibitors Simultaneously Targeting Jumonji C and Lysine-Specific Demethylases Display High Anticancer Activities. J. Med. Chem. 2013, 57, 42–55. [Google Scholar] [CrossRef] [PubMed]
- Schiller, R.; Scozzafava, G.; Tumber, A.; Wickens, J.R.; Bush, J.T.; Rai, G.; Lejeune, C.; Choi, H.; Yeh, T.L.; Chan, M.C.; et al. A Cell-Permeable Ester Derivative of the JmjC Histone Demethylase Inhibitor IOX1. Chem. Med. Chem. 2014, 9, 566–571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sliman, F.; Blairvacq, M.; Durieu, E.; Meijer, L.; Rodrigo, J.; Desmaële, D. Identification and structure-activity relationship of 8-hydroxy-quinoline-7-carboxylic acid derivatives as inhibitors of Pim-1 kinase. Bioorg. Med. Chem. Lett. 2010, 20, 2801–2805. [Google Scholar] [CrossRef] [PubMed]
- Li, X.M.; Wood, T.E.; Sprangers, R.; Jansen, G.; Franke, N.E.; Mao, X.L.; Wang, X.M.; Zhang, Y.; Verbrugge, S.E.; Adomat, H.; et al. Effect of Noncompetitive Proteasome Inhibition on Bortezomib Resistance. J. Natl. Cancer Inst. 2010, 102, 1069–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.C.; Yang, Z.Y. Synthesis, crystal structure; antioxidation and DNA binding properties of binuclear Ho(III) complexes of Schiff-base ligands derived from 8-hydroxyquinoline-2-carboxyaldehyde and four aroylhydrazines. J. Inorg. Biochem. 2009, 103, 1014–1022. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, J.A.; Fullagar, J.L.; Miller, M.T.; Cohen, S.M. Identifying Chelators for Metalloprotein Inhibitors Using a Fragment-Based Approach. J. Med. Chem. 2011, 54, 591–602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaw, A.Y.; Chang, C.Y.; Hsu, M.Y.; Lu, P.J.; Yang, C.N.; Chen, H.L.; Lo, C.W.; Shiau, C.W.; Chern, M.K. Synthesis and structure-activity relationship study of 8-hydroxyquinoline-derived Mannich bases as anticancer agents. Eur. J. Med. Chem. 2010, 45, 2860–2867. [Google Scholar] [CrossRef] [PubMed]
- Ariyasu, S.; Sawa, A.; Morita, A.; Hanaya, K.; Hoshi, M.; Takahashi, I.; Wang, B.; Aoki, S. Design and synthesis of 8-hydroxyquinoline-based radioprotective agents. Bioorg. Med. Chem. 2014, 22, 3891–3905. [Google Scholar] [CrossRef] [PubMed]
- Cacciatore, I.; Fornasari, E.; Baldassarre, L.; Cornacchia, C.; Fulle, S.; Di Filippo, E.S.; Pietrangelo, T.; Pinnen, F. A Potent (R)-alpha-bis-lipoyl Derivative Containing 8-Hydroxyquinoline Scaffold: Synthesis and Biological Evaluation of Its Neuroprotective Capabilities in SH-SY5Y Human Neuroblastoma Cells. Pharmaceuticals 2013, 6, 54–69. [Google Scholar] [CrossRef] [PubMed]
- Du Moulinet D’Hardemare, A.; Gellon, G.; Philouze, C.; Serratrice, G. Oxinobactin and Sulfoxinobactin; Abiotic Siderophore Analogues to Enterobactin Involving 8-Hydroxyquinoline Subunits: Thermodynamic and Structural Studies. Inorg. Chem. 2012, 51, 12142–12151. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Bachiller, M.I.; Pérez, C.; González-Munoz, G.C.; Conde, S.; López, M.G.; Villarroya, M.; García, A.G.; Rodriguez-Franco, M.I. Novel Tacrine−8-Hydroxyquinoline Hybrids as Multifunctional Agents for the Treatment of Alzheimer’s Disease; with Neuroprotective; Cholinergic; Antioxidant; and Copper-Complexing Properties. J. Med. Chem. 2010, 53, 4927–4937. [Google Scholar] [CrossRef] [PubMed]
- Calvaresi, E.C.; Hergenrother, P.J. Glucose conjugation for the specific targeting and treatment of cancer. Chem. Sci. 2013, 4, 2319–2333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveri, V. New Glycoconjugates for the Treatment of Diseases Related to Metal Dyshomeostasis. ChemistryOpen 2015, 4, 792–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, S.H.; Chui, C.H.; Chan, S.W.; Kok, S.H.L.; Chan, D.; Tsoi, M.Y.T.; Leung, P.H.M.; Lam, A.K.Y.; Chan, A.S.C.; Lam, K.H.; et al. Synthesis of 8-Hydroxyquinoline Derivatives as Novel Antitumor Agents. ACS Med. Chem. Lett. 2012, 4, 170–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Núñez, C.; Oliveira, E.; García-Prdo, J.; Diniza, M.; Lorenzof, J.; Capeloa, J.L.; Lodeiroa, C. A novel quinoline molecular probe and the derived functionalized gold nanoparticles: Sensing properties and cytotoxicity studies in MCF-7 human breast cancer cells. J. Inorg. Biochem. 2014, 137, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Kao, M.H.; Chen, T.Y.; Cai, Y.R.; Hu, C.H.; Liu, Y.W.; Jhong, Y.; Wu, A.T. A turn-on Schiff-base fluorescence sensor for Mg2+ ion and its practical application. J. Lumin. 2016, 169, 156–160. [Google Scholar] [CrossRef]
- Zhang, H.R.; Meng, T.; Liu, Y.C.; Qin, Q.P.; Chen, Z.F.; Liu, Y.N.; Liang, H. Synthesis, Structure Characterization and Antitumor Activity Study of a New Iron(III) Complex of 5-Nitro-8-hydroxylquinoline (HNOQ). Chem. Pharm. Bull. 2016, 64, 1208–1217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.R.; Meng, T.; Liu, Y.C.; Chen, Z.F.; Liu, Y.N.; Liang, H. Synthesis, characterization and biological evaluation of a cobalt(II) complex with 5-chloro-8-hydroxyquinoline as anticancer agent. Appl. Organomet. Chem. 2016, 30, 740–747. [Google Scholar] [CrossRef]
- Zhang, H.R.; Liu, Y.C.; Chen, Z.F.; Meng, T.; Zou, B.Q.; Liu, Y.N.; Liang, H. Studies on the structures, cytotoxicity and apoptosis mechanism of 8-hydroxylquinoline rhodium(III) complexes in T-24 cells. New J. Chem. 2016, 409, 6005–6014. [Google Scholar] [CrossRef]
- Heidary, D.K.; Howerton, B.S.; Glazer, E.C. Coordination of Hydroxyquinolines to a Ruthenium bis-dimethyl-phenanthroline Scaffold Radically Improves Potency for Potential as Antineoplastic Agents. J. Med. Chem. 2014, 57, 8936–8946. [Google Scholar] [CrossRef] [PubMed]
- Dömötör, O.; Pape, V.F.; May, N.V.; Szakács, G.; Enyedy, É.A. Comparative solution equilibrium studies of antitumor ruthenium(η6-p-cymene) and rhodium(η5-C5Me5) complexes of 8-hydroxyquinolines. Dalton Trans. 2017, 46, 4382–4396. [Google Scholar] [CrossRef] [PubMed]
- Martín-Santos, C.; Michelucci, E.; Marzo, T.; Messori, L.; Szumlas, P.; Bednarski, P.J.; Mas-Ballesté, R.; Navarro-Ranninger, C.; Cabrer, S. Gold(III) complexes with hydroxyquinoline, aminoquinoline and quinoline ligands: Synthesis, cytotoxicity, DNA and protein binding studies. J. Inorg. Biochem. 2015, 153, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.P.; Chen, Z.F.; Qin, J.L.; He, X.J.; Li, Y.L.; Liu, Y.C.; Liang, H. Studies on antitumor mechanism of two planar platinum(II) complexes with 8-hydroxyquinoline: Synthesis, characterization, cytotoxicity, cell cycle and apoptosis. Eur. J. Med. Chem. 2015, 92, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Tardito, S.; Barilli, A.; Bassanetti, I.; Tegoni, M.; Bussolati, O.; Franchi-Gazzola, R.; Marchiò, L. Copper-Dependent Cytotoxicity of 8-Hydroxyquinoline Derivatives Correlates with Their Hydrophobicity and Does Not Require Caspase Activation. J. Med. Chem. 2012, 55, 10448–10459. [Google Scholar] [CrossRef] [PubMed]
- Rogolino, D.; Cavazzoni, A.; Gatti, A.; Tegoni, M.; Pelosi, G.; Verdolino, V.; Carcelli, M. Anti-proliferative effects of copper(II) complexes with hydroxyquinoline-thiosemicarbazone ligands. Eur. J. Med. Chem. 2017, 128, 140–153. [Google Scholar] [CrossRef] [PubMed]
- Correia, I.; Adao, P.; Roy, S.; Wahba, M.; Matos, C.; Maurya, M.R.; Pessoa, J.C. Hydroxyquinoline derived vanadium (IV and V) and copper(II) complexes as potential anti-tuberculosis and anti-tumor agents. J. Inorg. Biochem. 2014, 141, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Laplante, S.R.; Fader, L.D.; Fandrick, K.R.; Fandrick, D.R.; Hucke, O.; Kemper, R.; Miller, S.P.F.; Edwards, P.J. Assessing Atropisomer Axial Chirality in Drug Discovery and Development. J. Med. Chem. 2016, 54, 7005–7022. [Google Scholar] [CrossRef] [PubMed]
- Sommerwerk, S.; Heller, L.; Kuhfs, J.; Csuk, R. Urea derivates of ursolic, oleanolic and maslinic acid induce apoptosis and are selective cytotoxic for several human tumor cell lines. Eur. J. Med. Chem. 2016, 119, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ooi, K.K.; Yeo, C.I.; Mahandaran, T.; Ang, K.P.; Akim, A.M.; Cheah, Y.K.; Seng, H.L.; Tiekink, E.R. G2/M cell cycle arrest on HT-29 cancer cells and toxicity assessment of triphenylphosphanegold(I) carbonimidothioates, Ph3PAu[SC(OR) = NPh], R = Me, Et, and iPr; during zebrafish development. J. Inorg. Biochem. 2017, 166, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Han, B.J.; Zeng, C.C.; Lai, S.H.; Li, W.; Tang, B.; Wan, D.; Jiang, G.B.; Liu, Y. Synthesis, characterization, in vitro cytotoxicity and anticancer effects of ruthenium(II) complexes on BEL-7402 cells. J. Inorg. Biochem. 2016, 157, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.P.; Liu, Y.N.; Liu, Y.; Qin, X.Y.; Zhou, Y.H.; Liu, J. Dinuclear ruthenium complexes display loop isomer selectivity to c-MYC DNA G-quadriplex and exhibit anti-tumour activity. J. Inorg. Biochem. 2016, 156, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Chen, J.; Zhang, S.; Hu, J.H.; Ling, H.; Li, X.S. Synthesis, Evaluation; and Mechanism Study of Novel Indole-Chalcone Derivatives Exerting Effective Antitumor Activity Through Microtubule Destabilization in Vitro and in Vivo. J. Med. Chem. 2016, 59, 5264–5283. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.L.; Qin, Q.P.; Cao, Q.Q.; Han, H.H.; Liu, Z.L.; Liu, Y.C.; Liang, H.; Chen, Z.F. Synthesis, crystal structure, cytotoxicity and action mechanism of a Rh(III) complex with 8-hydroxy-2-methylquinoline as a ligand. Med. Chem. Commun. 2017, 8, 184–190. [Google Scholar] [CrossRef]
- Horvat, A.; Zorec, R.; Vardjan, N. Adrenergic stimulation of single rat astrocytes results in distinct temporal changes in intracellular Ca2+ and cAMP-dependent PKA responses. Cell Calcium 2016, 59, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.Z.; Xu, Y.; Hu, D.C.; Hui, Y.; Yang, F.X. Apoptosis induction on human hepatoma cells Hep G2 of decabrominated diphenyl ether (PBDE-209). Toxicol. Lett. 2007, 171, 19–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawiak, A.; Zawacka-Pankau, J.; Wasilewska, A.; Stasilojc, G.; Bigda, J.; Lojkowska, E. Induction of Apoptosis in HL-60 Cells through the ROS-Mediated Mitochondrial Pathway by Ramentaceone from Drosera aliciae. J. Nat. Prod. 2012, 75, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Spierings, D.; McStay, G.; Saleh, M.; Bender, C.; Chipuk, J.; Maurer, U.; Green, D.R. Connected to Death: The (Unexpurgated) Mitochondrial Pathway of Apoptosis. Science 2005, 310, 66–67. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.L.; Qin, Q.P.; Wei, Z.Z.; Yu, C.C.; Meng, T.; Wu, C.X.; Liang, Y.L.; Liang, H.; Chen, Z.F. Stabilization of c-myc G-Quadruplex DNA, inhibition of telomerase activity, disruption of mitochondrial functions and tumor cell apoptosis by platinum(II) complex with 9-amino-oxoisoaporphine. Eur. J. Med. Chem. 2016, 124, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.H.; Cai, L.J.; Wang, Y.F.; Zhou, Y.H.; An, Y.F.; Liu, Y.C.; Peng, Y.; Chen, Z.F.; Liang, H. Novel compound PS-101 exhibits selective inhibition in non-small-cell lung cancer cell by blocking the EGFR-driven antiapoptotic pathway. Biochem. Pharmacol. 2013, 86, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.F.; Qin, Q.P.; Qin, J.L.; Liu, Y.C.; Huang, K.B.; Li, Y.L.; Meng, T.; Zhang, G.H.; Peng, Y.; Luo, X.J.; et al. Stabilization of G-Quadruplex DNA, Inhibition of Telomerase Activity, and Tumor Cell Apoptosis by Organoplatinum(II) Complexes with Oxoisoaporphine. J. Med. Chem. 2015, 58, 2159–2179. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.B.; Chen, Z.F.; Liu, Y.C.; Xie, X.L.; Liang, H. Dihydroisoquinoline copper(II) complexes: Crystal structures, cytotoxicity, and action mechanism. RSC Adv. 2015, 5, 81313–81323. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXS-97, Program for Solution of Crystal Structures; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Qin, Q.P.; Qin, J.L.; Meng, T.; Yang, G.A.; Wei, Z.Z.; Liu, Y.C.; Liang, H.; Chen, Z.F. Preparation of 6/8/11-Amino/Chloro-Oxoisoaporphine and Group-10 Metal Complexes and Evaluation of Their in Vitro and in Vivo Antitumor Activity. Sci. Rep. 2016, 6, 37644. [Google Scholar] [CrossRef] [PubMed] [Green Version]











| Compounds | Hep-G2 | SK-OV-3 | MGC80-3 | HeLa | T-24 | BEL-7402 | NCI-H460 | HL-7702 |
|---|---|---|---|---|---|---|---|---|
| L | 58.40 ± 0.69 | 42.94 ± 2.64 | 40.93 ± 0.94 | 58.73 ± 1.29 | 85.93 ± 15.11 | 47.85 ± 0.37 | 36.93 ± 3.93 | 48.63 ± 0.34 |
| 1 | 4.51 ± 0.38 | 3.69 ± 0.16 | 2.60 ± 0.17 | 3.62 ± 0.12 | 4.41 ± 0.06 | 5.92 ± 0.01 | 5.01 ± 0.16 | 12.78 ± 0.55 |
| 2 | >100 | 39.77 ± 2.15 | 38.99 ± 2.42 | 27.13 ± 6.51 | 18.97 ± 3.47 | 51.68 ± 0.66 | 36.31 ± 3.75 | 31.73 ± 2.11 |
| Cisplatin b | 9.55 ± 0.46 | 16.32 ± 1.37 | 12.37 ± 1.53 | 9.45 ± 2.05 | 28.15 ± 1.67 | 19.4 ± 0.58 | 9.59 ± 0.48 | 15.87 ± 0.36 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Q.-Y.; Cao, Q.-Q.; Qin, Q.-P.; Deng, C.-X.; Liang, H.; Chen, Z.-F. Syntheses, Crystal Structures, and Antitumor Activities of Copper(II) and Nickel(II) Complexes with 2-((2-(Pyridin-2-yl)hydrazono)methyl)quinolin-8-ol. Int. J. Mol. Sci. 2018, 19, 1874. https://doi.org/10.3390/ijms19071874
Yang Q-Y, Cao Q-Q, Qin Q-P, Deng C-X, Liang H, Chen Z-F. Syntheses, Crystal Structures, and Antitumor Activities of Copper(II) and Nickel(II) Complexes with 2-((2-(Pyridin-2-yl)hydrazono)methyl)quinolin-8-ol. International Journal of Molecular Sciences. 2018; 19(7):1874. https://doi.org/10.3390/ijms19071874
Chicago/Turabian StyleYang, Qi-Yuan, Qian-Qian Cao, Qi-Pin Qin, Cai-Xing Deng, Hong Liang, and Zhen-Feng Chen. 2018. "Syntheses, Crystal Structures, and Antitumor Activities of Copper(II) and Nickel(II) Complexes with 2-((2-(Pyridin-2-yl)hydrazono)methyl)quinolin-8-ol" International Journal of Molecular Sciences 19, no. 7: 1874. https://doi.org/10.3390/ijms19071874





