Abstract
Long non-coding RNAs (lncRNAs) are classified as a group of transcripts which regulate various biological processes, such as RNA processing, epigenetic control, and signaling pathways. According to recent studies, lncRNAs are dysregulated in cancer and play an important role in cancer incidence and spreading. There is also an association between lncRNAs and the overexpression of some tumor-associated proteins, including carbonic anhydrases II, IX, and XII (CA II, CA IX, and CA XII). Therefore, not only CA inhibition, but also lncRNA modulation, could represent an attractive strategy for cancer prevention and therapy. Experimental studies have suggested that herbal compounds regulate the expression of many lncRNAs involved in cancer, such as HOTAIR (HOX transcript antisense RNA), H19, MALAT1 (metastasis-associated lung adenocarcinoma transcript 1), PCGEM1 (Prostate cancer gene expression marker 1), PVT1, etc. These plant-derived drugs or phytochemicals include resveratrol, curcumin, genistein, quercetin, epigallocatechin-3-galate, camptothcin, and 3,3′-diindolylmethane. More comprehensive information about lncRNA modulation via phytochemicals would be helpful for the administration of new herbal derivatives in cancer therapy. In this review, we describe the state-of-the-art and potential of phytochemicals as modulators of lncRNAs in different types of cancers.
1. Introduction
It is known that only about 2% of the human genome is transcribed into proteins or regulatory elements, while the rest of the genome is either non-coding or transcribed into RNA, with no possibility for translation to any protein, although it is biologically active [1]. These transcribed RNAs are called non-coding RNAs (ncRNAs) [2]. ncRNAs are classified into two groups: (1) small non-coding RNAs, which are about 22 nucleotides, and (2) long non-coding RNAs (lncRNAs), which are longer than 200 nucleotides with no open reading frame (ORF) restriction [3]. H19 was the first lncRNA reported in 1990 by Brannan et al. [4]. H19 is an imprinted oncofetal RNA, the expression of which decreases after birth, while the overexpression of H19 lncRNA has been reported in many cancer types in humans [5]. The latest lncRNAs have been reported in NONCODE [6], which is a comprehensive database covering non-coding RNAs. It presents data for 17 species, including 172,216 (as of February 2019) human lncRNA transcripts, which are able to regulate cell growth, development, differentiation, and gene expression [7]. Furthermore, lncRNAs play an important role in the occurrence of various diseases, such as cancer, whenever they are dysregulated [8]. They take part in cellular proliferation, apoptosis, and migration in a variety of cancers [9], such as breast cancer [10], prostate cancer [11], renal cancer [12], pancreatic cancer [13], and lung cancer [14]. Recently, numerous studies have introduced new types of drugs derived from plants (phytochemicals), which regulated the expression of several lncRNAs in cancer cells with no side effects [15].
It is well-documented that healthy nutrition prevents cancer. In contrast, the consumption of red meat [16] and high-fat diets [17] are associated with cancer induction. On the other hand, it was shown that vitamins B, D, and E [18,19,20] prevent different kind of tumors, including colorectal adenomas and prostate cancer. In addition, many other factors have been associated with cancer prevention or therapy as potential targets. They also include carbonic anhydrase (CA) enzymes, especially CA II, CA IX, and CA XII, which are overexpressed in certain cancers. Cas, as the major regulators of pH homeostasis, are induced by hypoxia and aid cancer cell survival [21,22]. Studies have shown the overexpression of cancer-related CAs, such as CA IX, in tumor cells, while their expression in normal cells is often low [23]. These facts and a number of recent publications suggest that cancer-related CAs are, indeed, potential and promising anti-cancer targets [22]. These CAs can be efficiently inhibited using various types of inhibitors, such as 7-aryl-triazolyl-substituted sulfocoumarins [24], acetazolamide [25,26,27,28,29,30,31,32], 6-ethoxy-2-benzothiazolesulfonamide (EZA) [33], benzene sulfonamides [34], 1,3,4-thiadiazole-2-sulfonamide [35], and sulfamide-related compounds [36]. CAs may also be modulated by lncRNAs via the administration of phytochemical compounds.
Phytochemicals are non-nutritive chemical components taken from various vegetables, fruits, beverages, and other green plants. Generally, the mechanism of action of these compounds occurs through the simulation of hormones, while they are known by their anti-oxidant and anti-inflammatory activities in cells [37,38,39,40]. To date, many phytochemicals have been identified and several are considered potential drugs due to their anticancer properties. They can be used as single chemopreventive drugs or synergistically with other routine anticancer drugs. This kind of anticancer drug administration can improve the efficacy of the treatment strategy, and optimally, with minimal or no side effects [41,42]. It has been suggested that phytochemicals act through the modulation of different signaling pathways via the regulation of significant molecular targets [43,44]. We hypothesize that they could also function by modulating the expression of enzymes, such as CAs, which are important for carcinogenic processes. In this review, we describe the state-of-the-art of how lncRNAs and cancer-related CAs could be modulated and inhibited by defined phytochemicals as an additional option for cancer prevention and treatment.
1.1. Biogenesis of lncRNA
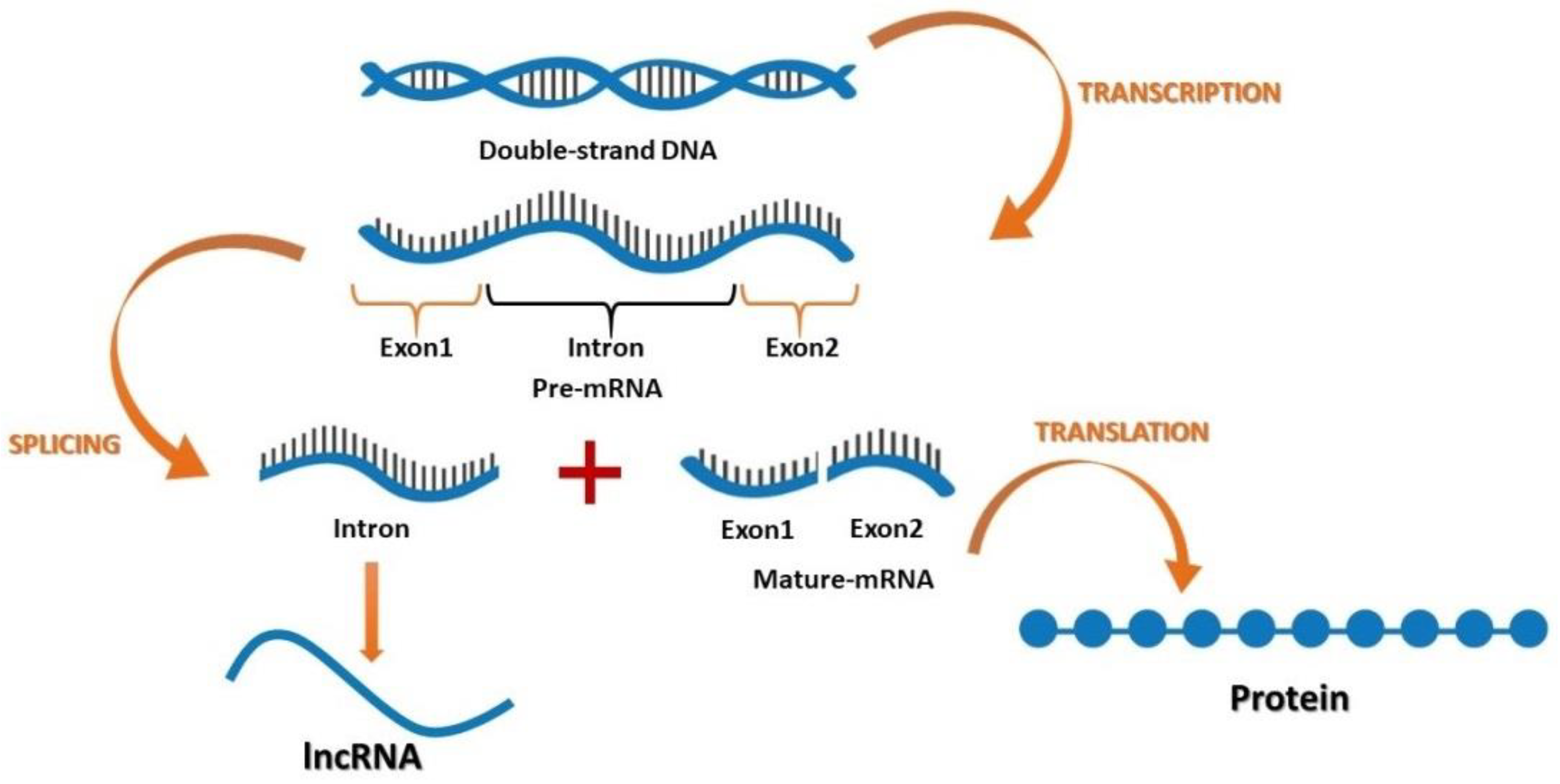
After the discovery of coding and non-coding parts of the genome, it was suggested that non-coding sections may play an important role in cellular activities [45]. Furthermore, recent findings have suggested that lncRNAs function in various cancers, where their contribution is based on developmental and tissue specific expression patterns [46,47,48,49,50,51,52,53,54]. Both coding and non-coding genes carry genetic information with different functions. According to their location in the genome, lncRNAs can be divided into four groups: (1) the intergenic lncRNAs, which are located between two genes; (2) the sense or antisense lncRNAs, which may overlap with an exon of another transcript in the same or opposite direction; (3) the intronic lncRNAs, which reside within an intron and do not overlap with any exon; and (4) the processed transcripts, which reside in a locus where none of the transcript has an ORF and thus, do not fit into any other categories because of structural complexity (Figure 1).
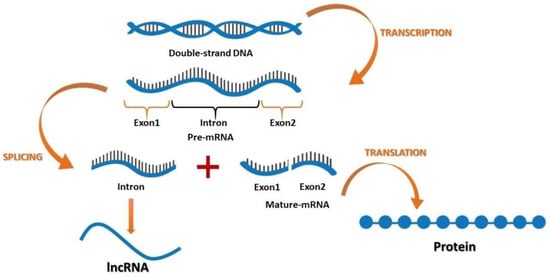
Figure 1.
The flow of genetic information encoding for mRNA and long non-coding RNA (lncRNA).
Kapranov et al. [55] declared that lncRNAs can be generated during RNA purification, where some sections appeared in a silica column. The researchers suggested that this demonstrated a random cutoff from RNA with more than 200 nucleotides which can bind to the RNA purification column, while it is not functional. Then, they identified three major properties of this RNA molecule: (1) it might have or does not have an ORF for coding more than 100 amino acids; (2) there is no need for this section to produce a protein, but is still functional [56] and ; (3) it can contain both coding and non-coding domains [57,58,59].
The current and widely accepted description of lncRNAs states that they are RNA molecules which do not fit in any other classes of RNAs [60,61], do not contain any ORF restriction, and do not encode any protein. According to various studies, lncRNAs include newly discovered segments of RNA, which are overexpressed in different cancer cell types (Table 1). Cancer studies have indicated that HOTAIR, as one of the best known lncRNAs, is overexpressed in numerous malignancies, including breast, colorectal, renal, and pancreatic cancers [62]. On the other hand, it has been shown that downregulation of HOTAIR expression leads to activation of the PI3K-AKT-mTOR1 signaling pathway in most cancers [63,64]. Inhibition of HOTAIR also upregulated miR-454-3p expression in chondrosarcoma, which consequently repressed the STAT3 signaling pathway [65]. The overexpression of HOTAIR in renal cancer decreased apoptosis and increased cell migration and invasion [66]. Tang et al. demonstrated that the knockdown of HOTAIR released caspase-2 through activation of the apoptosis signaling pathway during cancer treatment [67].

Table 1.
Studied long non-coding RNAs (lncRNAs) in different types of cancer.
Upregulation of MALAT1 was observed in many kinds of cancers, like oral, bladder, and colorectal carcinomas, as well as osteosarcoma [94,95,96,97]. The levels of H19 increased in various cancers, such as gastric and gallbladder carcinomas [89,90,91]. Other lncRNAs, such as PCGEM1, HOTAIR, and AK001796, were overexpressed in most cases of prostate cancer and phytochemical-induced anticancer activities [82,103,112,113]. Additionally, the role of HULC in promoting oral and liver cancers was proposed [75,76,114]. In pancreatic cancer, the expression of HOTAIR and PVT1 was upregulated [104,105,106]. Moreover, HOTAIR overexpression induced thyroid cancer in humans [83].
1.2. Modulation of lncRNA by phytochemicals
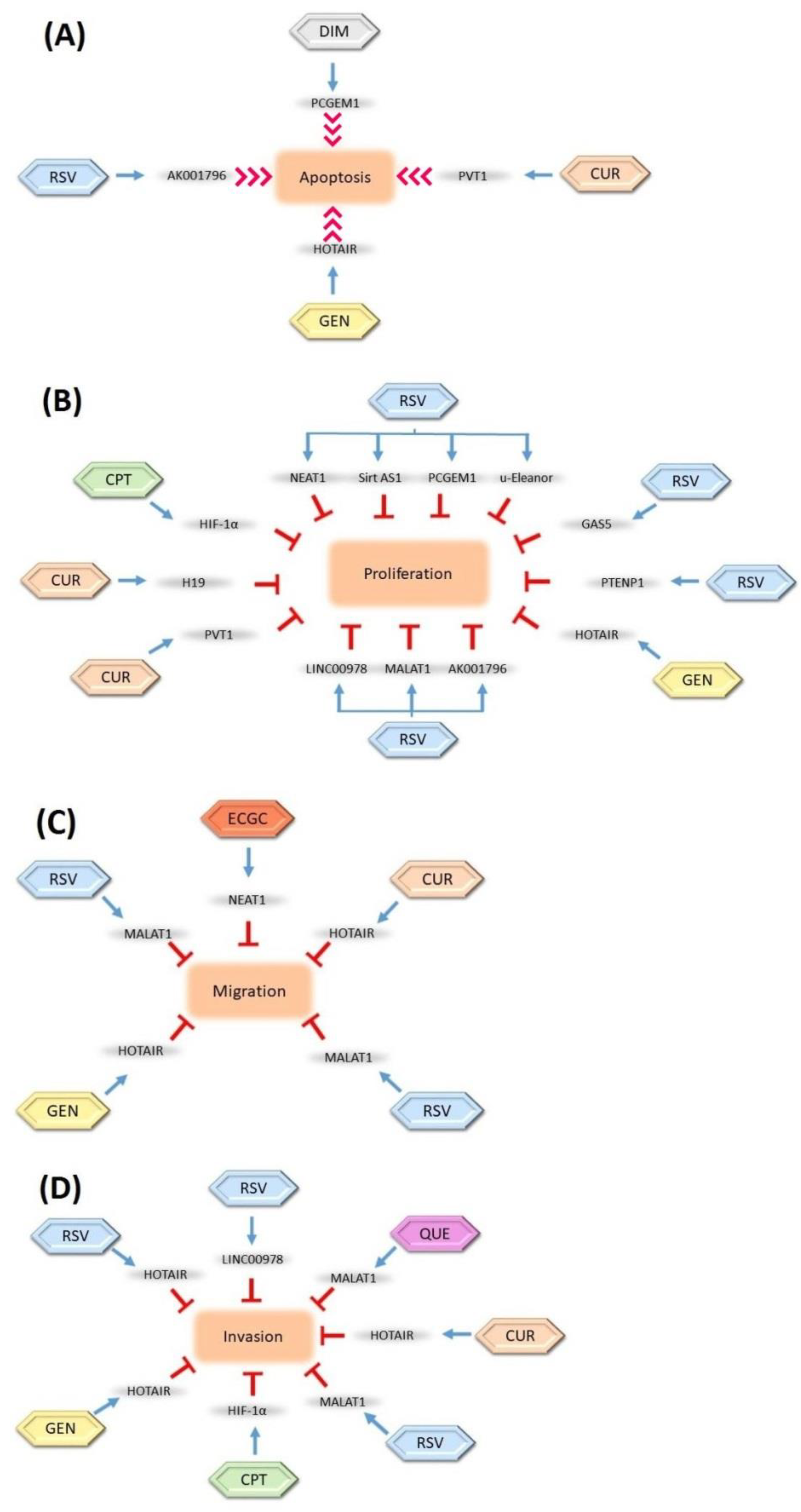
lncRNAs are considered great targets for anticancer studies due to their potential tumor suppressor abilities. Several studies have suggested that the modulation of lncRNAs with various phytochemicals could be a novel option in cancer therapy. It has been clearly indicated that these lncRNAs are regulated by defined phytochemicals (Figure 2).
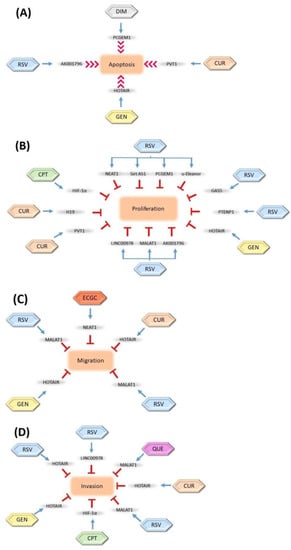
Figure 2.
Regulation of long non-coding RNAs (lncRNAs) by natural compounds and their inhibition effects on cell (A) apoptosis, (B) proliferation, (C) migration, and (D) invasion. The inhibition relationships are denoted as red stop symbols, whereas positive interactions are denoted as normal blue arrows. CUR: Curcumin, GEN: Genistein, RSV: Resveratrol, ECGC: Epigallocatechin-3-gallate, CPT: Camptothecin, DIM: 3,3-diindolylmethane, QUE: Quercetin. The blue arrows show the modulation roles of phytochemicals, the red arrows show the induction role of phytochemicals, and the T bars show the inhibition role of phytochemicals on the lncRNAs.
These phytochemicals include various compounds, some of which are presented in the following paragraphs.
1.3. Camptothecin (CPT)
Camptothecin (CPT, C20H16N2O4) is an alkaloid derived from a Chinese tree Camptotheca acuminate (happy tree). CPT has an inhibitory role in topoisomerase I and possesses antitumor activity [115,116,117]. CPT was demonstrated to suppress hypoxia-inducible factor 1 alpha (HIF-1α) -antisense RNA 1 in different human cancer types [77,106,115]. CPT also induces apoptosis in cardiovascular and kidney carcinomas, which results in an enhancement of the expression of antisense lncRNA. In another study, CPT treatment was shown to regulate the expression of lncRNA HIF-1α synergically with miR-17-5-p and miR-155 [78]. CPT has the ability to reduce CA IX expression in the cancer zone through the inhibition of angiogenesis and hypoxia. CPT has been conjugated to a linear, cyclodextrin-polyethylene glycol (CD-PEG) copolymer to form CRLX101 as a nanoparticle-drug conjugate (NDC). The conjugation step revealed that CRLX101 was more efficient than CPT in terms of the induction of apoptosis and supression of angiogenesis [118,119,120,121] (Table 2).

Table 2.
Long non-coding RNAs (lncRNAs) and carbonic anhydrases (CAs) affected by phytochemicals.
1.4. Curcumin
Curcumin (diferul[84]oylmethane) (C21H20O6 or C21H20O6) is a polyphenol derived from a perennial herbaceous plant, Curcuma longa [122]. This spicy yellow powder is used as an anti-inflammatory, antimicrobial, and antioxidant in traditional Asian medicine [123,124]. Curcumin acts as a chemopreventive and chemotherapeutic drug against various types of tumors, and is an important lncRNA regulator in cancers [125]. Petric et al. have shown that curcumin has an inhibitory effect on some oncogenic signaling pathways, including NF-kB, and induces apoptotic processes in breast cancer [38]. In another study, curcumin inhibited the overexpression of GAS5 in lung cancer by affecting signaling pathways, such as NF-kB, STAT3, and PI3K/Akt, to suppress tumor cell proliferation [126]. Curcumin also caused the modulation of tumor suppressor HOTAIR in pancreatic cancer [13], prostate cancer [88], hepatocellular carcinoma (HCC) [84,86], nasopharyngeal carcinoma (NPC) [81], breast cancer [87], lung cancer [80], and renal cancer [62,84,85,86,87,104,127]. It seems that the upregulation of HOTAIR has a controversial effect in terms of the occurrence of different cancer types and response to therapy methods, so radioresistance in breast cancer is enhanced by upregulated HOTAIR [87]. In addition, the expression level of HOTAIR is higher in renal cell carcinoma in comparison with normal kidney cells and a correlation has been shown between the upregulation of HOTAIR and distant metastasis in renal cell carcinoma malignancy [128]. Therefore, curcumin acts as a HOTAIR modulator, which consequently modulates the miR-19/PTEN/AKT/p53 axis in cancers [129].
Another tumor suppressor, H19, is induced by curcumin and directly inhibits p53 activation [90]. Overexpression of p53 can lead to colorectal and pancreatic cancer [130,131,132]. In nasopharyngeal carcinoma, high levels of expression have been demonstrated for six lncRNAs, including AF086415, AK095147, RP1-179N16.3, MUDENG, AK056098, and AK294004 [68,69]. Curcumin suppresses the expression of these lncRNAs and can tenderize cancer cells to radiotherapy [133]. On the other hand, different types of Cas, including isozymes I, II, IX, and XII, which are overexpressed in several cancers, are inhibited by curcumin and it’s phenolic compounds [134,135,136]. In addition, sulfonamides containing curcumin inhibited CA I [137]. The combination of curcumin with other factors can be a potent strategy in the treatment of tumor cells. This includes the combination of curcumin with glucose restriction [138] and dopamine-related compounds as phenolic sulfonamides and inhibitors of CA I and CA II [139] (Table 2).
1.5. 3,3′-diindolylmethane (DIM)
3,3′-Diindolylmethane (DIM, C17H14N2) is a known phytochemical compound derived from indole-3-carbinol (I3C) [140]. It is found in cruciferous vegetables like broccoli, cabbage, and kale [141]. DIM has an impact on signaling pathways and can regulate cell division, apoptosis, and angiogenesis in cancer cells [142]. It has been demonstrated that DIM inhibits PCGEM1 expression and induces apoptosis in prostate cancer [103]. Moreover, it has been observed that DIM indirectly suppresses the Akt/FOXM1 signaling cascade by regulating FOXM1 gene expression [143]. FOXM1 regulates various lncRNAs in some carcinomas [144]. Bioresponse formulated 3,3′-diindolylmethane (BR-DIM) decreases androgen receptor (AR) variants and AR3 expression in prostate cancer [103]. A study revealed that the combination of indolin-based compounds with sulfonamides can inhibit CA I, II, IV, and VII [145] (Table 2).
1.6. Epigallocatechin-3-galate (EGCG)
Epigallocatechin (EGCG, C15H14O7) is a known polyphenol flavonoid derived from almond and green tea [146,147,148,149,150,151]. This compound regulates the expression of non-coding RNAs in tumors and has notable anticancer, anti-inflammatory, and antioxidant features [38]. EGCG modulates various signaling pathways, such as NF-kB, MAPK, Akt, PI3K, PTEN, and mTORC1, as well as the expression of the estrogen receptor (ER) [152,153,154]. It has been shown that EGCG suppresses a lncRNA, AT102202, which downregulates the expression of 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR) in human hepatocytes, leading to the uptake of cholesterol by the liver [70]. A study showed that polyphenol Epigallocatechin upregulates CA IX in breast cancer cells, which may possess strong antioxidative and antiapoptotic properties [155]. It has also been demonstrated that EGCG as a content of flavonoids in green tea has a suppression effect on CA II [156] (Table 2).
1.7. Genistein
Genistein (C15H10O5), a dietary soy isoflavone, is another phytochemical compound with in vitro and in vivo antitumor effects [157]. It has shown some anti-proliferation effects on many types of human cancers, such as breast, renal, and prostate cancers [38,66,82,158,159,160]. Genistein modulates the expression level of HOTAIR in breast cancer, which consequently modulates the activity of the PI3K/Akt signaling pathway [161]. Genistein suppresses the progression of renal cancer by inhibiting HOTAIR [66]. It was found that the miR-141 expression was upregulated, while the HOTAIR expression was downregulated, by genistein in cancer cells [157]. In prostate cancer, genistein reduced the HOTAIR and miR-34a expression synergically. Another study also suggested that genistein has antitumor effects in colorectal cancer by affecting HOTAIR [162]. In addition, genistein induces apoptosis in cancer cells, including breast, prostate, gastric, lung, pancreatic, melanoma, and renal cancers, by inhibiting several signaling pathways, such as Wnt and Akt [82,163]. The decreased expression of HOTAIR leads to apoptosis, which has been induced by genistein in multiple types of cancer [88]. In this case, most studies considered the correlation between phyto-isoflavones and -oestrogens in cervix, ovariectomy, uterus, and liver cancers through the modulatory effect of genistein on CA II expression [164,165,166] (Table 2).
1.8. Quercetin
Quercetin (C15H10O7) is a polyphenolic flavonoid with chemopreventive properties. This dietary antioxidant is derived from several plants and fruits, such as red grapes, broccoli, and some berries. Quercetin downregulated the expression of DBH-AS1 in hepatocellular carcinoma through its antiproliferative and antioxidant activities [71,72,73]. It was reported that quercetin acts as an inhibitor in different signaling pathways like Akt/mTOR/P70S6K and PI3K/AkT [97,167,168]. Most studies have confirmed the inhibition activity of quercetin on CA isoforms, including CA I, II, III, IV, XII, and XIV [169,170,171]. Recently, quercetin-modified metal–organic frameworks (Zr-MOF-QU) as the novel type of Zr-MOF nanoparticles have shown excellent efficiency for CA IX inhibition in tumor cells [172] (Table 2). Zr-MOF-QU seems to be used successfully in radiotherapy.
1.9. Resveratrol
Resveratrol (3,4′,5 tri-hydroxystilbene) (C14H12O3) is a natural polyphenol compound found in various plants and herbs, including blueberries, raspberries, mulberries, and the skin of grapes [173]. Resveratrol has anti-inflammatory and antiproliferative properties, as well as antitumor effects on various human cancers [93,174], including prostate [113,175], thyroid [176], colorectal [177,178], breast [179,180], lung [181,182], and bladder cancers [93,95,111,113,183]. Resveratrol inhibits the AR signaling pathway in prostate cancer by affecting PCGEM1 and PRNCR1 [107,108,109,110]. Another prostate cancer study revealed that resveratrol is a reverse potent stimulator in the reduction of PCAT29 expression induced by a cancer cell line [175]. Synergistic growth inhibition activity of resveratrol and AK001796 has been demonstrated in lung cancer [14]. In another study, it was reported that resveratrol modulates the Wnt/β-Catenin signaling pathway by the downregulation of MALAT1 in colorectal cancer [95]. It has been demonstrated that the anticancer effect of resveratrol on estrogen receptor-α in breast cancer is due to the suppression of a lncRNA, u-Eleanor [111]. The aromatase inhibition property of resveratrol makes it a potential antitumor candidate in breast cancer treatment through the suppression of an oncogene, LINC00978 [92,93]. In the same study, LINC00978 functioned as a mediator for resveratrol to suppress the proliferation of breast cancer cells [93]. There is also evidence that the treatment of lung cancer with resveratrol results in the downregulation of AK001796 expression. Studies have revealed that polyphenol resveratrol could inhibit CA I‒XV in cancers, so CA II was inhibited more efficiently [135,184] (Table 2).
1.10. The Mechanisms of lncRNA Regulation by Phytochemicals
In recent years, several lncRNAs with interfering properties have been identified in different types of cancers. Thus far, the exact mechanism of lncRNA regulation in normal physiology or cancer cells is still unknown [185,186]. There is some evidence suggesting that lncRNAs are involved in the regulation of gene expression via transcriptional and post-transcriptional mechanisms and chromatin modification [9]. Furthermore, previous studies have defined that phytochemicals change the dysregulation of lncRNAs in various cancer types [187,188].
1.11. Transcriptional and Post-Transcriptional Regulation of lncRNAs
Experimental studies have revealed that there are several transcriptional factors regulating the expression of lncRNAs, and subsequently modulating pathological conditions in cancer. The studies have indicated that phytochemicals can adjust lncRNA expression via transcriptional regulation through various mechanisms. It has been shown that camptothecin decreases the transcription level of the HIF-1α gene in renal cancer [77]. Hypoxia inducible factor-1α is an important cell response modulator, which is regulated by lncRNAs and miRNAs [189,190].
TOP2A is a necessary element for the transcriptional activity of RNA polymerase II, which leads to a reduction of LS Pol II-mediated H19 transcription. Kujundzic and coworkers demonstrated that curcumin downregulates TOP2A expression and consequently inhibits H19 expression in tumor cell lines [191]. In another study, it was shown that curcumin regulates H19 through affecting the PI3K/Akt signaling pathway [192,193,194]. It was also shown that 3,3′-diindolylmethane inhibited the expression of PCGEM1 by banning its interaction with a nuclear RNA-binding protein, p54/nrb [103]. EGCG suppresses the promoter of the Cu(I) transport gene 1 (CTR1) in cancer cells, while it induces it through NEAT1, which is associated with hsa-miR-98-5p [195,196,197,198]. Furthermore, HOTAIR upregulates c-Myc in breast and ovarian cancers, which in turn promotes cancer cell proliferation [7]. Genistein downregulates the expression of HOTAIR at the transcription level in several cancers. The AR activation is a significant element in castration-resistant prostate cancer (CRPC) and increasing the expression level of HOTAIR [199].
1.12. Chromatin Modification by lncRNAs
lncRNAs are vital regulators of the genome structure, are able to interact with chromatin-modifying enzymes, and control the chromatin structure and accessibility to genetic information through reprogramming mechanisms [200,201]. The DNA methylation of genes inhibits the regulation of histone-modifying enzymes, which contributes to prostate cancer progression [175]. Several lncRNAs, such as PTENP1, Linc00963, PCGIM1, PRNCR1, CBR-3AS1, CTP1AS, GAS5, ANRIL, ANRASSF1, and PCAT1, upregulate the proliferation of cancer cells [59,74,102,175,202,203,204,205,206,207,208,209,210]. Resveratrol blocked the reduction of PCAT29 expression of this lncRNA in hepatocellular carcinoma [98]. HOTAIR can act as a mediator of proliferation, migration, invasion, and apoptosis in breast, liver, and colon cancer metastasis through genetic regulation [62,86,211]. Experimental studies have shown that curcumin can repress metastasis and invasion via epigenetic modulation [212]. Generally, lncRNAs are impartible vital molecules that are involved in gene modification and reprogramming. Phytochemicals, with their regulatory effects on lncRNAs, can be helpful as natural drugs for various cancer therapies.
2. Discussion
The idea of chemoprevention instead of chemotherapy was suggested by Moon et al. in 1979 for the prevention of breast cancer in rats using N-(4-Hydroxyphenyl)retinamide as a new retinoid compound [213]. Several studies proved this opinion sound during the subsequent decades, and studies were focused on discovering effective molecular targets for the modulatory function of phytochemicals.
lncRNAs are newly discovered regulators of cell functions, which have attracted considerable attention in biological sciences. Recently, researchers have discovered numerous lncRNAs in humans and animals, while their precise function is still unknown. At the moment, we are able to detect and analyze some regulatory functions of lncRNAs in cells. They are clearly crucial modulators of cell proliferation in cancer, and thus research on these molecules may open new avenues for cancer therapy. Previous studies have shown that lncRNA-low expression in tumors (lncRNA-LET) is involved in the inhibition of cell proliferation and cancer and metastasis suppression [99,100,214], while an opposite correlation was found between CA IX (an endogenous hypoxia marker and metabolism reprogramming factor) and lncRNA-LET during hypoxia in hepatocellular carcinoma [101,215]. Hypoxia induces cellular responses during cancer progression, including the overexpression of CA IX due to HIF-1α and HIF-1β stabilization through the protein kinase A (PKA) signaling pathway [216,217]. On the other hand, lncRNAs can induce the overexpression of CA XII through the PKA signaling pathway in fibrolamellar carcinoma [217]. In addition to these discoveries in cancer, an association has been found between CA II in the network of regulators and 16 lncRNAs in thoracic aortic dissection [218].
Phytochemicals have exhibited properties to change the level of lncRNAs involved in cancer induction and progression by regulating lncRNA expression through different signaling pathway mechanisms. These herbal drugs repress the proliferation and survival of cancer cells. Importantly, they are impressive natural compounds with no signs of toxicity or side effects. They can be applied simultaneously with some chemotherapy compounds in cancers, which may significantly improve the overall therapy outcome. The medicinal properties of phytochemicals have been shown not only in cancers, but also in a variety of chronic diseases, such as Alzheimer’s disease, cardiovascular diseases, diabetes, ocular diseases, and rheumatoid arthritis, in which they are capable of curing the dysregulation of lncRNAs [97,219,220].
Whithin this context, another recommended cancer therapeutic approach is called Acridine Orange-photodynamic therapy (AO-PDT), which was exploited by Kusuzaki et al. in 2017 [221]. In this cancer therapy method, AO was delivered by natural nanovesicles (exosomes) and released by macrophages, which consequently increased the uptake of AO by cancer cells. For the implementation of PDT in CA inhibition, a sulfonamide derivative of AO was delivered to tumor cells for the inhibition of cancer-related CA IX and XII [222,223,224].
One of the most important reasons for cancer relapse is the extracellular acidity of the tumor’s microenvironment, which can strongly influence cancer progression [21,225,226]. On the other hand, acidosis is a critical factor in the progression of tumors through promoting metastasis. One of the therapeutic strategies agaisnt tumor acidity is the inhibition or targeting of tumor acidity by diet buffers, sequestering agents in an acidic microenvironment, and developing proton pump inhibitors (PPIs). The acidic microenvironments are prepared by proton transporters, including V-ATPase, the Na+/H+ exchanger (NHE), monocarboxylate transporters (MCTs), and CA IX [227,228]. Since pKa for CA IX and XII was ˂6.5 and 7.1, respectively, CA IX is more active at a low pH. Hence, the inhibition of CA IX by low toxicity-inhibitors, including synthetic compounds and phytochemicals, or applying a combinational therapy method and their successful delivery to tumor acidic microenvironments by exosome nanovesicles is a therapeutic approach in cancers to overcome microenvironmental acidification [222]. This method has shown significant promise, which can accelerate the development of novel cancer treatment options.
On the other hand, there are some concerns about phytochemicals, like their poor bioavailability and limited efficiency, so novel formulations of these herbal drugs, such as combinations with adjuvants, liposomes, and nanoparticles, are needed to improve the efficacy of the phytochemicals for the modulation of lncRNAs in cancer. Although the phytochemicals have modulatory effects on lncRNAs, and could thus be useful in cancer prevention, the other biological effects of these compounds on other targets are inevitable. In parallel to the effects of phytochemicals on lncRNAs, the cancer-related CAs can also be inhibited efficiently. It reveals that some unprecedented targets are affected by phytochemicals within cancer prevention and/or treatment. On the other hand, it is obvious that cancer therapy with phytochemicals is a new area of science and research about its mechanisms of effect is still at a developing stage. More studies are needed to understand the relationship between the mentioned herbal drugs and their effects in normal and tumor cells. It is clear that more studies, including in vitro and in vivo tests, are needed to shed some new light on this research path.
Author Contributions
All authors participated in the design of the study. T.S., R.A.T., and R.Z.E. carried out the design of the study under the supervision of S.P. T.S. and R.Z.E. drafted the first version of the manuscript. All authors participated in writing, reading, and approving the final manuscript.
Funding
This research was funded by the National Institute of Genetic Engineering and Biotechnology (NIGEB) of Islamic Republic of Iran, the Academy of Finland, Finnish Cultural Foundation, Sigrid Juselius Foundation, Jane and Aatos Erkko Foundation, and Competitive Research Funding of the Tampere University Hospital. The APC was funded by Tampere University, Finland.
Conflicts of Interest
The authors declare that they have no competing interests.
References
- Kung, J.T.; Colognori, D.; Lee, J.T. Long noncoding RNAs: Past, present, and future. Genetics 2013, 193, 651–669. [Google Scholar] [CrossRef] [PubMed]
- Pickard, M.R.; Mourtada-Maarabouni, M.; Williams, G.T. Long non-coding RNA GAS5 regulates apoptosis in prostate cancer cell lines. Biochim. Biophys. Acta 2013, 1832, 1613–1623. [Google Scholar] [CrossRef]
- Harrow, J.; Frankish, A.; Gonzalez, J.M.; Tapanari, E.; Diekhans, M.; Kokocinski, F.; Aken, B.L.; Barrell, D.; Zadissa, A.; Searle, S. GENCODE: The reference human genome annotation for The ENCODE Project. Genome Res. 2012, 22, 1760–1774. [Google Scholar] [CrossRef] [PubMed]
- Brannan, C.I.; Dees, E.C.; Ingram, R.S.; Tilghman, S.M. The product of the H19 gene may function as an RNA. Mol. Cell. Biol. 1990, 10, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Pei, C.S.; Wu, H.Y.; Fan, F.T.; Wu, Y.; Shen, C.S.; Pan, L.Q. Influence of curcumin on HOTAIR-mediated migration of human renal cell carcinoma cells. Asian Pac. J. Cancer Prev. Apjcp 2014, 15, 4239–4243. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Zhang, L.; Guo, J.; Niu, Y.; Wu, Y.; Li, H.; Zhao, L.; Li, X.; Teng, X.; Sun, X.; et al. NONCODEV5: A comprehensive annotation database for long non-coding RNAs. Nucleic Acids Res. 2018, 46, D308–D314. [Google Scholar] [CrossRef] [PubMed]
- Bhan, A.; Soleimani, M.; Mandal, S.S. Long noncoding RNA and cancer: A new paradigm. Cancer Res. 2017, 77, 3965–3981. [Google Scholar] [CrossRef] [PubMed]
- Kapusta, A.; Feschotte, C. Volatile evolution of long noncoding RNA repertoires: Mechanisms and biological implications. Trends Genet. 2014, 30, 439–452. [Google Scholar] [CrossRef] [PubMed]
- Ponting, C.P.; Oliver, P.L.; Reik, W. Evolution and functions of long noncoding RNAs. Cell 2009, 136, 629–641. [Google Scholar] [CrossRef]
- Khan, N.; Mukhtar, H. Cancer and metastasis: Prevention and treatment by green tea. Cancer Metastasis Rev. 2010, 29, 435–445. [Google Scholar] [CrossRef]
- Kogo, R.; Shimamura, T.; Mimori, K.; Kawahara, K.; Imoto, S.; Sudo, T.; Tanaka, F.; Shibata, K.; Suzuki, A.; Komune, S.; et al. Long noncoding RNA HOTAIR regulates polycomb-dependent chromatin modification and is associated with poor prognosis in colorectal cancers. Cancer Res. 2011, 71, 6320–6326. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.C.; Kallin, E.M.; Zhang, Y. Role of H3K27 methylation in the regulation of lncRNA expression. Cell Res. 2010, 20, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Li, Z.; Zheng, S.; Chen, H.; Zhao, X.; Gao, W.; Bi, Z.; You, K.; Wang, Y.; Li, W.; et al. The long non-coding RNA HOTAIR affects the radiosensitivity of pancreatic ductal adenocarcinoma by regulating the expression of Wnt inhibitory factor 1. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2016, 37, 3957–3967. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Xu, E.; Dai, J.; Liu, B.; Han, Z.; Wu, J.; Zhang, S.; Peng, B.; Zhang, Y.; Jiang, Y. A novel long noncoding RNA AK001796 acts as an oncogene and is involved in cell growth inhibition by resveratrol in lung cancer. Toxicol. Appl. Pharmacol. 2015, 285, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Spizzo, R.; Almeida, M.I.; Colombatti, A.; Calin, G.A. Long non-coding RNAs and cancer: A new frontier of translational research? Oncogene 2012, 31, 4577–4587. [Google Scholar] [CrossRef]
- Bingham, S.A.; Hughes, R.; Cross, A.J. Effect of white versus red meat on endogenous N-nitrosation in the human colon and further evidence of a dose response. J. Nutr. 2002, 132, 3522S–3525S. [Google Scholar] [CrossRef]
- Sieri, S. Consuming a high-fat diet is associated with increased risk of certain types of BC. J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef]
- Johansson, M.; Relton, C.; Ueland, P.M.; Vollset, S.E.; Midttun, O.; Nygard, O.; Slimani, N.; Boffetta, P.; Jenab, M.; Clavel-Chapelon, F.; et al. Serum B vitamin levels and risk of lung cancer. JAMA 2010, 303, 2377–2385. [Google Scholar] [CrossRef]
- Jenab, M.; Bueno-de-Mesquita, H.B.; Ferrari, P.; van Duijnhoven, F.J.; Norat, T.; Pischon, T.; Jansen, E.H.; Slimani, N.; Byrnes, G.; Rinaldi, S.; et al. Association between pre-diagnostic circulating vitamin D concentration and risk of colorectal cancer in European populations:a nested case-control study. BMJ 2010, 340, b5500. [Google Scholar] [CrossRef]
- Klein, E.A.; Thompson, I.M., Jr.; Tangen, C.M.; Crowley, J.J.; Lucia, M.S.; Goodman, P.J.; Minasian, L.M.; Ford, L.G.; Parnes, H.L.; Gaziano, J.M.; et al. Vitamin E and the risk of prostate cancer: The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2011, 306, 1549–1556. [Google Scholar] [CrossRef]
- Pastorekova, S.; Gillies, R.J. The role of carbonic anhydrase IX in cancer development: Links to hypoxia, acidosis, and beyond. Cancer Metastasis Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Mboge, M.Y.; Mahon, B.P.; McKenna, R.; Frost, S.C. Carbonic Anhydrases: Role in pH Control and Cancer. Metabolites 2018, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Mahon, B.P.; Pinard, M.A.; McKenna, R. Targeting carbonic anhydrase IX activity and expression. Molecules 2015, 20, 2323–2348. [Google Scholar] [CrossRef] [PubMed]
- Nocentini, A.; Ceruso, M.; Carta, F.; Supuran, C.T. 7-Aryl-triazolyl-substituted sulfocoumarins are potent, selective inhibitors of the tumor-associated carbonic anhydrase IX and XII. J. Enzym. Inhib. Med. Chem. 2016, 31, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Parkkila, S.; Rajaniemi, H.; Parkkila, A.K.; Kivela, J.; Waheed, A.; Pastorekova, S.; Pastorek, J.; Sly, W.S. Carbonic anhydrase inhibitor suppresses invasion of renal cancer cells in vitro. Proc. Natl. Acad. Sci. USA 2000, 97, 2220–2224. [Google Scholar] [CrossRef] [PubMed]
- Zolfaghari Emameh, R.; Kuuslahti, M.; Vullo, D.; Barker, H.R.; Supuran, C.T.; Parkkila, S. Ascaris lumbricoides beta carbonic anhydrase: A potential target enzyme for treatment of ascariasis. Parasites Vectors 2015, 8, 479. [Google Scholar] [CrossRef]
- Bua, S.; Haapanen, S.; Kuuslahti, M.; Parkkila, S.; Supuran, C.T. Sulfonamide Inhibition Studies of a New beta-Carbonic Anhydrase from the Pathogenic Protozoan Entamoeba histolytica. Int. J. Mol. Sci. 2018, 19, 3946. [Google Scholar] [CrossRef]
- Zolfaghari Emameh, R.; Barker, H.R.; Syrjanen, L.; Urbanski, L.; Supuran, C.T.; Parkkila, S. Identification and inhibition of carbonic anhydrases from nematodes. J. Enzym. Inhib. Med. Chem. 2016, 31, 176–184. [Google Scholar] [CrossRef]
- Zolfaghari Emameh, R.; Syrjanen, L.; Barker, H.; Supuran, C.T.; Parkkila, S. Drosophila melanogaster: A model organism for controlling Dipteran vectors and pests. J. Enzym. Inhib. Med. Chem. 2015, 30, 505–513. [Google Scholar] [CrossRef]
- Zolfaghari Emameh, R.; Kuuslahti, M.; Nareaho, A.; Sukura, A.; Parkkila, S. Innovative molecular diagnosis of Trichinella species based on beta-carbonic anhydrase genomic sequence. Microb. Biotechnol. 2016, 9, 172–179. [Google Scholar] [CrossRef]
- Zolfaghari Emameh, R.; Barker, H.; Tolvanen, M.E.; Ortutay, C.; Parkkila, S. Bioinformatic analysis of beta carbonic anhydrase sequences from protozoans and metazoans. Parasit. Vectors 2014, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Zolfaghari Emameh, R.; Barker, H.; Hytonen, V.P.; Tolvanen, M.E.; Parkkila, S. Beta carbonic anhydrases: Novel targets for pesticides and anti-parasitic agents in agriculture and livestock husbandry. Parasit. Vectors 2014, 7, 403. [Google Scholar] [CrossRef] [PubMed]
- Noor, S.I.; Jamali, S.; Ames, S.; Langer, S.; Deitmer, J.W.; Becker, H.M. A surface proton antenna in carbonic anhydrase II supports lactate transport in cancer cells. Elife 2018, 7. [Google Scholar] [CrossRef]
- Mboge, M.Y.; Chen, Z.; Wolff, A.; Mathias, J.V.; Tu, C.; Brown, K.D.; Bozdag, M.; Carta, F.; Supuran, C.T.; McKenna, R.; et al. Selective inhibition of carbonic anhydrase IX over carbonic anhydrase XII in breast cancer cells using benzene sulfonamides: Disconnect between activity and growth inhibition. PLoS ONE 2018, 13, e0207417. [Google Scholar] [CrossRef] [PubMed]
- Supuran, C.T.; Scozzafava, A. Carbonic anhydrase inhibitors—Part 94. 1,3,4-thiadiazole-2-sulfonamidederivatives as antitumor agents? Eur. J. Med. Chem. 2000, 35, 867–874. [Google Scholar] [CrossRef]
- Gavernet, L.; Gonzalez Funes, J.L.; Palestro, P.H.; Bruno Blanch, L.E.; Estiu, G.L.; Maresca, A.; Barrios, I.; Supuran, C.T. Inhibition pattern of sulfamide-related compounds in binding to carbonic anhydrase isoforms I, II, VII, XII and XIV. Bioorg. Med. Chem. 2013, 21, 1410–1418. [Google Scholar] [CrossRef]
- Block, G.; Patterson, B.; Subar, A. Fruit, vegetables, and cancer prevention: A review of the epidemiological evidence. Nutr. Cancer 1992, 18, 1–29. [Google Scholar] [CrossRef]
- Petric, R.C.; Braicu, C.; Raduly, L.; Zanoaga, O.; Dragos, N.; Monroig, P.; Dumitrascu, D.; Berindan-Neagoe, I. Phytochemicals modulate carcinogenic signaling pathways in breast and hormone-related cancers. Oncotargets Ther. 2015, 8, 2053. [Google Scholar] [CrossRef]
- Reddy, L.; Odhav, B.; Bhoola, K. Natural products for cancer prevention: A global perspective. Pharmacol. Ther. 2003, 99, 1–13. [Google Scholar] [CrossRef]
- Steinmetz, K.A.; Potter, J.D. Vegetables, fruit, and cancer prevention: A review. J. Am. Diet. Assoc. 1996, 96, 1027–1039. [Google Scholar] [CrossRef]
- Siddique, Y.H.; Ara, G.; Beg, T.; Gupta, J.; Afzal, M. Assessment of cell viability, lipid peroxidation and quantification of DNA fragmentation after the treatment of anticancerous drug mitomycin C and curcumin in cultured human blood lymphocytes. Exp. Toxicol. Pathol. 2010, 62, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Lu, J.; Huang, M.; Li, Y.; Chen, M.; Wu, G.; Gong, J.; Zhong, Z.; Xu, Z.; Dang, Y. Anti-cancer natural products isolated from chinese medicinal herbs. Chin. Med. 2011, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, N.; Ghosh, P.C. Folate receptor mediated targeted delivery of ricin entrapped into sterically stabilized liposomes to human epidermoid carcinoma (KB) cells: Effect of monensin intercalated into folate-tagged liposomes. Eur. J. Pharm. Sci. 2011, 43, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, N.; Rathore, S.S.; Ghosh, P.C. Enhanced killing of human epidermoid carcinoma (KB) cells by treatment with ricin encapsulated into sterically stabilized liposomes in combination with monensin. Drug Deliv. 2011, 18, 394–404. [Google Scholar] [CrossRef]
- Gibb, E.A.; Brown, C.J.; Lam, W.L. The functional role of long non-coding RNA in human carcinomas. Mol. Cancer 2011, 10, 38. [Google Scholar] [CrossRef]
- Castle, J.C.; Armour, C.D.; Löwer, M.; Haynor, D.; Biery, M.; Bouzek, H.; Chen, R.; Jackson, S.; Johnson, J.M.; Rohl, C.A. Digital genome-wide ncRNA expression, including SnoRNAs, across 11 human tissues using polyA-neutral amplification. PLoS ONE 2010, 5, e11779. [Google Scholar] [CrossRef]
- Guffanti, A.; Iacono, M.; Pelucchi, P.; Kim, N.; Soldà, G.; Croft, L.J.; Taft, R.J.; Rizzi, E.; Askarian-Amiri, M.; Bonnal, R.J. A transcriptional sketch of a primary human breast cancer by 454 deep sequencing. BMC Genom. 2009, 10, 163. [Google Scholar] [CrossRef]
- Loewer, S.; Cabili, M.N.; Guttman, M.; Loh, Y.-H.; Thomas, K.; Park, I.H.; Garber, M.; Curran, M.; Onder, T.; Agarwal, S. Large intergenic non-coding RNA-RoR modulates reprogramming of human induced pluripotent stem cells. Nat. Genet. 2010, 42, 1113. [Google Scholar] [CrossRef]
- Maruyama, R.; Shipitsin, M.; Choudhury, S.; Wu, Z.; Protopopov, A.; Yao, J.; Lo, P.-K.; Bessarabova, M.; Ishkin, A.; Nikolsky, Y. Altered antisense-to-sense transcript ratios in breast cancer. Proc. Natl. Acad. Sci. USA 2012, 109, 2820–2824. [Google Scholar] [CrossRef]
- Mercer, T.R.; Dinger, M.E.; Sunkin, S.M.; Mehler, M.F.; Mattick, J.S. Specific expression of long noncoding RNAs in the mouse brain. Proc. Natl. Acad. Sci. USA 2008, 105, 716–721. [Google Scholar] [CrossRef]
- Perez, D.S.; Hoage, T.R.; Pritchett, J.R.; Ducharme-Smith, A.L.; Halling, M.L.; Ganapathiraju, S.C.; Streng, P.S.; Smith, D.I. Long, abundantly expressed non-coding transcripts are altered in cancer. Hum. Mol. Genet. 2007, 17, 642–655. [Google Scholar] [CrossRef] [PubMed]
- Rinn, J.L.; Kertesz, M.; Wang, J.K.; Squazzo, S.L.; Xu, X.; Brugmann, S.A.; Goodnough, L.H.; Helms, J.A.; Farnham, P.J.; Segal, E. Functional demarcation of active and silent chromatin domains in human HOX loci by noncoding RNAs. Cell 2007, 129, 1311–1323. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.M.; Perez, D.S.; Pritchett, J.R.; Halling, M.L.; Tang, H.; Smith, D.I. Identification of long stress-induced non-coding transcripts that have altered expression in cancer. Genomics 2010, 95, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Taft, R.J.; Pang, K.C.; Mercer, T.R.; Dinger, M.; Mattick, J.S. Non-coding RNAs: Regulators of disease. J. Pathol. 2010, 220, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Kapranov, P.; Cheng, J.; Dike, S.; Nix, D.A.; Duttagupta, R.; Willingham, A.T.; Stadler, P.F.; Hertel, J.; Hackermüller, J.; Hofacker, I.L. RNA maps reveal new RNA classes and a possible function for pervasive transcription. Science 2007, 316, 1484–1488. [Google Scholar] [CrossRef] [PubMed]
- Washietl, S.; Findeiß, S.; Müller, S.A.; Kalkhof, S.; von Bergen, M.; Hofacker, I.L.; Stadler, P.F.; Goldman, N. RNAcode: Robust discrimination of coding and noncoding regions in comparative sequence data. RNA 2011, 17, 578–594. [Google Scholar] [CrossRef] [PubMed]
- Candeias, M.M.; Malbert-Colas, L.; Powell, D.J.; Daskalogianni, C.; Maslon, M.M.; Naski, N.; Bourougaa, K.; Calvo, F.; Fåhraeus, R. P53 mRNA controls p53 activity by managing Mdm2 functions. Nat. Cell Biol. 2008, 10, 1098. [Google Scholar] [CrossRef]
- Martick, M.; Horan, L.H.; Noller, H.F.; Scott, W.G. A discontinuous hammerhead ribozyme embedded in a mammalian messenger RNA. Nature 2008, 454, 899. [Google Scholar] [CrossRef]
- Poliseno, L.; Salmena, L.; Zhang, J.; Carver, B.; Haveman, W.J.; Pandolfi, P.P. A coding-independent function of gene and pseudogene mRNAs regulates tumour biology. Nature 2010, 465, 1033. [Google Scholar] [CrossRef]
- Dinger, M.E.; Pang, K.C.; Mercer, T.R.; Mattick, J.S. Differentiating protein-coding and noncoding RNA: Challenges and ambiguities. PLoS Comput. Biol. 2008, 4, e1000176. [Google Scholar] [CrossRef]
- Mercer, T.R.; Dinger, M.E.; Mattick, J.S. Long non-coding RNAs: Insights into functions. Nat. Rev. Genet. 2009, 10, 155. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.A.; Shah, N.; Wang, K.C.; Kim, J.; Horlings, H.M.; Wong, D.J.; Tsai, M.-C.; Hung, T.; Argani, P.; Rinn, J.L. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature 2010, 464, 1071. [Google Scholar] [CrossRef] [PubMed]
- Hales, E.C.; Taub, J.W.; Matherly, L.H. New insights into Notch1 regulation of the PI3K–AKT–mTOR1 signaling axis: Targeted therapy of γ-secretase inhibitor resistant T-cell acute lymphoblastic leukemia. Cell. Signal. 2014, 26, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Liu, J.; Zheng, Y.; You, L.; Kuang, D.; Liu, T. Suppressed expression of long non-coding RNA HOTAIR inhibits proliferation and tumourigenicity of renal carcinoma cells. Tumor Biol. 2014, 35, 11887–11894. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Ren, T.; Huang, Y.; Sun, K.; Wang, S.; Liu, K.; Zheng, B.; Guo, W. Knockdown of long non-coding RNA HOTAIR increases miR-454-3p by targeting Stat3 and Atg12 to inhibit chondrosarcoma growth. Cell Death Dis. 2017, 8, e2605. [Google Scholar] [CrossRef] [PubMed]
- Imai-Sumida, M.; Majid, S.; Dasgupta, P.; Kulkarni, P.; Saini, S.; Bhagirath, D.; Kato, T.; Maekawa, S.; Hashimoto, Y.; Shiina, M. Genistein inhibits renal cancer progression through long non-coding RNA HOTAIR suppression. Cancer Res. 2017, 77, 3449. [Google Scholar]
- Tang, L.; Shen, H.; Li, X.; Li, Z.; Liu, Z.; Xu, J.; Ma, S.; Zhao, X.; Bai, X.; Li, M.; et al. MiR-125a-5p decreases after long non-coding RNA HOTAIR knockdown to promote cancer cell apoptosis by releasing caspase 2. Cell Death Dis. 2016, 7, e2137. [Google Scholar] [CrossRef]
- Musgrove, E.A.; Caldon, C.E.; Barraclough, J.; Stone, A.; Sutherland, R.L. Cyclin D as a therapeutic target in cancer. Nat. Rev. Cancer 2011, 11, 558. [Google Scholar] [CrossRef]
- Wang, Q.; Fan, H.; Liu, Y.; Yin, Z.; Cai, H.; Liu, J.; Wang, Z.; Shao, M.; Sun, X.; Diao, J. Curcumin enhances the radiosensitivity in nasopharyngeal carcinoma cells involving the reversal of differentially expressed long non-coding RNAs. Int. J. Oncol. 2014, 44, 858–864. [Google Scholar] [CrossRef]
- Salviano-Silva, A.; Lobo-Alves, S.C.; Almeida, R.C.; Malheiros, D.; Petzl-Erler, M.L. Besides Pathology: Long Non-Coding RNA in Cell and Tissue Homeostasis. Noncoding RNA 2018, 4, 3. [Google Scholar] [CrossRef]
- Russo, M.; Spagnuolo, C.; Tedesco, I.; Bilotto, S.; Russo, G.L. The flavonoid quercetin in disease prevention and therapy: Facts and fancies. Biochem. Pharmacol. 2012, 83, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Sudan, S.; Rupasinghe, H.V. Quercetin-3-O-glucoside induces human DNA topoisomerase II inhibition, cell cycle arrest and apoptosis in hepatocellular carcinoma cells. Anticancer Res. 2014, 34, 1691–1699. [Google Scholar] [PubMed]
- Zhao, J.-l.; Zhao, J.; Jiao, H.-J. Synergistic growth-suppressive effects of quercetin and cisplatin on HepG2 human hepatocellular carcinoma cells. Appl. Biochem. Biotechnol. 2014, 172, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Mourtada-Maarabouni, M.; Pickard, M.R.; Hedge, V.L.; Farzaneh, F.; Williams, G.T. GAS5, a non-protein-coding RNA, controls apoptosis and is downregulated in breast cancer. Oncogene 2009, 28, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Gandhy, S.U.; Imanirad, P.; Jin, U.H.; Nair, V.; Hedrick, E.; Cheng, Y.; Corton, J.C.; Kim, K.; Safe, S. Specificity protein (Sp) transcription factors and metformin regulate expression of the long non-coding RNA HULC. Oncotarget 2015, 6, 26359–26372. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zheng, H.; Chan, M.T.; Wu, W.K. HULC: An oncogenic long non-coding RNA in human cancer. J. Cell. Mol. Med. 2017, 21, 410–417. [Google Scholar] [CrossRef]
- Bertozzi, D.; Iurlaro, R.; Sordet, O.; Marinello, J.; Zaffaroni, N.; Capranico, G. Characterization of novel antisense HIF-1alpha transcripts in human cancers. Cell Cycle 2011, 10, 3189–3197. [Google Scholar] [CrossRef]
- Bertozzi, D.; Marinello, J.; Manzo, S.G.; Fornari, F.; Gramantieri, L.; Capranico, G. The natural inhibitor of DNA topoisomerase I, camptothecin, modulates HIF-1alpha activity by changing miR expression patterns in human cancer cells. Mol. Cancer Ther. 2014, 13, 239–248. [Google Scholar] [CrossRef]
- Huang, J.; Ke, P.; Guo, L.; Wang, W.; Tan, H.; Liang, Y.; Yao, S. Lentivirus-mediated RNA interference targeting the long noncoding RNA HOTAIR inhibits proliferation and invasion of endometrial carcinoma cells in vitro and in vivo. Int. J. Gynecol. Cancer Off. J. Int. Gynecol. Cancer Soc. 2014, 24, 635–642. [Google Scholar] [CrossRef]
- Chen, J.; Shen, Z.; Zheng, Y.; Wang, S.; Mao, W. Radiotherapy induced Lewis lung cancer cell apoptosis via inactivating beta-catenin mediated by upregulated HOTAIR. Int. J. Clin. Exp. Pathol. 2015, 8, 7878–7886. [Google Scholar]
- Ma, D.D.; Yuan, L.L.; Lin, L.Q. LncRNA HOTAIR contributes to the tumorigenesis of nasopharyngeal carcinoma via up-regulating FASN. Eur. Rev. Med Pharmacol. Sci. 2017, 21, 5143–5152. [Google Scholar] [CrossRef] [PubMed]
- Chiyomaru, T.; Yamamura, S.; Fukuhara, S.; Yoshino, H.; Kinoshita, T.; Majid, S.; Saini, S.; Chang, I.; Tanaka, Y.; Enokida, H.; et al. Genistein inhibits prostate cancer cell growth by targeting miR-34a and oncogenic HOTAIR. PLoS ONE 2013, 8, e70372. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, S.; Jiang, L.; Wang, X.; Song, X. HOTAIR is a promising novel biomarker in patients with thyroid cancer. Exp. Ther. Med. 2017, 13, 2274–2278. [Google Scholar] [CrossRef]
- Geng, Y.J.; Xie, S.L.; Li, Q.; Ma, J.; Wang, G.Y. Large intervening non-coding RNA HOTAIR is associated with hepatocellular carcinoma progression. J. Int. Med Res. 2011, 39, 2119–2128. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.Z.; Li, C.X.; Zhang, Y.; Weng, M.Z.; Zhang, M.D.; Qin, Y.Y.; Gong, W.; Quan, Z.W. Long non-coding RNA HOTAIR, a c-Myc activated driver of malignancy, negatively regulates miRNA-130a in gallbladder cancer. Mol. Cancer 2014, 13, 156. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhou, L.; Wu, L.M.; Lai, M.C.; Xie, H.Y.; Zhang, F.; Zheng, S.S. Overexpression of long non-coding RNA HOTAIR predicts tumor recurrence in hepatocellular carcinoma patients following liver transplantation. Ann. Surg. Oncol. 2011, 18, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wang, C.; Liu, X.; Wu, C.; Yin, H. Long non-coding RNA HOTAIR enhances radioresistance in MDA-MB231 breast cancer cells. Oncol. Lett. 2017, 13, 1143–1148. [Google Scholar] [CrossRef]
- Heubach, J.; Monsior, J.; Deenen, R.; Niegisch, G.; Szarvas, T.; Niedworok, C.; Schulz, W.A.; Hoffmann, M.J. The long noncoding RNA HOTAIR has tissue and cell type-dependent effects on HOX gene expression and phenotype of urothelial cancer cells. Mol. Cancer 2015, 14, 108. [Google Scholar] [CrossRef]
- Biersack, B. Interactions between anticancer active platinum complexes and non-coding RNAs/microRNAs. Non-Coding Rna Res. 2017, 2, 1–17. [Google Scholar] [CrossRef]
- Liu, G.; Xiang, T.; Wu, Q.F.; Wang, W.X. Curcumin suppresses the proliferation of gastric cancer cells by downregulating H19. Oncol. Lett. 2016, 12, 5156–5162. [Google Scholar] [CrossRef]
- Raveh, E.; Matouk, I.J.; Gilon, M.; Hochberg, A. The H19 Long non-coding RNA in cancer initiation, progression and metastasis–a proposed unifying theory. Mol. Cancer 2015, 14, 184. [Google Scholar] [CrossRef] [PubMed]
- Chottanapund, S.; Van Duursen, M.; Navasumrit, P.; Hunsonti, P.; Timtavorn, S.; Ruchirawat, M.; Van den Berg, M. Anti-aromatase effect of resveratrol and melatonin on hormonal positive breast cancer cells co-cultured with breast adipose fibroblasts. Toxicol. Vitr. 2014, 28, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.L.; Chi, Y.Y.; Liu, L.; Huang, N.S.; Wang, L.; Wu, J. LINC00978 predicts poor prognosis in breast cancer patients. Sci. Rep. 2016, 6, 37936. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.M.; Hu, W.W. Long non-coding RNA MALAT1 promotes oral squamous cell carcinoma development via microRNA-125b/STAT3 axis. J. Cell. Physiol. 2018, 233, 3384–3396. [Google Scholar] [CrossRef] [PubMed]
- Ji, Q.; Liu, X.; Fu, X.; Zhang, L.; Sui, H.; Zhou, L.; Sun, J.; Cai, J.; Qin, J.; Ren, J. Resveratrol inhibits invasion and metastasis of colorectal cancer cells via MALAT1 mediated Wnt/β-catenin signal pathway. PLoS ONE 2013, 8, e78700. [Google Scholar] [CrossRef]
- Meehan, M. Inhibition of Proliferation in Canine Osteosarcoma Cell Line 40 by Resveratrol is Correlated with an Increase in the Level of Long Noncoding RNA MALAT1. 2017. Available online: https://dspace.allegheny.edu/handle/10456/42720 (accessed on 3 April 2017).
- Pan, F.; Zhu, L.; Lv, H.; Pei, C. Quercetin promotes the apoptosis of fibroblast-like synoviocytes in rheumatoid arthritis by upregulating lncRNA MALAT1. Int. J. Mol. Med. 2016, 38, 1507–1514. [Google Scholar] [CrossRef]
- Zamani, M.; Sadeghizadeh, M.; Behmanesh, M.; Najafi, F. Dendrosomal curcumin increases expression of the long non-coding RNA gene MEG3 via up-regulation of epi-miRs in hepatocellular cancer. Phytomedicine 2015, 22, 961–967. [Google Scholar] [CrossRef]
- Liu, B.; Pan, C.F.; He, Z.C.; Wang, J.; Wang, P.L.; Ma, T.; Xia, Y.; Chen, Y.J. Long Noncoding RNA-LET Suppresses Tumor Growth and EMT in Lung Adenocarcinoma. Biomed. Res. Int. 2016, 2016, 4693471. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, H.; Li, L.; Zhang, S.; Liu, K.; Liu, Y.; Yang, C. Long noncoding RNA-LET, which is repressed by EZH2, inhibits cell proliferation and induces apoptosis of nasopharyngeal carcinoma cell. Med. Oncol. 2015, 32, 226. [Google Scholar] [CrossRef]
- Yang, F.; Huo, X.S.; Yuan, S.X.; Zhang, L.; Zhou, W.P.; Wang, F.; Sun, S.H. Repression of the long noncoding RNA-LET by histone deacetylase 3 contributes to hypoxia-mediated metastasis. Mol. Cell 2013, 49, 1083–1096. [Google Scholar] [CrossRef]
- Srikantan, V.; Zou, Z.; Petrovics, G.; Xu, L.; Augustus, M.; Davis, L.; Livezey, J.R.; Connell, T.; Sesterhenn, I.A.; Yoshino, K.; et al. PCGEM1, a prostate-specific gene, is overexpressed in prostate cancer. Proc. Natl. Acad. Sci. USA 2000, 97, 12216–12221. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.T.; Huang, J.; Zhou, N.; Zhang, Z.; Koirala, P.; Zhou, X.; Wu, F.; Ding, X.; Mo, Y.Y. Regulation of PCGEM1 by p54/nrb in prostate cancer. Sci. Rep. 2016, 6, 34529. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Toden, S.; Ravindranathan, P.; Han, H.; Goel, A. Curcumin sensitizes pancreatic cancer cells to gemcitabine by attenuating PRC2 subunit EZH2, and the lncRNA PVT1 expression. Carcinogenesis 2017, 38, 1036–1046. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Toden, S.; Weng, W.; Shigeyasu, K.; Han, H.; Becerra, C.; Boland, C.; Goel, A. Su2060 Curcumin Inhibits Polycomb Repressive Complex 2 through lncRNA-PVT1 and Enhances Gemcitabine Sensitivity in Chemoresistant Pancreatic Cancer. Gastroenterology 2016, 150, S624. [Google Scholar] [CrossRef]
- Yu, G.; Yao, W.; Wang, J.; Ma, X.; Xiao, W.; Li, H.; Xia, D.; Yang, Y.; Deng, K.; Xiao, H. LncRNAs expression signatures of renal clear cell carcinoma revealed by microarray. PLoS ONE 2012, 7, e42377. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Liu, G.Z.; Wang, Z. Modulation of androgen receptor-dependent transcription by resveratrol and genistein in prostate cancer cells. Prostate 2004, 59, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Harada, N.; Murata, Y.; Yamaji, R.; Miura, T.; Inui, H.; Nakano, Y. Resveratrol down-regulates the androgen receptor at the post-translational level in prostate cancer cells. J. Nutr. Sci. Vitaminol. 2007, 53, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, S.H.; Zhu, W.; Young, C.Y. Resveratrol inhibits the expression and function of the androgen receptor in LNCaP prostate cancer cells. Cancer Res. 1999, 59, 5892–5895. [Google Scholar] [PubMed]
- Shi, W.-F.; Leong, M.; Cho, E.; Farrell, J.; Chen, H.-C.; Tian, J.; Zhang, D. Repressive effects of resveratrol on androgen receptor transcriptional activity. PLoS ONE 2009, 4, e7398. [Google Scholar] [CrossRef] [PubMed]
- Tomita, S.; Abdalla, M.O.A.; Fujiwara, S.; Matsumori, H.; Maehara, K.; Ohkawa, Y.; Iwase, H.; Saitoh, N.; Nakao, M. A cluster of noncoding RNAs activates the ESR1 locus during breast cancer adaptation. Nat. Commun. 2015, 6, 6966. [Google Scholar] [CrossRef]
- Song, C. Darwinian Selection in Prostate Cancer and Medical Treatment. Clin. Med. 2017, 8, 353–367. [Google Scholar] [CrossRef][Green Version]
- Zook, P.A. Chemopreventive Effects of Pterostilbene in Metastatic Prostate Cancer Cells. Master’s Thesis, Philadelphia College of Osteopathic Medicine, Philadelphia, PA, USA, 2014. [Google Scholar]
- Chen, Z.-H.; Wang, W.-T.; Huang, W.; Fang, K.; Sun, Y.-M.; Liu, S.-R.; Luo, X.-Q.; Chen, Y.-Q. The lncRNA HOTAIRM1 regulates the degradation of PML-RARA oncoprotein and myeloid cell differentiation by enhancing the autophagy pathway. Cell Death Differ. 2017, 24, 212. [Google Scholar] [CrossRef] [PubMed]
- Lorenzen, J.M.; Thum, T. Long noncoding RNAs in kidney and cardiovascular diseases. Nat. Rev. Nephrol. 2016, 12, 360. [Google Scholar] [CrossRef] [PubMed]
- Moerter, C. Phase II study of camptothecin (NSC-100880) in the treatment of advanced gastrointestinal cancer. Cancer Chemother. Rep. 1972, 56, 95–101. [Google Scholar]
- Zeng, C.-W.; Zhang, X.-J.; Lin, K.-Y.; Ye, H.; Feng, S.-Y.; Zhang, H.; Chen, Y.-Q. Camptothecin induces apoptosis in cancer cells via microRNA-125b-mediated mitochondrial pathways. Mol. Pharmacol. 2012, 81, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.J.; Lin, Y.L.; Luh, F.; Yen, Y.; Chen, R.M. Preclinical effects of CRLX101, an investigational camptothecin-containing nanoparticle drug conjugate, on treating glioblastoma multiforme via apoptosis and antiangiogenesis. Oncotarget 2016, 7, 42408–42421. [Google Scholar] [CrossRef] [PubMed]
- Gaur, S.; Wang, Y.; Kretzner, L.; Chen, L.; Yen, T.; Wu, X.; Yuan, Y.C.; Davis, M.; Yen, Y. Pharmacodynamic and pharmacogenomic study of the nanoparticle conjugate of camptothecin CRLX101 for the treatment of cancer. Nanomedicine 2014, 10, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Eliasof, S.; Lazarus, D.; Peters, C.G.; Case, R.I.; Cole, R.O.; Hwang, J.; Schluep, T.; Chao, J.; Lin, J.; Yen, Y.; et al. Correlating preclinical animal studies and human clinical trials of a multifunctional, polymeric nanoparticle. Proc. Natl. Acad. Sci. USA 2013, 110, 15127–15132. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Khin, K.T.; Jensen, G.S.; Liu, A.; Davis, M.E. Synthesis of linear, beta-cyclodextrin-based polymers and their camptothecin conjugates. Bioconjug. Chem. 2003, 14, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Bolton, E.E.; Wang, Y.; Thiessen, P.A.; Bryant, S.H. PubChem: Integrated platform of small molecules and biological activities. In Annual Reports in Computational Chemistry; Elsevier: Amsterdam, The Netherlands, 2008; Volume 4, pp. 217–241. [Google Scholar]
- Esatbeyoglu, T.; Huebbe, P.; Ernst, I.; Chin, D.; Wagner, A.E.; Rimbach, G. Curcumin—From molecule to biological function. Angew. Chem. Int. Ed. 2012, 51, 5308–5332. [Google Scholar] [CrossRef] [PubMed]
- Grynkiewicz, G.; Ślifirski, P. Curcumin and curcuminoids in quest for medicinal status. Acta Biochim. Pol. 2012, 59, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef]
- De Bacco, F.; Luraghi, P.; Medico, E.; Reato, G.; Girolami, F.; Perera, T.; Gabriele, P.; Comoglio, P.M.; Boccaccio, C. Induction of MET by ionizing radiation and its role in radioresistance and invasive growth of cancer. Jnci J. Natl. Cancer Inst. 2011, 103, 645–661. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.-L.; Wang, L.-Y.; Yu, Y.-L.; Chen, H.-W.; Srivastava, S.; Petrovics, G.; Kung, H.-J. A long noncoding RNA connects c-Myc to tumor metabolism. Proc. Natl. Acad. Sci. USA 2014, 111, 18697–18702. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Wu, Y.; Hu, J.; Shan, Y.; Ma, J.; Ma, H.; Qi, X.; Jia, L. Long noncoding RNA HOTAIR promotes renal cell carcinoma malignancy through alpha-2, 8-sialyltransferase 4 by sponging microRNA-124. Cell Prolif. 2018, 51, e12507. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xie, W.; Xie, C.; Huang, C.; Zhu, J.; Liang, Z.; Deng, F.; Zhu, M.; Zhu, W.; Wu, R.; et al. Curcumin modulates miR-19/PTEN/AKT/p53 axis to suppress bisphenol A-induced MCF-7 breast cancer cell proliferation. Phytother. Res. 2014, 28, 1553–1560. [Google Scholar] [CrossRef] [PubMed]
- Carroll, R.E.; Benya, R.V.; Turgeon, D.K.; Vareed, S.; Neuman, M.; Rodriguez, L.; Kakarala, M.; Carpenter, P.M.; McLaren, C.; Meyskens, F.L. Phase IIa clinical trial of curcumin for the prevention of colorectal neoplasia. Cancer Prev. Res. 2011, 4, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, N.; Wolff, R.; Abbruzzese, J.; Hong, D.; Camacho, L.; Li, L.; Braiteh, F.; Kurzrock, R. Phase II clinical trial of curcumin in patients with advanced pancreatic cancer. J. Clin. Oncol. 2006, 24, 14151. [Google Scholar]
- Sharma, R.A.; Euden, S.A.; Platton, S.L.; Cooke, D.N.; Shafayat, A.; Hewitt, H.R.; Marczylo, T.H.; Morgan, B.; Hemingway, D.; Plummer, S.M. Phase I clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clin. Cancer Res. 2004, 10, 6847–6854. [Google Scholar] [CrossRef] [PubMed]
- Chi, H.-C.; Tsai, C.-Y.; Tsai, M.-M.; Yeh, C.-T.; Lin, K.-H. Roles of Long Noncoding RNAs in Recurrence and Metastasis of Radiotherapy-Resistant Cancer Stem Cells. Int. J. Mol. Sci. 2017, 18, 1903. [Google Scholar] [CrossRef] [PubMed]
- Ramya, P.V.S.; Angapelly, S.; Angeli, A.; Digwal, C.S.; Arifuddin, M.; Babu, B.N.; Supuran, C.T.; Kamal, A. Discovery of curcumin inspired sulfonamide derivatives as a new class of carbonic anhydrase isoforms I, II, IX, and XII inhibitors. J. Enzym. Inhib. Med. Chem. 2017, 32, 1274–1281. [Google Scholar] [CrossRef] [PubMed]
- Senturk, M.; Gulcin, I.; Beydemir, S.; Kufrevioglu, O.I.; Supuran, C.T. In Vitro inhibition of human carbonic anhydrase I and II isozymes with natural phenolic compounds. Chem. Biol. Drug Des. 2011, 77, 494–499. [Google Scholar] [CrossRef] [PubMed]
- Karioti, A.; Carta, F.; Supuran, C.T. Phenols and Polyphenols as Carbonic Anhydrase Inhibitors. Molecules 2016, 21, 1649. [Google Scholar] [CrossRef]
- Ahmed, M.; Qadir, M.A.; Hameed, A.; Arshad, M.N.; Asiri, A.M.; Muddassar, M. Sulfonamides containing curcumin scaffold: Synthesis, characterization, carbonic anhydrase inhibition and molecular docking studies. Bioorg. Chem. 2018, 76, 218–227. [Google Scholar] [CrossRef]
- Kim, S.W.; Cha, M.J.; Lee, S.K.; Song, B.W.; Jin, X.; Lee, J.M.; Park, J.H.; Lee, J.D. Curcumin Treatment in Combination with Glucose Restriction Inhibits Intracellular Alkalinization and Tumor Growth in Hepatoma Cells. Int. J. Mol. Sci. 2019, 20, 2375. [Google Scholar] [CrossRef] [PubMed]
- Aygul, I.; Yaylaci Karahalil, F.; Supuran, C.T. Investigation of the inhibitory properties of some phenolic standards and bee products against human carbonic anhydrase I and II. J. Enzym. Inhib. Med. Chem. 2016, 31, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Rakel, D. Integrative Medicine E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Minich, D.M.; Bland, J.S. A review of the clinical efficacy and safety of cruciferous vegetable phytochemicals. Nutr. Rev. 2007, 65, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Licznerska, B.; Baer-Dubowska, W. Indole-3-carbinol and its role in chronic diseases. In Anti-Inflammatory Nutraceuticals and Chronic Diseases; Springer: New York, NY, USA, 2016; pp. 131–154. [Google Scholar]
- Jin, H.; Park, M.H.; Kim, S.M. 3,3′-Diindolylmethane potentiates paclitaxel-induced antitumor effects on gastric cancer cells through the Akt/FOXM1 signaling cascade. Oncol. Rep. 2015, 33, 2031–2036. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Chen, J.; He, B.; Li, Q.; Li, Y.; Gao, Y. A FOXM1 related long non-coding RNA contributes to gastric cancer cell migration. Mol. Cell. Biochem. 2015, 406, 31–41. [Google Scholar] [CrossRef]
- Eldehna, W.M.; Al-Ansary, G.H.; Bua, S.; Nocentini, A.; Gratteri, P.; Altoukhy, A.; Ghabbour, H.; Ahmed, H.Y.; Supuran, C.T. Novel indolin-2-one-based sulfonamides as carbonic anhydrase inhibitors: Synthesis, in vitro biological evaluation against carbonic anhydrases isoforms I, II, IV and VII and molecular docking studies. Eur. J. Med. Chem. 2017, 127, 521–530. [Google Scholar] [CrossRef]
- Li, G.-X.; Chen, Y.-K.; Hou, Z.; Xiao, H.; Jin, H.; Lu, G.; Lee, M.-J.; Liu, B.; Guan, F.; Yang, Z. Pro-oxidative activities and dose–response relationship of (−)-epigallocatechin-3-gallate in the inhibition of lung cancer cell growth: A comparative study in vivo and in vitro. Carcinogenesis 2010, 31, 902–910. [Google Scholar] [CrossRef] [PubMed]
- McLarty, J.; Bigelow, R.L.; Smith, M.; Elmajian, D.; Ankem, M.; Cardelli, J.A. Tea polyphenols decrease serum levels of prostate-specific antigen, hepatocyte growth factor, and vascular endothelial growth factor in prostate cancer patients and inhibit production of hepatocyte growth factor and vascular endothelial growth factor in vitro. Cancer Prev. Res. 2009, 2, 673–682. [Google Scholar]
- Moiseeva, E.P.; Manson, M.M. Dietary chemopreventive phytochemicals: Too little or too much? Cancer Prev. Res. 2009, 2, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, M.; Deguchi, A.; Lim, J.T.; Moriwaki, H.; Kopelovich, L.; Weinstein, I.B. (−)-Epigallocatechin gallate and polyphenon E inhibit growth and activation of the epidermal growth factor receptor and human epidermal growth factor receptor-2 signaling pathways in human colon cancer cells. Clin. Cancer Res. 2005, 11, 2735–2746. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, X.; Lu, G.; Picinich, S.C. Cancer prevention by tea: Animal studies, molecular mechanisms and human relevance. Nat. Rev. Cancer 2009, 9, 429. [Google Scholar] [CrossRef] [PubMed]
- Hajipour, H.; Hamishehkar, H.; Nazari Soltan Ahmad, S.; Barghi, S.; Maroufi, N.F.; Taheri, R.A. Improved anticancer effects of epigallocatechin gallate using RGD-containing nanostructured lipid carriers. Artif. CellsNanomed. Biotechnol. 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kondo, A.; Takeda, T.; Li, B.; Tsuiji, K.; Kitamura, M.; Wong, T.F.; Yaegashi, N. Epigallocatechin-3-gallate potentiates curcumin’s ability to suppress uterine leiomyosarcoma cell growth and induce apoptosis. Int. J. Clin. Oncol. 2013, 18, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Kumazaki, M.; Noguchi, S.; Yasui, Y.; Iwasaki, J.; Shinohara, H.; Yamada, N.; Akao, Y. Anti-cancer effects of naturally occurring compounds through modulation of signal transduction and miRNA expression in human colon cancer cells. J. Nutr. Biochem. 2013, 24, 1849–1858. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.K.; Arora, S.; Averett, C.; Singh, S.; Singh, A.P. Modulation of microRNAs by phytochemicals in cancer: Underlying mechanisms and translational significance. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Yang, S.; Taylor, C.; Sonenshein, G.E. Green tea polyphenol epigallocatechin-3 gallate (EGCG) affects gene expression of breast cancer cells transformed by the carcinogen 7,12-dimethylbenz[a]anthracene. J. Nutr. 2005, 135, 2978S–2986S. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, N.; Cardoso, I.; Domingues, M.R.; Vitorino, R.; Bastos, M.; Bai, G.; Saraiva, M.J.; Almeida, M.R. Binding of epigallocatechin-3-gallate to transthyretin modulates its amyloidogenicity. FEBS Lett. 2009, 583, 3569–3576. [Google Scholar] [CrossRef] [PubMed]
- Chiyomaru, T.; Fukuhara, S.; Saini, S.; Majid, S.; Deng, G.; Shahryari, V.; Chang, I.; Tanaka, Y.; Enokida, H.; Nakagawa, M. Long non-coding RNA HOTAIR is targeted and regulated by miR-141 in human cancer cells. J. Biol. Chem. 2014, 289, 12550–12565. [Google Scholar] [CrossRef] [PubMed]
- Phuah, N.H.; Nagoor, N.H. Regulation of microRNAs by natural agents: New strategies in cancer therapies. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Li, J.; Wang, L. Large intervening non-coding RNA HOTAIR is an indicator of poor prognosis and a therapeutic target in human cancers. Int. J. Mol. Sci. 2014, 15, 18985–18999. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Zhao, J.C.; Kim, J.; Fong, K.-w.; Yang, Y.A.; Chakravarti, D.; Mo, Y.-Y.; Yu, J. LncRNA HOTAIR enhances the androgen-receptor-mediated transcriptional program and drives castration-resistant prostate cancer. Cell Rep. 2015, 13, 209–221. [Google Scholar] [CrossRef]
- Chen, J.; Lin, C.; Yong, W.; Ye, Y.; Huang, Z. Calycosin and genistein induce apoptosis by inactivation of HOTAIR/p-Akt signaling pathway in human breast cancer MCF-7 cells. Cell. Physiol. Biochem. 2015, 35, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Zhou Du, T.F.; Verhaak, R.G.; Su, Z.; Zhang, Y.; Brown, M.; Chen, Y.; Liu, X.S. Integrative genomic analyses reveal clinically relevant long non-coding RNA in human cancer. Nat. Struct. Mol. Biol. 2013, 20, 908. [Google Scholar]
- Banerjee, S.; Li, Y.; Wang, Z.; Sarkar, F.H. Multi-targeted therapy of cancer by genistein. Cancer Lett. 2008, 269, 226–242. [Google Scholar] [CrossRef] [PubMed]
- Caldarelli, A.; Diel, P.; Vollmer, G. Effect of phytoestrogens on gene expression of carbonic anhydrase II in rat uterus and liver. J. Steroid Biochem. Mol. Biol. 2005, 97, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Norrby, M.; Madej, A.; Ekstedt, E.; Holm, L. Effects of genistein on oestrogen and progesterone receptor, proliferative marker Ki-67 and carbonic anhydrase localisation in the uterus and cervix of gilts after insemination. Anim. Reprod. Sci. 2013, 138, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Q.; Wan, H.Y.; Helferich, W.G.; Wong, M.S. Genistein and a soy extract differentially affect three-dimensional bone parameters and bone-specific gene expression in ovariectomized mice. J. Nutr. 2009, 139, 2230–2236. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.-L.; Chen, Y.-G. Natural compounds regulate glycolysis in hypoxic tumor microenvironment. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Pratheeshkumar, P.; Son, Y.-O.; Divya, S.P.; Wang, L.; Turcios, L.; Roy, R.V.; Hitron, J.A.; Kim, D.; Dai, J.; Asha, P. Quercetin inhibits Cr (VI)-induced malignant cell transformation by targeting miR-21-PDCD4 signaling pathway. Oncotarget 2017, 8, 52118. [Google Scholar] [CrossRef] [PubMed]
- Ekinci, D.; Karagoz, L.; Ekinci, D.; Senturk, M.; Supuran, C.T. Carbonic anhydrase inhibitors: In vitro inhibition of alpha isoforms (hCA I, hCA II, bCA III, hCA IV) by flavonoids. J. Enzym. Inhib. Med. Chem. 2013, 28, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Sarikaya, S.B.; Gulcin, I.; Supuran, C.T. Carbonic anhydrase inhibitors: Inhibition of human erythrocyte isozymes I and II with a series of phenolic acids. Chem. Biol. Drug Des. 2010, 75, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Innocenti, A.; Beyza Ozturk Sarikaya, S.; Gulcin, I.; Supuran, C.T. Carbonic anhydrase inhibitors. Inhibition of mammalian isoforms I–XIV with a series of natural product polyphenols and phenolic acids. Bioorg. Med. Chem. 2010, 18, 2159–2164. [Google Scholar] [CrossRef]
- Ma, T.; Liu, Y.; Wu, Q.; Luo, L.; Cui, Y.; Wang, X.; Chen, X.; Tan, L.; Meng, X. Quercetin-Modified Metal-Organic Frameworks for Dual Sensitization of Radiotherapy in Tumor Tissues by Inhibiting the Carbonic Anhydrase IX. ACS Nano 2019, 13, 4209–4219. [Google Scholar] [CrossRef]
- Shrikanta, A.; Kumar, A.; Govindaswamy, V. Resveratrol content and antioxidant properties of underutilized fruits. J. Food Sci. Technol. 2015, 52, 383–390. [Google Scholar] [CrossRef]
- Sinha, D.; Sarkar, N.; Biswas, J.; Bishayee, A. Resveratrol for breast cancer prevention and therapy: Preclinical evidence and molecular mechanisms. Semin. Cancer Biol. 2016, 40–41, 209–232. [Google Scholar] [CrossRef]
- Al Aameri, R.F.H.; Sheth, S.; Alanisi, E.M.A.; Borse, V.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Tonic suppression of PCAT29 by the IL-6 signaling pathway in prostate cancer: Reversal by resveratrol. PLoS ONE 2017, 12, e0177198. [Google Scholar] [CrossRef]
- Li, Y.T.; Tian, X.T.; Wu, M.L.; Zheng, X.; Kong, Q.Y.; Cheng, X.X.; Zhu, G.W.; Liu, J.; Li, H. Resveratrol Suppresses the Growth and Enhances Retinoic Acid Sensitivity of Anaplastic Thyroid Cancer Cells. Int. J. Mol. Sci. 2018, 19, 1030. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Ma, X.; Song, Y.; Zhang, Y.; Xiong, W.; Li, L.; Zhou, L. Anti-colorectal cancer targets of resveratrol and biological molecular mechanism: Analyses of network pharmacology, human and experimental data. J. Cell. Biochem. 2019. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wu, X.; Lv, J.; Sun, H.; Zhou, F. Resveratrol induces p53 in colorectal cancer through SET7/9. Oncol. Lett. 2019, 17, 3783–3789. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Wang, Y.; Zhu, J.; Orloff, M.; Eng, C. Resveratrol enhances the anti-tumor activity of the mTOR inhibitor rapamycin in multiple breast cancer cell lines mainly by suppressing rapamycin-induced AKT signaling. Cancer Lett. 2011, 301, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Liu, Y.; Teng, M.; Jiao, K.; Zhen, J.; Wu, M.; Li, Z. Resveratrol inhibits the proliferation of estrogen receptor-positive breast cancer cells by suppressing EZH2 through the modulation of ERK1/2 signaling. Cell Biol. Toxicol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; He, B.; Lin, L.; Malhotra, A.; Yuan, N. Potential of curcumin and resveratrol as biochemical and biophysical modulators during lung cancer in rats. Drug Chem. Toxicol. 2019, 42, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Monteillier, A.; Voisin, A.; Furrer, P.; Allemann, E.; Cuendet, M. Intranasal administration of resveratrol successfully prevents lung cancer in A/J mice. Sci. Rep. 2018, 8, 14257. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Yang, X.; Li, H.; Chong, T. MP39-13 resveratrol sensitizes bladder cancer cells to trail-induced apoptosis. J. Urol. 2014, 191, e430–e431. [Google Scholar] [CrossRef]
- Innocenti, A.; Gulcin, I.; Scozzafava, A.; Supuran, C.T. Carbonic anhydrase inhibitors. Antioxidant polyphenols effectively inhibit mammalian isoforms I-XV. Bioorg. Med. Chem. Lett. 2010, 20, 5050–5053. [Google Scholar] [CrossRef]
- Cheetham, S.; Gruhl, F.; Mattick, J.; Dinger, M. Long noncoding RNAs and the genetics of cancer. Br. J. Cancer 2013, 108, 2419. [Google Scholar] [CrossRef]
- Prensner, J.R.; Chinnaiyan, A.M. The emergence of lncRNAs in cancer biology. Cancer Discov. 2011, 1, 391–407. [Google Scholar] [CrossRef] [PubMed]
- Budisan, L.; Gulei, D.; Zanoaga, O.M.; Irimie, A.I.; Sergiu, C.; Braicu, C.; Gherman, C.D.; Berindan-Neagoe, I. Dietary Intervention by Phytochemicals and Their Role in Modulating Coding and Non-Coding Genes in Cancer. Int. J. Mol. Sci. 2017, 18, 1178. [Google Scholar] [CrossRef] [PubMed]
- Debnath, T.; Deb Nath, N.C.; Kim, E.K.; Lee, K.G. Role of phytochemicals in the modulation of miRNA expression in cancer. Food Funct. 2017, 8, 3432–3442. [Google Scholar] [CrossRef] [PubMed]
- Darnell, J.E., Jr. STAT3, HIF-1, glucose addiction and Warburg effect. Aging (Albany Ny) 2010, 2, 890–891. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Demaria, M.; Giorgi, C.; Lebiedzinska, M.; Esposito, G.; D’Angeli, L.; Bartoli, A.; Gough, D.J.; Turkson, J.; Levy, D.E.; Watson, C.J. A STAT3-mediated metabolic switch is involved in tumour transformation and STAT3 addiction. Aging (Albany Ny) 2010, 2, 823–842. [Google Scholar] [CrossRef] [PubMed]
- Novak Kujundžić, R.; Grbeša, I.; Ivkić, M.; Katdare, M.; Gall-Trošelj, K. Curcumin downregulates H19 gene transcription in tumor cells. J. Cell. Biochem. 2008, 104, 1781–1792. [Google Scholar] [CrossRef] [PubMed]
- Celton-Morizur, S.; Merlen, G.; Couton, D.; Margall-Ducos, G.; Desdouets, C. The insulin/Akt pathway controls a specific cell division program that leads to generation of binucleated tetraploid liver cells in rodents. J. Clin. Investig. 2009, 119, 1880–1887. [Google Scholar] [CrossRef]
- Shoshani, O.; Zipori, D.; Shani, N. The tissue specific nature of mesenchymal stem/stromal cells: Gaining better understanding for improved clinical outcomes. Rna Dis. 2015, 2. [Google Scholar]
- Wang, G.; Lunardi, A.; Zhang, J.; Chen, Z.; Ala, U.; Webster, K.A.; Tay, Y.; Gonzalez-Billalabeitia, E.; Egia, A.; Shaffer, D.R. Zbtb7a suppresses prostate cancer through repression of a Sox9-dependent pathway for cellular senescence bypass and tumor invasion. Nat. Genet. 2013, 45, 739. [Google Scholar] [CrossRef]
- Jiang, P.; Wu, X.; Wang, X.; Huang, W.; Feng, Q. NEAT1 upregulates EGCG-induced CTR1 to enhance cisplatin sensitivity in lung cancer cells. Oncotarget 2016, 7, 43337. [Google Scholar] [CrossRef]
- Kalayda, G.V.; Wagner, C.H.; Jaehde, U. Relevance of copper transporter 1 for cisplatin resistance in human ovarian carcinoma cells. J. Inorg. Biochem. 2012, 116, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Larson, C.A.; Blair, B.G.; Safaei, R.; Howell, S.B. The role of the mammalian copper transporter 1 in the cellular accumulation of platinum-based drugs. Mol. Pharmacol. 2009, 75, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-Y.; Larson, C.A.; Safaei, R.; Howell, S.B. Molecular modulation of the copper and cisplatin transport function of CTR1 and its interaction with IRS-4. Biochem. Pharmacol. 2014, 90, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.-H.; Abdelmohsen, K.; Kim, J.; Yang, X.; Martindale, J.L.; Tominaga-Yamanaka, K.; White, E.J.; Orjalo, A.V.; Rinn, J.L.; Kreft, S.G. Scaffold function of long non-coding RNA HOTAIR in protein ubiquitination. Nat. Commun. 2013, 4, 2939. [Google Scholar] [CrossRef] [PubMed]
- Han, P.; Chang, C.-P. Long non-coding RNA and chromatin remodeling. Rna Biol. 2015, 12, 1094–1098. [Google Scholar] [CrossRef]
- Onder, T.T.; Kara, N.; Cherry, A.; Sinha, A.U.; Zhu, N.; Bernt, K.M.; Cahan, P.; Marcarci, B.O.; Unternaehrer, J.; Gupta, P.B.; et al. Chromatin-modifying enzymes as modulators of reprogramming. Nature 2012, 483, 598–602. [Google Scholar] [CrossRef]
- Beckedorff, F.C.; Ayupe, A.C.; Crocci-Souza, R.; Amaral, M.S.; Nakaya, H.I.; Soltys, D.T.; Menck, C.F.; Reis, E.M.; Verjovski-Almeida, S. The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation. PLoS Genet. 2013, 9, e1003705. [Google Scholar] [CrossRef]
- Chung, S.; Nakagawa, H.; Uemura, M.; Piao, L.; Ashikawa, K.; Hosono, N.; Takata, R.; Akamatsu, S.; Kawaguchi, T.; Morizono, T. Association of a novel long non-coding RNA in 8q24 with prostate cancer susceptibility. Cancer Sci. 2011, 102, 245–252. [Google Scholar] [CrossRef]
- Cui, Z.; Ren, S.; Lu, J.; Wang, F.; Xu, W.; Sun, Y.; Wei, M.; Chen, J.; Gao, X.; Xu, C. The prostate cancer-up-regulated long noncoding RNA PlncRNA-1 modulates apoptosis and proliferation through reciprocal regulation of androgen receptor. Urol. Oncol. 2003, 31, 1117–1123. [Google Scholar] [CrossRef]
- Malik, R.; Patel, L.; Prensner, J.R.; Shi, Y.; Iyer, M.K.; Subramaniyan, S.; Carley, A.; Niknafs, Y.S.; Sahu, A.; Han, S. The lncRNA PCAT29 inhibits oncogenic phenotypes in prostate cancer. Mol. Cancer Res. 2014, 12, 1081–1087. [Google Scholar] [CrossRef]
- Poliseno, L.; Salmena, L.; Riccardi, L.; Fornari, A.; Song, M.S.; Hobbs, R.M.; Sportoletti, P.; Varmeh, S.; Egia, A.; Fedele, G. Identification of the miR-106b~25 microRNA cluster as a proto-oncogenic PTEN-targeting intron that cooperates with its host gene MCM7 in transformation. Sci. Signal. 2010, 3, ra29. [Google Scholar] [CrossRef] [PubMed]
- Prensner, J.R.; Iyer, M.K.; Balbin, O.A.; Dhanasekaran, S.M.; Cao, Q.; Brenner, J.C.; Laxman, B.; Asangani, I.A.; Grasso, C.S.; Kominsky, H.D. Transcriptome sequencing across a prostate cancer cohort identifies PCAT-1, an unannotated lincRNA implicated in disease progression. Nat. Biotechnol. 2011, 29, 742. [Google Scholar] [CrossRef] [PubMed]
- Takayama, K.i.; Horie-Inoue, K.; Katayama, S.; Suzuki, T.; Tsutsumi, S.; Ikeda, K.; Urano, T.; Fujimura, T.; Takagi, K.; Takahashi, S. Androgen-responsive long noncoding RNA CTBP1-AS promotes prostate cancer. EMBO J. 2013, 32, 1665–1680. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Han, S.; Jin, G.; Zhou, X.; Li, M.; Ying, X.; Wang, L.; Wu, H.; Zhu, Q. Linc00963: A novel, long non-coding RNA involved in the transition of prostate cancer from androgen-dependence to androgen-independence. Int. J. Oncol. 2014, 44, 2041–2049. [Google Scholar] [CrossRef] [PubMed]
- Yap, K.L.; Li, S.; Muñoz-Cabello, A.M.; Raguz, S.; Zeng, L.; Mujtaba, S.; Gil, J.; Walsh, M.J.; Zhou, M.-M. Molecular interplay of the noncoding RNA ANRIL and methylated histone H3 lysine 27 by polycomb CBX7 in transcriptional silencing of INK4a. Mol. Cell 2010, 38, 662–674. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, P.; Wang, L.; Piao, H.-l.; Ma, L. Long non-coding RNA HOTAIR in carcinogenesis and metastasis. Acta Biochim. Biophys. Sin. 2013, 46, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Eckert, R.L. Keratinocyte proliferation, differentiation, and apoptosis—Differential mechanisms of regulation by curcumin, EGCG and apigenin. Toxicol. Appl. Pharmacol. 2007, 224, 214–219. [Google Scholar] [CrossRef]
- Moon, R.C.; Thompson, H.J.; Becci, P.J.; Grubbs, C.J.; Gander, R.J.; Newton, D.L.; Smith, J.M.; Phillips, S.L.; Henderson, W.R.; Mullen, L.T.; et al. N-(4-Hydroxyphenyl)retinamide, a new retinoid for prevention of breast cancer in the rat. Cancer Res. 1979, 39, 1339–1346. [Google Scholar]
- Jiang, S.; Wang, H.L.; Yang, J. Low expression of long non-coding RNA LET inhibits carcinogenesis of cervical cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 806–811. [Google Scholar]
- Serviss, J.T.; Johnsson, P.; Grander, D. An emerging role for long non-coding RNAs in cancer metastasis. Front. Genet. 2014, 5, 234. [Google Scholar] [CrossRef]
- Shih, J.W.; Chiang, W.F.; Wu, A.T.H.; Wu, M.H.; Wang, L.Y.; Yu, Y.L.; Hung, Y.W.; Wang, W.C.; Chu, C.Y.; Hung, C.L.; et al. Long noncoding RNA LncHIFCAR/MIR31HG is a HIF-1alpha co-activator driving oral cancer progression. Nat. Commun. 2017, 8, 15874. [Google Scholar] [CrossRef] [PubMed]
- Dinh, T.A.; Vitucci, E.C.; Wauthier, E.; Graham, R.P.; Pitman, W.A.; Oikawa, T.; Chen, M.; Silva, G.O.; Greene, K.G.; Torbenson, M.S.; et al. Comprehensive analysis of The Cancer Genome Atlas reveals a unique gene and non-coding RNA signature of fibrolamellar carcinoma. Sci. Rep. 2017, 7, 44653. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, N.; Zhou, X.; Bian, X.; Qiu, G.; Zhang, M.; Lin, H.; Li, D. LncRNA and mRNA interaction study based on transcriptome profiles reveals potential core genes in the pathogenesis of human thoracic aortic dissection. Mol. Med. Rep. 2018, 18, 3167–3176. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Verma, S.S.; Rai, V.; Awasthee, N.; Chava, S.; Hui, K.M.; Kumar, A.P.; Challagundla, K.B.; Sethi, G.; Gupta, S.C. Long non-coding RNAs are emerging targets of phytochemicals for cancer and other chronic diseases. Cell Mol. Life Sci. 2019, 76, 1947–1966. [Google Scholar] [CrossRef]
- McCubrey, J.A.; Lertpiriyapong, K.; Steelman, L.S.; Abrams, S.L.; Yang, L.V.; Murata, R.M.; Rosalen, P.L.; Scalisi, A.; Neri, L.M.; Cocco, L.; et al. Effects of resveratrol, curcumin, berberine and other nutraceuticals on aging, cancer development, cancer stem cells and microRNAs. Aging 2017, 9, 1477–1536. [Google Scholar] [CrossRef] [PubMed]
- Kusuzaki, K.; Matsubara, T.; Murata, H.; Logozzi, M.; Iessi, E.; Di Raimo, R.; Carta, F.; Supuran, C.T.; Fais, S. Natural extracellular nanovesicles and photodynamic molecules: Is there a future for drug delivery? J. Enzym. Inhib. Med. Chem. 2017, 32, 908–916. [Google Scholar] [CrossRef]
- Iessi, E.; Logozzi, M.; Lugini, L.; Azzarito, T.; Federici, C.; Spugnini, E.P.; Mizzoni, D.; Di Raimo, R.; Angelini, D.F.; Battistini, L.; et al. Acridine Orange/exosomes increase the delivery and the effectiveness of Acridine Orange in human melanoma cells: A new prototype for theranostics of tumors. J. Enzym. Inhib. Med. Chem. 2017, 32, 648–657. [Google Scholar] [CrossRef]
- Bragagni, M.; Carta, F.; Osman, S.M.; AlOthman, Z.; Supuran, C.T. Synthesis of an acridine orange sulfonamide derivative with potent carbonic anhydrase IX inhibitory action. J. Enzym. Inhib. Med. Chem. 2017, 32, 701–706. [Google Scholar] [CrossRef]
- Edgar, J.R. Q&A: What are exosomes, exactly? BMC Biol. 2016, 14, 46. [Google Scholar] [CrossRef]
- Logozzi, M.; Capasso, C.; Di Raimo, R.; Del Prete, S.; Mizzoni, D.; Falchi, M.; Supuran, C.T.; Fais, S. Prostate cancer cells and exosomes in acidic condition show increased carbonic anhydrase IX expression and activity. J. Enzym. Inhib. Med. Chem. 2019, 34, 272–278. [Google Scholar] [CrossRef]
- Gillies, R.J.; Pilot, C.; Marunaka, Y.; Fais, S. Targeting acidity in cancer and diabetes. Biochim. Biophys. Acta Rev. Cancer 2019, 1871, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Spugnini, E.P.; Sonveaux, P.; Stock, C.; Perez-Sayans, M.; De Milito, A.; Avnet, S.; Garcia, A.G.; Harguindey, S.; Fais, S. Proton channels and exchangers in cancer. Biochim. Biophys. Acta 2015, 1848, 2715–2726. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.; Spugnini, E.P.; Assaraf, Y.G.; Azzarito, T.; Rauch, C.; Fais, S. Microenvironment acidity as a major determinant of tumor chemoresistance: Proton pump inhibitors (PPIs) as a novel therapeutic approach. Drug Resist. Updat. 2015, 23, 69–78. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).