Insight into the Antifungal Mechanism of Action of Human RNase N-terminus Derived Peptides
Abstract
:1. Introduction
2. Results
2.1. Peptides Design and Physicochemical Characterization
2.2. Antifungal Activity against Planktonic Cells of hRNases Is Retained at the N-terminus
2.3. The Antifungal Mechanism of hRNase-Derived Peptides Relies on Membrane and Cell Wall Interactions
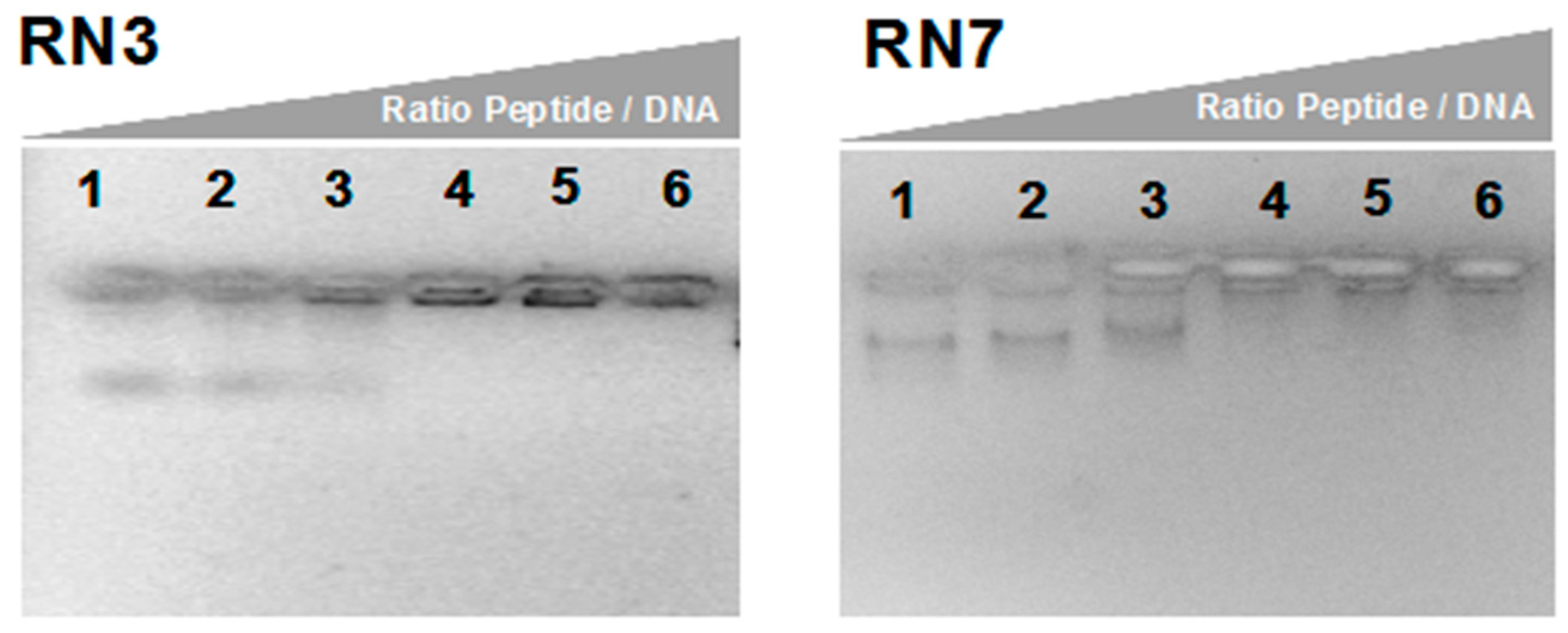
2.4. Nucleic Acids Binding of RN3 and RN7 the Potent Anticandidal Peptides
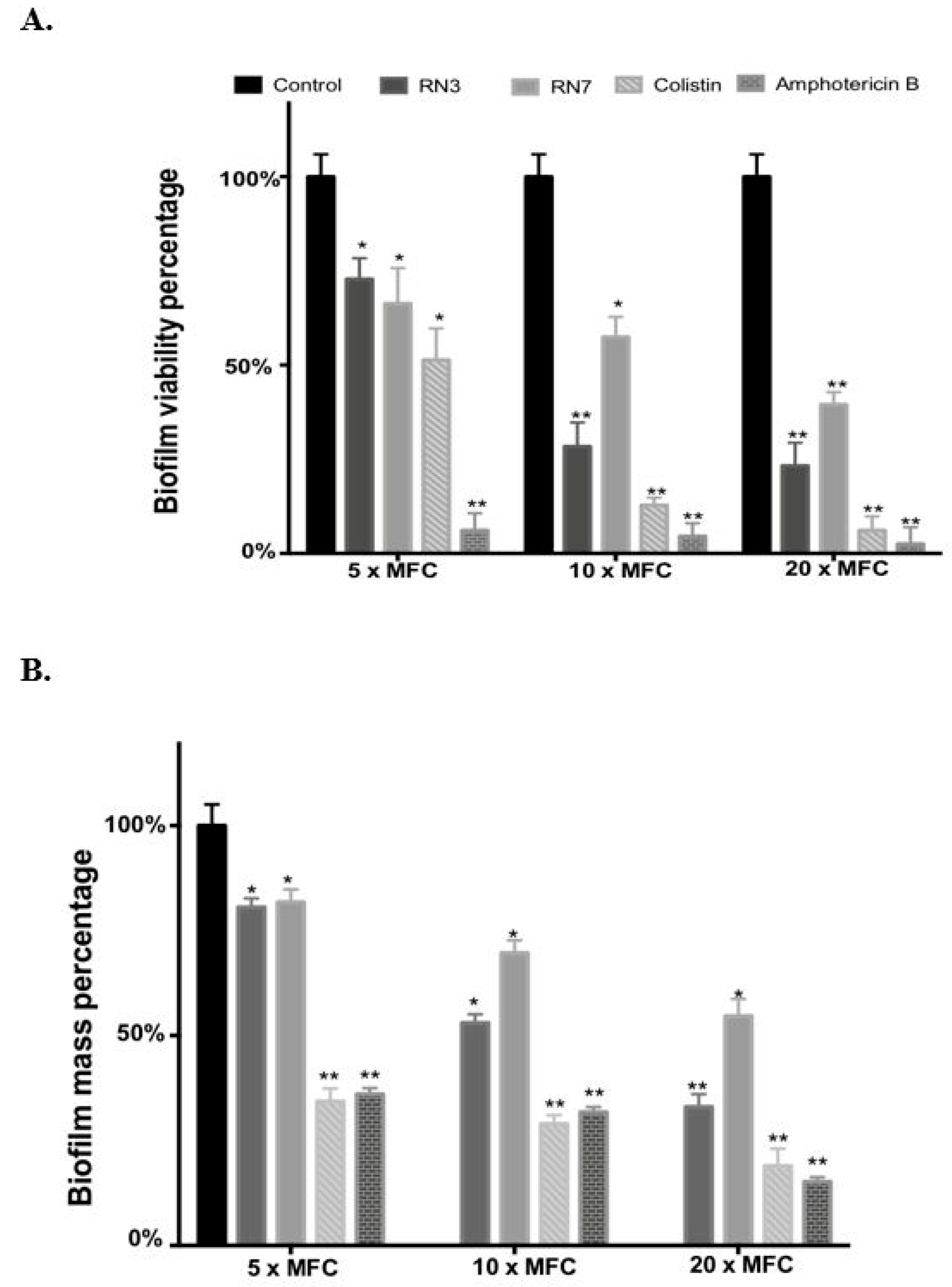
2.5. Evaluation of the Activities of RN3 and RN7 Peptides against C. albicans Biofilms
2.6. Modulation of the Gene Expression Profile Related to Cell Wall Synthesis and Biofilm Formation in Planktonic Cells
3. Discussion
4. Materials and Methods
4.1. Peptide Synthesis
4.2. C. albicans Growth Conditions
4.3. Minimum Fungicidal Concentration
4.4. Minimal Agglutination Activity (MAC)
4.5. Cell Viability Assay
4.6. Cell Survival Assay
4.7. Cell Membrane Depolarization Assay
4.8. Cell Membrane Permeabilization Activity
4.9. Cell Binding Assay
4.10. DNA Binding Assay
4.11. Gene Expression Analysis
4.12. Biofilm Growth
4.13. Biofilm Viability Test
4.14. Biofilm Mass Eradication
4.15. Confocal Microscopy
4.16. Cytotoxicity Assay
4.17. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CFU | Colony forming units |
| EDTA | Ethylenediaminetetraacetic acid |
| SDS-PAGE | Sodium dodecyl sulfate polyacrylamide gel electrophoresis |
| ATP | Adenosine triphosphate |
| DiSC3 | 3,3′-Dipropylthiadicarbocyanine Iodide |
| IC | Inhibition Concentration |
| MFC | Minimum Fungicidal Concentration |
| MAC | Minimum Agglutination Concentration |
| PI | Propidium iodide |
References
- Jabra-Rizk, M.A.; Kong, E.F.; Tsui, C.; Nguyen, M.H.; Clancy, C.J.; Fidel, P.L.; Noverr, M.; Noverr, M. Candida albicans Pathogenesis: Fitting within the Host-Microbe Damage Response Framework. Infect. Immun. 2016, 84, 2724–2739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gulati, M.; Nobile, C.J. Candida albicans biofilms: development, regulation, and molecular mechanisms. Microbes Infect. 2016, 18, 310–321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa-Orlandi, C.; Sardi, J.; Pitangui, N.; de Oliveira, H.; Scorzoni, L.; Galeane, M.; Medina-Alarcón, K.; Melo, W.; Marcelino, M.; Braz, J.; et al. Fungal Biofilms and Polymicrobial Diseases. J. Fungi 2017, 3, 22. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.J.; Lee, M.W.; Choi, J.S.; Lee, S.G.; Park, J.Y.; Kim, S.W. Inhibitory activity of hinokitiol against biofilm formation in fluconazole-resistant Candida species. PLoS ONE 2017, 12, e0171244. [Google Scholar] [CrossRef] [PubMed]
- Nobile, C.J.; Johnsonb, A.D. Candida albicans biofilms and human disease. Annu. Rev. Microbiol. 2015, 69, 71–92. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Zasloff, M. Antimicrobial peptides in health and disease. N. Engl. J. Med. 2002, 347, 1199–1200. [Google Scholar] [CrossRef] [PubMed]
- Gordon, Y.J.; Romanowski, E.G.; McDermott, A.M. A review of antimicrobial peptides and their therapeutic potential as anti-infective drugs. Curr. Eye Res. 2005, 30, 505–515. [Google Scholar] [CrossRef]
- Wang, G.; Li, X.; Wang, Z. APD2: The updated antimicrobial peptide database and its application in peptide design. Nucleic Acids Res. 2009, 37, D933-7. [Google Scholar] [CrossRef]
- Boman, H.G. Antibacterial peptides: Basic facts and emerging concepts. J. Intern. Med. 2003, 254, 197–215. [Google Scholar] [CrossRef]
- Fosgerau, K.; Hoffmann, T. Peptide therapeutics: Current status and future directions. Drug Discov. Today 2015, 20, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Sierra, J.M.; Fusté, E.; Rabanal, F.; Vinuesa, T.; Viñas, M. An overview of antimicrobial peptides and the latest advances in their development. Expert Opin. Biol. Ther. 2017, 17, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Mahlapuu, M.; Håkansson, J.; Ringstad, L.; Björn, C. Antimicrobial Peptides: An Emerging Category of Therapeutic Agents. Front. Cell. Infect. Microbiol. 2016, 6, 194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batoni, G.; Maisetta, G.; Lisa Brancatisano, F.; Esin, S.; Campa, M. Use of Antimicrobial Peptides Against Microbial Biofilms: Advantages and Limits. Curr. Med. Chem. 2011, 18, 256–279. [Google Scholar] [CrossRef] [PubMed]
- Lázár, V.; Martins, A.; Spohn, R.; Daruka, L.; Grézal, G.; Fekete, G.; Számel, M.; Jangir, P.K.; Kintses, B.; Csörgő, B.; et al. Antibiotic-resistant bacteria show widespread collateral sensitivity to antimicrobial peptides. Nat. Microbiol. 2018, 3, 718–731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bondaryk, M.; Staniszewska, M.; Zielińska, P.; Urbańczyk-Lipkowska, Z. Natural Antimicrobial Peptides as Inspiration for Design of a New Generation Antifungal Compounds. J. Fungi 2017, 3, 46. [Google Scholar] [CrossRef] [PubMed]
- Swidergall, M.; Ernst, J.F. Interplay between Candida albicans and the antimicrobial peptide armory. Eukaryot. Cell 2014, 13, 950–957. [Google Scholar] [CrossRef]
- Boix, E.; Nogués, M.V. Mammalian antimicrobial proteins and peptides: overview on the RNase A superfamily members involved in innate host defence. Mol. Biosyst. 2007, 3, 317. [Google Scholar] [CrossRef]
- Pulido, D.; Prats-Ejarque, G.; Villalba, C.; Albacar, M.; González-López, J.J.; Torrent, M.; Moussaoui, M.; Boix, E. A novel RNase 3/ECP peptide for Pseudomonas aeruginosa biofilm eradication that combines antimicrobial, lipopolysaccharide binding, and cell-agglutinating activities. Antimicrob. Agents Chemother. 2016, 60, 6313–6325. [Google Scholar] [CrossRef]
- Pulido, D.; Prats-Ejarque, G.; Villalba, C.; Albacar, M.; Moussaoui, M.; Andreu, D.; Volkmer, R.; Torrent, M.; Boix, E. Positional scanning library applied to the human eosinophil cationic protein/RNase3 N-terminus reveals novel and potent anti-biofilm peptides. Eur. J. Med. Chem. 2018, 152, 590–599. [Google Scholar] [CrossRef]
- Salazar, V.A.; Arranz-Trullén, J.; Navarro, S.; Blanco, J.A.; Sánchez, D.; Moussaoui, M.; Boix, E. Exploring the mechanisms of action of human secretory RNase 3 and RNase 7 against Candida albicans. Microbiologyopen 2016, 5, 830–845. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Li, J.; Moussaoui, M.; Boix, E. Immune modulation by human secreted RNases at the extracellular space. Front. Immunol. 2018, 9, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Torrent, M.; Pulido, D.; Valle, J.; Nogués, M.V.V.; Andreu, D.; Boix, E. Ribonucleases as a host-defence family: Evidence of evolutionarily conserved antimicrobial activity at the N-terminus. Biochem. J. 2013, 456, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Torrent, M.; de la Torre, B.G.; Nogués, V.M.; Andreu, D.; Boix, E. Bactericidal and membrane disruption activities of the eosinophil cationic protein are largely retained in an N-terminal fragment. Biochem. J. 2009, 421, 425–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torrent, M.; Pulido, D.; de la Torre, B.G.; Garcia-Mayoral, M.F.; Nogues, M.V.; Bruix, M.; Andreu, D.; Boix, E. Refining the eosinophil cationic protein antibacterial pharmacophore by rational structure minimization. J. Med. Chem. 2011, 54, 5237–5244. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, N.; Mishra, A.; Lai, G.H.; Wong, G.C.L. Arginine-rich cell-penetrating peptides. FEBS Lett. 2010, 584, 1806–1813. [Google Scholar] [CrossRef] [PubMed]
- Kerkis, A.; Hayashi, M.A.F.; Yamane, T.; Kerkis, I. Properties of cell penetrating peptides (CPPs). IUBMB Life 2006, 58, 7–13. [Google Scholar] [CrossRef]
- Wang, G.; Li, X.; Wang, Z. APD3: the antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 2016, 44. [Google Scholar] [CrossRef]
- Yin, L.M.; Edwards, M.A.; Li, J.; Yip, C.M.; Deber, C.M. Roles of hydrophobicity and charge distribution of cationic antimicrobial peptides in peptide-membrane interactions. J. Biol. Chem. 2012, 287, 7738–7745. [Google Scholar] [CrossRef]
- Boix, E.; Salazar, V.A.; Torrent, M.; Pulido, D.; Nogués, M.V.; Moussaoui, M. Structural determinants of the eosinophil cationic protein antimicrobial activity. Biol. Chem. 2012, 393, 801–815. [Google Scholar] [CrossRef]
- Torrent, M.; Sánchez, D.; Buzón, V.; Nogués, M.V.; Cladera, J.; Boix, E. Comparison of the membrane interaction mechanism of two antimicrobial RNases: RNase 3/ECP and RNase 7. Biochim. Biophys. Acta Biomembr. 2009, 1788, 1116–1125. [Google Scholar] [CrossRef] [Green Version]
- Torrent, M.; Badia, M.; Moussaoui, M.; Sanchez, D.; Nogués, M.V.; Boix, E. Comparison of human RNase 3 and RNase 7 bactericidal action at the Gram-negative and Gram-positive bacterial cell wall. FEBS J. 2010, 277, 1713–1725. [Google Scholar] [CrossRef] [Green Version]
- Torrent, M.; Navarro, S.; Moussaoui, M.; Nogues, M.V.; Boix, E. Eosinophil cationic protein high-affinity binding to bacteria-wall lipopolysaccharides and peptidoglycans. Biochemistry 2008, 47, 3544–3555. [Google Scholar] [CrossRef]
- García-Mayoral, M.F.; Moussaoui, M.; de la Torre, B.G.; Andreu, D.; Boix, E.; Nogués, M.V.; Rico, M.; Laurents, D.V.; Bruix, M. NMR structural determinants of eosinophil cationic protein binding to membrane and heparin mimetics. Biophys. J. 2010, 98, 2702–2711. [Google Scholar] [CrossRef]
- García-Mayoral, M.F.; Canales, Á.; Díaz, D.; López-Prados, J.; Moussaoui, M.; De Paz, J.L.; Angulo, J.; Nieto, P.M.; Jiménez-Barbero, J.; Boix, E.; et al. Insights into the glycosaminoglycan-mediated cytotoxic mechanism of eosinophil cationic protein revealed by NMR. ACS Chem. Biol. 2013, 8, 144–151. [Google Scholar] [CrossRef]
- Torrent, M.; Nogués, M.V.; Boix, E. Eosinophil cationic protein (ECP) can bind heparin and other glycosaminoglycans through its RNase active site. J. Mol. Recognit. 2011, 24, 90–100. [Google Scholar] [CrossRef]
- Avci, F.G. Membrane Active Peptides and Their Biophysical Characterization. Biomolecules 2018, 2017, 1–43. [Google Scholar] [CrossRef]
- Torrent, M.; Cuyás, E.; Carreras, E.; Navarro, S.; López, O.; De La Maza, A.; Nogués, M.V.; Reshetnyak, Y.K.; Boix, E. Topography studies on the membrane interaction mechanism of the eosinophil cationic protein. Biochemistry 2007, 46, 720–733. [Google Scholar] [CrossRef]
- Gow, N.A.R.; Van De Veerdonk, F.L.; Brown, A.J.P.; Netea, M.G.; Van De Veerdonk, F.L.; Brown, A.J.P.; Netea, M.G. Candida albicans morphogenesis and host defence:discriminating invasion from colonization. Nat. Rev. Microbiol. 2013, 10, 112–122. [Google Scholar] [CrossRef]
- Pierce, C.G.; Vila, T.; Romo, J.A.; Montelongo-jauregui, D.; Wall, G.; Ramasubramanian, A.; Lopez-ribot, J.L. The Candida albicans Biofilm Matrix: Composition, Structure and Function. J. Fungi 2017, 3, 14. [Google Scholar] [CrossRef]
- Borghi, E.; Borgo, F.; Morace, G. Fungal Biofilms: Update on Resistance. In Advances in Experimental Medicine and Biology; Springer Nature: New York, NY, USA, 2016; Volume 931, pp. 37–47. ISBN 22148019. [Google Scholar]
- Cavalheiro, M.; Teixeira, M.C. Candida Biofilms: Threats, Challenges, and Promising Strategies. Front. Med. 2018, 5, 28. [Google Scholar] [CrossRef] [Green Version]
- Lum, K.Y.; Tay, S.T.; Le, C.F.; Lee, V.S.; Sabri, N.H.; Velayuthan, R.D.; Hassan, H.; Sekaran, S.D. Activity of novel synthetic peptides against Candida albicans. Sci. Rep. 2015, 5, 9657. [Google Scholar] [CrossRef]
- Teixeira-Santos, R.; Ricardo, E.; Branco, R.J.; Azevedo, M.M.; Rodrigues, A.G.; Pina-Vaz, C. Unveiling the synergistic interaction between liposomal amphotericin B and colistin. Front. Microbiol. 2016, 7, 1439. [Google Scholar] [CrossRef]
- Maiolo, E.M.; Tafin, U.F.; Borens, O.; Trampuz, A. Activities of fluconazole, caspofungin, anidulafungin, and amphotericin b on planktonic and biofilm candida species determined by microcalorimetry. Antimicrob. Agents Chemother. 2014, 58, 2709–2717. [Google Scholar] [CrossRef]
- Hamill, R.J. Amphotericin B formulations: A comparative review of efficacy and toxicity. Drugs 2013, 73, 919–934. [Google Scholar] [CrossRef]
- Hancock, R.E.W.; Sahl, H.-G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 2006, 24, 1551–1557. [Google Scholar] [CrossRef]
- Li, J.; Koh, J.-J.; Liu, S.; Lakshminarayanan, R.; Verma, C.S.; Beuerman, R.W. Membrane Active Antimicrobial Peptides: Translating Mechanistic Insights to Design. Front. Neurosci. 2017, 11, 73. [Google Scholar] [CrossRef] [Green Version]
- Ciociola, T.; Giovati, L.; Conti, S.; Santinoli, C.; Polonelli, L. Natural and synthetic peptides with antifungal activity. Future Med. Chem. 2016, 8, 1413–1433. [Google Scholar] [CrossRef]
- Nett, J.E.; Zarnowski, R.; Cabezas-Olcoz, J.; Brooks, E.G.; Bernhardt, J.; Marchillo, K.; Mosher, D.F.; Andes, D.R. Host contributions to construction of three device-associated Candida albicans biofilms. Infect. Immun. 2015, 83, 4630–4638. [Google Scholar] [CrossRef]
- Pulido, D.; Arranz-Trullén, J.; Prats-Ejarque, G.; Velázquez, D.; Torrent, M.; Moussaoui, M.; Boix, E. Insights into the Antimicrobial Mechanism of Action of Human RNase6: Structural Determinants for Bacterial Cell Agglutination and Membrane Permeation. Int. J. Mol. Sci. 2016, 17, 552. [Google Scholar] [CrossRef]
- Pulido, D.; Torrent, M.; Andreu, D.; Nogués, M.V.; Boix, E.; Nogues, M.V.; Boix, E.; Nogués, M.V.; Boix, E. Two human host defense ribonucleases against mycobacteria, the eosinophil cationic protein (RNase 3) and RNase 7. Antimicrob. Agents Chemother. 2013, 57, 3797–3805. [Google Scholar] [CrossRef]
- Rosenberg, H.F. Recombinant human eosinophil cationic protein. Ribonuclease activity is not essential for cytotoxicity. J. Biol. Chem. 1995, 270, 7876–7881. [Google Scholar] [CrossRef]
- Torrent, M.; Pulido, D.; Nogués, M.V.; Boix, E. Exploring new biological functions of amyloids: bacteria cell agglutination mediated by host protein aggregation. PLoS Pathog. 2012, 8, 2012–2014. [Google Scholar] [CrossRef]
- Pizzo, E.; D’Alessio, G. The success of the RNase scaffold in the advance of biosciences and in evolution. Gene 2007, 406, 8–12. [Google Scholar] [CrossRef]
- Rosenberg, H.F. RNase A ribonucleases and host defense: An evolving story. J. Leukoc. Biol. 2008, 83, 1079–1087. [Google Scholar] [CrossRef]
- Becknell, B.; Eichler, T.E.; Beceiro, S.; Li, B.; Easterling, R.S.; Carpenter, A.R.; James, C.L.; McHugh, K.M.; Hains, D.S.; Partida-Sanchez, S.; et al. Ribonucleases 6 and 7 have antimicrobial function in the human and murine urinary tract. Kidney Int. 2014, 87, 151–161. [Google Scholar] [CrossRef]
- Pulido, D.; Moussaoui, M.; Andreu, D.; Nogués, M.V.; Torrent, M.; Boix, E. Antimicrobial action and cell agglutination by the eosinophil cationic protein are modulated by the cell wall lipopolysaccharide structure. Antimicrob. Agents Chemother. 2012, 56, 2378–2385. [Google Scholar] [CrossRef]
- Pulido, D.; Moussaoui, M.; Nogués, M.V.; Torrent, M.; Boix, E. Towards the rational design of antimicrobial proteins: Single point mutations can switch on bactericidal and agglutinating activities on the RNase A superfamily lineage. FEBS J. 2013, 1–12. [Google Scholar] [CrossRef]
- Chao, T.-Y.; Raines, R.T. Mechanism of ribonuclease A endocytosis: Analogies to cell-penetrating peptides. Biochemistry 2011, 50, 8374–8382. [Google Scholar] [CrossRef]
- Bahnsen, J.S.; Franzyk, H.; Sandberg-Schaal, A.; Nielsen, H.M. Antimicrobial and cell-penetrating properties of penetratin analogs: Effect of sequence and secondary structure. Biochim. Biophys. Acta Biomembr. 2013, 1828, 223–232. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Y.; Mohapatra, S.; Weisshaar, J.C. Rigidification of the Escherichia coli cytoplasm by the human antimicrobial peptide LL-37 revealed by superresolution fluorescence microscopy. Proc. Natl. Acad. Sci. USA 2018, 116, 201814924. [Google Scholar] [CrossRef]
- Muñoz-Camargo, C.; Salazar, V.; Barrero, L.; Camargo, S.; Mosquera, A.; Mitrani, E.; Groot, H.; Boix, E. Unveiling the multifaceted mechanisms of antibacterial activity of Buforin II and Frenatin 2.3S peptides from skin micro-organs of Sphaenorhynchus lacteus (Hylidae). Int. J. Mol. Sci. 2018, 19, 2170. [Google Scholar] [CrossRef]
- Li, L.; Sun, J.; Xia, S.; Tian, X.; Cheserek, M.J.; Le, G. Mechanism of antifungal activity of antimicrobial peptide APP, a cell-penetrating peptide derivative, against Candida albicans: intracellular DNA binding and cell cycle arrest. Appl. Microbiol. Biotechnol. 2016, 100, 3245–3253. [Google Scholar] [CrossRef]
- Tsai, P.W.; Cheng, Y.L.; Hsieh, W.P.; Lan, C.Y. Responses of Candida albicans to the human antimicrobial peptide LL-37. J. Microbiol. 2014, 52, 581–589. [Google Scholar] [CrossRef]
- Wang, T.; Xiu, J.; Zhang, Y.; Wu, J.; Ma, X.; Wang, Y.; Guo, G.; Shang, X. Transcriptional responses of Candida albicans to antimicrobial peptide MAF-1A. Front. Microbiol. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- Theberge, S.; Semlali, A.; Alamri, A.; Leung, K.P.; Rouabhia, M.C. albicans growth, transition, biofilm formation, and gene expression modulation by antimicrobial decapeptide KSL-W. BMC Microbiol. 2013, 13, 1. [Google Scholar] [CrossRef]
- Morici, P.; Fais, R.; Rizzato, C.; Tavanti, A.; Lupetti, A. Inhibition of Candida albicans biofilm formation by the synthetic lactoferricin derived peptide hLF1-11. PLoS ONE 2016, 11, 1–15. [Google Scholar] [CrossRef]
- Deveau, A.; Hogan, D.A. Linking Quorum Sensing Regulation and Biofilm Formation by Candida albicans. In Quorum Sensing; Humana Press: Totowa, NJ, USA, 2011; pp. 219–233. ISBN 978-1-60761-971-0. [Google Scholar]
- Kyriakidis, I.; Tragiannidis, A.; Munchen, S.; Groll, A.H. Clinical hepatotoxicity associated with antifungal agents. Expert Opin. Drug Saf. 2017, 16, 149–165. [Google Scholar] [CrossRef]
- Graham, C.E.; Cruz, M.R.; Garsin, D.A.; Lorenz, M.C. Enterococcus faecalis bacteriocin EntV inhibits hyphal morphogenesis, biofilm formation, and virulence of Candida albicans. Proc. Natl. Acad. Sci. USA 2017, 114, 4507–4512. [Google Scholar] [CrossRef]
- Pletzer, D.; Coleman, S.R.; Hancock, R.E.W. Anti-biofilm peptides as a new weapon in antimicrobial warfare. Curr. Opin. Microbiol. 2016, 33, 35–40. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, S.M.; Felício, M.R.; Boas, E.V.; Gonçalves, S.; Costa, F.F.; Samy, R.P.; Santos, N.C.; Franco, O.L. New frontiers for anti-biofilm drug development. Pharmacol. Ther. 2016, 160, 133–144. [Google Scholar] [CrossRef]
- Mathur, H.; Field, D.; Rea, M.C.; Cotter, P.D.; Hill, C.; Ross, R.P. Fighting biofilms with lantibiotics and other groups of bacteriocins. npj Biofilms Microbiomes 2018, 4, 1–13. [Google Scholar] [CrossRef]
- Paulone, S.; Ardizzoni, A.; Tavanti, A.; Piccinelli, S.; Rizzato, C.; Lupetti, A.; Colombari, B.; Pericolini, E.; Polonelli, L.; Magliani, W.; et al. The synthetic killer peptide KP impairs Candida albicans biofilm in vitro. PLoS ONE 2017, 12, 1–16. [Google Scholar] [CrossRef]
- Helmerhorst, E.J.; Troxler, R.F.; Oppenheim, F.G. The human salivary peptide histatin 5 exerts its antifungal activity through the formation of reactive oxygen species. Proc. Natl. Acad. Sci. USA 2001, 98, 14637–14642. [Google Scholar] [CrossRef] [Green Version]
- Lupetti, A.; Paulusma-Annema, A.; Senesi, S.; Campa, M.; Van Dissel, J.T.; Nibbering, P.H. Internal thiols and reactive oxygen species in candidacidal activity exerted by an N-terminal peptide of human lactoferrin. Antimicrob. Agents Chemother. 2002, 46, 1634–1639. [Google Scholar] [CrossRef]
- Hua, J.; Yamarthy, R.; Felsenstein, S.; Scott, R.W.; Markowitz, K.; Diamond, G. Activity of antimicrobial peptide mimetics in the oral cavity: I. Activity against biofilms of Candida albicans. Mol. Oral Microbiol. 2010, 25, 418–425. [Google Scholar] [CrossRef] [Green Version]
- Baker, O.J.; Edgerton, M.; Kramer, J.M.; Ruhl, S. Saliva-microbe interactions and salivary gland dysfunction. Adv. Dent. Res. 2014, 26, 7–14. [Google Scholar] [CrossRef]
- Ip, W.; Lau, Y. Role of mannose-binding lectin in the innate defense against Candida albicans: Enhancement of complement activation, but lack of opsonic function, in phagocytosis by human dendritic cells. J. Infect. Dis. 2004, 190, 632–640. [Google Scholar] [CrossRef]
- Clemons, K.V.; Martinez, M.; Axelsen, M.; Thiel, S.; Stevens, D.A. Efficacy of recombinant human mannose binding lectin alone and in combination with itraconazole against murine Candida albicans vaginitis. Immunol. Investig. 2011, 40, 553–568. [Google Scholar] [CrossRef]
- Hammad, N.M.; El Badawy, N.E.; Ghramh, H.A.; Al Kady, L.M. Mannose-Binding Lectin: A Potential Therapeutic Candidate against Candida Infection. Biomed Res. Int. 2018, 2018, 1–8. [Google Scholar] [CrossRef]
- Park, C.B.; Kim, H.S.; Kim, S.C. Mechanism of action of the antimicrobial peptide buforin II: buforin II kills microorganisms by penetrating the cell membrane and inhibiting cellular functions. Biochem. Biophys. Res. Commun. 1998, 244, 253–257. [Google Scholar] [CrossRef]
- Jang, S.A.; Kim, H.; Lee, J.Y.; Shin, J.R.; Kim, D.J.; Cho, J.H.; Kim, S.C. Mechanism of action and specificity of antimicrobial peptides designed based on buforin IIb. Peptides 2012, 34, 283–289. [Google Scholar] [CrossRef]
- Jin, Y.; Yip, H.K.; Samaranayake, Y.H.; Yau, J.Y.; Samaranayake, L.P. Biofilm-forming ability of Candida albicans is unlikely to contribute to high levels of oral yeast carriage in cases of human immunodeficiency virus infection. J. Clin. Microbiol. 2003, 41, 2961–2967. [Google Scholar] [CrossRef]
- Silva, S.; Henriques, M.; Oliveira, R.; Williams, D.; Azeredo, J. In vitro biofilm activity of non-Candida albicans Candida species. Curr. Microbiol. 2010, 61, 534–540. [Google Scholar] [CrossRef]
- Peeters, E.; Nelis, H.J.; Coenye, T. Comparison of multiple methods for quantification of microbial biofilms grown in microtiter plates. J. Microbiol. Methods 2008, 72, 157–165. [Google Scholar] [CrossRef] [Green Version]
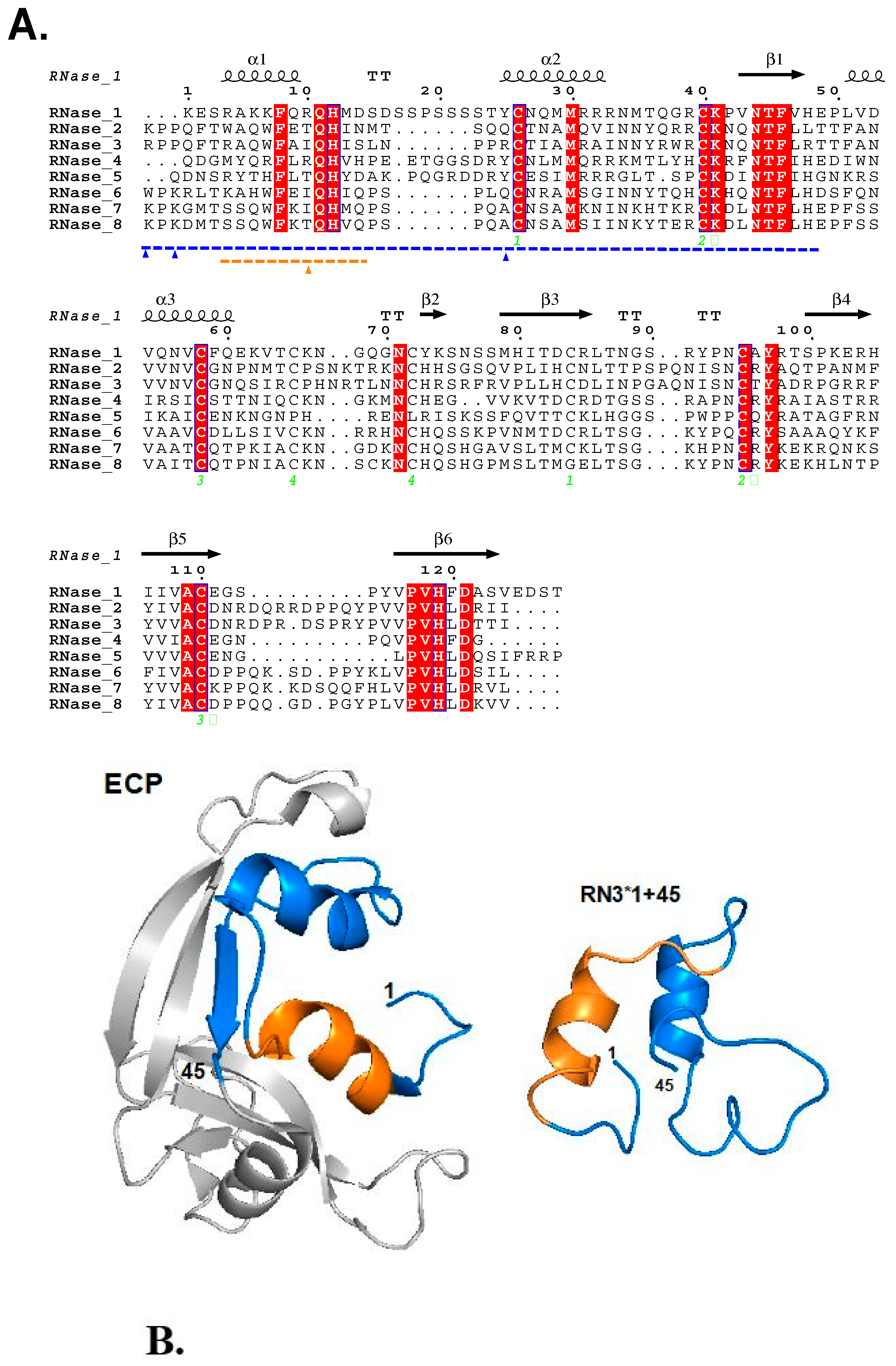




| Peptide † | Sequence | Hydrophobicity | pI | K/R | Protein Antimicrobial Activity * | Cytotoxicity (THP1) # GIC50 (μM) |
|---|---|---|---|---|---|---|
| RN1 | ---KESRAKKFQRQHMDSDSSPSSSSTYSNQMMRRRNMTQGRSKPVNTFVH | −1.575 | 11.40 | 4/6 | n.r | >50 |
| RN2 | KPPQFTWAQWFETQHINMTSQ------QSTNAMQVINNYQRRSKNQNTFLL | −1.069 | 10.28 | 2/2 | - | >50 |
| RN3 | RPPQFTRAQWFAIQHISLNPP------RSTIAMRAINNYRWRSKNQNTFLR | −0.764 | 11.88 | 1/7 | +++ Ŧ | >50 |
| RN3K | KPPQFTKAQWFAIQHISLNPP------KSTIAMKAINNYKWKSKNQNTFLK | −0.824 | 11.02 | 8/- | +++ | >50 |
| RN4 | --QDGMY-QRFLRQHVHPEET-GGSDRYSNLMMQRRKMTLYHSKRFNTFIH | −1.228 | 10.15 | 2/6 | n.r | >50 |
| RN5 | --QDNSRYTHFLTQHYDAKPQ-GRDDRYSESIMRRRGLTS-PSKDINTFIH | −1.430 | 9.40 | 2/6 | - | >50 |
| RN6 | WPKRLTKAHWFEIQHIQPSPL------QSNRAMSGINNYTQHSKHQNTFLH | −1.096 | 10.45 | 3/2 | +++ | >50 |
| RN7 | KPKGMTSSQWFKIQHMQPSPQ------ASNSAMKNINKHTKRSKDLNTFLH | −1.209 | 10.75 | 6/1 | +++ Ŧ | >50 |
| RN7R | RPRGMTSSQWFRIQHMQPSPQ------ASNSAMRNINRHTRRSRDLNTFLH | −1.302 | 12.88 | -/8 | +++ | >50 |
| RN8 | KPKDMTSSQWFKTQHVQPSPQ------ASNSAMSIINKYTERSKDLNTFLH | −1.044 | 9.70 | 5/1 | + | >50 |
| hRNase Peptide | MFC100 (μM) | IC50 (µM) | |
|---|---|---|---|
| Sabouraud Broth | PBS | ||
| RN1 | 3.75 ± 0.50 | 3.75 ± 0.30 | 2.10 ± 0.20 |
| RN2 | >20 | >20 | >20 |
| RN3 | 0.62 ± 0.20 | 0.62 ± 0.10 | 0.22 ± 0.05 |
| RN4 | 4.00 ± 0.50 | 3.75 ± 0.20 | 1.80 ± 0.30 |
| RN5 | >20 | >20 | >20 |
| RN6 | 2.00 ± 0.50 | 1.75 ± 0.30 | 1.80 ± 0.20 |
| RN7 | 0.93 ± 0.20 | 0.93 ± 0.10 | 0.45 ± 0.10 |
| RN8 | >10 | >10 | >10 |
| hRNase Peptide | Membrane Depolarization | Membrane Permeabilization | Cell Survival | ||
|---|---|---|---|---|---|
| ED50 (µM) | Depolmax * | Max. Perm. (AU) 1 | Perm. (%) 2 | % ¥ | |
| RN1 | 4.63 ± 0.70 | 65.2 ± 4.6 | 26.1 ± 0.4 | 8.3 ± 0.4 | 67 ± 1 |
| RN2 | 3.63 ± 1.20 | 28.6 ± 5.7 | 45.2 ± 0.8 | 12.4 ± 0.2 | 72 ± 0.5 |
| RN3 | 0.60 ± 0.40 | 100 ± 3.2 | 166 ± 2 | 73.2 ± 1 | 15 ± 2 |
| RN4 | 3.58 ± 1.60 | 76.2 ± 4.8 | 117 ± 2 | 38.7 ± 0.1 | 65 ± 10 |
| RN5 | 1.51 ± 0.10 | 39.1 ± 5.1 | 66.5 ± 0.6 | 21.8 ± 0.4 | 39 ± 0.5 |
| RN6 | 0.62 ± 0.10 | 86.2 ± 5.2 | 135 ± 1 | 48.4 ± 0.8 | 26 ± 2 |
| RN7 | 0.53 ± 0.18 | 71.5 ± 3.7 | 136 ± 0.4 | 53.9 ± 0.5 | 12 ± 1 |
| RN8 | 0.15 ± 0.20 | 36.1 ± 4.4 | 33.4 ± 0.4 | 11.2 ± 0.2 | 28 ± 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salazar, V.A.; Arranz-Trullén, J.; Prats-Ejarque, G.; Torrent, M.; Andreu, D.; Pulido, D.; Boix, E. Insight into the Antifungal Mechanism of Action of Human RNase N-terminus Derived Peptides. Int. J. Mol. Sci. 2019, 20, 4558. https://doi.org/10.3390/ijms20184558
Salazar VA, Arranz-Trullén J, Prats-Ejarque G, Torrent M, Andreu D, Pulido D, Boix E. Insight into the Antifungal Mechanism of Action of Human RNase N-terminus Derived Peptides. International Journal of Molecular Sciences. 2019; 20(18):4558. https://doi.org/10.3390/ijms20184558
Chicago/Turabian StyleSalazar, Vivian A., Javier Arranz-Trullén, Guillem Prats-Ejarque, Marc Torrent, David Andreu, David Pulido, and Ester Boix. 2019. "Insight into the Antifungal Mechanism of Action of Human RNase N-terminus Derived Peptides" International Journal of Molecular Sciences 20, no. 18: 4558. https://doi.org/10.3390/ijms20184558