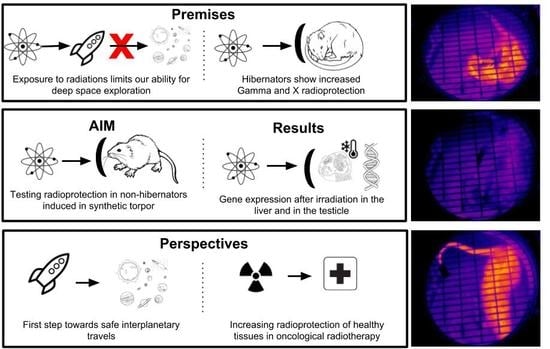
Hibernation and Radioprotection: Gene Expression in the Liver and Testicle of Rats Irradiated under Synthetic Torpor
Abstract
:1. Introduction
2. Results
3. Discussion
4. Methods
4.1. Animal Housing
4.2. Surgery
4.3. Experimental Protocol
- (1)
- Hypothermia (n = 5): After a 7-day recovery from surgery, animals were injected with the GABA-A agonist muscimol (1 mmoL) within the Raphe Pallidus (RPa). Each animal received, starting from 07:00 h, one injection/hour (100 nL). All animals entered synthetic torpor shortly after the first injection and maintained such condition until the end of the experiment.
- (2)
- Control (n = 5): After a 7-day recovery from surgery, animals were injected with artificial cerebrospinal fluid (ACSF) within the RPa. Each animal received, starting from 07:00 h, one injection/hour (100 nL).
4.4. Radiation Data
4.5. Gene Expression and Analysis
4.5.1. Liver
4.5.2. Testicle
4.5.3. Histology
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Heldmaier, G.; Ortmann, S.; Elvert, R. Natural hypometabolism during hibernation and daily torpor in mammals. Respir. Physiol. Neurobiol. 2004, 141, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Geiser, F. Metabolic rate and body temperature reduction during hibernation and daily torpor. Annu. Rev. Physiol. 2004, 66, 239–274. [Google Scholar] [CrossRef]
- Geiser, F. Ontogeny and phylogeny of endothermy and torpor in mammals and birds. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2008, 150, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Malan, A. The evolution of mammalian hibernation: Lessons from comparative acid-base physiology. Integr. Comp. Biol. 2014, 54, 484–496. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.C. Is human hibernation possible? Annu. Rev. Med. 2008, 59, 177–186. [Google Scholar] [CrossRef]
- Bouma, H.R.; Verhaag, E.M.; Otis, J.P.; Heldmaier, G.; Swoap, S.J.; Strijkstra, A.M.; Henning, R.H.; Carey, H.V. Induction of torpor: Mimicking natural metabolic suppression for biomedical applications. J. Cell. Physiol. 2012, 227, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Cerri, M. The Central Control of Energy Expenditure: Exploiting Torpor for Medical Applications. Annu. Rev. Physiol. 2017, 79, 167–186. [Google Scholar] [CrossRef]
- Cerri, M.; Tinganelli, W.; Negrini, M.; Helm, A.; Scifoni, E.; Tommasino, F.; Sioli, M.; Zoccoli, A.; Durante, M. Hibernation for space travel: Impact on radioprotection. Life Sci. Space Res. (Amst.) 2016, 11, 1–9. [Google Scholar] [CrossRef] [Green Version]
- ESA Advanced Concept Team. Available online: https://www.esa.int/gsp/ACT/projects/hibernation.html (accessed on 14 November 2018).
- Petit, G.; Koller, D.; Summerer, L.; Heldmaier, G.; Vyazovskiy, V.V.; Cerri, M.; Henning, R.H. Hibernation and Torpor: Prospects for Human Spaceflight. In Handbook of Life Support Systems for Spacecraft and Extraterrestrial Habitats; Seedhouse, E., Shayler, D., Eds.; Springer: Cham, Switzerland, 2018; pp. 1–15. ISBN 978-3-319-09575-2. [Google Scholar]
- Gemignani, J.; Gheysens, T.; Summerer, L. Beyond astronaut’s capabilities: The current state of the art. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2015, 2015, 3615–3618. [Google Scholar]
- Musacchia, X.J.; Barr, R.E. Survival of whole-body-irradiated hibernating and active ground squirrels; Citellus tridecemlineatus. Radiat. Res. 1968, 33, 348–356. [Google Scholar] [CrossRef]
- Durante, M.; Cucinotta, F.A. Heavy ion carcinogenesis and human space exploration. Nat. Rev. Cancer 2008, 8, 465–472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Durante, M. Space radiation protection: Destination Mars. Life Sci. Space Res. (Amst.) 2014, 1, 2–9. [Google Scholar] [CrossRef]
- Zeitlin, C.; Hassler, D.M.; Cucinotta, F.A.; Ehresmann, B.; Wimmer-Schweingruber, R.F.; Brinza, D.E.; Kang, S.; Weigle, G.; Bottcher, S.; Bohm, E.; et al. Measurements of energetic particle radiation in transit to Mars on the Mars Science Laboratory. Science 2013, 340, 1080–1084. [Google Scholar] [CrossRef] [PubMed]
- Hock, R.J. The potential application of hibernation to space travel. Aerosp. Med. 1960, 31, 485–489. [Google Scholar] [PubMed]
- Cockett, T.K.; Beehler, C.C. Protective effects of hypothermia in exploration of space. JAMA 1962, 182, 977–979. [Google Scholar] [CrossRef]
- Jaroslow, B.N.; Smith, D.E.; Williams, M.; Tyler, S.A. Survival of hibernating ground squirrels (Citellus tridecemlineatus) after single and fractionated doses of cobalt-60 gamma radiation. Radiat. Res. 1969, 38, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Barr, R.E.; Musacchia, X.J. The effect of body temperature and postirradiation cold exposure on the radiation response of the hibernator Citellus tridecemlineatus. Radiat. Res. 1969, 38, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Musacchia, X.J.; Volkert, W.A.; Barr, R.E. Radioresistance in hamsters during hypothermic depressed metabolism induced with helium and low temperatures. Radiat. Res. 1971, 46, 353–361. [Google Scholar] [CrossRef]
- Ghosh, S.; Indracanti, N.; Joshi, J.; Ray, J.; Indraganti, P.K. Pharmacologically induced reversible hypometabolic state mitigates radiation induced lethality in mice. Sci. Rep. 2017, 7, 14900. [Google Scholar] [CrossRef] [Green Version]
- Baird, B.J.; Dickey, J.S.; Nakamura, A.J.; Redon, C.E.; Parekh, P.; Griko, Y.V.; Aziz, K.; Georgakilas, A.G.; Bonner, W.M.; Martin, O.A. Hypothermia postpones DNA damage repair in irradiated cells and protects against cell killing. Mutat. Res. 2011, 711, 142–149. [Google Scholar] [CrossRef]
- Cerri, M.; Mastrotto, M.; Tupone, D.; Martelli, D.; Luppi, M.; Perez, E.; Zamboni, G.; Amici, R. The inhibition of neurons in the central nervous pathways for thermoregulatory cold defense induces a suspended animation state in the rat. J. Neurosci. 2013, 33, 2984–2993. [Google Scholar] [CrossRef]
- Morrison, S.F.; Madden, C.J.; Tupone, D. Central neural regulation of brown adipose tissue thermogenesis and energy expenditure. Cell Metab. 2014, 19, 741–756. [Google Scholar] [CrossRef]
- Griko, Y.; Regan, M.D. Synthetic torpor: A method for safely and practically transporting experimental animals aboard spaceflight missions to deep space. Life Sci. Space Res. (Amst.) 2018, 16, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Dang, L.; Lisowska, H.; Manesh, S.S.; Sollazzo, A.; Deperas-Kaminska, M.; Staaf, E.; Haghdoost, S.; Brehwens, K.; Wojcik, A. Effect of hypothermia on cells—A multiparametric approach to delineate the mechanisms. Int. J. Radiat. Biol. 2012, 88, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Lisowska, H.; Cheng, L.; Sollazzo, A.; Lundholm, L.; Wegierek-Ciuk, A.; Sommer, S.; Lankoff, A.; Wojcik, A. Hypothermia modulates the DNA damage response to ionizing radiation in human peripheral blood lymphocytes. Int. J. Radiat. Biol. 2018, 94, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Herzog, K.H.; Chong, M.J.; Kapsetaki, M.; Morgan, J.I.; McKinnon, P.J. Requirement for ATM in ionizing radiation-induced cell death in the developing central nervous system. Science 1998, 280, 1089–1091. [Google Scholar] [CrossRef] [PubMed]
- Heylmann, D.; Badura, J.; Becker, H.; Fahrer, J.; Kaina, B. Sensitivity of CD3/CD28-stimulated versus non-stimulated lymphocytes to ionizing radiation and genotoxic anticancer drugs: Key role of ATM in the differential radiation response. Cell Death Dis. 2018, 9, 1053. [Google Scholar] [CrossRef]
- Chen, N.; Jiang, J.; Gao, X.; Li, X.; Zhang, Y.; Liu, X.; Yang, H.; Bing, X.; Zhang, X. Histopathological analysis and the immune related gene expression profiles of mandarinfish (Siniperca chuatsi) infected with Aeromonas hydrophila. Fish Shellfish Immunol. 2018, 83, 410–415. [Google Scholar] [CrossRef]
- Abdelhalim, M.A.; Jarrar, B.M. Histological alterations in the liver of rats induced by different gold nanoparticle sizes, doses and exposure duration. J. Nanobiotechnol. 2012, 10, 5. [Google Scholar] [CrossRef] [Green Version]
- Khoronenkova, S.V.; Dianov, G.L. ATM prevents DSB formation by coordinating SSB repair and cell cycle progression. Proc. Natl. Acad. Sci. USA 2015, 112, 3997–4002. [Google Scholar] [CrossRef] [Green Version]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2007; ISBN 9780125476126. [Google Scholar]
- Nalivaiko, E.; Blessing, W.W. Raphe region mediates changes in cutaneous vascular tone elicited by stimulation of amygdala and hypothalamus in rabbits. Brain Res. 2001, 891, 130–137. [Google Scholar] [CrossRef]







© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tinganelli, W.; Hitrec, T.; Romani, F.; Simoniello, P.; Squarcio, F.; Stanzani, A.; Piscitiello, E.; Marchesano, V.; Luppi, M.; Sioli, M.; et al. Hibernation and Radioprotection: Gene Expression in the Liver and Testicle of Rats Irradiated under Synthetic Torpor. Int. J. Mol. Sci. 2019, 20, 352. https://doi.org/10.3390/ijms20020352
Tinganelli W, Hitrec T, Romani F, Simoniello P, Squarcio F, Stanzani A, Piscitiello E, Marchesano V, Luppi M, Sioli M, et al. Hibernation and Radioprotection: Gene Expression in the Liver and Testicle of Rats Irradiated under Synthetic Torpor. International Journal of Molecular Sciences. 2019; 20(2):352. https://doi.org/10.3390/ijms20020352
Chicago/Turabian StyleTinganelli, Walter, Timna Hitrec, Fabrizio Romani, Palma Simoniello, Fabio Squarcio, Agnese Stanzani, Emiliana Piscitiello, Valentina Marchesano, Marco Luppi, Maximiliano Sioli, and et al. 2019. "Hibernation and Radioprotection: Gene Expression in the Liver and Testicle of Rats Irradiated under Synthetic Torpor" International Journal of Molecular Sciences 20, no. 2: 352. https://doi.org/10.3390/ijms20020352