miRNAs and Novel Food Compounds Related to the Browning Process
Abstract
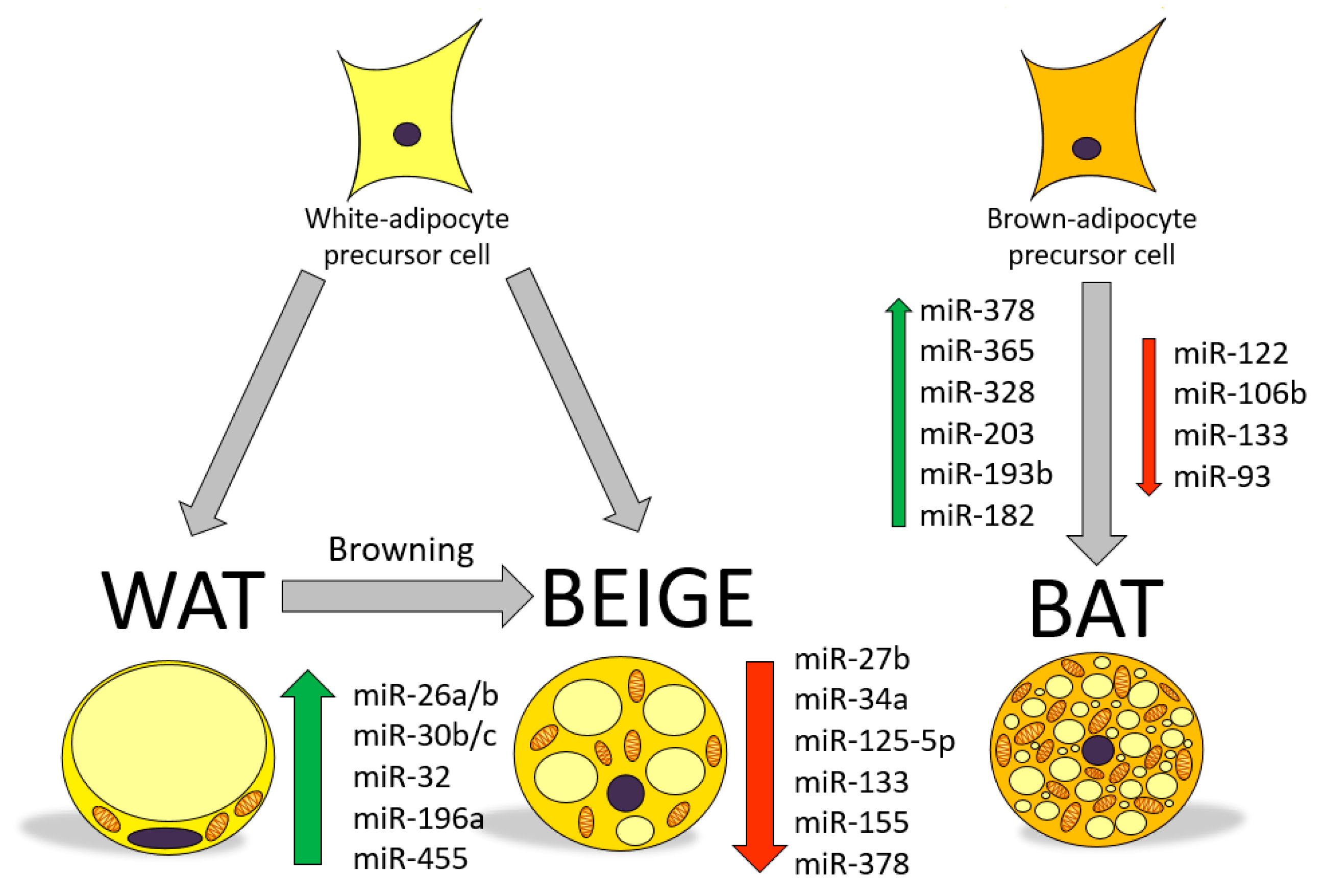
1. Brown and Beige Adipose Tissue in Energy Balance
2. MicroRNAs as Regulators of Gene Function and Metabolism
3. Involvement of miRNAs in the Regulation of Browning: Role of Nutritional Factors
4. Food Compounds Related to the Browning Process
4.1. Spices
4.1.1. Chili Peppers
4.1.2. Turmeric
4.1.3. Thyme
4.1.4. Cinnamon
4.1.5. Garlic
4.1.6. Onion
4.2. Other Herbal and Food Compounds
4.2.1. Magnolia Officinalis
4.2.2. Berberine
4.2.3. Panax ginseng
4.2.4. Mentha
4.2.5. Chrysin
4.2.6. Soy
4.2.7. Green Tea Polyphenols
4.3. PUFA’s
4.3.1. Conjugated Linoleic Acid
4.3.2. Eicosapentaenoic Acid
4.3.3. Docosahexaenoic Acid
4.4. Carotenoids
4.4.1. β-Carotene
4.4.2. Fucoxanthin
4.5. Citrus Fruits
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AMPK | AMP-activated protein kinase |
| AMPKα1 | 5-AMP-activated protein kinase catalytic subunit alpha-1 |
| BAT | Brown adipose tissue |
| BMP7 | Bone morphogenetic protein 7 |
| BW | Body weight |
| C/EBPα | CCAAT/enhancer binding protein alpha |
| Cd137 | 4-1BB |
| Cd40 | CD40 molecule |
| CIDEA | Cell death inducing DFFA like effector A |
| CITED1 | Cpb/P300 interacting transactivator with Glu/Asp rich carboxy-terminal |
| CLA | Conjugated linoleic acid |
| Cox8 | Cytochrome c oxidase subunit 8 |
| CPTB1β | Carnitine palmitoyltransferase I |
| DHA | Docosahexanoic acid |
| Dio2 | Iodothyronine deiodinase 2 |
| EGCG | Epigallocatechin gallate |
| Elovl3 | Elongation of very long-chain fatty acid 3 |
| EPA | Eicosapentanoic acid |
| ERK | Extracellular signal-regulated kinases |
| eWAT | Epididymal WAT |
| Fgf32 | Fibroblast growth factor 23 |
| FNDC5 | Fibronectin type III domain-containing protein 5 |
| Foxc2 | Forkhead box C2 |
| HFD | High-fat diet |
| HUVECs | Human umbilical vein endothelial cells |
| hWAT | Human white adipose tissue |
| iWAT | Inguinal WAT |
| LA | Linoleic acid |
| LFD | Low-fat diet |
| LPS | Lipopolysaccharides |
| MAPK | Mitogen-activated protein kinase |
| miRNAs | MicroRNAs |
| mWAT | Mesenteric WAT |
| Myf5 | Myogenic factor 5 |
| NAFLD | Nonalcoholic fatty liver disease |
| OPE | Onion peel extracti |
| Pgc-1α | Peroxisome proliferator-activated γ receptor co-activator 1 alpha |
| Ppargc1b | PPARG coactivator 1 beta |
| Pparα | Peroxisome proliferator-activated receptor α |
| Pparγ | Peroxisome proliferator-activated receptor gamma |
| Pparδ | Peroxisome proliferator-activated receptor delta |
| Prmd16 | PR-domain containing 16 |
| PUFA | Polyunsaturated fatty acids |
| RA | Retinoic acid |
| RAR | Retinoic acid receptor |
| RNA | Ribonucleic acid |
| rWAT | Retroperitoneal WAT |
| RXR | Retinoid X receptor |
| Sirt-1 | Sirtuin 1 |
| sWAT | Subcutaneous WAT |
| Tbx1 | T-box 1 |
| tCA | Trans-cinnamic acid |
| TG | Triglycerides |
| Tmem26 | Transmembrane protein 26 |
| TRPM8 | Transient receptor potential cation channel subfamily M (melastatin) member 8 |
| TRPV1 | Transient receptor potential vanilloid subfamily 1 |
| UCP-1 | Uncoupling protein-1 |
| UCP-2 | Uncoupling protein-2 |
| VEGFα | Vascular endothelial growth factor alpha |
| WAT | White adipose tissue |
| β3-AR | Beta3 adrenergic receptor |
References
- Villarroya, F.; Gavaldà-Navarro, A.; Peyrou, M.; Villarroya, J.; Giralt, M. Brown Adipokines. In Handbook of Experimental Pharmacology; Springer: Basel, Switzerland, 2019; Volume 251, pp. 239–256. ISBN 978-3-030-10512-9. [Google Scholar]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Shabalina, I.; Petrovic, N.; de Jong, J.M.A.; Kalinovich, A.; Cannon, B.; Nedergaard, J. UCP1 in Brite/Beige Adipose Tissue Mitochondria Is Functionally Thermogenic. Cell Rep. 2013, 5, 1196–1203. [Google Scholar] [CrossRef] [PubMed]
- Stojanović, O.; Kieser, S.; Trajkovski, M. Common traits between the beige fat-inducing stimuli. Curr. Opin. Cell Biol. 2018, 55, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Peirce, V.; Carobbio, S.; Vidal-Puig, A. The different shades of fat. Nature 2014, 510, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Gurmaches, J.; Hung, C.M.; Sparks, C.A.; Tang, Y.; Li, H.; Guertin, D.A. PTEN loss in the Myf5 lineage redistributes body fat and reveals subsets of white adipocytes that arise from Myf5 precursors. Cell Metab. 2012, 16, 348–362. [Google Scholar] [CrossRef] [PubMed]
- Kajimura, S.; Spiegelman, B.M.; Seale, P. Brown and Beige Fat: Physiological Roles beyond Heat Generation. Cell Metab. 2015, 22, 546–559. [Google Scholar] [CrossRef]
- Silvester, A.J.; Aseer, K.R.; Jang, H.J.; Ryu, E.Y.; Kwon, E.Y.; Park, J.G.; Cho, K.H.; Chaudhari, H.N.; Choi, M.S.; Suh, P.G.; et al. Loss of DJ-1 promotes browning of white adipose tissue in diet-induced obese mice. J. Nutr. Biochem. 2018, 61, 56–67. [Google Scholar] [CrossRef]
- Tamucci, K.A.; Namwanje, M.; Fan, L.; Qiang, L. The dark side of browning. Protein Cell 2018, 9, 152–163. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Bartel, D.P. Metazoan MicroRNAs. Cell 2018, 173, 20–51. [Google Scholar] [CrossRef]
- Arner, E.; Mejhert, N.; Kulyté, A.; Balwierz, P.J.; Pachkov, M.; Cormont, M.; Lorente-Cebrián, S.; Ehrlund, A.; Laurencikiene, J.; Hedén, P.; et al. Adipose tissue microRNAs as regulators of CCL2 production in human obesity. Diabetes 2012, 61, 1986–1993. [Google Scholar] [CrossRef] [PubMed]
- Trajkovski, M.; Ahmed, K.; Esau, C.C.; Stoffel, M. MyomiR-133 regulates brown fat differentiation through Prdm16. Nat. Cell Biol. 2012, 14, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Oskowitz, A.Z.; Lu, J.; Penfornis, P.; Ylostalo, J.; McBride, J.; Flemington, E.K.; Prockop, D.J.; Pochampally, R. Human multipotent stromal cells from bone marrow and microRNA: Regulation of differentiation and leukemia inhibitory factor expression. Proc. Natl. Acad. Sci. USA 2008, 105, 18372–18377. [Google Scholar] [CrossRef] [PubMed]
- Tay, Y.; Zhang, J.; Thomson, A.M.; Lim, B.; Rigoutsos, I. MicroRNAs to Nanog, Oct4 and Sox2 coding regions modulate embryonic stem cell differentiation. Nature 2008, 455, 1124–1128. [Google Scholar] [CrossRef]
- Broughton, J.P.; Lovci, M.T.; Huang, J.L.; Yeo, G.W.; Pasquinelli, A.E. Pairing beyond the Seed Supports MicroRNA Targeting Specificity. Mol. Cell 2016, 64, 320–333. [Google Scholar] [CrossRef]
- Vasudevan, S. Posttranscriptional upregulation by microRNAs. Wiley Interdiscip. Rev. RNA 2012, 3, 311–330. [Google Scholar] [CrossRef]
- Treiber, T.; Treiber, N.; Meister, G. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat. Rev. Mol. Cell Biol. 2019, 20, 5–20. [Google Scholar] [CrossRef]
- Arner, P.; Kulyté, A. MicroRNA regulatory networks in human adipose tissue and obesity. Nat. Rev. Endocrinol. 2015, 11, 276–288. [Google Scholar] [CrossRef]
- Shamsi, F.; Zhang, H.; Tseng, Y.H. MicroRNA Regulation of Brown Adipogenesis and Thermogenic Energy Expenditure. Front. Endocrinol. 2017, 8, 205. [Google Scholar] [CrossRef]
- Trajkovski, M.; Lodish, H. MicroRNA networks regulate development of brown adipocytes. Trends Endocrinol. Metab. TEM 2013, 24, 442–450. [Google Scholar] [CrossRef]
- Goody, D.; Pfeifer, A. MicroRNAs in brown and beige fat. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2019, 1864, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Lorente-Cebrián, S.; González-Muniesa, P.; Milagro, F.I.; Martínez, J.A. MicroRNAs and other non-coding RNAs in adipose tissue and obesity: Emerging roles as biomarkers and therapeutic targets. Clin. Sci. 2019, 133, 23–40. [Google Scholar] [CrossRef] [PubMed]
- Maciel-Dominguez, A.; Swan, D.; Ford, D.; Hesketh, J. Selenium alters miRNA profile in an intestinal cell line: Evidence that miR-185 regulates expression of GPX2 and SEP SH2. Mol. Nutr. Food Res. 2013, 57, 2195–2205. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Lacarte, M.; Martinez, J.A.; Zulet, M.A.; Milagro, F.I. Implication of miR-612 and miR-1976 in the regulation of TP53 and CD40 and their relationship in the response to specific weight-loss diets. PLoS ONE 2017, 13, e0201217. [Google Scholar] [CrossRef]
- Belarbi, Y.; Mejhert, N.; Gao, H.; Arner, P.; Rydén, M.; Kulyté, A. MicroRNAs-361-5p and miR-574-5p associate with human adipose morphology and regulate EBF1 expression in white adipose tissue. Mol. Cell. Endocrinol. 2018, 472, 50–56. [Google Scholar] [CrossRef]
- Xue, B.; Liu, Q.Y.; Massie, L.; Qualls, C.; Mao, J.T. Grape seed procyanidin extract against lung cancer: The role of microrna-106b, bioavailability, and bioactivity. Oncotarget 2018, 9, 15579–15590. [Google Scholar] [CrossRef]
- Schlörmann, W.; Naumann, S.; Renner, C.; Glei, M. Influence of miRNA-106b and miRNA-135a on butyrate-regulated expression of p21 and Cyclin D2 in human colon adenoma cells. Genes Nutr. 2015, 10, 50. [Google Scholar] [CrossRef]
- Liu, W.; Cao, H.; Ye, C.; Chang, C.; Lu, M.; Jing, Y.; Zhang, D.; Yao, X.; Duan, Z.; Xia, H.; et al. Hepatic miR-378 targets p110α and controls glucose and lipid homeostasis by modulating hepatic insulin signalling. Nat. Commun. 2014, 5, 5684. [Google Scholar] [CrossRef]
- Hu, S.; Dong, T.S.; Dalal, S.R.; Wu, F.; Bissonnette, M.; Kwon, J.H.; Chang, E.B. The Microbe-Derived Short Chain Fatty Acid Butyrate Targets miRNA-Dependent p21 Gene Expression in Human Colon Cancer. PLoS ONE 2011, 6, e16221. [Google Scholar] [CrossRef]
- Dhar, S.; Hicks, C.; Levenson, A. Resveratrol and prostate cancer: Promising role for microRNAs. Mol. Nutr. Food Res. 2011, 55, 1219–1229. [Google Scholar] [CrossRef]
- De Conti, A.; Ortega, J.F.; Tryndyak, V.; Dreval, K.; Moreno, F.S.; Rusyn, I.; Beland, F.A.; Pogribny, I.P. MicroRNA deregulation in nonalcoholic steatohepatitis-associated liver carcinogenesis. Oncotarget 2017, 8, 88517–88528. [Google Scholar] [CrossRef] [PubMed]
- McCann, M.J.; Rotjanapun, K.; Hesketh, J.E.; Roy, N.C. Expression profiling indicating low selenium-sensitive microRNA levels linked to cell cycle and cell stress response pathways in the Caco-2 cell line. Br. J. Nutr. 2017, 117, 1212–1221. [Google Scholar] [CrossRef] [PubMed]
- Okamatsu-Ogura, Y.; Matsushita, M.; Bariuan, J.V.; Nagaya, K.; Tsubota, A.; Saito, M. Association of circulating exosomal miR-122 levels with BAT activity in healthy humans. Sci. Rep. 2019, 9, 13243. [Google Scholar] [CrossRef] [PubMed]
- Ye, D.; Zhang, T.; Lou, G.; Xu, W.; Dong, F.; Chen, G.; Liu, Y. Plasma miR-17, miR-20a, miR-20b and miR-122 as potential biomarkers for diagnosis of NAFLD in type 2 diabetes mellitus patients. Life Sci. 2018, 208, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Adams, B.D.; Arem, H.; Hubal, M.J.; Cartmel, B.; Li, F.; Harrigan, M.; Sanft, T.; Cheng, C.J.; Pusztai, L.; Irwin, M.L. Excersice and weight loss interventions and miRNA expression in women with breast cancer. Breast Cancer Res. Treat. 2018, 170, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Satishchadran, A.; Ambade, A.; Rao, S.; Hsueh, Y.C.; Iracheta-Vellve, A.; Tornai, D.; Lowe, P.; Gyongyosi, B.; Li, J.; Catalano, D.; et al. MicroRNA 122, regulated by GRLH2, protects livers of mice and patients from ethanol-induced liver disease. Gastroenterology 2018, 154, 238–252. [Google Scholar] [CrossRef]
- Latorre, J.; Moreno-Navarrete, J.M.; Mercader, J.M.; Sabater, M.; Rovira, Ò.; Gironès, J.; Ricart, W.; Fernández-Real, M.; Ortega, F.J. Decreased lipid metabolism but increased FA biosynthesis are coupled with changes in liver microRNAs in obese subjects with NAFLD. Int. J. Obes. Lond. 2017, 41, 620–630. [Google Scholar] [CrossRef]
- Carreras-Badosa, G.; Bonmatí, A.; Ortega, F.J.; Mercader, J.M.; Guindo-Martínez, M.; Torrents, D.; Prats-Puig, A.; Martinez-Calcerrada, J.M.; Platero-Gutierrez, E.; De Zegher, F.; et al. Altered circulating miRNA expression profile in pregestational and gestational obesity. J. Clin. Endocrinol. Metab. 2015, 100, E1446–E1456. [Google Scholar] [CrossRef]
- Su, D.; Liu, H.; Qi, X.; Dong, L.; Zhang, R.; Zhang, J. Citrus peel flavonoids improve lipid metabolism by inhibiting miR-33 and miR-122 expression in HepG2 cells. Biosci. Biotechnol. Biochem. 2019, 83, 1747–1755. [Google Scholar] [CrossRef]
- Willeit, P.; Skroblin, P.; Moschen, A.R.; Yin, X.; Kaudewitz, D.; Zampetaki, A.; Barwari, T.; Whitehead, M.; Ramírez, C.M.; Goedeke, L.; et al. Circulating microRNA-122 is associated with the risk of new-onset metabolic syndrome and type 2 diabetes. Diabetes 2017, 66, 347–357. [Google Scholar] [CrossRef]
- Parasramka, M.A.; Ali, S.; Banerjee, S.; Deryavoush, T.; Sarkar, F.H.; Gupta, S. Garcinol sensitizes human pancreatic adenocarcinoma cells to gemcitabine in association with microRNA signatures. Mol. Nutr. Food Res. 2013, 57, 235–248. [Google Scholar] [CrossRef] [PubMed]
- Lam, T.K.; Shao, S.; Zhao, Y.; Marincola, F.; Pesatori, A.; Bertazzi, P.A.; Caporaso, N.E.; Wang, E.; Landi, M.T. Influence of quercetin-rich food intake on microRNA expression in lung cancer tissues. Cancer Epidemiol. Biomark. Biomark. 2012, 21, 2176–2184. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Dong, B.; Tian, Y.; Lefebvre, P.; Meng, Z.; Wang, X.; Pattou, F.; Han, W.; Wang, X.; Lou, F.; et al. MicroRNA-26a regulates insulin sensitivity and metabolism of glucose and lipids. J. Clin. Investig. 2015, 125, 2497–2509. [Google Scholar] [CrossRef] [PubMed]
- Malcomson, F.C.; Willis, N.D.; McCallum, I.; Xie, L.; Lagerwaard, B.; Kelly, S.; Bradburn, D.M.; Belshaw, N.J.; Johnson, I.T.; Mathers, J. Non-digestible carbohydrates supplementation increases miR-32 expression in the healthy human colorectal epithelium: A randomized controlled trial. Mol. Carcinog. 2017, 56, 2104–2111. [Google Scholar] [CrossRef]
- Gottmann, P.; Ouni, M.; Saussenthaler, S.; Roos, J.; Stirm, L.; Jähnert, M.; Kamitz, A.; Hallahan, N.; Jonas, W.; Fritsche, A.; et al. A computational biology approach of a genome-wide screen connected miRNAs to obesity and type 2 diabetes. Mol. Metab. 2018, 11, 145–159. [Google Scholar] [CrossRef]
- Zhang, T.; Cooper, S.; Brockdorff, N. The interplay of histone modifications—Writers that read. EMBO Rep. 2015, 16, 1467–1481. [Google Scholar] [CrossRef]
- Villard, A.; Marchand, L.; Thivolet, C.; Rome, S. Diagnostic Value of Cell-free Circulating MicroRNAs for Obesity and Type 2 Diabetes: A Meta-analysis. J. Mol. Biomark. Diagn. 2015, 6, 251. [Google Scholar] [CrossRef]
- Giardina, S.; Hernández-Alonso, P.; Salas-Salvadó, J.; Rabassa-Soler, A.; Bulló, M. Modulation of Human Subcutaneous Adipose Tissue MicroRNA Profile Associated with Changes in Adiposity-Related Parameters. Mol. Nutr. Food Res. 2018, 62, 1700594. [Google Scholar] [CrossRef]
- Wang, H.; Shao, Y.; Yuan, F.; Feng, H.; Li, N.; Zhang, H.; Wu, C.; Liu, Z. Fish Oil Feeding Modulates the Expression of Hepatic MicroRNAs in a Western-Style Diet-Induced Nonalcoholic Fatty Liver Disease Rat Model. BioMed Res. Int. 2017, 2017, 2503847. [Google Scholar] [CrossRef]
- Min, H.K.; Kapoor, A.; Fuchs, M.; Mirshahi, F.; Zhou, H.; Maher, J.; Kellum, J.; Warnick, R.; Contos, M.J.; Sanyal, A.J. Increased hepatic synthesis and dysregulation of cholesterol metabolism is associated with the severity of nonalcoholic fatty liver disease. Cell Metab. 2012, 15, 665–674. [Google Scholar] [CrossRef]
- Marques-Rocha, J.L.; Garcia-Lacarte, M.; Samblas, M.; Bressan, J.; Martínez, J.A.; Milagro, F.I. Regulatory roles of miR-155 and let-7b on the expression of inflammation-related genes in THP-1 cells: Effects of fatty acids. J. Physiol. Biochem. 2018, 74, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Hu, J.; Wang, X.; Zhao, X.; Li, Z.; Niu, J.; Steer, C.J.; Zheng, G.; Song, G. MicroRNA-378 promotes hepatic inflammation and fibrosis via modulation of the NF-kB-TNF-α pathway. J. Hepatol. 2019, 70, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhao, X.; Steer, C.J.; Yan, G.; Song, G. A negative feedback loop between microRNA-378 and Nrf1 promotes the development of hepatosteatosis in mice treated with a high fat diet. Metabolism. 2018, 85, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Camera, D.M.; Ong, J.N.; Coffey, V.G.; Hawley, J.A. Selective Modulation of MicroRNA Expression with Protein Ingestion Following Concurrent Resistance and Endurance Exercise in Human Skeletal Muscle. Front. Physiol. 2016, 7, 87. [Google Scholar] [CrossRef]
- Meale, S.J.; Beauchemin, K.A.; Hristov, A.N.; Chaves, A.V.; McAllister, T.A. Board-invited review: Opportunities and challenges in using exogenous enzymes to improve ruminant production. J. Anim. Sci. 2014, 92, 427–442. [Google Scholar] [CrossRef]
- Romao, J.M.; Jin, W.; He, M.; McAllister, T.; Guan, L.L. Altered MicroRNA Expression in Bovine Subcutaneous and Visceral Adipose Tissues from Cattle under Different Diet. PLoS ONE 2012, 7, e40605. [Google Scholar] [CrossRef]
- Wang, H.; Zhong, J.; Chai, Z.; Zhu, J.; Xin, J. Comparative expression profile of microRNAs and piRNAs in three ruminant species testes using next-generation sequencing. Reprod. Domest. Anim. 2018, 53, 963–970. [Google Scholar] [CrossRef]
- Casas-Agustench, P.; Fernandes, F.S.; Tavares do Carmo, M.G.; Visioli, F.; Herrera, E.; Dávalos, A. Consumption of Distinct Dietary Lipids during Early Pregnancy Differentially Modulates the Expression of microRNAs in Mothers and Offspring. PLoS ONE 2015, 10, e0117858. [Google Scholar] [CrossRef]
- Luceri, C.; Bigagli, E.; Pitozzi, V.; Giovannelli, L. A nutrigenomics approach for the study of anti-aging interventions: Olive oil phenols and the modulation of gene and microRNA expression profiles in mouse brain. Eur. J. Nutr. 2017, 56, 865–877. [Google Scholar] [CrossRef]
- Ma, H.; Hostuttler, M.; Wei, H.; Rexroad, C.E., III; Yao, J. Characterization of the Rainbow Trout Egg MicroRNA Transcriptome. PLoS ONE 2012, 7, e39649. [Google Scholar]
- Miranda, K.; Mehrpouya-Bahrami, P.; Nagarkatti, P.S.; Nagarkatti, M. Cannabinoid Receptor 1 Blockade Attenuates Obesity and Adipose Tissue Type 1 Inflammation Through miR-30e-5p Regulation of Delta-Like-4 in Macrophages and Consequently Downregulation of Th1 Cells. Front. Immunol. 2019, 10, 1049. [Google Scholar] [CrossRef]
- Miranda, K.; Yang, X.; Bam, M.; Murphy, E.A.; Nagarkatti, P.S.; Nagarkatti, M. MicroRNA-30 modulates metabolic inflammation by regulating Notch signaling in adipose tissue macrophages. Int. J. Obes. 2018, 42, 1140–1150. [Google Scholar] [CrossRef] [PubMed]
- Pahlavani, M.; Wijayatunga, N.N.; Kalupahana, N.S.; Ramalingam, L.; Gunaratne, P.H.; Coarfa, C.; Rajapakshe, K.; Kottapalli, P.; Moustaid-Moussa, N. Transcriptomic and microRNA analyses of gene networks regulated by eicosapentaenoic acid in brown adipose tissue of diet-induced obese mice. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2018, 1863, 1523–1531. [Google Scholar] [CrossRef] [PubMed]
- Pomar, C.A.; Castro, H.; Picó, C.; Palou, A.; Sánchez, J. Maternal Overfeeding during Lactation Impairs the Metabolic Response to Fed/Fasting Changing Conditions in the Postweaning Offspring. Mol. Nutr. Food Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Chang, H.; Chen, J.; Zhang, Q.; Xiaoping, Y.; Mantian, M. 3,6-Dihydroxyflavone regulates microRNA-34a through DNA methylation. BMC Cancer 2017, 17, 619. [Google Scholar] [CrossRef]
- Green, C.D.; Huang, Y.; Dou, X.; Yang, L.; Liu, Y.; Han, J.D.J. Impact of Dietary Interventions on Noncoding RNA Networks and mRNAs Encoding Chromatin-Related Factors. Cell Rep. 2017, 18, 2957–2968. [Google Scholar] [CrossRef]
- Lovis, P.; Roggli, E.; Laybutt, D.R.; Gattesco, S.; Yang, J.Y.; Widmann, C.; Abderrahmani, A.; Regazzi, R. Alterations in microRNA expression contribute to fatty acid-induced pancreatic beta-cell dysfunction. Diabetes 2008, 57, 2728–2736. [Google Scholar] [CrossRef]
- Roggli, E.; Britan, A.; Gattesco, S.; Lin-Marg, N.; Abderrahmani, A.; Meda, P.; Regazzi, R. Involvement of microRNAs in the cytotoxic effects exerted by proinflammatory cytokines on pancreatic beta-cells. Diabetes 2010, 59, 978–986. [Google Scholar] [CrossRef]
- Yin, H.; Pasut, A.; Soleimani, V.D.; Bentzinger, C.F.; Antoun, G.; Thorn, S.; Seale, P.; Fernando, P.; van Ijcken, W.; Grosveld, F.; et al. MicroRNA-133 controls brown adipose determination in skeletal muscle satellite cells by targeting Prdm16. Cell Metab. 2013, 17, 210–224. [Google Scholar] [CrossRef]
- Eseberri, I.; Lasa, A.; Miranda, J.; Garcia, A.; Portillo, M.P. Potential miRNA involvement in the anti-adipogenic effect of resveratrol and its metabolites. PLoS ONE 2017, 12, e0184875. [Google Scholar] [CrossRef]
- Olivo-Marston, S.E.; Hursting, S.D.; Perkins, S.N.; Schetter, A.; Khan, M.; Croce, C.; Haris, C.C.; Lavigne, J. Effects of calorie restriction and diet-induced obesity on murine colon carcinogenesis, growth and inflammatory factors, and microRNA expression. PLoS ONE 2014, 9, e94765. [Google Scholar] [CrossRef] [PubMed]
- Boesch-Saadatmandi, C.; Loboda, A.; Wagner, A.E.; Stachurska, A.; Jozkowicz, A.; Dulak, J.; Döring, F.; Wolffram, S.; Rimbach, G. Effect of quercetin and its metabolites isorhamnetin and quercetin-3-glucuronide on inflammatory gene expression: Role of miR-155. J. Nutr. Biochem. 2011, 22, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Croucher, N.J.; Thompson, C.M.; Trzciński, K.; Hanage, W.P.; Lipsitch, M. Identification of pneumococcal colonization determinants in the stringent response pathway facilitated by genomic diversity. BMC Genom. 2015, 16, 369. [Google Scholar] [CrossRef] [PubMed]
- Jeon, T.I.; Park, J.W.; Ahn, J.; Jung, C.H.; Ha, T.Y. Fisetin protects against hepatosteatosis in mice by inhibiting miR-378. Mol. Nutr. Food Res. 2013, 57, 1931–1937. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Okla, M.; Erickson, A.; Carr, T.; Natarajan, S.K.; Chung, S. Eicosapentaenoic Acid Potentiates Brown Thermogenesis through FFAR4-dependent Up-regulation of miR-30b and miR-378. J. Biol. Chem. 2016, 291, 20551–20562. [Google Scholar] [CrossRef]
- Thiele, R.; Mueller-Seitz, E.; Petz, M. Chili pepper fruits: Presumed precursors of fatty acids characteristic for capsaicinoids. J. Agric. Food Chem. 2008, 56, 4219–4224. [Google Scholar] [CrossRef]
- Luo, X.J.; Peng, J.; Li, Y.J. Recent advances in the study on capsaicinoids and capsinoids. Eur. J. Pharmacol. 2011, 650, 1–7. [Google Scholar] [CrossRef]
- El Hadi, H.; Di Vicenzo, A.; Vettor, R.; Rossato, M. Food Ingredients Involved in White-to-Brown Adipose Tissue Conversion and in Calorie Burning. Front. Physiol. 2018, 9, 1954. [Google Scholar] [CrossRef]
- Yoneshiro, T.; Aita, S.; Kawai, Y.; Iwanaga, T.; Saito, M. Nonpungent capsaicin analogs (capsinoids) increase energy expenditure through the activation of brown adipose tissue in humans. Am. J. Clin. Nutr. 2012, 95, 845–850. [Google Scholar] [CrossRef]
- Bonet, M.L.; Oliver, P.; Palou, A. Pharmacological and nutritional agents promoting browning of white adipose tissue. Biochim. Biophys. Acta 2013, 1831, 969–985. [Google Scholar] [CrossRef]
- Okla, M.; Kim, J.; Koehler, K.; Chung, S. Dietary Factors Promoting Brown and Beige Fat Development and Thermogenesis. Adv. Nutr. 2017, 8, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Baboota, R.K.; Murtaza, N.; Jagtap, S.; Singh, D.P.; Karmase, A.; Kaur, J.; Bhutani, K.K.; Bishnoi, M. Capsaicin-induced transcriptional changes in hypothalamus and alterations in gut microbial count in high fat diet fed mice. J. Nutr. Biochem. 2014, 5, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Baboota, R.K.; Singh, D.P.; Sarma, S.M.; Kaur, J.; Sandhir, R.; Boparai, R.K.; Kondepudi, K.K.; Bishnoi, M. Capsaicin induces “brite” phenotype in differentiating 3T3-L1 preadipocytes. PLoS ONE 2014, 9, e103093. [Google Scholar] [CrossRef] [PubMed]
- Baskaran, P.; Krishnan, V.; Ren, J.; Thyagarajan, B. Capsaicin induces browning of white adipose tissue and counters obesity by activating TRPV1 channel-dependent mechanisms. Br. J. Pharmacol. 2016, 173, 2369–2389. [Google Scholar] [CrossRef]
- Mosqueda-Solís, A.; Sánchez, J.; Portillo, M.P.; Palou, A.; Picó, C. Combination of Capsaicin and Hesperidin Reduces the Effectiveness of Each Compound to Decrease the Adipocyte Size and To Induce Browning Features in Adipose Tissue of Western Diet Fed Rats. J. Agric. Food Chem. 2018, 66, 9679–9689. [Google Scholar] [CrossRef]
- Fan, L.; Xu, H.; Yang, R.; Zang, Y.; Chen, J.; Qin, H. Combination of Capsaicin and Capsiate Induces Browning in 3T3-L1 White Adipocytes via Activation of the Peroxisome Proliferator-Activated Receptor gamma/beta3-Adrenergic Receptor Signaling Pathways. J. Agric. Food Chem. 2019, 67, 6232–6240. [Google Scholar] [CrossRef]
- Ohyama, K.; Nogusa, Y.; Shinoda, K.; Suzuki, K.; Bannai, M.; Kajimura, S. A Synergistic Antiobesity Effect by a Combination of Capsinoids and Cold Temperature Through Promoting Beige Adipocyte Biogenesis. Diabetes 2016, 65, 1410–1423. [Google Scholar] [CrossRef]
- Rohm, B.; Holik, A.K.; Kretschy, N.; Somoza, M.M.; Ley, J.P.; Widder, S.; Krammer, G.E.; Marko, D.; Somoza, V. Nonivamide enhances miRNA let-7d expression and decreases adipogenesis PPARγ expression in 3T3-L1 cells. J. Cell. Biochem. 2015, 116, 1153–1163. [Google Scholar] [CrossRef]
- Shishodia, S.; Sethi, G.; Aggarwal, B.B. Curcumin: Getting back to the roots. Ann. N. Y. Acad. Sci. 2005, 1056, 206–217. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Sundaram, C.; Malani, N.; Ichikawa, H. CURCUMIN: THE INDIAN SOLID GOLD. In Advances in Experimental Medicine and Biology; Aggarwal, B.B., Surh, Y.J., Shishodia, S., Eds.; Springer: Boston, MA, USA, 2007; Volume 595, ISBN 978-0-387-46400-8. [Google Scholar]
- Tønnesen, H.H.; Másson, M.; Loftsson, T. Studies of curcumin and curcuminoids. XXVII. Cyclodextrin complexation: Solubility, chemical and photochemical stability. Int. J. Pharm. 2002, 244, 127–135. [Google Scholar] [CrossRef]
- Yang, K.Y.; Lin, L.C.; Tseng, T.Y.; Wang, S.C.; Tsai, T.H. Oral bioavailability of curcumin in rat and the herbal analysis from Curcuma longa by LC-MS/MS. J. Chromatogr. B Analyt. Technol. Biomed. Life. Sci. 2007, 853, 183–189. [Google Scholar] [CrossRef]
- Wang, S.; Wang, X.; Ye, Z.; Xu, C.; Zhang, M.; Ruan, B.; Wei, M.; Jiang, Y.; Zhang, Y.; Wang, L.; et al. Curcumin promotes browning of white adipose tissue in a norepinephrine-dependent way. Biochem. Biophys. Res. Commun. 2015, 466, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, S.; Kamiya, M.; Aoyama, H.; Nomura, M.; Hyodo, T.; Ozeki, A.; Lee, H.; Takahashi, T.; Imaizumi, A.; Tsuda, T. Highly Dispersible and Bioavailable Curcumin but not Native Curcumin Induces Brown-Like Adipocyte Formation in Mice. Mol. Nutr. Food Res. 2018, 62, 1700731. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Revelo, X.; Shao, W.; Tian, L.; Zeng, K.; Lei, H.; Sun, H.S.; Woo, M.; Winer, D.; Jin, T. Dietary Curcumin Intervention Targets Mouse White Adipose Tissue Inflammation and Brown Adipose Tissue UCP1 Expression. Obesity 2018, 26, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Zhang, S.; Liu, C.; Liu, X. Curcumin promoted miR-34a expression and suppressed proliferation of gastric cancer cells. Cancer Biother. Radiopharm. 2019. [Google Scholar] [CrossRef]
- Kim, S.W.; Choi, J.H.; Mukherjee, R.; Hwang, K.C.; Yun, J.W. Proteomic identification of fat-browning markers in cultured white adipocytes treated with curcumin. Mol. Cell. Biochem. 2016, 415, 51–66. [Google Scholar] [CrossRef]
- Lone, J.; Choi, J.H.; Kim, S.W.; Yun, J.W. Curcumin induces brown fat-like phenotype in 3T3-L1 and primary white adipocytes. J. Nutr. Biochem. 2016, 27, 193–202. [Google Scholar] [CrossRef]
- Marchese, A.; Orhan, I.E.; Daglia, M.; Barbieri, R.; Di Lorenzo, A.; Nabavi, S.F.; Gortzi, O.; Izadi, M.; Nabavi, S.M. Antibacterial and antifungal activities of thymol: A brief review of the literature. Food Chem. 2016, 210, 402–414. [Google Scholar] [CrossRef]
- Salehi, B.; Mishra, A.P.; Shukla, I.; Sharifi-Rad, M.; Contreras, M.D.M.; Segura-Carretero, A.; Fathi, H.; Nasrabadi, N.N.; Kobarfard, F.; Sharifi-Rad, J. Thymol, thyme, and other plant sources: Health and potential uses. Phytother. Res. 2018, 32, 1688–1706. [Google Scholar] [CrossRef]
- Choi, J.H.; Kim, S.W.; Yu, R.; Yun, J.W. Monoterpene phenolic compound thymol promotes browning of 3T3-L1 adipocytes. Eur. J. Nutr. 2017, 56, 2329–2341. [Google Scholar] [CrossRef]
- De la Garza, A.L.; Milagro, F.I.; Boque, N.; Campión, J.; Martínez, J.A. Natural inhibitors of pancreatic lipase as new players in obesity treatment. Planta Med. 2011, 77, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.H.; Mukherjee, S.; Yun, J.W. Trans-Cinnamic Acid Stimulates White Fat Browning and Activates Brown Adipocytes. Nutrients 2019, 11, 577. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Yuan, H.D.; Kim, D.Y.; Quan, H.Y.; Chung, S.H. Cinnamaldehyde Prevents Adipocyte Differentiation and Adipogenesis via Regulation of Peroxisome Proliferator-Activated Receptor-γ (PPARγ) and AMP-Activated Protein Kinase (AMPK) Pathways. J. Agric. Food Chem. 2011, 59, 3666–3673. [Google Scholar] [CrossRef] [PubMed]
- Mnafgui, K.; Derbali, A.; Sayadi, S.; Gharsallah, N.; Elfeki, A.; Allouche, N. Anti-obesity and cardioprotective effects of cinnamic acid in high fat diet- induced obese rats. J. Food Sci. Technol. 2015, 52, 4369–4377. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Cao, Y.; Xiao, J.; Song, M.; Ho, C.T. Molecular mechanisms of the anti-obesity effect of bioactive ingredients in common spices: A review. Food Funct. 2018, 9, 4569–4581. [Google Scholar] [CrossRef]
- Zuo, J.; Zhao, D.; Yu, N.; Fang, X.; Mu, Q.; Ma, Y.; Mo, F.; Wu, R.; Ma, R.; Wang, L.; et al. Cinnamaldehyde Ameliorates Diet-Induced Obesity in Mice by Inducing Browning of White Adipose Tissue. Cell. Physiol. Biochem. 2017, 42, 1514–1525. [Google Scholar] [CrossRef]
- Qu, S.; Shen, Y.; Wang, M.; Wang, X.; Yang, Y. Suppression of miR-21 and miR-155 of macrophage by cinnamaldehyde ameliorates ulcerative colitis. Int. Immunopharmacol. 2019, 67, 22–34. [Google Scholar] [CrossRef]
- Chen, Y.; Siegel, F.; Kipschull, S.; Haas, B.; Fröhlich, H.; Meister, G.; Pfeifer, A. miR-155 regulates differentiation of brown and beige adipocytes via a bistable circuit. Nat. Commun. 2013, 4, 1769. [Google Scholar] [CrossRef]
- Shang, A.; Cao, S.Y.; Xu, X.Y.; Gan, R.Y.; Tang, G.Y.; Corke, H.; Mavumengwana, V.; Li, H.B. Bioactive Compounds and Biological Functions of Garlic (Allium sativum L.). Foods Basel Switz. 2019, 8, 246. [Google Scholar] [CrossRef]
- Ambati, S.; Yang, J.Y.; Rayalam, S.; Park, H.J.; Della-Fera, M.A.; Baile, C.A. Ajoene exerts potent effects in 3T3-L1 adipocytes by inhibiting adipogenesis and inducing apoptosis. Phytother. Res. 2009, 23, 513–518. [Google Scholar] [CrossRef]
- Kim, E.J.; Lee, D.H.; Kim, H.J.; Lee, S.J.; Ban, J.O.; Cho, M.C.; Jeong, H.S.; Yang, Y.; Hong, J.T.; Yoon, D.Y. Thiacremonone, a sulfur compound isolated from garlic, attenuates lipid accumulation partially mediated via AMPK activation in 3T3-L1 adipocytes. J. Nutr. Biochem. 2012, 23, 1552–1558. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Kim, H. Effect of garlic on high fat induced obesity. Acta Biol. Hung. 2011, 62, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Torres-Villarreal, D.; Camacho, A.; Milagro, F.I.; Ortiz-Lopez, R.; de la Garza, A.L. Quercetin-3-O-glucoside improves glucose tolerance in rats and decreases intestinal sugar uptake in caco-2 cells. Nat. Prod. Commun. 2017, 12, 1709–1712. [Google Scholar] [CrossRef]
- Peñarrieta, M.; Tejeda, L.; Mollinedo, P.; Vila, J.; Bravo, J. Phenolic Compounds in Food. Rev. Boliv. Quím. 2014, 31, 68–81. [Google Scholar]
- Lee, S.G.; Parks, J.S.; Kang, H.W. Quercetin, a functional compound of onion peel, remodels white adipocytes to brown-like adipocytes. J. Nutr. Biochem. 2017, 42, 62–71. [Google Scholar] [CrossRef]
- Kuipers, E.N.; Dam, A.D.V.; Held, N.M.; Mol, I.M.; Houtkooper, R.H.; Rensen, P.C.N.; Boom, M.R. Quercetin Lowers Plasma Triglycerides Accompanied by White Adipose Tissue Browning in Diet-Induced Obese Mice. Int. J. Mol. Sci. 2018, 19, 1786. [Google Scholar] [CrossRef]
- Akbari Kordkheyli, V.; Khonakdar Tarsi, A.; Mishan, M.A.; Tafazoli, A.; Bardania, H.; Zarpou, S.; Bagheri, A. Effects of quercetin on microRNAs: A mechanistic review. J. Cell. Biochem. 2019, 120, 12141–12155. [Google Scholar] [CrossRef]
- Boesch-Saadatmandi, C.; Wagner, A.E.; Wolffram, S.; Rimbach, G. Effect of quercetin on inflammatory gene expression in mice liver in vivo—Role of redox factor 1, miRNA-122 and miRNA-125b. Pharmacol. Res. 2012, 65, 523–530. [Google Scholar] [CrossRef]
- Arias, N.; Aguirre, L.; Fernández-Quintela, A.; González, M.; Lasa, A.; Miranda, J.; Macarulla, M.T.; Portillo, M.P. MicroRNAs involved in the browning process of adipocyte. J. Physiol. Biochem. 2016, 72, 509–521. [Google Scholar] [CrossRef]
- Galleggiante, V.; De Santis, S.; Liso, M.; Verna, G.; Sommella, E.; Mastronardi, M.; Campiglia, P.; Chieppa, M.; Serrino, G. Quercetin-Induced miR-369-3p Suppresses Chronic Inflammatory Response Targeting C/EBP-β. Mol. Nutr. Food Res. 2019, 63, 1801390. [Google Scholar] [CrossRef]
- Ranaware, A.M.; Banik, K.; Deshpande, V.; Padmavathi, G.; Roy, N.K.; Sethi, G.; Fan, L.; Kumar, A.P.; Kunnumakkara, A.B. Magnolol: A Neolignan from the Magnolia Family for the Prevention and Treatment of Cancer. Int. J. Mol. Sci. 2018, 19, 2362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, Z.; Huang, X.; Shi, W.; Zhang, R.; Chen, M.; Huang, H.; Wu, L. Insights on the Multifunctional Activities of Magnolol. BioMed Res. Int. 2019, 2019, 1847130. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Choi, M.S.; Cha, B.Y.; Woo, J.T.; Park, Y.B.; Kim, S.R.; Jung, U.J. Long-term supplementation of honokiol and magnolol ameliorates body fat accumulation, insulin resistance, and adipose inflammation in high-fat fed mice. Mol. Nutr. Food Res. 2013, 57, 1988–1998. [Google Scholar] [CrossRef] [PubMed]
- Parray, H.A.; Lone, J.; Park, J.P.; Choi, J.W.; Yun, J.W. Magnolol promotes thermogenesis and attenuates oxidative stress in 3T3-L1 adipocytes. Nutrition 2018, 50, 82–90. [Google Scholar] [CrossRef]
- Talarek, S.; Listos, J.; Barreca, D.; Tellone, E.; Sureda, A.; Nabavi, S.F.; Braidy, N.; Nabavi, S.M. Neuroprotective effects of honokiol: From chemistry to medicine. Biofactors 2017, 43, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Patel, S.; Imran, M.; Maalik, A.; Arshad, M.U.; Saeed, F.; Mabkhot, Y.N.; Al-Showiman, S.S.; Ahmad, N.; Elsharkawy, E. Honokiol: An anticancer lignan. Biomed. Pharmacother. 2018, 107, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Woodbury, A.; Yu, S.P.; Wei, L.; García, P. Neuro-modulating effects of honokiol: A review. Front. Neurol. 2013, 4, 130. [Google Scholar] [CrossRef] [PubMed]
- Prasad, R.; Katiyar, S.K. Honokiol, an Active Compound of Magnolia Plant, Inhibits Growth, and Progression of Cancers of Different Organs. Adv. Exp. Med. Biol. 2016, 928, 245–265. [Google Scholar]
- Lone, J.; Yun, J.W. Honokiol exerts dual effects on browning and apoptosis of adipocytes. Pharmacol. Rep. 2017, 69, 1357–1365. [Google Scholar] [CrossRef]
- Ross, S.R.; Choy, L.; Graves, R.A.; Fox, N.; Solevjeva, V.; Klaus, S.; Ricquier, D.; Spiegelman, B.M. Hibernoma formation in transgenic mice and isolation of a brown adipocyte cell line expressing the uncoupling protein gene. Proc. Natl. Acad. Sci. USA 1992, 89, 7561–7565. [Google Scholar] [CrossRef]
- Hagiwara, K.; Gailhouste, L.; Yasukawa, K.; Kosaka, N.; Ochiya, T. A robust screening method for dietary agents that activate tumour-suppressor microRNAs. Sci. Rep. 2015, 5, 14697. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zhang, Y.; Xue, Y.; Zhang, Z.; Wang, J. Berberine is a potential therapeutic agent for metabolic syndrome via brown adipose tissue activation and metabolism regulation. Am. J. Transl. Res. 2018, 10, 3322–3329. [Google Scholar] [PubMed]
- Zhang, Y.; Li, X.; Zou, D.; Liu, W.; Yang, J.; Zhu, N.; Huo, L.; Wang, M.; Hong, J.; Wu, P.; et al. Treatment of type 2 diabetes and dyslipidemia with the natural plant alkaloid berberine. J. Clin. Endocrinol. Metab. 2008, 93, 2559–2565. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Davies, G.E. Berberine inhibits adipogenesis in high-fat diet-induced obesity mice. Fitoterapia 2010, 81, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Fahmy, H.; Zjawiony, J.K.; Davies, G.E. Inhibitory effect and transcriptional impact of berberine and evodiamine on human white preadipocyte differentiation. Fitoterapia 2010, 81, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Ehli, E.A.; Kittelsrud, J.; Ronan, J.; Munger, K.; Downey, T.; Bohlen, K.; Callahan, L.; Munson, V.; Jahnke, M.; et al. Lipid-lowering effect of berberine in human subjects and rats. Phytomedicine 2012, 19, 861–867. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y.; Zhang, M.; Pang, X.; Xu, J.; Kang, C.; Li, M.; Zhang, C.; Zhang, Z.; Li, X.; et al. Structural changes of gut microbiota during berberine-mediated prevention of obesity and insulin resistance in high-fat diet-fed rats. PLoS ONE 2012, 7, e42529. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, H.; Li, B.; Meng, X.; Wang, J.; Zhang, Y.; Yao, S.; Ma, Q.; Jin, L.; Yang, J.; et al. Berberine activates thermogenesis in white and brown adipose tissue. Nat. Commun. 2014, 5, 5493. [Google Scholar] [CrossRef]
- Lin, Y.C.; Lee, Y.C.; Lin, Y.J.; Lin, J.C. Berberine Promotes Beige Adipogenic Signatures of 3T3-L1 Cells by Regulating Post-transcriptional Events. Cells 2019, 8, 632. [Google Scholar] [CrossRef]
- Lin, J.; Cai, Q.; Liang, B.; Wu, L.; Zhuang, Y.; He, Y.; Lin, W. Berberine, a Traditional Chinese Medicine, Reduces Inflammation in Adipose Tissue, Polarizes M2 Macrophages, and Increases Energy Expenditure in Mice Fed a High-Fat Diet. Med. Sci. Monit. 2019, 25, 87–97. [Google Scholar] [CrossRef]
- Wu, L.; Xia, M.; Duan, Y.; Zhang, L.; Jiang, H.; Hu, X.; Yan, H.; Gu, Y.; Shi, H.; Li, J.; et al. Berberine promotes the recruitment and activation of brown adipose tissue in mice and humans. Cell Death Dis. 2019, 10, 468. [Google Scholar] [CrossRef] [PubMed]
- Sanada, S.; Kondo, N.; Shoji, J.; Tanaka, O.; Shibata, S. Studies on the saponins of ginseng. II. Structures of ginsenoside-Re, -Rf and -Rg2. Chem. Pharm. Bull. (Tokyo) 2008, 22, 2407–2412. [Google Scholar] [CrossRef]
- Hong, H.D.; Choi, S.Y.; Kim, Y.C.; Lee, Y.C.; Cho, C.W. Rapid determination of ginsenosides Rb 1, Rf, and Rg 1 in Korean ginseng using HPLC. J. Ginseng Res. 2009, 33, 8–12. [Google Scholar]
- Washida, D.; Kitanaka, S. Determination of polyacetylenes and ginsenosides in Panax species using high performance liquid chromatography. Chem. Pharm. Bull. (Tokyo) 2003, 51, 1314–1317. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.; Gavrilova, O.; Pack, S.; Jou, W.; Mullen, S.; Summer, A.E.; Cushman, S.W.; Periwal, V. Hypertrophy and/or hyperplasia: Dynamics of adipose tissue growth. PLoS Comput. Biol. 2009, 5, e1000324. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Cha, B.Y.; Yamaguchi, K.; Choi, T.; Yonezawa, T.; Teruya, T.; Nagai, K.; Woo, J.T. Effects of Korean white ginseng extracts on obesity in high-fat diet-induced obese mice. Cytotechnology 2010, 62, 367–376. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Koh, E.J.; Kim, K.J.; Choi, J.; Jeon, H.J.; Seo, M.J.; Lee, B.Y. Ginsenoside Rg1 suppresses early stage of adipocyte development via activation of C/EBP homologous protein-10 in 3T3-L1 and attenuates fat accumulation in high fat diet-induced obese zebrafish. J. Ginseng Res. 2017, 41, 23–30. [Google Scholar] [CrossRef]
- Bijland, S.; Mancini, S.J.; Salt, I.P. Role of AMP-activated protein kinase in adipose tissue metabolism and inflammation. Clin. Sci. 2013, 124, 491–507. [Google Scholar] [CrossRef]
- Wang, S.; Liang, X.; Yang, Q.; Fu, X.; Rogers, C.J.; Zhu, M.; Rodgers, B.D.; Jiang, Q.; Dodson, M.V.; Du, M. Resveratrol induces brown-like adipocyte formation in white fat through activation of AMP-activated protein kinase (AMPK) alpha1. Int. J. Obes. 2015, 39, 967–976. [Google Scholar] [CrossRef]
- Lee, K.; Seo, Y.J.; Song, J.H.; Lee, B.Y. Ginsenoside Rg1 promotes browning by inducing UCP1 expression and mitochondrial activity in 3T3-L1 and subcutaneous white adipocytes. J. Ginseng Res. 2018. [Google Scholar] [CrossRef]
- Gu, X. Ginsenoside Rb2 Alleviates Obesity by Activation of Brown Fat and Induction of Browning of White Fat. Diabetes 2019, 68. [Google Scholar] [CrossRef]
- Hong, Y.; Lin, Y.; Si, Q.; Yang, L.; Dong, W.; Gu, X. Ginsenoside Rb2 Alleviates Obesity by Activation of Brown Fat and Induction of Browning of White Fat. Front. Endocrinol. 2019, 10, 153. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.S.; Yue, P.Y.; Kok, T.W.; Keung, M.H.; Mak, N.K.; Wong, R.N. Ginsenoside-Rb1 promotes adipogenesis through regulation of PPARγ and microRNA-27b. Horm. Metab. Res. 2012, 44, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, B.M. Mint: The Genus Mentha, Medicinal and Aromatic Plants—Industrial Profiles, 1st ed.; CRC Press: Boca Raton, FL, USA, 2006; ISBN 13:978-0-8493-0798-0. [Google Scholar]
- Kim, S.; Lee, J.Y. Menthol attenuates the magnitude of cold-induced vasodilation on the extremities of young females. J. Physiol. Anthropol. 2018, 37, 14. [Google Scholar] [CrossRef] [PubMed]
- Blanquart, S.; Borowiec, A.S.; Delcourt, P.; Figeac, M.; Emerling, C.A.; Meseguer, A.S.; Roudbaraki, M.; Prevarskaya, N.; Bidaux, G. Evolution of the human cold/menthol receptor, TRPM8. Mol. Phylogenet. Evol. 2019, 136, 104–118. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Yu, H.; Zhao, Z.; Luo, Z.; Chen, J.; Ni, Y.; Jin, R.; Ma, L.; Wang, P.; Zhu, Z.; et al. Activation of the cold-sensing TRPM8 channel triggers UCP1-dependent thermogenesis and prevents obesity. J. Mol. Cell Biol. 2012, 4, 88–96. [Google Scholar] [CrossRef]
- Khare, P.; Mangal, P.; Baboota, R.K.; Jagtap, S.; Kumar, V.; Singh, D.P.; Boparai, R.K.; Sharma, S.S.; Khardori, R.; Bhadada, S.K.; et al. Involvement of glucagon in preventive effect of menthol against high fat diet induced obesity in mice. Front. Pharmacol. 2018, 9, 1244. [Google Scholar] [CrossRef]
- Rossato, M.; Granzotto, M.; Macchi, V.; Porzionato, A.; Petrelli, L.; Calcagno, A.; Vencato, J.; De Stefani, D.; Silvestrin, V.; Rizzuto, R.; et al. Human white adipocytes express the cold receptor TRPM8 which activation induces UCP1 expression, mitochondrial activation and heat production. Mol. Cell. Endocrinol. 2014, 383, 137–146. [Google Scholar] [CrossRef]
- Goralczyk, A.; van Vijven, M.; Koch, M.; Badowski, C.; Yassin, M.S.; Toh, S.A.; Shabbir, A.; Franco-Obregón, A.; Raghunath, M. TRP channels in brown and white adipogenesis from human progenitors: New therapeutic targets and the caveats associated with the common antibiotic, streptomycin. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2017, 31, 3251–3266. [Google Scholar] [CrossRef]
- Khare, P.; Chauhan, A.; Kumar, V.; Kaur, J.; Mahajan, N.; Kumar, V.; Gesing, A.; Chopra, K.; Kondepudi, K.K.; Bishnoi, M. Bioavailable Menthol (Transient Receptor Potential Melastatin-8 Agonist) Induces Energy Expending Phenotype in Differentiating Adipocytes. Cells 2019, 8, E383. [Google Scholar] [CrossRef]
- Jiang, C.; Zhai, M.; Yan, D.; Li, D.; Li, C.; Zhang, Y.; Xiao, L.; Xiong, D.; Deng, Q.; Sun, W. Dietary menthol-induced TRPM8 activation enhances WAT “browning” and ameliorates diet-induced obesity. Oncotarget 2017, 8, 75114–75126. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.F.; Braidy, N.; Habtemariam, S.; Orhan, I.E.; Daglia, M.; Manayi, A.; Gortzi, O.; Nabavi, S.M. Neuroprotective effects of chrysin: From chemistry to medicine. Neurochem. Int. 2015, 90, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Mani, R.; Natesan, V. Chrysin: Sources, beneficial pharmacological activities, and molecular mechanism of action. Phytochemistry 2018, 145, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Farkhondeh, T.; Samarghandian, S.; Roshanravan, B. Impact of chrysin on the molecular mechanisms underlying diabetic complications. J. Cell. Physiol. 2019, 234, 17144–17158. [Google Scholar] [CrossRef]
- Choi, J.H.; Yun, J.W. Chrysin induces brown fat-like phenotype and enhances lipid metabolism in 3T3-L1 adipocytes. Nutrition 2016, 32, 1002–1010. [Google Scholar] [CrossRef]
- Ahn-Jarvis, J.H.; Riedl, K.M.; Schwartz, S.J.; Vodovotz, Y. Design and selection of soy breads used for evaluating isoflavone bioavailability in clinical trials. J. Agric. Food Chem. 2013, 61, 3111–3120. [Google Scholar] [CrossRef]
- Savini, I.; Catani, M.V.; Evangelista, D.; Gasperi, V.; Avigliano, L. Obesity-associated oxidative stress: Strategies finalized to improve redox state. Int. J. Mol. Sci. 2013, 14, 10497–10538. [Google Scholar] [CrossRef]
- Lephart, E.D.; Porter, J.P.; Lund, T.D.; Bu, L.; Setchell, K.D.; Ramoz, G.; Crowley, W.R. Dietary isoflavones alter regulatory behaviors, metabolic hormones and neuroendocrine function in Long-Evans male rats. Nutr. Metab. 2004, 1, 16. [Google Scholar] [CrossRef]
- Aziz, S.A.; Walkening, L.A.; Miwa, S.; Alberdi, G.; Hesketh, J.E.; Ford, D. Metabolic programming of a beige adipocyte phenotype by genistein. Mol. Nutr. Food Res. 2017, 61, 1600574. [Google Scholar] [CrossRef]
- Zhou, L.; Xiao, X.; Zhang, Q.; Zheng, J.; Li, M.; Deng, M. A Possible Mechanism: Genistein Improves Metabolism and Induces White Fat Browning Through Modulating Hypothalamic Expression of Ucn3, Depp, and Stc1. Front. Endocrinol. 2019, 10, 478. [Google Scholar] [CrossRef]
- Crespillo, A.; Alonso, M.; Vida, M.; Pavón, F.J.; Serrano, A.; Rivera, P.; Romero-Zerbo, Y.; Fernández-Llebrez, P.; Martínez, A.; Pérez-Valero, V.; et al. Reduction of body weight, liver steatosis and expression of stearoyl-CoA desaturase 1 by the isoflavone daidzein in diet-induced obesity. Br. J. Pharmacol. 2011, 164, 1899–1915. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhao, Z.; Pang, X.; Yang, J.; Yu, H.; Zhang, Y.; Zhou, H.; Zhao, J. Genistein Protects Against Ox-LDL-Induced Inflammation Through MicroRNA-155/SOCS1-Mediated Repression of NF-ĸB Signaling Pathway in HUVECs. Inflammation 2017, 40, 1450–1459. [Google Scholar] [CrossRef] [PubMed]
- De la Parra, C.; Castillo-Pichardo, L.; Cruz-Collazo, A.; Cubano, L.; Redis, R.; Calin, G.A.; Dharmawardhane, S. Soy Isoflavone Genistein-Mediated Downregulation of miR-155 Contributes to the Anticancer Effects of Genistein. Nutr. Cancer 2016, 68, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Thielecke, F.; Boschmann, M. The potential role of green tea catechins in the prevention of the metabolic syndrome—A review. Phytochemistry 2009, 70, 11–24. [Google Scholar] [CrossRef]
- USDA Database for the Flavonoid Content of Selected Foods. Nutrient Data Laboratory; Food Composition Laboratory; Beltsville Human Nutrition Research Center; Nutrient Data Laboratory. Available online: http://www.ars.usda.gov/SP2UserFiles/Place/12354500/Data/Flav/Flav02-1.pdf (accessed on 20 September 2019).
- Ahmad, M.; Ahmad, I.; Chattopadhyay, D. New Look to Phytomedicine: Advancements in Herbal Products as Novel Drug Leads, 1st ed.; Academic Press: London, UK, 2019; ISBN 978-0-12-814619-4. [Google Scholar]
- Wolfram, S. Effects of green tea and EGCG on cardiovascular and metabolic health. J. Am. Coll. Nutr. 2007, 26, 373S–388S. [Google Scholar] [CrossRef] [PubMed]
- Neyrinck, A.M.; Bindels, L.B.; Geurts, L.; Van Hul, M.; Cani, P.D.; Delzenne, N.M. A polyphenolic extract from green tea leaves activates fat browning in high-fat-diet-induced obese mice. J. Nutr. Biochem. 2017, 49, 15–21. [Google Scholar] [CrossRef]
- Chen, L.H.; Chien, Y.W.; Liang, C.T.; Chan, C.H.; Fan, M.H.; Huang, H.Y. Green tea extract induces genes related to browning of white adipose tissue and limits weight-gain in high energy diet-fed rat. Food Nutr. Res. 2017, 61, 1347480. [Google Scholar] [CrossRef]
- Yamashita, Y.; Wang, L.; Wang, L.; Tanaka, Y.; Zhang, T.; Ashida, H. Oolong, black and pu-erh tea suppresses adiposity in mice via activation of AMP-activated protein kinase. Food Funct. 2014, 5, 2420–2429. [Google Scholar] [CrossRef]
- Kudo, N.; Arai, Y.; Suhara, Y.; Ishii, T.; Nakayama, T.; Osakabe, N. A Single Oral Administration of Theaflavins Increases Energy Expenditure and the Expression of Metabolic Genes. PLoS ONE 2015, 10, e0137809. [Google Scholar] [CrossRef]
- Zhu, W.; Zou, B.; Nie, R.; Zhang, Y.; Li, C.M. A-type ECG and EGCG dimers disturb the structure of 3T3-L1 cell membrane and strongly inhibits its differentiation by targeting peroxisome proliferator-activated receptor γ with miR-27 involved mechanism. J. Nutr. Biochem. 2015, 26, 1124–1135. [Google Scholar] [CrossRef]
- Yoneshiro, T.; Matsushita, M.; Hibi, M.; Tone, H.; Takeshita, M.; Yasunaga, K.; Katsuragi, Y.; Kameya, T.; Sugie, H.; Saito, M. Tea catechin and caffeine activate brown adipose tissue and increase cold-induced thermogenic capacity in humans. Am. J. Clin. Nutr. 2017, 105, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Pang, K.; Zhu, Z.; Zhu, S.; Han, L. A high dose of conjugated linoleic acid increases fatty liver and insulin resistance in lactating mice. PLoS ONE 2019, 14, e0214903. [Google Scholar] [CrossRef]
- Roura-Guiberna, A.; Hernandez-Aranda, J.; Ramirez-Flores, C.J.; Mondragon-Flores, R.; Garibay-Nieto, N.; Queipo-Garcia, G.; Laresgoiti-Servitje, E.; Soh, J.W.; Olivares-Reyes, J.A. Isomers of conjugated linoleic acid induce insulin resistance through a mechanism involving activation of protein kinase Cε in liver cells. Cell Signal. 2019, 53, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A.; Banu, J.; Rahman, M.; Causey, J.; Fernandes, G. Biological effects of conjugated linoleic acids in health and disease. J. Nutr. Biochem. 2006, 17, 789–810. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; McIntosh, M.K. Conjugated linoleic acid in humans: Regulation of adiposity and insulin sensitivity. J. Nutr. 2003, 133, 3041–3046. [Google Scholar] [CrossRef]
- Shen, W.; Chuang, C.C.; Martinez, K.; Reid, T.; Brown, J.M.; Xi, L.; Hixson, L.; Hopkins, R.; Starnes, J.; McIntosh, M. Conjugated linoleic acid reduces adiposity and increases markers of browning and inflammation in white adipose tissue of mice. J. Lipid Res. 2013, 54, 909–922. [Google Scholar] [CrossRef] [PubMed]
- McDonald, M.E.; Li, C.; Bian, H.; Smith, B.D.; Layne, M.D.; Farmer, S.R. Myocardin-related transcription factor A regulates conversion of progenitors to beige adipocytes. Cell 2015, 160, 105–118. [Google Scholar] [CrossRef]
- Barneda, D.; Planas-Iglesias, J.; Gaspar, M.L.; Mohammadyani, D.; Prasannan, S.; Dormann, D.; Han, G.S.; Jesch, S.A.; Carman, G.M.; Kagan, V.; et al. The brown adipocyte protein CIDEA promotes lipid droplet fusion via a phosphatidic acid-binding amphipathic helix. eLife 2015, 4, e07485. [Google Scholar] [CrossRef]
- Jash, S.; Benerjee, S.; Lee, M.J.; Farmer, S.R.; Puri, V. CIDEA transcriptionally regulates UCP1 for britening and thermogenesis in human fat cells. iScience 2019, 20, 73–89. [Google Scholar] [CrossRef]
- Den Hartigh, L.J. Conjugated Linoleic Acid Effects on Cancer, Obesity, and Atherosclerosis: A Review of Pre-Clinical and Human Trials with Current Perspectives. Nutrients 2019, 11, E370. [Google Scholar] [CrossRef]
- Shen, W.; Baldwin, J.; Collins, B.; Hixson, L.; Lee, K.T.; Herberg, T.; Starnes, J.; Cooney, P.; Chuang, C.C.; Hopkins, R.; et al. Low level of trans-10, cis-12 conjugated linoleic acid decreases adiposity and increases browning independent of inflammatory signaling in overweight Sv129 mice. J. Nutr. Biochem. 2015, 26, 616–625. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.; Martinez, K.; Chung, S.; LaPoint, K.; Hopkins, R.; Schmidt, S.F.; Andersen, K.; Mandrup, S.; McIntosh, M. Inflammation and insulin resistance induced by trans-10, cis-12 conjugated linoleic acid depend on intracellular calcium levels in primary cultures of human adipocytes. J. Lipid Res. 2010, 51, 1906–1917. [Google Scholar] [CrossRef] [PubMed]
- Metges, C.C.; Lehmann, L.; Boeuf, S.; Petzke, K.J.; Müller, A.; Rickert, R.; Franke, W.; Steinhart, H.; Nümberg, G.; Klaus, S. cis-9,trans-11 and trans-10,cis-12 CLA affect lipid metabolism differently in primary white and brown adipocytes of Djungarian hamsters. Lipids 2003, 38, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Bruen, R.; Fitzsimons, S.; Belton, O. miR-155 in the Resolution of Atherosclerosis. Front. Pharmacol. 2019, 10, 463. [Google Scholar] [CrossRef] [PubMed]
- Albracht-Schulte, K.; Kalupahana, N.S.; Ramalingam, L.; Wang, S.; Rahman, S.M.; Robert-McComb, J.; Moustaid-Moussa, N. Omega-3 fatty acids in obesity and metabolic syndrome: A mechanistic update. J. Nutr. Biochem. 2018, 58, 1–16. [Google Scholar] [CrossRef]
- Zhao, M.; Chen, X. Eicosapentaenoic acid promotes thermogenic and fatty acid storage capacity in mouse subcutaneous adipocytes. Biochem. Biophys. Res. Commun. 2014, 450, 1446–1451. [Google Scholar] [CrossRef]
- Laiglesia, L.M.; Lorente-Cebrián, S.; Prieto-Hontoria, P.L.; Fernández-Galilea, M.; Ribeiro, S.M.R.; Sáinz, N.; Martínez, J.A.; Moreno-Aliaga, M.J. Eicosapentaenoic acid promotes mitochondrial biogenesis and beige-like features in subcutaneous adipocytes from overweight subjects. J. Nutr. Biochem. 2016, 37, 76–82. [Google Scholar] [CrossRef]
- Bargut, T.C.; Silva-e-Silva, A.C.; Souza-Mello, V.; Mandarim-de-Lacerda, C.A.; Aguila, M.B. Mice fed fish oil diet and upregulation of brown adipose tissue thermogenic markers. Eur. J. Nutr. 2016, 55, 159–169. [Google Scholar] [CrossRef]
- Kim, M.; Goto, T.; Yu, R.; Uchida, K.; Tominaga, M.; Kano, Y.; Takahashi, N.; Kawada, T. Fish oil intake induces UCP1 upregulation in brown and white adipose tissue via the sympathetic nervous system. Sci. Rep. 2015, 5, 18013. [Google Scholar] [CrossRef]
- Fleckenstein-Elsen, M.; Dinnies, D.; Jenelik, T.; Roden, M.; Romacho, T.; Eckel, J. Eicosapentaenoic acid and arachidonic acid differentially regulate adipogenesis, acquisition of a brite phenotype and mitochondrial function in primary human adipocytes. Mol. Nutr. Food Res. 2016, 60, 2065–2075. [Google Scholar] [CrossRef]
- Mori, M.A.; Thomou, T.; Boucher, J.; Lee, K.Y.; Lallukka, S.; Kim, J.K.; Torriani, M.; Yki-Järvinen, H.; Grinspoon, S.K.; Cypress, A.M.; et al. Altered miRNA processing disrupts brown/white adipocyte determination and associates with lipodystrophy. J. Clin. Investig. 2014, 124, 3339–3351. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The DHA content of a cell membrane can have a significant influence on cellular behavior and responsiveness to signals. Ann. Nutr. Metab. 2016, 69, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Juárez-Hernández, E.; Chávez-Tapia, N.C.; Uribe, M.; Barbero-Becerra, V.J. Role of bioactive fatty acids in nonalcoholic fatty liver disease. Nutr. J. 2016, 15, 72. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.; Lu, Y.; Shou, Q.; Mao, L.; He, L.; Wang, J.; Chen, J.; Zhang, Y.; Jiao, J. Differential Anti-Adipogenic Effects of Eicosapentaenoic and Docosahexaenoic Acids in Obesity. Mol. Nutr. Food Res. 2019, e1801135. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, T.E.; Castro, É.; Belchior, T.; Andrade, M.L.; Chaves-Filho, A.B.; Peixoto, A.S.; Moreno, M.F.; Ortiz-Silva, M.; Moreira, R.J.; Inague, A.; et al. Fish Oil Protects Wild Type and Uncoupling Protein 1-Deficient Mice from Obesity and Glucose Intolerance by Increasing Energy Expenditure. Mol. Nutr. Food Res. 2019, 63, e1800813. [Google Scholar] [CrossRef] [PubMed]
- Crescenzo, R.; Mazzoli, A.; Cancelliere, R.; Bianco, F.; Giacco, A.; Liverini, G.; Dullo, A.G.; Iossa, S. Polyunsaturated fatty acids stimulate de novo lipogenesis and improve glucose homeostasis during refeeding with high fat diet. Front. Physiol. 2017, 8, 178. [Google Scholar] [CrossRef]
- Kuda, O.; Rossmeisl, M.; Kopecky, J. Omega-3 fatty acids and adipose tissue biology. Mol. Aspects Med. 2018, 64, 147–160. [Google Scholar] [CrossRef]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 652, 18–26. [Google Scholar] [CrossRef]
- Coronel, J.; Pinos, I.; Amengual, J. β-carotene in Obesity Research: Technical Considerations and Current Status of the Field. Nutrients 2019, 11, E842. [Google Scholar] [CrossRef]
- Landrier, J.F.; Marcotorchino, J.; Tourniaire, F. Lipophilic micronutrients and adipose tissue biology. Nutrients 2012, 4, 1622–1649. [Google Scholar] [CrossRef]
- Gerhard, G.S.; Styer, A.M.; Strodel, W.E.; Roesch, S.L.; Yavorek, A.; Carey, D.J.; Wood, G.C.; Petrick, A.T.; Gabrielsen, J.; Ibele, A.; et al. Gene expression profiling in subcutaneous, visceral and epigastric adipose tissues of patients with extreme obesity. Int. J. Obes. 2014, 38, 371. [Google Scholar] [CrossRef] [PubMed]
- Bonet, M.L.; Canas, J.A.; Ribot, J.; Palou, A. Carotenoids and their conversion products in the control of adipocyte function, adiposity and obesity. Arch. Biochem. Biophys. 2015, 572, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Shabalina, I.G.; Backlund, E.C.; Bar-Tana, J.; Cannon, B.; Nedergaard, J. Within brown-fat cells, UCP1-mediated fatty acid-induced uncoupling is independent of fatty acid metabolism. Biochim. Biophys. Acta 2008, 1777, 642–650. [Google Scholar] [CrossRef] [PubMed]
- Serra, F.; Bonet, M.L.; Puigserver, P.; Oliver, J.; Palou, A. Stimulation of uncoupling protein 1 expression in brown adipocytes by naturally occurring carotenoids. Int. J. Obes. Relat. Metab. Disord. 1999, 23, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Foncea, R.; O’Byrne, S.M.; Jiang, H.; Zhang, Y.; Deis, J.A.; Blaner, W.S.; Bernlohr, D.A.; Chen, X. Lipocalin 2, a regulator of retinoid homeostasis and retinoidmediated thermogenic activation in adipose tissue. J. Biol. Chem. 2016, 291, 11216–11229. [Google Scholar] [CrossRef]
- Maeda, H. Nutraceutical effects of fucoxanthin for obesity and diabetes therapy: A review. J. Oleo Sci. 2015, 64, 125–132. [Google Scholar] [CrossRef]
- Sugawara, T.; Baskaran, V.; Tsuzuki, W.; Nagao, A. A Brown algae fucoxanthin is hydrolyzed to fucoxanthinol during absorption by Caco-2 human intestinal cells and mice. J. Nutr. 2002, 132, 946–951. [Google Scholar] [CrossRef]
- Peng, J.; Yuan, J.P.; Wu, C.F.; Wang, J.H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef]
- Abidov, M.; Ramazanov, Z.; Seifulla, R.; Grachev, S. The effects of Xanthigen in the weight management of obese premenopausal women with non-alcoholic fatty liver disease and normal liver fat. Diabetes Obes. Metab. 2010, 12, 72–81. [Google Scholar] [CrossRef]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Takahashi, N.; Kawada, T.; Miyashita, K. Fucoxanthin and its metabolite, fucoxanthinol, suppress adipocyte differentiation in 3T3-L1 cells. Int. J. Mol. Med. 2006, 18, 147–152. [Google Scholar] [CrossRef]
- Kim, S.M.; Jung, Y.J.; Kwon, O.N.; Cha, K.H.; Um, B.H.; Chung, D.; Pan, C.H. A potential commercial source of fucoxanthin extracted from the microalga Phaeodactylum tricornutum. Appl. Biochem. Biotechnol. 2012, 166, 1842–1855. [Google Scholar] [CrossRef]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Funayama, K.; Miyashita, K. Fucoxanthin from edible seaweed, Undaria pinnatifida, shows antiobesity effect through UCP1 expression in white adipose tissues. Biochem. Biophys. Res. Commun. 2005, 332, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Rebello, C.J.; Greenway, F.L.; Lau, F.H.; Lin, Y.; Stephens, J.M.; Johnson, W.D.; Coulter, A.A. Naringenin Promotes Thermogenic Gene Expression in Human White Adipose Tissue. Obesity 2019, 27, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Favela-Hernández, J.M.; González-Santiago, O.; Ramírez-Cabrera, M.; Esquivel-Ferriño, P.; Camacho-Corona, M. Chemistry and Pharmacology of Citrus sinensis. Mol. Nutr. Food Res. 2016, 21, 247. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Xi, W.; Hu, Y.; Nie, C.; Zhou, Z. Antioxidant activity of Citrus fruits. Food Chem. 2015, 196, 885–896. [Google Scholar] [CrossRef]
- De la Garza, A.L.; Etxeberria, U.; Haslberger, A.; Aumueller, E.; Martínez, J.A.; Milagro, F.I. Helichrysum and Grapefruit Extracts Boost Weight Loss in Overweight Rats Reducing Inflammation. J. Med. Food 2015, 18, 890–898. [Google Scholar] [CrossRef]
- Nakajima, V.M.; Macedo, G.A.; Macedo, J.A. Citrus bioactive phenolics: Role in the obesity treatment. LWT Food Sci. Technol. 2014, 59, 1205–1212. [Google Scholar] [CrossRef]
- Huong, D.T.T.; Takahashi, Y.; Ide, T. Activity and mRNA levels of enzymes involved in hepatic fatty acid oxidation in mice fed citrus flavonoids. Nutrition 2006, 22, 546–552. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef]
- Kim, G.S.; Park, H.J.; Woo, J.H.; Kim, M.K.; Koh, P.O.; Min, W.; Ko, Y.G.; Kim, C.H.; Won, C.K.; Cho, J.H. Citrus aurantium flavonoids inhibit adipogenesis through the Akt signaling pathway in 3T3-L1 cells. BMC Complement. Altern. Med. 2012, 12, 31. [Google Scholar] [CrossRef]
- Park, J.; Kim, H.L.; Jung, Y.; Ahn, K.S.; Kwak, H.J.; Um, J.Y. Bitter Orange (Citrus aurantium Linné) Improves Obesity by Regulating Adipogenesis and Thermogenesis through AMPK Activation. Nutrients 2019, 11, 1988. [Google Scholar] [CrossRef] [PubMed]
- Chou, Y.C.; Ho, C.T.; Pan, M.H. Immature Citrus reticulata Extract Promotes Browning of Beige Adipocytes in High-Fat Diet-Induced C57BL/6 Mice. J. Agric. Food Chem. 2018, 66, 969–9703. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, S.; Hyodo, T.; Nagao, T.; Nakanishi, A.; Tandia, M.; Tsuda, T. α-Monoglucosyl Hesperidin but Not Hesperidin Induces Brown-Like Adipocyte Formation and Suppresses White Adipose Tissue Accumulation in Mice. J. Agric. Food Chem. 2019, 67, 1948–1954. [Google Scholar] [CrossRef] [PubMed]


| Effect on Browning | miRNA | Model and/or Tissue Sample | Nutritional Factors | Effect of the Nutritional Factor on the Expression of the miRNA | Reference |
|---|---|---|---|---|---|
| Positive (+) | |||||
| 203 | Human colon adenocarcinoma cells (Caco-2) | Selenium (depletion/accurate levels) | ↓ miR-203 expression in Se depletion | [24] | |
| 193b | Human plasma | Weight loss dietary treatment (RESMENA trial) | ↑ miR-193b levels (and hypomethylated) in high responders to weight loss | [25] | |
| 365 | Human adipocytes | Obesity/adipose (WAT) hypertrophy | ↑ miR-365 in WAT hypertrophy | [26] | |
| Negative (−) | |||||
| 106b | Lung cancer cells | Seed procyanidin extract (GSE) cells | ↓ miR-106b in lung neoplastic cells | [27] | |
| LT97 cells (colon adenoma cells) | Butyrate (and trichostatin A) as histone deacetylase inhibitors | ↓ miR-106b levels: affects cycle-relevant genes and thus, cell proliferation | [28] | ||
| Plasma | Hyperlipidemia status/coronary artery disease (CAD) | ↓ miR-106b in patients with CAD. MiR-106b levels positively correlate with HDL-c & ApoA-I | [29] | ||
| Human colon adenocarcinoma cells (Caco-2) | Selenium (depletion/accurate levels) | ↓ miR-106b expression in Se depletion | [24] | ||
| HCT116 (colon cancer cells) | Butyrate (short chain FA) | ↓ miR-106b expression | [30] | ||
| Prostate cancer cells | Resveratrol treatment | ↓ miR-106b expression | [31] | ||
| 93 | Human hepatocellular carcinoma (HCC) | Nonalcoholic fatty liver disease (NAFLD) | ↑ miR-93 expression | [32] | |
| Human colon adenocarcinoma cells (Caco-2) | Low-selenium environment | ↓ miR-93 expression levels in low selenium | [33] | ||
| 122 | Exosomes from healthy males aged 20–30 y.o. | Fat mass/BAT activity | ↓ Exosomal miR-122-5p levels in high BAT activity group. Exosomal miR-12anti-obesityp correlates negatively with: BAT activity, serum HDL-cholesterol. Exosomal miR-122-5p correlates positively with: age, BMI, body fat mass, total cholesterol, and serum triglycerides. | [34] | |
| Plasma | Type 2 diabetes with/without NAFLD | ↑ miR-122 expression in T2DM patients with NAFLD as compared to those without NAFLD | [35] | ||
| Serum | Breast cancer patients (survivors) with different BMI ranges (obesity, weight gain) | miR-122 expression associated with BMI | [36] | ||
| Liver samples | Patients with alcoholic liver diseases (ALD) | miR-122 levels ↓ in liver samples from ALD patients ↑ levels of miR-122 target HIF1-α | [37] | ||
| Liver samples | Obese women following bariatric surgery with or without NAFLD | ↓ miR-122 in obese subjects with NAFLD ↓ miR-122 in liver associated with impaired FA usage | [38] | ||
| Plasma | Maternal (pre-gestational and gestational) obesity | ↓ miR-122 levels in pre-gestational obesity and gestational obesity | [39] | ||
| Human liver cancer cells (HepG2) | Flavonoid compounds (nobiletin, tangeretin, and hesperidin) from citrus peel | ↓ miR-122 expression and thus, affect FAS and CPT-1α, decreasing lipid accumulation | [40] | ||
| Serum and plasma | Individuals with risk factors for metabolic syndrome, T2D, CVD (Bruneck study) | Circulating miR-122 associated with ↑ levels of liver enzymes, adiposity, inflammation, and insulin resistance and an adverse lipid profile Circulating miR-122 levels correlated with lipid subspecies (monounsaturated and saturated fatty acids) and cholesterol esters | [41] |
| Effect on Browning | miRNA | Tissue Sample | Nutritional Factor | Effect of the Nutritional Factor on the Expression of the miRNA | Reference |
|---|---|---|---|---|---|
| Positive (+) | |||||
| 196a (specific) | Pancreatic cancer cells | Dietary phytochemicals (garcinol) | Modulated miR-196a expression | [42] | |
| 26a/b | Formalin-fixed paraffin-embedded lung cancer (144 adenocarcinomas and 120 squamous cell carcinomas) | Intake of quercetin-rich foods (evaluated through a food-frequency questionnaire) | miR-26 expression differentially expressed between highest and lowest quercetin consumers | [43] | |
| Liver (human) | HFD/obesity | ↓ miR-26 expression | [44] | ||
| 32 | Healthy human colorectal epithelium | Non-digestible carbohydrates (resistant starch and polydextrose) supplementation | ↑ miR-32 expression in rectal mucosa | [45] | |
| 455 | Human adipose tissue and blood cells | Obesity & T2D (computational framework miR-QTL-Scan) | BAT specific miR-455 play a role in adipogenesis | [46] | |
| Human adipose tissue (BAT and WAT) from neck | HFD/Obesity and cold-induced thermogenesis | MiR-455 identified as a BAT marker in humans | [47] | ||
| Negative (−) | |||||
| 125-5p (specific) | Blood from T2DM and obese patients | Meta-analysis including lifestyle intervention studies | ↓ miR-125-5p in obese patients | [48] | |
| 34a | Human sc WAT (48 subjects) | Three calorie-restricted diets (different amount and quality of carbohydrates): low glycemic index, high glycemic index, and low fat | ↓ miR-34a in waist circumference stratified (tertiles) cohort No changes on miRNA levels between the intervention groups. | [49] | |
| Liver of male Sprague-Dawley rats | High-fat high-cholesterol (WD) diet supplemented with fish oil (FOH) | ↓ miR-34a in FOH vs. WD | [50] | ||
| Huh-7 (human liver) cells | Cholesterol accumulation associated to nonalcoholic fatty liver disease (NAFLD) | ↑ miR-34a expression in liver | [51] | ||
| 155 | THP-1 (human) monocytes/macrophages | Oleic acid | ↑ miR-155 expression in monocytes (vs. DHA) | [52] | |
| 378 | Patients with NASH (liver biopsies) Human HepG2 with accumulated lipid (oleate) | N.A. Oleate | ↑ miR-378 expression ↑ miR-378 expression | [53,54] | |
| Muscle biopsies of healthy males | Single bout of concurrent resistance exercise (8 × 5 leg extension, 80% 1RM) + 30 min at ~70% VO2peak with either post-exercise (whey) protein (25 g) or placebo | ↑ miR-378 expression at 4 h post-exercise with protein | [55] |
| Effect on browning | miRNA | Tissue sample | Nutritional factor | Effect of the nutritional factor on the expression of the miRNA | Reference |
|---|---|---|---|---|---|
| Positive (+) | |||||
| 196a (specific) | Adipose tissue (sc and visceral) of lambs | DHA-G diet: barley-based finishing diet where algae meal (DHA-Gold; Schizochytrium spp.) replaced flax oil | ↑ miR-196a expression in SAT vs. PAT (perirenal) in DHA-G diet. Differential miRNA expression in each tissue depot depending on diet | [56] | |
| Bovine adipose tissue (sc and visceral) from cattle | HFD | ↓ miR-196a in HFD and higher expression in visceral tissue depot | [57] | ||
| 26a/b | Goat milk | Milk (fatty acid) composition | miR-26 expression associated with total fat yield and short-, medium and long-chain fatty acid content. No association with lactose or milk protein content. Positive correlation miR-26a family and C16:1 and C18:3 in milk fat. | [58] | |
| Liver and adipose tissue (pregnant) rats | Diets with different fatty acid types: soybean (SO), olive (OO), fish (FO), linseed (LO), or palm-oil (PO) diets from conception to day 12 of gestation and standard diet thereafter | MiR-26 (among others) differentially modulated by the different fatty acids during early pregnancy. | [59] | ||
| Liver (human) & mice | HFD/obesity | ↓ miR-26 expression (humans and two obesity mice models) | [44] | ||
| 30b/c | Cortex and cerebellum of middle-aged C57Bl/6J mice | Extra-virgin olive oil rich in phenols feeding for 6 months (H-EVOO, phenol dose/day: 6 mg/kg) vs. the same olive oil deprived of phenolics (L-EVOO) | ↓ miR-30 expression in H-EVOO ↑ miRNAs in old L-EVOO animals compared to young. | [60] | |
| Rainbow trout eggs | Trout egg quality and production (characterization of miRNA profile) | miR-30 among top-10 most abundant miRNAs | [61] | ||
| Mice adipose tissue macrophages (ATMs) | HFD (12 wk) in combination with CB1 antagonist AM251 (4 wk, 10 mg/kg) | ↑ miR-30e-5p in ATMs from HFD + AM251 mice | [62] | ||
| Male C57BL/6J mice | HFD (16 wk) | ↓ miR-30a, -30c, -30e expression in ATM from HFD mice vs. NCD through epigenetic (methylation) modifications | [63] | ||
| 455 | Male C57BL/6J (B6) mice | HFD (45% kcal fat) supplemented with EPA (6.75% kcal EPA) for 11 wk. | ↑ miR-455 expression in BAT | [64] | |
| Murine adipose tissue (BAT and WAT) | HFD/Obesity and cold-induced thermogenesis | MiR-455 identified as a BAT marker in rodents | [47] | ||
| Negative (−) | |||||
| 27b | Milk samples from rats (lactation) | Cafeteria and post-cafeteria diet | MiR-27 levels in milk decrease throughout lactation. | [65] | |
| Cortex and cerebellum of middle-aged C57Bl/6J mice | Extra-virgin olive oil rich in phenols feeding for 6 months (H-EVOO, phenol dose/day: 6 mg/kg) vs. the same olive oil deprived of phenolics (L-EVOO) | ↓ miR-27 levels in H-EVOO | [60] | ||
| 34a | Breast cancer cells and carcinogenesis model in rats | 3,6-dihydroxyflavone (flavonoid) | ↑ miR-34a in breast carcinogenesis | [66] | |
| Liver of male Sprague-Dawley rats | High-fat high-cholesterol (WD) diet supplemented with fish oil (FOH) | ↓ miR-34a in FOH vs. WD | [50] | ||
| Mice liver | Three dietary interventions affecting lifespan (LS): caloric restriction (CR), low fat or high fat plus voluntary exercise or 30% CR | ↑ miR-34a in livers of two models of obesity MiR-34a fold change negatively correlated with LS | [67] | ||
| Mouse pancreatic β-cells | Saturated fatty acids | ↑ miR-34a expression | [68,69] | ||
| 133 | C57BL/6 male mice | High fat diet concomitant with miR-133 ASO (anti-miR-133) | ↑ miR-133 expression in HFD ↓ miR-133 expression in cold-exposed mice HFD and miR-133 antagonism ↑ BAT activity | [70] | |
| 155 | 3T3-L1 (mouse) adipocytes | Resveratrol (25 µM) | ↑ miR-155 expression | [71] | |
| FVB mice (colon mucosa) | High fat diet (45%) and 30% caloric restriction (CR) | ↑ miR-155 expression in colon mucosa in HFD mice | [72] | ||
| RAW264.7 macrophages (LPS activated) | 10 µM quercetin, quercetin-3-glucuronide (Q3G) and isorhamnetin | ↓ miR-155 expression by quercetin and Q3G | [73] | ||
| 378 | Livers of dietary obese mice | HFD (60% cal.) | ↑ miR-378 expression | [53,54] | |
| Milk samples from dairy cows in mid lactation | Control diet (total mixed ration of corn:grass silages) for 28 days followed by a treatment period (control diet supplemented with 5% linseed or safflower oil) of 28 days. | ↑ miR-378 expression by both treatments | [74] | ||
| Mice livers Mouse primary hepatocytes | Fasting & re-feed Palmitic acid (PA), linoleic acid (LA), oleic acid (OA) | ↑ miR-378 expression in fasting & ↓ miR-378 upon re-feeding ↑ miR-378 expression | [29] | ||
| Mice liver | Fisetin (a flavonoid): normal diet, HFD, HFD + fisetin | ↑ miR-378 expression by HFD & ↓ miR-378 by fisetin sup. | [75] | ||
| Mice brown adipocytes Brown fat (BAT) from mice | Omega-3 eicosapentaenoic acid (EPA) Low fat diet (LF), iso-caloric high fat (HF, 50% cal.) enriched with palm oil (HF + PO), olive oil (HF + OO), fish oil (HF + FO) for 12 wk. | ↑ miR-378 expression by EPA during brown differentiation ↑ miR-378 expression in iBAT from mice treated with HF + FO vs. HF + OO or HF + PO. | [76] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lorente-Cebrián, S.; Herrera, K.; I. Milagro, F.; Sánchez, J.; de la Garza, A.L.; Castro, H. miRNAs and Novel Food Compounds Related to the Browning Process. Int. J. Mol. Sci. 2019, 20, 5998. https://doi.org/10.3390/ijms20235998
Lorente-Cebrián S, Herrera K, I. Milagro F, Sánchez J, de la Garza AL, Castro H. miRNAs and Novel Food Compounds Related to the Browning Process. International Journal of Molecular Sciences. 2019; 20(23):5998. https://doi.org/10.3390/ijms20235998
Chicago/Turabian StyleLorente-Cebrián, Silvia, Katya Herrera, Fermín I. Milagro, Juana Sánchez, Ana Laura de la Garza, and Heriberto Castro. 2019. "miRNAs and Novel Food Compounds Related to the Browning Process" International Journal of Molecular Sciences 20, no. 23: 5998. https://doi.org/10.3390/ijms20235998
APA StyleLorente-Cebrián, S., Herrera, K., I. Milagro, F., Sánchez, J., de la Garza, A. L., & Castro, H. (2019). miRNAs and Novel Food Compounds Related to the Browning Process. International Journal of Molecular Sciences, 20(23), 5998. https://doi.org/10.3390/ijms20235998









