Depression, GABA, and Age Correlate with Plasma Levels of Inflammatory Markers
Abstract
:1. Introduction
2. Results
2.1. Demographic Data
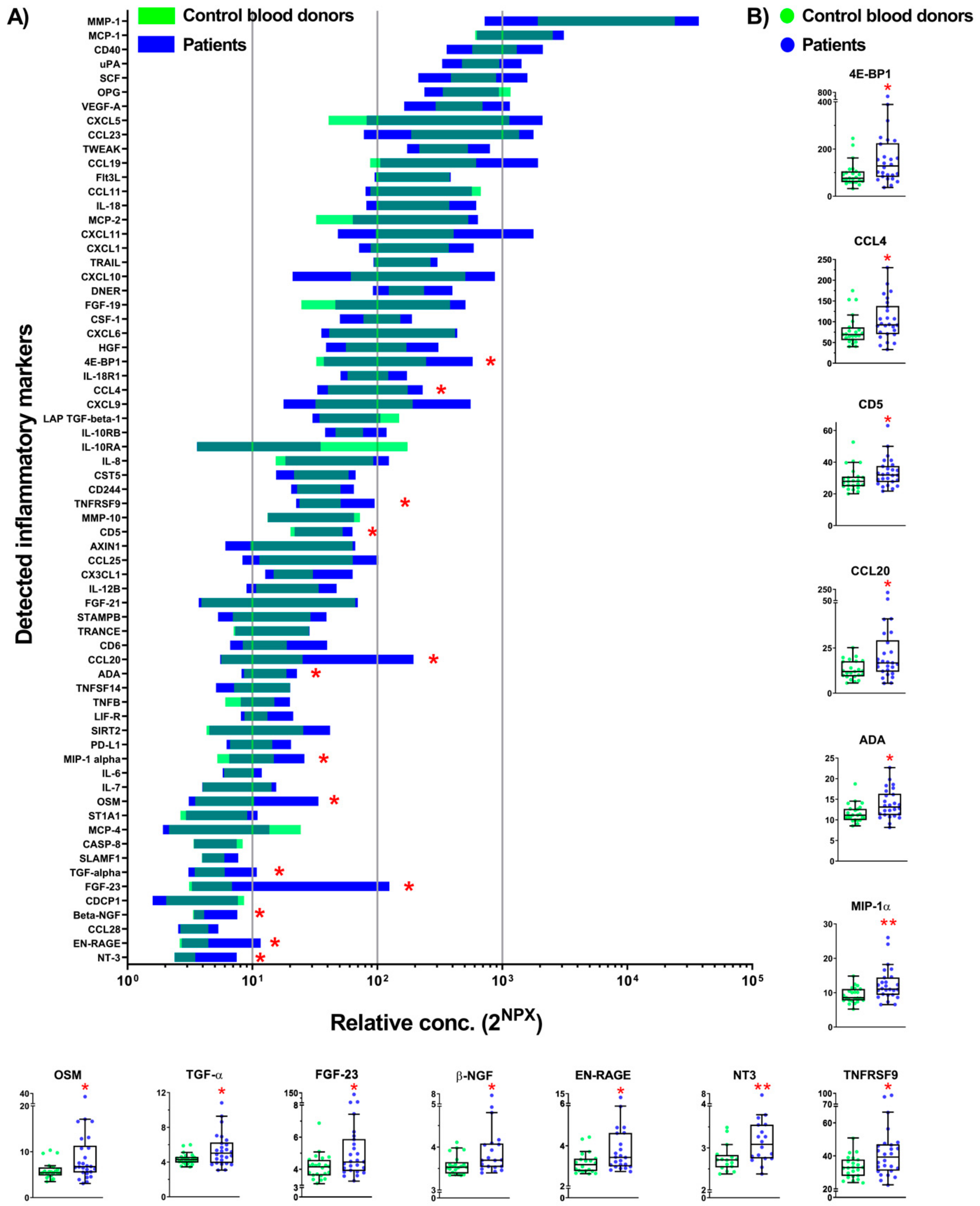
2.2. Inflammatory Markers in Plasma from Patients and CBD
2.3. Effects of Age on Levels of Inflammatory Markers in Plasma
2.4. The GABA Concentration in Plasma and Correlation of GABA or MADRS-S Score with Levels of Inflammatory Markers
2.5. The GABA Signaling System is Altered in PBMCs from the Patients
2.6. GABA Regulates the Release of Inflammatory Markers from Stimulated PBMCs from the Patients
3. Discussion
3.1. GABA and Age Modulate the Inflammatory Environment
3.2. Inflammatory Markers in Psychiatric Disorders
3.3. Plasma GABA Concentrations
3.4. Immunomodulation by GABA
3.5. Limitations of the Present Study
3.6. Conclusions and Further Studies
4. Materials and Methods
4.1. Study Individuals, Ethical Permits, and Blood Samples
4.2. Plasma and PBMC Isolation from Blood Samples
4.3. Multiplex PEA for Inflammatory Marker Measurements
4.4. Determination of the GABA Concentration
4.5. Real-Time Quantitative Reverse Transcription PCR
4.6. PBMC Supernatants for Multiplex PEA
4.7. Experimental Design and Statistical Rationale
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Olsen, R.W.; Sieghart, W. International Union of Pharmacology. LXX. Subtypes of gamma-aminobutyric acid(A) receptors: Classification on the basis of subunit composition, pharmacology, and function. Update. Pharm. Rev. 2008, 60, 243–260. [Google Scholar] [CrossRef] [Green Version]
- Levite, M. Neurotransmitters activate T-cells and elicit crucial functions via neurotransmitter receptors. Curr Opin Pharm. 2008, 8, 460–471. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Lu, Y.; Zhang, H.; Chau, C.H.; Dang, H.N.; Kaufman, D.L. Gamma-aminobutyric acid inhibits T cell autoimmunity and the development of inflammatory responses in a mouse type 1 diabetes model. J. Immunol. 2004, 173, 5298–5304. [Google Scholar] [CrossRef] [PubMed]
- Bjurstom, H.; Wang, J.; Ericsson, I.; Bengtsson, M.; Liu, Y.; Kumar-Mendu, S.; Issazadeh-Navikas, S.; Birnir, B. GABA, a natural immunomodulator of T lymphocytes. J. Neuroimmunol. 2008, 205, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Bhat, R.; Axtell, R.; Mitra, A.; Miranda, M.; Lock, C.; Tsien, R.W.; Steinman, L. Inhibitory role for GABA in autoimmune inflammation. Proc. Natl. Acad. Sci. USA 2010, 107, 2580–2585. [Google Scholar] [CrossRef] [Green Version]
- Jin, Z.; Mendu, S.K.; Birnir, B. GABA is an effective immunomodulatory molecule. Amino Acids 2013, 45, 87–94. [Google Scholar] [CrossRef] [Green Version]
- Bhandage, A.K.; Jin, Z.; Korol, S.V.; Shen, Q.; Pei, Y.; Deng, Q.; Espes, D.; Carlsson, P.O.; Kamali-Moghaddam, M.; Birnir, B. GABA Regulates Release of Inflammatory Cytokines From Peripheral Blood Mononuclear Cells and CD4(+) T Cells and Is Immunosuppressive in Type 1 Diabetes. EBioMedicine 2018, 30, 283–294. [Google Scholar] [CrossRef] [Green Version]
- Miller, A.H.; Raison, C.L. The role of inflammation in depression: From evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 2016, 16, 22–34. [Google Scholar] [CrossRef] [Green Version]
- Dantzer, R. Cytokine-induced sickness behavior: Mechanisms and implications. Ann. N.Y. Acad. Sci. 2001, 933, 222–234. [Google Scholar] [CrossRef]
- Udina, M.; Castellvi, P.; Moreno-Espana, J.; Navines, R.; Valdes, M.; Forns, X.; Langohr, K.; Sola, R.; Vieta, E.; Martin-Santos, R. Interferon-induced depression in chronic hepatitis C: A systematic review and meta-analysis. J. Clin. Psychiatry 2012, 73, 1128–1138. [Google Scholar] [CrossRef]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctot, K.L. A meta-analysis of cytokines in major depression. Biol. Psychiatry 2010, 67, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.H.; Maletic, V.; Raison, C.L. Inflammation and its discontents: The role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 2009, 65, 732–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, K.A.; Thomsen, C. The role of the innate immune system in psychiatric disorders. Mol. Cell Neurosci. 2013, 53, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Valkanova, V.; Ebmeier, K.P.; Allan, C.L. CRP, IL-6 and depression: A systematic review and meta-analysis of longitudinal studies. J. Affect. Disord. 2013, 150, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Raison, C.L.; Rutherford, R.E.; Woolwine, B.J.; Shuo, C.; Schettler, P.; Drake, D.F.; Haroon, E.; Miller, A.H. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: The role of baseline inflammatory biomarkers. Jama Psychiatry 2013, 70, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Strawbridge, R.; Arnone, D.; Danese, A.; Papadopoulos, A.; Herane Vives, A.; Cleare, A.J. Inflammation and clinical response to treatment in depression: A meta-analysis. Eur. Neuropsychopharmacol. 2015, 25, 1532–1543. [Google Scholar] [CrossRef] [PubMed]
- Kohler, C.A.; Freitas, T.H.; Stubbs, B.; Maes, M.; Solmi, M.; Veronese, N.; de Andrade, N.Q.; Morris, G.; Fernandes, B.S.; Brunoni, A.R.; et al. Peripheral Alterations in Cytokine and Chemokine Levels After Antidepressant Drug Treatment for Major Depressive Disorder: Systematic Review and Meta-Analysis. Mol. Neurobiol 2017, 55, 4195–4206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kohler, C.A.; Freitas, T.H.; Maes, M.; de Andrade, N.Q.; Liu, C.S.; Fernandes, B.S.; Stubbs, B.; Solmi, M.; Veronese, N.; Herrmann, N.; et al. Peripheral cytokine and chemokine alterations in depression: A meta-analysis of 82 studies. Acta Psychiatr. Scand. 2017, 135, 373–387. [Google Scholar] [CrossRef]
- Benedetti, F.; Poletti, S.; Hoogenboezem, T.A.; Locatelli, C.; de Wit, H.; Wijkhuijs, A.J.M.; Colombo, C.; Drexhage, H.A. Higher Baseline Proinflammatory Cytokines Mark Poor Antidepressant Response in Bipolar Disorder. J. Clin. Psychiatry 2017, 78, e986–e993. [Google Scholar] [CrossRef]
- Uher, R.; Tansey, K.E.; Dew, T.; Maier, W.; Mors, O.; Hauser, J.; Dernovsek, M.Z.; Henigsberg, N.; Souery, D.; Farmer, A.; et al. An inflammatory biomarker as a differential predictor of outcome of depression treatment with escitalopram and nortriptyline. Am. J. Psychiatry 2014, 171, 1278–1286. [Google Scholar] [CrossRef]
- Cattaneo, A.; Gennarelli, M.; Uher, R.; Breen, G.; Farmer, A.; Aitchison, K.J.; Craig, I.W.; Anacker, C.; Zunsztain, P.A.; McGuffin, P.; et al. Candidate genes expression profile associated with antidepressants response in the GENDEP study: Differentiating between baseline ‘predictors’ and longitudinal ‘targets’. Neuropsychopharmacology 2013, 38, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Bu, D.F.; Erlander, M.G.; Hitz, B.C.; Tillakaratne, N.J.; Kaufman, D.L.; Wagner-McPherson, C.B.; Evans, G.A.; Tobin, A.J. Two human glutamate decarboxylases, 65-kDa GAD and 67-kDa GAD, are each encoded by a single gene. Proc. Natl. Acad. Sci. USA 1992, 89, 2115–2119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, M.J.; Mirnics, K. Neurodevelopment, GABA system dysfunction, and schizophrenia. Neuropsychopharmacology 2015, 40, 190–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korol, S.V.J.Z.; Jin, Y.; Bhandage, A.K.; Tengholm, A.; Gandasi, N.; Barg, S.; Espes, D.; Carlsson, P.O.; Laver, D.; Birnir, B. Functional characterization of native, high-affinity GABAA receptors in human pancreatic beta cells. eBioMedicine 2018, 30, 273–282, Accepted for publication. [Google Scholar] [CrossRef] [Green Version]
- Birnir, B.; Korpi, E.R. The impact of sub-cellular location and intracellular neuronal proteins on properties of GABA(A) receptors. Curr. Pharm. Des. 2007, 13, 3169–3177. [Google Scholar] [CrossRef]
- Petty, F.; Sherman, A.D. Plasma GABA levels in psychiatric illness. J. Affect. Disord 1984, 6, 131–138. [Google Scholar] [CrossRef]
- Plog, B.A.; Nedergaard, M. The Glymphatic System in Central Nervous System Health and Disease: Past, Present, and Future. Annu. Rev. Pathol. 2018, 13, 379–394. [Google Scholar] [CrossRef] [Green Version]
- Bhandage, A.K.; Hellgren, C.; Jin, Z.; Olafsson, E.B.; Sundstrom-Poromaa, I.; Birnir, B. Expression of GABA receptors subunits in peripheral blood mononuclear cells is gender dependent, altered in pregnancy and modified by mental health. Acta Physiol (Oxf) 2015, 213, 575–585. [Google Scholar] [CrossRef]
- Conze, T.; Lammers, R.; Kuci, S.; Scherl-Mostageer, M.; Schweifer, N.; Kanz, L.; Buhring, H.J. CDCP1 is a novel marker for hematopoietic stem cells. Ann. N. Y. Acad. Sci. 2003, 996, 222–226. [Google Scholar] [CrossRef]
- Spassov, D.S.; Wong, C.H.; Sergina, N.; Ahuja, D.; Fried, M.; Sheppard, D.; Moasser, M.M. Phosphorylation of Trask by Src kinases inhibits integrin clustering and functions in exclusion with focal adhesion signaling. Mol. Cell Biol. 2011, 31, 766–782. [Google Scholar] [CrossRef] [Green Version]
- Enyindah-Asonye, G.; Li, Y.; Ruth, J.H.; Spassov, D.S.; Hebron, K.E.; Zijlstra, A.; Moasser, M.M.; Wang, B.; Singer, N.G.; Cui, H.; et al. CD318 is a ligand for CD6. Proc. Natl. Acad. Sci. USA 2017, 114, E6912–E6921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill, L.J.; Di Pietro, V.; Hazeldine, J.; Davies, D.; Toman, E.; Logan, A.; Belli, A. Cystatin D (CST5): An ultra-early inflammatory biomarker of traumatic brain injury. Sci. Rep. 2017, 7, 5002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrer-Mayorga, G.; Alvarez-Diaz, S.; Valle, N.; De Las Rivas, J.; Mendes, M.; Barderas, R.; Canals, F.; Tapia, O.; Casal, J.I.; Lafarga, M.; et al. Cystatin D locates in the nucleus at sites of active transcription and modulates gene and protein expression. J. Biol. Chem. 2015, 290, 26533–26548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marshall, F.H.; Jones, K.A.; Kaupmann, K.; Bettler, B. GABAB receptors-the first 7TM heterodimers. Trends Pharm. Sci. 1999, 20, 396–399. [Google Scholar] [CrossRef]
- Olsen, R.W.; Sieghart, W. GABA A receptors: Subtypes provide diversity of function and pharmacology. Neuropharmacology 2009, 56, 141–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gassmann, M.; Shaban, H.; Vigot, R.; Sansig, G.; Haller, C.; Barbieri, S.; Humeau, Y.; Schuler, V.; Muller, M.; Kinzel, B.; et al. Redistribution of GABAB(1) protein and atypical GABAB responses in GABAB(2)-deficient mice. J. Neurosci. 2004, 24, 6086–6097. [Google Scholar] [CrossRef] [Green Version]
- Pallavi, P.; Sagar, R.; Mehta, M.; Sharma, S.; Subramanium, A.; Shamshi, F.; Sengupta, U.; Qadri, R.; Pandey, R.M.; Mukhopadhyay, A.K. Serum neurotrophic factors in adolescent depression: Gender difference and correlation with clinical severity. J. Affect. Disord. 2013, 150, 415–423. [Google Scholar] [CrossRef]
- Oglodek, E.A.; Just, M.J.; Szromek, A.R.; Araszkiewicz, A. Melatonin and neurotrophins NT-3, BDNF, NGF in patients with varying levels of depression severity. Pharm. Rep. 2016, 68, 945–951. [Google Scholar] [CrossRef]
- Quiroz, J.A.; Machado-Vieira, R.; Zarate, C.A., Jr.; Manji, H.K. Novel insights into lithium’s mechanism of action: Neurotrophic and neuroprotective effects. Neuropsychobiology 2010, 62, 50–60. [Google Scholar] [CrossRef] [Green Version]
- Engel, D.; Zomkowski, A.D.; Lieberknecht, V.; Rodrigues, A.L.; Gabilan, N.H. Chronic administration of duloxetine and mirtazapine downregulates proapoptotic proteins and upregulates neurotrophin gene expression in the hippocampus and cerebral cortex of mice. J. Psychiatr. Res. 2013, 47, 802–808. [Google Scholar] [CrossRef]
- Angelucci, F.; Aloe, L.; Jimenez-Vasquez, P.; Mathe, A.A. Lithium treatment alters brain concentrations of nerve growth factor, brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor in a rat model of depression. Int J. Neuropsychopharmacol. 2003, 6, 225–231. [Google Scholar] [CrossRef] [Green Version]
- Hellweg, R.; Lang, U.E.; Nagel, M.; Baumgartner, A. Subchronic treatment with lithium increases nerve growth factor content in distinct brain regions of adult rats. Mol. Psychiatry 2002, 7, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Walz, J.C.; Frey, B.N.; Andreazza, A.C.; Cereser, K.M.; Cacilhas, A.A.; Valvassori, S.S.; Quevedo, J.; Kapczinski, F. Effects of lithium and valproate on serum and hippocampal neurotrophin-3 levels in an animal model of mania. J. Psychiatr. Res. 2008, 42, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Hannestad, J.; DellaGioia, N.; Bloch, M. The effect of antidepressant medication treatment on serum levels of inflammatory cytokines: A meta-analysis. Neuropsychopharmacology 2011, 36, 2452–2459. [Google Scholar] [CrossRef]
- Jansen, R.; Penninx, B.W.; Madar, V.; Xia, K.; Milaneschi, Y.; Hottenga, J.J.; Hammerschlag, A.R.; Beekman, A.; van der Wee, N.; Smit, J.H.; et al. Gene expression in major depressive disorder. Mol. Psychiatry 2016, 21, 339–347. [Google Scholar] [CrossRef]
- Svenningsson, P.; Berg, L.; Matthews, D.; Ionescu, D.F.; Richards, E.M.; Niciu, M.J.; Malinger, A.; Toups, M.; Manji, H.; Trivedi, M.H.; et al. Preliminary evidence that early reduction in p11 levels in natural killer cells and monocytes predicts the likelihood of antidepressant response to chronic citalopram. Mol. Psychiatry 2014, 19, 962–964. [Google Scholar] [CrossRef] [Green Version]
- Ryan, K.M.; McLoughlin, D.M. Vascular endothelial growth factor plasma levels in depression and following electroconvulsive therapy. Eur. Arch. Psychiatry Clin. Neurosci. 2018, 268, 839–848. [Google Scholar] [CrossRef]
- Maes, M. The immunoregulatory effects of antidepressants. Hum. Psychopharmacol. 2001, 16, 95–103. [Google Scholar] [CrossRef]
- Gadad, B.S.; Jha, M.K.; Grannemann, B.D.; Mayes, T.L.; Trivedi, M.H. Proteomics profiling reveals inflammatory biomarkers of antidepressant treatment response: Findings from the CO-MED trial. J. Psychiatr. Res. 2017, 94, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Ricken, R.; Busche, M.; Schlattmann, P.; Himmerich, H.; Bopp, S.; Bschor, T.; Richter, C.; Stamm, T.J.; Heinz, A.; Hellweg, R.; et al. Cytokine serum levels remain unchanged during lithium augmentation of antidepressants in major depression. J. Psychiatr. Res. 2018, 96, 203–208. [Google Scholar] [CrossRef]
- Leighton, S.P.; Nerurkar, L.; Krishnadas, R.; Johnman, C.; Graham, G.J.; Cavanagh, J. Chemokines in depression in health and in inflammatory illness: A systematic review and meta-analysis. Mol. Psychiatry 2018, 23, 48–58. [Google Scholar] [CrossRef] [Green Version]
- Ho, P.S.; Yeh, Y.W.; Huang, S.Y.; Liang, C.S. A shift toward T helper 2 responses and an increase in modulators of innate immunity in depressed patients treated with escitalopram. Psychoneuroendocrinology 2015, 53, 246–255. [Google Scholar] [CrossRef]
- Fan, N.; Luo, Y.; Ou, Y.; He, H. Altered serum levels of TNF-alpha, IL-6, and IL-18 in depressive disorder patients. Hum. Psychopharmacol. 2017, 32. [Google Scholar] [CrossRef]
- Brann, E.; Fransson, E.; White, R.A.; Papadopoulos, F.C.; Edvinsson, A.; Kamali-Moghaddam, M.; Cunningham, J.L.; Sundstrom-Poromaa, I.; Skalkidou, A. Inflammatory markers in women with postpartum depressive symptoms. J. Neurosci. Res. 2018. [Google Scholar] [CrossRef]
- Herder, C.; Hermanns, N. Subclinical inflammation and depressive symptoms in patients with type 1 and type 2 diabetes. Semin. Immunopathol. 2019. [Google Scholar] [CrossRef]
- Pietrzak, D.; Pietrzak, A.; Grywalska, E.; Kicinski, P.; Rolinski, J.; Donica, H.; Franciszkiewicz-Pietrzak, K.; Borzecki, A.; Socha, M.; Niedzialek, J.; et al. Serum concentrations of interleukin 18 and 25-hydroxyvitamin D3 correlate with depression severity in men with psoriasis. PLoS ONE 2018, 13, e0201589. [Google Scholar] [CrossRef]
- Antonelli, A.; Rotondi, M.; Fallahi, P.; Ferrari, S.M.; Paolicchi, A.; Romagnani, P.; Serio, M.; Ferrannini, E. Increase of CXC chemokine CXCL10 and CC chemokine CCL2 serum levels in normal ageing. Cytokine 2006, 34, 32–38. [Google Scholar] [CrossRef]
- Enroth, S.; Enroth, S.B.; Johansson, A.; Gyllensten, U. Protein profiling reveals consequences of lifestyle choices on predicted biological aging. Sci. Rep. 2015, 5, 17282. [Google Scholar] [CrossRef]
- Larsson, A.; Carlsson, L.; Gordh, T.; Lind, A.L.; Thulin, M.; Kamali-Moghaddam, M. The effects of age and gender on plasma levels of 63 cytokines. J. Immunol. Methods 2015, 425, 58–61. [Google Scholar] [CrossRef]
- Shurin, G.V.; Yurkovetsky, Z.R.; Chatta, G.S.; Tourkova, I.L.; Shurin, M.R.; Lokshin, A.E. Dynamic alteration of soluble serum biomarkers in healthy aging. Cytokine 2007, 39, 123–129. [Google Scholar] [CrossRef]
- Petty, F. GABA and mood disorders: A brief review and hypothesis. J. Affect. Disord. 1995, 34, 275–281. [Google Scholar] [CrossRef]
- Petty, F.; Kramer, G.L.; Dunnam, D.; Rush, A.J. Plasma GABA in mood disorders. Psychopharmacol. Bull. 1990, 26, 157–161. [Google Scholar]
- Petty, F.; Kramer, G.L.; Fulton, M.; Moeller, F.G.; Rush, A.J. Low plasma GABA is a trait-like marker for bipolar illness. Neuropsychopharmacology 1993, 9, 125–132. [Google Scholar] [CrossRef] [Green Version]
- Petty, F.; Schlesser, M.A. Plasma GABA in affective illness. A preliminary investigation. J. Affect. Disord. 1981, 3, 339–343. [Google Scholar] [CrossRef]
- Berrettini, W.H.; Nurnberger, J.I., Jr.; Hare, T.A.; Simmons-Alling, S.; Gershon, E.S.; Post, R.M. Reduced plasma and CSF gamma-aminobutyric acid in affective illness: Effect of lithium carbonate. Biol. Psychiatry 1983, 18, 185–194. [Google Scholar]
- Lu, Y.R.; Fu, X.Y.; Shi, L.G.; Jiang, Y.; Wu, J.L.; Weng, X.J.; Wang, Z.P.; Wu, X.Y.; Lin, Z.; Liu, W.B.; et al. Decreased plasma neuroactive amino acids and increased nitric oxide levels in melancholic major depressive disorder. BMC Psychiatry 2014, 14, 123. [Google Scholar] [CrossRef] [Green Version]
- Loscher, W. Anticonvulsant and biochemical effects of inhibitors of GABA aminotransferase and valproic acid during subchronic treatment in mice. Biochem. Pharm. 1982, 31, 837–842. [Google Scholar] [CrossRef]
- Barragan, A.; Weidner, J.M.; Jin, Z.; Korpi, E.R.; Birnir, B. GABAergic signalling in the immune system. Acta Physiol (Oxf) 2015, 213, 819–827. [Google Scholar] [CrossRef]
- Fuks, J.M.; Arrighi, R.B.; Weidner, J.M.; Kumar Mendu, S.; Jin, Z.; Wallin, R.P.; Rethi, B.; Birnir, B.; Barragan, A. GABAergic signaling is linked to a hypermigratory phenotype in dendritic cells infected by Toxoplasma gondii. PLoS Pathog. 2012, 8, e1003051. [Google Scholar] [CrossRef] [Green Version]
- Sheehan, D.V.; Lecrubier, Y.; Sheehan, K.H.; Amorim, P.; Janavs, J.; Weiller, E.; Hergueta, T.; Baker, R.; Dunbar, G.C. The Mini-International Neuropsychiatric Interview (M.I.N.I.): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 1998, 59 (Suppl. 20), 22–33, quiz 34-57. [Google Scholar]
- Svanborg, P.; Asberg, M. A new self-rating scale for depression and anxiety states based on the Comprehensive Psychopathological Rating Scale. Acta Psychiatr. Scand. 1994, 89, 21–28. [Google Scholar] [CrossRef]
- Cunningham, J.L.; Wernroth, L.; von Knorring, L.; Berglund, L.; Ekselius, L. Agreement between physicians’ and patients’ ratings on the Montgomery-Asberg Depression Rating Scale. J. Affect. Disord. 2011, 135, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Edvinsson, A.; Brann, E.; Hellgren, C.; Freyhult, E.; White, R.; Kamali-Moghaddam, M.; Olivier, J.; Bergquist, J.; Bostrom, A.E.; Schioth, H.B.; et al. Lower inflammatory markers in women with antenatal depression brings the M1/M2 balance into focus from a new direction. Psychoneuroendocrinology 2017, 80, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate-a Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. B 1995, 57, 289–300. [Google Scholar] [CrossRef]




| Participants (N) | 25 |
| Level of care at inclusion, n (%): | |
| Inpatient | 20 (80%) |
| Day program for depression | 5 (20%) |
| Age (Mean (SD)) | 43.96 (15.7) |
| Gender (M:F) | 12:13 |
| BMI (Mean (SD)) | 25.3 (6.4) |
| Diagnosis: n (%): | |
| Current depressive episode | 25 (100) |
| Major depressive disorder | 20 (80) |
| First depressive episode | 3 (12%) |
| Recurring unipolar depression | 17 (68%) |
| Bipolar disorder | 5 (20%) |
| Type I | 4 (16%) |
| Type II or uncategorized | 1 (4%) |
| Any anxiety disorder | 7 (28%) |
| Other psychiatric diagnoses * | 4 (16%) |
| Previous hospitalization for depression (n (%)) | 22 (88%) |
| MADRS-S score (mean (SD)) | 33.8 (7,4) |
| Medication, n (%): | |
| Other anxiolytic medications ** | 11 (44%) |
| Antidepressive treatment *** | 21 (84%) |
| Antipsychotics | 6 (24%) |
| Benzodiazepines | 5 (20%) |
| Z-analogues | 6 (24%) |
| CBD | Patients | |
|---|---|---|
| GABAA Receptor Subunits | ||
| GABRA1 (α1) | 0 | 0 |
| GABRA2 (α2) | 0 | 0 |
| GABRA3 (α3) | 3.8 | 4 |
| GABRA4 (α4) | 15.4 | 8 |
| GABRA5 (α5) | 19.2 | 4 |
| GABRA6 (α6) | 15.4 | 16 |
| GABRB1 (β1) | 30.8 | 8 |
| GABRB2 (β2) | 38.5 | 36 |
| GABRB3 (β3) | 0 | 0 |
| GABRG1 (γ1) | 0 | 4 |
| GABRG2 (γ2) | 0 | 0 |
| GABRG3 (γ3) | 0 | 0 |
| GABRD (δ) | 34.6 | 12 |
| GABRE (ε) | 42.3 | 20 |
| GABRQ (θ) | 0 | 0 |
| GABRP (π) | 3.8 | 4 |
| GABRR1 (ρ1) | 0 | 0 |
| GABRR2 (ρ2) | 100 | 96 |
| GABRR3 (ρ3) | 0 | 12 |
| GABAB Receptor Subunits | ||
| GABBR1 (GABA-B1) | 100 | 100 |
| GABBR2 (GABA-B2) | 0 | 0 |
| Chloride Transporters | ||
| SLC12A2 (NKCC1) | 100 | 100 |
| SLC12A1 (NKCC2) | 0 | 0 |
| SLC12A4 (KCC1) | 100 | 100 |
| SLC12A5 (KCC2) | 0 | 0 |
| SLC12A6 (KCC3) | 100 | 100 |
| SLC12A7 (KCC4) | 96 | 100 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhandage, A.K.; Cunningham, J.L.; Jin, Z.; Shen, Q.; Bongiovanni, S.; Korol, S.V.; Syk, M.; Kamali-Moghaddam, M.; Ekselius, L.; Birnir, B. Depression, GABA, and Age Correlate with Plasma Levels of Inflammatory Markers. Int. J. Mol. Sci. 2019, 20, 6172. https://doi.org/10.3390/ijms20246172
Bhandage AK, Cunningham JL, Jin Z, Shen Q, Bongiovanni S, Korol SV, Syk M, Kamali-Moghaddam M, Ekselius L, Birnir B. Depression, GABA, and Age Correlate with Plasma Levels of Inflammatory Markers. International Journal of Molecular Sciences. 2019; 20(24):6172. https://doi.org/10.3390/ijms20246172
Chicago/Turabian StyleBhandage, Amol K., Janet L. Cunningham, Zhe Jin, Qiujin Shen, Santiago Bongiovanni, Sergiy V. Korol, Mikaela Syk, Masood Kamali-Moghaddam, Lisa Ekselius, and Bryndis Birnir. 2019. "Depression, GABA, and Age Correlate with Plasma Levels of Inflammatory Markers" International Journal of Molecular Sciences 20, no. 24: 6172. https://doi.org/10.3390/ijms20246172





