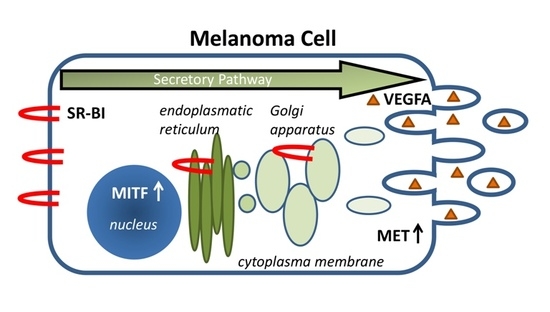
Loss of SR-BI Down-Regulates MITF and Suppresses Extracellular Vesicle Release in Human Melanoma
Abstract
:1. Introduction
2. Results
2.1. SR-BI Expression Pattern in Melanoma Is Associated with the Pigmentation Pathway
2.2. SR-BI Controls the Proto-Oncogene and Vesicle-Release-Driver MET
2.3. Extracellular Vesicle Secretion Is Altered after SR-BI Inhibition
2.4. SR-BI and MET Expression Correlates in Patient Data Sets
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Melanin Measurement
4.2. Transient SR-BI Overexpression
4.3. siRNA Knockdown
4.4. RNA Isolation and Real-Time PCR
4.5. Western Blotting
4.6. Immunocytochemistry and Immunofluorescence
4.7. ELISA
4.8. Vesicle Isolation
4.9. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATCC | American Type Culture Collection |
| CFSE | Carboxyfluorescein succinimidyl ester |
| DAPI | 4′,6-diamidino-2-phenylindole |
| FCS | fetal calf serum |
| FPKM | Fragments Per Kilobase Million |
| ICC | immunocytochemistry |
| IF | immunofluorescence |
| MIM | melanoma isolation medium |
| rel OD | relative optical density |
| RT-PCR | real-time polymerase chain reaction |
| SR-BI | scavenger receptor class B type I |
| TCGA | The Cancer Genome Atlas |
References
- Mooberry, L.K.; Sabnis, N.A.; Panchoo, M.; Nagarajan, B.; Lacko, A.G. Targeting the SR-B1 Receptor as a Gateway for Cancer Therapy and Imaging. Front. Pharmacol. 2016, 7, 466. [Google Scholar] [CrossRef] [PubMed]
- Cao, W.M.; Murao, K.; Imachi, H.; Yu, X.; Abe, H.; Yamauchi, A.; Niimi, M.; Miyauchi, A.; Wong, N.C.; Ishida, T. A mutant high-density lipoprotein receptor inhibits proliferation of human breast cancer cells. Cancer Res. 2004, 64, 1515–1521. [Google Scholar] [CrossRef] [PubMed]
- Twiddy, A.L.; Cox, M.E.; Wasan, K.M. Knockdown of scavenger receptor class B type I reduces prostate specific antigen secretion and viability of prostate cancer cells. Prostate 2012, 72, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Panchoo, M.; Lacko, A. Scavenger receptor class B type 1 regulates neuroblastoma cell proliferation, migration and invasion. Biochem. Biophys. Res. Commun. 2018, 495, 614–620. [Google Scholar] [PubMed]
- Velagapudi, S.; Schraml, P.; Yalcinkaya, M.; Bolck, H.A.; Rohrer, L.; Moch, H.; von Eckardstein, A. Scavenger receptor BI promotes cytoplasmic accumulation of lipoproteins in clear-cell renal cell carcinoma. J. Lipid Res. 2018, 59, 2188–2201. [Google Scholar] [PubMed]
- Li, J.; Wang, J.; Li, M.; Yin, L.; Li, X.A.; Zhang, T.G. Up-regulated expression of scavenger receptor class B type 1 (SR-B1) is associated with malignant behaviors and poor prognosis of breast cancer. Pathol. Res. Pract. 2016, 212, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Schorghofer, D.; Kinslechner, K.; Preitschopf, A.; Schutz, B.; Rohrl, C.; Hengstschlager, M.; Stangl, H.; Mikula, M. The HDL receptor SR-BI is associated with human prostate cancer progression and plays a possible role in establishing androgen independence. Reprod. Biol. Endocrinol. 2015, 13, 88. [Google Scholar] [CrossRef] [PubMed]
- Yuan, B.; Wu, C.; Wang, X.; Wang, D.; Liu, H.; Guo, L.; Li, X.A.; Han, J.; Feng, H. High scavenger receptor class B type I expression is related to tumor aggressiveness and poor prognosis in breast cancer. Tumour Biol. 2016, 37, 3581–3588. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Liu, Y.; Jin, H.; Pan, S.; Qian, Y.; Huang, C.; Zeng, Y.; Luo, Q.; Zeng, M.; Zhang, Z. Scavenger receptor B1 is a potential biomarker of human nasopharyngeal carcinoma and its growth is inhibited by HDL-mimetic nanoparticles. Theranostics 2013, 3, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Acton, S.; Rigotti, A.; Landschulz, K.T.; Xu, S.; Hobbs, H.H.; Krieger, M. Identification of scavenger receptor SR-BI as a high density lipoprotein receptor. Science 1996, 271, 518–520. [Google Scholar] [CrossRef] [PubMed]
- Krieger, M. Charting the fate of the “good cholesterol”: Identification and characterization of the high-density lipoprotein receptor SR-BI. Annu. Rev. Biochem. 1999, 68, 523–558. [Google Scholar] [CrossRef] [PubMed]
- Rohrl, C.; Stangl, H. HDL endocytosis and resecretion. Biochim. Biophys. Acta 2013, 1831, 1626–1633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pagler, T.A.; Rhode, S.; Neuhofer, A.; Laggner, H.; Strobl, W.; Hinterndorfer, C.; Volf, I.; Pavelka, M.; Eckhardt, E.R.; van der Westhuyzen, D.R.; et al. SR-BI-mediated high density lipoprotein (HDL) endocytosis leads to HDL resecretion facilitating cholesterol efflux. J. Biol. Chem. 2006, 281, 11193–11204. [Google Scholar] [CrossRef] [PubMed]
- Rohrl, C.; Pagler, T.A.; Strobl, W.; Ellinger, A.; Neumuller, J.; Pavelka, M.; Stangl, H.; Meisslitzer-Ruppitsch, C. Characterization of endocytic compartments after holo-high density lipoprotein particle uptake in HepG2 cells. Histochem. Cell Biol. 2010, 133, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Kinslechner, K.; Schorghofer, D.; Schutz, B.; Vallianou, M.; Wingelhofer, B.; Mikulits, W.; Rohrl, C.; Hengstschlager, M.; Moriggl, R.; Stangl, H.; et al. Malignant Phenotypes in Metastatic Melanoma are Governed by SR-BI and its Association with Glycosylation and STAT5 Activation. Mol. Cancer Res. 2018, 16, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Opdecamp, K.; Nakayama, A.; Nguyen, M.T.; Hodgkinson, C.A.; Pavan, W.J.; Arnheiter, H. Melanocyte development in vivo and in neural crest cell cultures: Crucial dependence on the Mitf basic-helix-loop-helix-zipper transcription factor. Development 1997, 124, 2377–2386. [Google Scholar] [PubMed]
- Cheli, Y.; Giuliano, S.; Botton, T.; Rocchi, S.; Hofman, V.; Hofman, P.; Bahadoran, P.; Bertolotto, C.; Ballotti, R. Mitf is the key molecular switch between mouse or human melanoma initiating cells and their differentiated progeny. Oncogene 2011, 30, 2307–2318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loercher, A.E.; Tank, E.M.; Delston, R.B.; Harbour, J.W. MITF links differentiation with cell cycle arrest in melanocytes by transcriptional activation of INK4A. J. Cell Biol. 2005, 168, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Antonellis, A.; Huynh, J.L.; Lee-Lin, S.Q.; Vinton, R.M.; Renaud, G.; Loftus, S.K.; Elliot, G.; Wolfsberg, T.G.; Green, E.D.; McCallion, A.S.; et al. Identification of neural crest and glial enhancers at the mouse Sox10 locus through transgenesis in zebrafish. PLoS Genet. 2008, 4, e1000174. [Google Scholar] [CrossRef] [PubMed]
- Carreira, S.; Goodall, J.; Denat, L.; Rodriguez, M.; Nuciforo, P.; Hoek, K.S.; Testori, A.; Larue, L.; Goding, C.R. Mitf regulation of Dia1 controls melanoma proliferation and invasiveness. Genes Dev. 2006, 20, 3426–3439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ennen, M.; Keime, C.; Gambi, G.; Kieny, A.; Coassolo, S.; Thibault-Carpentier, C.; Margerin-Schaller, F.; Davidson, G.; Vagne, C.; Lipsker, D.; et al. MITF-High and MITF-Low Cells and a Novel Subpopulation Expressing Genes of Both Cell States Contribute to Intra- and Intertumoral Heterogeneity of Primary Melanoma. Clin. Cancer Res. 2017, 23, 7097–7107. [Google Scholar] [CrossRef] [PubMed]
- Muller, J.; Krijgsman, O.; Tsoi, J.; Robert, L.; Hugo, W.; Song, C.; Kong, X.; Possik, P.A.; Cornelissen-Steijger, P.D.; Geukes Foppen, M.H.; et al. Low MITF/AXL ratio predicts early resistance to multiple targeted drugs in melanoma. Nat. Commun. 2014, 5, 5712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodall, J.; Carreira, S.; Denat, L.; Kobi, D.; Davidson, I.; Nuciforo, P.; Sturm, R.A.; Larue, L.; Goding, C.R. Brn-2 represses microphthalmia-associated transcription factor expression and marks a distinct subpopulation of microphthalmia-associated transcription factor-negative melanoma cells. Cancer Res. 2008, 68, 7788–7794. [Google Scholar] [CrossRef] [PubMed]
- Swoboda, A.; Schanab, O.; Tauber, S.; Bilban, M.; Berger, W.; Petzelbauer, P.; Mikula, M. MET expression in melanoma correlates with a lymphangiogenic phenotype. Hum. Mol. Genet. 2012, 21, 3387–3396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peinado, H.; Aleckovic, M.; Lavotshkin, S.; Matei, I.; Costa-Silva, B.; Moreno-Bueno, G.; Hergueta-Redondo, M.; Williams, C.; Garcia-Santos, G.; Ghajar, C.; et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 2012, 18, 883–891. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.; Afshari, A.; Sengupta, R.; Sebastiano, V.; Gupta, A.; Kim, Y.H.; Iorns, E.; Tsui, R.; Denis, A.; Perfito, N.; et al. Replication study: Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. eLife 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Hebert, D.N. Tyrosinase maturation through the mammalian secretory pathway: Bringing color to life. Pigment Cell Res. 2006, 19, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Garraway, L.A.; Widlund, H.R.; Rubin, M.A.; Getz, G.; Berger, A.J.; Ramaswamy, S.; Beroukhim, R.; Milner, D.A.; Granter, S.R.; Du, J.; et al. Integrative genomic analyses identify MITF as a lineage survival oncogene amplified in malignant melanoma. Nature 2005, 436, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Danilo, C.; Gutierrez-Pajares, J.L.; Mainieri, M.A.; Mercier, I.; Lisanti, M.P.; Frank, P.G. Scavenger receptor class B type I regulates cellular cholesterol metabolism and cell signaling associated with breast cancer development. Breast Cancer Res. 2013, 15, R87. [Google Scholar] [CrossRef] [PubMed]
- Fruhwurth, S.; Krieger, S.; Winter, K.; Rosner, M.; Mikula, M.; Weichhart, T.; Bittman, R.; Hengstschlager, M.; Stangl, H. Inhibition of mTOR down-regulates scavenger receptor, class B, type I (SR-BI) expression, reduces endothelial cell migration and impairs nitric oxide production. Biochim. Biophys. Acta 2014, 1841, 944–953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mineo, C.; Yuhanna, I.S.; Quon, M.J.; Shaul, P.W. High density lipoprotein-induced endothelial nitric-oxide synthase activation is mediated by Akt and MAP kinases. J. Biol. Chem. 2003, 278, 9142–9149. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Tyburczy, M.E.; Moss, J.; Darling, T.N.; Widlund, H.R.; Kwiatkowski, D.J. Tuberous sclerosis complex inactivation disrupts melanogenesis via mTORC1 activation. J. Clin. Investig. 2017, 127, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Stangl, H.; Cao, G.; Wyne, K.L.; Hobbs, H.H. Scavenger receptor, class B, type I-dependent stimulation of cholesterol esterification by high density lipoproteins, low density lipoproteins, and nonlipoprotein cholesterol. J. Biol. Chem. 1998, 273, 31002–31008. [Google Scholar] [CrossRef] [PubMed]
- Schallreuter, K.U.; Hasse, S.; Rokos, H.; Chavan, B.; Shalbaf, M.; Spencer, J.D.; Wood, J.M. Cholesterol regulates melanogenesis in human epidermal melanocytes and melanoma cells. Exp. Dermatol. 2009, 18, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Hoek, K.S.; Schlegel, N.C.; Eichhoff, O.M.; Widmer, D.S.; Praetorius, C.; Einarsson, S.O.; Valgeirsdottir, S.; Bergsteinsdottir, K.; Schepsky, A.; Dummer, R.; et al. Novel MITF targets identified using a two-step DNA microarray strategy. Pigment Cell Melanoma Res. 2008, 21, 665–676. [Google Scholar] [CrossRef] [PubMed]
- McGill, G.G.; Haq, R.; Nishimura, E.K.; Fisher, D.E. c-Met expression is regulated by Mitf in the melanocyte lineage. J. Biol. Chem. 2006, 281, 10365–10373. [Google Scholar] [CrossRef] [PubMed]
- Beer, K.B.; Wehman, A.M. Mechanisms and functions of extracellular vesicle release in vivo-What we can learn from flies and worms. Cell Adhes. Migr. 2017, 11, 135–150. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Chen, Y.; Zhu, S.; Li, F.; Zhao, S.; Wu, L.; Chen, X.; Su, J. RAB22A overexpression promotes the tumor growth of melanoma. Oncotarget 2016, 7, 71744–71753. [Google Scholar] [CrossRef] [PubMed]
- Im, Y.J.; Wollert, T.; Boura, E.; Hurley, J.H. Structure and function of the ESCRT-II-III interface in multivesicular body biogenesis. Dev. Cell 2009, 17, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Berger, W.; Hauptmann, E.; Elbling, L.; Vetterlein, M.; Kokoschka, E.M.; Micksche, M. Possible role of the multidrug resistance-associated protein (MRP) in chemoresistance of human melanoma cells. Int. J. Cancer 1997, 71, 108–115. [Google Scholar] [CrossRef] [Green Version]
- Bogunovic, D.; O’Neill, D.W.; Belitskaya-Levy, I.; Vacic, V.; Yu, Y.L.; Adams, S.; Darvishian, F.; Berman, R.; Shapiro, R.; Pavlick, A.C.; et al. Immune profile and mitotic index of metastatic melanoma lesions enhance clinical staging in predicting patient survival. Proc. Natl. Acad. Sci. USA 2009, 106, 20429–20434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riker, A.I.; Enkemann, S.A.; Fodstad, O.; Liu, S.; Ren, S.; Morris, C.; Xi, Y.; Howell, P.; Metge, B.; Samant, R.S.; et al. The gene expression profiles of primary and metastatic melanoma yields a transition point of tumor progression and metastasis. BMC Med. Genomics 2008, 1, 13. [Google Scholar] [CrossRef] [PubMed]
- Talantov, D.; Mazumder, A.; Yu, J.X.; Briggs, T.; Jiang, Y.; Backus, J.; Atkins, D.; Wang, Y. Novel genes associated with malignant melanoma but not benign melanocytic lesions. Clin. Cancer Res. 2005, 11, 7234–7242. [Google Scholar] [CrossRef] [PubMed]
- Akbani, R.; Akdemir, K.C.; Aksoy, B.A.; Albert, M.; Ally, A.; Amin, S.B.; Arachchi, H.; Arora, A.; Auman, J.T.; Ayala, B.; et al. Genomic Classification of Cutaneous Melanoma. Cell 2015, 161, 1681–1696. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| Primer Name | 5′ -> 3′ Forward | 5′ -> 3′ Reverse |
|---|---|---|
| β-Actin (ACTB) | CTATCCAGGCTGTGCTATCCCTGT | CCTTAATGTCACGCACGATTTCC |
| MET | GGCGACAGCTGACTTGCTGAGAG | GTAAACAGGAGCACGAGGATGCCAG |
| RAB22A | GCTTCGACAGCATGGCCCAC | AGATGGCAGGTTGGCGTCAGT |
| SNAP25 | CTGCGGGCTTTGTGTGTGTCC | TGACGGAGGTTCCCGATGATGC |
| SR-BI (SCARB1) | GTACGTCCTCCTGGCGCTGG | GCAGCACAGAGCCCTTGGGA |
| VPS25 | CATGTGGCGGAGGCCAGAAGA | GCCCGCAGTAGAGTGGCTTCA |
| Antibody | Company | Cat. No. | ICC/IF 1 | WB 2 |
|---|---|---|---|---|
| α-tubulin | Calbiochem | CP06 | 1:5000 | |
| β-Actin | Santa Cruz | sc-47778 | 1:1500 | |
| MET | Cell Signaling | #8198 | 1:200 | 1:1000 |
| MITF | Abcam | ab12039 | 1:1000 | |
| SR-BI | BDBiosciences | 610882 | 1:500 | 1:2000 |
| Biotinylated anti-rabbit | Vector Laboratories | BA-1100 | 1:500 | |
| Biotinylated anti-mouse | Vector Laboratories | BA-2000 | 1:500 | |
| Peroxidase conj. anti-rabbit | Thermo Scientific | 31460 | 1:5000 | |
| Peroxidase conj. anti-mouse | Thermo Scientific | 31430 | 1:5000 | |
| Dylight488 anti-rabbit | Abcam | ab96923 | 1:400 | |
| Dylight550 anti-mouse | Abcam | ab96876 | 1:400 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinslechner, K.; Schütz, B.; Pistek, M.; Rapolter, P.; Weitzenböck, H.P.; Hundsberger, H.; Mikulits, W.; Grillari, J.; Röhrl, C.; Hengstschläger, M.; et al. Loss of SR-BI Down-Regulates MITF and Suppresses Extracellular Vesicle Release in Human Melanoma. Int. J. Mol. Sci. 2019, 20, 1063. https://doi.org/10.3390/ijms20051063
Kinslechner K, Schütz B, Pistek M, Rapolter P, Weitzenböck HP, Hundsberger H, Mikulits W, Grillari J, Röhrl C, Hengstschläger M, et al. Loss of SR-BI Down-Regulates MITF and Suppresses Extracellular Vesicle Release in Human Melanoma. International Journal of Molecular Sciences. 2019; 20(5):1063. https://doi.org/10.3390/ijms20051063
Chicago/Turabian StyleKinslechner, Katharina, Birgit Schütz, Martina Pistek, Philipp Rapolter, Hans P. Weitzenböck, Harald Hundsberger, Wolfgang Mikulits, Johannes Grillari, Clemens Röhrl, Markus Hengstschläger, and et al. 2019. "Loss of SR-BI Down-Regulates MITF and Suppresses Extracellular Vesicle Release in Human Melanoma" International Journal of Molecular Sciences 20, no. 5: 1063. https://doi.org/10.3390/ijms20051063