Transcriptome Sequencing Reveals Potential Mechanisms of the Maternal Effect on Egg Diapause Induction of Locusta migratoria
Abstract
:1. Introduction
2. Results
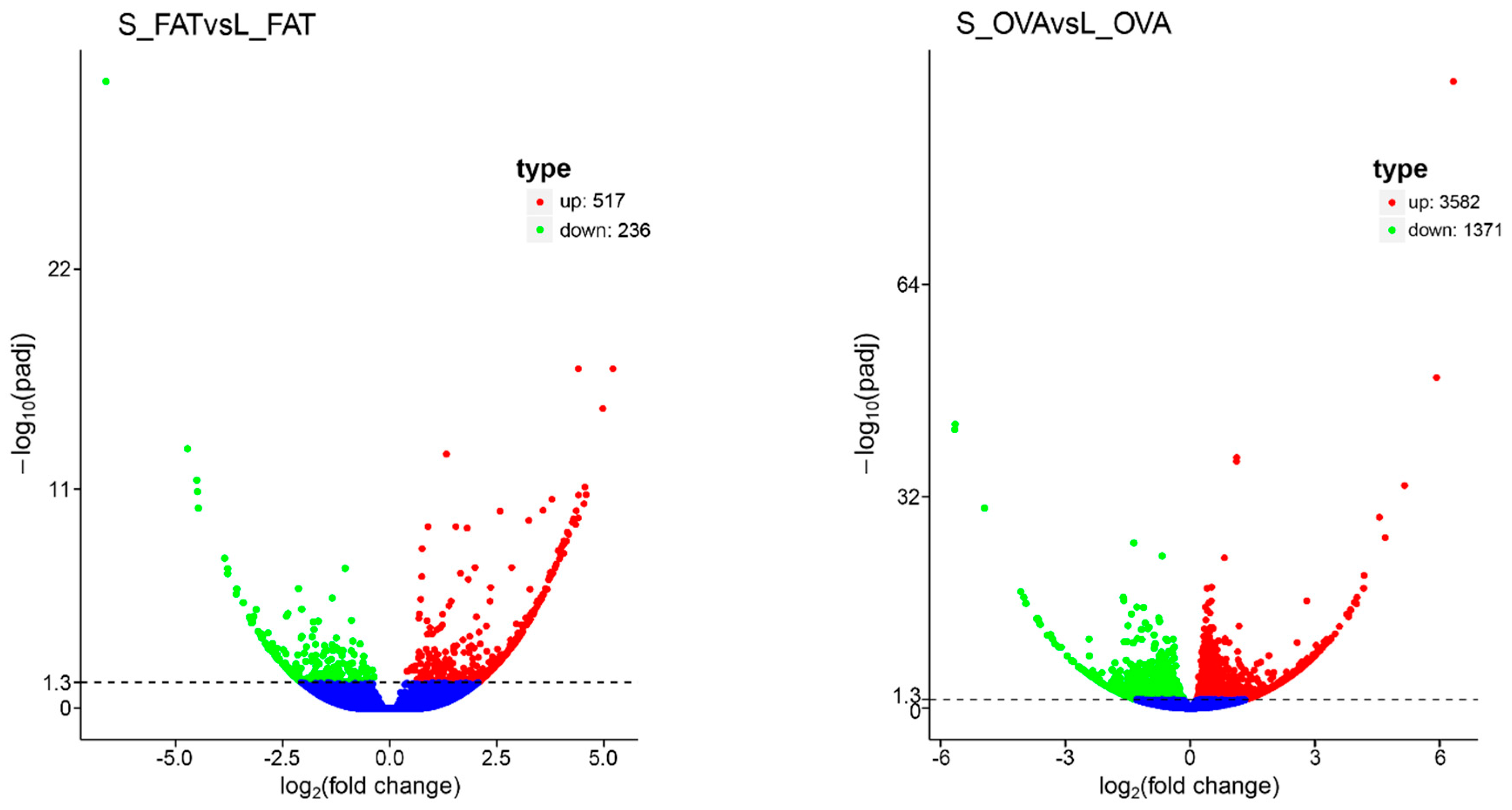
2.1. Transcriptomic Analyses
2.2. KEGG Pathways Analysis
2.3. Validations of DEGs Quantitative Real-Time PCR (qRT-PCR)
2.4. Rai1 and foxo Functions Identified by RNAi
2.5. SOD and ROS Activity Changes after RNAi rai1
3. Discussion
3.1. DEGs and Pathways Related to FOXO Signaling Pathway
3.2. Maternal rai1 Regulates Locust Diapause
4. Materials and Methods
4.1. Insect Rearing and Tissue Collection
4.2. RNA Extraction and RNA-Seq
4.3. Sequence Assembly, Annotation and DEGs Analysis
4.4. cDNA Synthesis and qRT-PCR
4.5. RNA Interference
4.6. Diapause Rate Detection
4.7. SOD and ROS Activity Detection
4.8. Statistical Analysis
4.9. Availability of Data and Materials
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| FAT | fat body |
| OVA | ovary |
| L | long photoperiod |
| S | short photperiod |
| DEGs | differentially expressed genes |
| vs | versus |
| FDR | false discovery rate |
| BLAST | basic local alignment search tool |
| GO | Gene Ontology |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| KO | KEGG Ortholog database |
| KOG/COG | Clusters of Orthologous Groups of proteins |
| NR | NCBI non-redundant protein sequences |
| NT | NCBI nucleotide sequences |
| Pfam | protein families |
| Swiss-Prot | a manually annotated and reviewed protein sequence database |
| TCA cycle | tricarboxylic acid cycle |
| fpps | farnesyl diphosphate synthase |
| JHamt | juvenile hormone acid O-methyltransferase |
| SOD | superoxide dismutase |
| PGM | phosphoglucomutase |
| GAPDH | glyceraldehyde 3-phosphate dehydrogenase |
| ADH | alcohol dehydrogenase |
| MDH | malate dehydrogenase |
| INSR | insulin receptor |
| IRS | insulin receptor substrate |
| EGFR | endothelial growth factor receptor |
| COX | cytochrome c oxidase |
| ND | NADH dehydrogenase |
| DR | diapause rate |
| foxo | foxo gene |
| FOXO | FOXO protein |
References
- Ring, R.A. Maternal induction of diapause in the larva of Lucilia caesar L. (Díptera: Calliphoridae). J. Exp. Biol. 1967, 46, 123–136. [Google Scholar]
- Andrewartha, H.G.; Miethke, P.M.; Wells, A. Induction of diapause in the pupa of Phalaenoides glycinae by a hormone from the suboesophageal ganglion. J. Insect Physiol. 1974, 20, 679–701. [Google Scholar] [CrossRef]
- Chino, H. Conversion of glycogen to sorbitol and glycerol in the diapause egg of the Bombyx silkworm. Nature 1957, 180, 606–607. [Google Scholar] [CrossRef]
- Benoit, J.B.; Denlinger, D.L. Suppression of water loss during adult diapause in the northern house mosquito (Culex pipiens). J Exp Biol. 2007, 210, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Denlinger, D.L. Regulation of diapause. Annu. Rev. Entomol. 2002, 47, 93–122. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Sokabe, T.; Kashio, M.; Yasukochi, Y.; Tominaga, M.; Shiomi, K. Embryonic thermosensitive TRPA1 determines transgenerational diapause phenotype of the silkworm, Bombyx mori. Proc. Natl. Acad. Sci. USA 2014, 111, E1249–E1255. [Google Scholar] [CrossRef] [PubMed]
- Moribe, Y.; Niimi, T.; Yamashita, O.; Yaginuma, T. Samui, a novel cold-inducible gene, encoding a protein with a BAG domain similar to silencer of death domains (SODD/BAG-4), isolated from Bombyx diapause eggs. Eur. J. Biochem. 2001, 268, 3432–3442. [Google Scholar] [CrossRef] [PubMed]
- Sim, C.; Denlinger, D.L. Insulin signaling and FOXO regulate the overwintering diapause of the mosquito Culex pipiens. Proc. Natl. Acad. Sci. USA 2008, 105, 6777–6781. [Google Scholar] [CrossRef] [PubMed]
- Hakomori, T.; Tanaka, S. Genetic control of diapause and other developmental traits in Japanese strains of the migratory locust, Locusta migratoria L.: Univoltine vs bivoltine. Jpn. J. Entomol. 1992, 60, 319–328. [Google Scholar]
- Tanaka, S. The significance of embryonic diapause in a Japanese strain of the migratory locust, Locusta migratoria (Orthoptera: Acrididae). Jpn. J. Entomol. 1992, 60, 503–520. [Google Scholar]
- Tanaka, H. Embryonic diapause and life cycle in the migratory locust, Locusta migratoria L. (Orthoptera: Acrididae), in Kyoto. Appl. Entomol. Zool. 1994, 29, 179–191. [Google Scholar] [CrossRef]
- Baker, D.A.; Russell, S. Gene expression during Drosophila melanogaster egg development before and after reproductive diapause. BMC Genomics 2009, 10, 242. [Google Scholar] [CrossRef]
- Wardhaugh, K.G. The effects of temperature and moisture on the inception of diapause in eggs of the Australian plague locust, Chortoicetes terminifera Walker (Orthoptera: Acrididae). J. Aus. Eco. 2010, 5, 187–191. [Google Scholar] [CrossRef]
- Xian, H.W.; Kang, L. Differences in egg thermotolerance between tropical and temperate populations of the migratory locust, Locusta migratoria (Orthoptera: Acridiidae). J. Insect Physiol. 2005, 51, 1277–1285. [Google Scholar]
- Tu, X.; Wang, J.; Hao, K.; Whitman, D.W.; Fan, Y.; Cao, G.; Zhang, Z. Transcriptomic and proteomic analysis of pre-diapause and non-diapause eggs of migratory locust, Locusta migratoria L. (Orthoptera: Acridoidea). Sci. Rep. 2015, 5, 11402. [Google Scholar] [CrossRef]
- Amsalem, E.; Galbraith, D.A.; Cnaani, J.; Teal, P.E.; Grozinger, C.M. Conservation and modification of genetic and physiological toolkits underpinning diapause in bumble bee queens. Mol. Ecol. 2015, 24, 5596–5615. [Google Scholar] [CrossRef] [PubMed]
- Adkisson, P.L.; Bell, R.A.; Wellso, S.G. Environmental factors controlling the induction of diapause in the pink bollworm, Pectinophora gossypiella (Saunders). J Insect Physiology. 1963, 9, 299–310. [Google Scholar] [CrossRef]
- Wardhaugh, K.G. The effects of temperature and photoperiod on the induction of diapause in eggs of the Australian plague locust, Chortoicetes terminifera (Walker) (Orthoptera: Acrididae). Bull. Entomol. Res. 1980, 70(4), 635–647. [Google Scholar] [CrossRef]
- Saunders, D.S. Larval Diapause induced by a Maternally-operating Photoperiod. Nature 1965, 206, 739–740. [Google Scholar] [CrossRef]
- Chen, Y.R.; Jiang, T.; Zhu, J.; Xie, Y.C.; Tan, Z.C.; Chen, Y.H.; Tang, S.M.; Hao, B.F.; Wang, S.P.; Huang, J.S.; et al. Transcriptome sequencing reveals potential mechanisms of diapause preparation in bivoltine silkworm Bombyx mori (Lepidoptera: Bombycidae). Comp. Biochem. Physiol. 2017, 24, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Kun, H.; Jie, W.; Tu, X.B.; Whitman, D.W.; Zhang, Z.H. Transcriptomic and proteomic analysis of Locusta migratoria eggs at different embryonic stages: Comparison for diapause and non-diapause regimes. J. Integr. Agric. 2017, 16, 1777–1788. [Google Scholar]
- Calixto, A.; Jara, J.S.; Court, F.A. Diapause formation and downregulation of insulin-like signaling via DAF-16/FOXO delays axonal degeneration and neuronal loss. PLoS Genet. 2012, 8, e1003141. [Google Scholar] [CrossRef]
- Almeida, M. Unraveling the role of FoxOs in bone—Insights from mouse models. Bone 2011, 49, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Chiu, J.F.; Mossman, B.T.; Fukagawa, N.K. Down-regulation of anganese-superoxide dismutase through phosphorylation of FOXO3a by Akt in explanted vascular smooth muscle cells from old rats. J. Biol. Chem. 2006, 81, 40429–40439. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.I.; Storey, K.B. Transcriptional regulation of antioxidant enzymes by FoxO1 under dehydration stress. Gene 2011, 485, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Tawfik, A.I.; Tanaka, Y.; Tanaka, S. Possible involvement of ecdysteroids in embryonic diapause of Locusta migratoria. J. Insect Physiol. 2002, 48, 743–749. [Google Scholar] [CrossRef]
- Kidokoro, K.; Iwata, K.I.; Fujiwara, Y.; Takeda, M. Effects of juvenile hormone analogs and 20-hydroxyecdysone on diapause termination in eggs of Locusta migratoria and Oxya yezoensis. J. Insect Physiol. 2006, 52, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.E.; Trobaugh, C.; Béliveau, C.; Richard, T.; Cusson, M. Cloning, expression and characterization of a dipteran farnesyl diphosphate synthase. Insect Bioch. Mol. Biol. 2007, 37, 1198–1206. [Google Scholar] [CrossRef]
- Taban, A.H.; Tittiger, C.; Blomquist, G.J.; Welch, W.H. Isolation and characterization of farnesyl diphosphate synthase from the cotton boll weevil, Anthonomus grandis. Arch. Insect Biochem. Phys. 2009, 71, 88–104. [Google Scholar] [CrossRef]
- Warren, J.T.; Petryk, A.; Marqués, G.; Jarcho, M.; Parvy, J.P.; Dauphin-Villemant, C.; Gilbert, L.I. Molecular and biochemical characterization of two P450 enzymes in the cdysteroidogenic pathway of Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2002, 9, 11043–11048. [Google Scholar] [CrossRef]
- Van Ekert, E.; Heylen, K.; Rougé, P.; Powell, C.A.; Shatters Jr, R.G.; Smagghe, G.; Borovsky, D. Aedes aegypti juvenile hormone acid methyl transferase, the ultimate enzyme in the biosynthetic pathway of juvenile hormone III, exhibits substrate control. J. Insect Phys. 2014, 64, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Shinoda, T.; Itoyama, K. Juvenile hormone acid methyltransferase: A key regulatory enzyme for insect metamorphosis. Proc. Natl. Acad. Sci. USA 2003, 100, 11986–11991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sim, C.; Denlinger, D.L. Insulin signaling and the regulation of insect diapause. Front. Phys. 2013, 4, 189. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.P.; Yin, C.M.; Tatar, M. Mutations in insulin signaling pathway alter juvenile hormone synthesis in Drosophila melanogaster. Gen. Comp. Endocrinol. 2005, 142, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Abrisqueta, M.; Sürencastillo, S.; Maestro, J.L. Insulin receptor-mediated nutritional signalling regulates juvenile hormone biosynthesis and vitellogenin production in the German cockroach. Insect Biochem. Mol. Biol. 2014, 49, 14–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.S.; Wang, T.; Lin, X.W.; Denlinger, D.L.; Xu, W.H. Reactive oxygen species extend insect life span using components of the insulin-signaling pathway. Proc. Natl. Acad. Sci. USA 2017, 114, E7832–E7840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Y.W. RIG-I, a Homolog Gene of RNA Helicase, Is Induced by Retinoic Acid during the Differentiation of Acute Promyelocytic Leukemia Cell. Ph.D. Thesis, Shanghai Second Medical University, Shanghai, China, 1997. AF038963. [Google Scholar]
- Seki, T.; Fujishita, S.; Ito, M.; Matsuoka, N.; Tsukida, K. Retinoid composition in the compound eyes of insects. Exp. Biol. 1987, 47, 95–103. [Google Scholar]
- Veerman, A.; Overmeer, W.P.J.; van Zon, A.Q.; De Boer, J.M.; de Waard, E.R.; Huisman, H.O. Vitamin A is essential for photoperiodic induction of diapause in an eyeless mite. Nature 1983, 302, 248. [Google Scholar] [CrossRef]
- Veerman, A.; Nunes, M.V. Photoperiod reception in spider mites: Photoreceptor, clock and counter. In Photoperiodic regulation of insect and molluscan hormones. Pitman London 1984, 104, 48–64. [Google Scholar]
- Williams, S.R.; Zies, D.; Mullegama, S.V.; Grotewiel, M.S.; Elsea, S.H. Smith-Magenis syndrome results in disruption of CLOCK gene transcription and reveals an integral role for RAI1 in the maintenance of circadian rhythmicity. J. Hum. Genet. 2012, 90, 941–949. [Google Scholar] [CrossRef]
- Ren, S.; Hao, Y.J.; Chen, B.; Yin, Y.P. Global transcriptome sequencing reveals molecular profiles of summer diapause induction stage of onion maggot, Delia antiqua (Diptera: Anthomyiidae). G3 2018, 8, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Poelchau, M.F.; Reynolds, J.A.; Elsik, C.G.; Denlinger, D.L.; Armbruster, P.A. RNA-Seq reveals early distinctions and late convergence of gene expression between diapause and quiescence in the Asian tiger mosquito, Aedes albopictus. J. Exp. Biol. 2013, 216, 4082–4090. [Google Scholar] [CrossRef]
- Kankare, M.; Parker, D.J.; Merisalo, M.; Salminen, T.S.; Hoikkala, A. Transcriptional differences between diapausing and non-diapausing D. montana females reared under the same photoperiod and temperature. PLoS ONE 2016, 11, e0161852. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Yang, Y.P.; Xu, W.H. PTEN expression responds to transcription factor POU and regulates p-AKT levels during diapause initiation in the cotton bollworm, Helicoverpa armigera. Insect Biochem. Mol. Biol. 2018, 100, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.W.; Xu, W.H. Hexokinase is a key regulator of energy metabolism and ROS activity in insect lifespan extension. Aging 2016, 8, 245–258. [Google Scholar] [CrossRef]
- Sim, C.; Denlinger, D.L. Catalase and superoxide dismutase-2 enhance survival and protect ovaries during overwintering diapause in the mosquito Culex pipiens. J. Insect Physiol. 2011, 57, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Sukiban, J.; Bräunig, P.; Mey, J.; Bui-Göbbels, K. Retinoic acid as a survival factor in neuronal development of the grasshopper, Locusta migratoria. Cell Tissue Res. 2014, 358, 303–312. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Chen, Z. Trinity reconstructing a full-length transcriptome without a genome from RNA-Seq data. Nat. Biotech. 2011, 29, 644. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Huber, W. Differential expression analysis for sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef]
- Young, M.D.; Wakefield, M.J.; Smyth, G.K.; Oshlack, A. Gene ontology analysis for RNA-seq accounting for selection bias. Genome Biol. 2010, 11, R14. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]





| Sample | Clean Reads | Clean Bases (G) | Q20 (%) | Number of Transcripts | Number of Unigenes |
|---|---|---|---|---|---|
| L_FAT1 | 78872568 | 11.83 | 95.61 | 260,779 | 102,273 |
| L_FAT2 | 79227948 | 11.88 | 95.62 | ||
| L_FAT3 | 88069778 | 13.21 | 95.86 | ||
| S_FAT1 | 77266106 | 11.59 | 96.06 | ||
| S_FAT2 | 77647066 | 11.65 | 96.78 | ||
| S_FAT3 | 76502530 | 11.48 | 95.71 | ||
| L_OVA1 | 80389650 | 12.06 | 95.58 | 323,527 | 132,147 |
| L_OVA2 | 90730566 | 13.61 | 95.84 | ||
| L_OVA3 | 71022586 | 10.65 | 95.30 | ||
| S_OVA1 | 75628016 | 11.34 | 95.92 | ||
| S_OVA2 | 88328188 | 13.25 | 96.35 | ||
| S_OVA3 | 76316318 | 11.45 | 94.97 |
| Group | Num. | Term | ID | p-Value |
|---|---|---|---|---|
| S_FAT vs. L_FAT up DEGs | 1 | Ribosome | ko03010 | 1.62 × 10−9 |
| 2 | Citrate cycle (TCA cycle) | ko00020 | 0.003078 | |
| 3 | Regulation of actin cytoskeleton | ko04810 | 0.012542 | |
| 4 | Glyoxylate and dicarboxylate metabolism | ko00630 | 0.015929 | |
| 5 | Glycosphingolipid biosynthesis | ko00603 | 0.019268 | |
| 6 | Protein processing in endoplasmic reticulum | ko04141 | 0.02064 | |
| 7 | Endocytosis | ko04144 | 0.021813 | |
| 8 | Glycolysis / Gluconeogenesis | ko00010 | 0.022633 | |
| 9 | Cysteine and methionine metabolism | ko00270 | 0.029743 | |
| 10 | Fc γ R-mediated phagocytosis | ko04666 | 0.029743 | |
| S_FAT vs. L_FAT down DEGs | 1 | Glycine, serine and threonine metabolism | ko00260 | 2.21 × 10−5 |
| 2 | Oxidative phosphorylation | ko00190 | 0.000119 | |
| 3 | Glyoxylate and dicarboxylate metabolism | ko00630 | 0.000133 | |
| 4 | Pentose phosphate pathway | ko00030 | 0.000141 | |
| 5 | Phenylalanine, tyrosine and tryptophan biosynthesis | ko00400 | 0.002921 | |
| 6 | Fructose and mannose metabolism | ko00051 | 0.006848 | |
| 7 | Phenylalanine metabolism | ko00360 | 0.013337 | |
| 8 | Alanine, aspartate and glutamate metabolism | ko00250 | 0.019824 | |
| 9 | Peroxisome | ko04146 | 0.036338 | |
| 10 | Arginine biosynthesis | ko00220 | 0.04393 | |
| S_OVA vs. L_OVA up DEGs | 1 | Protein processing in endoplasmic reticulum | ko04141 | 3.86 × 10−11 |
| 2 | Proteasome | ko03050 | 7.15 × 10−7 | |
| 3 | Spliceosome | ko03040 | 8.36 × 10−5 | |
| 4 | Cell cycle | ko04110 | 0.000107 | |
| 5 | Protein export | ko03060 | 0.000334 | |
| 6 | RNA transport | ko03013 | 0.000422 | |
| 7 | Antigen processing and presentation | ko04612 | 0.000672 | |
| 8 | Ubiquitin mediated proteolysis | ko04120 | 0.001973 | |
| 9 | Pyrimidine metabolism | ko00240 | 0.009155 | |
| 10 | NOD-like receptor signaling pathway | ko04621 | 0.010726 | |
| 11 | DNA replication | ko03030 | 0.013664 | |
| 12 | Toll-like receptor signaling pathway | ko04620 | 0.019418 | |
| 13 | RNA degradation | ko03018 | 0.021142 | |
| 14 | Oocyte meiosis | ko04114 | 0.021603 | |
| 15 | Ribosome | ko03010 | 0.027409 | |
| 16 | Cytosolic DNA-sensing pathway | ko04623 | 0.033442 | |
| 17 | Progesterone-mediated oocyte maturation | ko04914 | 0.034639 | |
| 18 | NF-kappa B signaling pathway | ko04064 | 0.042894 | |
| 19 | N-Glycan biosynthesis | ko00510 | 0.048725 | |
| S_OVA vs. L_OVA down DEGs | 1 | Thyroid hormone signaling pathway | ko04919 | 0.000812 |
| 2 | Focal adhesion | ko04510 | 0.000986 | |
| 3 | Hippo signaling pathway - fly | ko04391 | 0.00245 | |
| 4 | Notch signaling pathway | ko04330 | 0.009375 | |
| 5 | Tight junction | ko04530 | 0.01335 | |
| 6 | Dorso-ventral axis formation | ko04320 | 0.018136 | |
| 7 | Cardiac muscle contraction | ko04260 | 0.021591 | |
| 8 | ECM-receptor interaction | ko04512 | 0.027292 | |
| 9 | Longevity regulating pathway | ko04213 | 0.034335 | |
| 10 | cGMP-PKG signaling pathway | ko04022 | 0.042573 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, K.; Jarwar, A.R.; Ullah, H.; Tu, X.; Nong, X.; Zhang, Z. Transcriptome Sequencing Reveals Potential Mechanisms of the Maternal Effect on Egg Diapause Induction of Locusta migratoria. Int. J. Mol. Sci. 2019, 20, 1974. https://doi.org/10.3390/ijms20081974
Hao K, Jarwar AR, Ullah H, Tu X, Nong X, Zhang Z. Transcriptome Sequencing Reveals Potential Mechanisms of the Maternal Effect on Egg Diapause Induction of Locusta migratoria. International Journal of Molecular Sciences. 2019; 20(8):1974. https://doi.org/10.3390/ijms20081974
Chicago/Turabian StyleHao, Kun, Aftab Raza Jarwar, Hidayat Ullah, Xiongbing Tu, Xiangqun Nong, and Zehua Zhang. 2019. "Transcriptome Sequencing Reveals Potential Mechanisms of the Maternal Effect on Egg Diapause Induction of Locusta migratoria" International Journal of Molecular Sciences 20, no. 8: 1974. https://doi.org/10.3390/ijms20081974





