The Development of Hyaluronan/Fucoidan-Based Nanoparticles as Macrophages Targeting an Epigallocatechin-3-Gallate Delivery System
Abstract
:1. Introduction
2. Results
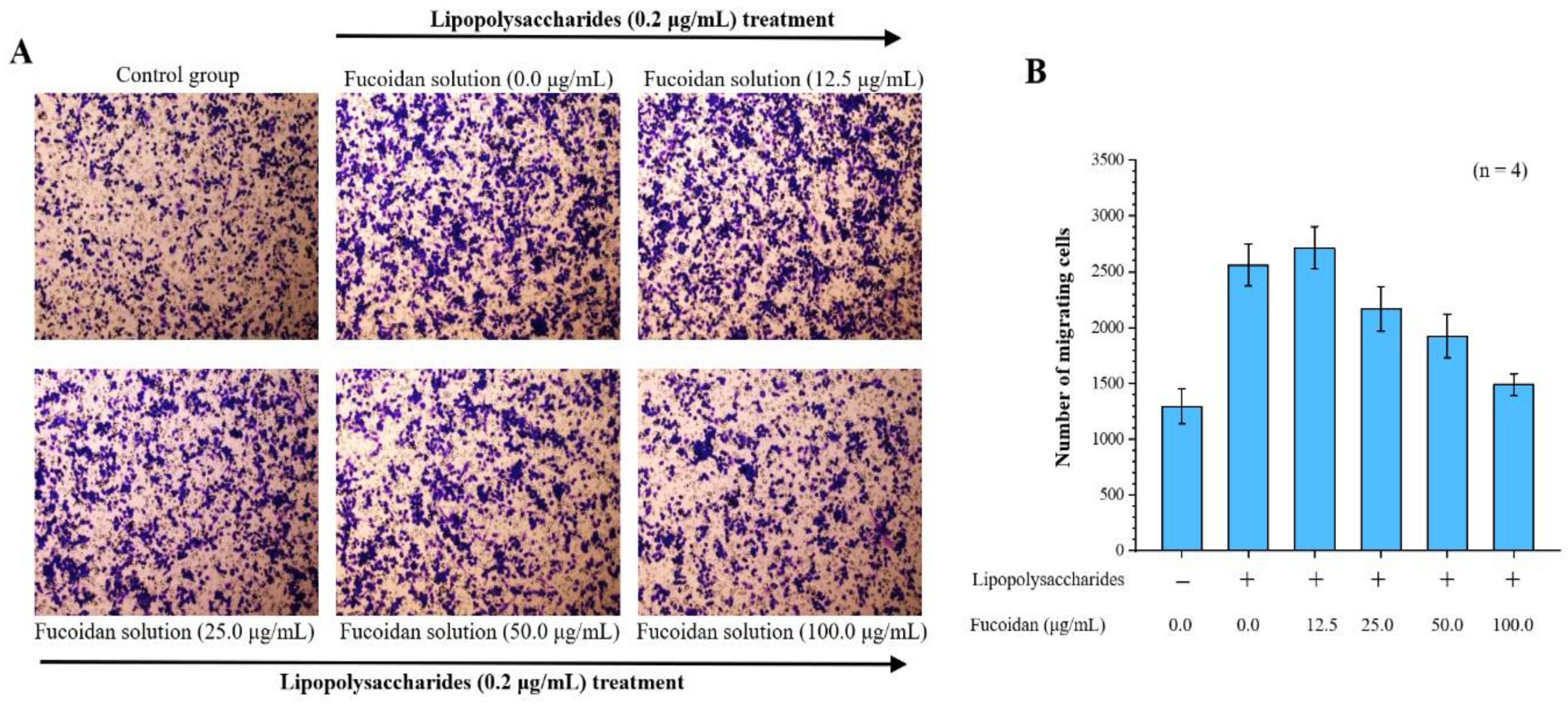
2.1. Effects of Fucoidan on Cell Migration Ability
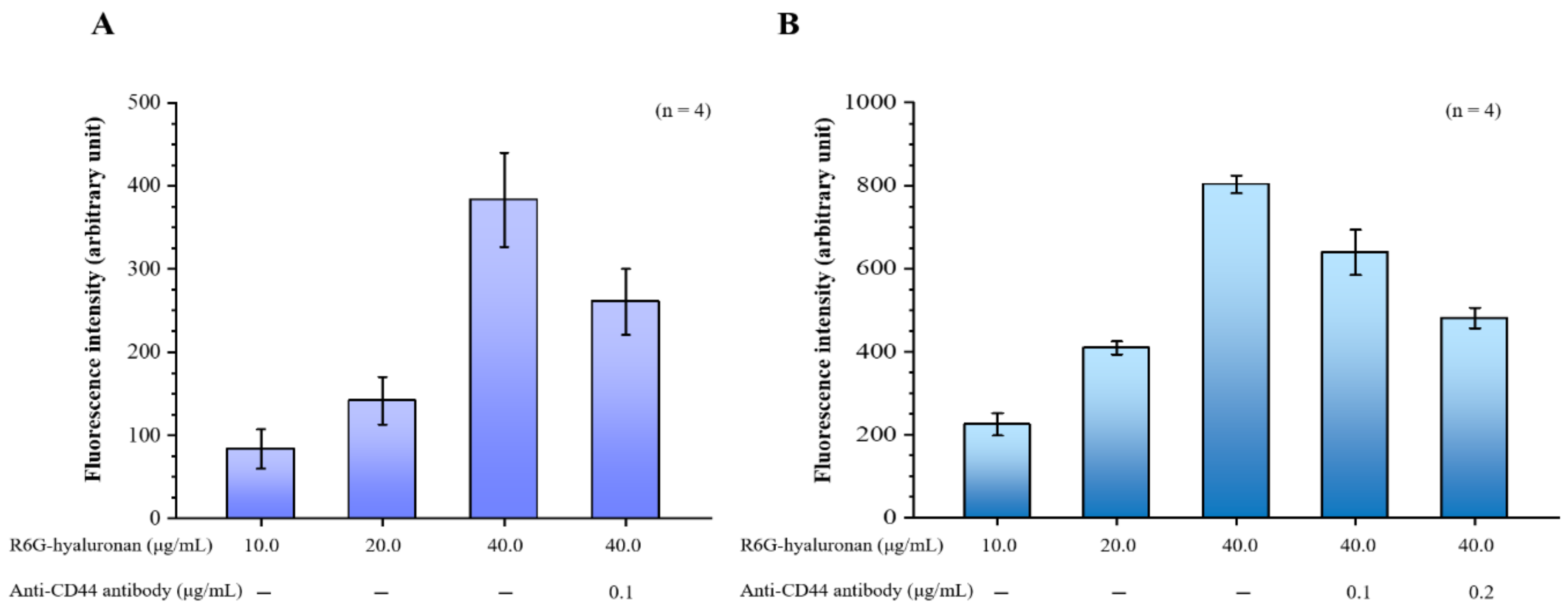
2.2. Effects of Hyaluronan on CD44-Targeting Ability
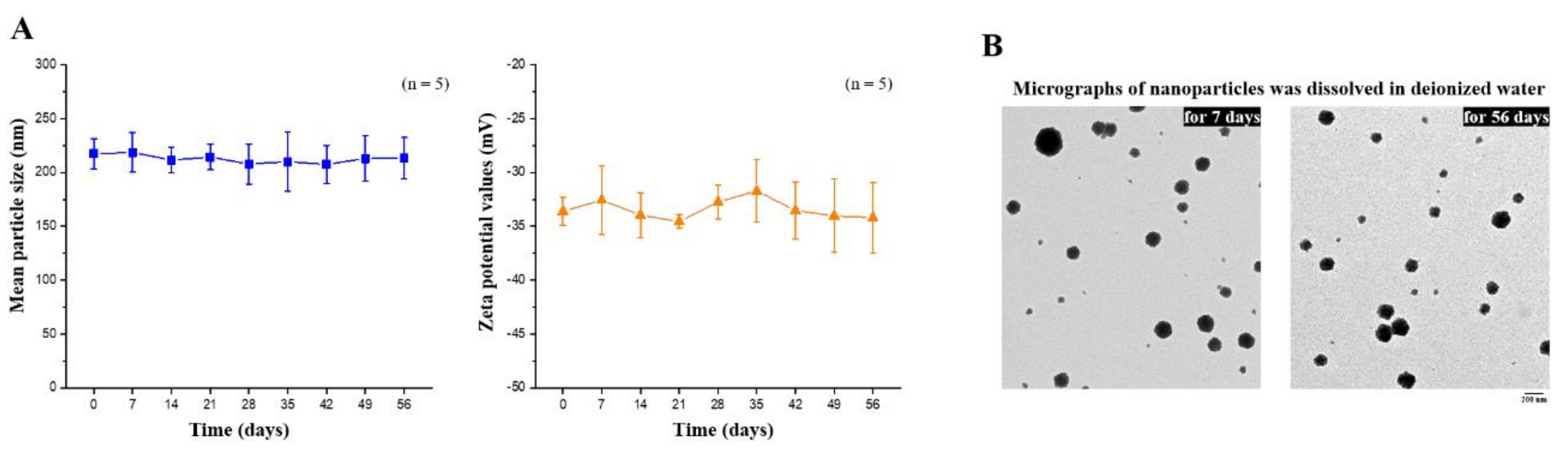
2.3. Synthesis and Characterization of the EGCG-Loaded Hyaluronan/Fucoidan/PEG–Gelatin Nanoparticles
2.4. Macrophage Cellular Internalization of Nanoparticles and Uptake of EGCG
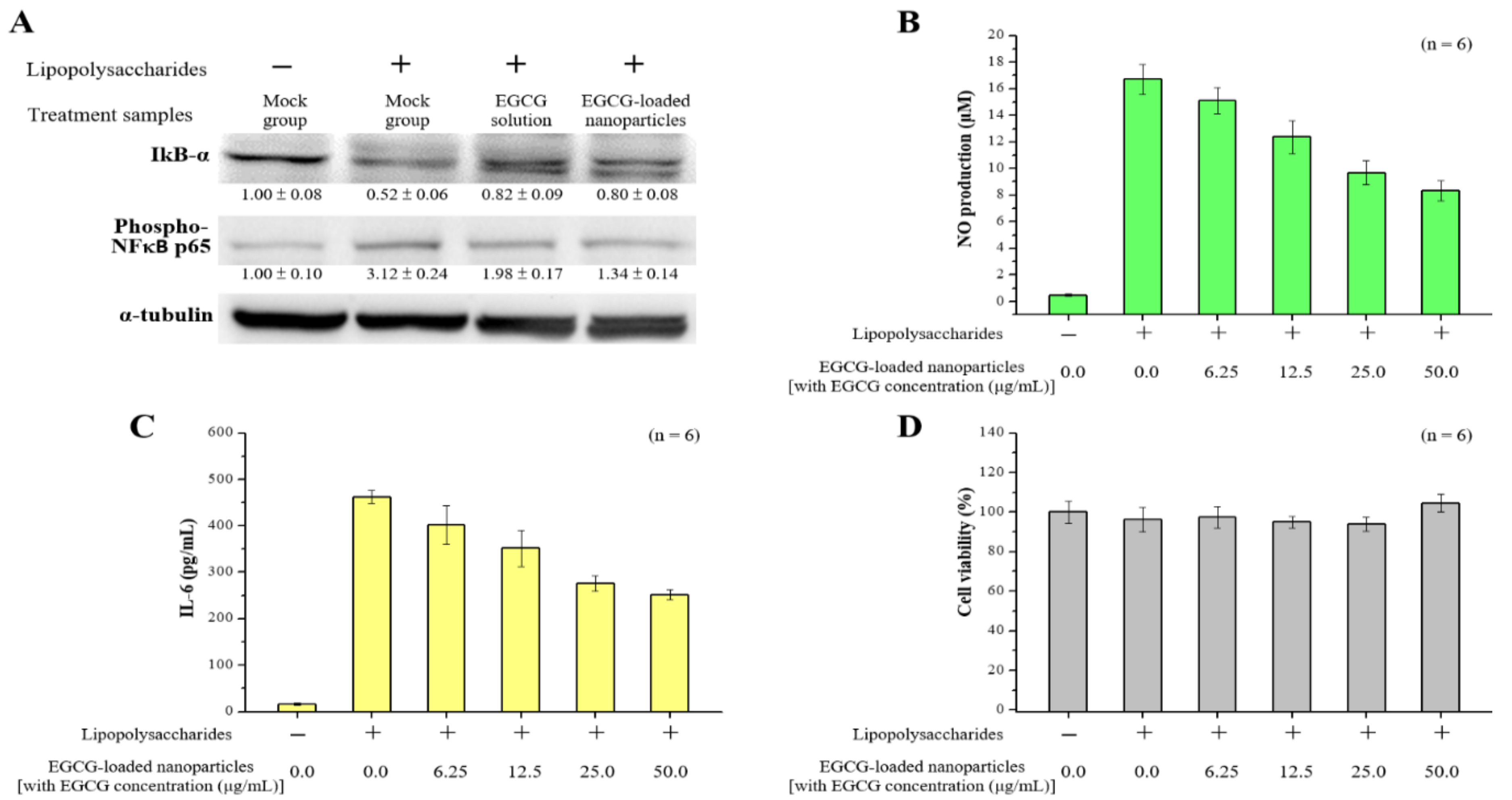
2.5. Anti-Inflammatory Effects of EGCG-Loaded Nanoparticles
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Assay of the Effects of Fucoidan on Macrophage Cell Migration
4.3. Assay of Hyaluronan Binding to CD44 Protein and Macrophage Cells
4.4. Preparation and Characterization of Nanoparticles
4.5. Cellular Internalization of Nanoparticles and the Uptake of EGCG
4.6. Western Blotting Analysis of Inflammatory-Related Proteins
4.7. Assay of Inflammatory Cytokine Interleukin-6 (IL-6) and Nitric Oxide (NO) Levels
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BSA | bovine serum albumin |
| Cy5 | cyanine 5 hydrazide |
| DAPI | 4′,6-diamidino-2-phenylindole |
| EGCG | (−)-epigallocatechin 3-gallate |
| FLA | fluoresceinamine |
| FBS | fetal bovine serum |
| FTIR | Fourier transform infrared spectroscopy |
| HPLC | high-performance liquid chromatography |
| IL-6 | interleukin-6 |
| LPS | lipopolysaccharides |
| NO | nitric oxide |
| NF-κB | nuclear factor-kappa B |
| PEG | polyethylene glycol |
| PBS | phosphate-buffered saline |
| R6G | rhodamine 6G |
| SD | standard deviation |
| TEM | transmission electron microscope |
| VT750 | vivotag 750-NHS ester |
References
- Caprara, G.; Allavena, P.; Erreni, M. Intestinal Macrophages at the Crossroad between Diet, Inflammation, and Cancer. Int. J. Mol. Sci. 2020, 21, 4825. [Google Scholar] [CrossRef] [PubMed]
- Lavin, Y.; Mortha, A.; Rahman, A.; Merad, M. Regulation of Macrophage Development and Function in Peripheral Tissues. Nat. Rev. Immunol. 2015, 15, 731–744. [Google Scholar] [CrossRef] [Green Version]
- Gregory, C.; Devitt, A. The Macrophage and the Apoptotic Cell: An Innate Immune Interaction Viewed Simplistically? Immunology 2004, 113, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sica, A.; Mantovani, A. Macrophage Plasticity and Polarization: In vivo Veritas. J. Clin. Investig. 2012, 122, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Shields, C.W., IV; Wang, L.L.W.; Evans, M.A.; Mitragotri, S. Materials for Immunotherapy. Adv. Mater. 2020, 32, e1901633. [Google Scholar] [CrossRef]
- He, W.; Kapate, N.; Shields, C.W.; Mitragotri, S. Drug Delivery to Macrophages: A Review of Targeting Drugs and Drug Carriers to Macrophages for Inflammatory Diseases. Adv. Drug Deliv. Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Antonov, A.S.; Kolodgie, F.D.; Munn, D.H.; Gerrity, R.G. Regulation of Macrophage Foam Cell Formation by alphaVbeta3 Integrin: Potential Role in Human Atherosclerosis. Am. J. Pathol. 2004, 165, 247–258. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen Recognition and Innate Immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef] [Green Version]
- Gee, K.; Lim, W.; Ma, W.; Nandan, D.; Diaz-Mitoma, F.; Kozlowski, M.; Kumar, A. Differential Regulation of CD44 Expression by Lipopolysaccharide (LPS) and TNF-α in Human Monocytic Cells: Distinct Involvement of c-Jun N-Terminal Kinase in LPS-induced CD44 Expression. J. Immunol. 2002, 169, 5660–5672. [Google Scholar] [CrossRef] [Green Version]
- Naor, D.; Sionov, R.V.; Ish-Shalom, D. CD44: Structure, Function, and Association with the Malignant Process. Adv. Cancer Res. 1997, 71, 241–319. [Google Scholar] [CrossRef]
- Puré, E.; Cuff, C.A. A Crucial Role for CD44 in Inflammation. Trends Mol. Med. 2001, 7, 213–221. [Google Scholar] [CrossRef]
- Siegelman, M.; DeGrendele, H.; Estess, P. Activation and Interaction of CD44 and Hyaluronan in Immunological Systems. J. Leukoc. Biol. 1999, 66, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Cunha, L.; Grenha, A. Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Mar. Drugs 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Zayed, A.; Ulber, R. Fucoidans: Downstream Processes and Recent Applications. Mar. Drugs 2020, 18, 170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakai, T.; Ishizuka, K.; Shimanaka, K.; Ikai, K.; Kato, I. Structures of oligosaccharides derived from Cladosiphon Okamuranus Fucoidan by Digestion with Marine Bacterial Enzymes. Mar. Biotechnol. 2003, 5, 536–544. [Google Scholar] [CrossRef]
- Zhao, Y.; Zheng, Y.; Wang, J.; Ma, S.; Yu, Y.; White, W.L.; Yang, S.; Yang, F.; Lu, J. Fucoidan Extracted from Undaria pinnatifida: Source for Nutraceuticals/Functional Foods. Mar. Drugs 2018, 16, 321. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.J.; Lee, B.Y. Fucoidan from the Sporophyll of Undaria Pinnatifida Suppresses Adipocyte Differentiation by Inhibition of Inflammation-Related Cytokines in 3T3-L1 Cells. Nutr. Res. 2012, 32, 439–447. [Google Scholar] [CrossRef]
- Synytsya, A.; Bleha, R.; Synytsya, A.; Pohl, R.; Hayashi, K.; Yoshinaga, K.; Nakano, T.; Hayashi, T. Mekabu Fucoidan: Structural Complexity and Defensive Effects Against Avian Influenza A viruses. Carbohydr. Polym. 2014, 111, 633–644. [Google Scholar] [CrossRef]
- Lu, M.K.; Cheng, J.J.; Lin, C.Y.; Chang, C.C. Purification, Structural Elucidation, and Anti-Inflammatory Effect of a Water-Soluble 1,6-Branched 1,3-α-d-Galactan from Cultured Mycelia of Poria Cocos. Food Chem. 2010, 118, 349–356. [Google Scholar] [CrossRef]
- Xue, M.; Ge, Y.; Zhang, J.; Liu, Y.; Wang, Q.; Hou, L.; Zheng, Z. Fucoidan Inhibited 4T1 Mouse Breast Cancer Cell Growth in Vivo and in Vitro via Downregulation of Wnt/β-catenin signaling. Nutr. Cancer 2013, 65, 460–468. [Google Scholar] [CrossRef]
- Park, J.H.; Choi, S.H.; Park, S.J.; Lee, Y.J.; Park, J.H.; Song, P.H.; Cho, C.M.; Ku, S.K.; Song, C.H. Promoting Wound Healing Using Low Molecular Weight Fucoidan in a Full-Thickness Dermal Excision Rat Model. Mar. Drugs 2017, 15, 112. [Google Scholar] [CrossRef]
- Li, P.; Wang, H.; Shao, Q.; Kong, B.; Qu, X. Fucoidan Modulates Cytokine Production and Migration of THP-1-Derived Macrophages via Colony-Stimulating Factor-1. Mol. Med. Rep. 2017, 15, 2325–2332. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.Q.; Kim, J.H.; Lee, G.S.; Pyo, H.B.; Shin, E.Y.; Kim, E.G.; Zhang, Y.H. In Vitro Antioxidant and In Vivo Anti-Inflammatory Activities of Ophioglossum thermale. Am. J. Chin. Med. 2012, 40, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Exarchou, V.; Nenadis, N.; Tsimidou, M.; Gerothanassis, I.P.; Troganis, A.; Boskou, D. Antioxidant Activities and Phenolic Composition of Extracts from Greek Oregano, Greek Sage, and Summer Savory. J. Agric. Food Chem. 2002, 50, 5294–5299. [Google Scholar] [CrossRef] [PubMed]
- Muzolf, M.; Szymusiak, H.; Gliszczyńska-Swigło, A.; Rietjens, I.M.; Tyrakowska, B. pH-Dependent Radical Scavenging Capacity of Green Tea Catechins. J. Agric. Food Chem. 2008, 56, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.W.; Yu, S.H.; Ho, Y.C.; Huang, B.Q.; Tsai, G.J.; Hsieh, H.Y.; Sung, H.W.; Mi, F.L. Characterization of Tea Catechins-Loaded Nanoparticles Prepared from Chitosan and an Edible Polypeptide. Food Hydrocoll. 2013, 30, 33–41. [Google Scholar] [CrossRef]
- Ananingsih, V.; Sharma, A.; Zhou, W. Green Tea Catechins during Food Processing and Storage: A Review on Stability and Detection. Food Res. Int. 2013, 50, 469–479. [Google Scholar] [CrossRef]
- Zhong, Y.; Chiou, Y.S.; Pan, M.H.; Shahidi, F. Anti-Inflammatory Activity of Lipophilic Epigallocatechin Gallate (EGCG) Derivatives in LPS-stimulated Murine Macrophages. Food Chem. 2012, 134, 742–748. [Google Scholar] [CrossRef]
- Syed, D.N.; Afaq, F.; Kweon, M.H.; Hadi, N.; Bhatia, N.; Spiegelman, V.S.; Mukhtar, H. Green Tea Polyphenol EGCG Suppresses Cigarette Smoke Condensate-Induced NF-kappaB Activation in Normal Human Bronchial Epithelial Cells. Oncogene 2007, 26, 673–682. [Google Scholar] [CrossRef]
- Chen, C.C.; Hsieh, D.S.; Huang, K.J.; Chan, Y.L.; Hong, P.D.; Yeh, M.K.; Wu, C.J. Improving Anticancer Efficacy of (-)-Epigallocatechin-3-Gallate Gold Nanoparticles in Murine B16F10 Melanoma Cells. Drug Des. Dev. Ther. 2014, 8, 459–474. [Google Scholar] [CrossRef] [Green Version]
- Landis-Piwowar, K.; Huo, C.; Chen, D.; Milacic, V.; Shi, G.; Chan, T.H.; Dou, Q.P. A Novel Prodrug of the Green Tea Polyphenol (-)-Epigallocatechin-3-Gallate as a Potential Anticancer Agent. Cancer Res. 2007, 67, 4303–4310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, W.Y.; Lin, J.N.; Hsieh, J.T.; Chou, S.C.; Lai, C.H.; Yun, E.J.; Lo, U.G.; Pong, R.C.; Lin, J.H.; Lin, Y.H. Nanoparticle Targeting CD44-Positive Cancer Cells for Site-Specific Drug Delivery in Prostate Cancer Therapy. ACS Appl. Mater. Interfaces 2016, 8, 30722–30734. [Google Scholar] [CrossRef] [PubMed]
- Shutava, T.G.; Balkundi, S.S.; Vangala, P.; Steffan, J.; Bigelow, R.L.; Cardelli, J.A.; O’Neal, D.P.; Lvov, Y.M. Layer-by-Layer-Coated Gelatin Nanoparticles as a Vehicle for Delivery of Natural Polyphenols. ACS Nano 2009, 3, 1877–1885. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Inflammation 2010: New Adventures of an Old Flame. Cell 2010, 140, 771–776. [Google Scholar] [CrossRef] [Green Version]
- Ferrero-Miliani, L.; Nielsen, O.H.; Andersen, P.S.; Girardin, S.E. Chronic Inflammation: Importance of NOD2 and NALP3 in Interleukin-1β Generation. Clin. Exp. Immunol. 2007, 147, 227–235. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2018, 9, 7204–7218. [Google Scholar] [CrossRef] [Green Version]
- Sladek, Z.; Rysanek, D. Expression of Macrophage CD44 Receptor in the Course of Experimental Inflammatory Response of Bovine Mammary Gland Induced by Lipopolysaccharide and Muramyl Dipeptide. Res. Vet. Sci. 2009, 86, 235–240. [Google Scholar] [CrossRef]
- Alstergren, P.; Zhu, B.; Glogauer, M.; Mak, T.W.; Ellen, R.P.; Sodek, J. Polarization and Directed Migration of Murine Neutrophils is Dependent on Cell Surface Expression of CD44. Cell Immunol. 2004, 231, 146–157. [Google Scholar] [CrossRef]
- Hollingsworth, J.W.; Li, Z.; Brass, D.M.; Garantziotis, S.; Timberlake, S.H.; Kim, A.; Hossain, I.; Savani, R.C.; Schwartz, D.A. CD44 Regulates Macrophage Recruitment to the Lung in Lipopolysaccharide-Induced Airway Disease. Am. J. Respir. Cell Mol. Biol. 2007, 37, 248–253. [Google Scholar] [CrossRef]
- Nagarajan, B.; Sankaranarayanan, N.V.; Desai, U.R. Perspective on Computational Simulations of Glycosaminoglycans. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2019, 9, e1388. [Google Scholar] [CrossRef]
- Harris, E.N.; Baker, E. Role of the Hyaluronan Receptor, Stabilin-2/HARE, in Health and Disease. Int. J. Mol. Sci. 2020, 21, 3504. [Google Scholar] [CrossRef] [PubMed]
- Platt, V.M.; Szoka, F.C. Anticancer Therapeutics: Targeting Macromolecules and Nanocarriers to Hyaluronan or CD44, a Hyaluronan Receptor. Mol. Pharm. 2008, 5, 474–486. [Google Scholar] [CrossRef] [Green Version]
- Shigeishi, H.; Fujimoto, S.; Hiraoka, M.; Ono, S.; Taki, M.; Ohta, K.; Higashikawa, K.; Kamata, N. Overexpression of the Receptor for Hyaluronan-Mediated Motility, Correlates with Expression of Microtubule-Associated Protein in Human Oral Squamous Cell Carcinomas. Int. J. Oncol. 2009, 34, 1565–1571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mizrahy, S.; Raz, S.R.; Hasgaard, M.; Liu, H.; Soffer-Tsur, N.; Cohen, K.; Dvash, R.; Landsman-Milo, D.; Bremer, M.G.E.G.; Moghimi, S.M.; et al. Hyaluronan-Coated Nanoparticles: The Influence of the Molecular Weight on CD44-Hyaluronan Interactions and on the Immune Response. J. Control. Release 2011, 156, 231–238. [Google Scholar] [CrossRef]
- Kamat, M.; El-Boubbou, K.; Zhu, D.C.; Lansdell, T.; Lu, X.; Li, W.; Huang, X. Hyaluronic Acid Immobilized Magnetic Nanoparticles for Active Targeting and Imaging of Macrophages. Bioconj. Chem. 2010, 21, 2128–2135. [Google Scholar] [CrossRef]
- Montanari, E.; Mancini, P.; Galli, F.; Varani, M.; Santino, I.; Coviello, T.; Mosca, L.; Matricardi, P.; Rancan, F.; Meo, C.D. Biodistribution and Intracellular Localization of Hyaluronan and its Nanogels. A Strategy to Target Intracellular, S. Aureus in Persistent Skin Infections. J. Control. Release 2020, 326, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lepperdinger, G.; Strobl, B.; Kreil, G. HYAL2, A Human Gene Expressed in Many Cells, Encodes a Lysosomal Hyaluronidase with a Novel Type of Specificity. J. Biol. Chem. 1998, 273, 22466–22470. [Google Scholar] [CrossRef] [Green Version]
- Evanko, S.P.; Wight, T.N. Intracellular Localization of Hyaluronan in Proliferating Cells. J. Histochem. Cytochem. 1999, 47, 1331–1341. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano Based Drug Delivery Systems: Recent Developments and Future Prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Xing, M.; Cao, Q.; Ji, A.; Liang, H.; Song, S. Biological Activities of Fucoidan and the Factors Mediating Its Therapeutic Effects: A Review of Recent Studies. Mar. Drugs 2019, 17, 183. [Google Scholar] [CrossRef] [Green Version]
- Kwak, J.Y. Fucoidan as a Marine Anticancer Agent in Preclinical Development. Mar. Drugs 2014, 12, 851–870. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghea, W.A.J.P.; Jeon, Y.J. Biological Activities and Potential Industrial Applications of Fucose Rich Sulfated Polysaccharides and Fucoidans Isolated from Brown Seaweeds: A Review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Tengdelius, M.; Gurav, D.; Konradsson, P.; Påhlsson, P.; Griffith, M.; Oommen, O.P. Synthesis and Anticancer Properties of Fucoidan-Mimetic Glycopolymer Coated Gold Nanoparticles. Chem. Commun. 2015, 51, 8532–8535. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.Y.; Chiu, S.L.; Wen, M.H.; Chen, K.Y.; Hua, K.F. Ligands of Macrophage Scavenger Receptor Induce Cytokine Expression via Differential Modulation of Protein Kinase Signaling Pathways. J. Biol. Chem. 2001, 276, 28719–28730. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.S.; Chung, J.Y.; Yang, G.; Chhabra, S.K.; Lee, M.J. Tea and Tea Polyphenols in Cancer Prevention. J. Nutr. 2000, 130, 472S–478S. [Google Scholar] [CrossRef]
- Lin, J.K.; Liang, Y.C.; Lin-Shiau, S.Y. Cancer Chemoprevention by Tea Polyphenols through Mitotic Signal Transduction Blockade. Biochem. Pharmacol. 1999, 58, 911–915. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [Green Version]
- Eberhardt, M.V.; Lee, C.Y.; Liu, R.H. Antioxidant Activity of Fresh Apples. Nature 2000, 405, 903–904. [Google Scholar] [CrossRef]
- Kanwar, J.; Taskeen, M.; Mohammad, I.; Huo, C.; Chan, T.H.; Dou, Q.P. Recent Advances on Tea Polyphenols. Front. Biosci. 2012, 4, 111–131. [Google Scholar] [CrossRef]
- Singh, B.N.; Shankar, S.; Srivastava, R.K. Green Tea Catechin, Epigallocatechin-3-Gallate (EGCG): Mechanisms, Perspectives and Clinical. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar] [CrossRef] [Green Version]
- Peairs, A.; Dai, R.; Gan, L.; Shimp, S.; Rylander, M.N.; Li, L.; Reilly, C.M. Epigallocatechin-3-Gallate (EGCG) Attenuates Inflammation in MRL/lpr Mouse Mesangial Cells. Cell Mol. Immunol. 2010, 7, 123–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nam, N.H. Naturally Occurring NF-κB Inhibitors. Mini-Rev. Med. Chem. 2006, 6, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Samp, M.A.; Iovanac, N.C.; Nolte, A.J. Sodium Alginate Toughening of Gelatin Hydrogels. ACS Biomater. Sci. Eng. 2017, 3, 3176–3182. [Google Scholar] [CrossRef]
- Amano, F.; Noda, T. Improved Detection of Nitric Oxide Radical (NO.) Production in an Activated Macrophage Culture with a Radical Scavenger, Carboxy PTIO, and Griess Reagent. FEBS Lett. 1995, 368, 425–428. [Google Scholar] [CrossRef] [Green Version]



| PEG–Gelatin 3.750 mg/mL; EGCG 2.000 mg/mL | ||||
|---|---|---|---|---|
| Hyaluronan/Fucoidan (mg/mL) | Mean Particle Size (nm) | Polydispersity Indices | Zeta Potential Value (mV) | Egcg Loading Efficiency (%) |
| 0.625:0.625 | 244.00 ± 12.00 | 0.30 ± 0.05 | −29.86 ± 1.56 | 39.86 ± 4.88 |
| 1.250:1.250 | 217.00 ± 14.00 | 0.28 ± 0.07 | −33.60 ± 1.30 | 52.08 ± 5.37 |
| 1.875:1.875 | 279.00 ± 16.00 | 0.38 ± 0.02 | −37.76 ± 0.67 | 62.66 ± 3.21 |
| 2.500:2.500 | 454.00 ± 28.00 | 0.45 ± 0.10 | −37.72 ± 1.16 | 70.12 ± 2.09 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ho, C.-H.; Chu, P.-Y.; Peng, S.-L.; Huang, S.-C.; Lin, Y.-H. The Development of Hyaluronan/Fucoidan-Based Nanoparticles as Macrophages Targeting an Epigallocatechin-3-Gallate Delivery System. Int. J. Mol. Sci. 2020, 21, 6327. https://doi.org/10.3390/ijms21176327
Ho C-H, Chu P-Y, Peng S-L, Huang S-C, Lin Y-H. The Development of Hyaluronan/Fucoidan-Based Nanoparticles as Macrophages Targeting an Epigallocatechin-3-Gallate Delivery System. International Journal of Molecular Sciences. 2020; 21(17):6327. https://doi.org/10.3390/ijms21176327
Chicago/Turabian StyleHo, Chang-Hsun, Pei-Yi Chu, Shin-Lei Peng, Shun-Chih Huang, and Yu-Hsin Lin. 2020. "The Development of Hyaluronan/Fucoidan-Based Nanoparticles as Macrophages Targeting an Epigallocatechin-3-Gallate Delivery System" International Journal of Molecular Sciences 21, no. 17: 6327. https://doi.org/10.3390/ijms21176327





