Spontaneous Right Ventricular Pseudoaneurysms and Increased Arrhythmogenicity in a Mouse Model of Marfan Syndrome
Abstract
1. Introduction
2. Results
2.1. Ultrasound Analysis of Thoracic Aorta Diameter and Cardiac Function
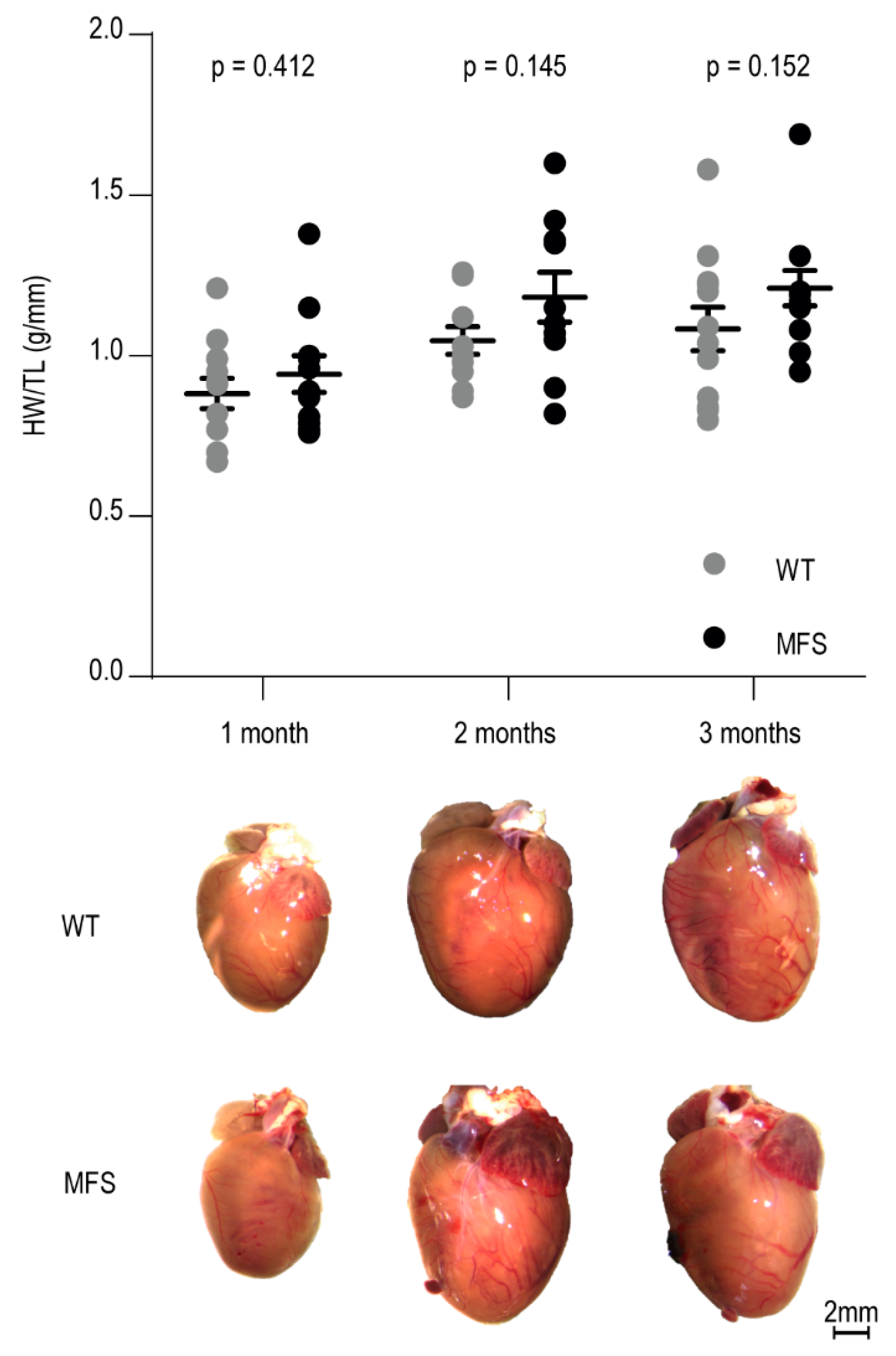
2.2. Macroscopic Analysis of Cardiac Morphology
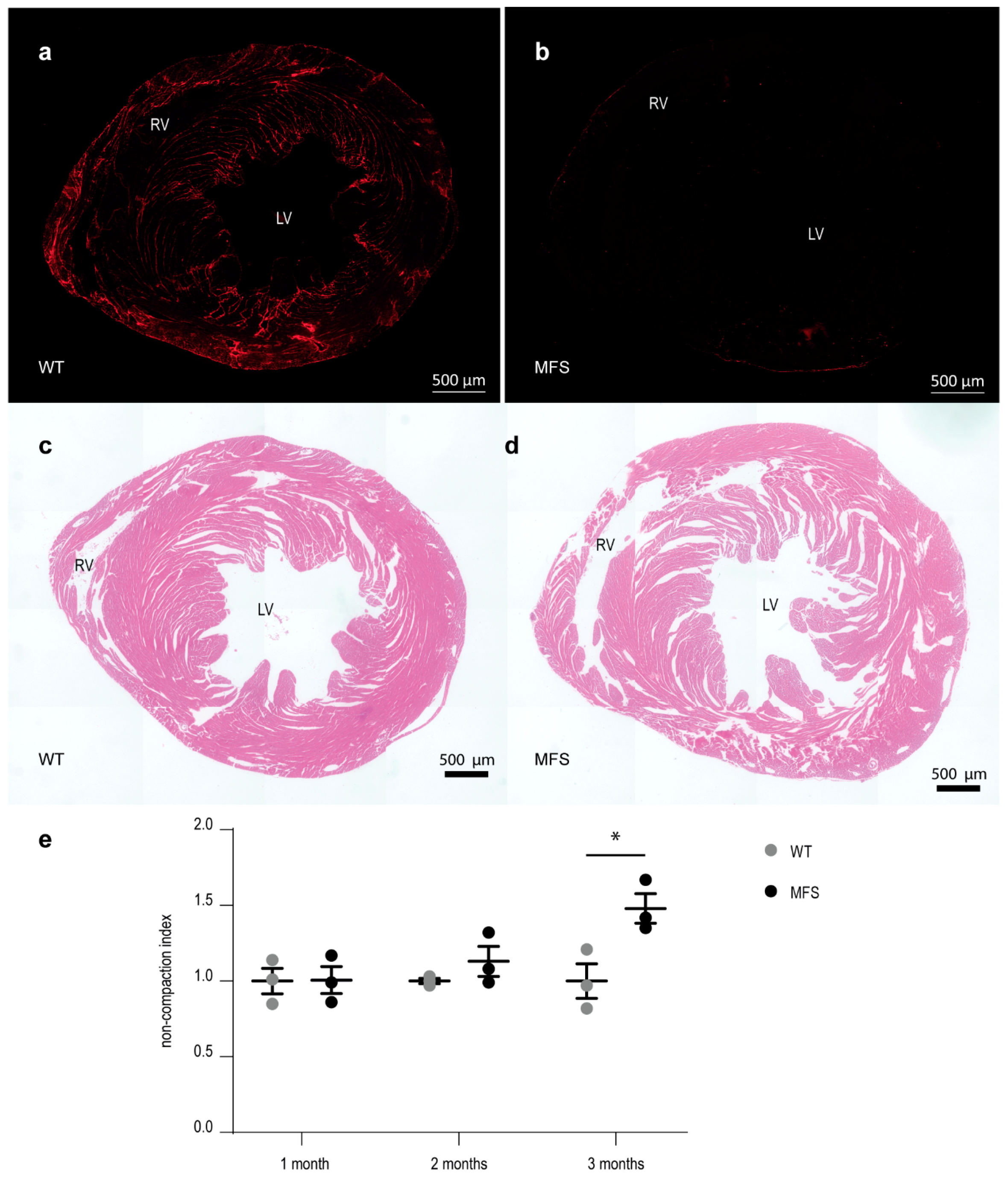
2.3. Myocardial Structure
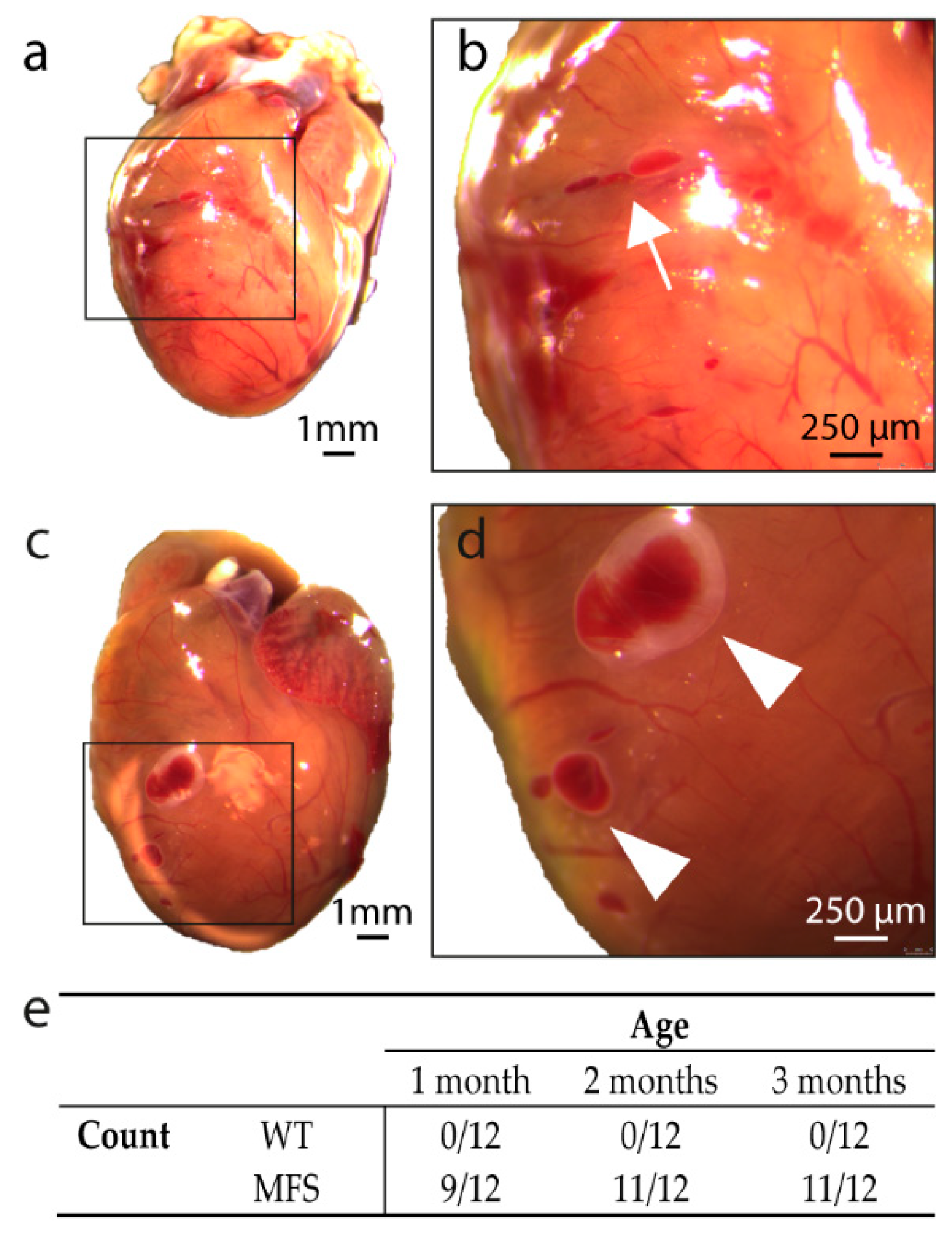
2.3.1. Macroscopic Analysis of Ventricular Wall Morphology
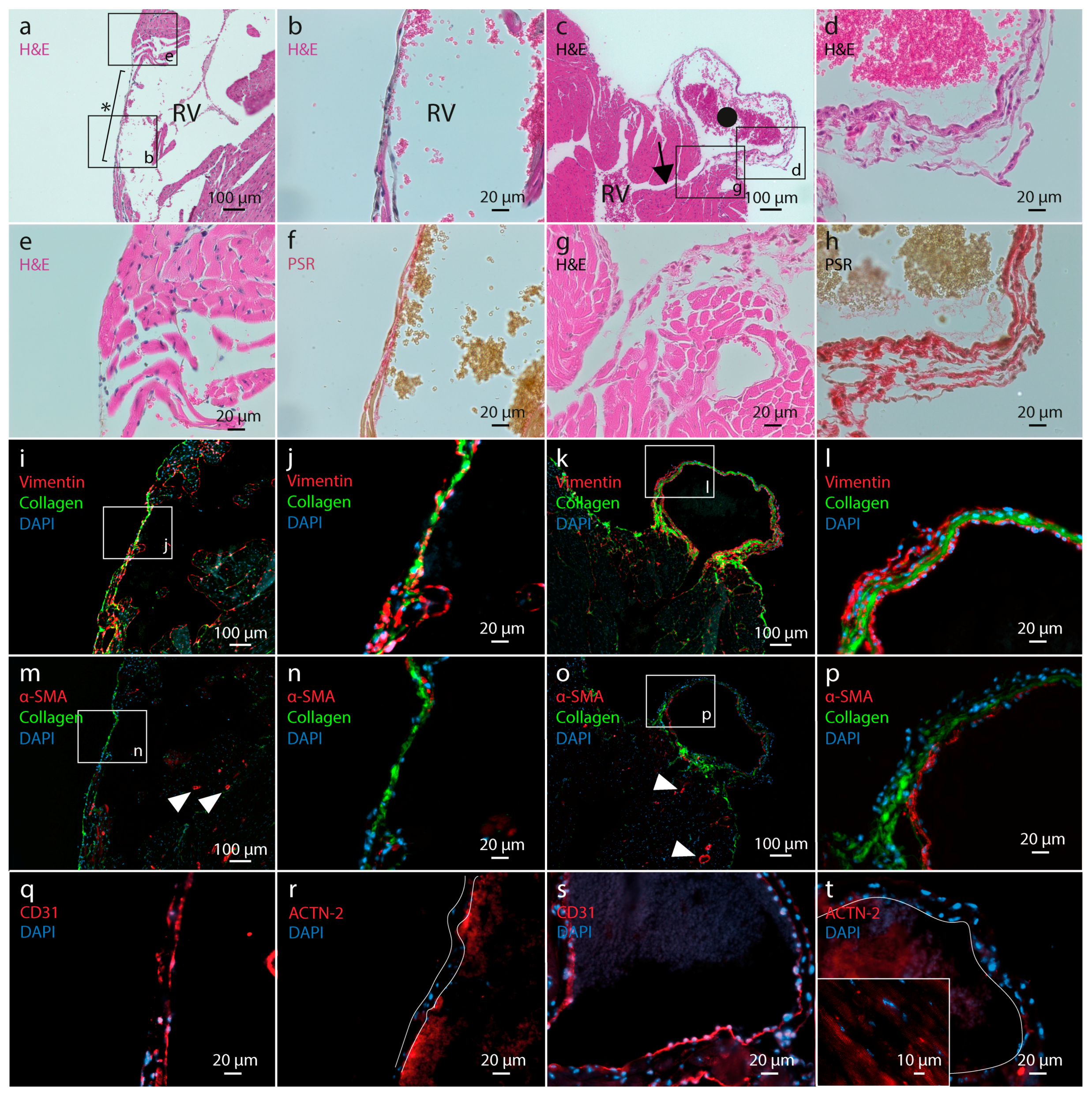
2.3.2. Histological Analysis of Ventricular Wall Lesions
2.3.3. Myocardial Wall Structure
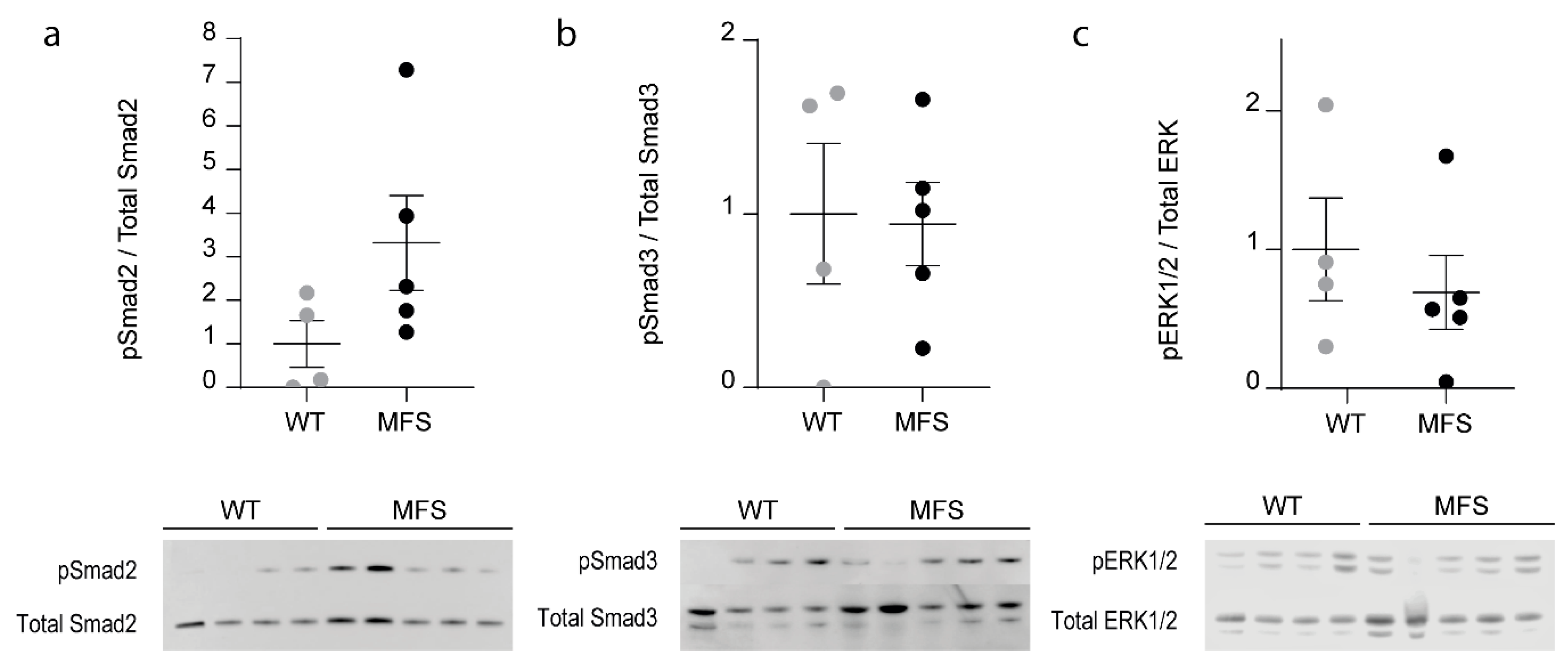
2.4. TGF-β-Dependent Signaling Pathways
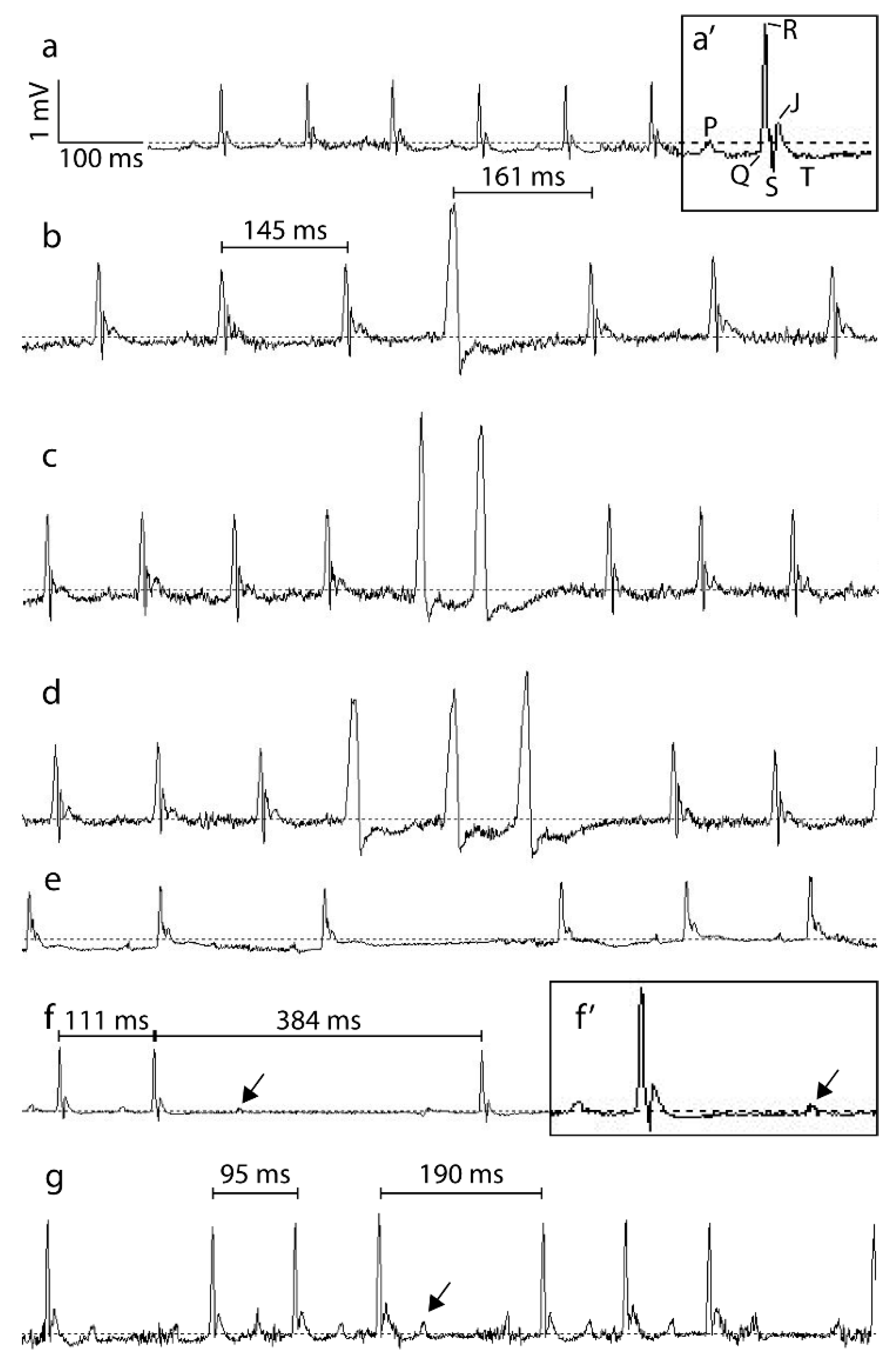
2.5. Electrocardiographic Analysis
2.5.1. Analysis of 24 h Recordings
2.5.2. Analysis of Short-Term Heart Rate Variability
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Ultrasound Analysis
4.3. Electrocardiographic Analysis
4.4. Tissue Isolation
4.5. Histological Analysis
4.6. Myocardial Compaction Calculation
4.7. TGFβ-Dependent Signalling
4.8. Statistics
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Ao | Aorta |
| ECG | Electrocardiogram |
| ECM | Extracellular Matrix |
| Fbn1 | Fibrillin-1 gene |
| FS | Fractional Shortening |
| H&E | Hematoxylin-Eosin |
| HF | High frequency |
| HRV | Heart rate variability |
| HW/TL | Heart weight to tibia length ratio |
| LF | Low frequency |
| LV | Left ventricle |
| LVEDD | Left ventricular end diastolic diameter |
| LVESD | Left ventricular end systolic diameter |
| LVNC | Left ventricular non-compaction |
| MFS | Marfan syndrome |
| PSD | Power spectral density |
| PSR | Picrosirius red |
| RV | Right ventricle |
| RVEDD | Right ventricular end diastolic diameter |
| SDNN | Standard deviation of normal RR-intervals |
| SE | Standard error |
| TGFβ | Transforming growth factor β |
| WT | Wild-type |
Appendix A
Appendix B
Appendix C
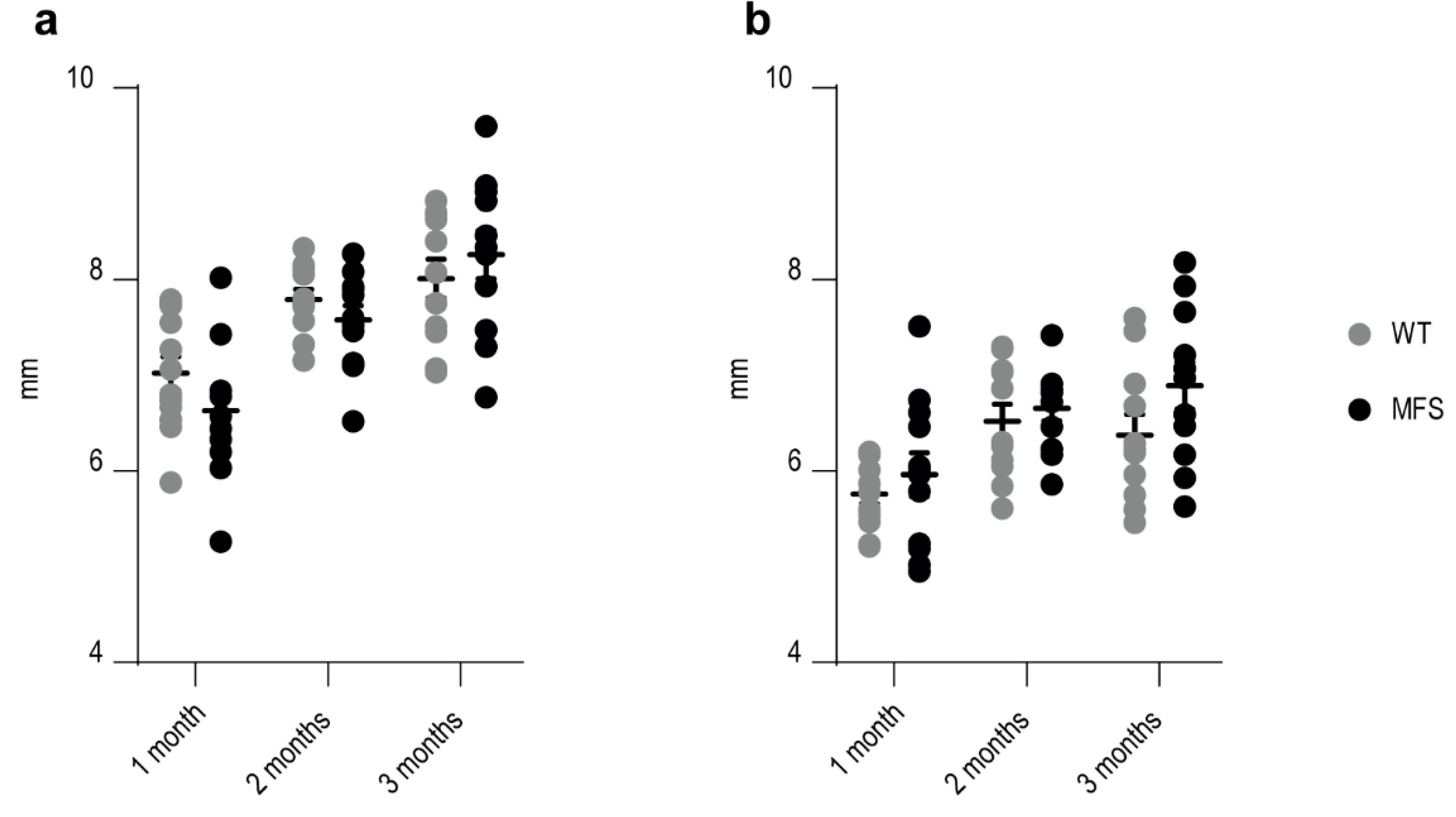
Appendix C.1. Ultrasound Analysis
Appendix C.2. Histological Analysis
Appendix C.3. Myocardial Compaction Calculation
References
- Rienks, M.; Papageorgiou, A.P.; Frangogiannis, N.G.; Heymans, S. Myocardial Extracellular Matrix an Ever-Changing and Diverse Entity. Circ. Res. 2014, 114, 872–888. [Google Scholar] [CrossRef] [PubMed]
- Steijns, F.; van Hengel, J.; Sips, P.; De Backer, J.; Renard, M. A heart for fibrillin: Spatial arrangement in adult wild-type murine myocardial tissue. Histochem. Cell Biol. 2018, 150, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Bouzeghrane, F.; Reinhardt, D.P.; Reudelhuber, T.L.; Thibault, G. Enhanced expression of fibrillin-1, a constituent of the myocardial extracellular matrix in fibrosis. Am. J. Physiol.-Heart Circ. Physiol. 2005, 289, H982–H991. [Google Scholar] [CrossRef]
- Vracko, R.; Thorning, D.; Frederickson, R.G. Spatial arrangements of microfibrils in myocardial scars—Application of antibody to fibrillin. J. Mol. Cell. Cardiol. 1990, 22, 749–757. [Google Scholar] [CrossRef]
- Groth, K.A.; Stochholm, K.; Hove, H.; Kyhl, K.; Gregersen, P.A.; Vejlstrup, N.; Ostergaard, J.R.; Gravholt, C.H.; Andersen, N.H. Aortic events in a nationwide Marfan syndrome cohort. Clin. Res. Cardiol. 2017, 106, 105–112. [Google Scholar] [CrossRef]
- Taub, C.C.; Stoler, J.M.; Perez-Sanz, T.; Chu, J.; Isselbacher, E.M.; Picard, M.H.; Weyman, A.E. Mitral Valve Prolapse in Marfan Syndrome: An Old Topic Revisited. Echocardiography 2009, 26, 357–364. [Google Scholar] [CrossRef]
- Silverman, D.I.; Burton, K.J.; Gray, J.; Bosner, M.S.; Kouchoukos, N.T.; Roman, M.J.; Boxer, M.; Devereux, R.B.; Tsipouras, P. Life expectancy in the Marfan-syndrome. Am. J. Cardiol. 1995, 75, 157–160. [Google Scholar] [CrossRef]
- De Backer, J.F.; Devos, D.; Segers, P.; Matthys, D.; Francois, K.; Gillebert, T.C.; De Paepe, A.M.; De Sutter, J. Primary impairment of left ventricular function in Marfan syndrome. Int. J. Cardiol. 2006, 112, 353–358. [Google Scholar] [CrossRef]
- Alpendurada, F.; Wong, J.; Kiotsekoglou, A.; Banya, W.; Child, A.; Prasad, S.K.; Pennell, D.J.; Mohiaddin, R.H. Evidence for Marfan cardiomyopathy. Eur. J. Heart Fail. 2010, 12, 1085–1091. [Google Scholar] [CrossRef]
- Hetzer, R.; Siegel, G.; Walter, E.M.D. Cardiomyopathy in Marfan syndrome. Eur. J. Cardio-Thorac. Surg. 2016, 49, 561–568. [Google Scholar] [CrossRef]
- Campens, L.; Renard, M.; Trachet, B.; Segers, P.; Mosquera, L.M.; De Sutter, J.; Sakai, L.; De Paepe, A.; De Backer, J. Intrinsic cardiomyopathy in Marfan syndrome: Results from in-vivo and ex-vivo studies of the Fbn1(C1039G/+) model and longitudinal findings in humans. Pediatric Res. 2015, 78, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Rybczynski, M.; Koschyk, D.H.; Aydin, M.A.; Robinson, P.N.; Brinken, T.; Franzen, O.; Berger, J.; Hofmann, T.; Meinertz, T.; von Kodolitsch, Y. Tissue Doppler imaging identifies myocardial dysfunction in adults with Marfan syndrome. Clin. Cardiol. 2007, 30, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Aydin, A.; Adsay, B.A.; Sheikhzadeh, S.; Keyser, B.; Rybczynski, M.; Sondermann, C.; Detter, C.; Steven, D.; Robinson, P.N.; Berger, J.; et al. Observational Cohort Study of Ventricular Arrhythmia in Adults with Marfan Syndrome Caused by FBN1 Mutations. PLoS ONE 2013, 8, e81281. [Google Scholar] [CrossRef] [PubMed]
- Yetman, A.T.; Bornemeier, R.A.; McCrindle, B.W. Long-term outcome in patients with Marfan syndrome: Is aortic dissection the only cause of sudden death? J. Am. Coll. Cardiol. 2003, 41, 329–332. [Google Scholar] [CrossRef]
- Beroukhim, R.S.; Reed, J.H.; Schaffer, M.S.; Yetman, A.T. Surgical correction of mitral valve prolapse—A cure for recurrent ventricular tachycardia in Marfan syndrome? Pediatric Cardiol. 2006, 27, 755–758. [Google Scholar] [CrossRef]
- Savolainen, A.; Kupari, M.; Toivonen, L.; Viitasalo, M. Abnormal ambulatory electrocardiographic findings in patients with the Marfan syndrome. J. Intern. Med. 1997, 241, 221–226. [Google Scholar] [CrossRef]
- Charbonneau, N.L.; Carlson, E.J.; Tufa, S.; Sengle, G.; Manalo, E.C.; Carlberg, V.M.; Ramirez, F.; Keene, D.R.; Sakai, L.Y. In Vivo Studies of Mutant Fibrillin-1 Microfibrils. J. Biol. Chem. 2010, 285, 24943–24955. [Google Scholar] [CrossRef]
- Cua, M.; Lin, E.; Lee, L.; Sheng, X.Y.; Wong, K.S.K.; Tibbits, G.F.; Beg, M.F.; Sarunic, M.V. Morphological phenotyping of mouse hearts using optical coherence tomography. J. Biomed. Opt. 2014, 19, 8. [Google Scholar] [CrossRef]
- Lee, L.; Cui, J.Z.; Cua, M.; Esfandiarei, M.; Sheng, X.Y.; Chui, W.A.; Xu, M.H.; Sarunic, M.V.; Beg, M.F.; van Breemen, C.; et al. Aortic and Cardiac Structure and Function Using High-Resolution Echocardiography and Optical Coherence Tomography in a Mouse Model of Marfan Syndrome. PLoS ONE 2016, 11, e0164778. [Google Scholar] [CrossRef]
- Cavanaugh, N.B.; Qian, L.; Westergaard, N.M.; Kutschke, W.J.; Born, E.J.; Turek, J.W. A Novel Murine Model of Marfan Syndrome Accelerates Aortopathy and Cardiomyopathy. Ann. Thorac. Surg. 2017, 104, 657–665. [Google Scholar] [CrossRef]
- Rouf, R.; MacFarlane, E.G.; Takimoto, E.; Chaudhary, R.; Nagpal, V.; Rainer, P.P.; Bindman, J.G.; Gerber, E.E.; Bedja, D.; Schiefer, C.; et al. Nonmyocyte ERK1/2 signaling contributes to load-induced cardiomyopathy in Marfan mice. JCI Insight 2017, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.R.; Carta, L.; Benard, L.; Chemaly, E.R.; Chiu, E.; Rao, S.K.; Hampton, T.G.; Yurchenco, P.; Costa, K.D.; Hajjar, R.J.; et al. Abnormal muscle mechanosignaling triggers cardiomyopathy in mice with Marfan syndrome. J. Clin. Investig. 2014, 124, 1329–1339. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.; Lee, S.Y.; Gayraud, B.; Andrikopoulos, K.; Shapiro, S.D.; Bunton, T.; Biery, N.J.; Dietz, H.C.; Sakai, L.Y.; Ramirez, F. Pathogenetic sequence for aneurysm revealed in mice underexpressing fibrillin-l. Proc. Natl. Acad. Sci. USA 1999, 96, 3819–3823. [Google Scholar] [CrossRef]
- Savolainen, A.; Nisula, L.; Keto, P.; Hekali, P.; Viitasalo, M.; Kaitila, I.; Kupari, M. Left-ventricular function in children with the Marfan-syndrome. Eur. Heart J. 1994, 15, 625–630. [Google Scholar] [CrossRef]
- Das, B.B.; Taylor, A.L.; Yetman, A.T. Left ventricular diastolic dysfunction in children and young adults with Marfan syndrome. Pediatric Cardiol. 2006, 27, 256–258. [Google Scholar] [CrossRef]
- Bendall, J.K.; Heymes, C.; Wright, T.J.F.; Wheatcroft, S.; Grieve, D.J.; Shah, A.M.; Cave, A.C. Strain-dependent variation in vascular responses to nitric oxide in the isolated murine heart. J. Mol. Cell. Cardiol. 2002, 34, 1325–1333. [Google Scholar] [CrossRef][Green Version]
- Hoit, B.D.; Kiatchoosakun, S.; Restivo, J.; Kirkpatrick, D.; Olszens, K.; Shao, H.F.; Pao, Y.H.; Nadeau, J.H. Naturally occurring variation in cardiovascular traits among inbred mouse strains. Genomics 2002, 79, 679–685. [Google Scholar] [CrossRef]
- Stull, L.B.; Hiranandani, N.; Kelley, M.A.; Leppo, M.K.; Marban, E.; Janssen, P.M.L. Murine strain differences in contractile function are temperature- and frequency-dependent. Pflug. Arch. Eur. J. Physiol. 2006, 452, 140–145. [Google Scholar] [CrossRef]
- Reichelt, M.E.; Willems, L.; Hack, B.A.; Peart, J.N.; Headrick, J.P. Cardiac and coronary function in the Langendorff-perfused mouse heart model. Exp. Physiol. 2009, 94, 54–70. [Google Scholar] [CrossRef]
- Barnabei, M.S.; Palpant, N.J.; Metzger, J.M. Influence of genetic background on ex vivo and in vivo cardiac function in several commonly used inbred mouse strains. Physiol. Genom. 2010, 42A, 103–113. [Google Scholar] [CrossRef]
- Buys, E.S.; Raher, M.J.; Kirby, A.; Mohd, S.; Baron, D.M.; Hayton, S.R.; Tainsh, L.T.; Sips, P.Y.; Rauwerdink, K.M.; Yan, Q.S.; et al. Genetic modifiers of hypertension in soluble guanylate cyclase alpha 1-deficient mice. J. Clin. Investig. 2012, 122, 2316–2325. [Google Scholar] [CrossRef]
- Lima, B.L.; Santos, E.J.C.; Fernandes, G.R.; Merkel, C.; Mello, M.R.B.; Gomes, J.P.A.; Soukoyan, M.; Kerkis, A.; Massironi, S.M.G.; Visintin, J.A.; et al. A New Mouse Model for Marfan Syndrome Presents Phenotypic Variability Associated with the Genetic Background and Overall Levels of Fbn1 Expression. PLoS ONE 2010, 5, e14136. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, G.R.; Massironi, S.M.G.; Pereira, L.V. Identification of Loci Modulating the Cardiovascular and Skeletal Phenotypes of Marfan Syndrome in Mice. Sci. Rep. 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Inayat, F.; Ghani, A.R.; Riaz, I.; Ali, N.S.; Sarwar, U.; Bonita, R.; Virk, H.U. Left Ventricular Pseudoaneurysm: An Overview of Diagnosis and Management. J. Investig. Med. High Impact Case Rep. 2018, 6, 2324709618792025. [Google Scholar] [CrossRef] [PubMed]
- Gaddipati, V.C.; Martin, A.I.; Valenzuela, M.O.; Mahmud, A.; Patel, A.A. An Incidentally Detected Right Ventricular Pseudoaneurysm. Case Rep. Cardiol. 2017, 2017, 4352474. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Yang, Y.K.; Yang, K.Q.; Zhang, Y.; Lu, P.P.; Fan, P.; Ma, L.H.; Zhou, X.L. Clinical characteristics and outcomes of left ventricular pseudoaneurysm A retrospective study in a single-center of China. Medicine 2017, 96, 18. [Google Scholar] [CrossRef]
- Janssen, J.; Leiner, T.; Cheriex, E.C. Bilobar apical pseudoaneurysm after left ventricular venting in a Marfan’s patient. Eur. J. Echocardiogr. 2009, 10, 154–155. [Google Scholar] [CrossRef]
- Parent, J.J.; Towbin, J.A.; Jefferies, J.L. Fibrillin-1 Gene Mutations in Left Ventricular Non-compaction Cardiomyopathy. Pediatric Cardiol. 2016, 37, 1123–1126. [Google Scholar] [CrossRef]
- Kwiatkowski, D.; Hagenbuch, S.; Meyer, R. A Teenager with Marfan Syndrome and Left Ventricular Noncompaction. Pediatric Cardiol. 2010, 31, 132–135. [Google Scholar] [CrossRef]
- Kodo, K.; Ong, S.G.; Jahanbani, F.; Termglinchan, V.; Hirono, K.; InanlooRahatloo, K.; Ebert, A.D.; Shukla, P.; Abilez, O.J.; Churko, J.M.; et al. iPSC-derived cardiomyocytes reveal abnormal TGF-beta signalling in left ventricular non-compaction cardiomyopathy. Nat. Cell Biol. 2016, 18, 1031–1042. [Google Scholar] [CrossRef]
- Chen, Q.; Chen, H.Y.; Zheng, D.W.; Kuang, C.Z.; Fang, H.; Zou, B.Y.; Zhu, W.Q.; Bu, G.X.; Jin, T.; Wang, Z.Z.; et al. Smad7 Is Required for the Development and Function of the Heart. J. Biol. Chem. 2009, 284, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Matt, P.; Schoenhoff, F.; Habashi, J.; Holm, T.; Van Erp, C.; Loch, D.; Carlson, O.D.; Griswold, B.F.; Fu, Q.; De Backer, J.; et al. Circulating Transforming Growth Factor-beta in Marfan Syndrome. Circulation 2009, 120, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, B.N.; Rybczynski, M.; Sheikhzadeh, S.; Akbulak, R.O.; Moser, J.; Jularic, M.; Schreiber, D.; Daubmann, A.; Willems, S.; von Kodolitsch, Y.; et al. Heart rate turbulence and deceleration capacity for risk prediction of serious arrhythmic events in Marfan syndrome. Clin. Res. Cardiol. 2015, 104, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Cherkas, A.; Zhuraev, R. A marked decrease in heart rate variability in Marfan syndrome patients with confirmed FBN1 mutations. Cardiol. J. 2016, 23, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Muiño-Mosquera, L.; Bauters, F.; Dhondt, K.; De Wilde, H.; Jordaens, L.; De Groote, K.; De Wolf, D.; Hertegonne, K.; De Backer, J. Sleep apnea and the impact on cardiovascular risk in patients with Marfan syndrome. Mol. Genet. Genom. Med. 2019, 7, e805. [Google Scholar] [CrossRef]
- Xhyheri, B.; Manfrini, O.; Mazzolini, M.; Pizzi, C.; Bugiardini, R. Heart Rate Variability Today. Prog. Cardiovasc. Dis. 2012, 55, 321–331. [Google Scholar] [CrossRef]
- Billman, G.E. Heart rate variability—A historical perspective. Front. Physiol. 2011, 2, 86. [Google Scholar] [CrossRef]
- Handlos, P.; Gruszka, T.; Stankova, M.; Marecova, K.; Joukal, M. Biventricular noncompaction cardiomyopathy with malignant arrhythmia as a cause of sudden death. Forensic Sci. Med. Pathol. 2017, 13, 495–499. [Google Scholar] [CrossRef]
- Stollberger, C.; Finsterer, J. Arrhythmias and Left Ventricular Hypertrabeculation/Noncompaction. Curr. Pharm. Des. 2010, 16, 2880–2894. [Google Scholar] [CrossRef]
- Steijns, F.; Toth, M.I.; Demolder, A.; Larsen, L.E.; Desloovere, J.; Renard, M.; Raedt, R.; Segers, P.; De Backer, J.; Sips, P. Ambulatory Electrocardiographic Monitoring and Ectopic Beat Detection in Conscious Mice. Sensors 2020, 20, 3867. [Google Scholar] [CrossRef]
- Behar, J.A.; Rosenberg, A.A.; Weiser-Bitoun, I.; Shemla, O.; Alexandrovich, A.; Konyukhov, E.; Yaniv, Y. PhysioZoo: A Novel Open Access Platform for Heart Rate Variability Analysis of Mammalian Electrocardiographic Data. Front. Physiol. 2018, 9, 1390. [Google Scholar] [CrossRef] [PubMed]
- Cardiff, R.D.; Miller, C.H.; Munn, R.J. Manual Hematoxylin and Eosin Staining of Mouse Tissue Sections; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2014; Volume 6. [Google Scholar]
- Junqueira, L.C.U.; Bignolas, G.; Brentani, R.R. Picrosirius staining plus polarization microscopy, a specific method for collagen detection in tissue-sections. Histochem. J. 1979, 11, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Gayraud, B.; Keene, D.R.; Sakai, L.Y.; Ramirez, F. New insights into the assembly of extracellular microfibrils from the analysis of the fibrillin 1 mutation in the Tight skin mouse. J. Cell Biol. 2000, 150, 667–679. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
| WT | MFS | p-Value | |
|---|---|---|---|
| Ao Diameters | |||
| Ao Sinus (mm) | 1.61 ± 0.10 | 2.15 ± 0.09 | 0.001 |
| Ao Asc (mm) | 1.55 ± 0.10 | 2.89 ± 0.10 | <0.001 |
| Arch (mm) | 1.43 ± 0.06 | 2.01 ± 0.06 | <0.001 |
| Ao Desc (mm) | 1.15 ± 0.07 | 1.37 ± 0.06 | 0.038 |
| LVESD (mm) | 3.27 ± 0.08 | 3.24 ± 0.16 | ns |
| LVEDD (mm) | 4.20 ± 0.11 | 4.49 ± 0.17 | ns |
| RVEDD (mm) | 1.92 ± 0.10 | 2.11 ± 0.09 | ns |
| LV FS (%) | 22.14 ± 1.30 | 27.71 ± 2.88 | ns |
| E (mm/s) | 625.3 ± 54.2 | 310.5 ± 56.8 | 0.002 |
| A (mm/s) | 386.3 ± 36.5 | 382.0 ± 35.3 | ns |
| E/A | 1.67 ± 0.14 | 0.84 ± 0.14 | 0.002 |
| Extrasystole | Sinus Arrest | Blocked p-Wave | AV-Block | Total | |||
|---|---|---|---|---|---|---|---|
| Single | Doublet | ≥3 | |||||
| WT A | 2 | 0 | 0 | 14 | 12 | 3 | 31 |
| WT B | 33 | 0 | 0 | 27 | 3 | 0 | 63 |
| WT C | 6 | 0 | 0 | 29 | 2 | 0 | 37 |
| WT D | 14 | 1 | 0 | 3 | 1 | 0 | 19 |
| Total Count WT (Relative %) | 55 (36.6%) | 1 (0.7%) | 0 (0.0%) | 73 (48.7%) | 18 (12.0%) | 3 (2.0%) | 150 |
| MFS A | 50 | 4 | 0 | 3 | 7 | 0 | 64 |
| MFS B | 894 | 275 | 2 | 9 | 1 | 0 | 1181 |
| MFS C | 204 | 27 | 1 | 10 | 7 | 0 | 249 |
| MFS D | 41 | 16 | 0 | 17 | 3 | 1 | 78 |
| Total Count MFS (Relative %) | 1189 (75.6%) | 322 (20.5%) | 3 (0.2%) | 39 (2.5%) | 18 (1.2%) | 1 (0.0%) | 1572 |
| p = 0.019 | |||||||
| WT | MFS | p-Value | |
|---|---|---|---|
| SDNN (ms) | 5.31 ± 0.42 | 2.77 ± 0.25 | <0.001 |
| PSD Analysis | |||
| Total Power | 31.37 ± 5.93 | 8.98 ± 2.17 | <0.001 |
| HF | 2.36 ± 0.33 | 0.55 ± 0.04 | <0.001 |
| LF | 10.84 ± 1.82 | 1.06 ± 0.22 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Steijns, F.; Renard, M.; Vanhomwegen, M.; Vermassen, P.; Desloovere, J.; Raedt, R.; Larsen, L.E.; Tóth, M.I.; De Backer, J.; Sips, P. Spontaneous Right Ventricular Pseudoaneurysms and Increased Arrhythmogenicity in a Mouse Model of Marfan Syndrome. Int. J. Mol. Sci. 2020, 21, 7024. https://doi.org/10.3390/ijms21197024
Steijns F, Renard M, Vanhomwegen M, Vermassen P, Desloovere J, Raedt R, Larsen LE, Tóth MI, De Backer J, Sips P. Spontaneous Right Ventricular Pseudoaneurysms and Increased Arrhythmogenicity in a Mouse Model of Marfan Syndrome. International Journal of Molecular Sciences. 2020; 21(19):7024. https://doi.org/10.3390/ijms21197024
Chicago/Turabian StyleSteijns, Felke, Marjolijn Renard, Marine Vanhomwegen, Petra Vermassen, Jana Desloovere, Robrecht Raedt, Lars E. Larsen, Máté I. Tóth, Julie De Backer, and Patrick Sips. 2020. "Spontaneous Right Ventricular Pseudoaneurysms and Increased Arrhythmogenicity in a Mouse Model of Marfan Syndrome" International Journal of Molecular Sciences 21, no. 19: 7024. https://doi.org/10.3390/ijms21197024
APA StyleSteijns, F., Renard, M., Vanhomwegen, M., Vermassen, P., Desloovere, J., Raedt, R., Larsen, L. E., Tóth, M. I., De Backer, J., & Sips, P. (2020). Spontaneous Right Ventricular Pseudoaneurysms and Increased Arrhythmogenicity in a Mouse Model of Marfan Syndrome. International Journal of Molecular Sciences, 21(19), 7024. https://doi.org/10.3390/ijms21197024






