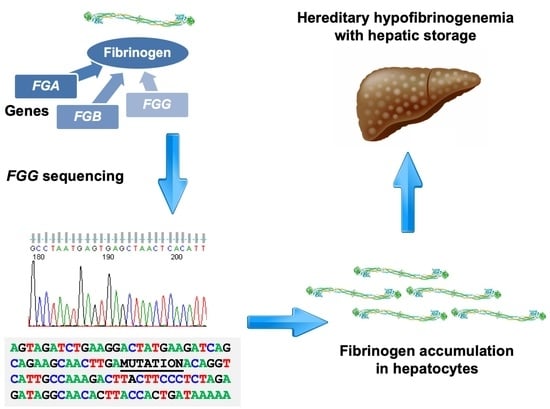
Hereditary Hypofibrinogenemia with Hepatic Storage
Abstract
:1. Fibrinogen and Related Disorders
1.1. Brief Overview on Biosynthesis, Structure, and Function of Fibrinogen
1.2. Fibrinogen-Related Inherited Disorders
2. Hereditary Hypofibrinogenemia with Hepatic Storage (HHHS)
2.1. Brief Historical Notes on HHHS
2.2. Histologic Characteristics of HHHS Inclusions
2.3. Clinic Characteristics of HHHS Patients
2.4. Genetic Basis of HHHS
2.5. Prevalence of HHHS Mutations Worldwide
2.6. Molecular Mechanisms
3. Open Questions and Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Aα | A alpha polypeptide chain |
| ALT | Alanine Aminotransferase |
| APOB | Apolipoprotein B |
| APTT | Activated Partial Thromboplastin Time |
| AST | Aspartate Aminotransferase |
| Bβ | B beta polypeptide chain |
| CBZ | Carbamazepine |
| ChinaMAP | China Metabolic Analytics Project |
| CNX | Calnexin |
| COVID-19 | Coronavirus Disease 2019 |
| CRT | Calreticulin |
| ER | Endoplasmic Reticulum |
| ERp57 | Endoplasmic Reticulum Resident Protein 57 (protein disulfide isomerase homologue) |
| ERSD | Endoplasmic Reticulum Storage Diseases |
| γ | Gamma polypeptide chain |
| FGA | Fibrinogen Alpha Chain Gene |
| FGB | Fibrinogen Beta Chain Gene |
| FGG | Fibrinogen Gamma Chain Gene |
| FSD | Fibrinogen Storage Disease |
| GME | Greater Middle East |
| GnomAD | Genome Aggregation Database |
| GTex | Genotype-Tissue Expression |
| HHHS | Hereditary Hypofibrinogenemia with Hepatic Storage |
| HFSD | Hepatic Fibrinogen Storage Disease |
| INR | International Normalized Ratio |
| MTTP | Microsomal Triglyceride Transfer Protein |
| PT | Prothrombin Time |
| PTT | Partial Thromboplastin Time |
| RCL | Reactive Centre Loop |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus-2 |
| TCT | Thrombin Clotting Time |
| UDCA | Ursodeoxycholic Acid |
| ∆∆G | Protein Folding Free Energy Change |
References
- Mosesson, M.W. Fibrinogen and fibrin structure and functions. J. Thromb. Haemost. 2005, 3, 1894–1904. [Google Scholar] [CrossRef] [PubMed]
- Genotype-Tissue Expression (GTex) Database, v8. Available online: https://www.gtexportal.org/home/ (accessed on 3 August 2020).
- Spraggon, G.; Everse, S.J.; Doolittle, R.F. Crystal structures of fragment D from human fibrinogen and its crosslinked counterpart from fibrin. Nature 1997, 389, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Yee, V.C.; Pratt, K.P.; Côté, H.C.; Trong, I.L.; Chung, D.W.; Davie, E.W.; Stenkamp, R.E.; Teller, D.C. Crystal structure of a 30 kDa C-terminal fragment from the gamma chain of human fibrinogen. Structure 1997, 5, 125–138. [Google Scholar] [CrossRef] [Green Version]
- Madrazo, J.; Brown, J.H.; Litvinovich, S.; Dominguez, R.; Yakovlev, S.; Medved, L.; Cohen, C. Crystal structure of the central region of bovine fibrinogen (E5 fragment) at 1.4-A resolution. Proc. Natl. Acad. Sci. USA 2001, 98, 11967–11972. [Google Scholar] [CrossRef] [Green Version]
- Redman, C.M.; Xia, H. Fibrinogen biosynthesis. Assembly, intracellular degradation, and association with lipid synthesis and secretion. Ann. N. Y. Acad. Sci. 2001, 936, 480–495. [Google Scholar] [CrossRef]
- Tamura, T.; Arai, S.; Nagaya, H.; Mizuguchi, J.; Wada, I. Stepwise assembly of fibrinogen is assisted by the endoplasmic reticulum lectin-chaperone system in HepG2 cells. PLoS ONE 2013, 8, e74580. [Google Scholar] [CrossRef] [Green Version]
- Laurens, N.; Koolwijk, P.; de Maat, M.P.M. Fibrin Structure and Wound Healing. J. Thromb. Haemost. 2006, 4, 932–939. [Google Scholar] [CrossRef]
- Yang, Z.; Mochalkin, I.; Doolittle, R.F. A model of fibrin formation based on crystal structures of fibrinogen and fibrin fragments complexed with synthetic peptides. Proc. Natl. Acad. Sci. USA 2000, 97, 14156–14161. [Google Scholar] [CrossRef] [Green Version]
- Lord, S.T. Molecular mechanisms affecting fibrin structure and stability. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 494–499. [Google Scholar] [CrossRef] [Green Version]
- Kattula, S.; Byrnes, J.R.; Wolberg, A.S. Fibrinogen and Fibrin in Hemostasis and Thrombosis. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e13–e21. [Google Scholar] [CrossRef] [Green Version]
- Wolberg, A.S.; Monroe, D.M.; Roberts, H.R.; Hoffman, M. Elevated prothrombin results in clots with an altered fiber structure: A possible mechanism of the increased thrombotic risk. Blood 2003, 101, 3008–3013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hämisch, B.; Büngeler, A.; Kielar, C.; Keller, A.; Strube, O.; Huber, K. Self-Assembly of Fibrinogen in Aqueous, Thrombin-Free Solutions of Variable Ionic Strengths. Langmuir 2019, 35, 12113–12122. [Google Scholar] [CrossRef] [PubMed]
- Lissitchkov, T.; Madan, B.; Djambas Khayat, C.; Zozulya, N.; Ross, C.; Karimi, M.; Kavakli, K.; De Angulo, G.R.; Almomen, A.; Subramanian, K.; et al. Fibrinogen concentrate for treatment of bleeding and surgical prophylaxis in congenital fibrinogen deficiency patients. J. Thromb. Haemost. 2020, 18, 815–824. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zierk, J.; Ganslandt, T.; Rauh, M.; Metzler, M.; Strasser, E. Data mining of reference intervals for coagulation screening tests in adult patients. Clin. Chim. Acta 2019, 499, 108–114. [Google Scholar] [CrossRef]
- Asselta, R.; Duga, S.; Tenchini, M.L. The molecular basis of quantitative fibrinogen disorders. J. Thromb. Haemost. 2006, 4, 2115–2129. [Google Scholar] [CrossRef] [PubMed]
- De Moerloose, P.; Casini, A.; Neerman-Arbez, M. Congenital fibrinogen disorders: An update. Semin. Thromb. Hemost. 2013, 39, 585–595. [Google Scholar] [CrossRef] [Green Version]
- Casini, A.; de Moerloose, P.; Neerman-Arbez, M. Clinical Features and Management of Congenital Fibrinogen Deficiencies. Semin. Thromb. Hemost. 2016, 42, 366–374. [Google Scholar] [CrossRef]
- Neerman-Arbez, M.; de Moerloose, P.; Casini, A. Laboratory and Genetic Investigation of Mutations Accounting for Congenital Fibrinogen Disorders. Semin. Thromb. Hemost. 2016, 42, 356–365. [Google Scholar] [CrossRef]
- Paraboschi, E.M.; Duga, S.; Asselta, R. Fibrinogen as a Pleiotropic Protein Causing Human Diseases: The Mutational Burden of Aα, Bβ, and γ Chains. Int. J. Mol. Sci. 2017, 18, 2711. [Google Scholar] [CrossRef] [Green Version]
- Casini, A.; Blondon, M.; Tintillier, V.; Goodyer, M.; Sezgin, M.E.; Gunes, A.M.; Hanss, M.; de Moerloose, P.; Neerman-Arbez, M. Mutational Epidemiology of Congenital Fibrinogen Disorders. Thromb. Haemost. 2018, 118, 1867–1874. [Google Scholar] [CrossRef] [Green Version]
- Callea, F.; De Vos, R.; Pinackat, J. Hereditary hypofibrinogenemia with hepatic storage of fibrinogen: A new endoplasmic reticulum storage disease, in Fibrinogen 2. In Biochemistry, Physiology, and Clinical Relevance; Lowe, G.D.O., Douglas, J.T., Forbes, C.D., Henschen, A., Eds.; Elsevier: Amsterdam, The Netherlands, 1987; pp. 75–78. [Google Scholar]
- Callea, F.; Brisigotti, M.; Fabbretti, G.; Bonino, F.; Desmet, V.J. Hepatic endoplasmic reticulum storage diseases. Liver 1992, 12, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, U.; Ormanns, W.; Klinge, O. Hepatocellular fibrinogen storage in familial hypofibrinogenemia. Virchows Arch. B Cell Pathol. Incl. Mol. Pathol. 1981, 36, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Wehinger, H.; Klinge, O.; Alexandrakis, E.; Schürmann, J.; Witt, J.; Seydewitz, H.H. Hereditary hypofibrinogenemia with fibrinogen storage in the liver. Eur. J. Pediatr. 1983, 141, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Brennan, S.O.; Wyatt, J.; Medicina, D.; Callea, F.; George, P.M. Fibrinogen brescia: Hepatic endoplasmic reticulumstorage and hypofibrinogenemia because of a gamma284 Gly→Arg mutation. Am. J. Pathol. 2000, 157, 189–196. [Google Scholar] [CrossRef]
- Zen, Y.; Nishigami, T. Rethinking fibrinogen storage disease of the liver: Ground glass and globular inclusions do not represent a congenital metabolic disorder but acquired collective retention of proteins. Hum. Pathol. 2020, 100, 1–9. [Google Scholar] [CrossRef]
- Callea, F.; Giovannoni, I.; Sari, S.; Guldal, E.; Dalgic, B.; Akyol, G.; Sogo, T.; Al-Hussaini, A.; Maggiore, G.; Bartuli, A.; et al. Fibrinogen Gamma Chain Mutations Provoke Fibrinogen and Apolipoprotein B Plasma Deficiency and Liver Storage. Int. J. Mol. Sci. 2017, 18, 2717. [Google Scholar] [CrossRef] [Green Version]
- Puls, F.; Goldschmidt, I.; Bantel, H.; Agne, C.; Brocker, V.; Dammrich, M.; Lehmann, U.; Berrang, J.; Pfister, E.D.; Kreipe, H.H.; et al. Autophagy-enhancing drug carbamazepine diminishes hepatocellular death in fibrinogen storage disease. J. Hepatol. 2013, 59, 626–630. [Google Scholar] [CrossRef] [Green Version]
- Brennan, S.O.; Davis, R.L.; Conard, K.; Savo, A.; Furuya, K.N. Novel fibrinogen mutation gammaThr-Pro (fibrinogen Al du Pont) associated with hepatic fibrinogen storage disease and hypofibrinogenemia. Liver Int. 2010, 30, 1541–1547. [Google Scholar] [CrossRef]
- Asselta, R.; Robusto, M.; Braidorri, P.; Pevvandi, E.; Nastasio, S.; D’Antiga, O.; Perisic, V.N.; Maggiore, G.; Caccia, S.; Duga, S. Hepatic fibrinogen storage disease: Identification of two novel mutations (p.Asp316Asn, fibrinogen Pisa and p.Gly366Ser, fibrinogen Beograd) impacting on the fibrinogen-module. J. Thromb. Hemost. 2015, 13, 1459–1467. [Google Scholar] [CrossRef] [Green Version]
- Callea, F.; Giovannoni, I.; Sari, S.; Aksu, A.U.; Esendaghy, G.; Dalgic, B.; Boldrini, R.; Akyol, G.; Francalanci, P.; Bellacchio, E. Fibrinogen gamma chain mutation (c.1096C>G p.His340Asp) fibrinogen Ankara, causing hypofibrinogenemia and hepatic storage. Pathology 2017, 49, 534–537. [Google Scholar] [CrossRef]
- Dib, N.; Queloin, F.; Ternisien, C.; Hanss, M.; Michalak, S.; De Mazancourt, P.; Rousselet, M.C.; Cales, P. Fibrinogen angers with a new deletion (gamma GVYY 346–350 causes hypofibrinogenemia with hepatic storage. J. Thromb. Hemost. 2007, 5, 1999–2005. [Google Scholar] [CrossRef] [PubMed]
- Burcu, G.; Bellacchio, E.; Sag, E.; Cebi, A.H.; Saygin, I.; Bahadir, A.; Yilmaz, G.; Corbeddu, M.; Cakir, M.; Callea, F. Structural Characteristics in the γ Chain Variants Associated with Fibrinogen Storage Disease Suggest the Underlying Pathogenic Mechanism. Int. J. Mol. Sci. 2020, 21, 5139. [Google Scholar] [CrossRef] [PubMed]
- Brennan, S.O.; Maghzal, G.; Schneider, B.L.; Gordon, R.; Magid, M.S.; George, P.M. Novel fibrinogen gamma375 Arg→Trp mutation (fibrinogen Aguadilla) causes hepatic endoplasmic reticulum storage and hypofibrinogenemia. Hepatology 2002, 36, 652–658. [Google Scholar] [CrossRef]
- Sari, S.; Yilmaz, G.; Gonul, I.I.; Dalgic, B.; Akyol, G.; Giovannoni, I.; Francalanci, P.; Callea, F. Fibrinogen storage disease and cirrhosis associated with hypobetalipoproteinemia owing to fibrinogen Aguadilla in a Turkish child. Liver Int. 2015, 35, 2501–2505. [Google Scholar] [CrossRef]
- Sogo, T.; Nagasaka, H.; Komatsu, H.; Inui, A.; Miida, T.; Callea, F.; Francalanci, P.; Hirano, K.; Kitamura, H.; Yorifuji, T.; et al. Fibrinogen storage disease caused by Aguadilla mutation presenting with hypobeta-lipoproteinemia and considerable liver disease. J. Pediatr. Gastroenterol. Nutr. 2009, 49, 133–136. [Google Scholar] [CrossRef]
- Rubbia-Brandt, L.; Neerman-Arbez, M.; Rougemont, A.L.; Male, P.J.; Spahr, L. Fibrinogen gamma375 arg→trp mutation (fibrinogen aguadilla) causes hereditary hypofibrinogenemia, hepatic endoplasmic reticulum storage disease and cirrhosis. Am. J. Surg. Pathol. 2006, 30, 906–911. [Google Scholar] [CrossRef]
- Francalanci, P.; Santorelli, F.M.; Talini, I.; Boldrini, R.; Devito, R.; Camassei, F.D.; Maggiore, G.; Callea, F. Severe liver disease in early childhood due to fibrinogen storage and de novo gamma375Arg→Trp gene mutation. J. Pediatr. 2006, 148, 396–398. [Google Scholar] [CrossRef] [PubMed]
- Al-Hussaini, A.; Altalhi, A.; El Hag, I.; AlHussaini, H.; Francalanci, P.; Giovannoni, I.; Callea, F. Hepatic fibrinogen storage disease due to the fibrinogen gamma375 Arg→Trp mutation “fibrinogen Aguadilla” is present in Arabs. Saudi J. Gastroenterol. 2014, 20, 255–261. [Google Scholar] [CrossRef]
- Casini, A.; Sokollik, C.; Lukowski, S.W.; Lurz, E.; Rieubland, C.; de Moerloose, P.; Neerman-Arbez, M. Hypofibrinogenemia and liver disease: A new case of Aguadilla fibrinogen and review of the literature. Haemophilia 2015, 21, 820–827. [Google Scholar] [CrossRef]
- Chang, M.H.; Knisely, A.S.; Wang, N.L.; Gong, J.Y.; Wang, J.S. Fibrinogen storage disease in a Chinese boy with de novo fibrinogen Aguadilla mutation: Incomplete response to carbamazepine and ursodeoxycholic acid. BMC Gastroenterol. 2016, 16, 92. [Google Scholar] [CrossRef] [Green Version]
- Gu, L.; Wang, B.; Liu, L.; Gan, Q.; Liu, X.; Chen, L.; Chen, L. Hepatic fibrinogen storage disease and hypofibrinogenemia caused by fibrinogen Aguadilla mutation: A case report. J. Int. Med. Res. 2020, 48, 300060519898033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maggiore, G.; Nastasio, S.; Sciveres, M. Long-term outcome of liver disease-related fibrinogen aguadilla storage disease in a child. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 699. [Google Scholar] [CrossRef]
- Lee, M.J.; Venick, R.; Bhuta, S.; Li, X.; Wang, H.L. Hepatic Fibrinogen Storage Disease in a Patient with Hypofibrinogenemia: Report of a Case with a Missense Mutation of the FGA Gene. Semin. Liver Dis. 2015, 35, 439–443. [Google Scholar] [CrossRef]
- The Human Fibrinogen Database. Available online: https://site.geht.org/base-de-donnees-fibrinogene/ (accessed on 3 August 2020).
- Peyvandi, F. Epidemiology and treatment of congenital fibrinogen deficiency. Thromb. Res. 2012, 130 (Suppl. 2), S7–S11. [Google Scholar] [CrossRef]
- Orphanet, the Portal for Rare Diseases and Orphan Drugs. Available online: http://www.orpha.net/consor/cgi-bin/index.php?lng=EN (accessed on 15 September 2020).
- The Genome Aggregation Database (GnomAD), Version 2.1.1. Available online: https://gnomad.broadinstitute.org/ (accessed on 3 September 2020).
- The Greater Middle East (GME) Variome Project. Available online: http://igm.ucsd.edu/gme/index.php (accessed on 3 September 2020).
- The China Metabolic Analytics Project (ChinaMAP). Available online: www.mBiobank.com (accessed on 3 September 2020).
- Sobrido, M.J.; Bauer, P.; de Koning, T.; Klopstock, T.; Nadjar, Y.; Patterson, M.C.; Synofzik, M.; Hendriksz, C.J. Recommendations for patient screening in ultra-rare inherited metabolic diseases: What have we learned from Niemann-Pick disease type C? Orphanet J. Rare Dis. 2019, 14, 20. [Google Scholar] [CrossRef] [Green Version]
- Medved, L.; Litvinovich, S.; Ugarova, T.; Matsuka, Y.; Ingham, K. Domain structure and functional activity of the recombinant human fibrinogen gamma-module (gamma148-411). Biochemistry 1997, 36, 4685–4693. [Google Scholar] [CrossRef] [PubMed]
- Gettins, P.G. Serpin structure, mechanism, and function. Chem. Rev. 2002, 102, 4751–4804. [Google Scholar] [CrossRef] [PubMed]
- Yakovlev, S.; Litvinovich, S.; Loukinov, D.; Medved, L. Role of the beta-strand insert in the central domain of the fibrinogen gamma-module. Biochemistry 2000, 39, 15721–15729. [Google Scholar] [CrossRef]
- Tsurupa, G.; Yakovlev, S.; McKee, P.; Medved, L. Noncovalent interaction of alpha(2)-antiplasmin with fibrin(ogen): Localization of alpha(2)-antiplasmin-binding sites. Biochemistry 2010, 49, 7643–7651. [Google Scholar] [CrossRef] [Green Version]
- Lishko, V.K.; Kudryk, B.; Yakubenko, V.P.; Yee, V.C.; Ugarova, T.P. Regulated unmasking of the cryptic binding site for integrin alpha M beta 2 in the gamma C-domain of fibrinogen. Biochemistry 2002, 41, 12942–12951. [Google Scholar] [CrossRef]
- Ekeowa, U.I.; Freeke, J.; Miranda, E.; Gooptu, B.; Bush, M.F.; Pérez, J.; Teckman, J.; Robinson, C.V.; Lomas, D.A. Defining the mechanism of polymerization in the serpinopathies. Proc. Natl. Acad. Sci. USA 2010, 107, 17146–17151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamasaki, M.; Sendall, T.J.; Pearce, M.C.; Whisstock, J.C.; Huntington, J.A. Molecular basis of α1-antitrypsin deficiency revealed by the structure of a domain-swapped trimer. EMBO Rep. 2011, 12, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Kruse, K.B.; Brodsky, J.L.; McCracken, A.A. Autophagy: An ER protein quality control process. Autophagy 2006, 2, 135–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Fourn, V.; Park, S.; Jang, I.; Gaplovska-Kysela, K.; Guhl, B.; Lee, Y.; Cho, J.W.; Zuber, C.; Roth, J. Large protein complexes retained in the ER are dislocated by non-COPII vesicles and degraded by selective autophagy. Cell. Mol. Life Sci. 2013, 70, 1985–2002. [Google Scholar] [CrossRef] [Green Version]
- Simsek, Z.; Ekinci, O.; Cindoruk, M.; Karakan, T.; Degertekin, B.; Akyol, G.; Unal, S. Fibrinogen storage disease without hypofibrinogenemia associated with estrogen therapy. BMC Gastroenterol. 2005, 5, 36. [Google Scholar] [CrossRef] [Green Version]
- Marucci, G.; Morandi, L.; Macchia, S.; Betts, C.M.; Tardio, M.L.; Dal Monte, P.R.; Pession, A.; Foschini, M.P. Fibrinogen storage disease without hypofibrinogenaemia associated with acute infection. Histopathology 2003, 42, 22–25. [Google Scholar] [CrossRef]
- Radhi, J.M.; Lukie, B.E. Pancreatic cancer and fibrinogen storage disease. J. Clin. Pathol. 1998, 51, 865–867. [Google Scholar] [CrossRef] [Green Version]
- Gabay, C.; Kushner, I. Acute-phase proteins and other systemic responses to inflammation. N. Engl. J. Med. 1999, 340, 448–454. [Google Scholar] [CrossRef]
- Fish, R.J.; Neerman-Arbez, M. A novel regulatory element between the human FGA and FGG genes. Thromb. Haemost. 2012, 108, 427–434. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.L.; Wang, X.G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.R.; Zhu, Y.; Li, B.; Huang, C.L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [Green Version]
- Jothimani, D.; Venugopal, R.; Abedin, M.F.; Kaliamoorthy, I.; Rela, M. COVID-19 and the liver. J. Hepatol. 2020, 73, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Covid-19 Recourses of the American Society of Hematology (ASH). Available online: https://www.hematology.org/covid-19/covid-19-and-coagulopathy (accessed on 17 September 2020).
- Fraga, M.; Moradpour, D.; Artru, F.; Romailler, E.; Tschopp, J.; Schneider, A.; Chtioui, H.; Neerman-Arbez, M.; Casini, A.; Alberio, L.; et al. Hepatocellular type II fibrinogen inclusions in a patient with severe COVID-19 and hepatitis. J. Hepatol. 2020, 73, 967–970. [Google Scholar] [CrossRef] [PubMed]

| Mutant Fibrinogen 1 | Country of Origin of the Index Case 1 | Sex | Age (Years) | ALT/AST 2 | Fibrinogen Levels (mg/dL) 3 | Other Coagulation Parameters | Liver Disease | Reference |
|---|---|---|---|---|---|---|---|---|
| Brescia | Italy * | M | 64 | Transaminase elevation | Clauss = 20 Immunoreactive = 100 | TCT = 47 sec | Cirrhosis | [26] |
| Italy | F | 49 | n.r. | 20 | n.r. | Cirrhosis | [28] | |
| Caucasian | M | 5 | Elevated ALT | n.r. | n.r. | Mild liver disease | [29] | |
| AI DuPont | US * | M | 4 | 94/67 | Functional = 47 | PT = 15.3 sec INR = 1.5 | Mild focal portal inflammatory infiltrate | [30] |
| Pisa | Italy * | F | 3 | 169/105 | Functional = 117 Antigenic = 136 | APTT = 33.7 sec TCT = 48 sec | No sign of liver disease | [31] |
| Ankara | Turkey * | F | 5.5 | 46/55 | 55 | PT = 14 sec PTT = 24 sec INR = 1.2 TCT = 15 sec | No sign of liver disease | [32] |
| Angers | France * | F | 35 | 73/51 | Functional = 96 Antigenic = 123 | PT = 18.4 sec | Severe chronic liver disease | [33] |
| Beograd | Serbia * | M | 3 | 308/187 | Functional = 66 Antigenic = 95 | PT = 62.8 sec APTT = 38.2 sec TCT = 28.3 sec | No sign of liver disease | [31] |
| Trabzon | Turkey * | M | 3.5 | 252/144 | Clauss = 36.8 | PT = 19.34 sec APTT = 35.7 sec INR = 1.69 | No sign of liver disease | [34] |
| Aguadilla | Puerto Rico * | F | 3 | 104/- | Clauss = 60 | PT = 17.1 sec | No sign of liver disease | [35] |
| Turkey | F | 2 | 151/77 | 74 | PT = 14.8 PTT = 26 sec INR = 1.27 | Portal and septal fibrosis | [28] | |
| Turkey | F | 5 | 223/411 | 48 | PT = 13.4 sec PTT = 25 sec | Advanced liver fibrosis | [36] | |
| Italy | M | 4 | n.r. | 43 | n.r. | Septal fibrosis | [28] | |
| Japan | M | 2 | 200/190 | 37.6 | PT = 61.1% (n.v., 70–120%) APTT = 32 sec | Early cirrhosis | [37] | |
| Switzerland | M | 61 | 131/109 | 70–80 | PT = 16.5 sec INR = 1.41 | No sign of chronic liver disease; portal hypertension | [38] | |
| Italy | M | 6 | 280/110 | Clauss = 57 | PT = 49% (n.v., 70–120%) PTT = 42 sec | Chronic liver disease | [39] | |
| Caucasian | F | 6 | Elevated ALT | n.r. | PT = prolonged | Mild liver disease | [29] | |
| Siria | F | 3 | 250/185 | 89 | PTT = 40 sec INR = 1 | Hepatomegaly | [40] | |
| Switzerland | F | 4.5 | 125/111 | Clauss = 70 Immunoreactive < 120 | n.r. | Mild liver disease | [41] | |
| China | M | 2 | 529.6/298.2 | 29 | PT = 17.1 sec APTT = 42.8 sec | Portal fibrosis and mild hepatitis | [42] | |
| China | M | 4 | 122/119 | Clauss = 64 | PT = 17.1 sec APTT = 43 sec INR = 1.38 | Hepatomegaly | [43] |
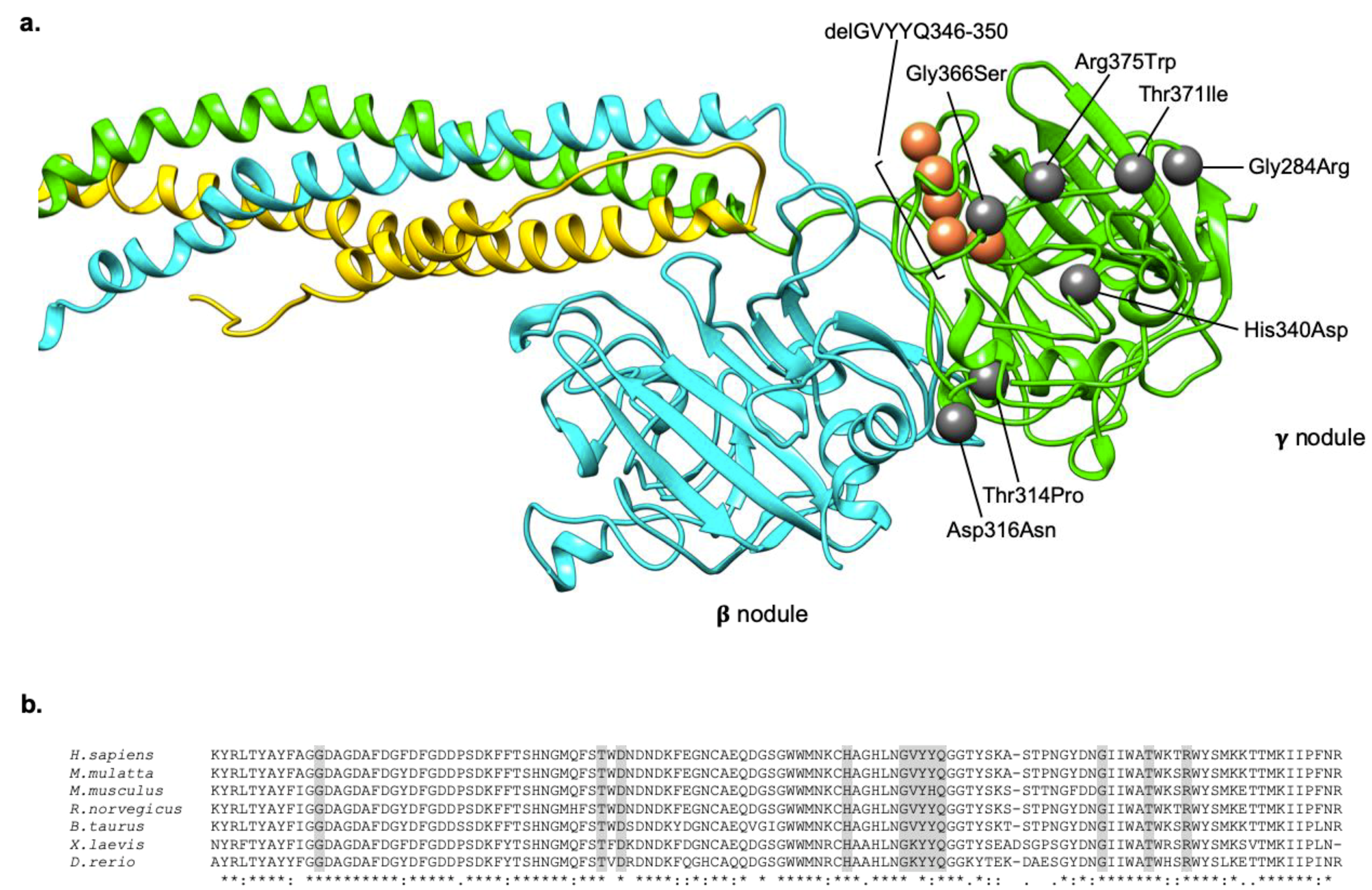
| Mutant Fibrinogen | Country 1 | FGG Exon | Type of Mutation | Chr 4 Position 2 | Nt Variation | Native Protein | Mature Protein | Status | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Brescia | Italy | 8 | Missense | 155,528,058 | G > C | p.Gly310Arg | p.Gly284Arg | Heterozygous | [26] |
| AI DuPont | US | 8 | Missense | 155,527,968 | C > A | p.Thr340Pro | p.Thr314Pro | Heterozygous | [30] |
| Pisa | Italy | 8 | Missense | 155,527,962 | G > A | p.Asp342Asn | p.Asp316Asn | Heterozygous | [31] |
| Ankara | Turkey | 8 | Missense | 155,527,890 | C > G | p.His366Asp | p.His340Asp | Heterozygous | [32] |
| Angers | France | 8 | Deletion + Splicing | 155,527,856–155,527,869 | GTTTATTACCAAGG deletion | p.delGVYYQ 372–376 | p.delGVYYQ 346–350 | Heterozygous | [33] |
| Beograd | Serbia | 9 | Missense | 155,526,174 | G > A | p.Gly392Ser | p.Gly366Ser | Heterozygous | [31] |
| Trabzon | Turkey | 9 | Missense | 155,526,159 | C > T | p.Thr397Ile | p.Thr371Ile | Heterozygous | [34] |
| Aguadilla | Puerto Rico | 9 | Missense | 155,526,147 | C > T | p.Arg401Trp | p.Arg375Trp | Heterozygous | [35] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asselta, R.; Paraboschi, E.M.; Duga, S. Hereditary Hypofibrinogenemia with Hepatic Storage. Int. J. Mol. Sci. 2020, 21, 7830. https://doi.org/10.3390/ijms21217830
Asselta R, Paraboschi EM, Duga S. Hereditary Hypofibrinogenemia with Hepatic Storage. International Journal of Molecular Sciences. 2020; 21(21):7830. https://doi.org/10.3390/ijms21217830
Chicago/Turabian StyleAsselta, Rosanna, Elvezia Maria Paraboschi, and Stefano Duga. 2020. "Hereditary Hypofibrinogenemia with Hepatic Storage" International Journal of Molecular Sciences 21, no. 21: 7830. https://doi.org/10.3390/ijms21217830