The Potential Anticancer Activity of Phytoconstituents against Gastric Cancer—A Review on In Vitro, In Vivo, and Clinical Studies
Abstract
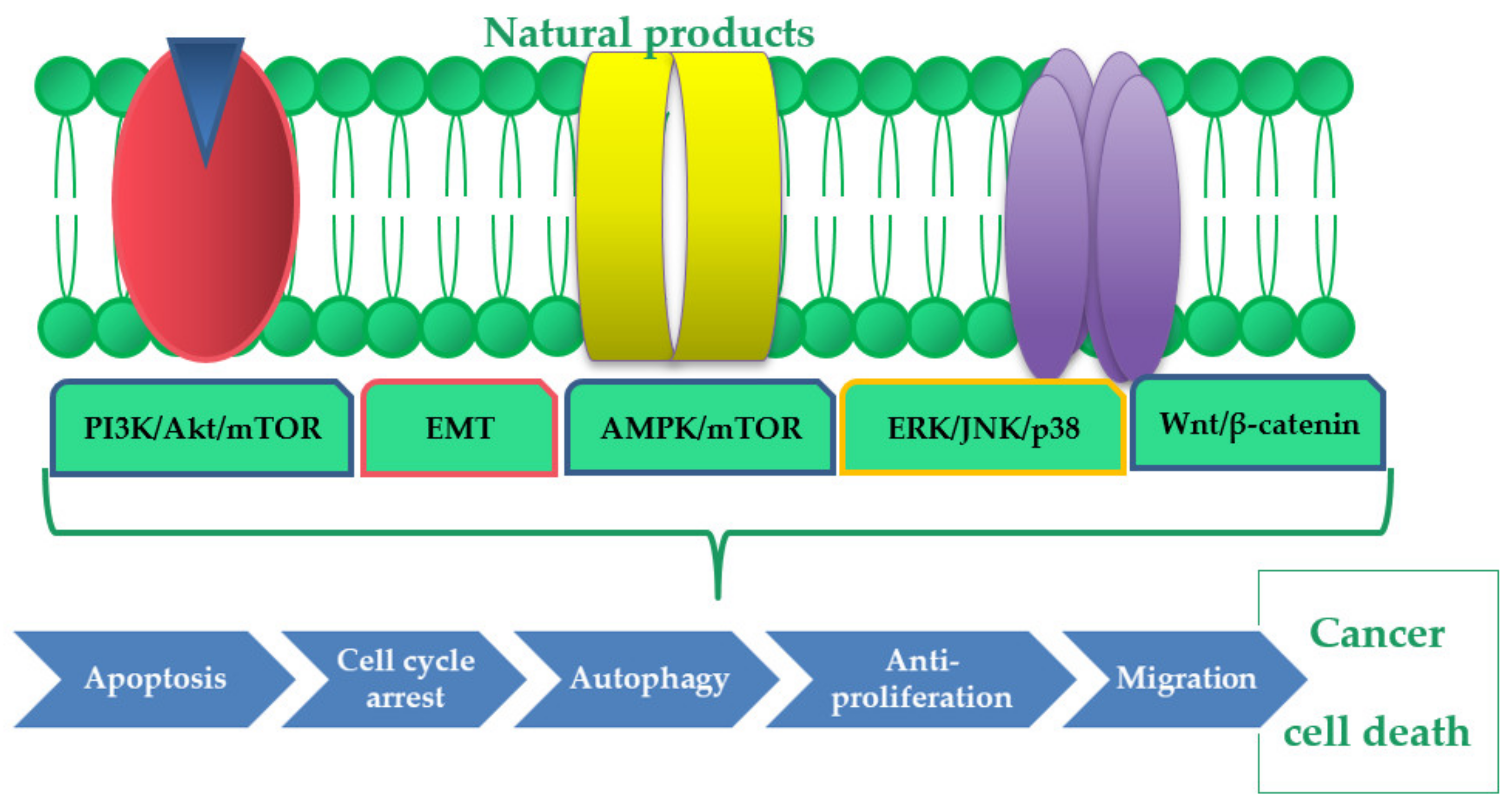
:1. Introduction
2. Biomarkers of Gastric Cancer
3. Phytocompounds with Potential Anticancer Activity on Gastric Cancer
3.1. Primary Metabolites
Cordyceps cicadae
3.2. Secondary Metabolites
3.2.1. Sulphur-Containing Compounds
Allium sativum
3.2.2. Polyphenols
Camellia sinensis
Cardiospermum halicacabum
Plumbago zeylanica
Chrysosplenium nudicaule
3.2.3. Terpenes
Saussurea lappa
Nigella sativa
Euphorbia lunulata
Euphorbia esula
Dioscorea bulbifera
3.2.4. Alkaloids
Coptis chinensis
Stephania tetrandra
Piper longum
Sophora spp.
4. Future Perspectives: In Vivo Studies and Clinical Trials
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GC | Gastric cancer |
| WHO | World Health Organization |
| DNA | deoxyribonucleic acid |
| VEGF | vascular endothelial growth factor |
| IL-8 | interleukin-8 |
| bFGF | basic fibroblast growth factor |
| PD | ECGF- platelet-derived endothelial cell growth factor |
| FGFs | fibroblast growth factors |
| EGF | epidermal growth factor |
| EGF | CFC -epidermal growth factor-CFC |
| TGF-α | transforming growth factor alpha |
| AR | amphiregulin |
| TGF-β | transforming growth factor β |
| IGF II | insulin-like factor II |
| EMT | The epithelial–mesenchymal transition |
| NRP-1 | neutrophilin-1 |
| IL-1 α | interleukin 1-α |
| IL-6 | interleukin 6 |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| IC50 | half-maximal inhibitory concentration |
| ECM | extracellular matrix |
| DAS | diallyl sulfide |
| DADS | diallyl disulfide |
| DATS | diallyltrisulfide |
| MAPK | mitogen-activated protein kinase |
| SH | sulfhydryl group |
| HSPs | heat shock proteins |
| CRP | C Reactive Protein |
| TLR | Toll-like receptor |
| EGCG | epigallocatechin-3-gallate |
| ROS | reactive oxygen species |
| AMPK | activated protein kinase |
| HIF-1α | Hypoxia-inducible factor 1 |
| Bcl-2 | antiapoptotic protein B-cell lymphoma-2 |
| CytC | cytochrome C |
| PARP | Poly (ADP-ribose) polymerase 1 |
| IGF-1R | insulin-like growth factor receptor |
| ERK1/2 | extracellular signal-regulated kinase |
| Akt | RAC-alpha serine/threonine-protein kinase |
| STAT3 | Signal Transducer and Activator of Transcription 3 |
| AP-1 | Activating Protein-1 |
| DNMT | DNA methyltransferase |
| HDAC | histone deacetylases |
| CH-AuNP | gold nanoparticles |
| IKKβ | IKB kinase protein |
| IAP1 | inhibitor of apoptosis 1 |
| XIAP | X-linked inhibitor of apoptosis |
| TTF | 6,7,3′-trimethoxy-3,5,4′-trihydroxy-flavone |
| DTFG | 5,4′-dihydroxy-3,6,3′-trimethoxy-flavone 7-O-β-D-glucoside |
| PARP | poly ADP ribose polymerase |
| TQ | thymoquinone |
| 5-FU | 5-fluorouracil |
| CDDP | cisplatin |
| MDR | cancer multidrug resistance |
| PTEN | phosphatase and tensin homolog |
| BBR | berberine |
| LC3-II | Microtubule-associated protein |
| mTOR | The mammalian target of rapamycin |
| MAPK | mitogen-activated protein kinases |
| MMP | matrix metalloproteinases |
| PL | piperlongumine |
| TrxR | Three thioredoxin reductase |
| MA | matrine |
| OMA | oxymatrine |
| IL-21R | Interleukin-21 receptor |
References
- Rawla, P.; Barsouk, A. Epidemiology of Gastric Cancer: Global Trends, Risk Factors and Prevention. Prz. Gastroenterol. 2019, 14, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Sitarz, R.; Skierucha, M.; Mielko, J.; Offerhaus, G.J.A.; Maciejewski, R.; Polkowski, W.P. Gastric Cancer: Epidemiology, Prevention, Classification, and Treatment. Cancer Manag. Res. 2018, 10, 239–248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ang, T.L.; Fock, K.M. Clinical Epidemiology of Gastric Cancer. Singap. Med. J. 2014, 55, 621–628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tétreault, M.-P.; Katz, J.P. Transgenic Animal Models of Gastrointestinal Function. In Physiology of the Gastrointestinal Tract; Academic Press: Cambridge, MA, USA, 2012; pp. 97–113. [Google Scholar]
- Gullo, I.; Carneiro, F. Gastric Cancer: Pathology and Genetic. Reference Module in Biomedical Sciences, Encyclopedia of Cancer, 3rd ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 77–98. [Google Scholar]
- Ansari, S.; Gantuya, B.; Tuan, V.P.; Yamaoka, Y. Diffuse Gastric Cancer: A Summary of Analogous Contributing Factors for Its Molecular Pathogenicity. Int. J. Mol. Sci. 2018, 19, 2424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, B.; El Hajj, N.; Sittler, S.; Lammert, N.; Barnes, R.; Meloni-Ehrig, A. Gastric Cancer: Classification, Histology and Application of Molecular Pathology. J. Gastrointest. Oncol. 2012, 3, 251–261. [Google Scholar]
- Orditura, M.; Galizia, G.; Sforza, V.; Gambardella, V.; Fabozzi, A.; Laterza, M.; Andreozzi, F.; Ventriglia, J.; Savastano, B.; Mabilia, A.; et al. Treatment of Gastric Cancer. World J. Gastroenterol. 2014, 20, 1635. [Google Scholar] [CrossRef] [PubMed]
- Waddingham, W.; Nieuwenburg, S.A.V.; Carlson, S.; Rodriguez-Justo, M.; Spaander, M.; Kuipers, E.J.; Jansen, M.; Graham, D.G.; Banks, M. Recent Advances in the Detection and Management of Early Gastric Cancer and Its Precursors. Front. Gastroenterol. 2020. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, W.; Jiang, X.; Liu, L.; Wei, K.; Du, H.; Wang, H.; Li, J. Anticancer Effects and Underlying Mechanism of Colchicine on Human Gastric Cancer Cell Lines In Vitro and In Vivo. Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef] [Green Version]
- Choudhari, A.; Mandave, P.; Deshpande, M.; Ranjekar, P.; Prakash, O. Phytochemicals in Cancer Treatment: From Preclinical Studies to Clinical Practice. Front. Pharmacol. 2020, 10, 1614. [Google Scholar] [CrossRef] [Green Version]
- Tahara, E. Growth Factors and Oncogenes in Gastrointestinal Cancers. In Encyclopedia of Molecular Cell Biology and Molecular Medicine; Wiley-VCH GmbH & Co. KGaA: Weinheim, Germany, 2005; Volume 6. [Google Scholar]
- Zavadil, J.; Böttinger, E. TGF-Beta and Epithelial-to-Mesenchymal Transitions. Oncogene 2005, 24, 5764–5774. [Google Scholar] [CrossRef] [Green Version]
- Smith, M.G.; Hold, G.L.; Tahara, E.; El-Omar, E.M. Cellular and Molecular Aspects of Gastric Cancer. World J. Gastroenterol. 2006, 12, 2979–2990. [Google Scholar] [CrossRef] [PubMed]
- Ashizawa, T.; Okada, R.; Suzuki, Y.; Takagi, M.; Yamazaki, T.; Sumi, T.; Aoki, T.; Ohnuma, S.; Aoki, T. Clinical Significance of Interleukin-6 (Il-6) in the Spread of Gastric Cancer: Role of IL-6 as a Prognostic Factor. Gastric Cancer 2005, 8, 124–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, S.C.; Kim, J.H.; Prasad, S.; Aggarwal, B.B. Regulation of Survival, Proliferation, Invasion, Angiogenesis, and Metastasis of Tumor Cells Through Modulation of Inflammatory Pathways by Nutraceuticals. Cancer Metastasis Rev. 2010, 29, 405–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sokolova, O.; Naumann, M. NF-κB Signaling in Gastric Cancer. Toxins 2017, 9, 119. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lv, Y.; Liu, T.S.; Di Yan, W.; Chen, L.Y.; Li, Z.H.; Piao, Y.S.; An, R.B.; Lin, Z.H.; Ren, X.S. Cordycepin Suppresses Cell Proliferation and Migration by Targeting CLEC2 in Human Gastric Cancer Cells via Akt Signaling Pathway. Life Sci. 2019, 223, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Tao, M.; Gao, L.; Pan, J.; Wang, X. Study on the Inhibitory Effect of Allicin on Human Gastric Cancer Cell Line SGC-7901 and Its Mechanism. Afr. J. Tradit. Complement. Altern. Med. 2013, 11, 176–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Ha, M.; Gong, Y.; Xu, Y.; Dong, N.; Yuan, Y. Allicin Induces Apoptosis in Gastric Cancer Cells Through Activation of Both Extrinsic and Intrinsic Pathways. Oncol. Rep. 2010, 24. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Zhu, Y.; Duan, W.; Feng, C.; He, X. Allicin Induces Apoptosis of the MGC-803 Human Gastric Carcinoma Cell Line Through the p38 Mitogen-Activated Protein Kinase/Caspase-3 Signaling Pathway. Mol. Med. Rep. 2014, 11, 2755–2760. [Google Scholar] [CrossRef] [Green Version]
- Hayakawa, S.; Oishi, Y.; Tanabe, H.; Isemura, M.; Suzuki, Y. Tea, Coffee and Health Benefits; Springer International Publishing: Cham, Switzerland, 2018. [Google Scholar]
- Fu, J.-D.; Yao, J.-J.; Wang, H.; Cui, W.-G.; Leng, J.; Ding, L.-Y.; Fan, K.-Y. Effects of EGCG on Proliferation and Apoptosis of Gastric Cancer SGC7901 Cells via Down-Regulation of HIF-1α and VEGF under a Hypoxic State. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 155–161. [Google Scholar]
- Zan, L.; Chen, Q.; Zhang, L.; Li, X. Epigallocatechin Gallate (EGCG) Suppresses Growth and Tumorigenicity in Breast Cancer Cells by Downregulation of miR-25. Bioengineered 2019, 10, 374–382. [Google Scholar] [CrossRef] [Green Version]
- Kwak, T.W.; Park, S.-B.; Kim, H.-J.; Jeong, Y.-I.; Kang, D.H. Anticancer Activities of Epigallocatechin-3-Gallate Against Cholangiocarcinoma Cells. Onco Targets Ther. 2016, 10, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Liu, Z.; Li, J.; Zhang, Q.; Zhong, P.; Teng, T.; Chen, M.; Xie, Z.; Ji, A.; Li, Y. Epigallocatechin-3-Gallate Inhibits the Growth and Increases the Apoptosis of Human Thyroid Carcinoma Cells Through Suppression of EGFR/RAS/RAF/MEK/ERK Signaling Pathway. Cancer Cell Int. 2019, 19, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.; Du, W.; Yang, D. Inhibition of Green Tea Polyphenol EGCG((−)-Epigallocatechin-3-Gallate) on the Proliferation of Gastric Cancer Cells by Suppressing Canonical Wnt/β-Catenin Signalling Pathway. Int. J. Food Sci. Nutr. 2016, 67, 818–827. [Google Scholar] [CrossRef]
- Pal, D.; Sur, S.; Roy, R.; Mandal, S.; Panda, C.K. Epigallocatechin Gallate in Combination with Eugenol or Amarogentin Shows Synergistic Chemotherapeutic Potential in Cervical Cancer Cell Line. J. Cell. Physiol. 2018, 234, 825–836. [Google Scholar] [CrossRef] [Green Version]
- Khan, M.A.; Hussain, A.; Sundaram, M.K.; Alalami, U.; Gunasekera, D.; Ramesh, L.; Hamza, A.; Quraishi, U. (−)-Epigallocatechin-3-Gallate Reverses the Expression of Various Tumor-Suppressor Genes by Inhibiting DNA Methyltransferases and Histone Deacetylases in Human Cervical Cancer Cells. Oncol. Rep. 2015, 33, 1976–1984. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Wang, Y.; Zhang, H.; Li, M.; Zhu, Z.; Xue, Y. An Investigation on the Cytotoxicity and Caspase-Mediated Apoptotic Effect of Biologically Synthesized Gold Nanoparticles Using Cardiospermum Halicacabum on AGS Gastric Carcinoma Cells. Int. J. Nanomed. 2019, 14, 951–962. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Li, J.; Cai, G.; Shen, L.; Lu, F. Proapoptotic and Growth-inhibitory Effects of Plumbagin on Human Gastric Cancer Cells Via Suppression of Signal Transducer and Activator of Transcription 3 and Protein Kinase B. Altern. Ther. Health Med. 2017, 23, 42–48. [Google Scholar]
- Li, J.; Shen, L.; Lu, F.-R.; Qin, Y.; Chen, R.; Li, J.; Li, Y.; Zhan, H.-Z.; He, Y.-Q. Plumbagin Inhibits Cell Growth and Potentiates Apoptosis in Human Gastric Cancer Cells In Vitro Through the NF-κB Signaling Pathway. Acta Pharmacol. Sin. 2012, 33, 242–249. [Google Scholar] [CrossRef]
- Li, X.; Lu, X.; Xu, H.; Zhu, Z.; Yin, H.; Qian, X.; Li, R.; Jiang, X.; Liu, B. Paclitaxel/Tetrandrine Coloaded Nanoparticles Effectively Promote the Apoptosis of Gastric Cancer Cells Based on “Oxidation Therapy”. Mol. Pharm. 2012, 9, 222–229. [Google Scholar] [CrossRef]
- Luo, Y.; Yu, H.; Yang, Y.; Tian, W.; Dong, K.; Shan, J.; Ma, X. A Flavonoid Compound from Chrysosplenium Nudicaule Inhibits Growth and Induces Apoptosis of the Human Stomach Cancer Cell Line SGC-7901. Pharm. Biol. 2015, 54, 1133–1139. [Google Scholar] [CrossRef] [Green Version]
- Ko, S.G.; Kim, H.-P.; Jin, N.-H.; Bae, H.; Kim, S.H.; Park, C.-H.; Lee, J.W. Saussurea Lappa Induces G2-Growth Arrest and Apoptosis in AGS Gastric Cancer Cells. Cancer Lett. 2005, 220, 11–19. [Google Scholar] [CrossRef] [Green Version]
- Ko, S.-G.; Koh, S.-H.; Jun, C.-Y.; Nam, C.-G.; Bae, H.; Shin, M.-K. Induction of Apoptosis by Saussurea Lappa and Pharbitis Nil on AGS Gastric Cancer Cells. Biol. Pharm. Bull. 2004, 27, 1604–1610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, Z.; Xu, T.; An, Z.; Hu, Y.; Chen, W.; Ma, J.; Shao, C.; Zhu, F. Costunolide Induces Mitochondria-Mediated Apoptosis in Human Gastric Adenocarcinoma BGC-823 Cells. BMC Complement. Altern. Med. 2019, 19, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, D.Y.; Choi, B.Y. Costunolide—A Bioactive Sesquiterpene Lactone with Diverse Therapeutic Potential. Int. J. Mol. Sci. 2019, 20, 2926. [Google Scholar] [CrossRef] [Green Version]
- Mahfouz, N.; Tahtouh, R.; Alaaeddine, N.; El Hajj, J.; Sarkis, R.; Hachem, R.; Raad, I.; Hilal, G. Gastrointestinal Cancer Cells Treatment With Bevacizumab Activates a VEGF Autoregulatory Mechanism Involving Telomerase Catalytic Subunit hTERT via PI3K-AKT, HIF-1α and VEGF Receptors. PLoS ONE 2017, 12, e0179202. [Google Scholar] [CrossRef]
- Lei, X.; Lv, X.; Liu, M.; Yang, Z.; Ji, M.; Guo, X.; Dong, W. Thymoquinone Inhibits Growth and Augments 5-Fluorouracil-Induced Apoptosis in Gastric Cancer Cells Both In Vitro and In Vivo. Biochem. Biophys. Res. Commun. 2012, 417, 864–868. [Google Scholar] [CrossRef]
- Ma, J.; Hu, X.; Li, J.; Wu, D.; Lan, Q.; Wang, Q.; Tian, S.; Dong, W. Enhancing Conventional Chemotherapy Drug Cisplatin-Induced Anti-Tumor Effects on Human Gastric Cancer Cells Both in Vitro and in Vivo by Thymoquinone Targeting PTEN Gene. Oncotarget 2017, 8, 85926–85939. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.-M.; Wang, X.-F.; Huang, Q.-X. Thymoquinone Induces Cytotoxicity and Reprogramming of EMT in Gastric Cancer Cells by Targeting PI3K/Akt/mTOR Pathway. J. Biosci. 2017, 42, 547–554. [Google Scholar] [CrossRef]
- El-Far, A.H. Thymoquinone Anticancer Discovery: Possible Mechanisms. Curr. Drug Discov. Technol. 2015, 12, 80–89. [Google Scholar] [CrossRef]
- Zhu, W.; Wang, J.; Guo, X.; Liu, Z.; Dong, W. Thymoquinone Inhibits Proliferation in Gastric Cancer via the stat3 Pathway In Vivo and In Vitro. World J. Gastroenterol. 2016, 22, 4149. [Google Scholar] [CrossRef]
- Sanjarin, F.; Sabouni, F.; Rashid, M. Thymoquinone Effects on Cell Viability, Apoptosis and VEGF-A Gene Expression Level in AGS(CRL-1739) Cell Line. Anti-Cancer Agents Med. Chem. 2019, 19, 820–826. [Google Scholar] [CrossRef]
- Fu, Z.; Han, X.; Du, J.; Han, X.; Liu, W.; Shao, S.; Liu, X. Euphorbia Lunulata Extract Acts on Multidrug Resistant Gastric Cancer Cells to Inhibit Cell Proliferation, Migration and Invasion, Arrest Cell Cycle Progression, and Induce Apoptosis. J. Ethnopharmacol. 2018, 212, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Han, X.; Wang, A.; Liu, X. Apoptosis of Human Gastric Carcinoma Cells Induced by Euphorbia Esula Latex. World J. Gastroenterol. 2016, 22, 3564. [Google Scholar] [CrossRef]
- Chen, X.; Wu, S.; Zeng, X.; Jiang, X.; Yuan, J.; Lu, B.; Li, J. Antioxidant and SGC-7901 Cell Inhibition Activities of Rhizoma Dioscoreae Bulbiferae. Ethanol Extracts. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 261–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Wang, X.; Cao, S.; Sun, Y.; He, X.; Jiang, B.; Yu, Y.; Duan, J.; Qiu, F.; Kang, N. Berberine Represses Human Gastric Cancer Cell Growth in Vitro and in Vivo by Inducing Cytostatic Autophagy via Inhibition of MAPK/mTOR/p70s6k and Akt Signaling Pathways. Biomed. Pharmacother. 2020, 128, 110245. [Google Scholar] [CrossRef]
- Yi, T.; Zhuang, L.; Song, G.; Zhang, B.; Li, G.; Hu, T. Akt Signaling Is Associated with T He Berberine-Induced Apoptosis of Human Gastric Cancer Cells. Nutr. Cancer 2015, 67, 523–531. [Google Scholar] [CrossRef]
- Hu, Q.; Li, L.; Zou, X.; Xu, L.; Yi, P. Berberine Attenuated Proliferation, Invasion and Migration by Targeting the AMPK/HNF4α/WNT5A Pathway in Gastric Carcinoma. Front. Pharmacol. 2018, 9, 9. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, M.; Shang, D. Berberine Inhibits Human Gastric Cancer Cell Growth via Deactivation of p38/Jnk Pathway, Induction of Mitochondrial-Mediated Apoptosis, Caspase Activation and NF-κB Inhibition. J. BU ON Off. J. Balk. Union Oncol. 2020, 25, 314–318. [Google Scholar]
- Liu, D.; Meng, X.; Wu, D.; Qiu, Z.; Luo, H. A Natural Isoquinoline Alkaloid with Antitumor Activity: Studies of the Biological Activities of Berberine. Front. Pharmacol. 2019, 10, 9. [Google Scholar] [CrossRef] [Green Version]
- Pandey, A.; Vishnoi, K.; Mahata, S.; Tripathi, S.C.; Misra, S.P.; Misra, V.; Mehrotra, R.; Dwivedi, M.; Bharti, A.C. Berberine and Curcumin Target Survivin and STAT3 in Gastric Cancer Cells and Synergize Actions of Standard Chemotherapeutic 5-Fluorouracil. Nutr. Cancer 2015, 67, 1295–1306. [Google Scholar] [CrossRef]
- Wei, J.; Liu, B.-R.; Wang, L.; Qian, X.; Ding, Y.; Yu, L. Synergistic Interaction Between Tetrandrine and Chemotherapeutic Agents and Influence of Tetrandrine on Chemotherapeutic Agent-Associated Genes in Human Gastric Cancer Cell Lines. Cancer Chemother. Pharmacol. 2007, 60, 703–711. [Google Scholar] [CrossRef]
- Bai, X.; Liu, Y.-G.; Song, W.; Li, Y.; Hou, D.-S.; Luo, H.-M.; Liu, P. Anticancer Activity of Tetrandrine by Inducing Pro-Death Apoptosis and Autophagy in Human Gastric Cancer Cells. J. Pharm. Pharmacol. 2018, 70, 1048–1058. [Google Scholar] [CrossRef] [PubMed]
- Qin, R.; Shen, H.; Cao, Y.; Fang, Y.; Li, H.; Chen, Q.; Xu, W. Tetrandrine Induces Mitochondria-Mediated Apoptosis in Human Gastric Cancer BGC-823 Cells. PLoS ONE 2013, 8, e76486. [Google Scholar] [CrossRef] [Green Version]
- Zhang, P.; Shi, L.; Zhang, T.; Hong, L.; He, W.; Cao, P.; Shen, X.; Zheng, P.; Xia, Y.; Zou, P. Piperlongumine Potentiates the Antitumor Efficacy of Oxaliplatin through Ros Induction in Gastric Cancer Cells. Cell. Oncol. 2019, 42, 847–860. [Google Scholar] [CrossRef]
- Zou, P.; Xia, Y.; Ji, J.; Chen, W.; Zhang, J.; Chen, X.; Rajamanickam, V.; Chen, G.; Wang, Z.; Chen, L.; et al. Piperlongumine as a Direct TrxR1 Inhibitor with Suppressive Activity against Gastric Cancer. Cancer Lett. 2016, 375, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Duan, C.; Zhang, B.; Deng, C.; Cao, Y.; Zhou, F.; Wu, L.; Chen, M.; Shen, S.; Xu, G.; Zhang, S.; et al. Piperlongumine Induces Gastric Cancer Cell Apoptosis and g2/M Cell Cycle Arrest Both In Vitro and In Vivo. Tumor Biol. 2016, 37, 10793–10804. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Sramkoski, R.M.; Jacobberger, J.W. The Kinetics of G2 and M Transitions Regulated by B Cyclins. PLoS ONE 2013, 8, e80861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, B.; Zhan, H.; Bian, Q.; Gu, J. Piperlongumine Inhibits Gastric Cancer Cells via Suppression of the JAK1, 2/STAT3 Signaling Pathway. Mol. Med. Rep. 2016, 13, 4475–4480. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Shi, Z.; Lin, J.; Zhao, S.; Hao, M.; Xu, J.; Li, Y.; Zhao, Q.; Tao, L.; Diao, A. Piperlongumine-Induced Nuclear Translocation of the FOXO3A Transcription Factor Triggers BIM-Mediated Apoptosis in Cancer Cells. Biochem. Pharmacol. 2019, 163, 101–110. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Chen, X.; Liu, T.; Chen, Y.; He, W.; Zhang, Q.; Liu, S. Autophagy Is Involved in Anticancer Effects of Matrine on SGC-7901 Human Gastric Cancer Cells. Oncol. Rep. 2011, 26, 115–124. [Google Scholar] [CrossRef] [Green Version]
- Song, S.; Zhu, S.; Zhang, Z.; Mo, Z.; Ke, Q.; Luo, Z. A Study on the Inhibitory Effect of Matrine on Gastric Cancer SGC-7901 Cells. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 435–438. [Google Scholar] [CrossRef]
- Luo, C.; Zhu, Y.; Jiang, T.; Lu, X.; Zhang, W.; Jing, Q.; Li, J.; Pang, L.; Chen, K.; Qiu, F.; et al. Matrine Induced Gastric Cancer MKN45 Cells Apoptosis via Increasing Pro-Apoptotic Molecules of BCL-2 Family. Toxicology 2007, 229, 245–252. [Google Scholar] [CrossRef]
- Zhang, J.-W.; Su, K.; Shi, W.-T.; Wang, Y.; Hu, P.-C.; Wang, Y.; Wei, L.; Xiang, J.; Yang, F. Matrine Inhibits the Adhesion and Migration of BCG823 Gastric Cancer Cells by Affecting the Structure and Function of the Vasodilator-Stimulated Phosphoprotein (VASP). Acta Pharmacol. Sin. 2013, 34, 1084–1092. [Google Scholar] [CrossRef] [Green Version]
- Hsu, J.-H.; Jhou, B.-Y.; Yeh, S.-H.; Chen, Y.-L. Healthcare Functions of Cordyceps cicadae. J. Nutr. Food Sci. 2015, 5, 432. [Google Scholar] [CrossRef]
- Hur, H. Chemical Ingredients of Cordyceps militaris. Mycobiology 2008, 36, 233–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, J.S.L.; Barseghyan, G.S.; Asatiani, M.D.; Wasser, S.P. Chemical Composition and Medicinal Value of Fruiting Bodies and Submerged Cultured Mycelia of Caterpillar Medicinal Fungus Cordyceps militaris CBS-132098 (Ascomycetes). Int. J. Med. Mushrooms 2015, 17, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Li, X.; Chen, Y.; Lang, M.; Shen, Z.; Shi, L.-G. Ethanolic Extract of Cordyceps cicadae Exerts Antitumor Effect on Human Gastric Cancer SGC-7901 Cells by Inducing Apoptosis, Cell Cycle Arrest and Endoplasmic Reticulum Stress. J. Ethnopharmacol. 2018, 231, 230–240. [Google Scholar] [CrossRef]
- Dillekås, H.; Rogers, M.S.; Straume, O. Are 90% of Deaths from Cancer Caused by Metastases? Cancer Med. 2019, 8, 5574–5576. [Google Scholar] [CrossRef] [Green Version]
- Ho, C.-C.; Lai, K.; Hsu, S.-C.; Kuo, C.-L.; Ma, C.-Y.; Lin, M.-L.; Yang, J.-S.; Chung, J.-G. Benzyl Isothiocyanate (BITC) Inhibits Migration and Invasion of Human Gastric Cancer AGS Cells via Suppressing ERK Signal Pathways. Hum. Exp. Toxicol. 2010, 30, 296–306. [Google Scholar] [CrossRef]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular Principles of Metastasis: A Hallmark of Cancer Revisited. Signal Transduct. Target. Ther. 2020, 5, 1–17. [Google Scholar] [CrossRef]
- Chen, T.; You, Y.; Jiang, H.; Wang, Z.Z. Epithelial-Mesenchymal Transition (EMT): A Biological Process in the Development, Stem Cell Differentiation, and Tumorigenesis. J. Cell. Physiol. 2017, 232, 3261–3272. [Google Scholar] [CrossRef]
- Powolny, A.A.; Singh, S.V. Multitargeted Prevention and Therapy of Cancer by Diallyl Trisulfide and Related Allium Vegetable-Derived Organosulfur Compounds. Cancer Lett. 2008, 269, 305–314. [Google Scholar] [CrossRef] [Green Version]
- Charron, C.; Milner, J.; Novotny, J. Garlic. In Encyclopedia of Food and Health; Academic Press: Oxford, UK, 2016. [Google Scholar]
- Rahman, M.S. Allicin and Other Functional Active Components in Garlic: Health Benefits and Bioavailability. Int. J. Food Prop. 2007, 10, 245–268. [Google Scholar] [CrossRef]
- Belman, S. Onion and Garlic Oils Inhibit Tumor Promotion. Carcinogenesis 1983, 4, 1063–1065. [Google Scholar] [CrossRef]
- Setiawan, V.W.; Yu, G.-P.; Lu, Q.-Y.; Lu, M.-L.; Yu, S.-Z.; Mu, L.; Zhang, J.-G.; Kurtz, R.C.; Cai, L.; Hsieh, C.-C.; et al. Allium Vegetables and Stomach Cancer Risk in China. Asian Pac. J. Cancer Prev. 2005, 6, 387–395. [Google Scholar]
- Alizadeh-Navaei, R.; Shamshirian, A.; Hedayatizadeh-Omran, A.; Ghadimi, R.; Janbabai, G. Effect of Garlic in Gastric Cancer Prognosis: A Systematic Review and Meta-Analysis. World Cancer Res. J. 2018, 5, e1184. [Google Scholar]
- Hekmatdoost, A.; Ghobeh, M.; Shaker-Hosseini, R.; MirSattari, D.; Rastmanesh, R.; Rashidkhani, B.; Navai, L. The Effect of Garlic Consumption on Helicobacter Pylori Treatment Using Urea Breath Test: A Randomized Clinical Trial. J. Nutr. Sci. Diet. 2015, 1, 21–27. [Google Scholar]
- Zardast, M.; Namakin, K.; Kaho, J.E.; Hashemi, S.S. Assessment of Antibacterial Effect of Garlic in Patients Infected with Helicobacter Pylori Using Urease Breath Test. Avicenna J. Phytomedicine 2016, 6, 495–501. [Google Scholar]
- Wang, Y.; Wu, K.; Yang, Z.; Zhao, Q.; Fan, N.; Xu, P.; Nie, Y.; Fan, D. Multidrug-Resistance Related Long Non-Coding RNA Expression Profile Analysis of Gastric Cancer. PLoS ONE 2015, 10, e0135461. [Google Scholar] [CrossRef]
- Hayakawa, S.; Ohishi, T.; Miyoshi, N.; Oishi, Y.; Nakamura, Y.; Isemura, M. Anti-Cancer Effects of Green Tea Epigallocatchin-3-Gallate and Coffee Chlorogenic Acid. Molecules 2020, 25, 4553. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Q.; Lu, J.-L.; Liang, Y.-R.; Li, Q.-S. Suppressive Effects of EGCG on Cervical Cancer. Molecules 2018, 23, 2334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molaei, F.; Forghanifard, M.M.; Fahim, Y.; Abbaszadegan, M.R. Molecular Signaling in Tumorigenesis of Gastric Cancer. Iran. Biomed. J. 2018, 22, 217–230. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; Chen, H.; Zhou, L.; Li, G.; Yi, D.; Zhang, Y.; Wu, Y.; Liu, X.; Wu, X.; Song, Q.; et al. Association Between Green Tea Intake and Risk of Gastric Cancer: A Systematic Review and Dose–Response Meta-Analysis of Observational Studies. Public Health Nutr. 2017, 20, 3183–3192. [Google Scholar] [CrossRef] [Green Version]
- Zalke, A.S.; Duraiswamy, B.; Gandagule, U.B.; Singh, N. Pharmacognostical Evaluation of Cardiospermum Halicacabum Linn. Leaf and Stem. Anc. Sci. Life 2013, 33, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cai, Y.; He, C.; Chen, M.; Li, H. Anticancer Properties and Pharmaceutical Applications of Plumbagin: A Review. Am. J. Chin. Med. 2017, 45, 423–441. [Google Scholar] [CrossRef]
- Xia, L.; Tan, S.; Zhou, Y.; Lin, J.; Wang, H.; Oyang, L.; Tian, Y.; Liu, L.; Su, M.; Wang, H.; et al. Role of the NFκB-Signaling Pathway in Cancer. Onco Targets Ther. 2018, 11, 2063–2073. [Google Scholar] [CrossRef] [Green Version]
- Ma, X.; Yin, X.; Liu, H.; Chen, Q.; Feng, Y.; Ma, X.; Liu, W. Antiproliferative Activity of Plumbagin (5-Hydroxy-2-Methyl-1,4-Naphthoquinone) in Human Gastric Carcinoma Cells Is Facilitated via Activation of Autophagic Pathway, Mitochondrial-Mediated Programmed Cell Death and Inhibition of Cell Migration and Invasion. J. BU ON Off. J. Balk. Union Oncol. 2019, 24, 2000–2005. [Google Scholar]
- Zong, Y.; Dang, H.; Luo, G.; Zhang, X.; Che, Z. Antitumor Screening Research from 110 Tibetan Medicines. J. Pharm. Pract. 2000, 18, 290–291. [Google Scholar]
- Wang, Y.; Yang, Y.; Yang, L. K562 Apoptosis Induced by Flavone from Tibetan Medicine Chrysosplenium Nudicaule Bunge and Its Molecular Mechanism. Pract. J. Cancer 2005, 20, 374–376. [Google Scholar]
- Lee, M.-G.; Lee, K.; Chi, S.-G.; Park, J.-H. Costunolide Induces Apoptosis by ROS-mediated Mitochondrial Permeability Transition and Cytochrome C Release. Biol. Pharm. Bull. 2001, 24, 303–306. [Google Scholar] [CrossRef] [Green Version]
- Venkatachallam, S.K.T.; Pattekhan, H.; Divakar, S.; Kadimi, U.S. Chemical Composition of Nigella Sativa L. Seed Extracts Obtained by Supercritical Carbon Dioxide. J. Food Sci. Technol. 2010, 47, 598–605. [Google Scholar] [CrossRef] [Green Version]
- Mansi, K.M. Effects of Oral Administration of Water Extract of Nigella sativa on Serum Concentrations of Insulin and Testosterone in Alloxan—Induced Diabetic Rats. Pak. J. Biol. Sci. 2005, 8, 1152–1156. [Google Scholar]
- Majidinia, M.; Mirza-Aghazadeh-Attari, M.; Rahimi, M.; Mihanfar, A.; Karimian, A.; Safa, A.; Yousefi, B. Overcoming Multidrug Resistance in Cancer: Recent Progress in Nanotechnology and New Horizons. IUBMB Life 2020, 72, 855–871. [Google Scholar] [CrossRef]
- Yang, X.; Liu, K. P-gp Inhibition-Based Strategies for Modulating Pharmacokinetics of Anticancer Drugs: An Update. Curr. Drug Metab. 2016, 17, 806–826. [Google Scholar] [CrossRef]
- Shi, X.; Wang, J.; Lei, Y.; Cong, C.; Tan, D.; Zhou, X. Research Progress on the PI3K/AKT Signaling Pathway in Gynecological Cancer. Mol. Med. Rep. 2019, 19, 4529–4535. [Google Scholar] [CrossRef] [Green Version]
- Arshad, S.; Naveed, M.; Ullia, M.; Javed, K.; Butt, A.; Khawar, M.; Amjad, F. Targeting STAT-3 Signaling Pathway in Cancer for Development of Novel Drugs: Advancements and Challenges. Genet. Mol. Biol. 2020, 43, e20180160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Groner, B.; Lucks, P.; Borghouts, C. The Function of Stat3 in Tumor Cells and Their Microenvironment. Semin. Cell Dev. Biol. 2008, 19, 341–350. [Google Scholar] [CrossRef]
- Wang, Y.-B.; Ji, P.; Wang, H.-B.; Qin, G.-W. Diterpenoids from Euphorbia esula. Chin. J. Nat. Med. 2010, 8, 94–96. [Google Scholar] [CrossRef]
- Zhou, B.; Wu, Y.; Dalal, S.; Cassera, M.B.; Yue, J.-M. Euphorbesulins A–P, Structurally Diverse Diterpenoids from Euphorbia esula. J. Nat. Prod. 2016, 79, 1952–1961. [Google Scholar] [CrossRef]
- Akaberi, M.; Mehri, S.; Iranshahi, M. Multiple Pro-Apoptotic Targets of Abietane Diterpenoids from Salvia Species. Fitoterapia 2015, 100, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Liao, Z.; Liu, S.-J.; Qu, Y.-B.; Wang, H.-S. Two New Diterpene Derivatives from Euphorbia Lunulata Bge and Their Anti-Proliferative Activities. Fitoterapia 2014, 96, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yu, X.; Wang, L.; Zhang, F.; Zhang, Y.-Q. Research Progress on Chemical Constituents and Anticancer Pharmacological Activities of Euphorbia lunulata Bunge. BioMed Res. Int. 2020, 2020, 1–11. [Google Scholar] [CrossRef]
- Wang, J.-M.; Ji, L.; Branford-White, C.J.; Wang, Z.-Y.; Shen, K.-K.; Liu, H.; Hu, F. Antitumor Activity of Dioscorea bulbifera L. Rhizome In Vivo. Fitoterapia 2012, 83, 388–394. [Google Scholar] [CrossRef]
- Kukula-Koch, W.; Widelski, J. Pharmacognosy. Fundamentals, Applications and Strategies; Academic Press: Cambridge, MA, USA, 2017; pp. 163–198. [Google Scholar]
- Sun, C.; Li, J.; Wang, X.; Duan, W.; Zhang, T.; Ito, Y. Preparative Separation of Quaternary Ammonium Alkaloids from Coptis Chinensis Franch by pH-Zone-Refining Counter-Current Chromatography. J. Chromatogr. 2014, 1370, 156–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Wang, L.; Lou, G.-H.; Zeng, H.-R.; Hu, J.; Huang, Q.-W.; Peng, W.; Yang, X.-B. Coptidis Rhizoma: A Comprehensive Review of Its Traditional Uses, Botany, Phytochemistry, Pharmacology and Toxicology. Pharm. Biol. 2019, 57, 193–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, A.; Chopra, K.; Mukherjee, M.; Pottabathini, R.; Dhull, D.K. Current Knowledge and Pharmacological Profile of Berberine: An Update. Eur. J. Pharmacol. 2015, 761, 288–297. [Google Scholar] [CrossRef]
- Huang, J.-F.; Du, W.-X.; Chen, J.-J. Elevated Expression of Matrix Metalloproteinase-3 in Human Osteosarcoma and Its Association with Tumor Metastasis. J. BUON 2016, 21, 1279–1286. [Google Scholar]
- Chu, C.; Liu, X.; Bai, X.; Zhao, T.; Wang, M.; Xu, R.; Li, M.; Hu, Y.; Li, W.; Yang, L.; et al. MiR-519d Suppresses Breast Cancer Tumorigenesis and Metastasis via Targeting MMP3. Int. J. Biol. Sci. 2018, 14, 228–236. [Google Scholar] [CrossRef] [Green Version]
- Simi, A.K.; Anlaş, A.A.; Stallings-Mann, M.; Zhang, S.; Hsia, T.; Cichon, M.A.; Radisky, D.C.; Nelson, C.M. A Soft Microenvironment Protects from Failure of Midbody Abscission and Multinucleation Downstream of the EMT-Promoting Transcription Factor Snail. Cancer Res. 2018, 78, 2277–2289. [Google Scholar] [CrossRef] [Green Version]
- Van Der Post, R.S.; Bult, P.; Vogelaar, I.P.; Ligtenberg, M.J.L.; Hoogerbrugge, N.; Van Krieken, J.H. HNF4A Immunohistochemistry Facilitates Distinction Between Primary and Metastatic Breast and Gastric Carcinoma. Virchows Arch. 2014, 464, 673–679. [Google Scholar] [CrossRef]
- Nam, S.; Chang, H.R.; Kim, K.-T.; Kook, M.-C.; Hong, D.; Kwon, C.H.; Jung, H.R.; Park, H.S.; Powis, G.; Liang, H.; et al. PATHOME: An Algorithm for Accurately Detecting Differentially Expressed Subpathways. Oncogene 2014, 33, 4941–4951. [Google Scholar] [CrossRef] [Green Version]
- You, H.-Y.; Xie, X.-M.; Zhang, W.-J.; Zhu, H.-L.; Jiang, F.-Z. Berberine Modulates Cisplatin Sensitivity of Human Gastric Cancer Cells by Upregulation of miR-203. Vitr. Cell. Dev. Biol. Anim. 2016, 52, 857–863. [Google Scholar] [CrossRef]
- Fan, R.; Xiao, C.; Wan, X.; Cha, W.; Miao, Y.; Zhou, Y.; Qin, C.; Cui, T.; Su, F.; Shan, X. Small Molecules with Big Roles in microRNA Chemical Biology and microRNA-Targeted Therapeutics. RNA Biol. 2019, 16, 707–718. [Google Scholar] [CrossRef]
- Yu, M.; Ren, L.; Liang, F.; Zhang, Y.; Jiang, L.; Ma, W.; Li, C.; Li, X.; Ye, X. Effect of Epiberberine from Coptis Chinensis Franch on Inhibition of Tumor Growth in MKN-45 Xenograft Mice. Phytomedicine 2020, 76, 153216. [Google Scholar] [CrossRef]
- Chen, J. The Cell-Cycle Arrest and Apoptotic Functions of p53 in Tumor Initiation and Progression. Cold Spring Harb. Perspect. Med. 2016, 6, a026104. [Google Scholar] [CrossRef] [PubMed]
- Bhagya, N.; Chandrashekar, K. Tetrandrine–A Molecule of Wide Bioactivity. Phytochemistry 2016, 125, 5–13. [Google Scholar] [CrossRef]
- Luan, F.; He, X.; Zeng, N. Tetrandrine: A Review of Its Anticancer Potentials, Clinical Settings, Pharmacokinetics and Drug Delivery Systems. J. Pharm. Pharmacol. 2020. [Google Scholar] [CrossRef]
- Han, D.; Wan, C.; Liu, F.; Xu, X.; Jiang, L.; Xu, J. Jujuboside A Protects H9C2 Cells from Isoproterenol-Induced Injury via Activating PI3K/Akt/mTOR Signaling Pathway. Evid. Based Complement. Altern. Med. 2016, 2016, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, S. Historical Spice as a Future Drug: Therapeutic Potential of Piperlongumine. Curr. Pharm. Des. 2016, 22, 4151–4159. [Google Scholar] [CrossRef]
- Valette, O.; Tran, T.T.T.; Cavazza, C.; Caudeville, E.; Brasseur, G.; Dolla, A.; Talla, E.; Pieulle, L. Biochemical Function, Molecular Structure and Evolution of an Atypical Thioredoxin Reductase from Desulfovibrio vulgaris. Front. Microbiol. 2017, 8, 1855. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ao, X.; Ding, W.; Ponnusamy, M.; Wu, W.; Hao, X.; Yu, W.; Wang, Y.; Li, P.; Wang, J. Critical Role of FOXO3a in Carcinogenesis. Mol. Cancer 2018, 17, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Eijkelenboom, A.; Burgering, B.M.T. FOXOs: Signalling Integrators for Homeostasis Maintenance. Nat. Rev. Mol. Cell Biol. 2013, 14, 83–97. [Google Scholar] [CrossRef]
- Cardoso, D.; Pennington, R.; De Queiroz, L.; Boatwright, J.S.; Van Wyk, B.E.; Wojciechowski, M.; Lavin, M. Reconstructing the Deep-Branching Relationships of the Papilionoid Legumes. S. Afr. J. Bot. 2013, 89, 58–75. [Google Scholar] [CrossRef] [Green Version]
- Tan, C.-J.; Zhao, Y.; Goto, M.; Hsieh, K.-Y.; Yang, X.-M.; Morris-Natschke, S.L.; Liu, L.-N.; Zhao, B.-Y.; Lee, K.-H.; And, B.-Y.Z. ChemInform Abstract: Alkaloids from Oxytropis ochrocephala and Antiproliferative Activity of Sophoridine Derivatives Against Cancer Cell Lines. Bioorganic Med. Chem. Lett. 2016, 47. [Google Scholar] [CrossRef]
- Li, J.-J.; Zhang, X.; Shen, X.-C.; Long, Q.-D.; Xu, C.-Y.; Tan, C.-J.; Lin, Y. Phytochemistry and Biological Properties of Isoprenoid Flavonoids from Sophora Flavescens Ait. Fitoterapia 2020, 143, 104556. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, X.; Fan, H.; Zhu, D.; Wu, X.; Huang, X.; Tang, J. Separation and Purification of Alkaloids from Sophora Flavescens Ait. by Focused Microwave-Assisted Aqueous Two-Phase Extraction Coupled with Reversed Micellar Extraction. Ind. Crop. Prod. 2016, 86, 231–238. [Google Scholar] [CrossRef]
- Dai, Z.-J.; Gao, J.; Ji, Z.-Z.; Wang, X.-J.; Ren, H.-T.; Liu, X.-X.; Wu, W.-Y.; Kang, H.; Guan, H.-T. Matrine Induces Apoptosis in Gastric Carcinoma Cells via Alteration of FAS/FASL and Activation of Caspase-3. J. Ethnopharmacol. 2009, 123, 91–96. [Google Scholar] [CrossRef]
- Peng, X.; Zhou, D.; Wang, X.; Hu, Z.; Yan, Y.; Huang, J.R. Matrine Suppresses Proliferation and Invasion of SGC7901 Cells through Inactivation of PI3K/Akt/uPA Pathway. Ann. Clin. Lab. Sci. 2016, 46, 457–462. [Google Scholar]
- Mao, Q.-Q.; Xu, X.-Y.; Shang, A.; Gan, R.-Y.; Wu, D.; Atanasov, A.G.; Li, H.-B. Phytochemicals for the Prevention and Treatment of Gastric Cancer: Effects and Mechanisms. Int. J. Mol. Sci. 2020, 21, 570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mu, J.; Liu, T.; Jiang, L.; Wu, X.; Cao, Y.; Li, M.; Dong, Q.; Liu, Y.; Xu, H. The Traditional Chinese Medicine Baicalein Potently Inhibits Gastric Cancer Cells. J. Cancer 2016, 7, 453–461. [Google Scholar] [CrossRef] [Green Version]
- Wen, C.; Wang, H.; Wu, X.; He, L.; Zhou, Q.; Wang, F.; Chen, S.; Huang, L.; Chen, J.; Wang, H.; et al. ROS-Mediated Inactivation of the PI3K/AKT Pathway Is Involved in the Antigastric Cancer Effects of Thioredoxin Reductase-1 Inhibitor Chaetocin. Cell Death Dis. 2019, 10, 809–816. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Chen, Y.; Yang, R.; Zhou, T.; Ke, W.; Si, Y.; Yang, S.; Zhang, T.; Liu, X.; Zhang, L.; et al. Cucurbitacin B Inhibits Gastric Cancer Progression by Suppressing STAT3 Activity. Arch. Biochem. Biophys. 2020, 684, 108314. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Fang, Y.; Xu, W.; Wang, Q.; Zhou, J.; Lu, R. 3,3′-Diindolylmethane Induces Anti-Human Gastric Cancer Cells by the miR-30e-ATG5 Modulating Autophagy. Biochem. Pharmacol. 2016, 115, 77–84. [Google Scholar] [CrossRef]
- Zhao, X.; Xu, L.; Zheng, L.; Yin, L.; Qi, Y.; Han, X.; Xu, Y.; Peng, J. Potent Effects of Dioscin Against Gastric Cancer In Vitro and In Vivo. Phytomedicine 2016, 23, 274–282. [Google Scholar] [CrossRef]
- Yao, J.-N.; Zhang, X.-X.; Zhang, Y.-Z.; Li, J.-H.; Zhao, D.-Y.; Gao, B.; Zhou, H.-N.; Gao, S.-L.; Zhang, L.-F. Discovery and Anticancer Evaluation of a Formononetin Derivative Against Gastric Cancer SGC7901 Cells. Investig. New Drugs 2019, 37, 1300–1308. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, J.; Lin, L.; He, L.; Wu, Y.; Zhang, L.; Yi, Z.; Chen, Y.; Pang, X.; Liu, M. Inhibition of STAT3 Signaling Pathway by Nitidine Chloride Suppressed the Angiogenesis and Growth of Human Gastric Cancer. Mol. Cancer Ther. 2011, 11, 277–287. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Liu, S.; Feng, Q.; Huang, X.; Wang, X.; Peng, Y.; Zhao, Z.; Liu, Z. Perilaldehyde Activates AMP-Activated Protein Kinase to Suppress the Growth of Gastric Cancer via Induction of Autophagy. J. Cell. Biochem. 2018, 120, 1716–1725. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Jiang, X.; Li, A.; Sun, Y.; Liu, Y.; Sun, X.; Feng, X.; Li, S.; Zhao, Z. S-a Llylmercaptocysteine Suppresses the Growth of Human Gastric Cancer Xenografts Through Induction of Apoptosis and Regulation of MAPK and PI3K/Akt Signaling Pathways. Biochem. Biophys. Res. Commun. 2017, 491, 821–826. [Google Scholar] [CrossRef]
- Ishiguro, K.; Ando, T.; Maeda, O.; Ohmiya, N.; Niwa, Y.; Kadomatsu, K.; Goto, H. Ginger Ingredients Reduce Viability of Gastric Cancer Cells via Distinct Mechanisms. Biochem. Biophys. Res. Commun. 2007, 362, 218–223. [Google Scholar] [CrossRef]
- Li, W.-Q.; Zhang, J.-Y.; Ma, J.-L.; Li, Z.-X.; Zhang, L.; Zhang, Y.; Guo, Y.; Zhou, T.; Li, J.-Y.; Shen, L.; et al. Effects of Helicobacter Pylori Treatment and Vitamin and Garlic Supplementation on Gastric Cancer Incidence and Mortality: Follow-up of a Randomized Intervention Trial. BMJ 2019, 366, l5016. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.-H.; Choi, W.-C.; Kim, K.-S.; Park, J.-W.; Yoon, S.-W. Shrinkage of Gastric Cancer in an Elderly Patient Who Received Rhus Verniciflua Stokes Extract. J. Altern. Complement. Med. 2010, 16, 497–500. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, M.; Ren, Q.; Zhu, W.-F.; Wang, Y.; Chen, H.; Chen, J. Oral and Injectable Marsdenia Tenacissima Extract (MTE) as Adjuvant Therapy to Chemotherapy for Gastric Cancer: A Systematic Review. BMC Complement. Altern. Med. 2019, 19, 366. [Google Scholar] [CrossRef] [Green Version]
- Cheng, A.L.; Hsu, C.-H.; Lin, J.K.; Hsu, M.; Ho, Y.F.; Shen, T.S.; Ko, J.Y.; Lin, J.T.; Lin, B.R.; Ming-Shiang, W.; et al. Phase I Clinical Trial of Curcumin, a Chemopreventive Agent, in Patients with High-Risk or Pre-Malignant Lesions. Anticancer Res. 2001, 21, 2895–2900. [Google Scholar]
- Lissoni, P.; Rovelli, F.; Brivio, F.; Zago, R.; Colciago, M.; Messina, G.; Mora, A.; Porro, G. A Randomized Study of Chemotherapy Versus Biochemotherapy With Chemotherapy Plus Aloe Arborescens in Patients with Metastatic Cancer. In Vivo 2009, 23, 171–175. [Google Scholar]
| Plant Species | Active Compound | Cell Line | Bioactive Effect | Ref. |
|---|---|---|---|---|
| Cordyceps cicadae | Cordicepine | SGC-7901 MGC-803 HGC-27 | 1. Antiproliferative effects
| [18] |
| Allium sativum | Allicin | SGC-7901 MGC-803 BGC-823 | 1. Antiproliferative effects
| [19,20,21] |
| Camellia sinensis | Epigallocatechin gallate | SGC-7901 | 1. Antiproliferative effects
| [22,23,24,25,26,27,28,29] |
| Cardiospermum halicacabum (water extract) | Synthesized gold nanoparticles (CH-AuNP) | AGS SNU-5 SNU-16 | 1. Proapoptotic effects
| [30] |
| Plumbago zeylanica | Plumbagin | AGS SGC-7901 MKN-28 | 1. Proapoptotic effects
3. Antimetastatic effects
| [31,32,33] |
| Chrysosplenium nudicaule (ethanol extract) | TTF and DTFG | SGC-7901 |
| [34] |
| Saussurea lappa | Costunolide | SGC-7901 MGC-803 BGC-823 | 1. Antiproliferative effects
| [35,36,37,38,39] |
| Nigella sativa | Thymoquinone | BGC-823 SGC-7901 MGC-803 HGC-27 | 1. Antimetastatic effects
| [40,41,42,43,44,45] |
| Euphorbia lunulata | Diterpenoids | SGC-7901/ADR | 1. Antiproliferative effects
| [46] |
| Euphorbia esula (water extract) | Total extract | SGC-7901 | 1. Proapoptotic effects
| [47] |
| Dioscorea bulbifera(ethanol extract) | Diosbulbine B | SGC-7901 |
| [48] |
| Coptis chinesis | Berberine | BGC-823 AGS SGC-7901 SNU-1 | 1. Antiproliferative effects
| [49,50,51,52,53,54] |
| Stephania tetrandra | Tetrandrine | HGC-27 BGC-823 | 1. Proapoptotic effects
| [33,55,56,57] |
| Piper longum | Piperlongumine | SGC-7901 BGC-823 AGS MKN-45 MGC-803 | 1. Antiproliferative effects
| [58,59,60,61,62,63] |
| Sophora spp. | Matrine | BGC-823 SGC-7901 MKN-28 | 1. Antiproliferative effects
| [64,65,66,67] |
| Plant | Bioactive Compound | Molecular Formula | Structure |
|---|---|---|---|
| Allium sativum | Allicin | C6H10OS2 |  |
| Coptis chinesis | Berberine | C20H18NO4+ |  |
| Epiberberine | C20H18NO4 |  | |
| Cordyceps cicadae | Cordycepin | C10H13N5O3 |  |
| Dioscorea bulbifera | Diosbulbin B | C19H20O6 |  |
| Nigella sativa | Thymoquinone | C10H12O2 |  |
| Piper longum | Piperlongumine | C17H19NO5 |  |
| Plumbago zeylanica | Plumbagin | C11H8O3 |  |
| Saussurea lappa | Costunolide | C15H20O2 |  |
| Sophora spp. | Matrine | C15H24N2O |  |
| Oxymatrine | C15H24N2O2 |  | |
| Stephania tetrandra | Tetrandrine | C38H42N2O6 |  |
| Natural Product | Plant of Origin | Model | Mechanism of Action | Molecular Mechanism | References |
|---|---|---|---|---|---|
| Baicalein | Scutellaria spp. | Nude mice | Reduced tumor volume | Induced apoptosis, S phase arrest | [136] |
| Chaetocin | Chaetomium spp. fungi | Nude mice | Inhibited proliferation, induced cell cycle arrest (G2/M) and apoptosis | Inactivation of PI3K/AKT pathway | [137] |
| Cucurbitacin C | Cucurbita spp. | Female and male nude mice | Reduced tumor volume | Inhibition of STAT3 | [138] |
| 3,3′-diindolylmethane | Cruciferous plants | female nude mice | Inhibited tumor growth | Increase of LC3 levels (microtubule-associated protein light chain) | [139] |
| Dioscin | Dioscorea spp. | Nude mice | Induced apoptosis, cell arrest (S phase), inhibited tumor growth | Induced ROS generation and Ca2+ release | [140] |
| Formononetin-coumarin hybrid | Nude mice | Inhibited tumor growth | Inhibition of SIRT-1, Wnt/β-Catenin and AKT/mTOR pathways | [141] | |
| Nitidine chloride | Zanthoxylum nitidum | Male BALB/cA nude mice | Reduced tumor volume | Decreased STAT3 and VEGF | [142] |
| Perillaldehyde | Perilla frutescens | Female BALB/c nude mice | Inhibited tumor growth | Increased caspase-3, p53, catepsin and LC3-II | [143] |
| S-allylmercaptocysteine | Garlic | Female BALB/e nude mice | Inhibited tumor growth | - | [144] |
| 6-shogaol | Zingiber officinale | Athymic nude mice | Inhibited tumor growth | [145] |
| Natural Product/Extract | Study Group | Dosage | Comments | Reference | |
|---|---|---|---|---|---|
| Gastric cancer | Garlic extract and oil | 3365 residents with high risk of gastric cancer | 200 mg aged garlic extract and 1 mg steam distilled garlic oil for 7 years | Garlic supplementation was associated with a significantly reduced risk of incidence of gastric cancer and death | [146] |
| Rhus verniciflua extract rich in flavonoids | An 82-year old woman | 900 mg of extract for 5 months | Marked decrease in the polyploidy mass of the tumor and decreased lesion of the prepyloric antrum | [147] | |
| Marsdenia tenacissima extract | 1329 patients (51–68 years old) | Injection: 40–80 mL/dose, 7–21 doses/session orally: 6–7.2 g/dose, 30 doses/session | Adjuvant therapy with anticancer treatment (typically folinic acid + fluorouracil + oxaliplatin) improved the response to chemotherapy and reduced thrombocytopenia, anemia, nausea, peripheral neurotoxicity and hepatic injury | [148] | |
| Curcumin from Curcuma longa | 25 patients | 500 mg/ day, 3 months | Histologic improvement of lesions | [149] | |
| Aloe arborescens rich in antraquinonic and anthracenic molecules | 240 patients (58–79 years old) | 10 mL/ thrice daily orally (300 g of leaves in 500 g of honey in 40 mL of 40% alcohol) 6 days before and during chemotherapy | Adjuvant therapy of metastatic solid tumor with 5-florouracil. Increased tumor regression | [150] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakonieczna, S.; Grabarska, A.; Kukula-Koch, W. The Potential Anticancer Activity of Phytoconstituents against Gastric Cancer—A Review on In Vitro, In Vivo, and Clinical Studies. Int. J. Mol. Sci. 2020, 21, 8307. https://doi.org/10.3390/ijms21218307
Nakonieczna S, Grabarska A, Kukula-Koch W. The Potential Anticancer Activity of Phytoconstituents against Gastric Cancer—A Review on In Vitro, In Vivo, and Clinical Studies. International Journal of Molecular Sciences. 2020; 21(21):8307. https://doi.org/10.3390/ijms21218307
Chicago/Turabian StyleNakonieczna, Sylwia, Aneta Grabarska, and Wirginia Kukula-Koch. 2020. "The Potential Anticancer Activity of Phytoconstituents against Gastric Cancer—A Review on In Vitro, In Vivo, and Clinical Studies" International Journal of Molecular Sciences 21, no. 21: 8307. https://doi.org/10.3390/ijms21218307






