Regulation of Glutamatergic Activity via Bidirectional Activation of Two Select Receptors as a Novel Approach in Antipsychotic Drug Discovery
Abstract
:1. Introduction
2. Malfunction of Receptors in Patients with Schizophrenia
| (A) | ||||
| Receptor | Method | Brain Structure | Change | |
| M1/M4 | ||||
| [3H] pirenzepine binding | caudate-putamen | decrease | [68] | |
| [3H] pirenzepine binding | hippocampal formation | decrease | [69] | |
| [3H] pirenzepine binding | Brodmann area 9 | decrease | [70] | |
| [3H] pirenzepine binding | Brodmann area 40 | no change | [70] | |
| [3H] pirenzepine binding | Brodmann area 9 | decrease | [71] | |
| [3H] pirenzepine binding | Brodmann area 46 | decrease | [71] | |
| [3H] pirenzepine binding | anterior cingulate cortex | decrease | [72] | |
| [3H] pirenzepine binding | superior temporal gyrus | decrease | [73] | |
| [3H] pirenzepine binding | posterior cingulate cortex | decrease | [74] | |
| [3H] pirenzepine binding | hippocampal formation | decrease | [75] | |
| [3H] pirenzepine binding | Brodmann area 6 | decrease | [76] | |
| M1 | ||||
| in situ hybridization | caudate-putamen | no change | [77] | |
| in situ hybridization, Western blot | Brodmann area 9 | decrease | [70] | |
| in situ hybridization, Western blot | Brodmann area 40 | decrease no change | [70] | |
| cDNA | Brodmann area 6 | decrease | [78] | |
| in situ hybridization, Western blot | thalamus | no change | [79] | |
| in situ hybridization | hippocampal formation | no change | [75] | |
| immunohistochemistry | Brodmann area 9 | decrease | [80] | |
| immunohistochemistry | Brodmann area 17 | decrease | [80] | |
| immunohistochemistry | thalamus | no change | [80] | |
| immunohistochemistry | hippocampal formation | no change | [80] | |
| M4 | ||||
| in situ hybridization, Western blot | Brodmann area 9 | no change | [70] | |
| in situ hybridization, Western blot | Brodmann area 40 | decrease no change | [70] | |
| in situ hybridization, Western blot | thalamus | no change | [79] | |
| in situ hybridization | hippocampal formation | decrease | [75] | |
| M2/M4 | ||||
| [3H]AF-DX 384 | anterior cingulate cortex | no change | [81] | |
| (B) | ||||
| Receptor | Method | Brain Structure | Change | |
| GABAB | ||||
| immunohistochemistry | hippocampal formation | decrease (not quantified) | [82] | |
| immunohistochemistry | entorhinal cortex, inferior temporal cortex | decrease (not quantified) | [83] | |
| immunohistochemistry, Western blot | Brodmann area 9 | decrease (not quantified), decrease (GABAB1a) | [84] | |
| Western blot | lateral cerebellum | decrease | [85] | |
| Western blot | Brodmann area 9 | decrease | [86] | |
| (C) | ||||
| Receptor | Method | Brain Structure | Change | |
| mGlu5 | ||||
| [3H]MPEP binding | Brodmann area 46 | no change | [87] | |
| [3H]MPEP binding | Brodmann area 24 | no change | [88] | |
| in situ hybridization | Brodmann area 9 | no change | [89] | |
| in situ hybridization | Brodmann area 10 | no change | [89] | |
| in situ hybridization | Brodmann area 11 | increase | [89] | |
| in situ hybridization | hippocampal formation | no change | [90] | |
| in situ hybridization | parahippocampal gyrus | no change | [90] | |
| in situ hybridization | thalamus | no change | [91] | |
| Western blot | Brodmann area 9 | no change | [92] | |
| Western blot | Brodmann area 11 | no change | [92] | |
| Western blot | Brodmann area 32 | no change | [92] | |
| Western blot | Brodmann area 46 | no change | [92] | |
| Western blot | nucleus accumbens | no change | [92] | |
| Western blot | caudate nucleus | no change | [92] | |
| Western blot | putamen | no change | [92] | |
| Western blot | Brodmann area 10 | no change | [93] | |
| Western blot | lateral cerebellum | decrease (monomer) | [94] | |
| Western blot | Brodmann area 9 | decrease (monomer) | [94] | |
| Western blot | Brodmann area 46 | no change (monomer) | [87] | |
| Western blot | Brodmann area 46 | increase (total and dimer) | [95] | |
| RT-PCR | Brodmann area 9 | no change | [96] | |
| qRT-PCR | lateral cerebellum | decrease | [94] | |
| qRT-PCR | Brodmann area 46 | no change | [95] | |
| qPCR | Brodmann area 10 | no change | [97] | |
| qPCR | Brodmann area 46 | no change | [97] | |
| mGlu2/3 | ||||
| [3H]LY341495 binding | Brodmann area 24 | no change | [88] | |
| [3H]LY341495 binding | Brodmann area 17 | no change | [98] | |
| [3H]LY341495 binding | Brodmann area 24 | no change | [98] | |
| [3H]LY341495 binding | Brodmann area 46 | no change | [98] | |
| [3H]LY341495 binding | Brodmann area 46 | no change | [99] | |
| Western blot | Brodmann area 46 | no change | [100] | |
| Western blot | PFC | increase | [92] | |
| mGlu2 | ||||
| in situ hybridization | dentate gyrus | decrease | [101] | |
| in situ hybridization | CA3 | decrease | [101] | |
| in situ hybridization | CA2 | decrease | [101] | |
| in situ hybridization | subiculum | decrease | [101] | |
| in situ hybridization | parahipocampal gyrus | decrease | [101] | |
| in situ hybridization | thalamus | no change | [91] | |
| in situ hybridization | prefrontal cortex (white matter) | increase | [102] | |
| in situ hybridization | paranigral nucleus | increase | [102] | |
| Western blot | prefrontal cortex | no change | [103] | |
| Western blot | temporal cortex | no change | [103] | |
| Western blot | motor cortex | no change | [103] | |
| mGlu4 | ||||
| in situ hybridization | thalamus | no change | [91] | |
| Western blot | Brodmann area 9 | no change | [92] | |
| Western blot | Brodmann area 11 | no change | [92] | |
| Western blot | Brodmann area 32 | no change | [92] | |
| Western blot | Brodmann area 46 | no change | [92] | |
| Western blot | nucleus accumbens | no change | [92] | |
| Western blot | caudate nucleus | no change | [92] | |
| Western blot | putamen | no change | [92] | |
| mGlu7 | ||||
| in situ hybridization | thalamus | no change | [91] | |
3. Regulation of Glutamate Release
3.1. Glutamatergic Network in the Brain
3.2. Presynaptic Regulation of Glutamate Release—Autoreceptors
3.2.1. mGlu2 Receptors
3.2.2. Group III mGlu Receptors
3.3. Presynaptic Regulation of Glutamate Release—Heteroreceptors
3.3.1. GABAB Receptor
3.3.2. Muscarinic M4 Receptor
3.4. Postsynaptic Regulation of Neuronal Circuits in Patients with Schizophrenia
3.4.1. mGlu5 Receptor
3.4.2. Muscarinic M1 Receptor
3.4.3. Muscarinic M5 Receptor
3.4.4. Comparative Assessment of Receptor Expression
| (A) | ||||
| Receptor | Protein | mRNA | ||
| M1 | cortex (including: mPFC, entorhinal cortex) | [162,170,177,178] | cortex (including piriform cortex, visual cortex) | [171,179,180,181,182,183] |
| nucleus accumbens | [171] | |||
| hippocampus | [162,170] | caudate-putamen | [171,179,183] | |
| caudate-putamen | [162] | basolateral amygdala | [182] | |
| nucleus accumbens | [162,184] | olfactory tubercule | [179] | |
| thalamus | [162] | primary olfactory cortex | [182,183] | |
| amygdala | [162] | hippocampus | [182] | |
| brainstem | [162] | olfactory nuclei | [182] | |
| olfactory tubercule | [162] | olfactory bulb | [171,182,183] | |
| M4 | cortex | [162,185] | ||
| caudate-putamen | [162,184,185] | |||
| nucleus accumbens | [162] | cortex (including primary olfactory cortex, visual cortex, piriform cortex) | [171,179,180,181,182,183,185] | |
| thalamus | [162] | nucleus accumbens | [171] | |
| hippocampus | [185] | caudate-putamen | [171,179,182,183,185] | |
| substantia nigra | [162] | hippocampus | [182,183,185] | |
| brainstem | [162] | olfactory tubercule | [171,179,183] | |
| olfactory tubercule | [162] | olfactory bulb | [182,185] | |
| olfactory bulb | [185] | |||
| islands of Calleja | [162] | |||
| M5 | brainstem | [162] | substantia nigra (pc) | [171,186] |
| ventral tegmental area | [171,186] | |||
| hippocampus (CA1) | [186] | |||
| ventral subiculum | [186] | |||
| GABAB | cortex | [187,188] | cortex (including piriform cortex) | [189] |
| caudate-putamen | [187,188] | hippocampus | [189] | |
| globus pallidus | [188] | nucleus accumbens | [189] | |
| nucleus accumbens | [188] | caudate-putamen | [189] | |
| amygdala | [188] | thalamus | [189] | |
| hippocampus | [187,188] | hypothalamus | [189] | |
| thalamus | [187,188] | substantia nigra (pc) | [189] | |
| hypothalamus | [188] | ventral tegmental area | [189] | |
| ventral tegmental area | [188] | cerebellum | [189] | |
| substantia nigra | [188] | pons | [189] | |
| cerebellum | [187,188] | |||
| olfactory bulb | [187] | (GABAB1) | ||
| medulla/pons | [187] | |||
| cortex (including piriform cortex) | [190,191] | |||
| (GABAB1A, GABAB1B, GABAB2) | ||||
| caudate-putamen | [190] | |||
| nucleus accumbens | [190] | |||
| globus pallidus | [190] | |||
| substantia nigra | [190] | |||
| amygdala | [190] | |||
| hippocampus | [190,191] | |||
| hypothalamus | [190] | |||
| thalamus | [190,191] | |||
| cerebellum | [190,191] | |||
| ventral tegmental area | [190] | |||
| pons | [190] | |||
| (GABAB2) | ||||
| cortex (including piriform cortex, frontal cortex, occipital cortex, retrosplenial cortex, | [192] | |||
| temporal cortex) | ||||
| hippocampus | [192] | |||
| thalamus | [192] | |||
| hypothalamus | [192] | |||
| striatum | [192] | |||
| nucleus accumbens | [192] | |||
| substantia nigra | [192] | |||
| amygdala, | [192] | |||
| cerebellum | [192] | |||
| (GABAB1A, GABAB2) | ||||
| mGlu5 | cortex (including piriform cortex) | [159,193] | cortex (including entorhinal cortex) | [194,195,196,197,198] |
| caudate-putamen | [159,193,199] | hippocampus | [194,195,196,197,198,200] | |
| nucleus accumbens | [159,193] | caudate-putamen | [194,195,196,197,198,201] | |
| hippocampus | [159,193,202,203] | nucleus accumbens | [196,197,198] | |
| thalamus | [159] | subiculum | [196,197] | |
| hypothalamus | [159] | thalamus | [196,198] | |
| subiculum | [159] | hypothalamus | [196] | |
| cerebellum | [159] | inferior and superior colliculi | [196,198] | |
| inferior colliculus | [193] | amygdala | [200] | |
| olfactory bulb | [159,193] | olfactory bulb | [196,198] | |
| olfactory tubercule | [159,193] | olfactory tubercule | [196,197] | |
| mGlu2 | cortex (including piriform cortex, entorhinal cortex) | [114,204,205] | ||
| hippocampus | [114,202,204,205] | cortex (including piriform cortex, entorhinal cortex) | [194,197,206,207] | |
| thalamus | [114,204,205] | hippocampus | [194,197,207] | |
| basolateral amygdala | [114,204] | thalamus | [197,206,207] | |
| caudate-putamen | [114,204,205] | basolateral amygdala | [206,207] | |
| nucleus accumbens | [114,204] | caudate-putamen | [206] | |
| globus pallidus | [204] | nucleus accumbens | [206] | |
| substantia nigra | [204] | globus pallidus | [206] | |
| ventral tegmental area | [114] | cerebellum | [194,197,206,208] | |
| cerebellum | [114,204] | olfactory tubercule | [206] | |
| olfactory bulb | [114,204] | |||
| olfactory tubercule | [114,204] | |||
| mGlu4 | cortex (including piriform cortex) | [209] | cortex (including entorhinal cortex) | [194,197,210,211,212] |
| caudate-putamen | [209] | caudate-putamen | [197,210,212,213] | |
| substantia nigra | [209] | substantia nigra | [197] | |
| hippocampus | [202,209] | nucleus accumbens | [197,212,213] | |
| thalamus | [209] | thalamus | [194,197,210,212,213,214] | |
| hypothalamus | [209] | hypothalamus | [212] | |
| amygdala | [209] | hippocampus | [194,210,214] | |
| superior colliculus | [209] | amygdala | [212] | |
| cerebellum | [209,215,216] | lateral septum | [210,214] | |
| olfactory bulb | [209] | cerebellum | [194,197,208,212,214] | |
| olfactory tubercule | [209] | olfactory bulb | [210,212,214] | |
| olfactory tubercule | [197,214] | |||
| mGlu7 | cortex (including piriform cortex) | [136,217] | ||
| caudate-putamen | [136] | |||
| nucleus accumbens | [136] | |||
| globus pallidus | [136] | |||
| substantia nigra | [136] | |||
| thalamus | [136] | |||
| hypothalamus | [136] | |||
| hippocampus | [136] | |||
| subiculum | [136] | |||
| amygdala | [136] | cortex | [212,218,219,220] | |
| ventral tegmental area | [136] | caudate-putamen | [212,213,218,219,220] | |
| olfactory bulb | [136] | globus pallidus | [212] | |
| olfactory tubercule | [217] | nucleus accumbens | [212,213,218,220] | |
| substantia nigra | [212] | |||
| (mGlu7a) | thalamus | [212,213,218,219,220] | ||
| cortex | [136] | hypothalamus | [212,219,220] | |
| hippocampus | [136] | amygdala | [212,220] | |
| substantia nigra | [136] | hippocampus | [218,219,220] | |
| globus pallidus | [136] | ventral tegmental area | [212] | |
| amygdala | [136] | superior and inferior colliculi | [219] | |
| cerebellum | [136] | locus coeruleus | [218] | |
| (mGlu7b) | cerebellum | [208,212,218,219,220] | ||
| cortex (including piriform cortex) | [221,222] | olfactory bulb | [212,218,219] | |
| hippocampus | [202,221,222,223] | olfactory tubercule | [219,220] | |
| thalamus | [222] | |||
| caudate-putamen | [222] | |||
| globus pallidus | [222] | |||
| nucleus accumbens | [222] | |||
| locus coeruleus | [222] | |||
| cerebellum | [222] | |||
| olfactory bulb | [221] | |||
| mGlu8 | cortex (including piriform cortex) | [218,224,225] | ||
| striatum | [213,218,225] | |||
| nucleus accumbens | [213,225] | |||
| globus pallidus | [225] | |||
| piriform cortex | [216] | substantia nigra | [225] | |
| entorhinal cortex | [216] | thalamus | [213,218,224,225] | |
| hippocampus | [202,226] | hypothalamus | [225] | |
| olfactory bulb | [216] | hippocampus | [218,224,225,226] | |
| amygdala | [218,225] | |||
| cerebellum | [208,218,224,225] | |||
| olfactory bulb | [218,224,227] | |||
| olfactory tubercule | [227] | |||
| (B) | ||||
| Receptor | Protein | mRNA | ||
| M1 | frontal cortex | [69,79,227] | ||
| parietal cortex | [70,228] | |||
| temporal cortex | [228] | frontal cortex | [70] | |
| occipital cortex | [228] | parietal cortex | [70] | |
| primary visual cortex | [80] | thalamus | [79] | |
| thalamus | [79,80] | hippocampus | [75] | |
| hippocampus | [80,228] | caudate-putamen | [77] | |
| nucleus basalis | [228] | |||
| putamen | [228] | |||
| M4 | frontal cortex | [70,228] | ||
| temporal cortex | [228] | |||
| parietal cortex | [70,228] | frontal cortex | [70] | |
| occipital cortex | [228] | parietal cortex | [70] | |
| thalamus | [79] | thalamus | [79] | |
| hippocampus | [228] | hippocampus | [75] | |
| nucleus basalis | [228] | |||
| putamen | [228] | |||
| M5 | frontal cortex | [228] | ||
| temporal cortex | [228] | |||
| parietal cortex | [228] | |||
| occipital cortex | [228] | |||
| nucleus basalis | [228] | |||
| GABAB | prefrontal cortex | [229] | ||
| frontal cortex | [192] | |||
| occipital cortex | [192] | |||
| temporal cortex | [192] | |||
| caudate nucleus | [192,229] | |||
| putamen | [192,229] | |||
| globus pallidus | [229] | |||
| substantia nigra | [192,229] | |||
| nucleus accumbens | [192] | |||
| entorhinal cortex | [230] | thalamus | [192] | |
| caudate | [230] | hypothalamus | [192] | |
| putamen | [230] | hippocampus | [192,229] | |
| globus pallidus | [230] | amygdala | [192] | |
| thalamus | [230] | corpus callosum | [192] | |
| hippocampus | [230] | cerebellum | [192,229] | |
| substantia nigra | [230] | |||
| cerebellum | [230] | |||
| cortex | [191] | |||
| (GABAB1, GABAB2) | putamen | [191] | ||
| caudate nucleus | [191] | |||
| substantia nigra | [191] | |||
| thalamus | [191] | |||
| hippocampus | [191] | |||
| amygdala | [191] | |||
| cerebellum | [191] | |||
| (GABAB2) | ||||
| mGlu5 | frontal cortex | [94] | cortex (including frontal cortex, prefrontal cortex) | [89,94,153] |
| hippocampus | [231] | hippocampus | [90,153] | |
| lateral cerebellum | [94] | parahippocampal gyrus | [90] | |
| cerebellum | [94,153] | |||
| mGlu2 | prefrontal cortex | [103] | prefrontal cortex | [102] |
| temporal cortex | [103] | thalamus | [91] | |
| dorsolateral prefrontal cortex | [100] | hippocampus | [101] | |
| motor cortex | [103] | ventral mesencephalon (including substantia nigra) | [102] | |
| hippocampus | [231] | |||
| mGlu4 | hippocampus | [231] | cortex | [232] |
| putamen | [232] | |||
| substantia nigra | [232] | |||
| caudate nucleus | [232] | |||
| thalamus | [91,232,233] | |||
| hypothalamus | [232,233] | |||
| hippocampus | [232,233] | |||
| amygdala | [232] | |||
| corpus callosum | [232] | |||
| cerebellum | [232,233,234] | |||
| mGlu7 | cortex (including entorhinal cortex) | [235] | ||
| thalamus | [91,234,235] | |||
| hypothalamus | [234] | |||
| hippocampus | [234,235] | |||
| caudate-putamen | [235] | |||
| cerebellum | [235] | |||
| mGlu8 | cortex | [133] | ||
| putamen | [133,225] | |||
| caudate nucleus | [133,225] | |||
| globus pallidus | [225] | |||
| nucleus accumbens | [225] | |||
| substantia nigra | [225] | |||
| cingulate gyrus | [225] | |||
| thalamus | [91,133,225] | |||
| hypothalamus | [225] | |||
| hippocampus | [225] | |||
| amygdala | [133,225] | |||
| locus coeruleus | [225] | |||
| cerebellum | [133,225] | |||
4. Strategies Based on Bidirectional Inhibition of Glutamate Release
4.1. Simultaneous Administration of Ligands Activating Receptors Associated with Adenyl Cyclase Activity
4.1.1. Heterodimerization
4.2. Simultaneous Administration of Ligands Activating Receptors Associated with Adenyl Cyclase and the Inositol Phosphate Signaling Pathway
5. Conclusions
- −
- the dose of each compound may be reduced and the antipsychotic-like efficacy is the same as the highest dose of each compound administered alone (this approach may potentially allow us to avoid putative adverse effects or unnecessary exposure of the prodrug to patients, as shown previously for mGlu2/3 agonists);
- −
- the action of the combined treatment might be selective in specific areas and thus may target a specific group of symptoms;
- −
- the ligands administered in combinations may complement the action of the other ligand and compensate for possible receptor dysfunctions, activating both homodimers and heterodimers/heterocomplexes.
Funding
Conflicts of Interest
References
- Tandon, R.; Gaebel, W.; Barch, D.M.; Bustillo, J.; Gur, R.E.; Heckers, S.; Malaspina, D.; Owen, M.J.; Schultz, S.; Tsuang, M.; et al. Definition and description of schizophrenia in the DSM-5. Schizophr. Res. 2013, 150, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Braff, D.L.; Ryan, J.; Rissling, A.J.; Carpenter, W.T. Lack of use in the literature from the last 20 years supports dropping traditional schizophrenia subtypes from DSM-5 ICD-11. Schizophr. Bull. 2013, 39, 751–753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mcglashan, T.H.; Fenton, W.S. Classical subtypes for schizophrenia: Literature review for DSM-IV. Schizophr. Bull. 1991, 17, 609–632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jauhar, S.; McKenna, P.J.; Radua, J.; Fung, E.; Salvador, R.; Laws, K.R. Cognitive-behavioural therapy for the symptoms of schizophrenia: Systematic review and meta-analysis with examination of potential bias. Br. J. Psychiatry 2014, 204, 20–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wood, L.; Williams, C.; Billings, J.; Johnson, S. A systematic review and meta-analysis of cognitive behavioural informed psychological interventions for psychiatric inpatients with psychosis. Schizophr. Res. 2020, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Sykes, D.A.; Moore, H.; Stott, L.; Holliday, N.; Javitch, J.A.; Robert Lane, J.; Charlton, S.J. Extrapyramidal side effects of antipsychotics are linked to their association kinetics at dopamine D2 receptors. Nat. Commun. 2017, 8, 763. [Google Scholar] [CrossRef] [Green Version]
- Seeman, P.; Lee, T.; Chau-Wong, M.; Wong, K. Antipsychotic drug doses and neuroleptic/dopamine receptors. Nature 1976, 261, 717–719. [Google Scholar] [CrossRef]
- Kapur, S.; Mamo, D. Half a century of antipsychotics and still a central role for dopamine D2 receptors. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2003, 27, 1081–1090. [Google Scholar] [CrossRef]
- Aringhieri, S.; Carli, M.; Kolachalam, S.; Verdesca, V.; Cini, E.; Rossi, M.; McCormick, P.J.; Corsini, G.U.; Maggio, R.; Scarselli, M. Molecular targets of atypical antipsychotics: From mechanism of action to clinical differences. Pharmacol. Ther. 2018, 192, 20–41. [Google Scholar] [CrossRef]
- Aringhieri, S.; Kolachalam, S.; Gerace, C.; Carli, M.; Verdesca, V.; Brunacci, M.G.; Rossi, C.; Ippolito, C.; Solini, A.; Corsini, G.U.; et al. Clozapine as the most efficacious antipsychotic for activating ERK 1/2 kinases: Role of 5-HT2A receptor agonism. Eur. Neuropsychopharmacol. 2017, 27, 383–398. [Google Scholar] [CrossRef]
- Schmid, C.L.; Streicher, J.M.; Meltzer, H.Y.; Bohn, L.M. Clozapine acts as an agonist at serotonin 2A receptors to counter MK-801-induced behaviors through a βarrestin2-independent activation of akt. Neuropsychopharmacology 2014, 39, 1902–1913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leucht, S.; Corves, C.; Arbter, D.; Engel, R.R.; Li, C.; Davis, J.M. Second-generation versus first-generation antipsychotic drugs for schizophrenia: A meta-analysis. Lancet 2009, 373, 31–41. [Google Scholar] [CrossRef]
- Campden-Main, B.C.; Wegielski, Z. The Control of Deviant Behavior in Chronically Disturbed Psychotic Patients by the Oral Administration of Reserpine. Ann. N. Y. Acad. Sci. 1955, 61, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Kinross-Wright, V. Chlorpromazine and Reserpine in the Treatment of Psychoses. Ann. N. Y. Acad. Sci. 1955, 61, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Howes, O.; McCutcheon, R.; Stone, J. Glutamate and dopamine in schizophrenia: An update for the 21st century. J. Psychopharmacol. 2015, 29, 97–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Howes, O.D.; Kapur, S. A neurobiological hypothesis for the classification of schizophrenia: Type a (hyperdopaminergic) and type b (normodopaminergic). Br. J. Psychiatry 2014, 205, 1–3. [Google Scholar] [CrossRef]
- Reynolds, G.P. The pharmacogenetics of symptom response to Antipsychotic drugs. Psychiatry Investig. 2012, 9, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Stern, S.; Linker, S.; Vadodaria, K.C.; Marchetto, M.C.; Gage, F.H. Prediction of response to drug therapy in psychiatric disorders. Open Biol. 2018, 8, 180031. [Google Scholar] [CrossRef] [Green Version]
- Pouget, J.G.; Shams, T.A.; Tiwari, A.K.; Müller, D.J. Pharmacogenetics and outcome with antipsychotic drugs. Dialogues Clin. Neurosci. 2014, 16, 555–566. [Google Scholar]
- Luby, E.; Cohen, B.; Rosenbaum, G.; Gottlieb, J.S.; Kelley, R. Study Schizophrenomimetic Drug Sernyl. Arch. Neurol. Psychiatr. 1959, 81, 363–369. [Google Scholar] [CrossRef]
- Javitt, D.C.; Zukin, S.R. Recent Advances in the Phencyclidine Model of Schizophrenia. Am. J. Psychiatry 1991, 148, 1301–1308. [Google Scholar] [PubMed]
- Krystal, J.H.; Karper, L.P.; Seibyl, J.P.; Freeman, G.K.; Delaney, R.; Bremner, J.D.; Heninger, G.R.; Bowers, M.B.; Charney, D.S. Subanesthetic Effects of the Noncompetitive NMDA Antagonist, Ketamine, in Humans: Psychotomimetic, Perceptual, Cognitive, and Neuroendocrine Responses. Arch. Gen. Psychiatry 1994, 51, 199–214. [Google Scholar] [CrossRef] [PubMed]
- Domino, E.F.; Luby, E.D. Phencyclidine/schizophrenia: One view toward the past, the other to the future. Schizophr. Bull. 2012, 38, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Javitt, D.C. Negative schizophrenic symptomatology and the PCP (phencyclidine) model of schizophrenia. Hillside J. Clin. Psychiatry 1987, 9, 12–35. [Google Scholar] [PubMed]
- Conn, P.J.; Lindsley, C.W.; Jones, C.K. Activation of metabotropic glutamate receptors as a novel approach for the treatment of schizophrenia. Trends Pharmacol. Sci. 2009, 30, 25–31. [Google Scholar] [CrossRef] [Green Version]
- Coyle, J.T. Glutamate and Schizophrenia: Beyond the Dopamine Hypothesis. Cell. Mol. Neurobiol. 2006, 26, 4–6. [Google Scholar] [CrossRef]
- Singh, S.P.; Singh, V. Meta-analysis of the efficacy of adjunctive NMDA receptor modulators in chronic schizophrenia. CNS Drugs 2011, 25, 859–885. [Google Scholar] [CrossRef]
- Coyle, J.T.; Tsai, G. The NMDA receptor glycine modulatory site: A therapeutic target for improving cognition and reducing negative symptoms in schizophrenia. Psychopharmacology 2004, 174, 38. [Google Scholar] [CrossRef]
- Duncan, E.J.; Szilagyi, S.; Schwartz, M.P.; Bugarski-Kirola, D.; Kunzova, A.; Negi, S.; Stephanides, M.; Efferen, T.R.; Angrist, B.; Peselow, E.; et al. Effects of D-cycloserine on negative symptoms in schizophrenia. Schizophr. Res. 2004, 71, 239–248. [Google Scholar] [CrossRef]
- Evins, A.E.; Fitzgerald, S.M.; Wine, L.; Rosselli, R.; Goff, D.C. Placebo-controlled trial of glycine added to clozapine in schizophrenia. Am. J. Psychiatry 2000, 157, 826–828. [Google Scholar] [CrossRef]
- Goff, D.C.; Cather, C.; Gottlieb, J.D.; Evins, A.E.; Walsh, J.; Raeke, L.; Otto, M.W.; Schoenfeld, D.; Green, M.F. Once-weekly d-cycloserine effects on negative symptoms and cognition in schizophrenia: An exploratory study. Schizophr. Res. 2008, 106, 320–327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heresco-Levy, U.; Javitt, D.C. Comparative effects of glycine and D-cycloserine on persistent negative symptoms in schizophrenia: A retrospective analysis. Schizophr. Res. 2004, 66, 89–96. [Google Scholar] [CrossRef]
- Javitt, D.C. Is the glycine site half saturated or half unsaturated? Effects of glutamatergic drugs in schizophrenia patients. Curr. Opin. Psychiatry 2006, 19, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Lane, H.Y.; Tseng, P.T.; Chen, S.J.; Liu, C.Y.; Lin, C.H. Effect of N-methyl-D-aspartate-receptor-enhancing agents on cognition in patients with schizophrenia: A systematic review and meta-analysis of double-blind randomised controlled trials. J. Psychopharmacol. 2019, 33, 436–448. [Google Scholar] [CrossRef]
- Chaki, S. Group II metabotropic glutamate receptor agonists as a potential drug for schizophrenia. Eur. J. Pharmacol. 2010, 639, 59–66. [Google Scholar] [CrossRef]
- Lesage, A.; Steckler, T. Metabotropic glutamate mGlu1 receptor stimulation and blockade: Therapeutic opportunities in psychiatric illness. Eur. J. Pharmacol. 2010, 639, 2–16. [Google Scholar] [CrossRef]
- Marek, G.J. Metabotropic glutamate 2/3 (mGlu2/3) receptors, schizophrenia and cognition. Eur. J. Pharmacol. 2010, 639, 81–90. [Google Scholar] [CrossRef]
- Yasuhara, A.; Chaki, S. Metabotropic glutamate receptors: Potential drug targets for psychiatric disorders. Open Med. Chem. J. 2010, 4, 20–36. [Google Scholar] [CrossRef]
- Chaki, S.; Hikichi, H. Targeting of Metabotropic Glutamate Receptors for the Treatment of Schizophrenia. Curr. Pharm. Des. 2011, 17, 94–102. [Google Scholar] [CrossRef]
- Gregory, K.J.; Dong, E.N.; Meiler, J.; Conn, P.J. Allosteric modulation of metabotropic glutamate receptors: Structural insights and therapeutic potential. Neuropharmacology 2011, 60, 66–81. [Google Scholar] [CrossRef] [Green Version]
- Nicoletti, F.; Bockaert, J.; Collingridge, G.L.; Conn, P.J.; Ferraguti, F.; Schoepp, D.D.; Wroblewski, J.T.; Pin, J.P. Metabotropic glutamate receptors: From the workbench to the bedside. Neuropharmacology 2011, 60, 1017–1041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheffler, D.J.; Gregory, K.J.; Rook, J.M.; Conn, P.J. Allosteric Modulation of Metabotropic Glutamate Receptors. Adv. Pharmacol. 2011, 62, 37–77. [Google Scholar] [PubMed] [Green Version]
- Fell, M.J.; McKinzie, D.L.; Monn, J.A.; Svensson, K.A. Group II metabotropic glutamate receptor agonists and positive allosteric modulators as novel treatments for schizophrenia. Neuropharmacology 2012, 62, 1473–1483. [Google Scholar] [CrossRef]
- Vinson, P.N.; Conn, P.J. Metabotropic glutamate receptors as therapeutic targets for schizophrenia. Neuropharmacology 2012, 62, 1461–1472. [Google Scholar] [CrossRef] [Green Version]
- Gregory, K.J.; Noetzel, M.J.; Niswender, C.M. Pharmacology of metabotropic glutamate receptor allosteric modulators: Structural basis and therapeutic potential for CNS disorders. Prog. Mol. Biol. Transl. Sci. 2013, 115, 61–121. [Google Scholar]
- Nickols, H.; Conn, P.J. Development of allosteric modulators of GPCRs for treatment of CNS disorders. Neurobiol. Dis. 2014, 61, 55–71. [Google Scholar] [CrossRef] [Green Version]
- Li, M.-L.; Hu, X.-Q.; Li, F.; Gao, W.-J. Perspectives on the mGluR2/3 agonists as a therapeutic target for schizophrenia: Still promising or a dead end? Prog. Neuropsychopharmacol. Biol. Psychiatry 2015, 60, 66–76. [Google Scholar] [CrossRef] [Green Version]
- Golubeva, A.V.; Moloney, R.D.; O’Connor, R.M.; Dinan, T.G.; Cryan, J.F. Metabotropic Glutamate Receptors in Central Nervous System Diseases. Curr. Drug Targets 2016, 17, 538–616. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.G.; Conn, P.J. Group I and group II metabotropic glutamate receptor allosteric modulators as novel potential antipsychotics. Curr. Opin. Pharmacol. 2015, 20, 40–45. [Google Scholar] [CrossRef] [Green Version]
- Muguruza, C.; Meana, J.J.; Callado, L.F. Group II metabotropic glutamate receptors as targets for novel antipsychotic drugs. Front. Pharmacol. 2016, 7, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Wierońska, J.M.; Zorn, S.H.; Doller, D.; Pilc, A. Metabotropic glutamate receptors as targets for new antipsychotic drugs: Historical perspective and critical comparative assessment. Pharmacol. Ther. 2016, 157, 10–27. [Google Scholar] [CrossRef] [PubMed]
- Foster, D.J.; Conn, P.J. Allosteric Modulation of GPCRs: New Insights and Potential Utility for Treatment of Schizophrenia and Other CNS Disorders. Neuron 2017, 94, 431–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maksymetz, J.; Moran, S.P.; Conn, P.J. Targeting metabotropic glutamate receptors for novel treatments of schizophrenia. Mol. Brain 2017, 10, 1–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nicoletti, F.; Orlando, R.; Menna, L.D.; Cannella, M.; Notartomaso, S.; Mascio, G.; Iacovelli, L.; Matrisciano, F.; Fazio, F.; Caraci, F.; et al. Targeting mGlu receptors for optimization of antipsychotic activity and disease-modifying effect in schizophrenia. Front. Psychiatry 2019, 10, 49. [Google Scholar] [CrossRef]
- Stansley, B.J.; Conn, P.J. Neuropharmacological Insight from Allosteric Modulation of mGlu Receptors. Trends Pharmacol. Sci. 2019, 40, 240–252. [Google Scholar] [CrossRef]
- Kinon, B.J.; Zhang, L.; Millen, B.A.; Osuntokun, O.O.; Williams, J.E.; Kollack-Walker, S.; Jackson, K.; Kryzhanovskaya, L.; Jarkova, N. A multicenter, inpatient, phase 2, double-blind, placebo-controlled dose-ranging study of LY2140023 monohydrate in patients with DSM-IV schizophrenia. J. Clin. Psychopharmacol. 2011, 31, 349–355. [Google Scholar] [CrossRef]
- Patil, S.T.; Zhang, L.; Martenyi, F.; Lowe, S.L.; Jackson, K.A.L.; Andreev, B.V.; Avedisova, A.S.; Bardenstein, L.M.; Gurovich, I.Y.; Morozova, M.A.; et al. Activation of mGlu2/3 receptors as a new approach to treat schizophrenia: A randomized Phase 2 clinical trial. Nat. Med. 2007, 13, 1102–1107. [Google Scholar] [CrossRef]
- Erdely, H.A.; Tamminga, C.A.; Roberts, R.C.; Vogel, M.W. Regional alterations in RGS4 protein in Schizophrenia. Synapse 2006, 59, 472–479. [Google Scholar] [CrossRef]
- Bowden, N.A.; Scott, R.J.; Tooney, P.A. Altered expression of regulator of G-protein signalling 4 (RGS4) mRNA in the superior temporal gyrus in schizophrenia. Schizophr. Res. 2007, 89, 165–168. [Google Scholar] [CrossRef]
- Ding, L.; Hegde, A.N. Expression of RGS4 Splice Variants in Dorsolateral Prefrontal Cortex of Schizophrenic and Bipolar Disorder Patients. Biol. Psychiatry 2009, 65, 541–545. [Google Scholar] [CrossRef]
- Mirnics, K.; Middleton, F.A.; Stanwood, G.D.; Lewis, D.A.; Levitt, P. Disease-specific changes in regulator of G-protein signaling 4 (RGS4) expression in schizophrenia. Mol. Psychiatry 2001, 6, 293–301. [Google Scholar] [CrossRef] [PubMed]
- De Vries, L.; Zheng, B.; Fischer, T.; Elenko, E.; Farquhar, M.G. The regulator of G protein signaling family. Annu. Rev. Pharmacol. Toxicol. 2000, 40, 235–271. [Google Scholar] [CrossRef] [PubMed]
- Schwendt, M.; Sigmon, S.A.; McGinty, J.F. RGS4 overexpression in the rat dorsal striatum modulates mGluR5- and amphetamine-mediated behavior and signaling. Psychopharmacology 2012, 221, 621–635. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Jung, S.; Son, H.; Kim, S.; Choi, J.; Lee, D.H.; Roh, G.S.; Kang, S.S.; Cho, G.J.; Choi, W.S.; et al. The GABAB receptor associates with regulators of G-protein signaling 4 protein in the mouse prefrontal cortex and hypothalamus. BMB Rep. 2014, 47, 324–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, J.; Guzman, J.N.; Tkatch, T.; Chen, S.; Goldberg, J.A.; Ebert, P.J.; Levitt, P.; Wilson, C.J.; Hamm, H.E.; Surmeier, D.J. RGS4-dependent attenuation of M4 autoreceptor function in striatal cholinergic interneurons following dopamine depletion. Nat. Neurosci. 2006, 9, 832–842. [Google Scholar] [CrossRef] [PubMed]
- Carbon, M.; Correll, C.U. Clinical predictors of therapeutic response to antipsychotics in schizophrenia. Dialogues Clin. Neurosci. 2014, 16, 505–524. [Google Scholar]
- Potkin, S.G.; Kane, J.M.; Correll, C.U.; Lindenmayer, J.P.; Agid, O.; Marder, S.R.; Olfson, M.; Howes, O.D. The neurobiology of treatment-resistant schizophrenia: Paths to antipsychotic resistance and a roadmap for future research. npj Schizophr. 2020, 6, 1. [Google Scholar] [CrossRef]
- Dean, B.; Crook, J.M.; Opeskin, K.; Hill, C.; Keks, N.; Copolov, D.L. The density of muscarinic M1 receptors is decreased in the caudate-putamen of subjects with schizophrenia. Mol. Psychiatry 1996, 1, 54–58. [Google Scholar]
- Crook, J.M.; Tomaskovic-Crook, E.; Copolov, D.L.; Dean, B. Decreased muscarinic receptor binding in subjects with schizophrenia: A study of the human hippocampal formation. Biol Psychiatry 2000, 48, 381–388. [Google Scholar] [CrossRef]
- Dean, B.; McLeod, M.; Keriakous, D.; McKenzie, J.; Scarr, E. Decreased muscarinic 1 receptors in the dorsolateral prefrontal cortex of subjects with schizophrenia. Mol. Psychiatry 2002, 7, 1083–1091. [Google Scholar] [CrossRef] [Green Version]
- Crook, J.M.; Tomaskovic-Crook, E.; Copolov, D.L.; Dean, B. Low muscarinic receptor binding in prefrontal cortex from subjects with schizophrenia: A study of brodmann’s areas 8, 9, 10, and 46 and the effects of neuroleptic drug treatment. Am. J. Psychiatry 2001, 158, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Zavitsanou, K.; Katsifis, A.; Mattner, F.; Huang, X.-F. Investigation of M1/M4 muscarinic receptors in the anterior cingulate cortex in schizophrenia, bipolar disorder, and major depression disorder. Neuropsychopharmacology 2004, 29, 619–625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, C.; Huang, X.F. Decreased density of muscarinic receptors in the superior temporal gyrus in schizophrenia. J. Neurosci. Res. 2005, 81, 883–890. [Google Scholar] [CrossRef]
- Newell, K.A.; Zavitsanou, K.; Jew, S.K.; Huang, X.-F. Alterations of muscarinic and GABA receptor binding in the posterior cingulate cortex in schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 225–233. [Google Scholar] [CrossRef]
- Scarr, E.; Sundram, S.; Keriakous, D.; Dean, B. Altered Hippocampal Muscarinic M4, but Not M1, Receptor Expression from Subjects with Schizophrenia. Biol. Psychiatry 2007, 61, 1161–1170. [Google Scholar] [CrossRef]
- Dean, B.; Soulby, A.; Evin, G.M.; Scarr, E. Levels of [3H]pirenzepine binding in Brodmann’s area 6 from subjects with schizophrenia is not associated with changes in the transcription factor SP1 or BACE1. Schizophr. Res. 2008, 106, 229–236. [Google Scholar] [CrossRef]
- Dean, B.; Crook, J.M.; Pavey, G.; Opeskin, K.; Copolov, D.L. Muscarinic 1 and 2 receptor mRNA in the human caudate-putamen: No change in m1 mRNA in schizophrenia. Mol. Psychiatry 2000, 5, 203–207. [Google Scholar] [CrossRef] [Green Version]
- Mancama, D.; Arranz, M.J.; Landau, S.; Kerwin, R. Reduced expression of the muscarinic 1 receptor cortical subtype in schizophrenia. Am. J. Med. Genet. Neuropsychiatr. Genet. 2003, 119B, 2–6. [Google Scholar] [CrossRef]
- Dean, B.; Gray, L.; Keriakous, D.; Scarr, E. A comparison of M1 and M4 muscarinic receptors in the thalamus from control subjects and subjects with schizophrenia. Thalamus Relat. Syst. 2004, 2, 287–295. [Google Scholar] [CrossRef]
- Scarr, E.; Hopper, S.; Vos, V.; Suk Seo, M.; Everall, I.P.; Aumann, T.D.; Chana, G.; Dean, B. Low levels of muscarinic M1 receptor–positive neurons in cortical layers III and V in Brodmann areas 9 and 17 from individuals with schizophrenia. J. Psychiatry Neurosci. 2018, 43, 338–346. [Google Scholar] [CrossRef] [Green Version]
- Zavitsanou, K.; Katsifis, A.; Yu, Y.; Huang, X.F. M2/M4 muscarinic receptor binding in the anterior cingulate cortex in schizophrenia and mood disorders. Brain Res. Bull. 2005, 65, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Mizukami, K.; Sasaki, M.; Ishikawa, M.; Iwakiri, M.; Hidaka, S.; Shiraishi, H.; Iritani, S. Immunohistochemical localization of gamma-aminobutyric acid(B) receptor in the hippocampus of subjects with schizophrenia. Neurosci. Lett. 2000, 283, 101–104. [Google Scholar] [CrossRef]
- Mizukami, K.; Ishikawa, M.; Hidaka, S.; Iwakiri, M.; Sasaki, M.; Iritani, S. Immunohistochemical localization of GABA B receptor in the entorhinal cortex and inferior temporal cortex of schizophrenic brain. Prog. Neuropsychopharmacol. Biol. Psychiatry 2002, 26, 393–396. [Google Scholar] [CrossRef]
- Ishikawa, M.; Mizukami, K.; Iwakiri, M.; Asada, T. Immunohistochemical and immunoblot analysis of γ-aminobutyric acid B receptor in the prefrontal cortex of subjects with schizophrenia and bipolar disorder. Neurosci. Lett. 2005, 383, 272–277. [Google Scholar] [CrossRef]
- Fatemi, S.H.; Folsom, T.D.; Thuras, P.D. Deficits in GABA(B) receptor system in schizophrenia and mood disorders: A postmortem study. Schizophr. Res. 2011, 128, 37–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fatemi, S.H.; Folsom, T.D.; Thuras, P.D. GABAA and GABAB receptor dysregulation in superior frontal cortex of subjects with schizophrenia and bipolar disorder. Synapse 2017, 71, e21973. [Google Scholar] [CrossRef]
- Matosin, N.; Frank, E.; Deng, C.; Huang, X.F.; Newell, K.A. Metabotropic glutamate receptor 5 binding and protein expression in schizophrenia and following antipsychotic drug treatment. Schizophr. Res. 2013, 146, 1–3. [Google Scholar] [CrossRef] [Green Version]
- Matosin, N.; Fernandez-Enright, F.; Frank, E.; Deng, C.; Wong, J.; Huang, X.F.; Newell, K.A. Metabotropic glutamate receptor mGluR2/3 and mGluR5 binding in the anterior cingulate cortex in psychotic and nonpsychotic depression, bipolar disorder and schizophrenia: Implications for novel mGluR-based therapeutics. J. Psychiatry Neurosci. 2014, 39, 407–416. [Google Scholar] [CrossRef] [Green Version]
- Ohnuma, T.; Augood, S.J.; Arai, H.; McKenna, P.J.; Emson, P.C. Expression of the human excitatory amino acid transporter 2 and metabotropic glutamate receptors 3 and 5 in the prefrontal cortex from normal individuals and patients with schizophrenia. Mol. Brain Res. 1998, 56, 207–217. [Google Scholar] [CrossRef]
- Ohnuma, T.; Tessler, S.; Arai, H.; Faull, R.L.M.; McKenna, P.J.; Emson, P.C. Gene expression of metabotropic glutamate receptor 5 and excitatory amino acid transporter 2 in the schizophrenic hippocampus. Mol. Brain Res. 2000, 85, 24–31. [Google Scholar] [CrossRef]
- Richardson-Burns, S.M.; Haroutunian, V.; Davis, K.L.; Watson, S.J.; Meador-Woodruff, J.H. Metabotropic glutamate receptor mRNA expression in the schizophrenic thalamus. Biol. Psychiatry 2000, 47, 22–28. [Google Scholar] [CrossRef]
- Gupta, D.S.; McCullumsmith, R.E.; Beneyto, M.; Haroutunian, V.; Davis, K.L.; Meador-Woodruff, J.H. Metabotropic glutamate receptor protein expression in the prefrontal cortex and striatum in schizophrenia. Synapse 2005, 57, 123–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corti, C.; Xuereb, J.H.; Crepaldi, L.; Corsi, M.; Michielin, F.; Ferraguti, F. Altered levels of glutamatergic receptors and Na+/K+ ATPase-α1 in the prefrontal cortex of subjects with schizophrenia. Schizophr. Res. 2011, 128, 7–14. [Google Scholar] [CrossRef]
- Fatemi, S.H.; Folsom, T.D.; Rooney, R.J.; Thuras, P.D. MRNA and protein expression for novel GABA A receptors θ and ρ2 are altered in schizophrenia and mood disorders; Relevance to FMRP-mGluR5 signaling pathway. Transl. Psychiatry 2013, 3, e271. [Google Scholar] [CrossRef] [PubMed]
- Matosin, N.; Fernandez-Enright, F.; Fung, S.J.; Lum, J.S.; Engel, M.; Andrews, J.L.; Huang, X.F.; Weickert, C.S.; Newell, K.A. Alterations of mGluR5 and its endogenous regulators Norbin, Tamalin and Preso1 in schizophrenia: Towards a model of mGluR5 dysregulation. Acta Neuropathol. 2015, 130, 119–129. [Google Scholar] [CrossRef] [Green Version]
- Volk, D.W.; Eggan, S.M.; Lewis, D.A. Alterations in metabotropic glutamate receptor 1α and regulator of G protein signaling 4 in the prefrontal cortex in schizophrenia. Am. J. Psychiatry 2010, 167, 1489–1498. [Google Scholar] [CrossRef] [Green Version]
- Parkin, G.M.; Gibbons, A.; Udawela, M.; Dean, B. Excitatory amino acid transporter (EAAT)1 and EAAT2 mRNA levels are altered in the prefrontal cortex of subjects with schizophrenia. J. Psychiatr. Res. 2020, 123, 151–158. [Google Scholar] [CrossRef]
- McOmish, C.E.; Pavey, G.; Gibbons, A.; Hopper, S.; Udawela, M.; Scarr, E.; Dean, B. Lower [3H]LY341495 binding to mGlu2/3 receptors in the anterior cingulate of subjects with major depressive disorder but not bipolar disorder or schizophrenia. J. Affect. Disord. 2016, 190, 241–248. [Google Scholar] [CrossRef]
- Frank, E.; Newell, K.A.; Huang, X.-F. Density of metabotropic glutamate receptors 2 and 3 (mGluR2/3) in the dorsolateral prefrontal cortex does not differ with schizophrenia diagnosis but decreases with age. Schizophr. Res. 2011, 128, 56–60. [Google Scholar] [CrossRef] [Green Version]
- Crook, J.M.; Akil, M.; Law, B.C.W.; Hyde, T.M.; Kleinman, J.E. Comparative analysis of group II metabotropic glutamate receptor immunoreactivity in Brodmann’s area 46 of the dorsolateral prefrontal cortex from patients with schizophrenia and normal subjects. Mol. Psychiatry 2002, 7, 157–164. [Google Scholar] [CrossRef] [Green Version]
- Eastwood, S.L.; McDonald, B.; Burnet, P.W.J.; Beckwith, J.P.; Kerwin, R.W.; Harrison, P.J. Decreased expression of mRNAs encoding non-NMDA glutamate receptors GluRl and GluR2 in medial temporal lobe neurons in schizophrenia. Mol. Brain Res. 1995, 29, 211–223. [Google Scholar] [CrossRef]
- Ghose, S.; Crook, J.M.; Bartus, C.L.; Sherman, T.G.; Herman, M.M.; Hyde, T.M.; Kleinman, J.E.; Akil, M. Metabotropic glutamate receptor 2 and 3 gene expression in the human prefrontal cortex and mesencephalon in schizophrenia. Int. J. Neurosci. 2008, 118, 1609–1627. [Google Scholar] [CrossRef] [Green Version]
- Ghose, S.; Gleason, K.A.; Potts, B.W.; Lewis-Amezcua, K.; Tamminga, C.A. Differential expression of metabotropic glutamate receptors 2 and 3 in schizophrenia: A mechanism for antipsychotic drug action? Am. J. Psychiatry 2009, 166, 812–820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schousboe, A. Transport and metabolism of glutamate and gaba in neurons and glial cells. Int. Rev. Neurobiol. 1981, 22, 1–45. [Google Scholar] [PubMed]
- Conn, P.J.; Pin, J.P. Pharmacology and functions of metabotropic glutamate receptors. Annu. Rev. Pharmacol. Toxicol. 1997, 37, 205–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwartz, T.L.; Sachdeva, S.; Stahl, S.M. Glutamate neurocircuitry: Theoretical underpinnings in: Schizophrenia. Front. Pharmacol. 2012, 3, 195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carlsson, A.; Waters, N.; Waters, S.; Carlsson, M.L. Network interactions in schizophrenia - Therapeutic implications. Brain Res. Rev. 2000, 31, 342–349. [Google Scholar] [CrossRef]
- Kehrer, C.; Maziashvili, N.; Dugladze, T.; Gloveli, T. Altered excitatory-inhibitory balance in the NMDA-hypofunction model of schizophrenia. Front. Mol. Neurosci. 2008, 1, 6. [Google Scholar] [CrossRef] [Green Version]
- Sohal, V.S.; Zhang, F.; Yizhar, O.; Deisseroth, K. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 2009, 459, 698–702. [Google Scholar] [CrossRef] [Green Version]
- Cartmell, J.; Schoepp, D.D. Regulation of neurotransmitter release by metabotropic glutamate receptors. J. Neurochem. 2000, 75, 889–907. [Google Scholar] [CrossRef]
- Schoepp, D.D.; Jane, D.E.; Monn, J.A. Pharmacological agents acting at subtypes of metabotropic glutamate receptors. Neuropharmacology 1999, 38, 1431–1476. [Google Scholar] [CrossRef]
- Moussawi, K.; Riegel, A.; Nair, S.; Kalivas, P.W.; Bargas, J.; Nacional, U. Extracellular glutamate: Functional compartments operate in different concentration ranges. Front. Syst. Neurosci. 2011, 5, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pin, J.-P.; Duvoisin, R. The metabotropic glutamate receptors: Structure and functions. Neuropharmacology 1995, 34, 1–26. [Google Scholar] [CrossRef]
- Neki, A.; Ohishi, H.; Kaneko, T.; Shigemoto, R.; Nakanishi, S.; Mizuno, N. Pre- and postsynaptic localization of a metabotropic glutamate receptor, mGluR2, in the rat brain: An immunohistochemical study with a monoclonal antibody. Neurosci. Lett. 1996, 202, 197–200. [Google Scholar] [CrossRef]
- Weinberger, D.R. Schizophrenia drug says goodbye to dopamine. Nat. Med. 2007, 13, 1018–1019. [Google Scholar] [CrossRef] [PubMed]
- Connell, P.H. Amphetamine Psychosis. Maudsley Monographs, No. 5.; Oxford University Press: London, UK, 1958. [Google Scholar]
- Kinon, B.J.; Millen, B.A.; Zhang, L.; McKinzie, D.L. Exploratory Analysis for a Targeted Patient Population Responsive to the Metabotropic Glutamate 2/3 Receptor Agonist Pomaglumetad Methionil in Schizophrenia. Biol. Psychiatry 2015, 78, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Nisenbaum, L.K.; Downing, A.C.M.; Zhao, F.; Millen, B.A.; Munsie, L.; Kinon, B.J.; Adams, D.H.; Gomez, J.C.; Penny, M.A. Serotonin 2A Receptor SNP rs7330461 association with treatment response to pomaglumetad methionil in patients with schizophrenia. J. Pers. Med. 2016, 6, 9. [Google Scholar] [CrossRef] [Green Version]
- Bueno, A.B.; Collado, I.; De Dios, A.; Domínguez, C.; Martín, J.A.; Martín, L.M.; Martínez-Grau, M.A.; Montero, C.; Pedregal, C.; Catlow, J.; et al. Dipeptides as effective prodrugs of the unnatural amino acid (+)-2-aminobicyclo[3.1.0]hexane-2,6-dicarboxylic acid (LY354740), a selective group II metabotropic glutamate receptor agonist. J. Med. Chem. 2005, 48, 5305–5320. [Google Scholar] [CrossRef]
- Urabe, H.; Miyakoshi, N.; Ohtake, N.; Nozoe, A.; Ochi, M.; Fukasawa, M.; Kinoshita, K.; Yamaguchi, J.; Marumo, T.; Hikichi, H.; et al. Discovery of MGS0274, an ester prodrug of a metabotropic glutamate receptor 2/3 agonist with improved oral bioavailability. Eur. J. Med. Chem. 2020, 203, 112521. [Google Scholar] [CrossRef]
- Annes, W.F.; Long, A.; Witcher, J.W.; Ayan-Oshodi, M.A.; Knadler, M.P.; Zhang, W.; Mitchell, M.I.; Cornelissen, K.; Hall, S.D. Relative contributions of presystemic and systemic peptidases to oral exposure of a novel metabotropic glutamate 2/3 receptor agonist (LY404039) after oral administration of prodrug Pomaglumetad methionil (LY2140023). J. Pharm. Sci. 2015, 104, 207–214. [Google Scholar] [CrossRef]
- Mezler, M.; Geneste, H.; Gault, L.; Marek, G.J. LY-2140023, a prodrug of the group II metabotropic glutamate receptor agonist LY-404039 for the potential treatment of schizophrenia. Curr. Opin. Investig. Drugs 2010, 11, 833–845. [Google Scholar] [PubMed]
- Kinoshita, K.; Ochi, M.; Iwata, K.; Fukasawa, M.; Yamaguchi, J. ichi Preclinical disposition of MGS0274 besylate, a prodrug of a potent group II metabotropic glutamate receptor agonist MGS0008 for the treatment of schizophrenia. Pharmacol. Res. Perspect. 2019, 7, e00520. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, M.; Marcy, B.; Kinoshita, K.; Fukasawa, M.; Hikichi, H.; Chaki, S.; Okuyama, S.; Gevorkyan, H.; Yoshida, S. Safety and pharmacokinetic profiles of MGS0274 besylate (TS-134), a novel metabotropic glutamate 2/3 receptor agonist prodrug, in healthy subjects. Br. J. Clin. Pharmacol. 2020, 86, 2286–2301. [Google Scholar] [CrossRef] [PubMed]
- Bocchio, M.; Lukacs, I.P.; Stacey, R.; Plaha, P.; Apostolopoulos, V.; Livermore, L.; Sen, A.; Ansorge, O.; Gillies, M.J.; Somogyi, P.; et al. Group II metabotropic glutamate receptors mediate presynaptic inhibition of excitatory transmission in pyramidal neurons of the human cerebral cortex. Front. Cell. Neurosci. 2019, 12, 508. [Google Scholar] [CrossRef] [PubMed]
- Gereau IV, R.W.; Conn, P.J. Multiple presynaptic metabotropic glutamate receptors modulate excitatory and inhibitory synaptic transmission in hippocampal area CA1. J. Neurosci. 1995, 15, 6879–6889. [Google Scholar] [CrossRef]
- Jones, P.J.; Xiang, Z.; Conn, P.J. Metabotropic glutamate receptors mGluR4 and mGluR8 regulate transmission in the lateral olfactory tract-piriform cortex synapse. Neuropharmacology 2008, 55, 440–446. [Google Scholar] [CrossRef] [Green Version]
- Thomsen, C.; Pekhletski, R.; Haldeman, B.; Gilbert, T.A.; O’Hara, P.; Hampson, D.R. Cloning and characterization of a metabotropic glutamate receptor, mGluR4b. Neuropharmacology 1997, 36, 21–30. [Google Scholar] [CrossRef]
- Woźniak, M.; Gołembiowska, K.; Noworyta-Sokołowska, K.; Acher, F.; Cieślik, P.; Kusek, M.; Tokarski, K.; Pilc, A.; Wierońska, J.M. Neurochemical and behavioral studies on the 5-HT1A-dependent antipsychotic action of the mGlu4 receptor agonist LSP4-2022. Neuropharmacology 2017, 115, 149–165. [Google Scholar] [CrossRef]
- Sławińska, A.; Wierońska, J.M.; Stachowicz, K.; Marciniak, M.; Łasoń-Tyburkiewicz, M.; Gruca, P.; Papp, M.; Kusek, M.; Tokarski, K.; Doller, D.; et al. The antipsychotic-like effects of positive allosteric modulators of metabotropic glutamate mGlu4 receptors in rodents. Br. J. Pharmacol. 2013, 169, 1824–1839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klodzinska, A.; Bijak, M.; Tokarski, K.; Pilc, A. Group II mGlu receptor agonists inhibit behavioural and electrophysiological effects of DOI in mice. Pharmacol. Biochem. Behav. 2002, 73, 327–332. [Google Scholar] [CrossRef]
- Schulz, H.L.; Stohr, H.; Weber, B.H.F. Characterization of three novel isoforms of the metabotrobic glutamate receptor 7 (GRM7). Neurosci. Lett. 2002, 326, 37–40. [Google Scholar] [CrossRef]
- Malherbe, P.; Kratzeisen, C.; Lundstrom, K.; Richards, J.G.; Faull, R.L.M.; Mutel, V. Cloning and functional expression of alternative spliced variants of the human metabotropic glutamate receptor 8. Mol. Brain Res. 1999, 67, 201–210. [Google Scholar] [CrossRef]
- Wierońska, J.M.; Stachowicz, K.; Acher, F.; Lech, T.; Pilc, A. Opposing efficacy of group III mGlu receptor activators, LSP1-2111 and AMN082, in animal models of positive symptoms of schizophrenia. Psychopharmacology 2012, 220, 481–494. [Google Scholar]
- Cieślik, P.; Woźniak, M.; Kaczorowska, K.; Brański, P.; Burnat, G.; Chocyk, A.; Bobula, B.; Gruca, P.; Litwa, E.; Pałucha-Poniewiera, A.; et al. Negative Allosteric Modulators of mGlu7 Receptor as Putative Antipsychotic Drugs. Front. Mol. Neurosci. 2018, 11, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kinoshita, A.; Shigemoto, R.; Ohishi, H.; Mizuno, N. Immunohistochemical Localization of Metabotropic Glutamate Receptors, mGluR7a and mGluR7b, in the Central Nervous System of the Adult Rat and Mouse: A Light and Electron Microscopic Study. J. Comp. Neurol. 1998, 352, 332–352. [Google Scholar] [CrossRef]
- Wierońska, J.M.; Kusek, M.; Tokarski, K.; Wabno, J.; Froestl, W.; Pilc, A. The GABA B receptor agonist CGP44532 and the positive modulator GS39783 reverse some behavioural changes related to positive syndromes of psychosis in mice. Br. J. Pharmacol. 2011, 163, 1034–1047. [Google Scholar] [CrossRef] [Green Version]
- Wierońska, J.M.; Kłeczek, N.; Woźniak, M.; Gruca, P.; Łasoń-Tyburkiewicz, M.; Papp, M.; Brański, P.; Burnat, G.; Pilc, A. mGlu5-GABAB interplay in animal models of positive, negative and cognitive symptoms of schizophrenia. Neurochem. Int. 2015, 88, 97–109. [Google Scholar] [CrossRef]
- May, C.R. Baclofen overdose. Ann. Emerg. Med. 1983, 12, 171–173. [Google Scholar] [CrossRef]
- Garabedian-Ruffalo, S.M.; Ruffalo, R.L. Adverse effects secondary to baclofen withdrawal. Drug Intell. Clin. Pharm. 1985, 19, 304–306. [Google Scholar] [CrossRef]
- Shekhar, A.; Potter, W.Z.; Lightfoot, J.; Lienemann, J.; Dubé, S.; Mallinckrodt, C.; Bymaster, F.P.; McKinzie, D.L.; Felder, C.C. Selective muscarinic receptor agonist xanomeline as a novel treatment approach for schizophrenia. Am. J. Psychiatry 2008, 165, 1033–1039. [Google Scholar] [CrossRef]
- Mirza, N.R.; Peters, D.; Sparks, R.G. Xanomeline and the antipsychotic potential of muscarinic receptor subtype selective agonists. CNS Drug Rev. 2003, 9, 159–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melancon, B.J.; Tarr, J.C.; Panarese, J.D.; Wood, M.R.; Lindsley, C.W. Allosteric modulation of the M1 muscarinic acetylcholine receptor: Improving cognition and a potential treatment for schizophrenia and Alzheimer’s disease. Drug Discov. Today 2013, 18, 1185–1199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pancani, T.; Foster, D.J.; Moehle, M.S.; Bichell, T.J.; Bradley, E.; Bridges, T.M.; Klar, R.; Poslusney, M.; Rook, J.M.; Daniels, J.S.; et al. Allosteric activation of M4 muscarinic receptors improve behavioral and physiological alterations in early symptomatic YAC128 mice. Proc. Natl. Acad. Sci. USA 2015, 112, 14078–14083. [Google Scholar] [CrossRef] [Green Version]
- Rouse, S.T.; Marino, M.J.; Potter, L.T.; Conn, P.J.; Levey, A.I. Muscarinic receptor subtypes involved in hippocampal circuits. Life Sci. 1999, 64, 501–509. [Google Scholar] [CrossRef]
- Klawonn, A.M.; Wilhelms, D.B.; Lindström, S.H.; Singh, A.K.; Jaarola, M.; Wess, J.; Fritz, M.; Engblom, D. Muscarinic M4 receptors on cholinergic and dopamine D1 receptor-expressing neurons have opposing functionality for positive reinforcement and influence impulsivity. Front. Mol. Neurosci. 2018, 11, 1–11. [Google Scholar] [CrossRef]
- Thorn, C.A.; Popiolek, M.; Stark, E.; Edgerton, J.R. Effects of M1 and M4 activation on excitatory synaptic transmission in CA1. Hippocampus 2017, 27, 794–810. [Google Scholar] [CrossRef]
- Dasari, S.; Gulledge, A.T. M1 and M4 Receptors Modulate Hippocampal Pyramidal Neurons. J. Neurophysiol. 2011, 105, 779–792. [Google Scholar] [CrossRef] [Green Version]
- Radnikow, G.; Feldmeyer, D. Layer- and cell type-specific modulation of excitatory neuronal activity in the neocortex. Front. Neuroanat. 2018, 12, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Yang, D.; Günter, R.; Qi, G.; Radnikow, G.; Feldmeyer, D. Muscarinic and Nicotinic Modulation of Neocortical Layer 6A Synaptic Microcircuits Is Cooperative and Cell-Specific. Cereb. Cortex 2020, 30, 3528–3542. [Google Scholar] [CrossRef]
- Amar, M.; Lucas-Meunier, E.; Baux, G.; Fossier, P. Blockade of different muscarinic receptor subtypes changes the equilibrium between excitation and inhibition in rat visual cortex. Neuroscience 2010, 169, 1610–1620. [Google Scholar] [CrossRef]
- Cieślik, P.; Woźniak, M.; Rook, J.M.; Tantawy, M.N.; Conn, P.J.; Acher, F.; Tokarski, K.; Kusek, M.; Pilc, A.; Wierońska, J.M. Mutual activation of glutamatergic mGlu4 and muscarinic M4 receptors reverses schizophrenia-related changes in rodents. Psychopharmacology 2018, 235, 2897–2913. [Google Scholar]
- Malherbe, P.; Kew, J.N.C.; Richards, J.G.; Knoflach, F.; Kratzeisen, C.; Zenner, M.T.; Faull, R.L.M.; Kemp, J.A.; Mutel, V. Identification and characterization of a novel splice variant of the metabotropic glutamate receptor 5 gene in human hippocampus and cerebellum. Mol. Brain Res. 2002, 109, 168–178. [Google Scholar] [CrossRef]
- Tu, J.C.; Xiao, B.; Naisbitt, S.; Yuan, J.P.; Petralia, R.S.; Brakeman, P.; Doan, A.; Aakalu, V.K.; Lanahan, A.A.; Sheng, M.; et al. Coupling of mGluR/Homer and PSD-95 complexes by the Shank family of postsynaptic density proteins. Neuron 1999, 23, 583–592. [Google Scholar] [CrossRef] [Green Version]
- Doherty, A.J.; Palmer, M.J.; Henley, J.M.; Collingridge, G.L.; Jane, D.E. (RS)-2-chloro-5-hydroxyphenylglycine (CHPG) activates mGlu5, but not mGlu1, receptors expressed in CHO cells and potentiates NMDA responses in the hippocampus. Neuropharmacology 1997, 36, 265–267. [Google Scholar] [CrossRef]
- Attucci, S.; Carlá, V.; Mannaioni, G.; Moroni, F. Activation of type 5 metabotropic glutamate receptors enhances NMDA responses in mice cortical wedges. Br. J. Pharmacol. 2001, 132, 799–806. [Google Scholar] [CrossRef] [Green Version]
- Mannaioni, G.; Marino, M.J.; Valenti, O.; Traynelis, S.F.; Conn, P.J. Metabotropic glutamate receptors 1 and 5 differentially regulate CA1 pyramidal cell function. J. Neurosci. 2001, 21, 5925–5934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rook, J.M.; Xiang, Z.; Lv, X.; Ghoshal, A.; Dickerson, J.W.; Bridges, T.M.; Johnson, K.A.; Foster, D.J.; Gregory, K.J.; Vinson, P.N.; et al. Biased mGlu5-Positive Allosteric Modulators Provide InVivo Efficacy without Potentiating mGlu5 Modulation of NMDAR Currents. Neuron 2015, 86, 1029–1040. [Google Scholar] [CrossRef] [Green Version]
- Romano, C.; Sesma, M.A.; McDonald, C.T.; O’malley, K.; van den Pol, A.N.; Olney, J.W. Distribution of metabotropic glutamate receptor mGluR5 immunoreactivity in rat brain. J. Comp. Neurol. 1995, 355, 455–469. [Google Scholar] [CrossRef]
- Bragina, L.; Bonifacino, T.; Bassi, S.; Milanese, M.; Bonanno, G.; Conti, F. Differential expression of metabotropic glutamate and GABA receptors at neocortical glutamatergic and GABAergic axon terminals. Front. Cell. Neurosci. 2015, 9, 345. [Google Scholar] [CrossRef] [Green Version]
- Levey, A.I.; Edmunds, S.M.; Koliatsos, V.; Wiley, R.G.; Heilman, C.J. Expression of m1-m4 muscarinic acetylcholine receptor proteins in rat hippocampus and regulation by cholinergic innervation. J. Neurosci. Off. J. Soc. Neurosci. 1995, 15, 4077–4092. [Google Scholar] [CrossRef]
- Levey, A.I.; Kitt, C.A.; Simonds, W.F.; Price, D.L.; Brann, M.R. Identification and localization of muscarinic acetylcholine receptor proteins in brain with subtype-specific antibodies. J. Neurosci. Off. J. Soc. Neurosci. 1991, 11, 3218–3226. [Google Scholar] [CrossRef] [Green Version]
- Wei, J.; Walton, E.A.; Milici, A.; Buccafusco, J.J. m1-m5 Muscarinic Receptor Distribution in Rat CNS by RT-PCR and HPLC. J. Neurochem. 1994, 63, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, R.P.; Ciesla, W.; Flores, L.R.; Wall, S.J.; Li, M.; Satkus, S.A.; Weisstein, J.S.; Spagnola, B.V.; Wolfe, B.B. Development of antisera selective for m4 and m5 muscarinic cholinergic receptors: Distribution of m4 and m5 receptors in rat brain. Mol. Pharmacol. 1993, 43, 149–157. [Google Scholar]
- Hamilton, S.E.; Nathanson, N.M. The M1 Receptor is Required for Muscarinic Activation of Mitogen-activated Protein (MAP) Kinase in Murine Cerebral Cortical Neurons. J. Biol. Chem. 2001, 276, 15850–15853. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anagnostaras, S.G.; Murphy, G.G.; Hamilton, S.E.; Mitchell, S.L.; Rahnama, N.P.; Nathanson, N.M.; Silva, A.J. Selective cognitive dysfunction in acetylcholine M1 muscarinic receptor mutant mice. Nat. Neurosci. 2003, 6, 51–58. [Google Scholar] [CrossRef]
- Miyakawa, T.; Yamada, M.; Duttaroy, A.; Wess, J. Hyperactivity and intact hippocampus-dependent learning in mice lacking the M1 muscarinic acetylcholine receptor. J. Neurosci. 2001, 21, 5239–5250. [Google Scholar] [CrossRef] [Green Version]
- Gerber, D.J.; Sotnikova, T.D.; Gainetdinov, R.R.; Huang, S.Y.; Caron, M.G.; Tonegawa, S. Hyperactivity, elevated dopaminergic transmission, and response to amphetamine in M1 muscarinic acetylcholine receptor-deficient mice. Proc. Natl. Acad. Sci. USA 2001, 98, 15312–15317. [Google Scholar] [CrossRef] [Green Version]
- Cieślik, P.; Woźniak, M.; Tokarski, K.; Kusek, M.; Pilc, A.; Płoska, A.; Radulska, A.; Pelikant-Małecka, I.; Żołnowska, B.; Sławiński, J.; et al. Simultaneous activation of muscarinic and GABA B receptors as a bidirectional target for novel antipsychotics. Behav. Brain Res. 2019, 359, 671–685. [Google Scholar] [CrossRef]
- Cieślik, P.; Domin, H.; Chocyk, A.; Gruca, P.; Litwa, E.; Płoska, A.; Radulska, A.; Pelikant-Małecka, I.; Brański, P.; Kalinowski, L.; et al. Simultaneous activation of mGlu2 and muscarinic receptors reverses MK-801-induced cognitive decline in rodents. Neuropharmacology 2020, 174, 107866. [Google Scholar] [CrossRef]
- Weiner, D.M.; Levey, A.I.; Brann, M.R. Expression of muscarinic acetylcholine and dopamine receptor mRNAs in rat basal ganglia. Proc. Natl. Acad. Sci. USA 1990, 87, 7050–7054. [Google Scholar] [CrossRef] [Green Version]
- Araya, R.; Noguchi, T.; Yuhki, M.; Kitamura, N.; Higuchi, M.; Saido, T.C.; Seki, K.; Itohara, S.; Kawano, M.; Tanemura, K.; et al. Loss of M5 muscarinic acetylcholine receptors leads to cerebrovascular and neuronal abnormalities and cognitive deficits in mice. Neurobiol. Dis. 2006, 24, 334–344. [Google Scholar] [CrossRef] [PubMed]
- Bymaster, F.P.; McKinzie, D.L.; Felder, C.C.; Wess, J. M1-M5 muscarinic receptor knockout mice as novel tools to study the physiological roles of the muscarinic cholinergic system. Neurochem. Res. 2003, 28, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Foster, D.J.; Gentry, P.R.; Lizardi-Ortiz, J.E.; Bridges, T.M.; Wood, M.R.; Niswender, C.M.; Sulzer, D.; Lindsley, C.W.; Xiang, Z.; Conn, P.J. M5 Receptor Activation Produces Opposing Physiological Outcomes in Dopamine Neurons Depending on the Receptor’s Location. J. Neurosci. 2014, 34, 3253–3262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamada, M.; Lamping, K.G.; Duttaroy, A.; Zhang, W.; Cui, Y.; Bymaster, F.P.; McKinzie, D.L.; Felder, C.C.; Deng, C.X.; Faraci, F.M.; et al. Cholinergic dilation of cerebral blood vessels is abolished in M5 muscarinic acetylcholine receptor knockout mice. Proc. Natl. Acad. Sci. USA 2001, 98, 14096–14101. [Google Scholar] [CrossRef] [Green Version]
- Thomsen, M.; Wörtwein, G.; Fink-Jensen, A.; Woldbye, D.P.D.; Wess, J.; Caine, S.B. Decreased prepulse inhibition and increased sensitivity to muscarinic, but not dopaminergic drugs in M5 muscarinic acetylcholine receptor knockout mice. Psychopharmacology 2007, 192, 97–110. [Google Scholar] [CrossRef]
- Rouse, S.T.; Levey, A.I. Expression of m1-m4 muscarinic acetylcholine receptor immunoreactivity in septohippocampal neurons and other identified hippocampal afferents. J. Comp. Neurol. 1996, 375, 406–416. [Google Scholar] [CrossRef]
- Oda, S.; Tsuneoka, Y.; Yoshida, S.; Adachi-Akahane, S.; Ito, M.; Kuroda, M.; Funato, H. Immunolocalization of muscarinic M1 receptor in the rat medial prefrontal cortex. J. Comp. Neurol. 2018, 526, 1329–1350. [Google Scholar] [CrossRef] [Green Version]
- Vilaró, M.T.; Wiederhold, K.H.; Palacios, J.M.; Mengod, G. Muscarinic cholinergic receptors in the rat caudate-putamen and olfactory tubercle belong predominantly to the m4 class: In situ hybridization and receptor autoradiography evidence. Neuroscience 1991, 159–167. [Google Scholar] [CrossRef]
- Roßner, S.; Kues, W.; Witzemann, V.; Schliebs, R. Laminar expression of m1-, m3- and m4-muscarinic cholinergic receptor genes in the developing rat visual cortex using in situ hybridization histochemistry. Effect of monocular visual deprivation. Int. J. Dev. Neurosci. 1993, 11, 369–378. [Google Scholar]
- Schliebs, R.; Rossner, S.; Kumar, A.; Bigl, V. Muscarinic acetylcholine receptor subtypes in rat visual cortex—A comparative study using quantitative receptor autoradiography and in situ hybridization. Indian J. Exp. Biol. 1994, 32, 25–30. [Google Scholar]
- Buckley, N.J.; Bonner, T.I.; Brann, M.R. Localization of a family of muscarinic receptor mRNAs in rat brain. J. Neurosci. 1988, 8, 4646–4652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brann, M.R.; Buckley, N.J.; Bonner, T.I. The striatum and cerebral cortex express different muscarinic receptor mRNAs. FEBS Lett. 1988, 230, 90–94. [Google Scholar] [CrossRef] [Green Version]
- Hersch, S.M.; Gutekunst, C.A.; Rees, H.D.; Heilman, C.J.; Levey, A.I. Distribution of m1-m4 muscarinic receptor proteins in the rat striatum: Light and electron microscopic immunocytochemistry using subtype-specific antibodies. J. Neurosci. 1994, 14, 3351–3363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomeza, J.; Zhang, L.; Kostenis, E.; Felder, C.; Bymaster, F.; Brodkin, J.; Shannon, H.; Xia, B.; Deng, C.; Wess, J. Enhancement of D1 dopamine receptor-mediated locomotor stimulation in M4 muscarinic acetylcholine receptor knockout mice. Proc. Natl. Acad. Sci. USA 1999, 96, 10483–10488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vilaró, M.T.; Palacios, J.M.; Mengod, G. Localization of m5 muscarinic receptor mRNA in rat brain examined by in situ hybridization histochemistry. Neurosci. Lett. 1990, 114, 154–159. [Google Scholar] [CrossRef]
- Benke, D.; Honer, M.; Michel, C.; Bettler, B.; Mohler, H. γ-Aminobutyric acid type B receptor splice variant proteins GBR1a and GBR1b are both associated with GBR2 in situ and display differential regional and subcellular distribution. J. Biol. Chem. 1999, 274, 27323–27330. [Google Scholar] [CrossRef] [Green Version]
- Charles, K.J.; Evans, M.L.; Robbins, M.J.; Calver, A.R.; Leslie, R.A.; Pangalos, M.N. Comparative immunohistochemical localisation of GABAB1a, GABAB1b and GABAB2 subunits in rat brain, spinal cord and dorsal root ganglion. Neuroscience 2001, 106, 447–467. [Google Scholar] [CrossRef]
- Lu, X.Y.; Ghasemzadeh, M.B.; Kalivas, P.W. Regional distribution and cellular localization of γ-aminobutyric acid subtype 1 receptor mRNA in the rat brain. J. Comp. Neurol. 1999, 407, 166–182. [Google Scholar] [CrossRef]
- Durkin, M.M.; Gunwaldsen, C.A.; Borowsky, B.; Jones, K.A.; Branchek, T.A. An in situ hybridization study of the distribution of the GABA(B2) protein mRNA in the rat CNS. Mol. Brain Res. 1999, 71, 185–200. [Google Scholar] [CrossRef]
- Clark, J.A.; Mezey, É.; Lam, A.S.; Bonner, T.I. Distribution of the GABAB receptor subunit gb2 in rat CNS. Brain Res. 2000, 860, 41–52. [Google Scholar] [CrossRef] [Green Version]
- Calver, A.R.; Medhurst, A.D.; Robbins, M.J.; Charles, K.J.; Evans, M.L.; Harrison, D.C.; Stammers, M.; Hughes, S.A.; Hervieu, G.; Couve, A.; et al. The expression of GABA(B1) and GABA(B2) receptor subunits in the CNS differs from that in peripheral tissues. Neuroscience 2000, 100, 155–170. [Google Scholar] [CrossRef]
- Shigemoto, R.; Nomura, S.; Ohishi, H.; Sugihara, H.; Nakanishi, S.; Mizuno, N. Immunohistochemical localization of a metabotropic glutamate receptor, mGluR5, in the rat brain. Neurosci. Lett. 1993, 163, 53–57. [Google Scholar] [CrossRef]
- Fotuhi, M.; Standaert, D.G.; Testa, C.M.; Penney, J.B.; Young, A.B. Differential expression of metabotropic glutamate receptors in the hippocampus and entorhinal cortex of the rat. Mol. Brain Res. 1994, 21, 283–292. [Google Scholar] [CrossRef] [Green Version]
- Kerner, J.A.; Standaert, D.G.; Penney, J.B.; Young, A.B.; Landwehrmeyer, G.B. Expression of group one metabotropic glutamate receptor subunit mRNAs in neurochemically identified neurons in the rat neostriatum, neocortex, and hippocampus. Mol. Brain Res. 1997, 48, 259–269. [Google Scholar] [CrossRef]
- Abe, T.; Sugihara, H.; Nawa, H.; Shigemoto, R.; Mizuno, N.; Nakanishi, S. Molecular characterization of a novel metabotropic glutamate receptor mGluR5 coupled to inositol phosphate/Ca2+ signal transduction. J. Biol. Chem. 1992, 267, 13361–13368. [Google Scholar]
- Testa, C.M.; Standaert, D.G.; Young, A.B.; Penney, J.B. Metabotropic glutamate receptor mRNA expression in the basal ganglia of the rat. J. Neurosci. 1994, 14, 3005–3018. [Google Scholar] [CrossRef] [Green Version]
- Mutel, V.; Ellis, G.J.; Adam, G.; Chaboz, S.; Nilly, A.; Messer, J.; Bleuel, Z.; Metzler, V.; Malherbe, P.; Schlaeger, E.J.; et al. Characterization of [3H]quisqualate binding to recombinant rat metabotropic glutamate 1a and 5a receptors and to rat and human brain sections. J. Neurochem. 2000, 75, 2590–2601. [Google Scholar] [CrossRef] [Green Version]
- Tallaksen-Greene, S.J.; Kaatz, K.W.; Romano, C.; Albin, R.L. Localization of mGluR1a-like immunoreactivity and mGluR5-like immunoreactivity in identified populations of striatal neurons. Brain Res. 1998, 780, 210–217. [Google Scholar] [CrossRef]
- Śmiałowska, M.; Wierońska, J.M.; Brański, P.; Obuchowicz, E.; Pilc, A. MPEP, mGLU5 receptor antagonist, regulates NPYmRNA expression in hippocampal and amygdalar neurons. Pol. J. Pharmacol. 2004, 56, 709–718. [Google Scholar]
- Testa, C.M.; Standaert, D.G.; Landwehrmeyer, G.B.; Penney, J.B.; Young, A.B. Differential expression of mGluR5 metabotropic glutamate receptor mRNA by rat striatal neurons. J. Comp. Neurol. 1995, 354, 241–252. [Google Scholar] [CrossRef]
- Shigemoto, R.; Kinoshita, A.; Wada, E.; Nomura, S.; Ohishi, H.; Takada, M.; Flor, P.J.; Neki, A.; Abe, T.; Nakanishi, S.; et al. Differential presynaptic localization of metabotropic glutamate receptor subtypes in the rat hippocampus. J. Neurosci. 1997, 17, 7503–7522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luján, R.; Nusser, Z.; Roberts, J.D.B.; Shigemoto, R.; Somogyi, P. Perisynaptic location of metabotropic glutamate receptors mGluR1 and mGluR5 on dendrites and dendritic spines in the rat hippocampus. Eur. J. Neurosci. 1996, 8, 1488–1500. [Google Scholar] [CrossRef] [PubMed]
- Ohishi, H.; Neki, A.; Mizuno, N. Distribution of a metabotropic glutamate receptor, mGluR2, in the central nervous system of the rat and mouse: An immunohistochemical study with a monoclonal antibody. Neurosci. Res. 1998, 30, 65–82. [Google Scholar] [CrossRef]
- Venkatadri, P.S.; Lee, C.C. Differential Expression of mGluR2 in the Developing Cerebral Cortex of the Mouse. J. Biomed. Sci. Eng. 2014, 7, 1030–1037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohishi, H.; Shigemoto, R.; Nakanishi, S.; Mizuno, N. Distribution of the messenger RNA for a metabotropic glutamate receptor, mGluR2, in the central nervous system of the rat. Neuroscience 1993, 53, 1009–1018. [Google Scholar] [CrossRef]
- McOmish, C.E.; Demireva, E.Y.; Gingrich, J.A. Developmental expression of mGlu2 and mGlu3 in the mouse brain. Gene Expr. Patterns 2016, 22, 46–53. [Google Scholar] [CrossRef] [Green Version]
- Berthele, A.; Platzer, S.; Laurie, D.J.; Weis, S.; Sommer, B.; Zieglgänsberger, W.; Conrad, B.; Tölle, T.R. Expression of metabotropic glutamate receptor subtype mRNA (mGluR1-8) in human cerebellum. Neuroreport 1999, 10, 3861–3867. [Google Scholar] [CrossRef]
- Corti, C.; Aldegheri, L.; Somogyi, P.; Ferraguti, F. Distribution and synaptic localisation of the metabotropic glutamate receptor 4 (mGluR4) in the rodent CNS. Neuroscience 2002, 110, 403–420. [Google Scholar] [CrossRef]
- Kristensen, P.; Suzdak, P.D.; Thomsen, C. Expression pattern and pharmacology of the rat type IV metabotropic glutamate receptor. Neurosci. Lett. 1993, 155, 159–162. [Google Scholar] [CrossRef]
- Thomsen, C.; Kristensen, P.; Mulvihill, E.; Haldeman, B.; Suzdak, P.D. L-2-amino-4-phosphonobutyrate (L-AP4) is an agonist at the type iv metabotropic glutamate receptor which is negatively coupled to adenylate cyclase. Eur. J. Pharmacol. Mol. Pharmacol. 1992, 227, 361–362. [Google Scholar] [CrossRef]
- Ohishi, H.; Akazawa, C.; Shigemoto, R.; Nakanishi, S.; Mizuno, N. Distributions of the mRNAs for L-2-amino-4-phosphonobutyrate-sensitive metabotropic glutamate receptors, mGluR4 and mGluR7, in the rat brain. J. Comp. Neurol. 1995, 360, 555–570. [Google Scholar] [CrossRef] [PubMed]
- Messenger, M.J.; Dawson, L.G.; Duty, S. Changes in metabotropic glutamate receptor 1-8 gene expression in the rodent basal ganglia motor loop following lesion of the nigrostriatal tract. Neuropharmacology 2002, 43, 261–271. [Google Scholar] [CrossRef]
- Tanabe, Y.; Nomura, A.; Masu, M.; Shigemoto, R.; Mizuno, N.; Nakanishi, S. Signal transduction, pharmacological properties, and expression patterns of two rat metabotropic glutamate receptors, mGluR3 and mGluR4. J. Neurosci. 1993, 13, 1372–1378. [Google Scholar] [CrossRef] [PubMed]
- Mateos, J.M.; Azkue, J.; Sarría, R.; Kuhn, R.; Grandes, P.; Knöpfel, T. Localization of the mGlu4a metabotropic glutamate receptor in rat cerebellar cortex. Histochem. Cell Biol. 1998, 109, 135–139. [Google Scholar] [CrossRef]
- Kinoshita, A.; Ohishi, H.; Nomura, S.; Shigemoto, R.; Nakanishi, S.; Mizuno, N. Presynaptic localization of a metabotropic glutamate receptor, mGluR4a, in the cerebellar cortex: A light and electron microscope study in the rat. Neurosci. Lett. 1996, 207, 199–202. [Google Scholar] [CrossRef]
- Wada, E.; Shigemoto, R.; Kinoshita, A.; Ohishi, H.; Mizuno, N. Metabotropic glutamate receptor subtypes in axon terminals of projection fibers from the main and accessory olfactory bulbs: A light and electron microscopic immunohistochemical study in the rat. J. Comp. Neurol. 1998, 393, 493–504. [Google Scholar] [CrossRef]
- Corti, C.; Restituito, S.; Rimland, J.M.; Brabet, I.; Corsi, M.; Pin, J.P.; Ferraguti, F. Cloning and characterization of alternative mRNA forms for the rat metabotropic glutamate receptors mGluR7 and mGluR8. Eur. J. Neurosci. 1998, 10, 3629–3641. [Google Scholar] [CrossRef]
- Okamoto, N.; Hori, S.; Akazawa, C.; Hayashi, Y.; Shigemoto, R.; Mizuno, N.; Nakanishi, S. Molecular characterization of a new metabotropic glutamate receptor mGluR7 coupled to inhibitory cyclic AMP signal transduction. J. Biol. Chem. 1994, 269, 1231–1236. [Google Scholar]
- Kinzie, J.M.; Saugstad, J.A.; Westbrook, G.L.; Segerson, T.P. Distribution of metabotropic glutamate receptor 7 messenger RNA in the developing and adult rat brain. Neuroscience 1995, 69, 167–176. [Google Scholar] [CrossRef]
- Kinzie, J.M.; Shinohara, M.M.; Van Den Pol, A.N.; Westbrook, G.L.; Segerson, T.P. Immunolocalization of metabotropic glutamate receptor 7 in the rat olfactory bulb. J. Comp. Neurol. 1997, 385, 372–384. [Google Scholar] [CrossRef]
- Bradley, S.R.; Rees, H.D.; Yi, H.; Levey, A.I.; Conn, P.J. Distribution and developmental regulation of metabotropic glutamate receptor 7a in rat brain. J. Neurochem. 1998, 71, 636–645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shigemoto, R.; Kulik, A.; Roberts, J.D.B.; Ohishi, H.; Nusser, Z.; Kaneko, T.; Somogyi, P. Target-cell-specific concentration of a metabotropic glutamate receptor in the presynaptic active zone. Nature 1996, 381, 523–525. [Google Scholar] [CrossRef]
- Saugstad, J.A.; Kinzie, J.M.; Shinohara, M.M.; Segerson, T.P.; Westbrook, G.L. Cloning and expression of rat metabotropic glutamate receptor 8 reveals a distinct pharmacological profile. Mol. Pharmacol. 1997, 51, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Robbins, M.J.; Starr, K.R.; Honey, A.; Soffin, E.M.; Rourke, C.; Jones, G.A.; Kelly, F.M.; Strum, J.; Melarange, R.A.; Harris, A.J.; et al. Evaluation of the mGlu8 receptor as a putative therapeutic target in schizophrenia. Brain Res. 2007, 1152, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Ferraguti, F.; Klausberger, T.; Cobden, P.; Baude, A.; Roberts, J.D.B.; Szucs, P.; Kinoshita, A.; Shigemoto, R.; Somogyi, P.; Dalezios, Y. Metabotropic glutamate receptor 8-expressing nerve terminals target subsets of GABAergic neurons in the hippocampus. J. Neurosci. 2005, 25, 10520–10536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duvoisin, R.M.; Zhang, C.; Ramonell, K. A novel metabotropic glutamate receptor expressed in the retina and olfactory bulb. J. Neurosci. 1995, 15, 3075–3083. [Google Scholar] [CrossRef] [Green Version]
- Flynn, D.D.; Ferrari-DiLeo, G.; Mash, D.C.; Levey, A.I. Differential Regulation of Molecular Subtypes of Muscarinic Receptors in Alzheimer’s Disease. J. Neurochem. 1995, 64, 1888–1891. [Google Scholar] [CrossRef]
- Berthele, A.; Platzer, S.; Weis, S.; Conrad, B.; Tölle, T.R. Expression of GABA(B1) and GABA(B2) mRNA in the human brain. Neuroreport 2001, 12, 3269–3275. [Google Scholar] [CrossRef]
- Billinton, A.; Ige, A.O.; Wise, A.; White, J.H.; Disney, G.H.; Marshall, F.H.; Waldvogel, H.J.; Faull, R.L.M.; Emson, P.C. GABA(B) receptor heterodimer-component localisation in human brain. Mol. Brain Res. 2000, 77, 111–124. [Google Scholar] [CrossRef]
- Blümcke, I.; Behle, K.; Malitschek, B.; Kuhn, R.; Knöpfel, T.; Wolf, H.K.; Wiestler, O.D. Immunohistochemical distribution of metabotropic glutamate receptor subtypes mGluR1b, mGluR2/3, mGluR4a and mGluR5 in human hippocampus. Brain Res. 1996, 736, 217–226. [Google Scholar] [CrossRef]
- Makoff, A.; Lelchuk, R.; Oxer, M.; Harrington, K.; Emson, P. Molecular characterization and localization of human metabotropic glutamate receptor type 4. Mol. Brain Res. 1996, 37, 239–248. [Google Scholar] [CrossRef]
- Flor, P.J.; Lukic, S.; Rüegg, D.; Leonhardt, T.; Knöpfel, T.; Kühn, R. Molecular cloning, functional expression and pharmacological characterization of the human metabotropic glutamate receptor type 4. Neuropharmacology 1995, 34, 149–155. [Google Scholar] [CrossRef]
- Flor, P.J.; Van Der Putten, H.; Rüegg, D.; Lukic, S.; Leonhardt, T.; Bence, M.; Sansig, G.; Knöpfel, T.; Kuhn, R. A novel splice variant of a metabotropic glutamate receptor, human mGluR7b. Neuropharmacology 1997, 36, 153–159. [Google Scholar] [CrossRef]
- Makoff, A.; Pilling, C.; Harrington, K.; Emson, P. Human metabotropic glutamate receptor type 7: Molecular cloning and mRNA distribution in the CNS. Mol. Brain Res. 1996, 40, 165–170. [Google Scholar] [CrossRef]
- Woźniak, M.; Acher, F.; Marciniak, M.; Lasoń-Tyburkiewicz, M.; Gruca, P.; Papp, M.; Pilc, A.; Wierońska, J.M. Involvement of GABAB Receptor Signaling in Antipsychotic-like Action of the Novel Orthosteric Agonist of the mGlu4 Receptor, LSP4-2022. Curr. Neuropharmacol. 2016, 14, 413–426. [Google Scholar] [CrossRef] [Green Version]
- Charvin, D. mGlu4 allosteric modulation for treating Parkinson’s disease. Neuropharmacology 2018, 135, 308–315. [Google Scholar] [CrossRef]
- Levey, A.I. Immunological localization of m1-m5 muscarinic acetylcholine receptors in peripheral tissues and brain. Life Sci. 1993, 52, 441–448. [Google Scholar] [CrossRef]
- Cieślik, P.; Radulska, A.; Pelikant-Małecka, I.; Płoska, A.; Kalinowski, L.; Wierońska, J.M. Reversal of MK-801-Induced Disruptions in Social Interactions and Working Memory with Simultaneous Administration of LY487379 and VU152100 in Mice. Int. J. Mol. Sci. 2019, 20, 2781. [Google Scholar] [CrossRef] [Green Version]
- Jones, K.A.; Borowsky, B.; Tamm, J.A.; Craig, D.A.; Durkin, M.M.; Dai, M.; Yao, W.J.; Johnson, M.; Gunwaldsen, C.; Huang, L.Y.; et al. GABA(B) receptors function as a heteromeric assembly of the subunits GABA(B)R1 and GABA(B)R2. Nature 1998, 396, 674–679. [Google Scholar] [CrossRef]
- Carriba, P.; Navarro, G.; Ciruela, F.; Ferré, S.; Casadó, V.; Agnati, L.; Cortés, A.; Mallol, J.; Fuxe, K.; Canela, E.I.; et al. Detection of heteromerization of more than two proteins by sequential BRET-FRET. Nat. Methods 2008, 5, 727–733. [Google Scholar] [CrossRef]
- Dupré, D.J.; Robitaille, M.; Éthier, N.; Villeneuve, L.R.; Mamarbachi, A.M.; Hébert, T.E. Seven transmembrane receptor core signaling complexes are assembled prior to plasma membrane trafficking. J. Biol. Chem. 2006, 281, 34561–34573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gandia, J.; Galino, J.; Amaral, O.B.; Soriano, A.; Lluís, C.; Franco, R.; Ciruela, F. Detection of higher-order G protein-coupled receptor oligomers by a combined BRET-BiFC technique. FEBS Lett. 2008, 582, 2979–2984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, W.; Urizar, E.; Kralikova, M.; Mobarec, J.C.; Shi, L.; Filizola, M.; Javitch, J.A. Dopamine D2 receptors form higher order oligomers at physiological expression levels. EMBO J. 2008, 27, 2293–2304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopez-Gimenez, J.F.; Canals, M.; Pediani, J.D.; Milligan, G. The α1b-adrenoceptor exists as a higher-order oligomer: Effective oligomerization is required for receptor maturation, surface delivery, and function. Mol. Pharmacol. 2007, 71, 1015–1029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navarro, G.; Carriba, P.; Gandía, J.; Ciruela, F.; Casadó, V.; Cortés, A.; Mallol, J.; Canela, E.I.; Lluis, C.; Franco, R. Detection of heteromers formed by cannabinoid CB1, dopamine D2, and adenosine A2A G-protein-coupled receptors by combining bimolecular fluorescence complementation and bioluminescence energy transfer. Sci. World J. 2008, 8, 1088–1097. [Google Scholar] [CrossRef] [Green Version]
- Vidi, P.A.; Chemel, B.R.; Hu, C.D.; Watts, V.J. Ligand-dependent oligomerization of dopamine D2 and adenosine A2A receptors in living neuronal cells? Mol. Pharmacol. 2008, 74, 544–551. [Google Scholar] [CrossRef] [Green Version]
- González-Maeso, J.; Ang, R.; Yuen, T.; Chan, P.; Weisstaub, N.V.; López-Giménez, J.F.; Zhou, M.; Okawa, Y.; Callado, L.F.; Milligan, G.; et al. Identification of a Novel Serotonin/Glutamate Receptor Complex Implicated in Psychosis. Nature 2008, 452, 93–97. [Google Scholar] [CrossRef] [Green Version]
- Shah, U.H.; González-Maeso, J. Serotonin and glutamate interactions in preclinical schizophrenia models. ACS Chem. Neurosci. 2019, 10, 3068–3077. [Google Scholar] [CrossRef]
- Hideshima, K.S.; Hojati, A.; Saunders, J.M.; On, D.M.; de la Fuente Revenga, M.; Shin, J.M.; Sánchez-González, A.; Dunn, C.M.; Pais, A.B.; Pais, A.C.; et al. Role of mGlu2 in the 5-HT 2A receptor-dependent antipsychotic activity of clozapine in mice. Psychopharmacology 2018, 235, 3149–3165. [Google Scholar] [CrossRef]
- Cabello, N.; Gandía, J.; Bertarelli, D.C.G.; Watanabe, M.; Lluís, C.; Franco, R.; Ferré, S.; Luján, R.; Ciruela, F. Metabotropic glutamate type 5, dopamine D2 and adenosine A 2a receptors form higher-order oligomers in living cells. J. Neurochem. 2009, 109, 1497–1507. [Google Scholar] [CrossRef]
- Fuxe, K.; Marcellino, D.; Leo, G.; Agnati, L.F. Molecular integration via allosteric interactions in receptor heteromers. A working hypothesis. Curr. Opin. Pharmacol. 2010, 10, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Delgado, D.M.; Møller, T.C.; Ster, J.; Giraldo, J.; Maurel, D.; Rovira, X.; Scholler, P.; Zwier, J.M.; Perroy, J.; Durroux, T.; et al. Pharmacological evidence for a metabotropic glutamate receptor heterodimer in neuronal cells. eLife 2017, 6, e25233. [Google Scholar] [CrossRef] [PubMed]
- Fulton, M.G.; Loch, M.T.; Cuoco, C.A.; Rodriguez, A.L.; Days, E.; Vinson, P.N.; Kozek, K.A.; Weaver, C.D.; Blobaum, A.L.; Conn, P.J.; et al. Challenges in the Discovery and Optimization of mGlu2/4 Heterodimer Positive Allosteric Modulators. Lett. Drug Des. Discov. 2019, 16, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, Z.; Moreno-Delgado, D.; Dalton, J.A.R.; Rovira, X.; Trapero, A.; Goudet, C.; Llebaria, A.; Giraldo, J.; Yuan, Q.; et al. Allosteric control of an asymmetric transduction in a g protein-coupled receptor heterodimer. eLife 2017, 6, e26985. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Noetzel, M.J.; Johnson, K.A.; Zamorano, R.; Jalan-Sakrikar, N.; Gregory, K.J.; Jeffrey Conn, P.; Niswender, C.M. Selective actions of novel allosteric modulators reveal functional heteromers of Metabotropic glutamate receptors in the CNS. J. Neurosci. 2014, 34, 79–94. [Google Scholar] [CrossRef]
- Colavita, M.; Terral, G.; Lemercier, C.E.; Drago, F.; Marsicano, G.; Massa, F. Layer-specific potentiation of network GABAergic inhibition in the CA1 area of the hippocampus. Sci. Rep. 2016, 6, 28454. [Google Scholar] [CrossRef] [Green Version]
- González, J.C.; Albiñana, E.; Baldelli, P.; García, A.G.; Hernández-Guijo, J.M. Presynaptic muscarinic receptor subtypes involved in the enhancement of spontaneous GABAergic postsynaptic currents in hippocampal neurons. Eur. J. Neurosci. 2011, 33, 69–81. [Google Scholar] [CrossRef]
- Yi, F.; Ball, J.; Stoll, K.E.; Satpute, V.C.; Mitchell, S.M.; Pauli, J.L.; Holloway, B.B.; Johnston, A.D.; Nathanson, N.M.; Deisseroth, K.; et al. Direct excitation of parvalbumin-positive interneurons by M1muscarinic acetylcholine receptors: Roles in cellular excitability, inhibitory transmission and cognition. J. Physiol. 2014, 592, 3463–3494. [Google Scholar] [CrossRef]
- Niswender, C.M.; Conn, P.J. Metabotropic Glutamate Receptors: Physiology, Pharmacology, and Disease. Annu. Rev. Pharmacol. Toxicol. 2010, 50, 295–322. [Google Scholar] [CrossRef] [Green Version]
- Leo, R.J.; Baer, D. Delirium associated with baclofen withdrawal: A review of common presentations and management strategies. Psychosomatics 2005, 46, 503–507. [Google Scholar] [CrossRef]
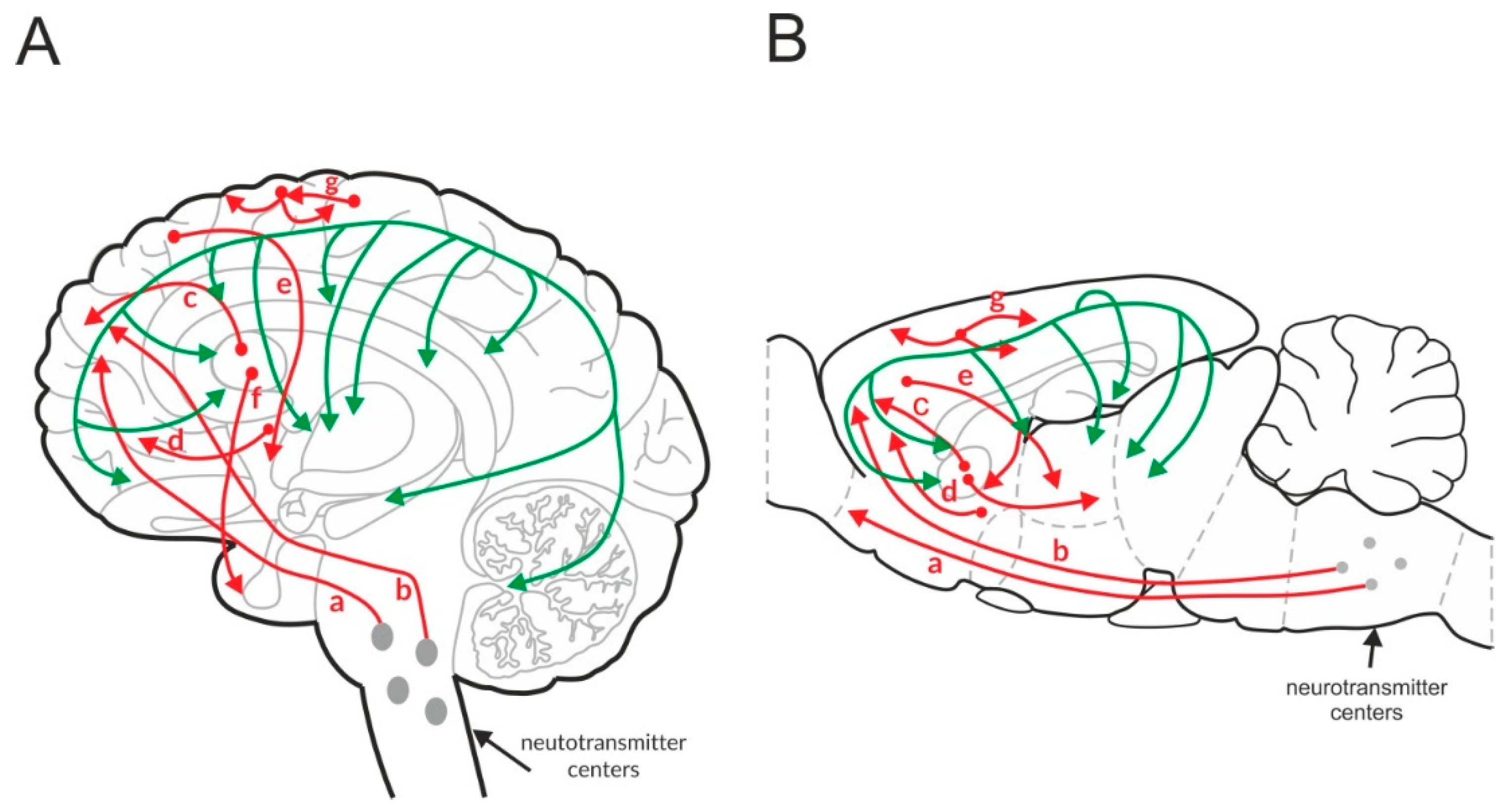










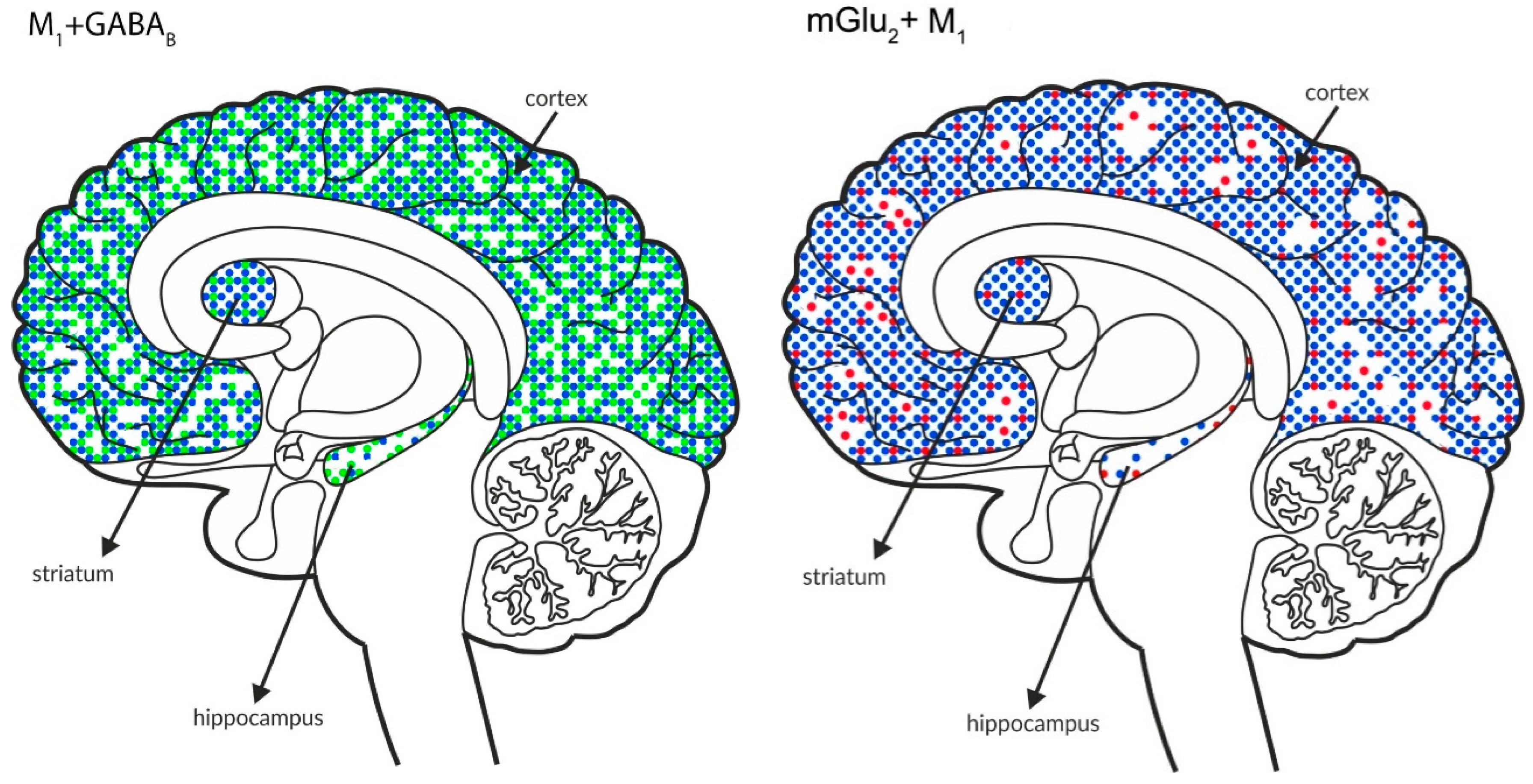

| Schizophrenic Subtype/Disorder | Positive Symptoms | Negative Symptoms | Cognitive Symptoms | Psychomotor Disturbances |
|---|---|---|---|---|
| paranoid | +++++ | − | − | − |
| disorganized | − | ++ | +++++ | − |
| catatonic | − | ++ | ++ | +++++ |
| unspecified | +++ | +++ | +++ | ++ |
| residual | − | ++++ | + | |
| Schizoaffective disorder | +++ | +++ | − | − |
| brief psychotic disorder | ++ | +++++ | − | ++ |
| Chaki et al., 2010 | [35] | mGlu2/3 |
| Lesage et al., 2010 | [36] | mGlu1 |
| Marek, 2010 | [37] | mGlu2/3 |
| Yasuhara et al., 2010 | [38] | mGlu1, mGlu2, mGlu2/3, mGlu5 |
| Chaki et al., 2011 | [39] | mGlu1, mGlu2, mGlu2/3 |
| Gregory et al., 2011 | [40] | mGlu2, mGlu5 |
| Nicoletti et al., 2011 | [41] | mGlu1, mGlu2/3, mGlu5 |
| Sheffler et al., 2011 | [42] | mGlu2, mGlu5 |
| Fell et al., 2012 | [43] | mGlu2, mGlu2/3 |
| Vinson et al., 2012 | [44] | mGlu2, mGlu3, mGlu5 |
| Gregory et al., 2013 | [45] | mGlu2/3, mGlu5 |
| Nickols et al., 2014 | [46] | mGlu2, mGlu2/3, mGlu4, mGlu5 |
| Li et al., 2015 | [47] | mGlu2/3 |
| Golubeva et al., 2016 | [48] | mGlu2, mGlu2/3, mGlu4, mGlu5, mGlu7 |
| Walker et al., 2015 | [49] | mGlu1, mGlu2, mGlu3, mGlu2/3, mGlu5 |
| Muguruza et al., 2016 | [50] | mGlu2/3 |
| Wierońska et al., 2016 | [51] | mGlu2/3, mGlu5, mGlu4, mGlu7 |
| Foster et al., 2017 | [52] | mGlu1, mGlu2, mGlu3, mGlu2/3, mGlu5 |
| Maksymetz et al., 2017 | [53] | mGlu1, mGlu2, mGlu3, mGlu4, mGlu5, mGlu7, mGlu8 |
| Nicoletti et al., 2019 | [54] | mGlu1, mGlu2, mGlu2/3, mGlu4, mGlu5 |
| Stansley et al., 2019 | [55] | mGlu1, mGlu3 |
| M1 | M4 | M5 | GABAB | mGlu2 | mGlu4 | mGlu5 | |
|---|---|---|---|---|---|---|---|
| cortex | +++++ | +++ | + | ++++ | ++ | ++ | ++/+++ |
| hippocampus | +++ | nd | + | +++++ | +++ | + | ++++ |
| striatum | +++++ | +++ | + | +++/++++ | ++ | +++ | ++ |
| hypothalamus | nd | nd | nd | ++++ | 0/+ | +++ | + |
| thalamus | ++ | +++ | + | ++++ | ++ | +++ | ++ |
| amygdala | nd | nd | nd | +++ | +/++ | +++ | +/++ |
| cerebellum | nd | nd | nd | ++++ | +++ | +++++ | ++ |
| Positive Symptoms | Negative Symptoms | Cognitive Symptoms |
|---|---|---|
| DOI-induced head twitches Amphetamine-induced hyperlocomotion MK-801-induced hyperlocomotion | Social interactions Modified forced swim test | Novel object recognition Spatial alterations Prepulse inhibition |
| Synaptic Localization | Behavioral Test | Activity | |
|---|---|---|---|
| Pre | Pre | ||
| mGlu2 | M4 | social interaction test | + |
| novel object recognition test | + | ||
| mGlu4 | M4 | DOI-induced head twitches | −/+ |
| MK-801-induced hyperactivity | + | ||
| AMPH-induced hyperactivity | + | ||
| modified forced swim test | + | ||
| social interaction test | + | ||
| novel object recognition test | + | ||
| GABAB | mGlu4 | DOI-induced head twitches | + |
| MK-801-induced hyperactivity | + | ||
| social interaction test | − | ||
| novel object recognition test | − | ||
| GABAB | M4 | DOI-induced head twitches | + |
| social interaction test | − | ||
| novel object recognition test | + | ||
| Synaptic Localization | Behavioral Test | Activity | |
|---|---|---|---|
| Pre | Post | ||
| mGlu2 | M1 | novel object recognition test | + |
| prepulse inhibition | + | ||
| spatial-delayed alternation test | + | ||
| mGlu2 | M5 | novel object recognition test | + |
| prepulse inhibition | + | ||
| spatial-delayed alternation test | + | ||
| GABAB | mGlu5 | modified forced swim test | + |
| social interaction test | + | ||
| novel object recognition test | + | ||
| GABAB | M1 | DOI-induced head twitches | − |
| novel object recognition test | + | ||
| GABAB | M5 | DOI-induced head twitches | − |
| novel object recognition test | + | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cieślik, P.; Wierońska, J.M. Regulation of Glutamatergic Activity via Bidirectional Activation of Two Select Receptors as a Novel Approach in Antipsychotic Drug Discovery. Int. J. Mol. Sci. 2020, 21, 8811. https://doi.org/10.3390/ijms21228811
Cieślik P, Wierońska JM. Regulation of Glutamatergic Activity via Bidirectional Activation of Two Select Receptors as a Novel Approach in Antipsychotic Drug Discovery. International Journal of Molecular Sciences. 2020; 21(22):8811. https://doi.org/10.3390/ijms21228811
Chicago/Turabian StyleCieślik, Paulina, and Joanna M. Wierońska. 2020. "Regulation of Glutamatergic Activity via Bidirectional Activation of Two Select Receptors as a Novel Approach in Antipsychotic Drug Discovery" International Journal of Molecular Sciences 21, no. 22: 8811. https://doi.org/10.3390/ijms21228811





