Abstract
(1) Background: The atopic march is defined by the increased prevalence of allergic diseases after atopic dermatitis onset. In fact, atopic dermatitis is believed to play an important role in allergen sensitization via the damaged skin barrier, leading to allergic diseases such as allergic asthma and allergic rhinitis. The eosinophil, a pro-inflammatory cell that contributes to epithelial damage, is one of the various cells recruited in the inflammatory reactions characterizing these diseases. Few studies were conducted on the transcriptome of this cell type and even less on their specific microRNA (miRNA) profile, which could modulate pathogenesis of allergic diseases and clinical manifestations post-transcriptionally. Actually, their implication in allergic diseases is not fully understood, but they are believed to play a role in inflammation-related patterns and epithelial cell proliferation. (2) Methods: Next-generation sequencing was performed on RNA samples from eosinophils of individuals with atopic dermatitis, atopy, allergic rhinitis and asthma to obtain differential counts of primary miRNA (pri-miRNA); these were also analyzed for asthma-related phenotypes such as forced expiratory volume in one second (FEV1), immunoglobulin E (IgE) and provocative concentration of methacholine inducing a 20% fall in forced expiratory volume in 1 s (PC20) levels, as well as FEV1 to forced vital capacity (FEV1/FVC) ratio. (3) Results: Eighteen miRNAs from eosinophils were identified to be significantly different between affected individuals and unaffected ones. Based on counts from these miRNAs, individuals were then clustered into groups using Ward’s method on Euclidian distances. Groups were found to be explained by asthma diagnosis, familial history of respiratory diseases and allergic rhinitis as well as neutrophil counts. (4) Conclusions: The 18 differential miRNA counts for the studying phenotypes allow a better understanding of the epigenetic mechanisms underlying the development of the allergic diseases included in the atopic march.
Keywords:
microRNAs; miRNAs; eosinophils; atopic march; asthma; allergy; gene expression; sequencing 1. Introduction
Allergic diseases (which include atopic dermatitis, food allergy, allergic asthma, and allergic rhinitis) are noncommunicable diseases that have seen the greatest increase in prevalence in the last decades, a phenomenon described as the “allergy epidemic” [1]. In the past ten years, allergy prevalence and hospitalization rates for severe allergic reactions has tripled [2] and our understanding of the root causes of allergic diseases has seen a complete paradigm shift [3]. In fact, the atopic march is defined by an increased prevalence of allergic diseases in individuals with atopic dermatitis. The latter, long considered only an epiphenomenon of allergies, has now clearly been shown to play a central role in their development. The inflamed and disrupted skin barrier of infants with atopic dermatitis allows the penetration of food and environmental allergenic proteins, leading to sensitization, the first step in becoming allergic. This explains why children with atopic dermatitis are at a high risk of developing food allergy (35% prevalence), allergic asthma (50%), and allergic rhinitis (75%) later in life [4]. Not only is the incidence of these diseases increased with atopic dermatitis, but they are also much more severe when compared to children without a history of atopic dermatitis [4].
Allergic asthma, which can happen at the final stages of the atopic march, is a chronic inflammatory disease characterized by symptoms of wheezing, shortness of breath, chest tightness, cough and variable expiratory airflow limitation [5]. Asthma is often associated with airway hyperresponsiveness and chronic inflammation in reaction to various stimuli including allergens, infections and air pollutants. Causes of asthma are multiple; they include environmental and genetic factors [6]. Indeed, familial history of asthma was found to be an important risk factor for the development of the disease, which is characterized by a calculated heritability of 55 to 90% [7]. However, heritability only partially explains the disease prevalence in the atopic march process. Another part of this prevalence could also be explained by epigenetic mechanisms such as DNA methylation, histone modification and non-coding RNAs. [8] More studies need to be conducted to better understand the influence of these mechanisms on diseases of the atopic march, especially concerning miRNA profiles.
miRNAs are small non-coding RNAs with 19–25 nucleotides that regulate gene expression post-transcriptionally. They silence complementary genes by targeting the 3′ untranslated region of mRNA, repressing their expression [9]. Their maturation includes primary (pri-miRNA, up to 1000 nucleotides long) and precursor (pre-miRNA, 60–120 nucleotides long) steps [10]. miRNAs act on various pro-inflammatory mechanisms in asthma and on smooth muscle cell proliferation, promoting airway hyperresponsiveness and playing an essential role in its pathogenesis [11]. Some miRNAs were also found to influence both the differentiation of T helper cells [12] and the innate immune response in keratinocytes of atopic dermatitis patients, [13] and to be differentially expressed in subjects with rhinitis and other allergic diseases [14,15].
Difference in miRNA profiles was also associated with other asthma-related phenotypes. Methacholine challenge (PC20), forced expiratory volume in one second (FEV1) and FEV1 to forced vital capacity (FEV1/FVC) ratio are all measures used as diagnostic tools or indicators of asthma severity [16,17]. Few studies have focused on miRNA expression predicting these phenotypes, but all three were previously associated with differential counts for certain miRNAs [18,19,20]. Finally, the differences in miRNA patterns that were observed for various diseases could lead to the identification of potential biomarkers for atopic dermatitis, asthma or other allergic diseases and even of potential therapeutic targets [8,11,21,22,23,24,25,26,27].
Moreover, few studies have demonstrated differential miRNA patterns in eosinophils, a type of pro-inflammatory cells whose proliferation in airways is characteristic of certain asthma phenotypes [28]. A study by Rodrigo-Muñoz et al. found that miRNAs in these cells that are peripheral can serve as potential diagnostic tools [29]. Another study by Allantaz et al. has shown a down-regulation of miR-155 in eosinophils that could be implicated in inflammatory processes [30]. Studying miRNA profiles from cell types well-known for their implication in allergic asthma is important considering the cell-type-specific characteristics of the transcriptome.
In this study, we sought to determine if some miRNAs were differentially expressed between individuals with allergic diseases included in the atopic march process and unaffected ones. We also wanted to see if they could be grouped based on their miRNA expression counts associated with diseases of the atopic march and phenotypes.
2. Results
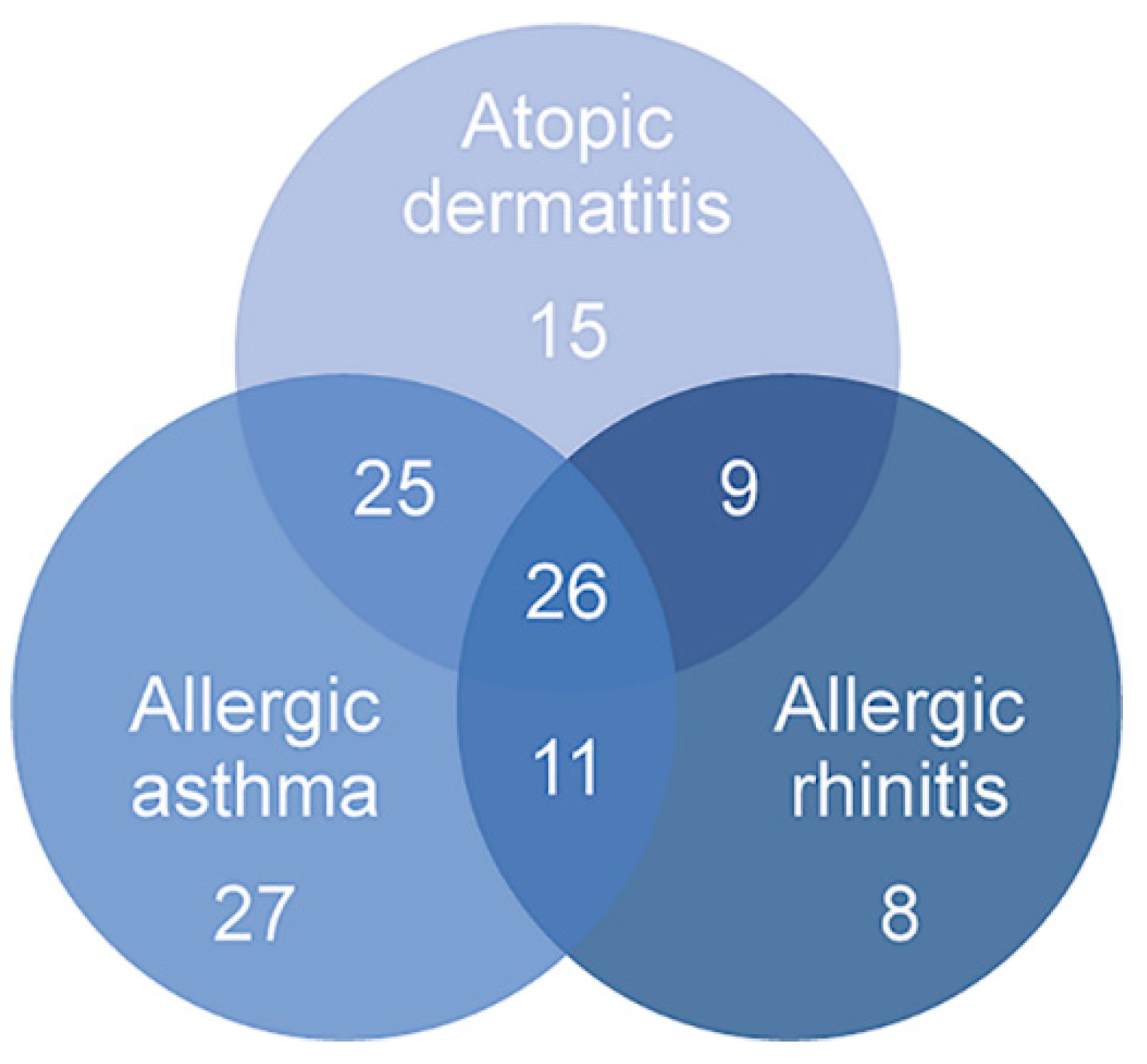
This study used pri-miRNA expression counts from 145 individuals of the Saguenay‒Lac-Saint-Jean (SLSJ) asthma familial cohort sampled for eosinophils in order to better understand underlying epigenetic mechanisms of diseases included in the atopic march process. The counts were analyzed for atopic dermatitis, atopy, allergic rhinitis, asthma, and asthma-related phenotypes (FEV1, IgE levels, PC20 and FEV1/FVC ratio). A clustering approach with significant miRNAs was then applied to identify potential explanations regarding the phenotypes for the differential counts of pri-miRNA between individuals. The phenotypic characteristics of the 145 individuals are accessible in Table 1 and number of individuals presenting overlapping conditions are represented in Figure 1.

Table 1.
Phenotypic characteristics of the individuals included in the eosinophil analysis of the miRNAs.
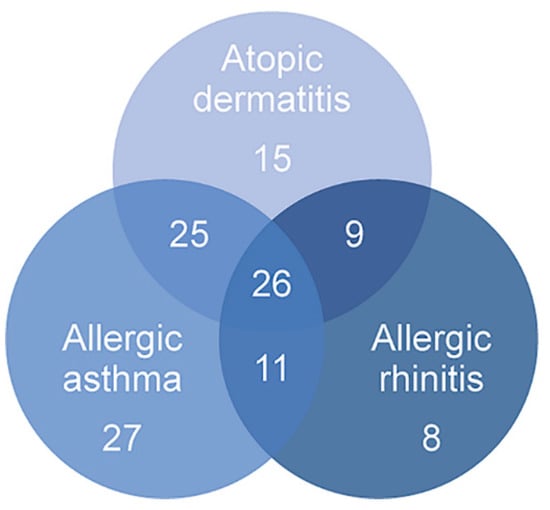
Figure 1.
Number of individuals presenting overlapping conditions of the atopic march. Overall, 75 individuals presented atopic dermatitis, 89 had allergic asthma and 54 had allergic rhinitis. Several of these individuals had overlapping conditions, highlighting the increased risk of presenting an allergic disease as allergic asthma or allergic rhinitis if an individual has atopic dermatitis.
2.1. Differential Counts of Pri-miRNA Associated with Allergic Diseases Included in the Atopic March
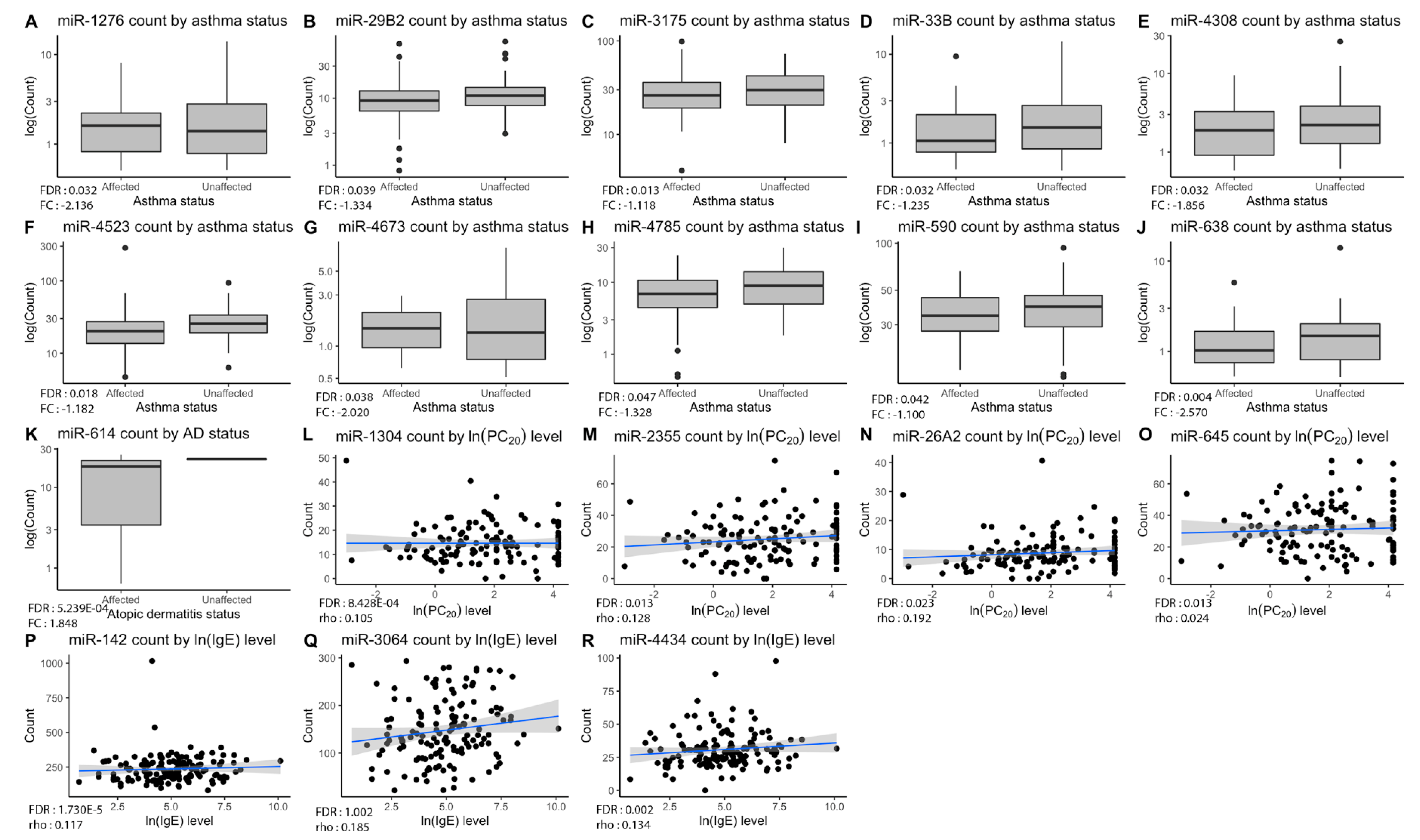
The negative binomial regression model performed for each trait (atopic dermatitis, atopy, asthma, allergic rhinitis, FEV1, IgE levels, PC20 and FEV1/FVC ratio) allowed us to find 18 miRNAs with significant differences in expression counts for the eosinophil cell type (Table 2). One miRNA was found to be up-regulated in atopic dermatitis (fold change (FC) = 1.85), ten miRNAs were down-regulated in asthma (FC from −2.57 to −1.10), three miRNAs were positively correlated with IgE level (rho from 0.02 to 0.19) as well as four miRNAs with PC20 level (rho from 0.11 to 0.18). The distribution of counts for each significant miRNA in accordance with the testing phenotypes is represented in Figure 2.

Table 2.
Significant associations (FDR < 0.05) between pri-miRNAs expression counts and diseases included in the atopic march process or related phenotypes.
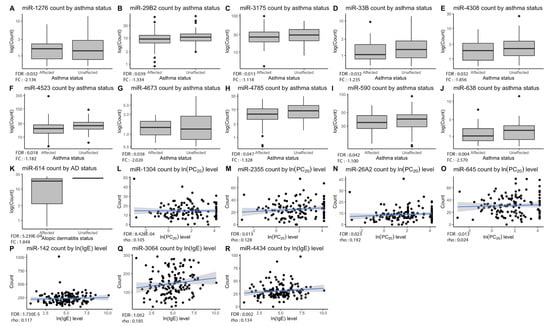
Figure 2.
Significant pri-miRNA expression counts by phenotypes. Plots (A–J) show down-regulation of ten pri-miRNAs in asthmatic patients compared to non-asthmatics, while plot (K) shows up-regulation of miR-614 for individuals with atopic dermatitis in comparison with unaffected ones. As for plots (L–O), they display positive correlations between miRNAs and PC20 levels. Lastly, plots (P–R) show positive correlation between miRNAs and IgE levels. Overall p-value of the analysis is in lower-left corner of each graph with corresponding fold change or Spearman’s rho value.
2.2. Clustering of Significant miRNAs
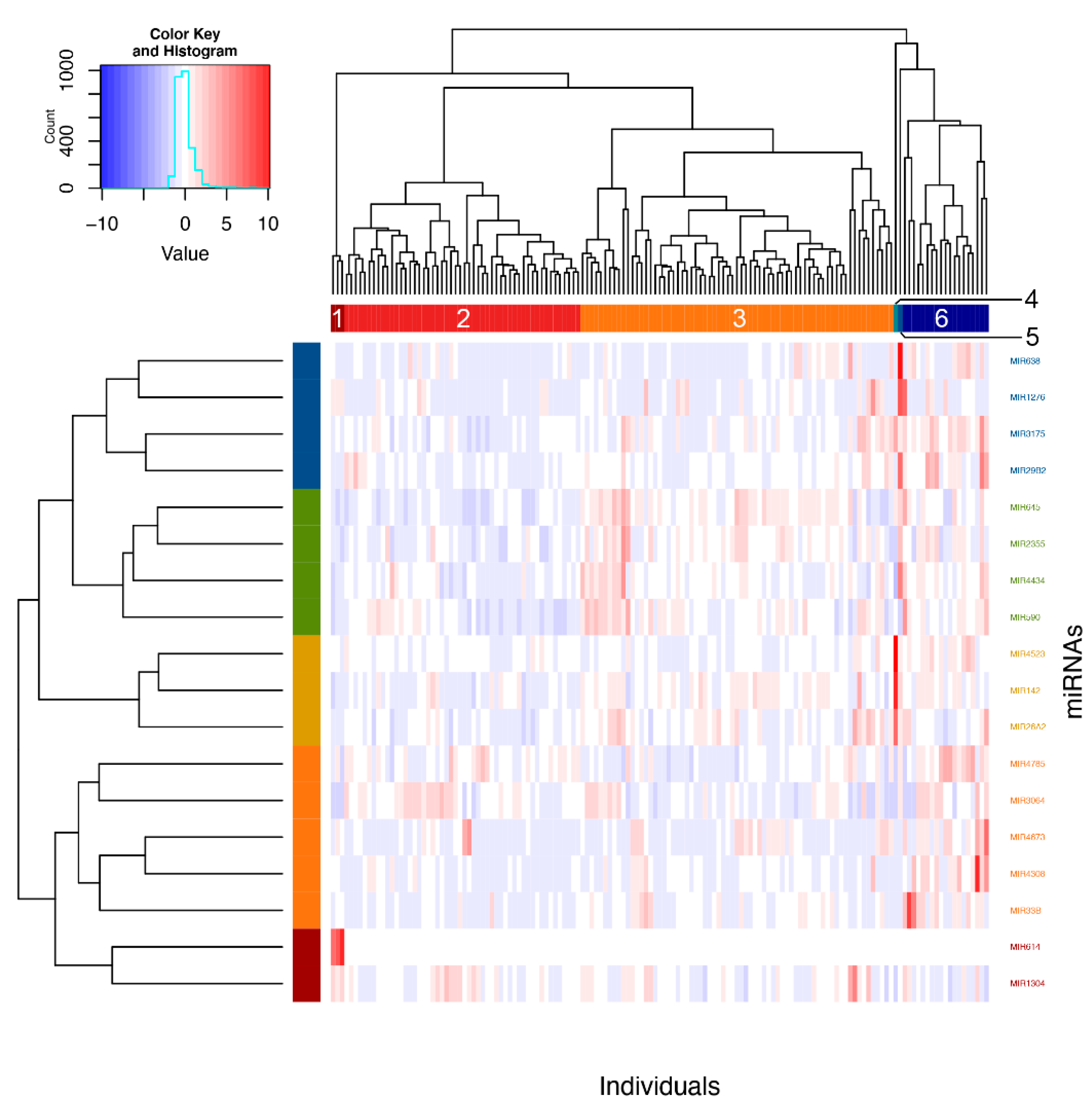
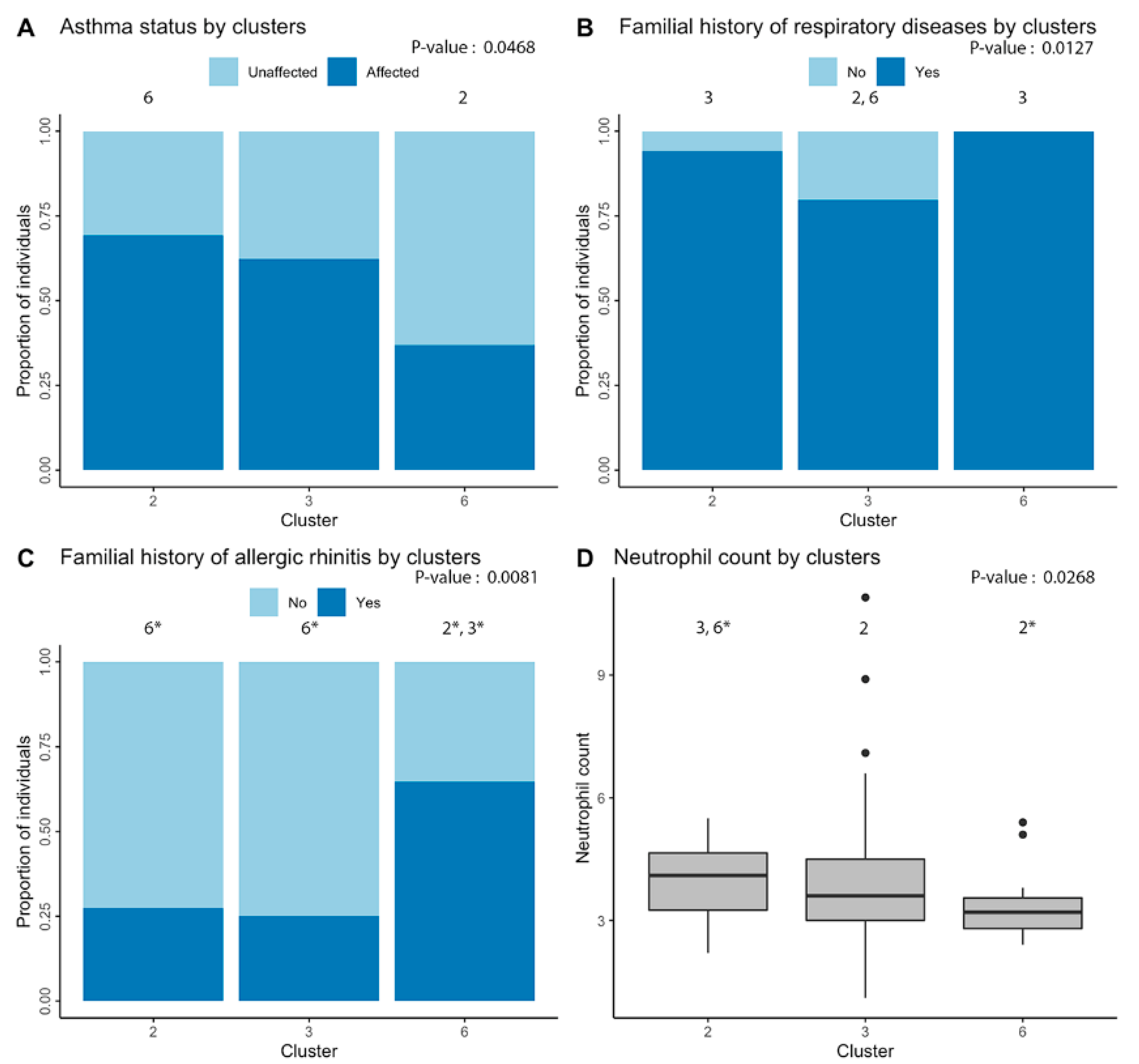
Using Ward’s method on Euclidian distances for the significant miRNAs, six clusters were obtained (Figure 3). Analyses were then performed to define which phenotypic elements characterize them. Clusters 1, 4 and 5 were ignored in analyses because they contained three individuals or less. As for clusters 2, 3 and 6, they included, respectively, 52, 69 and 19 individuals and were kept for subsequent steps. See Table S1 for a detailed view of all the phenotypes for comparing clusters. These were found to be explained by asthma phenotype, familial history of respiratory diseases and allergic rhinitis as well as neutrophil counts (Figure 4).
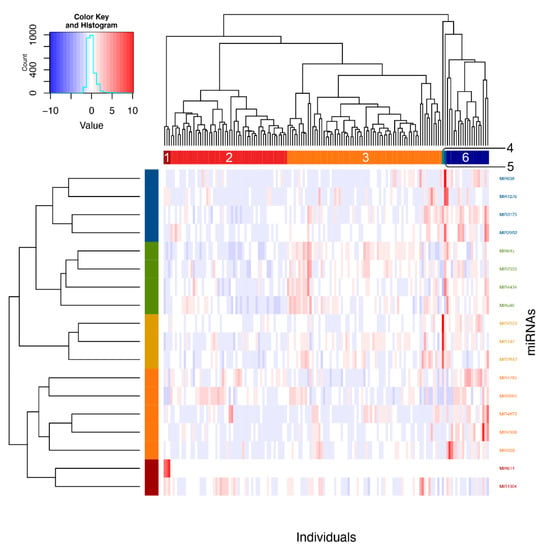
Figure 3.
Clustering of individuals according to significant pri-miRNAs counts. Individuals were clustered according to expression counts from the 18 associated miRNAs using Ward’s method on Euclidian distances. Six clusters were then obtained by visual inspection of the dendrogram and confirmed with the NbClust package in R.
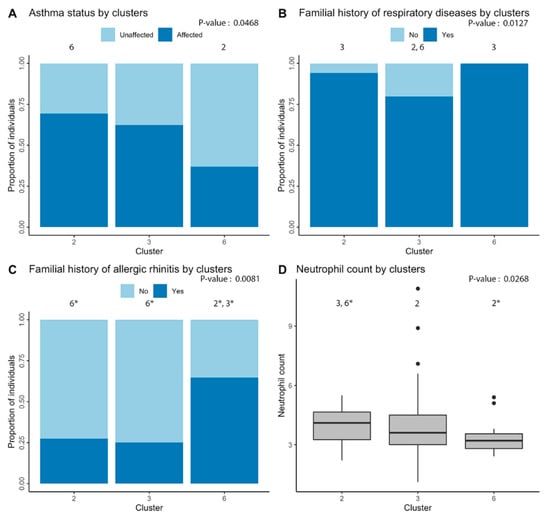
Figure 4.
Phenotypic differences underlying clustering of individuals. Significant differences between individuals of the three main clusters (including more than three people) were found for: (A) asthma; (B) familial history of respiratory diseases; (C) familial history of allergic rhinitis; and (D) neutrophil cell count. Overall p-value of the analysis is in the upper-right corner of each graph and the number above each cluster indicates the other clusters that are significantly different in post-hoc analyses. An asterisk indicates significant difference after Bonferroni correction.
Moreover, looking at Figure 4 and according to post-hoc analyses, cluster 2 was associated with higher rates of diagnosis of asthma as compared with cluster 6 (p-value = 0.027). Cluster 3 was less related to familial history of respiratory diseases as clusters 2 (p-value = 0.033) and 6 (p-value = 0.034). Cluster 6 also had a stronger association with familial history of allergic rhinitis when compared with clusters 2 (p-value = 0.009) and 3 (p-value = 0.003). Finally, cluster 2 had a higher neutrophil count in comparison with clusters 3 (p-value = 0.035) and 6 (p-value = 0.005).
2.3. Identifiaction of Possible Gene Targets
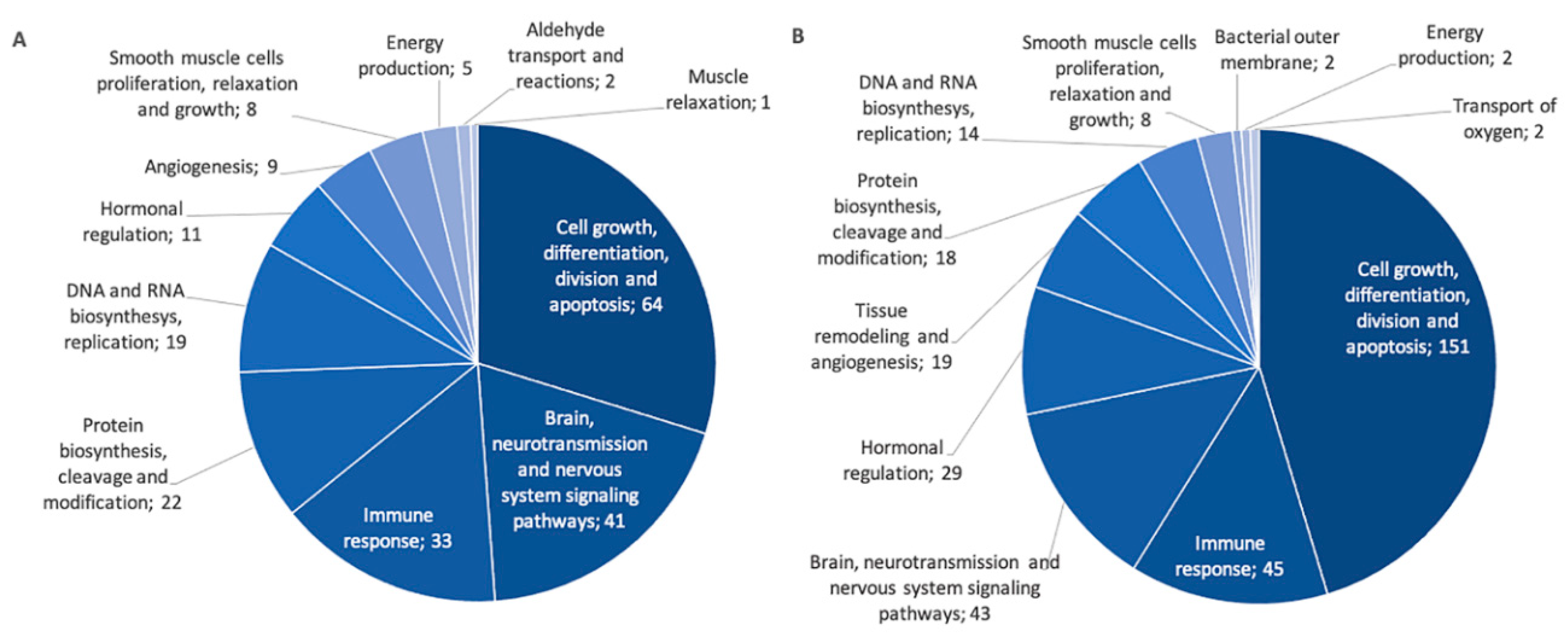
In order to better understand underlying mechanisms explaining associations observed between pri-miRNA expression and diseases of the atopic march or clinical measures, correlations were performed to identify possible gene targets. miRNA–gene target pairs with false discovery rate (FDR)-values inferior to 0.05 and absolute correlation coefficients >0.2 were deemed significant and further validated using miRTArBase v8.0 [31]. A total of 540 validated miRNA–gene target pairs were found using negative correlations and 663 using positive correlations. According to the inhibition function of miRNAs, results of negative correlations are usually prioritized. However, a recent study highlighted the mechanisms underlying positive correlations with gene targets, such as in feedback loops and co-transcription of intronic miRNAs and their host genes [32], motivating the presentation of positive correlations in this study. The top 25 best negative and positive correlations are displayed in Table S2. They were then classified into PANTHER biological pathways [33]. Among the gene targets identified, 215 from the negative correlations and 353 from the positive ones have been successfully classified by PANTHER. Biological pathways identified were grouped into more general categories shown in Figure 5 (see Table S3 for the list of PANTHER pathways in each general category).
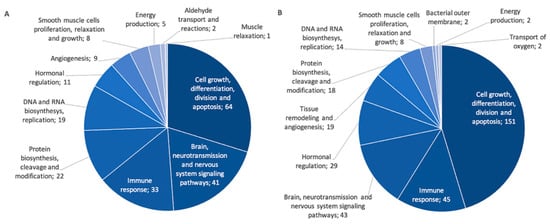
Figure 5.
General categories of PANTHER pathways for gene targets identified in negative and positive correlations with associated miRNAs. The number of genes is indicated for each category. (A) Pathways for negatively correlated miRNA–gene target pairs. (B) Pathways for positively correlated miRNA–gene target pairs.
Finally, gene targets found were compared to the genes already associated with diseases of the atopic march in the GWAS Catalogue (42 for negative correlations and 53 for positives ones) and the SLSJ asthma familial cohort (13 for the negative correlations and 22 for the positive ones; Table S4). Table 3 represents those gene targets that are included in PANTHER biological pathways. Negative correlations included eight gene targets associated in GWAS analyses and four were associated in the SLSJ cohort. Positive correlations included 19 gene targets associated in GWAS and nine were associated in the SLSJ cohort. Total resulting number of targets for positive and negative correlations before and after validation, as well as number of genes found in GWAS Catalogue and literature about the SLSJ asthma familial cohort are detailed in Table S4.

Table 3.
miRNAs’ gene targets previously associated in GWAS as listed by the GWAS Catalogue or in the Saguenay‒Lac-Saint-Jean (SLSJ) cohort and classified by PANTHER pathways.
3. Discussion
This research used pri-miRNA counts obtained by next-generation sequencing of the whole eosinophil transcriptome, a cell known to play an inflammatory role in asthma [28], for finding differential expression of certain miRNAs between patients having allergic diseases often associated with the atopic march and controls in order to better understand epigenetic mechanisms underlying these phenomena. Specifically, it was previously noted that eosinophils can carry miRNAs via exosomes to other cells; they are becoming not only a pro-inflammatory cell type, but also a vector for miRNAs and a potential regulator of gene expression [34]. In this sense, extracting miRNAs from eosinophils allows a better understanding of their effect on diseases in the atopic march process as compared to those from whole blood. Overall, miRNAs extracted from eosinophils improve the knowledge of their possible roles in proliferation of the inflammatory cells and immune functions in these diseases [35,36].
The miRNA levels measured from eosinophil cells extracted as part of this study allowed us to identify 18 that were differentially expressed between individuals with allergic diseases and unaffected ones, as well as for IgE levels and PC20. Five of them were associated in the past with asthma and allergic diseases (miR-142 [37,38], -26a [39,40], -29b [41,42], -590 [29,43] and -638 [44]). In fact, the miRNA expression counts of these five miRNAs were all differentially regulated in the same direction as that is found in literature. Three were previously associated with other respiratory conditions including lung cancer (miR-1276 [45], -1304 [46] and -33b [47]), hypopharyngeal cancer (miR-1304 [48]) and cystic fibrosis (miR-1276 [49]). Finally, three were previously associated with diseases involving inflammatory components (miR-2355 [50], -3175 [51], -33b [52]). This study led to the identification of 13 miRNAs not previously associated with allergic diseases or asthma. The remaining five miRNAs were already found to have a differential expression in diseases of the atopic march, but this study allowed the confirmation that these were also differentially expressed in eosinophils. There are very few studies on miRNA expression in eosinophil samples for allergic diseases. A study by Rodrigo-Muñoz et al. found 21 miRNAs differentially expressed in eosinophils in asthma, from which miR-590 was also down-regulated [29]. No significant differences were observed for the 20 other miRNAs in our study. This could be explained by their smaller sample size (29 asthmatic and 10 healthy individuals), the fact that they did not study an allergic endotype of asthma as done in this project, and also because we measured differential pri-miRNA counts as compared to mature miRNAs in their study. However, measuring pri-miRNA counts allow a more direct understanding of miRNA transcription. Finally, even though no mature miRNA data were available, several pri-miRNAs were previously associated with mature miRNAs in allergic diseases included in the atopic march process. Moreover, the target analysis of the significant miRNAs associated in this study found CITED2 as a target of miR-590, a gene regulating TGF-beta pathways acting on airway smooth muscle cell proliferation. In fact, eosinophils have previously been shown to enhance gene expression of TGF-beta1 and to increase airway smooth muscle cell proliferation [53]. In this sense, it is expected that this miRNA can be differentially regulated in both allergic and non-allergic endotypes of asthma. These findings point toward a regulatory role in these diseases. Finally, considering modest coefficients of correlations found between pri-miRNA counts and PC20 and IgE levels, we hypothesize that such correlations are more complex than mere direct interaction. In fact, Davis et al. have found that certain miRNAs could modulate PC20 by increasing airway smooth muscle cell diameter [18]. Therefore, more studies need to be done in order to better define links between these miRNAs and biological measures.
Actually, some well-known miRNAs in asthma and allergic diseases such as miR-221, -485-3p, -1248, -126, -146a/b, -28-5p, -181a, -133a or -10a [23] were not replicated in this study. Among these, four miRNAs were expressed in eosinophil samples, but their corresponding pri-miRNAs did not show significant differences according to the phenotype status. However, based on the cell-type specificity of transcriptome measurements, it was expected that eosinophils will have a distinct profile in comparison to whole blood. It was demonstrated in the past that whole blood miRNAs are derived from various exosomes specific to cell types; this means previously found differential miRNA counts associated with diseases in the atopic march process could be derived from other ones like lymphocytes, dendritic cells, platelets, mastocytes, epithelial cells, endothelial cells or neurons [54].
To further understand the links between pri-miRNA expression counts and phenotypic differences in asthma and allergic diseases, individuals were clustered into three distinct groups based on the pri-miRNA counts for each of the 18 previously associated miRNAs. These clusters featured significant differences, which is consistent with the proportion of subjects having asthma phenotype as well as familial history of respiratory diseases and allergic rhinitis, further confirming the family component in them [6]. Moreover, earlier studies have associated asthma prevalence with allergic rhinitis [55,56,57]; these findings tend to demonstrate a link between the two. However, more studies need to be done in regard to the similarities between the eosinophil miRNA patterns between asthma and allergic rhinitis to reach such a conclusion. A difference in counts of circulating neutrophils, a cell type characterizing an asthma endotype not typically linked with allergies [58], was also observed. With a higher proportion of individuals with familial history of allergic rhinitis and a smaller neutrophil count, cluster 6 shows a higher number of allergic individuals, even though its proportion of asthmatics is lower. However, it is important to note the smaller number of individuals included in this cluster (19 compared to 52 and 69). Interestingly, many miRNAs turned out to be differentially regulated in cluster 6, as compared to clusters 2 and 3. These miRNAs were previously found to play a role in diseases of the atopic march. It is the case for miR-142 [37,38], -26a [39,40], -29b [41,42], -590 [29,43] and -638 [44] which all had higher counts in cluster 6, in comparison with clusters 2 and 3. Similarly, both clusters 2 and 3 showed higher values for miR-1276 and -1304 whereas cluster 3 showed higher values for miR-33b and -2355, four miRNAs known for their implication in non-allergic respiratory diseases [45,46,47,48,49,50]. Furthermore, no association was made with eosinophil counts in the clustering approach. This could be explained by the fact that the eosinophil cell counts were similar between affected (by one or more allergic disease) and non-affected (by none of the allergic diseases tested) individuals. However, there is indeed a difference in eosinophil cell counts when we compare for a single disease such as asthma, with a mean of 0.29 × 109/L eosinophils in individuals with asthma and 0.18 × 109/L in unaffected ones. However, miRNAs associated in eosinophil samples were not necessarily linked with biological pathways involved in eosinophil recruitment or proliferation.
To further understand the implication of these miRNAs on diseases of the atopic march, an analysis to identify possible gene targets was performed. The 18 associated pri-miRNA expression counts were significantly correlated with several targets previously identified in diseases of the atopic march. Overall, 95 gene targets were already associated with diseases of the atopic march in the GWAS Catalogue and 35 with the SLSJ asthmatic cohort. Among gene targets that were linked with biological pathways, 42 were also associated with diseases of the atopic march, either in the GWAS Catalogue or in the SLSJ cohort. Those genes were classified into pathways including cell regulation, immune response, smooth muscle cell proliferation and angiogenesis, further confirming the importance of miRNAs in these diseases. However, further studies are needed to better understand the specific link between these 18 miRNAs and the different diseases of the atopic march.
This study sought to find a differential miRNA pattern in eosinophils from patients presenting with diseases in the atopic march process and unaffected individuals. Eighteen miRNAs turned out to be differentially expressed in eosinophil samples in case of either atopic dermatitis or asthmatic statuses than in unaffected individuals, or according to the PC20 or IgE levels. Moreover, the clustering approach used with the associated miRNAs revealed a link between these and fine phenotypic information defining the individuals included in this study and allowed identifying the miRNAs that are more likely to be involved specifically in the atopic component of the studied diseases among the 18 in association.
Overall, these miRNAs could help improve the knowledge of post-transcriptional regulation leading to allergic diseases included in the atopic march process. In fact, their differential regulation found in eosinophils help refine our understanding of miRNAs in these diseases and their important role in gene expression regulation. Finally, the presence of multiple atopic diseases in one patient may imply the relevance of miRNAs in the atopic march. However, birth cohorts and longitudinal studies will need to be performed to support this hypothesis. Despite this, it is interesting to note that a previous study including the SLSJ cohort and a birth cohort aiming at developing a polygenic risk score for moderate-to-severe atopic dermatitis, the sub-phenotype of atopic dermatitis associated with the highest risk to develop the atopic march, also showed good discriminative values for allergies, allergic asthma and allergic rhinitis in the SLSJ cohort [59].
4. Materials and Methods
4.1. SLSJ Asthma Cohort
The SLSJ asthma familial cohort comprises 1394 individuals distributed in 271 families from which 1214 subjects have genotypic information. Pulmonary health of each individual was evaluated according to the American Thoracic Society (ATS) Clinical Practice Guidelines using a standardized questionnaire and pulmonary function testing [60]. The subpopulation used for this study was selected using siblings with discordant asthma status and from trios of affected probands and discordant parents regarding asthma status. Using these criteria, 215 subjects were obtained to isolate their eosinophils for RNA sequencing. Asthma and atopy phenotypes were defined according to ATS standards [61]. Participants were considered as asthmatic if: (1) they had a reported history of asthma (validated by a physician), or (2) they presented asthma-related symptoms and positive PC20 (<8 mg/mL) at recruitment. Individuals were deemed atopic if they had at least one positive response on skin prick tests (wheal diameter ≥ 3 mm or larger than the wheal diameter elicited by the negative control (glycerin)) and have a physician diagnosis. Atopic dermatitis and allergic rhinitis were self-reported and were considered as positive if past or present occurrence of these diseases was reported. For children, cross validation was done using questionnaires filled out by their parents. Moreover, validation in medical records of these self-reported phenotypes were done for a subset of the SLSJ Cohort (n = 217), giving 89% concordance. Proportions of white blood cells were obtained using a Coulter LH 780 hematology analyzer (Beckman Coulter, Mississauga, ON, Canada) to estimate proportion of five different types of white blood cells: eosinophils, lymphocytes, neutrophils, monocytes and basophils. Respiratory measures such as PC20 and FEV1 were taken using a Morgan spirometer (Morgan Spiro 232, P.K. Morgan Ltd., Kent, UK) [62]. Complete descriptions of both recruitment and evaluation used for the SLSJ cohort can be found in Laprise et al. [62] All participants gave informed consent and the study was approved by the Centre intégré universitaire de santé et de services sociaux du Saguenay–Lac-Saint-Jean ethics committee (project #0002-001, 08-11-2005).
4.2. Isolation of RNA from Eosinophils and Sequencing
Complete description of the procedure for eosinophil isolation can be found in Madore et al. [63] Briefly, eosinophils were isolated by negative selection from 200 mL blood samples with anti-CD16, anti-CD3 and anti-CD19 MicroBeads (Miltenyi Biotec, Auburn, CA, USA) and a proportion of cells (2 × 106) was used for total RNA extraction (molecules ≥ 200 nucleotides). This step was performed with the RNeasy Mini Kit following manufacturer’s instructions (Qiagen, Toronto, ON, Canada). RNA sequencing was carried out at the McGill University and Genome Québec Innovation Center using TruSeq Stranded Total RNA Sample Prep kit (Illumina, Vancouver, BC, Canada). Final libraries were quality controlled on a Bioanalyzer (Agilent Technologies, Mississauga, ON, Canada) and underwent 100 bp paired-end sequencing on the Illumina HiSeq2000 System (Illumina, Vancouver, BC, Canada). Generated raw reads were filtered for quality (phred33 ≥ 30) and length (n ≥ 32) as well as adapter sequences were removed using Trimmomatic v.0.32 as previously described [63]. Considering the length of the extracted RNAs (≥200 nucleotides), expression counts were available for pri-miRNAs. After quality control filtering, 441 pri-miRNA were available for analyses. From the 215 initial individuals, considering the large amount of blood necessary for RNA sequencing, quality filtering applied and the covariates availability for statistical analysis, pri-miRNA counts were accessible for 145 of them. These results reflect the transcription of miRNA sequences without necessarily quantifying the abundance of the mature miRNAs [10].
4.3. Statistical Methods
4.3.1. Analyses of Pri-miRNA Expression Counts between Individuals
Pri-miRNA expression counts were compared with a negative binomial regression model built with pri-miRNA normalized expression counts and phenotypes using the MASS package in R. Studied phenotypes were atopic dermatitis (75 affected, 70 healthy), atopy (94 affected, 51 healthy), allergic rhinitis (54 affected, 91 healthy), asthma (89 affected, 56 healthy), FEV1, IgE levels, PC20 and FEV1/FVC ratio. Age, sex, smoking history, eosinophil proportion and surrogate variables were used as covariates. Proportions of eosinophils were calculated working with methylome data from the same eosinophil samples as well as the method by Houseman implemented in the R package RnBeads as described above (Table S5) [64,65]. Surrogate variables were evaluated using the R package sva in order to account for batch effects, relatedness between samples and other hidden confounders [66]. Normalization of miRNAs expression counts on library size was done with the DESeq2 package. Differences in these with a FDR-value <0.05 were deemed significant. FC were calculated using the mean ratios for affected and non-affected individuals in case of analyses taking into account the disease status whereas Spearman’s rank correlation was performed between pri-miRNA counts and continuous phenotypes. miRNAs with positive FC were considered up-regulated while those with negative FC were down-regulated in the affected group of each allergic disease analyzed.
4.3.2. Clustering of miRNAs
Significant miRNAs were used to cluster individuals with Ward’s method on Euclidian distances in R, as described by O’Sullivan et al. [67] Groups were determined by visual inspection of the dendrogram and confirmed using the NbClust package in R. An explanation for these clusters was then searched comparing the phenotypic measures between groups with Fisher’s exact test for binary data, one-way ANOVA for continuous normally distributed variables and Kruskal–Wallis test for continuous non-normally distributed ones. Post-hoc analyses were performed afterward to find pairwise differences between clusters using Fisher’s exact test, Tukey’s test and Dunn’s test, respectively, followed by Bonferroni corrections. Clusters with three individuals or less were ignored from this testing (data available for 139 subjects on the 145 included in the pri-miRNA analysis).
4.3.3. Identification of Possible Gene Targets for Associated Pri-miRNAs
Negative and positive correlations were applied between the 18 associated pri-miRNA and gene log-transformed expression count in order to find which genes are possible gene targets. Correlations that reached an FDR-value <0.05 and an absolute correlation coefficient > 0.2 were deemed significant. These miRNA–gene target pairs were then validated with miRTarBase v8.0 [31], a database that list gene targets of miRNAs that were observed using technical approaches (e.g., qRT-PCR, next-generation sequencing), in order to find which of the significant genes are more likely to be real targets. These targets were classified according to their PANTHER pathways [33]. Gene targets that were previously associated with diseases of the atopic march were identified using the GWAS Catalogue (https://www.ebi.ac.uk/gwas/) and studies from the SLSJ asthma familial cohort. The GWAS Catalogue list all human genome-wide associations for studies targeting 100,000 single nucleotide polymorphisms (SNPs) or more and with p-values <10 × 10−5.
Supplementary Materials
Supplementary Materials can be found at https://www.mdpi.com/1422-0067/21/23/9011/s1.
Author Contributions
Conceptualization and supervision, C.L.; funding acquisition, C.L. and T.P.; resources, A.-M.B.-L., M.-M.S., T.K., T.P. and C.L.; formal analysis, É.B. and A.-M.M.; writing—original draft preparation, É.B.; writing—review and editing, A.-M.M. and C.L. All authors have read and agreed to the published version of the manuscript.
Funding
Anne-Marie Boucher-Lafleur was supported by a Fonds de recherche du Québec—Santé (FRQS) and the Quebec Respiratory Health Network (RHN) Master training awards. This project was supported by operating grants from the Canadian Institute of Health Research (CIHR; Laprise & Pastinen, #133605). Catherine Laprise is part of the Quebec Respiratory Health Network (RHN; https://rsr-qc.ca/en/), the investigator of CHILD Study, the director of the Centre intersectoriel en santé durable de l’UQAC and the chairholder of the Canada Research Chair in the Environment and Genetics of Respiratory Diseases and Allergy (http://www.chairs.gc.ca).
Acknowledgments
We wish to thank the participants recruited in the SLSJ asthma cohort for their valuable input in this study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Platts-Mills, T.A. The allergy epidemics: 1870–2010. J. Allergy Clin. Immunol. 2015, 136, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Dear, K.B.; Tang, M.L. Time trends in Australian hospital anaphylaxis admissions in 1998–1999 to 2011–2012. J. Allergy Clin. Immunol. 2015, 136, 367–375. [Google Scholar] [CrossRef]
- Du Toit, G.; Tsakok, T.; Lack, S.; Lack, G. Prevention of food allergy. J. Allergy Clin. Immunol. 2016, 137, 998–1010. [Google Scholar] [CrossRef] [PubMed]
- Carlsten, C.; Dimich-Ward, H.; Ferguson, A.; Watson, W.; Rousseau, R.; Dybuncio, A.; Becker, A.; Chan-Yeung, M. Atopic dermatitis in a high-risk cohort: Natural history, associated allergic outcomes, and risk factors. Ann. Allergy Asthma Immunol. 2013, 110, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. 2019. Available online: https://ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-June-2019-wms.pdf (accessed on 23 April 2020).
- Mims, J.W. Asthma: Definitions and pathophysiology. Int. Forum Allergy Rhinol. 2015, 5 (Suppl. 1), S2–S6. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Pacheco, N.; Pino-Yanes, M.; Flores, C. Genomic Predictors of Asthma Phenotypes and Treatment Response. Front. Pediatr. 2019, 7, 6. [Google Scholar] [CrossRef]
- Lovinsky-Desir, S.; Miller, R.L. Epigenetics, asthma, and allergic diseases: A review of the latest advancements. Curr. Allergy Asthma Rep. 2012, 12, 211–220. [Google Scholar] [CrossRef]
- Lu, T.X.; Rothenberg, M.E. MicroRNA. J. Allergy Clin. Immunol. 2018, 141, 1202–1207. [Google Scholar] [CrossRef]
- Lee, Y.; Jeon, K.; Lee, J.T.; Kim, S.; Kim, V.N. MicroRNA maturation: Stepwise processing and subcellular localization. EMBO J. 2002, 21, 4663–4670. [Google Scholar] [CrossRef]
- Maneechotesuwan, K. Role of microRNA in severe asthma. Respir. Investig. 2019, 57, 9–19. [Google Scholar] [CrossRef]
- Ma, L.; Xue, H.B.; Wang, F.; Shu, C.M.; Zhang, J.H. MicroRNA-155 may be involved in the pathogenesis of atopic dermatitis by modulating the differentiation and function of T helper type 17 (Th17) cells. Clin. Exp. Immunol. 2015, 181, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Rebane, A.; Runnel, T.; Aab, A.; Maslovskaja, J.; Rückert, B.; Zimmermann, M.; Plaas, M.; Kärner, J.; Treis, A.; Pihlap, M.; et al. MicroRNA-146a alleviates chronic skin inflammation in atopic dermatitis through suppression of innate immune responses in keratinocytes. J. Allergy Clin. Immunol. 2014, 134, 836–847.e11. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Yang, G.; Zhang, R.; Xu, G.; Zhang, L.; Wen, W.; Lu, J.; Liu, J.; Yu, Y. Altered microRNA Expression Profiles of Extracellular Vesicles in Nasal Mucus From Patients With Allergic Rhinitis. Allergy Asthma Immunol. Res. 2015, 7, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Suojalehto, H.; Toskala, E.; Kilpeläinen, M.; Majuri, M.L.; Mitts, C.; Lindström, I.; Puustinen, A.; Plosila, T.; Sipilä, J.; Wolff, H.; et al. MicroRNA profiles in nasal mucosa of patients with allergic and nonallergic rhinitis and asthma. Int. Forum Allergy Rhinol. 2013, 3, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Nair, P.; Martin, J.G.; Cockcroft, D.C.; Dolovich, M.; Lemiere, C.; Boulet, L.P.; O’Byrne, P.M. Airway Hyperresponsiveness in Asthma: Measurement and Clinical Relevance. J. Allergy Clin. Immunol. Pract. 2017, 5, 649–659.e2. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.; Gindner, L.; Tilemann, L.; Schermer, T.; Dinant, G.J.; Meyer, F.J.; Szecsenyi, J. Diagnostic accuracy of spirometry in primary care. BMC Pulm. Med. 2009, 9, 31. [Google Scholar] [CrossRef]
- Davis, J.S.; Sun, M.; Kho, A.T.; Moore, K.G.; Sylvia, J.M.; Weiss, S.T.; Lu, Q.; Tantisira, K.G. Circulating microRNAs and association with methacholine PC20 in the Childhood Asthma Management Program (CAMP) cohort. PLoS ONE 2017, 12, e0180329. [Google Scholar] [CrossRef]
- Trinh, H.K.T.; Pham, D.L.; Kim, S.C.; Kim, R.Y.; Park, H.S.; Kim, S.H. Association of the miR-196a2, miR-146a, and miR-499 Polymorphisms with Asthma Phenotypes in a Korean Population. Mol. Diagn. Ther. 2017, 21, 547–554. [Google Scholar] [CrossRef]
- Kho, A.T.; Sharma, S.; Davis, J.S.; Spina, J.; Howard, D.; McEnroy, K.; Moore, K.; Sylvia, J.; Qiu, W.; Weiss, S.T.; et al. Circulating MicroRNAs: Association with Lung Function in Asthma. PLoS ONE 2016, 11, e0157998. [Google Scholar] [CrossRef]
- Mousavi, S.R.; Ahmadi, A.; Jamalkandi, S.A.; Salimian, J. Involvement of microRNAs in physiological and pathological processes in asthma. J. Cell. Physiol. 2019, 234, 21547–21559. [Google Scholar] [CrossRef]
- Levänen, B.; Bhakta, N.R.; Paredes, P.T.; Barbeau, R.; Hiltbrunner, S.; Pollack, J.L.; Sköld, C.M.; Svartengren, M.; Grunewald, J.; Gabrielsson, S.; et al. Altered microRNA profiles in bronchoalveolar lavage fluid exosomes in asthmatic patients. J. Allergy Clin. Immunol. 2013, 131, 894–903. [Google Scholar] [CrossRef]
- Kai, W.; Qian, X.U.; Qun, W.U. MicroRNAs and Asthma Regulation. Iran. J. Allergy Asthma Immunol. 2015, 14, 120–125. [Google Scholar] [PubMed]
- Wang, Y.; Yang, L.; Li, P.; Huang, H.; Liu, T.; He, H.; Lin, Z.; Jiang, Y.; Ren, N.; Wu, B.; et al. Circulating microRNA Signatures Associated with Childhood Asthma. Clin. Lab. 2015, 61, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Bin, L.; Leung, D.Y. Genetic and epigenetic studies of atopic dermatitis. Allergy Asthma Clin. Immunol. 2016, 12, 52. [Google Scholar] [CrossRef]
- Zhang, X.-H.; Zhang, Y.-N.; Liu, Z. MicroRNA in chronic rhinosinusitis and allergic rhinitis. Curr. Allergy Asthma Rep. 2014, 14, 415. [Google Scholar] [CrossRef]
- Dissanayake, E.; Inoue, Y. MicroRNAs in Allergic Disease. Curr. Allergy Asthma Rep. 2016, 16, 67. [Google Scholar] [CrossRef]
- Drake, M.G.; Lebold, K.M.; Roth-Carter, Q.R.; Pincus, A.B.; Blum, E.D.; Proskocil, B.J.; Jacoby, D.B.; Fryer, A.D.; Nie, Z. Eosinophil and airway nerve interactions in asthma. J. Leukoc. Biol. 2018, 104, 61–67. [Google Scholar] [CrossRef]
- Rodrigo-Muñoz, J.M.; Cañas, J.A.; Sastre, B.; Rego, N.; Greif, G.; Rial, M.; Mínguez, P.; Mahíllo-Fernández, I.; Fernández-Nieto, M.; Mora, I.; et al. Asthma diagnosis using integrated analysis of eosinophil microRNAs. Allergy 2019, 74, 507–517. [Google Scholar] [CrossRef]
- Allantaz, F.; Cheng, D.T.; Bergauer, T.; Ravindran, P.; Rossier, M.F.; Ebeling, M.; Badi, L.; Reis, B.; Bitter, H.; D’Asaro, M.; et al. Expression profiling of human immune cell subsets identifies miRNA-mRNA regulatory relationships correlated with cell type specific expression. PLoS ONE 2012, 7, e29979. [Google Scholar] [CrossRef]
- Huang, H.-Y.; Lin, Y.-C.-D.; Li, J.; Huang, K.-Y.; Shrestha, S.; Hong, H.-C.; Tang, Y.; Chen, Y.-G.; Jin, C.-N.; Yu, Y.; et al. miRTarBase 2020: Updates to the experimentally validated microRNA–target interaction database. Nucleic Acids Res. 2019, 48, D148–D154. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, J.; Deng, F.Y.; Wu, L.F.; Mo, X.B.; Zhu, X.W.; Xia, W.; Xie, F.F.; He, P.; Bing, P.F.; et al. Correlation analyses revealed global microRNA-mRNA expression associations in human peripheral blood mononuclear cells. Mol. Genet. Genom. 2018, 293, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Mi, H.; Lazareva-Ulitsky, B.; Loo, R.; Kejariwal, A.; Vandergriff, J.; Rabkin, S.; Guo, N.; Muruganujan, A.; Doremieux, O.; Campbell, M.J.; et al. The PANTHER database of protein families, subfamilies, functions and pathways. Nucleic Acids Res. 2005, 33, D284–D288. [Google Scholar] [CrossRef] [PubMed]
- Cañas, J.A.; Sastre, B.; Rodrigo-Muñoz, J.M.; del Pozo, V. Exosomes: A new approach to asthma pathology. Clin. Chim. Acta 2019, 495, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Heffler, E.; Allegra, A.; Pioggia, G.; Picardi, G.; Musolino, C.; Gangemi, S. MicroRNA Profiling in Asthma: Potential Biomarkers and Therapeutic Targets. Am. J. Respir. Cell Mol. Biol. 2017, 57, 642–650. [Google Scholar] [CrossRef]
- Lu, T.X.; Rothenberg, M.E. Diagnostic, functional, and therapeutic roles of microRNA in allergic diseases. J. Allergy Clin. Immunol. 2013, 132, 3–13. [Google Scholar] [CrossRef]
- Badalzadeh, M.; Mazinani, M.; Pourpak, Z.; Heidarnazhad, H.; Mortaz, E.; Moin, M.; Farazmand, A. In Vitro Analysis of Nine MicroRNAs in CD8+ T Cells of Asthmatic Patients and the Effects of Two FDA-approved Drugs. Iran. J. Allergy Asthma Immunol. 2019, 18, 358–368. [Google Scholar] [CrossRef]
- Sharma, S. Immunomodulation: A definitive role of microRNA-142. Dev. Comp. Immunol. 2017, 77, 150–156. [Google Scholar] [CrossRef]
- Panganiban, R.P.; Pinkerton, M.H.; Maru, S.Y.; Jefferson, S.J.; Roff, A.N.; Ishmael, F.T. Differential microRNA epression in asthma and the role of miR-1248 in regulation of IL-5. Am. J. Clin. Exp. Immunol. 2012, 1, 154–165. [Google Scholar]
- Li, H.M.; Xiao, Y.J.; Min, Z.S.; Tan, C. Identification and interaction analysis of key genes and microRNAs in atopic dermatitis by bioinformatics analysis. Clin. Exp. Dermatol. 2019, 44, 257–264. [Google Scholar] [CrossRef]
- Wardzyńska, A.; Pawełczyk, M.; Rywaniak, J.; Kurowski, M.; Makowska, J.S.; Kowalski, M.L. Circulating MicroRNAs and T-Cell Cytokine Expression Are Associated With the Characteristics of Asthma Exacerbation. Allergy Asthma Immunol. Res. 2020, 12, 125–136. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, X.; Sun, S.; Yang, T.; Yang, J.; Wu, G.; Qiu, Y.; Yin, Y.; Xu, W. miR-29b Reverses T helper 1 cells/T helper 2 cells Imbalance and Alleviates Airway Eosinophils Recruitment in OVA-Induced Murine Asthma by Targeting Inducible Co-Stimulator. Int. Arch. Allergy Immunol. 2019, 180, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Jin, L.; Zhang, S.; Li, H.; Zhang, B.; Sun, M. MicroRNA-590-5p represses proliferation of human fetal airway smooth muscle cells by targeting signal transducer and activator of transcription 3. Arch. Med. Sci. 2018, 14, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yao, H.; Yi, B.; Kazama, K.; Liu, Y.; Deshpande, D.; Zhang, J.; Sun, J. MicroRNA-638 inhibits human airway smooth muscle cell proliferation and migration through targeting cyclin D1 and NOR1. J. Cell. Physiol. 2018, 234, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Zhan, J.W.; Jiao, D.M.; Wang, Y.; Song, J.; Wu, J.H.; Wu, L.J.; Chen, Q.Y.; Ma, S.L. Integrated microRNA and gene expression profiling reveals the crucial miRNAs in curcumin anti-lung cancer cell invasion. Thorac. Cancer 2017, 8, 461–470. [Google Scholar] [CrossRef]
- Li, C.G.; Pu, M.F.; Li, C.Z.; Gao, M.; Liu, M.X.; Yu, C.Z.; Yan, H.; Peng, C.; Zhao, Y.; Li, Y.; et al. MicroRNA-1304 suppresses human non-small cell lung cancer cell growth in vitro by targeting heme oxygenase-1. Acta Pharmacol. Sin. 2017, 38, 110–119. [Google Scholar] [CrossRef]
- Qu, J.; Li, M.; An, J.; Zhao, B.; Zhong, W.; Gu, Q.; Cao, L.; Yang, H.; Hu, C. MicroRNA-33b inhibits lung adenocarcinoma cell growth, invasion, and epithelial-mesenchymal transition by suppressing Wnt/β-catenin/ZEB1 signaling. Int. J. Oncol. 2015, 47, 2141–2152. [Google Scholar] [CrossRef]
- Xu, C.Z.; Shi, R.J.; Chen, D.; Sun, Y.Y.; Wu, Q.W.; Wang, T.; Wang, P.H. Potential biomarkers for paclitaxel sensitivity in hypopharynx cancer cell. Int. J. Clin. Exp. Pathol. 2013, 6, 2745–2756. [Google Scholar]
- Fesen, K.; Silveyra, P.; Fuentes, N.; Nicoleau, M.; Rivera, L.; Kitch, D.; Graff, G.R.; Siddaiah, R. The role of microRNAs in chronic pseudomonas lung infection in Cystic fibrosis. Respir. Med. 2019, 151, 133–138. [Google Scholar] [CrossRef]
- Lin, C.E.; Kaptein, J.S.; Sheikh, J. Differential expression of microRNAs and their possible roles in patients with chronic idiopathic urticaria and active hives. Allergy Rhinol. 2017, 8, 67–80. [Google Scholar] [CrossRef]
- Pan, K.; Wang, Y.; Pan, P.; Xu, G.; Mo, L.; Cao, L.; Wu, C.; Shen, X. The regulatory role of microRNA-mRNA co-expression in hepatitis B virus-associated acute liver failure. Ann. Hepatol. 2019, 18, 883–892. [Google Scholar] [CrossRef]
- Ono, K. A Novel Link between Plasma MicroRNA-33b Levels and Lipid Disorders in Diabetes Mellitus. J. Atheroscler. Thromb. 2016, 23, 1259–1260. [Google Scholar] [CrossRef] [PubMed]
- Januskevicius, A.; Vaitkiene, S.; Gosens, R.; Janulaityte, I.; Hoppenot, D.; Sakalauskas, R.; Malakauskas, K. Eosinophils enhance WNT-5a and TGF-β1 genes expression in airway smooth muscle cells and promote their proliferation by increased extracellular matrix proteins production in asthma. BMC Pulm. Med. 2016, 16, 94. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Li, J.; Huang, B.; Liu, J.; Chen, X.; Chen, X.M.; Xu, Y.M.; Huang, L.F.; Wang, X.Z. Exosomes: Novel biomarkers for clinical diagnosis. Sci. World J. 2015, 2015, 657086. [Google Scholar] [CrossRef] [PubMed]
- Tohidinik, H.R.; Mallah, N.; Takkouche, B. History of allergic rhinitis and risk of asthma; a systematic review and meta-analysis. World Allergy Organ. J. 2019, 12, 100069. [Google Scholar] [CrossRef]
- Gon, Y.; Hashimoto, S. Role of airway epithelial barrier dysfunction in pathogenesis of asthma. Allergol Int. 2018, 67, 12–17. [Google Scholar] [CrossRef]
- Rosati, M.G.; Peters, A.T. Relationships among allergic rhinitis, asthma, and chronic rhinosinusitis. Am. J. Rhinol. Allergy 2016, 30, 44–47. [Google Scholar] [CrossRef]
- Boonpiyathad, T.; Sözener, Z.C.; Satitsuksanoa, P.; Akdis, C.A. Immunologic mechanisms in asthma. Semin. Immunol. 2019, 46, 101333. [Google Scholar] [CrossRef]
- Simard, M.; Madore, A.M.; Girard, S.; Waserman, S.; Duan, Q.; Subbarao, P.; Sears, M.R.; Moraes, T.J.; Becker, A.B.; Turvey, S.E.; et al. Polygenic risk score for atopic dermatitis in the Canadian population. J. Allergy Clin. Immunol. 2020. [Google Scholar] [CrossRef]
- American Thoracic Society. Standardization of Spirometry, 1994 Update. Am. J. Respir. Crit. Care Med. 1995, 152, 1107–1136. [Google Scholar] [CrossRef]
- Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease (COPD) and asthma. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, November 1986. Am. Rev. Respir. Dis. 1987, 136, 225–244. [Google Scholar] [CrossRef]
- Laprise, C. The Saguenay-Lac-Saint-Jean asthma familial collection: The genetics of asthma in a young founder population. Genes Immun. 2014, 15, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Madore, A.M.; Pain, L.; Boucher-Lafleur, A.M.; Morin, A.; Meloche, J.; Simon, M.M.; Ge, B.; Kwan, T.; Cheung, W.A.; Pastinen, T.; et al. Asthma-associated polymorphisms in 17q12-21 locus modulate methylation and gene expression of GSDMA in naïve CD4(+) T cells. J. Genet. Genom. 2020, 47, 171–174. [Google Scholar] [CrossRef]
- Houseman, E.A.; Accomando, W.P.; Koestler, D.C.; Christensen, B.C.; Marsit, C.J.; Nelson, H.H.; Wiencke, J.K.; Kelsey, K.T. DNA methylation arrays as surrogate measures of cell mixture distribution. BMC Bioinform. 2012, 13, 86. [Google Scholar] [CrossRef] [PubMed]
- Reinius, L.E.; Acevedo, N.; Joerink, M.; Pershagen, G.; Dahlén, S.E.; Greco, D.; Söderhäll, C.; Scheynius, A.; Kere, J. Differential DNA methylation in purified human blood cells: Implications for cell lineage and studies on disease susceptibility. PLoS ONE 2012, 7, e41361. [Google Scholar] [CrossRef] [PubMed]
- Leek, J.T.; Storey, J.D. Capturing heterogeneity in gene expression studies by surrogate variable analysis. PLoS Genet. 2007, 3, 1724–1735. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, F.; Keenan, J.; Aherne, S.; O’Neill, F.; Clarke, C.; Henry, M.; Meleady, P.; Breen, L.; Barron, N.; Clynes, M.; et al. Parallel mRNA, proteomics and miRNA expression analysis in cell line models of the intestine. World J. Gastroenterol. 2017, 23, 7369–7386. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).