New 1,3,4-Oxadiazole Derivatives of Pyridothiazine-1,1-Dioxide with Anti-Inflammatory Activity
Abstract
:1. Introduction
2. Results and Discussion
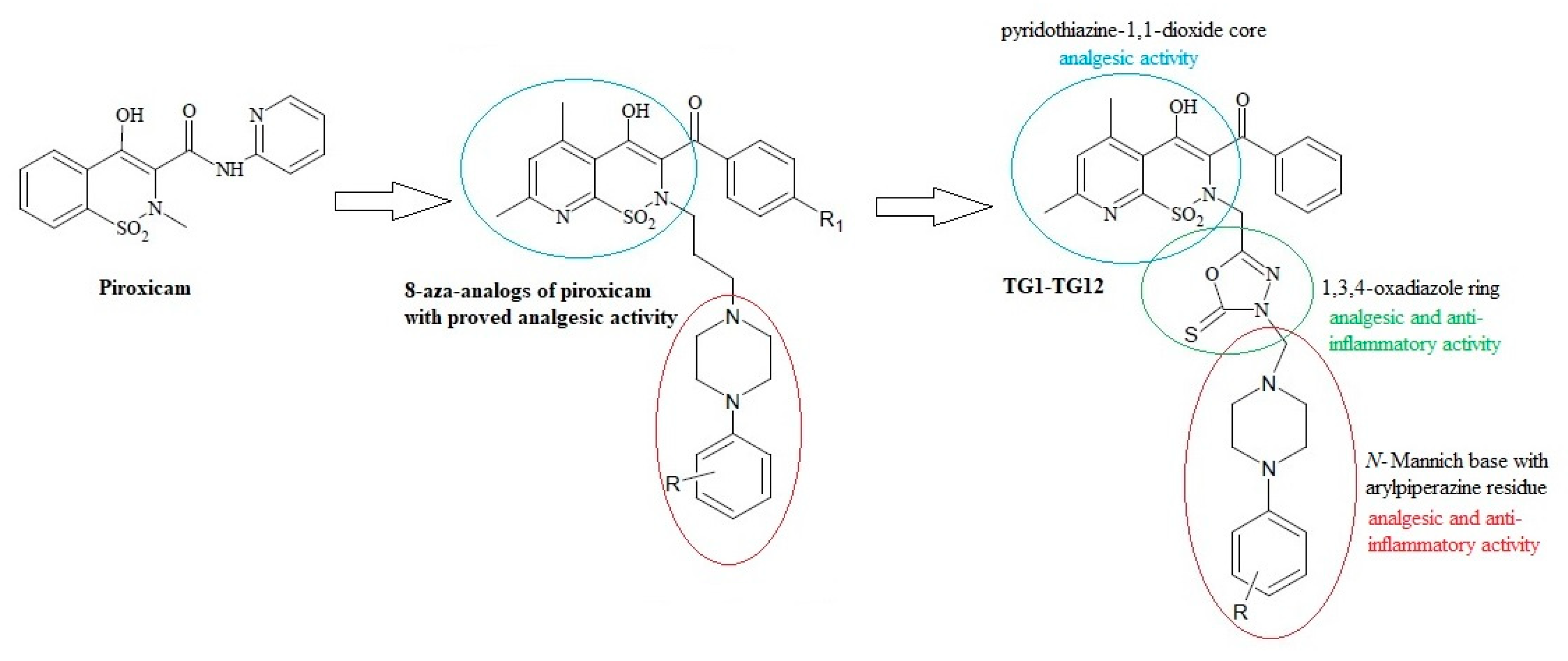
2.1. Chemistry
Synthesis of New 1,3,4-Oxadiazole Derivatives of Pyridothiazine-1,1-Dioxide
2.2. Biological Tests
2.2.1. Cyclooxygenase Inhibition
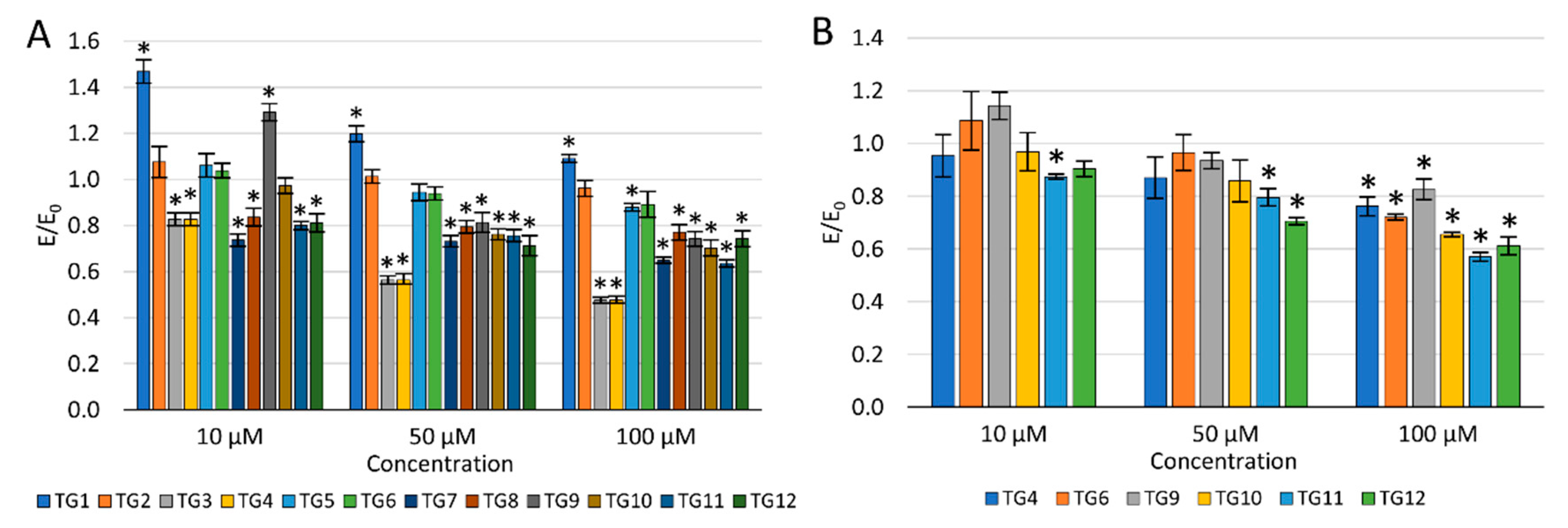
2.2.2. Viability of Cell Cultures
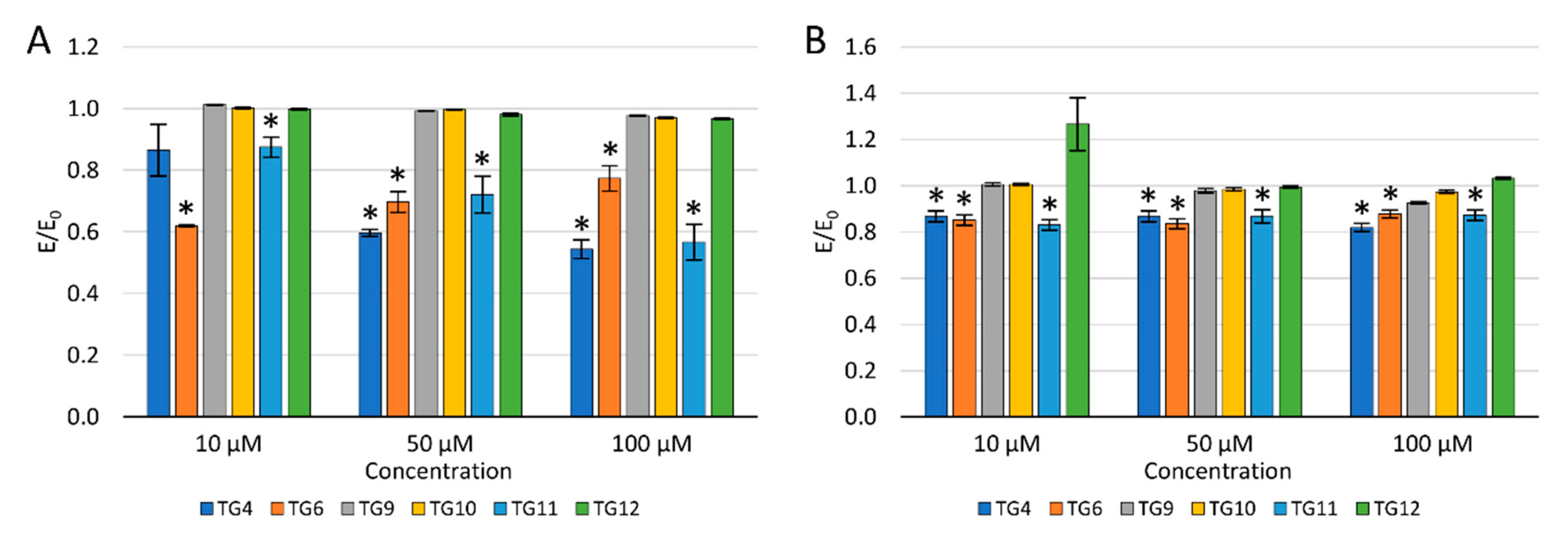
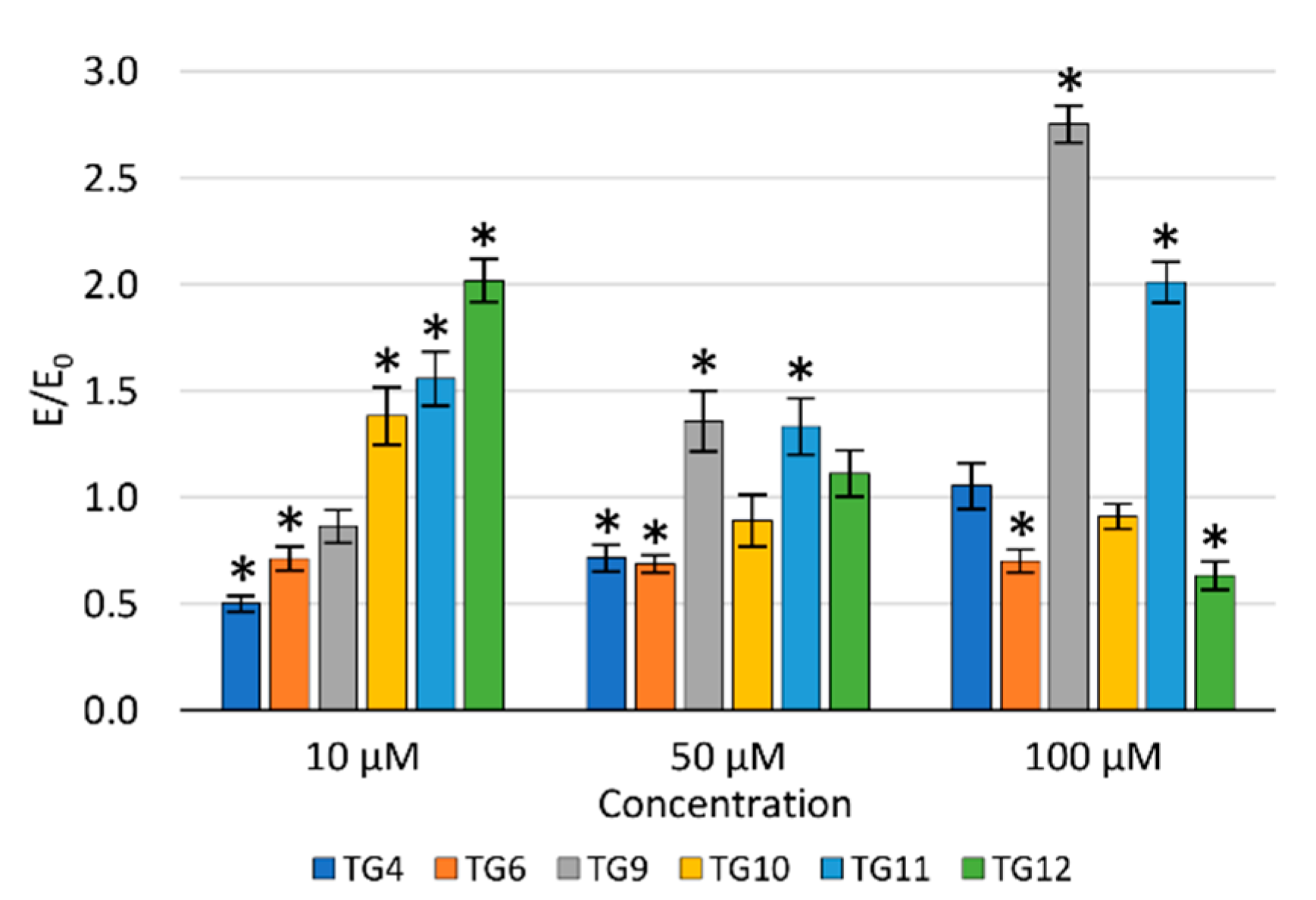
2.2.3. Reactive Oxygen Species and Nitric Oxide
2.2.4. DNA Damage
2.2.5. Multiple-Criteria Decision Analysis
2.3. Molecular Docking Study
3. Material and Methods
3.1. Chemistry
3.1.1. Chemicals
3.1.2. Preparation and Experimental Properties of Compounds 2, 3, and TG1-TG12
Benzoyl-4-Hydroxy-5,7-Dimethyl-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide-2-Acetic Acid Hydrazide 2
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[5-Tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide 3
Mannich Bases TG1-TG12
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-Phenyl-1-Piperazinyl)Methyl)-5-tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG1
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(4-Methylphenyl)-1-Piperazinyl)Methyl)-5-tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG2
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(2-Methoxyphenyl)-1-Piperazinyl)Methyl)-5-tioxo-1,3,4-Oxadiazol-2-ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG3
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(3-Triflouomethylphenyl)-1-Piperazinyl)Methyl)-5-Tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG4
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(2-Fluorophenyl)-1-Piperazinyl)Methyl)-5-tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG5
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(2,4-Difluorophenyl)-1-Piperazinyl)Methyl)-5-Tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG6
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(4-Bromophenyl)-1-Piperazinyl)Methyl)-5-Tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG7
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(2,4-Dichlorophenyl)-1-Piperazinyl)Methyl)-5-tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG8
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(2-cyanophenyl)-1-Piperazinyl)Methyl)-5-Tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG9
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(4-Nitrophenyl)-1-Piperazinyl)Methyl)-5-tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG10
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(2-Pyridyl)-1-Piperazinyl)Methyl)-5-Tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG11
3-Benzoyl-4-Hydroxy-5,7-Dimethyl-2-{[4-((4-(2-Pyrimidyl)-1-Piperazinyl)Methyl)-5-Tioxo-1,3,4-Oxadiazol-2-Ilo]Methyl}-2H-Pyrido[3,2-e]-1,2-Thiazine-1,1-Dioxide TG12
3.2. Biological Section
3.2.1. Cell Line
3.2.2. Tested Compounds
3.2.3. Experimental Design
3.2.4. Sulforhodamine B (SRB) Assay
3.2.5. Reactive Oxygen Species and Nitric Oxide
3.2.6. Fast-Halo Assay
3.2.7. Cyclooxygenase Inhibition Assay
3.2.8. Statistical Analysis
3.3. Molecular Modeling
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kirkby, N.S.; Chan, M.V.; Zaiss, A.K.; Garcia-Vaz, E.; Jiao, J.; Berglund, L.M.; Verdu, E.F.; Ahmetaj-Shala, B.; Wallace, J.L.; Herschman, H.R.; et al. Systematic study of constitutive cyclooxygenase-2 expression: Role of NF-κB and NFAT transcriptional pathways. Proc. Natl. Acad. Sci. USA 2016, 113, 434–439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kam, P.C.A.; See, A.U.-L. Cyclo-oxygenase isoenzymes: Physiological and pharmacological role. Anaesthesia 2000, 55, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, P.; Tacconelli, S.; Bruno, A.; Sostres, C.; Lanas, A. Managing the adverse effects of nonsteroidal anti-inflammatory drugs. Expert Rev. Clin. Pharmacol. 2011, 4, 605–621. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, C. COX-1 and COX-2 inhibitors. Best Pr. Res. Clin. Gastroenterol. 2001, 15, 801–820. [Google Scholar] [CrossRef] [PubMed]
- Blobaum, A.L.; Marnett, L.J. Structural and Functional Basis of Cyclooxygenase Inhibition. J. Med. Chem. 2007, 50, 1425–1441. [Google Scholar] [CrossRef] [Green Version]
- Ricciotti, E.; Fitzgerald, G.A. Prostaglandins and Inflammation. Arter. Thromb. Vasc. Biol. 2011, 31, 986–1000. [Google Scholar] [CrossRef]
- Süleyman, H.; Demircan, B.; Karagöz, Y. Anti-inflammatory and side effects of cyclooxygenase inhibitors. Pharmacol. Rep. 2007, 59, 247–258. [Google Scholar]
- Vardeny, O.; Solomon, S.D. Cyclooxygenase-2 Inhibitors, Nonsteroidal Anti-inflammatory Drugs, and Cardiovascular Risk. Cardiol. Clin. 2008, 26, 589–601. [Google Scholar] [CrossRef]
- Parada, B.; Sereno, J.; Reis, F.; Teixeira, F.; Garrido, P.; Pinto, A.F.; Cunha, M.F.; Pinto, R.M.A.; Mota, A.; Figueiredo, A.; et al. Anti-inflammatory, anti-proliferative and antioxidant profiles of selective cyclooxygenase-2 inhibition as chemoprevention for rat bladder carcinogenesis. Cancer Biol. Ther. 2009, 8, 1615–1622. [Google Scholar] [CrossRef] [Green Version]
- Sereno, J.; Parada, B.; Reis, F.; Cunha, F.X.; Teixeira, F.; Garrido, P.; Pinto, R.M.A.; Rocha-Pereira, P.; Neto, P.; Ruivo, J.; et al. Preventive but Not Curative Efficacy of Celecoxib on Bladder Carcinogenesis in a Rat Model. Mediat. Inflamm. 2011, 2010, 1–11. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F.R. Cancer-related inflammation. Nat. Cell Biol. 2008, 454, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Schetter, A.J.; Heegaard, N.H.H.; Harris, C.C. Inflammation and cancer: Interweaving microRNA, free radical, cytokine and p53 pathways. Carcinogenesis 2010, 31, 37–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGarry, T.; Biniecka, M.; Veale, D.J.; Fearon, U. Hypoxia, oxidative stress and inflammation. Free. Radic. Biol. Med. 2018, 125, 15–24. [Google Scholar] [CrossRef]
- Rathore, A.; Rahman, M.U.; Siddiqui, A.A.; Ali, A.; Shaharyar, M. Design and Synthesis of Benzimidazole Analogs Endowed with Oxadiazole as Selective COX-2 Inhibitor. Arch. Pharm. 2014, 347, 923–935. [Google Scholar] [CrossRef]
- Banerjee, A.G.; Das, N.; Shengule, S.A.; Sharma, P.A.; Srivastava, R.S.; Shrivastava, S.K. Design, synthesis, evaluation and molecular modelling studies of some novel 5,6-diphenyl-1,2,4-triazin-3(2H)-ones bearing five-member heterocyclic moieties as potential COX-2 inhibitors: A hybrid pharmacophore approach. Bioorg. Chem. 2016, 69, 102–120. [Google Scholar] [CrossRef]
- Boström, J.; Hogner, A.; Llinàs, A.; Wellner, E.; Plowright, A.T. Oxadiazoles in Medicinal Chemistry. J. Med. Chem. 2012, 55, 1817–1830. [Google Scholar] [CrossRef]
- Tresse, C.; Radigue, R.; Von Borowski, R.G.; Thepaut, M.; Le, H.H.; DeMay, F.; Georgeault, S.; Dhalluin, A.; Trautwetter, A.; Ermel, G.; et al. Synthesis and evaluation of 1,3,4-oxadiazole derivatives for development as broad-spectrum antibiotics. Bioorg. Med. Chem. 2019, 27, 115097. [Google Scholar] [CrossRef] [PubMed]
- Telehoiu, A.T.B.; Nuță, D.C.; Căproiu, M.T.; Dumitrascu, F.; Zarafu, I.; Ionita, P.; Bădiceanu, C.D.; Avram, S.; Chifiriuc, M.C.; Bleotu, C.; et al. Design, Synthesis and In Vitro Characterization of Novel Antimicrobial Agents Based on 6-Chloro-9H-carbazol Derivatives and 1,3,4-Oxadiazole Scaffolds. Molecules 2020, 25, 266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levent, S.; Çavuşoğlu, B.K.; Sağlık, B.N.; Osmaniye, D.; Çevik, U.A.; Atlı-Eklioğlu, Ö.; Özkay, Y.; Kaplancıklı, Z.A. Synthesis of Oxadiazole-Thiadiazole Hybrids and Their Anticandidal Activity. Molecules 2017, 22, 2004. [Google Scholar] [CrossRef] [Green Version]
- Song, Z.-L.; Zhu, Y.; Liu, J.-R.; Guo, S.-K.; Gu, Y.-C.; Han, X.-Y.; Dong, H.-Q.; Sun, Q.; Zhang, W.-H.; Zhang, M.-Z. Diversity-oriented synthesis and antifungal activities of novel pimprinine derivative bearing a 1,3,4-oxadiazole-5-thioether moiety. Mol. Divers. 2020, 1–17. [Google Scholar] [CrossRef]
- Sonawane, A.D.; Rode, N.D.; Nawale, L.; Joshi, R.R.; Joshi, R.A.; Likhite, A.P.; Sarkar, D. Synthesis and biological evaluation of 1,2,4-triazole-3-thione and 1,3,4-oxadiazole-2-thione as antimycobacterial agents. Chem. Biol. Drug Des. 2017, 90, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Karabanovich, G.; Němeček, J.; Valášková, L.; Carazo, A.; Konečná, K.; Stolaříková, J.; Hrabálek, A.; Pavliš, O.; Pávek, P.; Vávrová, K.; et al. S-substituted 3,5-dinitrophenyl 1,3,4-oxadiazole-2-thiols and tetrazole-5-thiols as highly efficient antitubercular agents. Eur. J. Med. Chem. 2017, 126, 369–383. [Google Scholar] [CrossRef] [PubMed]
- Gan, X.; Hu, D.; Chen, Z.; Wang, Y.; Song, B. Synthesis and antiviral evaluation of novel 1,3,4-oxadiazole/thiadiazole-chalcone conjugates. Bioorg. Med. Chem. Lett. 2017, 27, 4298–4301. [Google Scholar] [CrossRef] [PubMed]
- Glomb, T.; Szymankiewicz, K.; Świątek, P. Anti-Cancer Activity of Derivatives of 1,3,4-Oxadiazole. Molecules 2018, 23, 3361. [Google Scholar] [CrossRef] [Green Version]
- Ananth, A.H.; Manikandan, N.; Rajan, R.K.; Elancheran, R.; Lakshmithendral, K.; Ramanathan, M.; Bhattacharjee, A.; Kabilan, S. Design, Synthesis, and Biological Evaluation of 2-(2-Bromo-3-nitrophenyl)-5-phenyl-1,3,4-oxadiazole Derivatives as Possible Anti-Breast Cancer Agents. Chem. Biodivers. 2020, 17, e1900659. [Google Scholar] [CrossRef]
- Shyma, C.P.; Balakrishna, K.; Peethambar, K.S.; Vijesh, M.A. Synthesis, Characterization, Antidiabetic and Antioxidant Activity of 1,3,4-Oxadiazole Derivatives Bearing 6-Methyl Pyridine Moiety. Der Pharma Chem. 2015, 7, 137–145. [Google Scholar]
- Kaur, J.; Soto-Velasquez, M.; Ding, Z.; Ghanbarpour, A.; Lill, M.A.; Van Rijn, R.M.; Watts, V.J.; Flaherty, D.P. Optimization of a 1,3,4-oxadiazole series for inhibition of Ca2+/calmodulin-stimulated activity of adenylyl cyclases 1 and 8 for the treatment of chronic pain. Eur. J. Med. Chem. 2019, 162, 568–585. [Google Scholar] [CrossRef]
- Banerjee, A.G.; Das, N.; Shengule, S.A.; Srivastava, R.S.; Shrivastava, S.K. Synthesis, characterization, evaluation and molecular dynamics studies of 5, 6–diphenyl–1,2,4–triazin–3(2 H )–one derivatives bearing 5–substituted 1,3,4–oxadiazole as potential anti–inflammatory and analgesic agents. Eur. J. Med. Chem. 2015, 101, 81–95. [Google Scholar] [CrossRef]
- Akhter, M.; Husain, A.; Azad, B.; Ajmal, M. Aroylpropionic acid based 2,5-disubstituted-1,3,4-oxadiazoles: Synthesis and their anti-inflammatory and analgesic activities. Eur. J. Med. Chem. 2009, 44, 2372–2378. [Google Scholar] [CrossRef]
- Bansal, S.; Bala, M.; Suthar, S.K.; Choudhary, S.; Bhattacharya, S.; Bhardwaj, V.; Singla, S.; Joseph, A. Design and synthesis of novel 2-phenyl-5-(1,3-diphenyl-1H-pyrazol-4-yl)-1,3,4-oxadiazoles as selective COX-2 inhibitors with potent anti-inflammatory activity. Eur. J. Med. Chem. 2014, 80, 167–174. [Google Scholar] [CrossRef]
- Gulnaz, A.R.; Mohammed, Y.H.; Khanum, S.A. Design, synthesis and molecular docking of benzophenone conjugated with oxadiazole sulphur bridge pyrazole pharmacophores as anti inflammatory and analgesic agents. Bioorg. Chem. 2019, 92, 103220. [Google Scholar] [CrossRef] [PubMed]
- Grover, J.; Bhatt, N.; Kumar, V.; Patel, N.K.; Gondaliya, B.J.; Sobhia, M.E.; Bhutani, K.K.; Jachak, S.M. 2,5-Diaryl-1,3,4-oxadiazoles as selective COX-2 inhibitors and anti-inflammatory agents. RSC Adv. 2015, 5, 45535–45544. [Google Scholar] [CrossRef]
- Bhandari, S.V.; Bothara, K.G.; Raut, M.K.; Patil, A.A.; Sarkate, A.P.; Mokale, V.J. Design, Synthesis and Evaluation of Antiinflammatory, Analgesic and Ulcerogenicity studies of Novel S-Substituted phenacyl-1,3,4-oxadiazole-2-thiol and Schiff bases of Diclofenac acid as Nonulcerogenic Derivatives. Bioorg. Med. Chem. 2008, 16, 1822–1831. [Google Scholar] [CrossRef] [PubMed]
- Amir, M.; Kumar, H.; Javed, S.A. Non-carboxylic Analogues of Naproxen: Design, Synthesis, and Pharmacological Evaluation of some 1,3,4-Oxadiazole/Thiadiazole and 1,2,4-Triazole Derivatives. Arch. Pharm. 2007, 340, 577–585. [Google Scholar] [CrossRef]
- Bala, S.; Kamboj, S.; Saini, V.; Prasad, D.N. Anti-Inflammatory, Analgesic Evaluation and Molecular Docking Studies of N-Phenyl Anthranilic Acid-Based 1,3,4-Oxadiazole Analogues. J. Chem. 2013, 2013, 1–6. [Google Scholar] [CrossRef] [Green Version]
- PalkarMalleshappa, M.B.; Singhai, A.S.; Ronad, P.M.; Vishwanathswamy, A.; Boreddy, T.S.; Veerapur, V.P.; Shaikh, M.S.; Rane, R.A.; Karpoormath, R. Synthesis, pharmacological screening and in silico studies of new class of Diclofenac analogues as a promising anti-inflammatory agents. Bioorg. Med. Chem. 2014, 22, 2855–2866. [Google Scholar] [CrossRef]
- Malinka, W.; Kaczmarz, M.; Filipek, B.; Sapa, J.; Glod, B. Preparation of novel derivatives of pyridothiazine-1,1-dioxide and their CNS and antioxidant properties. Farmaco 2002, 57, 737–746. [Google Scholar] [CrossRef]
- Saeed, M.M.; Khalil, N.A.; Ahmed, E.M.; Eissa, K.I. Synthesis and anti-inflammatory activity of novel pyridazine and pyridazinone derivatives as non-ulcerogenic agents. Arch. Pharmacal Res. 2012, 35, 2077–2092. [Google Scholar] [CrossRef]
- Hatnapure, G.D.; Keche, A.P.; Rodge, A.H.; Birajdar, S.S.; Tale, R.H.; Kamble, V.M. Synthesis and biological evaluation of novel piperazine derivatives of flavone as potent anti-inflammatory and antimicrobial agent. Bioorg. Med. Chem. Lett. 2012, 22, 6385–6390. [Google Scholar] [CrossRef]
- Dündar, Y.; Gökçe, M.; Kupeli, E.; Sahin, M. Synthesis and Analgesic and Anti-inflammatory Activity of Ethyl (6-Substituted-3(2H)-pyridazinone-2-yl)acetate Derivatives. Arzneimittelforschung 2011, 57, 777–781. [Google Scholar] [CrossRef]
- Li, J.; Li, D.; Xu, Y.; Guo, Z.; Liu, X.; Yang, H.; Wu, L.; Wang, L. Design, synthesis, biological evaluation, and molecular docking of chalcone derivatives as anti-inflammatory agents. Bioorg. Med. Chem. Lett. 2017, 27, 602–606. [Google Scholar] [CrossRef] [PubMed]
- Nimbalkar, U.D.; Tupe, S.; Seijas, J.A.; Khan, F.A.K.; Sangshetti, J.N.; Nikalje, A.P.G. Ultrasound- and Molecular Sieves-Assisted Synthesis, Molecular Docking and Antifungal Evaluation of 5-(4-(Benzyloxy)-substituted phenyl)-3-((phenylamino)methyl)-1,3,4-oxadiazole-2(3H)-thiones. Molecules 2016, 21, 484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aggarwal, N.; Kumar, R.; Dureja, P.; Khurana, J.M. Synthesis of Novel Nalidixic Acid-Based 1,3,4-Thiadiazole and 1,3,4-Oxadiazole Derivatives as Potent Antibacterial Agents. Chem. Biol. Drug Des. 2012, 79, 384–397. [Google Scholar] [CrossRef] [PubMed]
- Bhutani, R.; Pathak, D.P.; Kapoor, G.; Husain, A.; Kant, R.; Iqbal, A. Synthesis, molecular modelling studies and ADME prediction of benzothiazole clubbed oxadiazole-Mannich bases, and evaluation of their anti-diabetic activity through in vivo model. Bioorg. Chem. 2018, 77, 6–15. [Google Scholar] [CrossRef]
- Caneschi, W.; Enes, K.B.; De Mendonça, C.C.; Fernandes, F.D.S.; Miguel, F.B.; Martins, J.S.; Le Hyaric, M.; Pinho, R.R.; Duarte, L.M.; De Oliveira, M.A.L.; et al. Synthesis and anticancer evaluation of new lipophilic 1,2,4 and 1,3,4-oxadiazoles. Eur. J. Med. Chem. 2019, 165, 18–30. [Google Scholar] [CrossRef]
- Bajaj, S.; Roy, P.P.; Singh, J. Synthesis, thymidine phosphorylase inhibitory and computational study of novel 1,3,4-oxadiazole-2-thione derivatives as potential anticancer agents. Comput. Biol. Chem. 2018, 76, 151–160. [Google Scholar] [CrossRef]
- Koksal, M.; Ozkan-Dagliyan, I.; Ozyazici, T.; Kadioglu, B.; Sipahi, H.; Bozkurt, A.; Bilge, S.S. Some Novel Mannich Bases of 5-(3,4-Dichlorophenyl)-1,3,4-oxadiazole-2(3H )-one and Their Anti-Inflammatory Activity. Arch. der Pharm. 2017, 350, 1700153. [Google Scholar] [CrossRef]
- Manjunatha, K.; Poojary, B.; Lobo, P.L.; Fernandes, J.; Kumari, N.S. Synthesis and biological evaluation of some 1,3,4-oxadiazole derivatives. Eur. J. Med. Chem. 2010, 45, 5225–5233. [Google Scholar] [CrossRef]
- Viveka, S.; Shama, P.; Nagaraja, G.K.; Deepa, N.; Sreenivasa, M.Y. Design, synthesis, and pharmacological studies of some new Mannich bases and S-alkylated analogs of pyrazole integrated 1,3,4-oxadiazole. Res. Chem. Intermed. 2015, 42, 2597–2617. [Google Scholar] [CrossRef]
- Zawisza, T.; Malinka, W. Synthesis and properties of some derivatives of 2H-4,6-dimethylpyrido[3,2-d]isothiazolin-3-one-1,1-dioxide. Farm. Ed. Sci. 1986, 41, 676–683. [Google Scholar] [CrossRef]
- Zawisza, T.; Malinka, W. A novel system: 2H-pyrido[3,2-e]-1,2-thiazine-1,1-dioxide. Synthesis and properties of some derivatives. Farm. Ed. Sci. 1986, 41, 819–826. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, S.; Hermanson, D.J.; Banerjee, S.; Ghebreselasie, K.; Clayton, G.M.; Garavito, R.M.; Marnett, L.J. Oxicams Bind in a Novel Mode to the Cyclooxygenase Active Site via a Two-water-mediated H-bonding Network. J. Biol. Chem. 2014, 289, 6799–6808. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gautam, R.; Jachak, S.M.; Kumar, V.; Mohan, C.G. Synthesis, biological evaluation and molecular docking studies of stellatin derivatives as cyclooxygenase (COX-1, COX-2) inhibitors and anti-inflammatory agents. Bioorg. Med. Chem. Lett. 2011, 21, 1612–1616. [Google Scholar] [CrossRef] [PubMed]
- Świątek, P.; Strzelecka, M.; Urniaz, R.; Gębczak, K.; Gębarowski, T.; Gąsiorowski, K.; Malinka, W. Synthesis, COX-1/2 inhibition activities and molecular docking study of isothiazolopyridine derivatives. Bioorg. Med. Chem. 2017, 25, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Gierse, J.K.; McDonald, J.J.; Hauser, S.D.; Rangwala, S.H.; Koboldt, C.M.; Seibert, K. A Single Amino Acid Difference between Cyclooxygenase-1 (COX-1) and −2 (COX-2) Reverses the Selectivity of COX-2 Specific Inhibitors. J. Biol. Chem. 1996, 271, 15810–15814. [Google Scholar] [CrossRef] [Green Version]
- Wakulik, K.; Wiatrak, B.; Szczukowski, Ł.; Bodetko, D.; Szandruk, M.; Dobosz, A.; Świątek, P.; Gąsiorowski, K. Effect of Novel Pyrrolo[3,4-d]pyridazinone Derivatives on Lipopolysaccharide-Induced Neuroinflammation. Int. J. Mol. Sci. 2020, 21, 2575. [Google Scholar] [CrossRef] [Green Version]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef] [Green Version]
- Vosko, S.H.; Wilk, L.; Nusair, M. Accurate spin-dependent electron liquid correlation energies for local spin density calculations: A critical analysis. Can. J. Phys. 1980, 58, 1200–1211. [Google Scholar] [CrossRef] [Green Version]
- Stephens, P.J.; Devlin, F.J.; Chabalowski, C.F.; Frisch, M.J. Ab Initio Calculation of Vibrational Absorption and Circular Dichroism Spectra Using Density Functional Force Fields. J. Phys. Chem. 1994, 98, 11623–11627. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum Mechanical Continuum Solvation Models. Chem. Rev. 2005, 105, 2999–3094. [Google Scholar] [CrossRef] [PubMed]
- Tomasi, J.; Mennucci, B.; Cancès, E. The IEF version of the PCM solvation method: An overview of a new method addressed to study molecular solutes at the QM ab initio level. J. Mol. Struct. THEOCHEM 1999, 464, 211–226. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Morris, G.M.; Ruth, H.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [Green Version]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, A.C.; Laskowski, R.A.; Thornton, J.M. LIGPLOT: A program to generate schematic diagrams of protein-ligand interactions. Protein Eng. Des. Sel. 1995, 8, 127–134. [Google Scholar] [CrossRef] [PubMed]

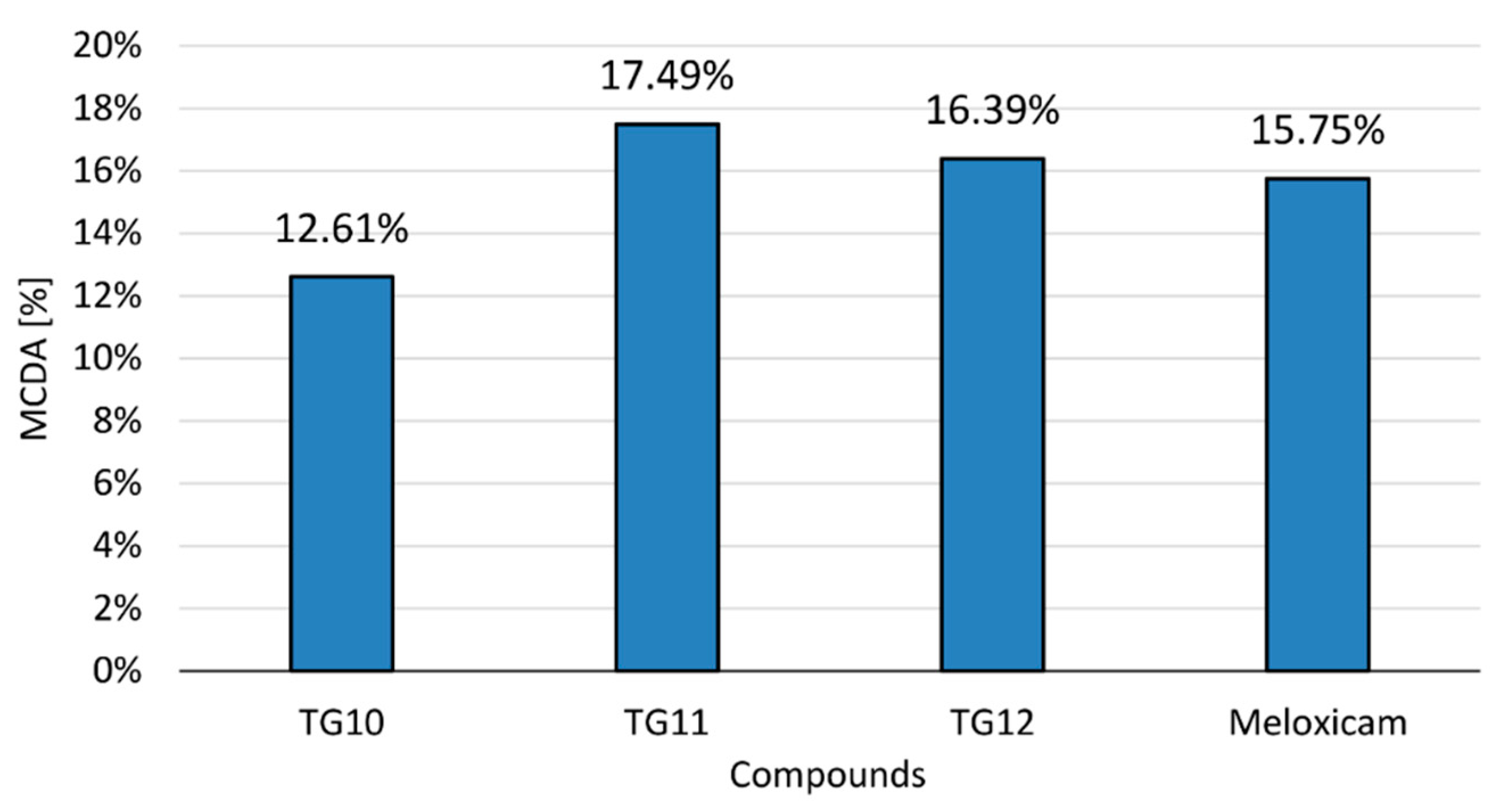
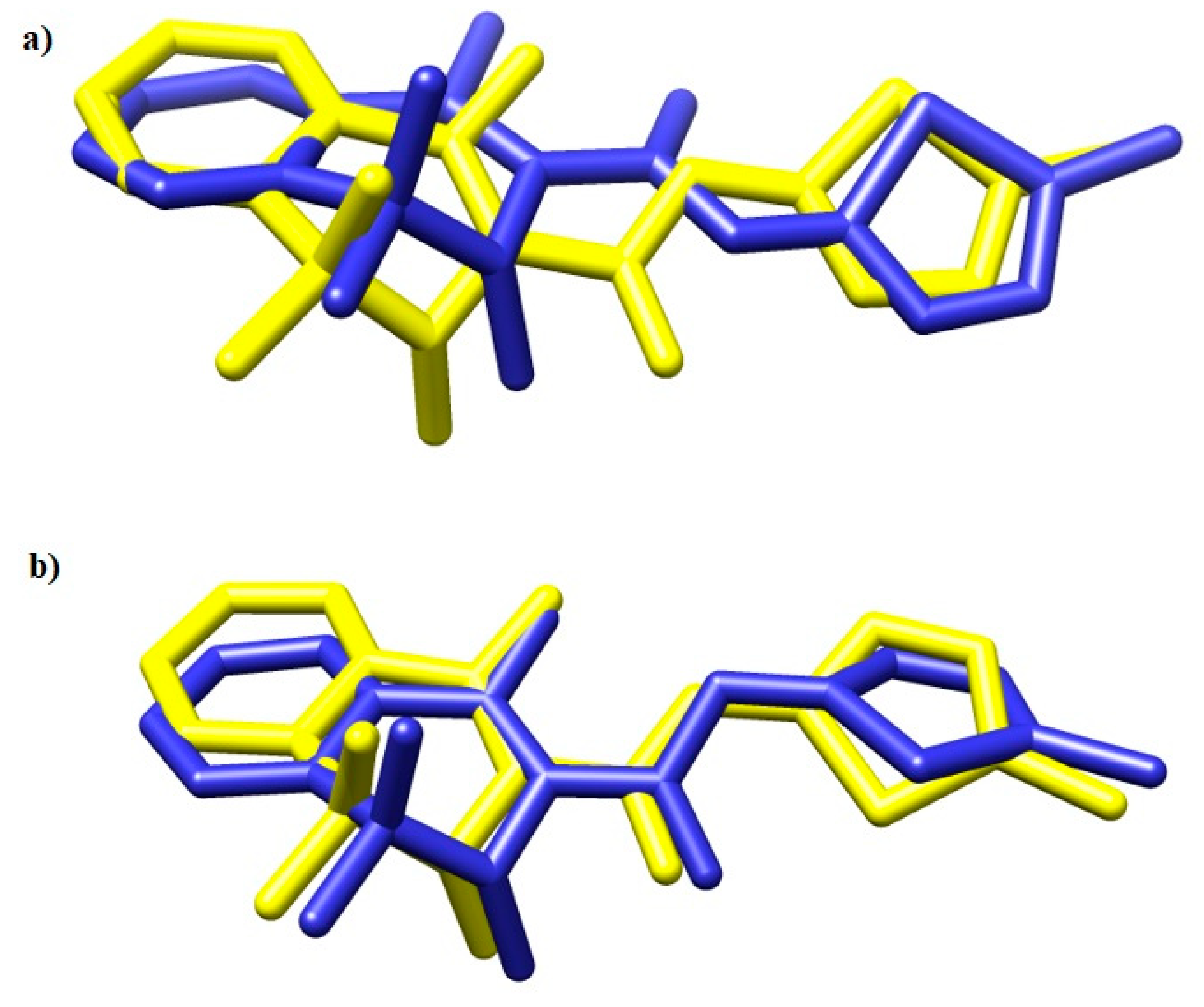
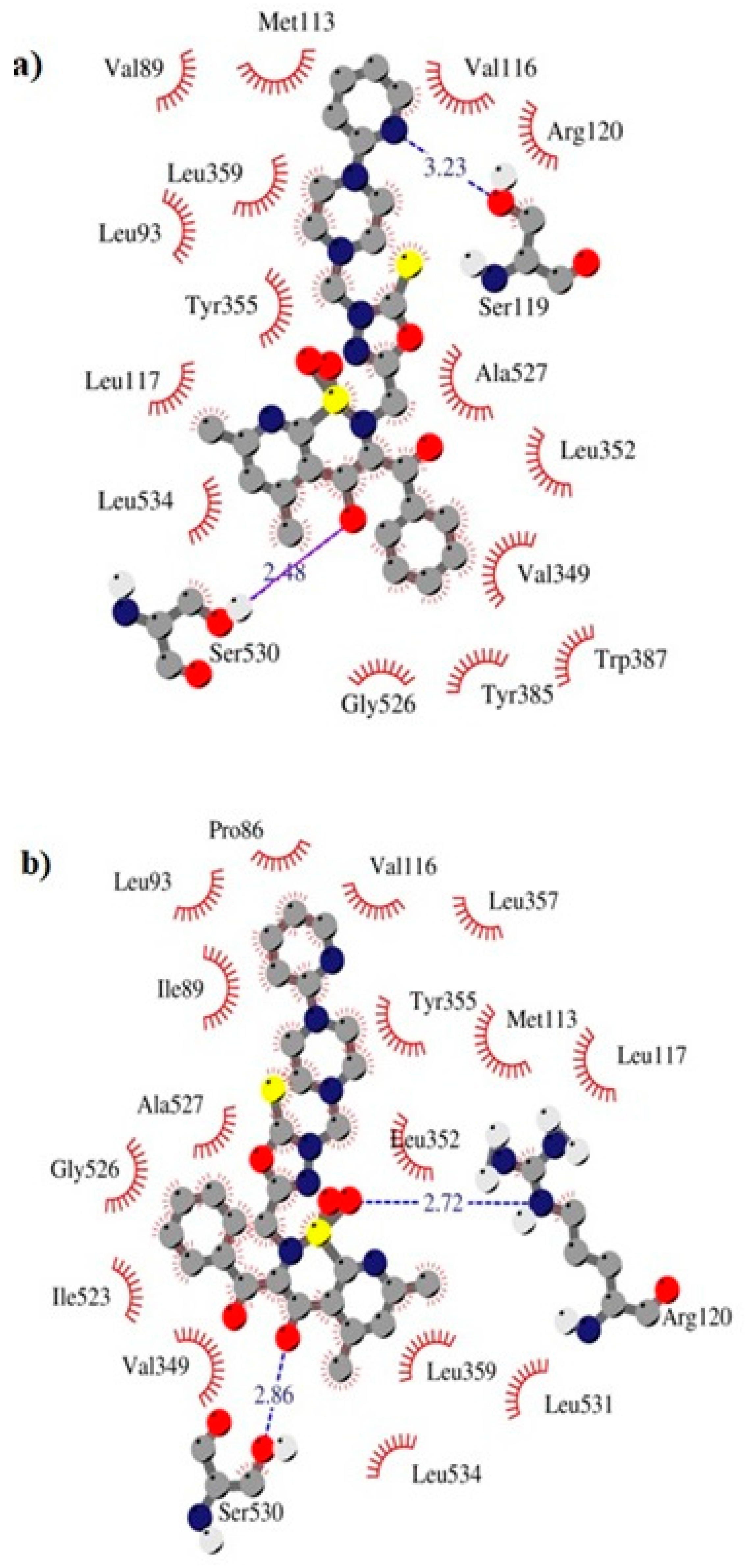
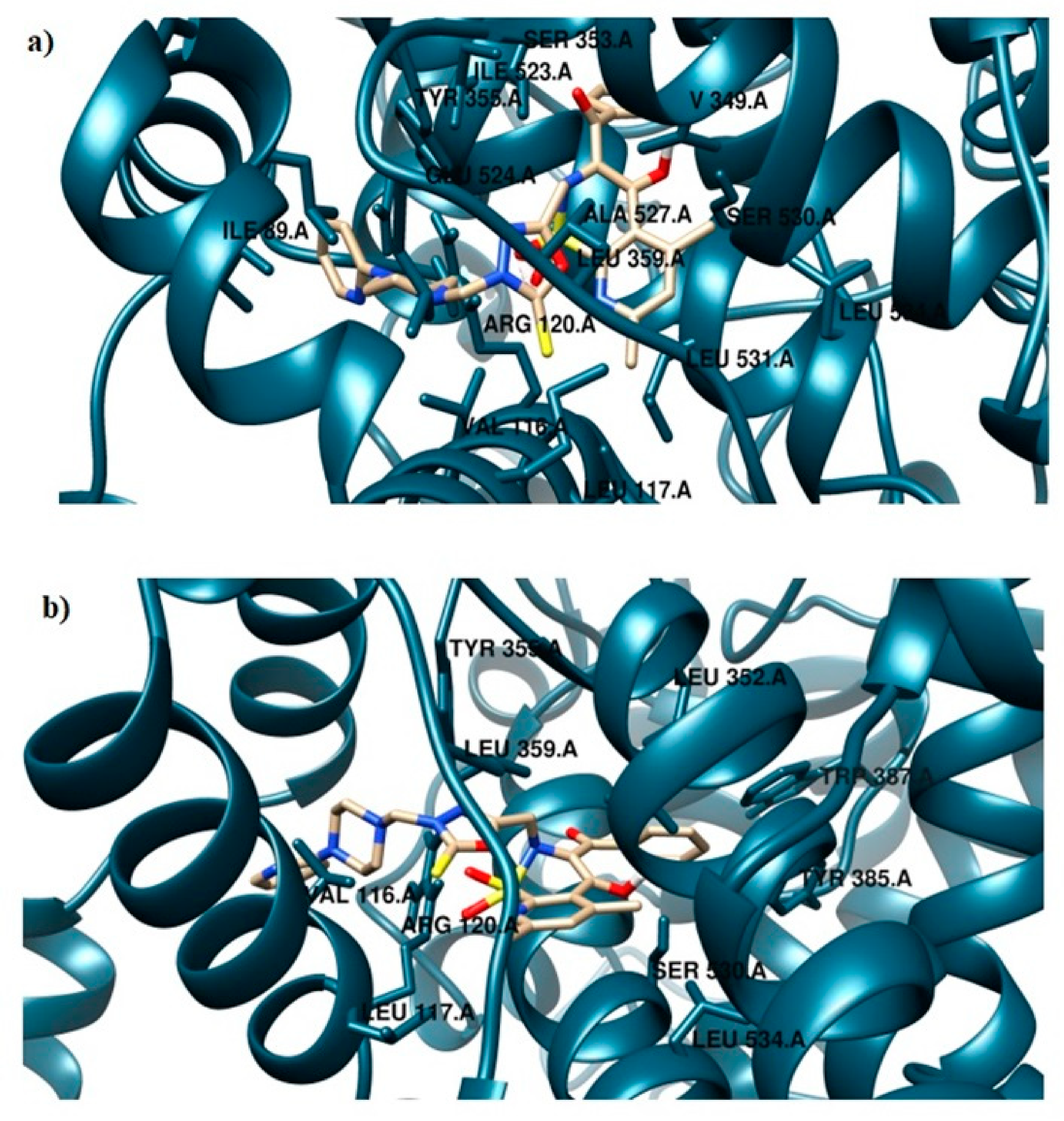
| Compound | COX Inhibition at a Concentration of 100 μM | COX Selectivity Ratio %inh. COX-2/%inh. COX-1 | |
|---|---|---|---|
| COX-1 | COX-2 | ||
| TG1 | N/A | N/A | - |
| TG2 | N/A | N/A | - |
| TG3 | N/A | N/A | - |
| TG4 | N/A | 41.60% | - |
| TG5 | N/A | N/A | - |
| TG6 | N/A | 24.43% | - |
| TG7 | N/A | N/A | - |
| TG8 | N/A | N/A | - |
| TG9 | 1.20% | N/A | - |
| TG10 | 42.79% | 10.05% | 0.23 |
| TG11 | 12.21% | 27.52% | 2.25 |
| TG12 | 31.93% | 31.38% | 0.98 |
| Meloxicam | 30.33% | 44.95% | 1.48 |
| Compound | COX Inhibition at a Concentration of 100 μM | COX-1 Free Energy of Binding (kJ/mol) | COX-2 Free Energy of Binding (kJ/mol) | |
|---|---|---|---|---|
| COX-1 | COX-2 | |||
| TG4 | N/A | 41.60% | 2.1 | −46.5 |
| TG6 | N/A | 24.43% | 13.3 | −44.4 |
| TG10 | 42.79% | 10.05% | −46.5 | −41.4 |
| TG11 | 12.21% | 27.52% | −38.4 | −45.2 |
| TG12 | 31.93% | 31.38% | −45.6 | −45.2 |
| Meloxicam | 30.33% | 44.95% | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glomb, T.; Wiatrak, B.; Gębczak, K.; Gębarowski, T.; Bodetko, D.; Czyżnikowska, Ż.; Świątek, P. New 1,3,4-Oxadiazole Derivatives of Pyridothiazine-1,1-Dioxide with Anti-Inflammatory Activity. Int. J. Mol. Sci. 2020, 21, 9122. https://doi.org/10.3390/ijms21239122
Glomb T, Wiatrak B, Gębczak K, Gębarowski T, Bodetko D, Czyżnikowska Ż, Świątek P. New 1,3,4-Oxadiazole Derivatives of Pyridothiazine-1,1-Dioxide with Anti-Inflammatory Activity. International Journal of Molecular Sciences. 2020; 21(23):9122. https://doi.org/10.3390/ijms21239122
Chicago/Turabian StyleGlomb, Teresa, Benita Wiatrak, Katarzyna Gębczak, Tomasz Gębarowski, Dorota Bodetko, Żaneta Czyżnikowska, and Piotr Świątek. 2020. "New 1,3,4-Oxadiazole Derivatives of Pyridothiazine-1,1-Dioxide with Anti-Inflammatory Activity" International Journal of Molecular Sciences 21, no. 23: 9122. https://doi.org/10.3390/ijms21239122








