Abstract
Using nanoparticles to carry and delivery anticancer drugs holds much promise in cancer therapy, but nanoparticles per se are lacking specificity. Active targeting, that is, using specific ligands to functionalize nanoparticles, is attracting much attention in recent years. Aptamers, with their several favorable features like high specificity and affinity, small size, very low immunogenicity, relatively low cost for production, and easiness to store, are one of the best candidates for the specific ligands of nanoparticle functionalization. This review discusses the benefits and challenges of using aptamers to functionalize nanoparticles for active targeting and especially presents nearly all of the published works that address the topic of using aptamers to functionalize nanoparticles for targeted drug delivery and cancer therapy.
1. Introduction
The ideal cancer therapeutics should be capable of exerting maximum destruction on cancer cells while being able to keep damage to healthy tissues at a minimum. Many anticancer drugs are toxic to cancer cells and healthy cells largely non-differentially, and the major reason that they cause more damage to cancer is because the cancer cells grow/divide more quickly. Besides, most anticancer drugs are in general evenly distributed throughout the body when administered systemically and the result is that only a very small fraction of the drugs reach the diseased site. Therefore, it is not surprising that selective delivery of anticancer drugs to cancer cells has long been a vigorous pursuit of cancer scientists.
Nanoparticles have the potential to encapsulate and transport anticancer drugs to tumor tissue more effectively [1]. However, nanoparticles per se do not have specificity to cancer cells; the fact that nanoparticles accumulate preferentially in cancer sites is basically due to the enhanced permeability and retention (EPR) effect of the tumor tissue [2]. On the other hand, if nanoparticles could be functionalized by ligands capable of recognizing cancer cells specifically, they will be able to target and deliver cargoes selectively to cancer cells and thus greatly increase the therapeutic index (increasing therapeutic efficacy while reducing toxicity). To date, a number of moieties have been studied to functionalize nanoparticles for specific targeting and aptamer is one of them [3].
This paper discusses aptamer-functionalized nanoparticles in targeted delivery for cancer therapy. It first compares passive and active targeting of nanoparticles, then describes the advantages of using aptamers to functionalize nanoparticles for active targeting, explains the strategies to conjugate aptamers to nanoparticles, and summarizes nearly all of the existing aptamer-functionalized nanoparticles used thus far to study targeted delivery to cancer cells. It finally briefly discusses the challenges facing active targeting.
2. Passive vs. Active Targeting of Nanoparticles
Passive targeting of nanoparticles refers to the passive accumulation of nanoparticles in the tumor tissue, which is generally attributed to the enhanced permeability and retention effect. The concept of EPR was first introduced more than 30 years ago when Maeda and colleagues found that certain macromolecules accumulate preferentially in the tumor tissue [4]. EPR is mainly the result of leakiness of the discontinuous endothelium of angiogenic tumor vasculature combined with defective lymphatic drainage of the tumor matrix, which facilitates the extravasation and accumulation of nanoparticles in tumor. It has been shown that the number of nanoparticles accumulated in tumor tissue may be 10–200 times higher than in normal tissue as a result of EPR. The EPR effect is considered to be the primary element to improve the efficacy and safety of nanotherapeutics. In fact, most of the nanomedicines marketed thus far base their increased therapeutic index mainly on the EPR effect [5].
Nevertheless, the EPR effect alone is insufficient for adequate nanoparticle accumulation, particularly in some circumstances. The EPR effect is not effective for some cancers because of tumor heterogeneity and cancer stage, is even not applicable to some types of cancers, and it is not effective in some patients because of individual differences. A survey of the literature in this area from 2005 to 2015 that included 232 data sets showed that only a median of 0.7% of the systemically administered nanoparticle dose could reach the solid tumor in mouse models [6]; multivariate analysis of the pertinent parameters indicated that tumor type, tumor model, and nanomaterial properties are the major factors to affect the delivery efficiency of the nanoparticles. Research also found that the high interstitial fluid pressure of tumor tissue impedes the extravasation of nanoparticles [7]; some particles that have entered the tumor intercellular space via EPR effect may be forced back into the blood circulation because of the high fluid pressure within the tumor interstitium. It is manifest that blood cancers, very early stage tumors, and small metastasized cancers do not have or have only insignificant EPR effect. In addition, because of tumor heterogeneity, the EPR effect is very poor or not shown in some types of cancers and even in different regions of the same tumor [8]. Clinical observations have also indicated that the EPR effect exhibits significant individual variations among patients; the nanomedicines do not increase the therapeutic efficacy in some subpopulations of the patients [9]. Finally, and most importantly, it is now reckoned that the EPR effect chiefly works in animal models rather than in humans [10]; in patients, their effects are just uncertain (because of interpatient variability); these uncertainties pose the most serious challenge to the rationale of nanomedicine development based on the EPR effect and to the clinical translation of the nanotherapeutics. All the above problems warrant the development of a more effective way to deliver nanoparticles to the site of interest.
Active targeting, which is achieved by conjugating tumor specific ligands to the surface of nanoparticles, can provide a means to complement the EPR effect or solve the aforementioned problems. Common classes of targeting ligands that can functionalize nanoparticles include antibodies or antibody fragments, aptamers, carbohydrates, human transferrin protein, peptides, and vitamins such as folate, etc. Representative tumor biomarkers that can be recognized by the targeting ligands include epidermal growth factor receptor (EGFR), epithelial cell adhesion molecule (EpCAM), human epidermal growth factor receptor 2 (HER2), Mucin-1 (MUC1), nucleolin, platelet-derived growth factor receptor β (PGFRβ), prostate specific membrane antigen (PSMA), transferrin receptor, folate receptor, and so on.
The foremost advantage of actively targeted nanoparticles over passively targeted nanoparticles is that they can add on to or improve the EPR effect. An actively targeted nanoparticle can first enter the tumor tissue via the EPR effect and then target cancer cells through specific ligand recognition of the tumor biomarker. In addition, active targeting can augment the EPR effect by having more particles entering than leaving the tumor interstitium because the particles that already enter stick to the cancer cells and thus lower the concentration of the free nanoparticles in the interstitial space. Studies have already demonstrated that actively targeted nanoparticles tend to accumulate more efficiently in the tumor tissue through their selective binding to receptors on the cancer cells when they enter the tumor interstitium [11].
The ligand-mediated active targeting not only helps nanoparticles selectively reach the tumor; it may also promote cellular internalization of the nanoparticles through receptor-mediated endocytosis since some receptors have the intrinsic property to internalize when bound by a ligand. The importance of cellular internalization should be obvious when we think of the fact that most anticancer drugs exert their actions inside cancer cells. Although nanoparticles themselves can get into the cell through clathirin-mediated endocytosis or fluid-phase pinocytosis, conjugation of active ligands to them may boost the process. Receptor-mediated engulfment has already been observed in many specific ligand conjugated nanoparticles; typical examples of aptamer-mediated cellular internalization include the PSMA-targeting A10 aptamer mediated as well as the nucleolin-targeting AS1411 aptamer mediated internalizations [12,13].
Although the targeting ligands can be conjugated with the anticancer agents such as siRNAs and chemotherapeutics directly, the advantage of using nanoparticles is that they can deliver large amounts of drug payload or diversified therapeutics to cancer cells per delivery and biorecognition event [14]. Having a nanoparticle encapsulate diverse therapeutic ingredients could potentially offer synergistic tumor killing effects (e.g., combining any of these anticancer strategies like chemotherapy, gene silencing, immunotherapy, photodynamic therapy, photothermal therapy, and thermodynamic therapy, etc.). Encapsulating different therapeutics within a nanoparticle may also help to overcome or reduce multiple drug resistance (MDR) because MDR usually does not occur to different drugs at the same time or at the same degree, and the mechanisms of MDR differs with different drugs. One example is that nanoparticle-mediated combination of chemotherapy and photodynamic therapy can overcome drug resistance through invoking multiple anticancer mechanisms including cytotoxicity and significantly enhanced production of reactive oxygen species [15].
Active targeting of nanoparticles could also have additive therapeutic effects by exploiting the drug-carrying and receptor-inhibiting actions at the same time. For instance, anticancer reagent-containing nanoparticles functionalized with HER2-targeting ligand, in addition to delivering the therapeutic ingredients into the target cells can, meanwhile, inhibit the activity of the targeted receptors or remove the receptors from the cell surface by means of internalization [16].
3. Aptamer-Functionalized Nanoparticles in Actively Targeted Drug Delivery
Aptamers are short single-stranded DNA or RNA molecules with defined three-dimensional structures that can selectively bind to target molecules with high affinity [17]. Aptamers are usually produced by selecting them from a large random sequence pool with the technology systematic evolution of ligands by exponential enrichment (SELEX). In addition to their superb binding specificity and affinity, aptamers have a number of other favorable features that together make them very suitable molecules to functionalize nanoparticles for actively targeted delivery. Aptamer functionalized nanoparticles have already demonstrated their effectiveness in targeted delivery of anticancer drugs in numerous preclinical and animal studies, though none of them have as yet entered clinical trial or application.
3.1. The Advantages of Using Aptamers to Functionalize Nanoparticles
Aptamers have a very broad spectrum of target recognition and binding; they have little or no immunogenicity; they can easily be end-attached with a chemical group to conjugate nanoparticles; they are small (only a few nanometers in diameter) and will not increase nanoparticle size significantly after coupling; they are relatively easy to make and to store [17]. Those are the general properties of aptamers that make them one of the best choices to functionalize nanoparticles. Up to now, quite a few aptamers have been used to functionalize nanoparticles for targeted delivery to cancer cells (Table 1).

Table 1.
Aptamer-functionalized nanoparticles designed for actively targeted drug delivery and cancer therapy in laboratory investigation stage.
Apart from the abovementioned characteristics, aptamers have a unique advantage that is related to their production—the establishment of the cell-SELEX technique and its improvements have made the aptamer an especially useful ligand to be used to construct the cancer-targeting nanocarriers (Figure 1).
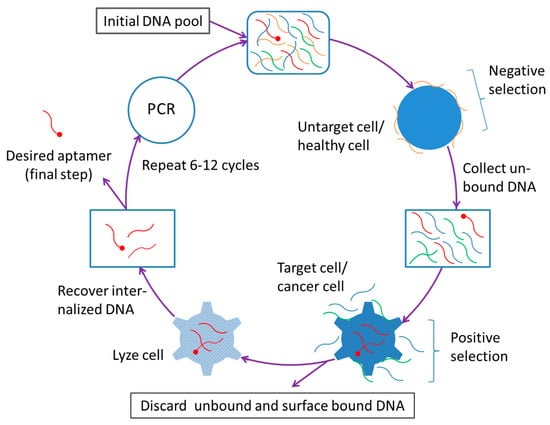
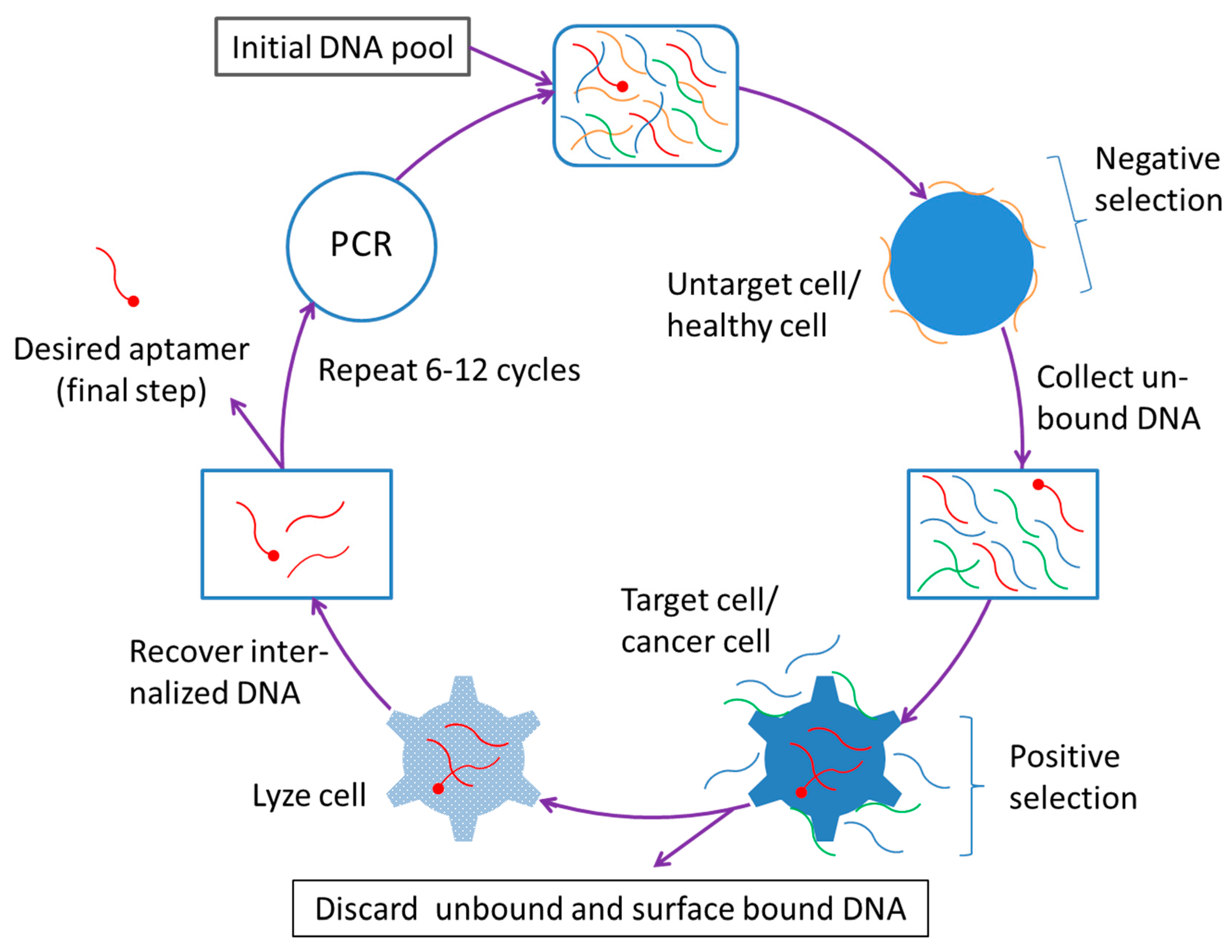
Figure 1.
Selection procedure of cell-internalizing DNA aptamer using cell-SELEX.
After the setting up of the prototype SELEX technology in 1990, a selection strategy known as cell-SELEX was developed in 2003 that uses whole (living) cells to select aptamers targeting cell surface molecules [215]. This technique allows for the isolation of cell-recognizing aptamers without prior knowledge of the target molecule(s). In 2006, a negative selection (or counter-selection) process was integrated into the original cell-SELEX strategy, which makes it possible to obtain cell-specific aptamers on researcher’s will [216]. In the new cell-SELEX procedure, the negative selection is performed first, wherein the negative-selection cells (these may be normal cells or any untargeted cells and several different types of cells may be used) are used to absorb the undesired or non-specific aptamers (In this step, the undesired or non-specific oligonucleotides in the pool are removed as they bind to the negative-selection cells). The negative selection is followed by positive selection that is conducted basically in the same way as the conventional cell-SELEX strategy and aims to discard the oligonucleotides that do not bind to the positive-selection cells (usually, certain types of cancer cells or any researcher-intended cells are used for this purpose). Thus, by employing the new cell-SELEX technique, one is able to generate aptamers that can specifically recognize cell surface receptors (or molecules) and thus can effectively differentiate cancer cells from normal cells. More importantly, with certain added steps, the cell-SELEX technique can still select aptamers that not only specifically recognize or target cell surface receptors but also get into the cells through receptor mediated internalization [217].
3.2. Strategies of Conjugating Aptamers to Nanoparticles
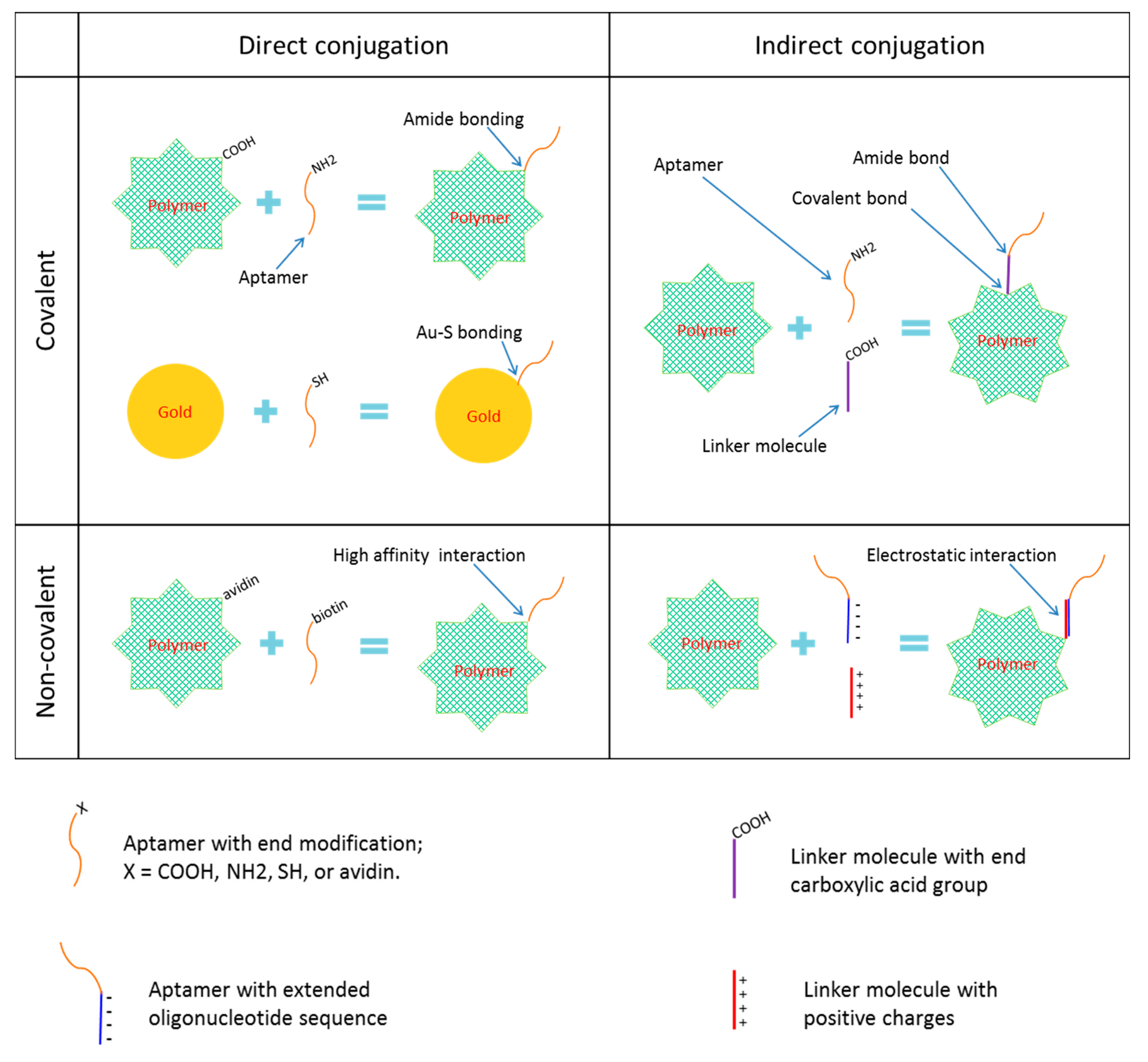
Aptamers can be conjugated to nanoparticles directly or indirectly via a linker molecule (a bridge or spacer). Both direct and indirect conjugation can be achieved either covalently or non-covalently (Figure 2).
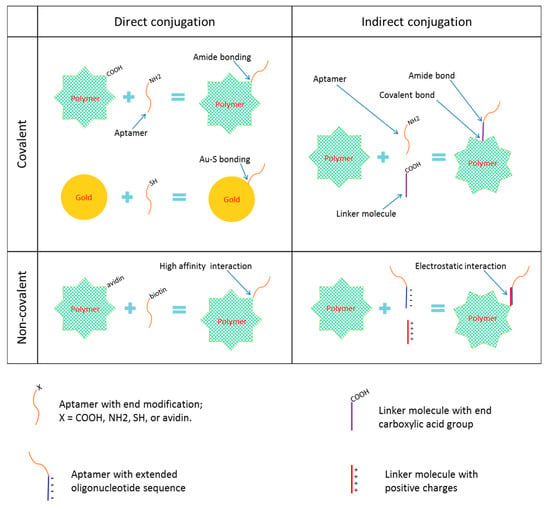
Figure 2.
Common strategies of nanoparticle-aptamer conjugation.
In covalent conjugation, a functional group (such as a primary amino group or a thiol group) is usually attached to one terminus of the aptamer, which can react with the functional group (such as the carboxylic acid group, the maleimide group, and the aldehyde group) on the surface of the nanoparticle or at one end of the linker molecule, or react with the gold or other metal element or inorganic molecule for inorganic nanoparticles. Common examples of covalent conjugation include the carboxylic acid group and the amino group interaction that results in an amide (or carboxamide) linkage, the carboxylic acid group and the thiol group interaction that results in a thioester bond, the carboxylic acid group and the alcohol group interaction that results in an ester bond, the primary amine group and thiol group interaction that results in a thioamide bond, the thiol group and the thiol group interaction that results in a disulfide bond, and the thiol group and the gold or silver interaction that results in a Au–S or Ag-S bond.
Non-covalent conjugation strategies include high affinity interactions and electrostatic interactions. The former includes avidin–biotin and streptavidin–biotin interactions. The latter are commonly seen when a linker molecule is used, in which case the opposite charges on the linker molecule and on the extended oligonucleotide sequence of the aptamer interact, but also include the using of histidine tags.
Most of the aptamer–nanoparticle conjugates reported thus far utilized the direct and covalent strategy. According to Farokhzad and colleagues [11], “covalently linked bioconjugates may result in enhanced stability in physiological salt and pH whilst avoiding the unnecessary addition of biological components (i.e., streptavidin); thus minimizing immunological reactions and potential toxicity”. Fewer studies used bridge or spacer molecule to link aptamer and nanoparticle together. These are in consideration of avoiding any possible steric or spatial restrictions on aptamer’s binding to target molecule, but an associated problem is the increased size of the conjugates. Several aptamer-nanoparticle constructions, including both direct and indirect linkage, used the avidin–biotin or the streptavidin–biotin system. These interactions are very stable but the bulk of the formulation may increase considerably and potential immunological rejection problems might also result.
3.3. Aptamer-Functionalized Nanoparticles in Pre-Clinical Studies
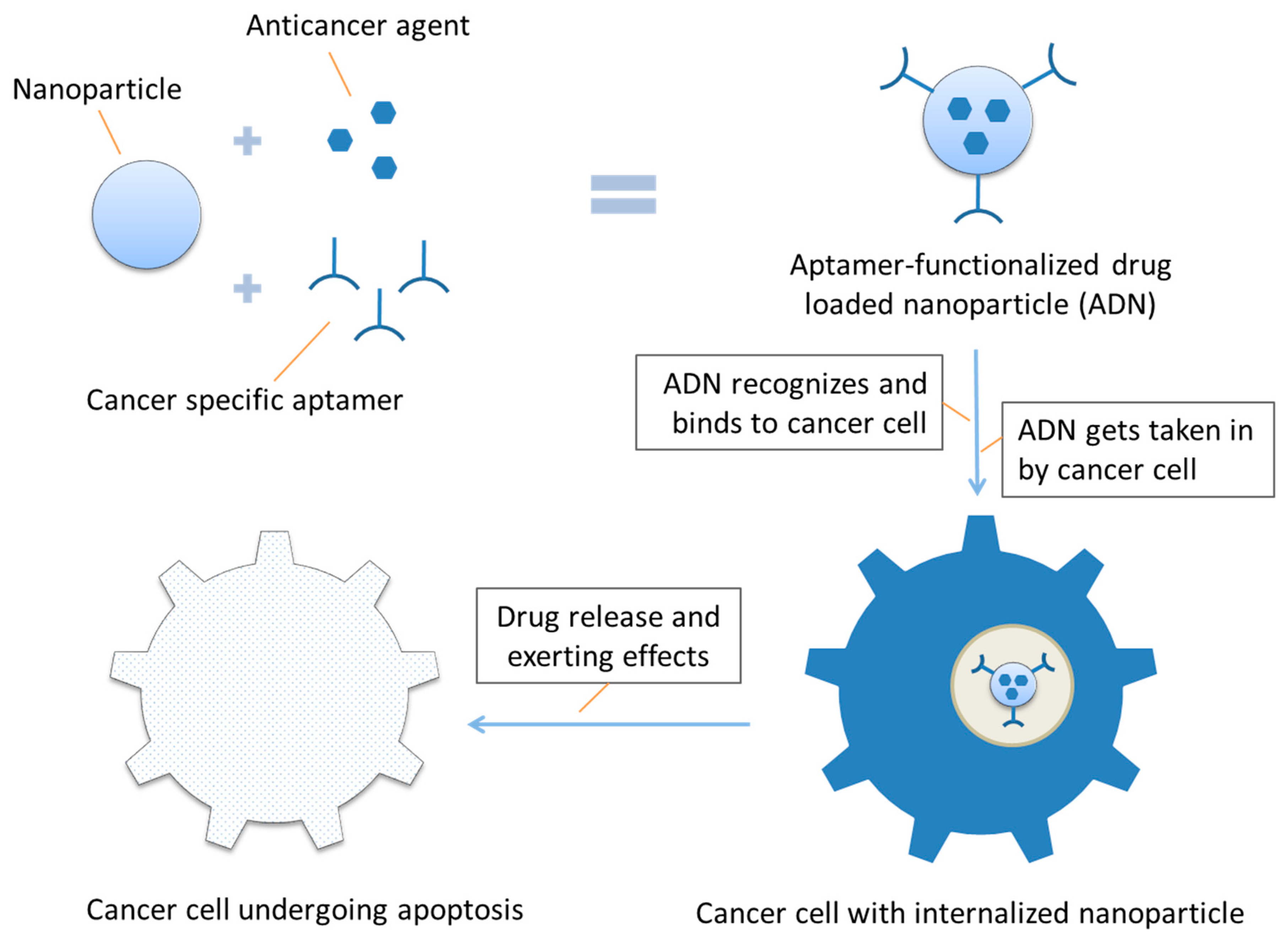
Up till now, quite a lot of aptamer-conjugated nanoparticles have been developed that can target specific cancer cells, deliver various therapeutic agents into cancer cells, and result in cancer cell toxicity in vitro (e.g., inhibit cell proliferation and induce apoptosis of cultivated cancer cells) and/or anticancer effects in vivo (e.g., inhibit xenograft tumor formation in nude mouse model). An inclusive list of nearly all aptamer-conjugated drug-delivering nanoparticles that have been studied thus far with their characteristics and sources is provided in Table 1. A schematic representation of the action process of aptamer-functionalized nanoparticles acting on a cancer cell is shown in Figure 3.
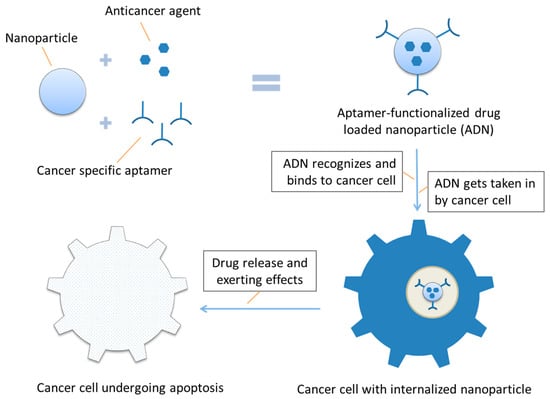
Figure 3.
Schematic representation of aptamer-functionalized nanoparticle acting on a cancer cell.
Farokhzad and Langer et al. [18] first performed the proof of concept study of using the aptamer to functionalize nanoparticles for actively targeted drug delivery in 2004. The authors synthesized the nanoparticles of poly (lactic acid)-block-polyethylene glycol copolymer with a terminal carboxylic acid functional group (PLA-b-PEG-COOH) and encapsulated the nanoparticles with rhodamine-labeled dextran as a model drug; they then covalently attached the PSMA-targeting A10 RNA aptamer to the nanoparticles through the reaction of the amino groups on the 3′ end of the aptamers with the carboxylic acid groups on the surface of the nanoparticles. These aptamer–nanoparticle conjugates were demonstrated to be able to target the PSMA-positive prostate LNCaP cells significantly more efficiently compared with the same PEGylated nanoparticles without aptamer conjugation and could get internalized into the cells. The uptake of these conjugates was not boosted in the PC3 cells that are also prostate-derived but do not express PSMA.
A similar nanoparticle-aptamer construction, which used the same PSMA-targeting aptamer but used poly (lactic-co-glycolic acid)-block-polyethylene glycol copolymer with a terminal carboxylic acid group (PLGA-b-PEG-COOH) as nanomaterial and encapsulated the anticancer drug Docetaxel within the nanoparticles, was later assessed both in vitro and in vivo by the same laboratory. The in vivo results showed that the aptamer-targeted drug-loaded nanoparticles exhibited significantly more reduced toxicity (side effects) in the nude mice as measured by mean body weight loss than non-targeted nanoparticles, and intratumoral injection of these aptamer-targeted drug-loaded nanoparticles resulted in complete tumor reduction in five of seven LNCaP xenograft nude mice compared with two of five for non-targeted nanoparticles [19].
Up to the present time, polymers, which include miscellaneous classes with PLGA-PEG being the most frequently used, remain the most used nanomaterials to construct aptamer functionalized nanoparticles to study targeted delivery for cancer therapy, followed by lipid based materials, particularly liposomes and nucleic acid based nanoparticles, including either DNA or RNA. Other organic nanomaterials that have been used include dendrimers, chitosan, proteins/peptides, or hybrids of the above. There are also many inorganic nanomaterials that have been studied in this area, including gold (Au) compounds, silver (Ag), mesoporous silica, graphene based, Calcium carbonate, ZnO, iron, etc. Other and special inorganic nanomaterials include magnetic nanomaterials, quantum dot based nanoparticles, and so on. In addition, organic and inorganic hybrids have also been used. Refer to Table 2 for a classified list of these nanoparticles and nanomaterials with their payloads, targets, related cancers, etc.

Table 2.
Aptamer-functionalized nanoparticles classified by nanomaterials and payloads.
4. Challenges Facing Actively Targeted Delivery
Although active targeting holds much promise, several challenges exist. These include the increased complexity of synthesis and purification, the increased cost to make the conjugants, the alterations of nanoparticle properties, choosing a suitable tumor marker or receptor to target, and so forth.
4.1. Potential Alterations of Nanoparticle and Ligand Properties after Conjugation
Ligand conjugation may alter the properties of the nanoparticle. Not only will it increase nanoparticle size; it can also change the charge and modify the conformation of the nanoparticle. The change of nanoparticle size is likely to affect their pharmacokinetics; the change of nanoparticle charge will probably complicate their cellular uptake; the change of nanoparticle conformation may influence the binding feature of the attached ligand because of inadequate steric freedom or decreased orientation. All these must be taken into consideration in making actively targeting nanoparticles.
Although conjugating ligands to nanoparticles might change the pharmacokinetic property of the nanoparticles, this may not be a problem for aptamer conjugation because aptamers are very small, about 2–3 nm in length, in comparison with the drug-carrying nanoparticles, which is typically around 100 nm or larger in diameter. In fact, no literature has reported any alterations in the pharmacokinetics of nanoparticles following aptamer coupling.
Aptamers are commonly modified before therapeutic use. The purpose of modification is to increase their stability against nuclease degradation or prolong their half-life against kidney filtration. Aptamer modification can be performed during selection or after selection. The former aims at stabilizing the aptamers against nucleases. The latter aims at prolonging renal retention and is frequently done with PEGylation, covalent attachment of PEG to one end of the aptamer. Therefore, the attachment of aptamers to a nanoparticle will favorably increase their stability.
However, conjugation of aptamers to a nanoparticle might interfere with their proper folding and change their binding specificity and affinity. For example, the surface charge of the nanoparticle and the density of the attached aptamers on the nanoparticle may both affect their folding and three-dimensional structure. In addition, aptamers that are coupled directly to a nanoparticle may not recognize and bind their target effectively because there is no sufficient space (stereo-interference effect). Sometimes, the orientation of aptamer immobilization may also affect aptamer binding. All these problems should be considered by the researchers and optimum parameters or corresponding resolving measures be taken. For instance, the density and the orientation of attached aptamers can be investigated and optimized, and when stereo-interference occurs, the researchers can consider the use of a spacer molecule.
4.2. Selection of Suitable Tumor Marker or Receptor
The ideal receptor for targeted therapy is one that is exclusively presented on the tumor cells but not on the healthy cells. However, such a receptor may not exist in reality. What we can do is to choose the receptors that have a higher expression level on tumor cells than on healthy cells. The expression of the target receptors on healthy cells, though at a lower level, still carries a potential risk of off target binding. What is more, binding to these receptors may consume or waste the therapeutic nanoparticles and lower its concentration to reach the tumor.
4.3. The “Binding Site Barrier” Effect
Aside from the challenges mentioned above, there may also be the “binding site barrier” problem, which refers to a situation wherein high affinity binding to target cells prevents in-depth and uniform penetration of the targeted therapeutics into the tumor tissue. This phenomenon was first observed by Weinstein and colleagues [218,219] with antibodies, which showed that (1) antibody–antigen binding in tumor-retarded antibody percolation and (2) high antibody affinity had a tendency to decrease antibody percolation. The explanation to the phenomenon that higher-affinity antibodies penetrate the tumor tissue less efficiently than lower-affinity antibodies is that during tissue penetration, the higher-affinity antibodies bind tightly to the cells they first meet and so there are fewer free antibody molecules available; in contrast, lower-affinity antibodies tend to bypass these target cells and can penetrate deeper. Although the “binding site barrier” was originally demonstrated in antigen–antibody interaction, it may be reasonably extrapolated to the actively targeted nanoparticles and a similar phenomenon has in fact be observed by Miao et al. [220] using anisamide ligand targeted lipid-coated calcium phosphate nanoparticles. Therefore, it is essential to seek a balance between the affinity of active tumor targeting and the depth of nanoparticle penetration; trial and error may be necessary [221].
5. Conclusions
The first nanotechnology-based anticancer medicine was approved by the United States Food and Drug Administration (FDA) in 1996, which used PEGylated liposomes to encapsulate the chemotherapeutic drug doxorubicin. Today, about ten nanoparticle based medications are on the market (approved by FDA or other agencies) for cancer therapy [14,222]. All of them are non-targeted or passively targeted. These nanodrugs could delay the clearance or prolong the half-life of the drugs and reduce side-effects to a certain degree. However, only a modest increase in therapeutic efficacy could be observed and the undesired off-target problem still exists, which calls for the development of active targeting of nanoparticles. At the present time, more than a dozen nanoparticles for cancer therapy are undergoing clinical trials [2], of which several are actively targeted, but none of them are aptamer-functionalized. Actively targeted, especially aptamer-functionalized, nanoparticles hold great promise for future nanodrug development and applications. Therefore, more efforts are needed to further the investigation in this area, to refine the experiments and overcome the obstacles for clinical translation. Some obstacles for developing aptamer conjugated-nanoparticles into clinical use include insufficient data about their off-target effects and toxicity either in animals or in human. Venditto and Szoka once notified in their review paper titled Cancer nanomedicines: so many papers and so few drugs published in 2013 that “if we are truly interested in bringing more drugs into the clinic we should focus less on our publication record and more on devising scientific progress that translates into patient treatment” [223]. The same situation exists in the investigation of aptamer-functionalized nanoparticles when we take notice of the fact that more than two hundred papers have been published so far but none of the aptamer-functionalized nanoparticles have entered clinical trials, not to mention clinical application.
Funding
This work was supported by NSFC, grant number 81760732.
Conflicts of Interest
The author declares no competing interests.
Abbreviations
| 3WJ-RNA | a highly stable three-way junction (3WJ) motif from phi29 packaging RNA |
| 5-FU | 5-fluorouracil |
| ALCL | anaplastic large cell lymphoma |
| ALK | anaplastic lymphoma kinase |
| ALL | acute lymphoblastic leukemia, also known as T-cell acute lymphoblastic leukemia |
| AML-M2 | acute myeloid leukemia subtype 2 |
| APTES | (3-aminopropyl) triethoxysilane |
| BSA | bovine serum albumin |
| cMet | hepatocyte growth factor receptor |
| COOH | (terminal) carboxylic acid group |
| CSC | cancer stem cell |
| CTC | circulating tumor cell |
| CUR-NP | curcumin-loaded lipid-polymer-lecithin hybrid nanoparticle |
| Cyt c | cytochrome c |
| DAU | daunorubicin |
| DGL | dendrigraftpoly-L-lysines |
| DOTAP | 1,2-dioleoyl-3-trimethylammonium-propane |
| DNR | daunorubicin |
| DOX | doxorubicin |
| dsDNA | double-stranded DNA |
| DSPE | 1,2-distearoyl-sn-glycero-3-phosphoethanolamine |
| EGFR | Epidermal growth factor receptor |
| EHH | electrostatic adsorption, hydrogen bonding, and hydrophobic interaction |
| Ehrlich’s ACC | Ehrlich’s ascites carcinoma cell |
| ELP | elastin-like polypeptide |
| EpCAM | epithelial cell adhesion molecule |
| EPI | epirubicin |
| FGFR1 | fibroblast growth factor receptor type-1 |
| FMSN | fluorescent mesoporous silica nanoparticle |
| FN | fibronectin |
| FO | Ferric oxide |
| FoxM1 | Forkhead box M1 |
| Gd:SrHap | gadolinium-doped luminescent and mesoporous strontium hydroxyapatite |
| GMNP | gold-coated magnetic nanoparticle |
| GNP | gold nanoparticle |
| GO | Graphene oxide |
| GPN | gefitinib-loaded poly (lactic co-glycolic acid) nanoparticle |
| GQD | graphene quantum dot |
| GST | glutathione S-transferase |
| HA | Hyaluronic acid |
| HAS-CS | human serum albumin coated with chitosan |
| HBLL | human B cell leukemia and lymphoma |
| HCC | Hepatocellular carcinoma |
| HER3 | human epidermal growth factor receptor 3 |
| His | hexahistidine |
| HMME | is a photosensitizer |
| HPA | heparanase |
| HPAEG | poly(2-((2-(acryloyloxy)ethyl)disulfanyl)ethyl 4-cyano-4-(((propylthio)carbonothioyl)-thio)-pentanoate-co-poly(ethylene glycol) methacrylate) |
| HSP71 | heat shock cognate 71 kDa protein |
| HTT | hyperthermia therapy |
| IL-6R | interleukin-6 receptor |
| IONP | Iron oxide nanoparticle |
| KG6E | glutamic acid-modified dendritic poly(L-lysine) system |
| KLA | (KLAKLAK)2 peptide |
| LP-DNA | liposome-polycation-DNA |
| MAA | methacrylamide |
| MAGE | melanoma-associated peptide antigen |
| MAL | maleimide |
| MASI | N-(methacryloxy)succinimide |
| MCS | Myristylated Chitosan |
| MMA | methyl methacrylate |
| MOF | (mesoporous) metal-organic framework |
| MPC | mesoporous carbon |
| MPEG | Poly(ethylene glycol) methyl ether |
| M-PLGA | mannitol-functionalized poly(lactide-co-glycolide) |
| MSN | Mesoporous silica nanoparticle |
| mTEC | mouse tumor endothelial cell |
| MDR | multiple drug resistance |
| MUC1 | Mucin-1 |
| NHS | N-hydroxysuccinimide |
| NIR PLN | near infrared-persistent luminescence nanomaterials |
| NMOF | amino-triphenyl dicarboxylate-bridged Zr4+ metal-organic framework nanoparticle |
| NP | nanoparticle |
| NSCLC | non-small cell lung cancer |
| ONT | oligonucleotide |
| PAA | polyacrylic acid |
| PAM | Peptide amphiphile micelle |
| PAMAM | polyamidoamine |
| PBABT | poly (butylene adipate-co-butylene terephthalate) |
| PCL | poly(ε-capro-lactone) |
| PDA | hydrochloride dopamine |
| PDGFR | platelet-derived growth factor receptor |
| PEC | polyelectrolyte complexe |
| PEEUA | polyethylenimine-urocanic acid |
| PEG | polyethylene glycol |
| PEI | polyethylene imine |
| PF127 | Pluronic F127 |
| PFK15 | 1-(4-pyridyl)-3-(2-quinoline)-2-propyl-1-one (an aerobic glycolysis inhibitor) |
| PFOB | Perfluorooctylbromide |
| PGFRβ | platelet-derived growth factor receptor β |
| P-gp | P-glycoprotein |
| PLA | poly(lactic acid) |
| PLGA | poly(lactic-co-glycolic acid) |
| PLK1 | Polo-Like Kinase 1 |
| PLL | poly (L-lysine) |
| pPEGMA-PCL-pPEGMA | poly(poly(ethylene glycol) methacrylate)-poly(caprolactone)-poly(poly(ethylene glycol) methacrylate) |
| PTK7 | protein tyrosine kinase-7 |
| PTT | Photothermal therapy |
| PVP | poly (N-vinylpyrrolidone) |
| PβAE | poly (β amino ester) |
| QD | quantum dot |
| RBCm | red blood cell membrane |
| SATB1 | special AT-rich sequence binding protein 1 |
| SPION | superparamagnetic iron oxide nanoparticles |
| SPMFN | Superparamagnetic Ferroarabinogalactan Nanoparticles |
| TAG-72 | tumor-associated glycoprotein 72 |
| TD | thiolated dextran |
| TiO2 | titanium dioxide |
| TLR | Toll-like receptor TLR4-siRNA |
| TM-JM1/2 | transmembrane-juxtamembrane 1/2 domain |
| TMPyP | 5, 10, 15, 20-tetra (phenyl-4-N-methyl-4-pyridyl) porphyrin |
| TMPyP4 | 5,10,15,20-tetrakis(1-methylpyridinium-4-yl) porphyrin |
| TNBC | triple-negative breast cancer |
| TPGS | D-α-tocopheryl polyethylene glycol 1000 succinate |
| TSP | thermosensitive polymer |
| UCNP | up-conversion luminescent |
| NaYF4 | Yb(3+)/Er(3+) nanoparticle |
| VEGF | vascular endothelial growth factor |
| β-CD | β-cyclodextrin |
References
- Quader, S.; Kataoka, K. Nanomaterial-Enabled Cancer Therapy. Mol. Ther. 2017, 25, 1501–1513. [Google Scholar] [CrossRef]
- Shi, J.; Kantoff, P.W.; Wooster, R.; Farokhzad, O.C. Cancer nanomedicine: Progress, challenges and opportunities. Nat. Rev. Cancer 2017, 17, 20–37. [Google Scholar] [CrossRef]
- Guan, B.; Zhang, X. Aptamers as Versatile Ligands for Biomedical and Pharmaceutical Applications. Int. J. Nanomed. 2020, 15, 1059–1071. [Google Scholar] [CrossRef]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar]
- Sun, H.; Zu, Y. Aptamers and Their Applications in Nanomedicine. Small 2015, 11, 2352–2364. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.; Tavares, A.J.; Dai, Q.; Ohta, S.; Audet, J.; Dvorak, H.F.; Chan, W.C.W. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 2016, 1, 16014. [Google Scholar] [CrossRef]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Ali, E.S.; Sharker, S.M.; Islam, M.T.; Khan, I.N.; Shaw, S.; Rahman, A.; Uddin, S.J.; Shill, M.C.; Rehman, S.; Das, N.; et al. Targeting cancer cells with nanotherapeutics and nanodiagnostics: Current status and future perspectives. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, N.; Wu, J.; Xu, X.; Kamaly, N.; Farokhzad, O.C. Cancer nanotechnology: The impact of passive and active targeting in the era of modern cancer biology. Adv. Drug Deliv. Rev. 2014, 66, 2–25. [Google Scholar] [CrossRef] [PubMed]
- Danhier, F. To exploit the tumor microenvironment: Since the EPR effect fails in the clinic, what is the future of nanomedicine? J. Control. Release 2016, 244, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Farokhzad, O.C.; Karp, J.M.; Langer, R. Nanoparticle–aptamer bioconjugates for cancer targeting. Expert Opin. Drug Deliv. 2006, 3, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Kolishetti, N.; Dhar, S.; Valencia, P.M.; Lin, L.Q.; Karnik, R.; Lippard, S.J.; Langer, R.; Farokhzad, O.C. Engineering of self-assembled nanoparticle platform for precisely controlled combination drug therapy. Proc. Natl. Acad. Sci. USA 2010, 107, 17939–17944. [Google Scholar] [CrossRef] [PubMed]
- Ireson, C.R.; Kelland, L.R. Discovery and development of anticancer aptamers. Mol. Cancer Ther. 2006, 5, 2957–2962. [Google Scholar] [CrossRef]
- Cheng, Z.; Al Zaki, A.; Hui, J.Z.; Muzykantov, V.R.; Tsourkas, A. Multifunctional Nanoparticles: Cost Versus Benefit of Adding Targeting and Imaging Capabilities. Science 2012, 338, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Shiao, Y.-S.; Chiu, H.-H.; Wu, P.-H.; Huang, Y.-F. Aptamer-Functionalized Gold Nanoparticles As Photoresponsive Nanoplatform for Co-Drug Delivery. ACS Appl. Mater. Interfaces 2014, 6, 21832–21841. [Google Scholar] [CrossRef]
- Shen, Y.; Li, M.; Liu, T.; Liu, J.; Xie, Y.; Zhang, J.; Xu, S.; Liu, H. A dual-functional HER2 aptamer-conjugated, pH-activated mesoporous silica nanocarrier-based drug delivery system provides in vitro synergistic cytotoxicity in HER2-positive breast cancer cells. Int. J. Nanomed. 2019, 14, 4029–4044. [Google Scholar] [CrossRef]
- Fu, Z.; Xiang, J. Aptamers, the Nucleic Acid Antibodies, in Cancer Therapy. Int. J. Mol. Sci. 2020, 21, 2793. [Google Scholar] [CrossRef]
- Farokhzad, O.C.; Jon, S.; Khademhosseini, A.; Tran, T.-N.T.; LaVan, D.A.; Langer, R. Nanoparticle-Aptamer Bioconjugates. Cancer Res. 2004, 64, 7668–7672. [Google Scholar] [CrossRef]
- Farokhzad, O.C.; Cheng, J.; Teply, B.A.; Sherifi, I.; Jon, S.; Kantoff, P.W.; Richie, J.P.; Langer, R. Targeted nanoparticle-aptamer bioconjugates for cancer chemotherapy in vivo. Proc. Natl. Acad. Sci. USA 2006, 103, 6315–6320. [Google Scholar] [CrossRef]
- Dhar, S.; Gu, F.X.; Langer, R.; Farokhzad, O.C.; Lippard, S.J. Targeted delivery of cisplatin to prostate cancer cells by aptamer functionalized Pt(IV) prodrug-PLGA-PEG nanoparticles. Proc. Natl. Acad. Sci. USA 2008, 105, 17356–17361. [Google Scholar] [CrossRef]
- Huang, Y.-F.; Sefah, K.; Bamrungsap, S.; Chang, H.-T.; Tan, W. Selective Photothermal Therapy for Mixed Cancer Cells Using Aptamer-Conjugated Nanorods. Langmuir ACS J. Surf. Colloids 2008, 24, 11860–11865. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Z.; Bagalkot, V.; Vasilliou, C.C.; Gu, F.; Alexis, F.; Zhang, L.; Shaikh, M.; Yuet, K.; Cima, M.J.; Langer, R.; et al. Superparamagnetic Iron Oxide Nanoparticle-Aptamer Bioconjugates for Combined Prostate Cancer Imaging and Therapy. ChemMedChem 2008, 3, 1311–1315. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Soontornworajit, B.; Martin, J.; Sullenger, B.A.; Gilboa, E.; Wang, Y. A Hybrid DNA Aptamer-Dendrimer Nanomaterial for Targeted Cell Labeling. Macromol. Biosci. 2009, 9, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Tong, R.; Mishra, A.; Xu, W.; Wong, G.C.; Cheng, J.; Lu, Y. Reversible cell-specific drug delivery with aptamer-functionalized liposomes. Angew. Chem. (Int. Ed. Engl.) 2009, 48, 6494–6498. [Google Scholar] [CrossRef]
- Yu, C.; Hu, Y.; Duan, J.; Yuan, W.; Wang, C.; Xu, H.; Yang, X.-D. Novel Aptamer-Nanoparticle Bioconjugates Enhances Delivery of Anticancer Drug to MUC1-Positive Cancer Cells In Vitro. PLoS ONE 2011, 6, e24077. [Google Scholar] [CrossRef]
- Guo, J.; Gao, X.; Su, L.; Xia, H.; Gu, G.; Pang, Z.; Jiang, X.; Yao, L.; Chen, J.; Chen, H.-Z. Aptamer-functionalized PEG–PLGA nanoparticles for enhanced anti-glioma drug delivery. Biomaterials 2011, 32, 8010–8020. [Google Scholar] [CrossRef]
- Chang, M.; Yang, C.-S.; Huang, D.-M. Aptamer-Conjugated DNA Icosahedral Nanoparticles As a Carrier of Doxorubicin for Cancer Therapy. ACS Nano 2011, 5, 6156–6163. [Google Scholar] [CrossRef]
- Yu, M.K.; Kim, D.; Lee, I.-H.; So, J.-S.; Jeong, Y.Y.; Jon, S. Image-Guided Prostate Cancer Therapy Using Aptamer-Functionalized Thermally Cross-Linked Superparamagnetic Iron Oxide Nanoparticles. Small 2011, 7, 2241–2249. [Google Scholar] [CrossRef]
- Lee, I.-H.; An, S.; Yu, M.K.; Kwon, H.-K.; Im, S.-H.; Jon, S. Targeted chemoimmunotherapy using drug-loaded aptamer–dendrimer bioconjugates. J. Control. Release 2011, 155, 435–441. [Google Scholar] [CrossRef]
- Bagalkot, V.; Gao, X. siRNA-Aptamer Chimeras on Nanoparticles: Preserving Targeting Functionality for Effective Gene Silencing. ACS Nano 2011, 5, 8131–8139. [Google Scholar] [CrossRef]
- Xiao, Z.; Levy-Nissenbaum, E.; Alexis, F.; Lupták, A.; Teply, B.A.; Chan, J.M.; Shi, J.; Digga, E.; Cheng, J.; Langer, R.; et al. Engineering of Targeted Nanoparticles for Cancer Therapy Using Internalizing Aptamers Isolated by Cell-Uptake Selection. ACS Nano 2012, 6, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Aravind, A.; Jeyamohan, P.; Nair, R.; Veeranarayanan, S.; Nagaoka, Y.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. AS1411 aptamer tagged PLGA-lecithin-PEG nanoparticles for tumor cell targeting and drug delivery. Biotechnol. Bioeng. 2012, 109, 2920–2931. [Google Scholar] [CrossRef] [PubMed]
- Aravind, A.; Varghese, S.H.; Veeranarayanan, S.; Mathew, A.; Nagaoka, Y.; Iwai, S.; Fukuda, T.; Hasumura, T.; Yoshida, Y.; Maekawa, T.; et al. Aptamer-labeled PLGA nanoparticles for targeting cancer cells. Cancer Nanotechnol. 2012, 3, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Qian, J.; Cao, S.; Yang, Z.; Pang, Z.; Pan, S.; Fan, L.; Xi, Z.; Jiang, X.; Zhang, Q. Precise glioma targeting of and penetration by aptamer and peptide dual-functioned nanoparticles. Biomaterials 2012, 33, 5115–5123. [Google Scholar] [CrossRef]
- Yang, X.; Liu, X.; Liu, Z.; Pu, F.; Ren, J.; Qu, X. Near-Infrared Light-Triggered, Targeted Drug Delivery to Cancer Cells by Aptamer Gated Nanovehicles. Adv. Mater. 2012, 24, 2890–2895. [Google Scholar] [CrossRef]
- Gao, H.; Qian, J.; Yang, Z.; Pang, Z.; Xi, Z.; Cao, S.; Wang, Y.; Pan, S.; Zhang, S.; Wang, W.; et al. Whole-cell SELEX aptamer-functionalised poly(ethyleneglycol)-poly(epsilon-caprolactone) nanoparticles for enhanced targeted glioblastoma therapy. Biomaterials 2012, 33, 6264–6272. [Google Scholar] [CrossRef]
- Li, Z.; Liu, Z.; Yin, M.; Yang, X.; Yuan, Q.; Ren, J.; Qu, X. Aptamer-Capped Multifunctional Mesoporous Strontium Hydroxyapatite Nanovehicle for Cancer-Cell-Responsive Drug Delivery and Imaging. Biomacromolecules 2012, 13, 4257–4263. [Google Scholar] [CrossRef]
- Li, L.-L.; Yin, Q.; Cheng, J.; Lu, Y. Polyvalent Mesoporous Silica Nanoparticle-Aptamer Bioconjugates Target Breast Cancer Cells. Adv. Healthc. Mater. 2012, 1, 567–572. [Google Scholar] [CrossRef]
- Hernandez, F.J.; Hernandez, L.I.; Pinto, A.; Schäfer, T.; Özalp, V.C. Targeting cancer cells with controlled release nanocapsules based on a single aptamer. Chem. Commun. 2013, 49, 1285–1287. [Google Scholar] [CrossRef]
- Li, L.-L.; Xie, M.; Wang, J.; Li, X.; Wang, C.; Xueyuan, C.; Pang, D.-W.; Lu, Y.; Tan, W. A vitamin-responsive mesoporous nanocarrier with DNA aptamer-mediated cell targeting. Chem. Commun. 2013, 49, 5823–5825. [Google Scholar] [CrossRef]
- Xing, H.; Tang, L.; Yang, X.; Hwang, K.; Wang, W.; Yin, Q.; Wong, N.Y.; Dobrucki, L.W.; Yasui, N.; Katzenellenbogen, J.A.; et al. Selective delivery of an anticancer drug with aptamer-functionalized liposomes to breast cancer cells In Vitro and In Vivo. J. Mater. Chem. B 2013, 1, 5288–5297. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Siddiqui, I.A.; Nihal, M.; Pilla, S.; Rosenthal, K.; Mukhtar, H.; Gong, S. Aptamer-conjugated and doxorubicin-loaded unimolecular micelles for targeted therapy of prostate cancer. Biomaterials 2013, 34, 5244–5253. [Google Scholar] [CrossRef] [PubMed]
- Jalalian, S.H.; Taghdisi, S.M.; Hamedani, N.S.; Kalat, S.A.M.; Lavaee, P.; Zandkarimi, M.; Ghows, N.; Jaafari, M.R.; Naghibi, S.; Danesh, N.M.; et al. Epirubicin loaded super paramagnetic iron oxide nanoparticle-aptamer bioconjugate for combined colon cancer therapy and imaging in vivo. Eur. J. Pharm. Sci. 2013, 50, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; You, J.; Zeng, Z.; Li, C.; Zu, Y. An Ultra pH-Sensitive and Aptamer-Equipped Nanoscale Drug-Delivery System for Selective Killing of Tumor Cells. Small 2013, 9, 3477–3484. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Han, D.; Chen, T.; Peng, L.; Zhu, G.; You, M.; Qiu, L.; Sefah, K.; Zhang, X.; Tan, W. Building a Multifunctional Aptamer-Based DNA Nanoassembly for Targeted Cancer Therapy. J. Am. Chem. Soc. 2013, 135, 18644–18650. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Zheng, J.; Song, E.; Donovan, M.; Zhang, K.; Liu, C.; Tan, W. Self-assembled, aptamer-tethered DNA nanotrains for targeted transport of molecular drugs in cancer theranostics. Proc. Natl. Acad. Sci. USA 2013, 110, 7998–8003. [Google Scholar] [CrossRef]
- Pala, K.; Serwotka, A.; Jelen, F.; Jakimowicz, P.; Otlewski, J. Tumor-specific hyperthermia with aptamer-tagged superparamagnetic nanoparticles. Int. J. Nanomed. 2013, 9, 67–76. [Google Scholar] [CrossRef]
- Zhou, W.; Zhou, Y.; Wu, J.; Liu, Z.; Zhao, H.; Liu, J.; Ding, J. Aptamer-nanoparticle bioconjugates enhance intracellular delivery of vinorelbine to breast cancer cells. J. Drug Target. 2014, 22, 57–66. [Google Scholar] [CrossRef]
- Ninomiya, K.; Yamashita, T.; Kawabata, S.; Shimizu, N. Targeted and ultrasound-triggered drug delivery using liposomes co-modified with cancer cell-targeting aptamers and a thermosensitive polymer. Ultrason. Sonochem. 2014, 21, 1482–1488. [Google Scholar] [CrossRef]
- Li, L.; Hou, J.; Liu, X.; Guo, Y.; Wu, Y.; Zhang, L.; Yang, Z. Nucleolin-targeting liposomes guided by aptamer AS1411 for the delivery of siRNA for the treatment of malignant melanomas. Biomaterials 2014, 35, 3840–3850. [Google Scholar] [CrossRef]
- Xiang, D.; Shigdar, S.; Yang, W.; Duan, W.; Li, Q.; Lin, J.; Liu, K.; Li, L. Epithelial cell adhesion molecule aptamer functionalized PLGA-lecithin-curcumin-PEG nanoparticles for targeted drug delivery to human colorectal adenocarcinoma cells. Int. J. Nanomed. 2014, 9, 1083–1096. [Google Scholar] [CrossRef]
- Lale, S.V.; G, A.R.; Aravind, A.; Kumar, D.S.; Koul, V. AS1411 Aptamer and Folic Acid Functionalized pH-Responsive ATRP Fabricated pPEGMA–PCL–pPEGMA Polymeric Nanoparticles for Targeted Drug Delivery in Cancer Therapy. Biomacromolecules 2014, 15, 1737–1752. [Google Scholar] [CrossRef] [PubMed]
- Latorre, A.; Posch, C.; Garcimartín, Y.; Celli, A.; Sanlorenzo, M.; Vujic, I.; Ma, J.; Zekhtser, M.; Rappersberger, K.; Ortiz-Urda, S.; et al. DNA and aptamer stabilized gold nanoparticles for targeted delivery of anticancer therapeutics. Nanoscale 2014, 6, 7436–7442. [Google Scholar] [CrossRef] [PubMed]
- Ban, C.; Youn, H.; Lee, S.; Ban, C. Ultra-effective photothermal therapy for prostate cancer cells using dual aptamer-modified gold nanostars. J. Mater. Chem. B 2014, 2, 4862–4867. [Google Scholar] [CrossRef]
- Sayari, E.; Dinarvand, M.; Amini, M.; Azhdarzadeh, M.; Mollarazi, E.; Ghasemi, Z.; Atyabi, F. MUC1 aptamer conjugated to chitosan nanoparticles, an efficient targeted carrier designed for anticancer SN38 delivery. Int. J. Pharm. 2014, 473, 304–315. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.E.; Lee, K.H.; Park, Y.S.; Oh, D.-K.; Oh, S.; Kim, K.-S.; Kim, D.-E. RNA aptamer-conjugated liposome as an efficient anticancer drug delivery vehicle targeting cancer cells in vivo. J. Control. Release 2014, 196, 234–242. [Google Scholar] [CrossRef]
- Ryou, S.-M.; Yeom, J.-H.; Kang, H.J.; Won, M.; Kim, J.-S.; Lee, B.; Seong, M.-J.; Ha, N.-C.; Bae, J.; Lee, K. Gold nanoparticle–DNA aptamer composites as a universal carrier for in vivo delivery of biologically functional proteins. J. Control. Release 2014, 196, 287–294. [Google Scholar] [CrossRef]
- Gao, S.; Wu, X.; Tai, Z.; Zhu, Q.; Fan, W.; Ding, B.; Zhang, L.; Yao, C.; Wang, X.; Zhang, W.; et al. Study on the prostate cancer-targeting mechanism of aptamer-modified nanoparticles and their potential anticancer effect in vivo. Int. J. Nanomed. 2014, 9, 5431–5440. [Google Scholar] [CrossRef]
- Hao, Z.; Fan, W.; Hao, J.; Wu, X.; Zeng, G.Q.; Zhang, L.J.; Nie, S.F.; Wang, X. Efficient delivery of micro RNA to bone-metastatic prostate tumors by using aptamer-conjugated atelocollagen in vitro and in vivo. Drug Deliv. 2014, 23, 864–871. [Google Scholar] [CrossRef]
- Li, X.; Yu, Y.; Ji, Q.; Qiu, L. Targeted delivery of anticancer drugs by aptamer AS1411 mediated Pluronic F127/cyclodextrin-linked polymer composite micelles. Nanomed. Nanotechnol. Boil. Med. 2015, 11, 175–184. [Google Scholar] [CrossRef]
- Das, M.; Duan, W.; Sahoo, S.K. Multifunctional nanoparticle–EpCAM aptamer bioconjugates: A paradigm for targeted drug delivery and imaging in cancer therapy. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Alibolandi, M.; Ramezani, M.; Abnous, K.; Sadeghi, F.; Atyabi, F.; Asouri, M.; Ahmadi, A.A.; Hadizadeh, F. In vitro and in vivo evaluation of therapy targeting epithelial-cell adhesion-molecule aptamers for non-small cell lung cancer. J. Control. Release 2015, 209, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, K.; Kanwar, J.R.; Athalya, P.K.; Janakiraman, N.; Khetan, V.; Kanwar, R.K.; Eluchuri, S.; Krishnakumar, S. EpCAM aptamer mediated cancer cell specific delivery of EpCAM siRNA using polymeric nanocomplex. J. Biomed. Sci. 2015, 22, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Yao, H.; Meng, Y.; Wang, Y.; Yan, X.; Huang, R. Specific aptamer-conjugated mesoporous silica–carbon nanoparticles for HER2-targeted chemo-photothermal combined therapy. Acta Biomater. 2015, 16, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Tseng, Y.-T.; Suo, G.; Chen, L.; Yu-Ting, T.; Chiu, W.-J.; Huang, C.-C.; Lin, C.-H. Photothermal Therapeutic Response of Cancer Cells to Aptamer–Gold Nanoparticle-Hybridized Graphene Oxide under NIR Illumination. ACS Appl. Mater. Interfaces 2015, 7, 5097–5106. [Google Scholar] [CrossRef]
- Yasun, E.; Li, C.; Barut, I.; Janvier, D.; Qiu, L.; Cui, C.; Tan, W. BSA modification to reduce CTAB induced nonspecificity and cytotoxicity of aptamer-conjugated gold nanorods. Nanoscale 2015, 7, 10240–10248. [Google Scholar] [CrossRef]
- Alibolandi, M.; Ramezani, M.; Sadeghi, F.; Abnous, K.; Hadizadeh, F. Epithelial cell adhesion molecule aptamer conjugated PEG–PLGA nanopolymersomes for targeted delivery of doxorubicin to human breast adenocarcinoma cell line in vitro. Int. J. Pharm. 2015, 479, 241–251. [Google Scholar] [CrossRef]
- Deng, K.; Hou, Z.; Li, X.; Li, C.; Zhang, Y.; Deng, X.; Cheng, Z.; Lin, J. Aptamer-Mediated Up-conversion Core/MOF Shell Nanocomposites for Targeted Drug Delivery and Cell Imaging. Sci. Rep. 2015, 5, 7851. [Google Scholar] [CrossRef]
- Kaur, J.; Tikoo, K. Ets1 identified as a novel molecular target of RNA aptamer selected against metastatic cells for targeted delivery of nano-formulation. Oncogene 2015, 34, 5216–5228. [Google Scholar] [CrossRef]
- Ghasemi, Z.; Dinarvand, R.; Mottaghitalab, F.; Esfandyari-Manesh, M.; Sayari, E.; Atyabi, F. Aptamer decorated hyaluronan/chitosan nanoparticles for targeted delivery of 5-fluorouracil to MUC1 overexpressing adenocarcinomas. Carbohydr. Polym. 2015, 121, 190–198. [Google Scholar] [CrossRef]
- Tang, Y.; Hu, H.; Zhang, M.G.; Song, J.; Nie, L.; Wang, S.; Niu, G.; Huang, P.; Lu, G.; Chen, X. An aptamer-targeting photoresponsive drug delivery system using “off–on” graphene oxide wrapped mesoporous silica nanoparticles. Nanoscale 2015, 7, 6304–6310. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Ni, M.; Xiong, M.; Zhang, X.; Cai, G.; Chen, H.; Zeng, Q. Poly(lactic-co-glycolic acid) nanoparticles conjugated with CD133 aptamers for targeted salinomycin delivery to CD133+ osteosarcoma cancer stem cells. Int. J. Nanomed. 2015, 10, 2537–2554. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Han, Q.; Yu, N.; Li, J.; Yang, L.; Yang, R.; Wang, C. Aptamer–conjugated graphene oxide–gold nanocomposites for targeted chemo-photothermal therapy of cancer cells. J. Mater. Chem. B 2015, 3, 4036–4042. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Chen, H.; Yu, C.; Zhang, Y.; Chen, M.; Tian, S.; Sun, C. The promotion of salinomycin delivery to hepatocellular carcinoma cells through EGFR and CD133 aptamers conjugation by PLGA nanoparticles. Nanomedicine (Lond. Engl.) 2015, 10, 1863–1879. [Google Scholar] [CrossRef] [PubMed]
- Heng, C.; Wang, X.; Heng, C.; Han, Q.; Cai, S.; Li, J.; Qi, C.; Liang, W.; Yang, R.; Wang, C. Synergistically enhanced photocatalytic and chemotherapeutic effects of aptamer-functionalized ZnO nanoparticles towards cancer cells. Phys. Chem. Chem. Phys. 2015, 17, 21576–21582. [Google Scholar] [CrossRef]
- Song, X.; Ren, Y.; Zhang, J.; Wang, G.; Han, X.; Zheng, W.; Zhen, L. Targeted delivery of doxorubicin to breast cancer cells by aptamer functionalized DOTAP/DOPE liposomes. Oncol. Rep. 2015, 34, 1953–1960. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, R.; Chen, F.; Chen, M.; Wang, Y. Nucleolin targeting AS1411 aptamer modified pH-sensitive micelles: A dual-functional strategy for paclitaxel delivery. J. Control. Release 2015, 213, e137–e138. [Google Scholar] [CrossRef] [PubMed]
- Varnamkhasti, B.S.; Hosseinzadeh, H.; Azhdarzadeh, M.; Vafaei, S.Y.; Esfandyari-Manesh, M.; Mirzaie, Z.H.; Amini, M.; Ostad, S.N.; Atyabi, F.; Dinarvand, R. Protein corona hampers targeting potential of MUC1 aptamer functionalized SN-38 core–shell nanoparticles. Int. J. Pharm. 2015, 494, 430–444. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, X.; Guo, C.; Ren, D.; Zhao, Y.; Xiao, W.; Jiao, W. Aptamer-Dendrimer Bioconjugates for Targeted Delivery of miR-34a Expressing Plasmid and Antitumor Effects in Non-Small Cell Lung Cancer Cells. PLoS ONE 2015, 10, e0139136. [Google Scholar] [CrossRef]
- Askarian, S.; Abnous, K.; Taghavi, S.; Oskuee, R.K.; Ramezani, M. Cellular delivery of shRNA using aptamer-conjugated PLL-alkyl-PEI nanoparticles. Colloids Surf. B Biointerfaces 2015, 136, 355–364. [Google Scholar] [CrossRef]
- Zheng, F.-F.; Zhang, P.-H.; Xi, Y.; Chen, J.-J.; Li, L.-L.; Zhu, J. Aptamer/Graphene Quantum Dots Nanocomposite Capped Fluorescent Mesoporous Silica Nanoparticles for Intracellular Drug Delivery and Real-Time Monitoring of Drug Release. Anal. Chem. 2015, 87, 11739–11745. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, R.; Lee, A.S.W.; Yap, L.W.; Jans, D.A.; Wagstaff, K.M.; Cheng, W. Tumor cell-specific photothermal killing by SELEX-derived DNA aptamer-targeted gold nanorods. Nanoscale 2016, 8, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Azhdarzadeh, M.; Atyabi, F.; Saei, A.A.; Varnamkhasti, B.S.; Omidi, Y.; Fateh, M.; Ghavami, M.; Shanehsazzadeh, S.; Dinarvand, R. Theranostic MUC-1 aptamer targeted gold coated superparamagnetic iron oxide nanoparticles for magnetic resonance imaging and photothermal therapy of colon cancer. Colloids Surf. B Biointerfaces 2016, 143, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Hu, Y.; Yu, L.; Deng, X.; Meng, J.; Wang, C.; Yang, X.-D.; Fangqin, G.; Yan, H.; Lianyuan, Y.; et al. Enhancement of Thermal Damage to Adenocarcinoma Cells by Iron Nanoparticles Modified with MUC1 Aptamer. J. Nanosci. Nanotechnol. 2016, 16, 2246–2253. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, J.; Wang, J.; Wang, L.; Tan, X.; Tu, K.; Tong, X.; Qi, L. Aptamer Functionalized Cisplatin-Albumin Nanoparticles for Targeted Delivery to Epidermal Growth Factor Receptor Positive Cervical Cancer. J. Biomed. Nanotechnol. 2016, 12, 656–666. [Google Scholar] [CrossRef] [PubMed]
- Shoyele, S.A.; Maher, C.; Lakshmikuttyamma, A.; Perepelyuk, M. Aptamer-hybrid nanoparticle bioconjugate efficiently delivers miRNA-29b to non-small-cell lung cancer cells and inhibits growth by downregulating essential oncoproteins. Int. J. Nanomed. 2016, 11, 3533–3544. [Google Scholar] [CrossRef]
- Taghdisi, S.M.; Danesh, N.M.; Lavaee, P.; Emrani, A.S.; Hassanabad, K.Y.; Ramezani, M.; Abnous, K. Double targeting, controlled release and reversible delivery of daunorubicin to cancer cells by polyvalent aptamers-modified gold nanoparticles. Mater. Sci. Eng. C 2016, 61, 753–761. [Google Scholar] [CrossRef]
- Zaimy, M.A.; Jebali, A.; Bazrafshan, B.; Mehrtashfar, S.; Shabani, S.; Tavakoli, A.; Hekmatimoghaddam, S.H.; Sarli, A.; Azizi, H.; Izadi, P.; et al. Coinhibition of overexpressed genes in acute myeloid leukemia subtype M2 by gold nanoparticles functionalized with five antisense oligonucleotides and one anti-CD33(+)/CD34(+) aptamer. Cancer Gene Ther. 2016, 23, 315–320. [Google Scholar] [CrossRef]
- Xie, X.; Li, F.; Zhang, H.; Lu, Y.; Lian, S.; Lin, H.; Gao, Y.; Jia, L. EpCAM aptamer-functionalized mesoporous silica nanoparticles for efficient colon cancer cell-targeted drug delivery. Eur. J. Pharm. Sci. 2016, 83, 28–35. [Google Scholar] [CrossRef]
- Moosavian, S.A.; Abnous, K.; Badiee, A.; Jaafari, M.R. Improvement in the drug delivery and anti-tumor efficacy of PEGylated liposomal doxorubicin by targeting RNA aptamers in mice bearing breast tumor model. Colloids Surf. B Biointerfaces 2016, 139, 228–236. [Google Scholar] [CrossRef]
- Leach, J.C.; Wang, A.; Ye, K.; Jin, S. A RNA-DNA Hybrid Aptamer for Nanoparticle-Based Prostate Tumor Targeted Drug Delivery. Int. J. Mol. Sci. 2016, 17, 380. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xiaowei, M.; Zhao, J.; Huang, Y.; Deng, H.; Kumar, A.; Wang, C.; Liang, Z.; Ma, X.; Liang, X.-J. Multifunctional aptamer-based nanoparticles for targeted drug delivery to circumvent cancer resistance. Biomaterials 2016, 91, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Alibolandi, M.; Ramezani, M.; Abnous, K.; Hadizadeh, F. AS1411 Aptamer-Decorated Biodegradable Polyethylene Glycol–Poly(lactic-co-glycolic acid) Nanopolymersomes for the Targeted Delivery of Gemcitabine to Non–Small Cell Lung Cancer In Vitro. J. Pharm. Sci. 2016, 105, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.; Deng, H.; Su, Y.; He, L.; Wang, R.; Tong, G.; He, D.; Zhu, X. Aptamer-Functionalized and Backbone Redox-Responsive Hyperbranched Polymer for Targeted Drug Delivery in Cancer Therapy. Biomacromolecules 2016, 17, 2050–2062. [Google Scholar] [CrossRef] [PubMed]
- Taghdisi, S.M.; Danesh, N.M.; Ramezani, M.; Lavaee, P.; Jalalian, S.H.; Robati, R.Y.; Abnous, K. Double targeting and aptamer-assisted controlled release delivery of epirubicin to cancer cells by aptamers-based dendrimer in vitro and in vivo. Eur. J. Pharm. Biopharm. 2016, 102, 152–158. [Google Scholar] [CrossRef]
- Jiao, J.; Zou, Q.; Zou, M.H.; Zhu, S.; Guo, R.M.; Zhang, Y. Aptamer-modified PLGA nanoparticle delivery of triplex forming oligonucleotide for targeted prostate cancer therapy. Neoplasma 2016, 63, 569–575. [Google Scholar] [CrossRef]
- Chen, Z.; Tai, Z.; Gu, F.; Hu, C.; Zhu, Q.; Gao, S. Aptamer-mediated delivery of docetaxel to prostate cancer through polymeric nanoparticles for enhancement of antitumor efficacy. Eur. J. Pharm. Biopharm. 2016, 107, 130–141. [Google Scholar] [CrossRef]
- Tao, W.; Xu, G.; Yu, X.; Zhang, J.; Sheng, Y.; Liu, G.; Mei, L. Robust aptamer–polydopamine-functionalized M-PLGA–TPGS nanoparticles for targeted delivery of docetaxel and enhanced cervical cancer therapy. Int. J. Nanomed. 2016, 11, 2953–2965. [Google Scholar] [CrossRef]
- Pi, F.; Zhang, H.; Li, H.; Thiviyanathan, V.; Gorenstein, D.G.; Sood, A.K.; Guo, P. RNA nanoparticles harboring annexin A2 aptamer can target ovarian cancer for tumor-specific doxorubicin delivery. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 1183–1193. [Google Scholar] [CrossRef]
- Esfandyari-Manesh, M.; Mohammadi, A.; Atyabi, F.; Nabavi, S.M.; Ebrahimi, S.M.; Shahmoradi, E.; Varnamkhasti, B.S.; Ghahremani, M.H.; Dinarvand, R. Specific targeting delivery to MUC1 overexpressing tumors by albumin-chitosan nanoparticles conjugated to DNA aptamer. Int. J. Pharm. 2016, 515, 607–615. [Google Scholar] [CrossRef]
- Behrooz, A.B.; Nabavizadeh, F.; Adiban, J.; Ardestani, M.S.; Vahabpour, R.; Aghasadeghi, M.R.; Sohanaki, H. Smart bomb AS1411 aptamer-functionalized/PAMAM dendrimer nanocarriers for targeted drug delivery in the treatment of gastric cancer. Clin. Exp. Pharmacol. Physiol. 2016, 44, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Tian, J.; Liu, D.; He, W.; Guo, Z. Dual aptamer modified dendrigraft poly-l-lysine nanoparticles for overcoming multi-drug resistance through mitochondrial targeting. J. Mater. Chem. B 2017, 5, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Jurek, P.M.; Zabłocki, K.; Waśko, U.; Mazurek, M.P.; Otlewski, J.; Jeleń, F. Anti-FGFR1 aptamer-tagged superparamagnetic conjugates for anticancer hyperthermia therapy. Int. J. Nanomed. 2017, 12, 2941–2950. [Google Scholar] [CrossRef] [PubMed]
- Zhen, S.; Takahashi, Y.; Narita, S.; Yang, Y.-C.; Li, X. Targeted delivery of CRISPR/Cas9 to prostate cancer by modified gRNA using a flexible aptamer-cationic liposome. Oncotarget 2016, 8, 9375–9387. [Google Scholar] [CrossRef] [PubMed]
- Alibolandi, M.; Taghdisi, S.M.; Ramezani, P.; Shamili, F.H.; Farzad, S.A.; Abnous, K.; Ramezani, M. Smart AS1411-aptamer conjugated pegylated PAMAM dendrimer for the superior delivery of camptothecin to colon adenocarcinoma In Vitro and In Vivo. Int. J. Pharm. 2017, 519, 352–364. [Google Scholar] [CrossRef]
- Powell, D.; Chandra, S.; Dodson, K.; Shaheen, F.; Wiltz, K.; Ireland, S.; Syed, M.; Dash, S.; Wiese, T.; Mandal, T.; et al. Aptamer-functionalized hybrid nanoparticle for the treatment of breast cancer. Eur. J. Pharm. Biopharm. 2017, 114, 108–118. [Google Scholar] [CrossRef]
- Li, F.; Mei, H.; Xie, X.; Zhang, H.; Liu, J.; Lv, T.; Nie, H.; Gao, Y.; Jia, L. Aptamer-Conjugated Chitosan-Anchored Liposomal Complexes for Targeted Delivery of Erlotinib to EGFR-Mutated Lung Cancer Cells. AAPS J. 2017, 19, 814–826. [Google Scholar] [CrossRef]
- Kolovskaya, O.S.; Zamay, T.N.; Belyanina, I.V.; Karlova, E.; Garanzha, I.; Aleksandrovsky, A.; Kirichenko, A.; Dubynina, A.V.; Sokolov, A.E.; Zamay, G.S.; et al. Aptamer-Targeted Plasmonic Photothermal Therapy of Cancer. Mol. Ther. Nucleic Acids 2017, 9, 12–21. [Google Scholar] [CrossRef]
- Atabi, F.; Gargari, S.M.; Hashemi, M.; Yaghmaei, P. Doxorubicin Loaded DNA Aptamer Linked Myristilated Chitosan Nanogel for Targeted Drug Delivery to Prostate Cancer. Iran. J. Pharm. Res. IJPR 2017, 16, 35–49. [Google Scholar]
- Zhao, F.; Zhou, J.; Su, X.; Wang, Y.; Yan, X.; Jia, S.; Du, B. A Smart Responsive Dual Aptamers-Targeted Bubble-Generating Nanosystem for Cancer Triplex Therapy and Ultrasound Imaging. Small 2017, 13. [Google Scholar] [CrossRef]
- Taghavi, S.; Ramezani, M.; Alibolandi, M.; Abnous, K.; Taghdisi, S.M. Chitosan-modified PLGA nanoparticles tagged with 5TR1 aptamer for in vivo tumor-targeted drug delivery. Cancer Lett. 2017, 400, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ayatollahi, S.; Salmasi, Z.; Hashemi, M.; Askarian, S.; Oskuee, R.K.; Abnous, K.; Ramezani, M. Aptamer-targeted delivery of Bcl-xL shRNA using alkyl modified PAMAM dendrimers into lung cancer cells. Int. J. Biochem. Cell Biol. 2017, 92, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Monaco, I.; Camorani, S.; Colecchia, D.; Locatelli, E.; Calandro, P.; Oudin, A.; Niclou, S.; Arra, C.; Chiariello, M.; Cerchia, L.; et al. Aptamer Functionalization of Nanosystems for Glioblastoma Targeting through the Blood–Brain Barrier. J. Med. Chem. 2017, 60, 4510–4516. [Google Scholar] [CrossRef] [PubMed]
- Hanafi-Bojd, M.Y.; Kalat, S.A.M.; Taghdisi, S.M.; Ansari, L.; Abnous, K.; Malaekeh-Nikouei, B. MUC1 aptamer-conjugated mesoporous silica nanoparticles effectively target breast cancer cells. Drug Dev. Ind. Pharm. 2018, 44, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Pascual, L.; Cerqueira-Coutinho, C.; García-Fernández, A.; De Luis, B.; Bernardes, E.S.; Albernaz, M.S.; Missailidis, S.; Martínez-Máñez, R.; Santos-Oliveira, R.; Orzaez, M.; et al. MUC1 aptamer-capped mesoporous silica nanoparticles for controlled drug delivery and radio-imaging applications. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2495–2505. [Google Scholar] [CrossRef]
- Li, F.; Mei, H.; Gao, Y.; Xie, X.; Nie, H.; Li, T.; Zhang, H.; Jia, L. Co-delivery of oxygen and erlotinib by aptamer-modified liposomal complexes to reverse hypoxia-induced drug resistance in lung cancer. Biomaterials 2017, 145, 56–71. [Google Scholar] [CrossRef]
- Zhao, J.; Tu, K.; Liu, Y.; Qin, Y.; Wang, X.; Qi, L.; Shi, D. Photo-controlled aptamers delivery by dual surface gold-magnetic nanoparticles for targeted cancer therapy. Mater. Sci. Eng. C 2017, 80, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chang, Y.-Q.; Han, L.; Zhang, Y.; Wang, W.-J.; Shu, Y.; Wang, J.-H. Aptamer-anchored di-polymer shell-capped mesoporous carbon as a drug carrier for bi-trigger targeted drug delivery. J. Mater. Chem. B 2017, 5, 6882–6889. [Google Scholar] [CrossRef]
- Guo, X.; Zhu, X.; Gao, J.; Liu, D.; Dong, C.; Jin, X. PLGA nanoparticles with CD133 aptamers for targeted delivery and sustained release of propranolol to hemangioma. Nanomedicine 2017, 12, 2611–2624. [Google Scholar] [CrossRef]
- Prisner, L.; Bohn, N.; Hahn, U.; Mews, A. Size dependent targeted delivery of gold nanoparticles modified with the IL-6R-specific aptamer AIR-3A to IL-6R-carrying cells. Nanoscale 2017, 9, 14486–14498. [Google Scholar] [CrossRef]
- Li, Y.; Duo, Y.; Bao, S.; He, L.; Ling, K.; Luo, J.; Zhang, Y.; Huang, H.; Zhang, H.; Yu, X. EpCAM aptamer-functionalized polydopamine-coated mesoporous silica nanoparticles loaded with DM1 for targeted therapy in colorectal cancer. Int. J. Nanomed. 2017, 12, 6239–6257. [Google Scholar] [CrossRef]
- Belyanina, I.V.; Zamay, T.N.; Zamay, G.S.; Zamay, S.S.; Kolovskaya, O.S.; Ivanchenko, T.I.; Denisenko, V.V.; Kirichenko, A.K.; Glazyrin, Y.E.; Garanzha, I.V.; et al. In Vivo Cancer Cells Elimination Guided by Aptamer-Functionalized Gold-Coated Magnetic Nanoparticles and Controlled with Low Frequency Alternating Magnetic Field. Theranostics 2017, 7, 3326–3337. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Ren, Z.; Chen, M.; Wang, Y.; You, B.; Chen, W.; Qu, C.; Liu, Y.; Zhang, X.-N. Nucleolin-Targeting AS1411-Aptamer-Modified Graft Polymeric Micelle with Dual pH/Redox Sensitivity Designed to Enhance Tumor Therapy through the Codelivery of Doxorubicin/TLR4 siRNA and Suppression of Invasion. Mol. Pharm. 2018, 15, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.A.; Pearce, T.R.; Pengo, T.; Kuang, H.; Forster, C.; Kokkoli, E. Aptamer micelles targeting fractalkine-expressing cancer cells In Vitro and In Vivo. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Han, L.; Guo, Y.; Zheng, G.; Fan, L.; Shen, Z.; Zhao, R.; Shao, J. A carrier-free dual-drug nanodelivery system functionalized with aptamer specific targeting HER2-overexpressing cancer cells. J. Mater. Chem. B 2017, 5, 9121–9129. [Google Scholar] [CrossRef] [PubMed]
- Aghanejad, A.; Babamiri, H.; Adibkia, K.; Barar, J.; Omidi, Y. Mucin-1 aptamer-armed superparamagnetic iron oxide nanoparticles for targeted delivery of doxorubicin to breast cancer cells. BioImpacts 2018, 8, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Willner, I.; Sung, S.Y.; Fadeev, M.; Cecconello, A.; Nechushtai, R.; Willner, I. Targeted VEGF-triggered release of an anti-cancer drug from aptamer-functionalized metal–organic framework nanoparticles. Nanoscale 2018, 10, 4650–4657. [Google Scholar] [CrossRef]
- Jalalian, S.H.; Ramezani, M.; Abnous, K.; Taghdisi, S.M. Targeted co-delivery of epirubicin and NAS-24 aptamer to cancer cells using selenium nanoparticles for enhancing tumor response in vitro and in vivo. Cancer Lett. 2018, 416, 87–93. [Google Scholar] [CrossRef]
- Dou, X.-Q.; Wang, H.; Zhang, J.; Wang, F.; Xu, G.-L.; Xu, C.-C.; Xu, H.-H.; Xiang, S.-S.; Fu, J.; Song, H.-F. Aptamer–drug conjugate: Targeted delivery of doxorubicin in a HER3 aptamer-functionalized liposomal delivery system reduces cardiotoxicity. Int. J. Nanomed. 2018, 13, 763–776. [Google Scholar] [CrossRef]
- Srivithya, V.; Roun, H.; Babu, M.S.; Hyung, P.J.; Ha, P.S. Aptamer-conjugated DNA nano-ring as the carrier of drug molecules. Nanotechnology 2018, 29, 095602. [Google Scholar] [CrossRef]
- Wu, M.; Zhao, H.; Guo, L.; Wang, Y.; Song, J.; Zhao, X.; Li, C.; Hao, L.; Wang, D.; Tang, J. Ultrasound-mediated nanobubble destruction (UMND) facilitates the delivery of A10-3.2 aptamer targeted and siRNA-loaded cationic nanobubbles for therapy of prostate cancer. Drug Deliv. 2018, 25, 226–240. [Google Scholar] [CrossRef] [PubMed]
- Charbgoo, F.; Alibolandi, M.; Taghdisi, S.M.; Abnous, K.; Soltani, F.; Ramezani, M. MUC1 aptamer-targeted DNA micelles for dual tumor therapy using doxorubicin and KLA peptide. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 685–697. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.-B.; Yu, Z.-C.; He, Y.-N.; Zhang, T.; Du, L.-B.; Dong, Y.-M.; Chen, H.-W.; Zhang, Y.-Y.; Wang, W.-Q. Salinomycin-loaded lipid-polymer nanoparticles with anti-CD20 aptamers selectively suppress human CD20+ melanoma stem cells. Acta Pharmacol. Sin. 2017, 39, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Chen, F.; Qi, X.; Dong, Y.; Zhang, Y.; Ge, Z.; Cai, G.; Zhang, X. Epidermal growth factor receptor aptamer‑conjugated polymer‑lipid hybrid nanoparticles enhance salinomycin delivery to osteosarcoma and cancer stem cells. Exp. Ther. Med. 2017, 15, 1247–1256. [Google Scholar] [CrossRef]
- Prusty, D.K.; Adam, V.; Zadegan, R.; Irsen, S.; Famulok, M. Supramolecular aptamer nano-constructs for receptor-mediated targeting and light-triggered release of chemotherapeutics into cancer cells. Nat. Commun. 2018, 9, 535. [Google Scholar] [CrossRef]
- Cheng, Y.; Ou, Z.; Li, Q.; Yang, J.; Hu, M.; Zhou, Y.; Zhuang, X.; Zhang, Z.J.; Guan, S. Cabazitaxel liposomes with aptamer modification enhance tumor‑targeting efficacy in nude mice. Mol. Med. Rep. 2018, 19, 490–498. [Google Scholar] [CrossRef]
- Ghassami, E.; Varshosaz, J.; Jahanian-Najafabadi, A.; Minaiyan, M.; Rajabi, P.; Hayati, E. Pharmacokinetics and In Vitro/In Vivo antitumor efficacy of aptamer-targeted Ecoflex((R)) nanoparticles for docetaxel delivery in ovarian cancer. Int. J. Nanomed. 2018, 13, 493–504. [Google Scholar] [CrossRef]
- Yang, H.; Lu, W.-L.; Huang, T.; Chen, Q.-Y.; Gao, J.; Zhao, Y. An aptamer-Fe3+ modified nanoparticle for lactate oxidation and tumor photodynamic therapy. Colloids Surf. B Biointerfaces 2018, 164, 192–200. [Google Scholar] [CrossRef]
- Tekie, F.S.M.; Soleimani, M.; Zakerian, A.; Dinarvand, M.; Amini, M.; Dinarvand, R.; Arefian, E.; Atyabi, F. Glutathione responsive chitosan-thiolated dextran conjugated miR-145 nanoparticles targeted with AS1411 aptamer for cancer treatment. Carbohydr. Polym. 2018, 201, 131–140. [Google Scholar] [CrossRef]
- Zhao, N.; Zeng, Z.; Zu, Y. Self-Assembled Aptamer-Nanomedicine for Targeted Chemotherapy and Gene Therapy. Small 2017, 14, 1702103. [Google Scholar] [CrossRef]
- Perepelyuk, M.; Sacko, K.; Thangavel, K.; Shoyele, S.A. Evaluation of MUC1-Aptamer Functionalized Hybrid Nanoparticles for Targeted Delivery of miRNA-29b to Nonsmall Cell Lung Cancer. Mol. Pharm. 2018, 15, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Engelberg, S.; Modrejewski, J.; Walter, J.G.; Livney, Y.D.; Assaraf, Y.G. Cancer cell-selective, clathrin-mediated endocytosis of aptamer decorated nanoparticles. Oncotarget 2018, 9, 20993–21006. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Zeng, Y.; Qi, X.; Chen, Y.; Ge, Z.; Jiang, Z.; Zhang, X.; Dong, Y.; Chen, H.; Yu, Z. Targeted salinomycin delivery with EGFR and CD133 aptamers based dual-ligand lipid-polymer nanoparticles to both osteosarcoma cells and cancer stem cells. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2115–2127. [Google Scholar] [CrossRef] [PubMed]
- Alibolandi, M.; Hoseini, F.; Mohammadi, M.; Ramezani, P.; Einafshar, E.; Taghdisi, S.M.; Ramezani, M.; Abnous, K. Curcumin-entrapped MUC-1 aptamer targeted dendrimer-gold hybrid nanostructure as a theranostic system for colon adenocarcinoma. Int. J. Pharm. 2018, 549, 67–75. [Google Scholar] [CrossRef]
- Ma, J.; Zhuang, H.; Zhuang, Z.; Lu, Y.; Xia, R.; Gan, L.; Wu, Y. Development of docetaxel liposome surface modified with CD133 aptamers for lung cancer targeting. Artif. Cells Nanomed. Biotechnol. 2017, 46, 1–8. [Google Scholar] [CrossRef]
- Moosavian, S.A.; Abnous, K.; Akhtari, J.; Arabi, L.; Dewin, A.G.; Jaafari, M.R. 5TR1 aptamer-PEGylated liposomal doxorubicin enhances cellular uptake and suppresses tumour growth by targeting MUC1 on the surface of cancer cells. Artif. Cells Nanomed. Biotechnol. 2017, 46, 1–12. [Google Scholar] [CrossRef]
- Xu, L.; He, X.-Y.; Liu, B.-Y.; Xu, C.; Ai, S.-L.; Zhuo, R.-X.; Cheng, S.-X. Aptamer-functionalized albumin-based nanoparticles for targeted drug delivery. Colloids Surf. B Biointerfaces 2018, 171, 24–30. [Google Scholar] [CrossRef]
- Wu, D.; Wang, W.; He, X.; Jiang, M.; Lai, C.; Hu, X.; Xi, J.; Wang, M. Biofabrication of nano copper oxide and its aptamer bioconjugate for delivery of mRNA 29b to lung cancer cells. Mater. Sci. Eng. C 2019, 97, 827–832. [Google Scholar] [CrossRef]
- Zhaoa, C.; Songa, X.; Jinc, W.; Wub, F.; Zhangb, Q.; Zhangab, M.; Zhou, N.; Shenb, J. Image-guided cancer therapy using aptamer-functionalized cross-linked magnetic-responsive Fe3O4@carbon nanoparticles. Anal. Chim. Acta 2019, 1056, 108–116. [Google Scholar] [CrossRef]
- Guan, Z.; Zhang, T.; Zhu, H.; Lyu, D.; Sarangapani, S.; Xu, Q.-H.; Lang, M.J. Simultaneous Imaging and Selective Photothermal Therapy through Aptamer-Driven Au Nanosphere Clustering. J. Phys. Chem. Lett. 2018, 10, 183–188. [Google Scholar] [CrossRef]
- Kalinowska, D.; Grabowska-Jadach, I.; Liwińska, M.; Drozd, M.; Pietrzak, M.; Dybko, A.; Brzózka, Z. Studies on effectiveness of PTT on 3D tumor model under microfluidic conditions using aptamer-modified nanoshells. Biosens. Bioelectron. 2019, 126, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.D.; Cardwell, L.N.; Porciani, D.; A Nguyen, J.; Zhang, R.; Gallazzi, F.; Tata, R.R.; Burke, D.H.; Daniels, M.A.; Ulery, B.D. Aptamer-displaying peptide amphiphile micelles as a cell-targeted delivery vehicle of peptide cargoes. Phys. Biol. 2018, 15, 065006. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Cao, Y.; Shen, H.; Ma, Q.; Mao, S.; Li, S.; Sun, J. EGFR aptamer-conjugated liposome-polycation-DNA complex for targeted delivery of SATB1 small interfering RNA to choriocarcinoma cells. Biomed. Pharmacother. 2018, 107, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Vandghanooni, S.; Eskandani, M.; Barar, J.; Omidi, Y. AS1411 aptamer-decorated cisplatin-loaded poly(lactic-co-glycolic acid) nanoparticles for targeted therapy of miR-21-inhibited ovarian cancer cells. Nanomedicine 2018, 13, 2729–2758. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, J.; Sun, J.; Huang, L.; Li, Q. Targeting lung cancer initiating cells by all‑trans retinoic acid‑loaded lipid‑PLGA nanoparticles with CD133 aptamers. Exp. Ther. Med. 2018, 16, 4639–4649. [Google Scholar] [CrossRef] [PubMed]
- Engelberg, S.; Netzer, E.; Assaraf, Y.G.; Livney, Y.D. Selective eradication of human non-small cell lung cancer cells using aptamer-decorated nanoparticles harboring a cytotoxic drug cargo. Cell Death Dis. 2019, 10, 1–14. [Google Scholar] [CrossRef]
- Mie, M.; Matsumoto, R.; Mashimo, Y.; Cass, A.E.G.; Kobatake, E. Development of drug-loaded protein nanoparticles displaying enzymatically-conjugated DNA aptamers for cancer cell targeting. Mol. Biol. Rep. 2018, 46, 261–269. [Google Scholar] [CrossRef]
- Bahreyni, A.; Alibolandi, M.; Ramezani, M.; Sarafan Sadeghi, A.; Abnous, K.; Taghdisi, S.M. A novel MUC1 aptamer-modified PLGA-epirubicin-PbetaAE-antimir-21 nanocomplex platform for targeted co-delivery of anticancer agents In Vitro and In Vivo. Colloids Surf. B Biointerfaces 2019, 175, 231–238. [Google Scholar] [CrossRef]
- Gui, K.; Zhang, X.; Chen, F.; Ge, Z.; Zhang, S.; Qi, X.; Sun, J.; Yu, Z. Lipid-polymer nanoparticles with CD133 aptamers for targeted delivery of all-trans retinoic acid to osteosarcoma initiating cells. Biomed. Pharmacother. 2019, 111, 751–764. [Google Scholar] [CrossRef]
- Yazdian-Robati, R.; Arab, A.; Ramezani, M.; Rafatpanah, H.; Bahreyni, A.; Nabavinia, M.S.; Abnous, K.; Taghdisi, S.M. Smart aptamer-modified calcium carbonate nanoparticles for controlled release and targeted delivery of epirubicin and melittin into cancer cells In Vitro and In Vivo. Drug Dev. Ind. Pharm. 2019, 45, 603–610. [Google Scholar] [CrossRef]
- Doerflinger, A.; Quang, N.N.; Gravel, E.; Ducongé, F.; Doris, E. Aptamer-decorated polydiacetylene micelles with improved targeting of cancer cells. Int. J. Pharm. 2019, 565, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Sacko, K.; Thangavel, K.; Shoyele, S.A. Codelivery of Genistein and miRNA-29b to A549 Cells Using Aptamer-Hybrid Nanoparticle Bioconjugates. Nanomaterials 2019, 9, 1052. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.W.; Jeong, H.Y.; Kang, S.J.; Jeong, I.H.; Choi, M.J.; You, Y.M.; Im, C.S.; Song, I.H.; Lee, T.S.; Lee, J.S.; et al. Anti-EGF Receptor Aptamer-Guided Co-Delivery of Anti-Cancer siRNAs and Quantum Dots for Theranostics of Triple-Negative Breast Cancer. Theranostics 2019, 9, 837–852. [Google Scholar] [CrossRef] [PubMed]
- Saleh, T.; Soudi, T.; Shojaosadati, S.A. Aptamer functionalized curcumin-loaded human serum albumin (HSA) nanoparticles for targeted delivery to HER-2 positive breast cancer cells. Int. J. Biol. Macromol. 2019, 130, 109–116. [Google Scholar] [CrossRef]
- Sakhtianchi, R.; Darvishi, B.; Mirzaie, Z.; Dorkoosh, F.; Shanehsazzadeh, S.; Dinarvand, R. Pegylated magnetic mesoporous silica nanoparticles decorated with AS1411 Aptamer as a targeting delivery system for cytotoxic agents. Pharm. Dev. Technol. 2019, 24, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Baneshi, M.; Dadfarnia, S.; Shabani, A.M.H.; Sabbagh, S.K.; Haghgoo, S.; Bardania, H. A novel theranostic system of AS1411 aptamer-functionalized albumin nanoparticles loaded on iron oxide and gold nanoparticles for doxorubicin delivery. Int. J. Pharm. 2019, 564, 145–152. [Google Scholar] [CrossRef]
- Luo, X.; Yang, Y.; Kong, F.; Zhang, L.; Wei, K. CD30 aptamer-functionalized PEG-PLGA nanoparticles for the superior delivery of doxorubicin to anaplastic large cell lymphoma cells. Int. J. Pharm. 2019, 564, 340–349. [Google Scholar] [CrossRef]
- Yu, S.; Bi, X.; Yang, L.; Wu, S.; Yu, Y.; Jiang, B.; Zhang, A.; Lan, K.; Duan, S. Co-Delivery of Paclitaxel and PLK1-Targeted siRNA Using Aptamer-Functionalized Cationic Liposome for Synergistic Anti-Breast Cancer Effects In Vivo. J. Biomed. Nanotechnol. 2019, 15, 1135–1148. [Google Scholar] [CrossRef]
- Li, X.; Wu, X.; Yang, H.; Li, L.; Ye, Z.; Rao, Y.-F. A nuclear targeted Dox-aptamer loaded liposome delivery platform for the circumvention of drug resistance in breast cancer. Biomed. Pharmacother. 2019, 117, 109072. [Google Scholar] [CrossRef]
- Cadinoiu, A.N.; Rata, D.M.; Atanase, L.-I.; Daraba, O.M.; Gherghel, D.; Vochita, G.; Popa, M. Aptamer-Functionalized Liposomes as a Potential Treatment for Basal Cell Carcinoma. Polymers 2019, 11, 1515. [Google Scholar] [CrossRef]
- Sun, X.; Liu, B.; Chen, X.; Lin, H.; Peng, Y.; Li, Y.; Zheng, H.; Xu, Y.; Ou, X.; Yan, S.; et al. Aptamer-assisted superparamagnetic iron oxide nanoparticles as multifunctional drug delivery platform for chemo-photodynamic combination therapy. J. Mater. Sci. Mater. Med. 2019, 30, 76. [Google Scholar] [CrossRef] [PubMed]
- Duan, T.; Xu, Z.; Sun, F.; Wang, Y.; Zhang, J.; Luo, C.; Wang, M. HPA aptamer functionalized paclitaxel-loaded PLGA nanoparticles for enhanced anticancer therapy through targeted effects and microenvironment modulation. Biomed. Pharmacother. 2019, 117, 109121. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, K.; Zhuang, L.; Liu, J.; Zeng, W.; Shi, J.; Zhang, Z. Aptamer/photosensitizer hybridized mesoporous MnO2 based tumor cell activated ROS regulator for precise photodynamic therapy of breast cancer. Colloids Surf. B Biointerfaces 2019, 184, 110536. [Google Scholar] [CrossRef] [PubMed]
- Saravanakumar, K.; Hu, X.; Shanmugam, S.; Chelliah, R.; Sekar, P.; Oh, D.-H.; Vijayakumar, S.; Kathiresan, K.; Wang, M.-H. Enhanced cancer therapy with pH-dependent and aptamer functionalized doxorubicin loaded polymeric (poly D, L-lactic-co-glycolic acid) nanoparticles. Arch. Biochem. Biophys. 2019, 671, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Zhao, Z.; Wang, Y.; Zou, J.; Lin, Q.; Duan, Y. One-Step Self-Assembly of Multifunctional DNA Nanohydrogels: An Enhanced and Harmless Strategy for Guiding Combined Antitumor Therapy. ACS Appl. Mater. Interfaces 2019, 11, 46479–46489. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhu, Z.; Zhang, G.; Lin, F.; Liu, Y.; Zhang, Y.; Feng, J.; Chen, W.; Meng, Q.; Xu, B. AS1411 Aptamer/Hyaluronic Acid-Bifunctionalized Microemulsion Co-Loading Shikonin and Docetaxel for Enhanced Antiglioma Therapy. J. Pharm. Sci. 2019, 108, 3684–3694. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, J.; Le, V.M.; Gong, Q.; Li, S.; Gao, F.; Ni, L.; Liu, J.; Liang, X. EpCAM Aptamer-Functionalized Cationic Liposome-Based Nanoparticles Loaded with miR-139-5p for Targeted Therapy in Colorectal Cancer. Mol. Pharm. 2019, 16, 4696–4710. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, W.; Tan, W.; Lai, Z.; Fang, D.; Jiang, L.; Zuo, C.; Yang, N.; Lai, Y. An Efficient Cell-Targeting Drug Delivery System Based on Aptamer-Modified Mesoporous Silica Nanoparticles. Nanoscale Res. Lett. 2019, 14, 390. [Google Scholar] [CrossRef]
- Shi, S.; Fu, W.; Lin, S.; Tian, T.; Li, S.; Shao, X.; Zhang, Y.; Zhang, T.; Tang, Z.; Zhou, Y.; et al. Targeted and effective glioblastoma therapy via aptamer-modified tetrahedral framework nucleic acid-paclitaxel nanoconjugates that can pass the blood brain barrier. Nanomed. Nanotechnol. Biol. Med. 2019, 21, 102061. [Google Scholar] [CrossRef]
- Wang, T.; Luo, Y.; Lv, H.; Wang, J.; Zhang, Y.; Pei, R. Aptamer-Based Erythrocyte-Derived Mimic Vesicles Loaded with siRNA and Doxorubicin for the Targeted Treatment of Multidrug-Resistant Tumors. ACS Appl. Mater. Interfaces 2019, 11, 45455–45466. [Google Scholar] [CrossRef]
- Xu, Z.; Ni, R.; Chen, Y. Targeting breast cancer stem cells by a self-assembled, aptamer-conjugated DNA nanotrain with preloading doxorubicin. Int. J. Nanomed. 2019, 14, 6831–6842. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Qian, W.; Tao, W.; Zhou, Y.; Xue, B. Delivery Of Curcumin Nanoliposomes Using Surface Modified With CD133 Aptamers For Prostate Cancer. Drug Des. Dev. Ther. 2019, 13, 4021–4033. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Liu, P.; Ma, J.; Li, D.; Yang, H.; Chen, W.; Jiang, Y. Enhancement of Radiosensitization by Silver Nanoparticles Functionalized with Polyethylene Glycol and Aptamer As1411 for Glioma Irradiation Therapy. Int. J. Nanomed. 2019, 14, 9483–9496. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Liang, Y.; Zhong, X.; Liang, Z.; Tian, Y.; Li, S.; Liang, J.; Wang, R.; Zhong, Y.; Shi, Y.; et al. Aptamer-Conjugated Gold Nanoparticles Targeting Epidermal Growth Factor Receptor Variant III for the Treatment of Glioblastoma. Int. J. Nanomed. 2020, 15, 1363–1372. [Google Scholar] [CrossRef] [PubMed]
- Erin, N.; Dilmaç, S.; Curry, A.; Duymuş, Ö.; Tanriover, G.; Prodeus, A.; Gariepy, J.; Gorczynski, R.M. CD200 mimetic aptamer PEG-M49 markedly increases the therapeutic effects of pegylated liposomal doxorubicin in a mouse model of metastatic breast carcinoma: An effect independent of CD200 receptor 1. Cancer Immunol. Immunother. 2019, 69, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, C.; Zhang, S.; Xue, C.; Yu, X.; Xu, H.; Wang, Z.; Lu, Y.; Wu, Z.-S. Precision-Guided Missile-Like DNA Nanostructure Containing Warhead and Guidance Control for Aptamer-Based Targeted Drug Delivery into Cancer Cells In Vitro and In Vivo. J. Am. Chem. Soc. 2020, 142, 1265–1277. [Google Scholar] [CrossRef]
- Pan, Q.; Nie, C.; Hu, Y.; Yi, J.; Liu, C.; Zhang, J.; He, M.; He, M.; Chen, T.-T.; Chu, X. Aptamer-Functionalized DNA Origami for Targeted Codelivery of Antisense Oligonucleotides and Doxorubicin to Enhance Therapy in Drug-Resistant Cancer Cells. ACS Appl. Mater. Interfaces 2019, 12, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, S.; Yang, Z.; Hoshika, S.; Xie, S.; Li, J.; Chen, X.; Wan, S.; Li, L.; A Benner, S.; et al. An Aptamer-Nanotrain Assembled from Six-Letter DNA Delivers Doxorubicin Selectively to Liver Cancer Cells. Angew. Chem. Int. Ed. 2020, 59, 663–668. [Google Scholar] [CrossRef]
- Siminzar, P.; Omidi, Y.; Golchin, A.; Aghanejad, A.; Barar, J. Targeted delivery of doxorubicin by magnetic mesoporous silica nanoparticles armed with mucin-1 aptamer. J. Drug Target. 2019, 28, 92–101. [Google Scholar] [CrossRef]
- Lin, H.-C.; Li, W.-T.; Madanayake, T.W.; Tao, C.; Niu, Q.; Yan, S.-Q.; Gao, B.-A.; Ping, Z. Aptamer-guided upconversion nanoplatform for targeted drug delivery and near-infrared light-triggered photodynamic therapy. J. Biomater. Appl. 2019, 34, 875–888. [Google Scholar] [CrossRef]
- Kolovskaya, O.S.; Zamay, T.N.; Zamay, G.S.; Babkin, V.A.; Medvedeva, E.N.; Neverova, N.A.; Kirichenko, A.K.; Zamay, S.; Lapin, I.; Morozov, E.V.; et al. Aptamer-Conjugated Superparamagnetic Ferroarabinogalactan Nanoparticles for Targeted Magnetodynamic Therapy of Cancer. Cancers 2020, 12, 216. [Google Scholar] [CrossRef] [PubMed]
- Kardani, A.; Yaghoobi, H.; Alibakhshi, A.; Khatami, M. Inhibition of miR-155 in MCF-7 breast cancer cell line by gold nanoparticles functionalized with antagomir and AS1411 aptamer. J. Cell. Physiol. 2020, 235, 6887–6895. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Dlie, Z.Y.; Mukherjee, B.; Besra, S.E.; Sengupta, S.; Sen, R.; Mukherjee, A. A Comparative Investigation of the Ability of Various Aptamer-Functionalized Drug Nanocarriers to Induce Selective Apoptosis in Neoplastic Hepatocytes: In Vitro and In Vivo Outcome. AAPS PharmSciTech 2020, 21, 89. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Dlie, Z.Y.; Chakraborty, S.; Roy, S.; Mukherjee, B.; Besra, S.E.; Dewanjee, S.; Mukherjee, A.; Ojha, P.K.; Kumar, V.; et al. Aptamer-Functionalized Drug Nanocarrier Improves Hepatocellular Carcinoma toward Normal by Targeting Neoplastic Hepatocytes. Mol. Ther. Nucleic Acids 2020, 20, 34–49. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Li, J.; Wu, C.; Yan, F.; Li, X.; Zhang, S. A self-assembled RNA-triple helix hydrogel drug delivery system targeting triple-negative breast cancer. J. Mater. Chem. B 2020, 8, 3527–3533. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Ren, X.; Peng, Y.; Zhang, J.; Ai, S.; Liu, B.; Xu, C.; Cheng, S.-X. Aptamer/Peptide-Functionalized Genome-Editing System for Effective Immune Restoration through Reversal of PD-L1-Mediated Cancer Immunosuppression. Adv. Mater. 2020, 32, e2000208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Cheng, W.; Pan, Y.; Jia, L. An anticancer agent-loaded PLGA nanomedicine with glutathione-response and targeted delivery for the treatment of lung cancer. J. Mater. Chem. B 2020, 8, 655–665. [Google Scholar] [CrossRef]
- Chen, R.; Sun, P.; Chu, X.; Pu, X.; Yang, Y.; Zhang, N.; Zhao, Y. Synergistic Treatment of Tumor by Targeted Biotherapy and Chemotherapy via Site-Specific Anchoring of Aptamers on DNA Nanotubes. Int. J. Nanomed. 2020, 15, 1309–1320. [Google Scholar] [CrossRef]
- Guo, W.; Mashimo, Y.; Kobatake, E.; Mie, M. Construction of DNA-displaying nanoparticles by enzymatic conjugation of DNA and elastin-like polypeptides using a replication initiation protein. Nanotechnology 2020, 31, 255102. [Google Scholar] [CrossRef]
- Xu, C.-M.; Tang, M.; Feng, J.; Xia, H.-F.; Wu, L.-L.; Pang, D.-W.; Chen, G.; Zhang, Z.-L. A liquid biopsy-guided drug release system for cancer theranostics: Integrating rapid circulating tumor cell detection and precision tumor therapy. Lab Chip 2020, 20, 1418–1425. [Google Scholar] [CrossRef]
- Pei, W.; Liu, M.; Wu, Y.; Zhao, Y.; Liu, T.; Sun, B.; Liu, Y.; Wang, Q.; Han, J. High payload and targeted release of anthracyclines by aptamer-tethered DNA nanotrains—Thermodynamic and release kinetic study. Eur. J. Pharm. Sci. 2020, 148, 105319. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Zhang, W.; Zu, Y. Aptamer-Equipped Protamine Nanomedicine for Precision Lymphoma Therapy. Cancers 2020, 12, 780. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Sun, N.; Ding, P.; Chen, C.; Wu, Z.; Zhu, W.; Liu, L.; Wang, Z.; Pei, R. Fabrication of aptamer modified TiO2 nanofibers for specific capture of circulating tumor cells. Colloids Surf. B Biointerfaces 2020, 191, 110985. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Yin, C.; Zhang, W.; Sun, Y.; Zhang, Z.; Yang, E.; Sun, D.; Wang, W. Designing Aptamer-Gold Nanoparticle-Loaded pH-Sensitive Liposomes Encapsulate Morin for Treating Cancer. Nanoscale Res. Lett. 2020, 15, 68. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Yan, J.H.; Long, S.; Xiong, H.; Wen, N.; Cai, S.; Wang, Y.; Peng, D.; Liu, Z.; Liu, Y. Tumor microenvironment and NIR laser dual-responsive release of berberine 9-O-pyrazole alkyl derivative loaded in graphene oxide nanosheets for chemo-photothermal synergetic cancer therapy. J. Mater. Chem. B 2020, 8, 4046–4055. [Google Scholar] [CrossRef]
- Yao, F.; An, Y.; Li, X.; Li, Z.; Duan, J.; Yang, X.-D. Targeted Therapy of Colon Cancer by Aptamer-Guided Holliday Junctions Loaded with Doxorubicin. Int. J. Nanomed. 2020, 15, 2119–2129. [Google Scholar] [CrossRef]
- Mashreghi, M.; Zamani, P.; Moosavian, S.A.; Jaafari, M.R. Anti-Epcam Aptamer (Syl3c)-Functionalized Liposome for Targeted Delivery Of Doxorubicin: In Vitro And In Vivo Antitumor Studies in Mice Bearing C26 Colon Carcinoma. Nanoscale Res. Lett. 2020, 15, 1–13. [Google Scholar] [CrossRef]
- Zhao, Q.; Li, J.; Wu, B.; Shang, Y.; Huang, X.; Dong, H.; Liu, H.; Chen, W.; Gui, R.; Nie, X. Smart Biomimetic Nanocomposites Mediate Mitochondrial Outcome through Aerobic Glycolysis Reprogramming: A Promising Treatment for Lymphoma. ACS Appl. Mater. Interfaces 2020, 12, 22687–22701. [Google Scholar] [CrossRef]
- Khezrian, S.; Khoee, S.; Caceres, M. Synthesis of combinatorial Janus nanoparticles based on EpCAM-PEG/PCL for targeted therapy of human colorectal adenocarcinoma. J. Biomed. Mater. Res. Part A 2020, 108, 2291–2304. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, J.; Cai, X.; Ding, P.; Lv, H.; Pei, R. Metal–Organic Frameworks with Enhanced Photodynamic Therapy: Synthesis, Erythrocyte Membrane Camouflage, and Aptamer-Targeted Aggregation. ACS Appl. Mater. Interfaces 2020, 12, 23697–23706. [Google Scholar] [CrossRef]
- Chan, M.; Huang, W.; Wang, J.; Liu, R.-S.; Hsiao, M. Next-Generation Cancer-Specific Hybrid Theranostic Nanomaterials: MAGE-A3 NIR Persistent Luminescence Nanoparticles Conjugated to Afatinib for In Situ Suppression of Lung Adenocarcinoma Growth and Metastasis. Adv. Sci. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Deng, Y.; Zhu, C.; Xiang, C. Anti prostate cancer therapy: Aptamer-functionalized, curcumin and cabazitaxel co-delivered, tumor targeted lipid-polymer hybrid nanoparticles. Biomed. Pharmacother. 2020, 127, 110181. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Nguyen, H.M.; Wiltz, K.; Hall, N.; Chaudhry, S.; Olverson, G.; Mandal, T.; Dash, S.; Kundu, A. Aptamer-functionalized Hybrid Nanoparticles to Enhance the Delivery of Doxorubicin into Breast Cancer Cells by Silencing P-glycoprotein. J. Cancer Treat. Diagn. 2020, 4, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Lin, S.; Yang, F.; Situ, J.; Lin, S.; Luo, Y. Aptamer-Conjugated Multifunctional Polymeric Nanoparticles as Cancer-Targeted, MRI-Ultrasensitive Drug Delivery Systems for Treatment of Castration-Resistant Prostate Cancer. BioMed Res. Int. 2020, 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Daniels, D.A.; Chen, H.; Hicke, B.J.; Swiderek, K.M.; Gold, L. A tenascin-C aptamer identified by tumor cell SELEX: Systematic evolution of ligands by exponential enrichment. Proc. Natl. Acad. Sci. USA 2003, 100, 15416–15421. [Google Scholar] [CrossRef] [PubMed]
- Shangguan, D.; Li, Y.; Tang, Z.; Cao, Z.C.; Chen, H.W.; Mallikaratchy, P.; Sefah, K.; Yang, C.J.; Tan, W. Aptamers evolved from live cells as effective molecular probes for cancer study. Proc. Natl. Acad. Sci. USA 2006, 103, 11838–11843. [Google Scholar] [CrossRef]
- Xiao, Z.; Shangguan, D.; Cao, Z.; Fang, X.; Tan, W. Cell-Specific Internalization Study of an Aptamer from Whole Cell Selection. Chemistry (Weinh. Bergstr. Ger.) 2008, 14, 1769–1775. [Google Scholar] [CrossRef]
- Fujimori, K.; Covell, D.G.; E Fletcher, J.; Weinstein, J.N. Modeling analysis of the global and microscopic distribution of immunoglobulin G, F(ab′)2, and Fab in tumors. Cancer Res. 1989, 49, 5656–5663. [Google Scholar]
- Fujimori, K.; Covell, D.G.; E Fletcher, J.; Weinstein, J.N. A modeling analysis of monoclonal antibody percolation through tumors: A binding-site barrier. J. Nucl. Med. 1990, 31, 1191–1198. [Google Scholar]
- Miao, L.; Newby, J.M.; Lin, C.M.; Zhang, L.; Xu, F.; Kim, W.Y.; Forest, M.G.; Lai, S.K.; Milowsky, M.I.; Wobker, S.E.; et al. The Binding Site Barrier Elicited by Tumor-Associated Fibroblasts Interferes Disposition of Nanoparticles in Stroma-Vessel Type Tumors. ACS Nano 2016, 10, 9243–9258. [Google Scholar] [CrossRef]
- Jin, Y.; Wu, Z.; Wu, C.; Zi, Y.; Chu, X.; Liu, J.; Zhang, W. Size-adaptable and ligand (biotin)-sheddable nanocarriers equipped with avidin scavenging technology for deep tumor penetration and reduced toxicity. J. Control. Release 2020, 320, 142–158. [Google Scholar] [CrossRef] [PubMed]
- Salvioni, L.; Rizzuto, M.A.; Bertolini, J.A.; Pandolfi, L.; Colombo, M.; Prosperi, D. Thirty Years of Cancer Nanomedicine: Success, Frustration, and Hope. Cancers 2019, 11, 1855. [Google Scholar] [CrossRef] [PubMed]
- Venditto, V.J.; Szoka, F.C. Cancer nanomedicines: So many papers and so few drugs! Adv. Drug Deliv. Rev. 2013, 65, 80–88. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).