Application of WES towards Molecular Investigation of Congenital Cataracts: Identification of Novel Alleles and Genes in a Hospital-Based Cohort of South India
Abstract
1. Introduction
2. Results
2.1. Clinical Documentation and Pattern of Inheritance
2.2. Gene Mutation Identification From Whole-Exome Sequencing and Validation Through Sanger Sequencing
2.3. Congenital Cataract Families Resolved for Genetic Mutations Using WES
2.4. Family Code: DKEC4
2.5. Family Code: C343
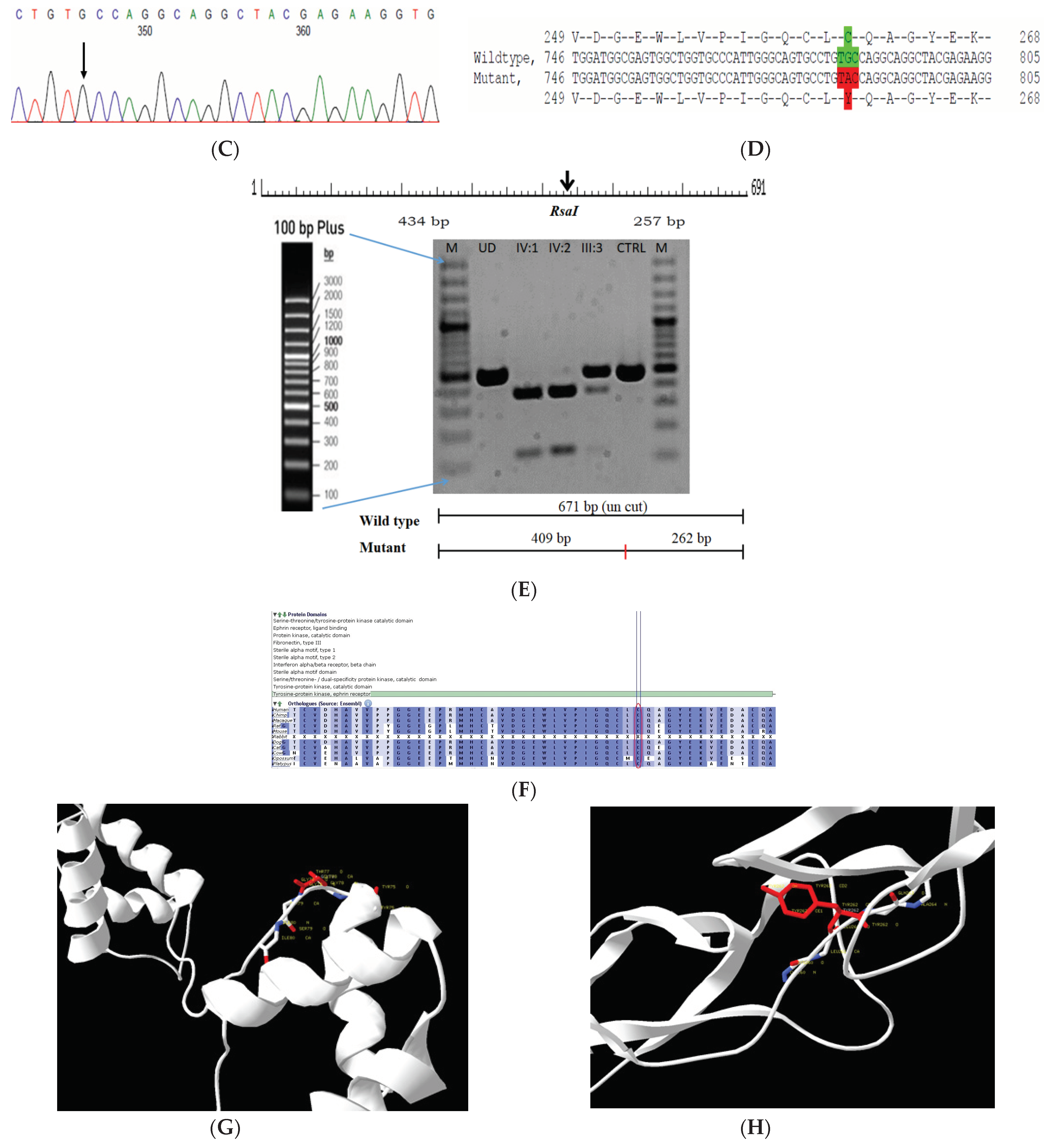
2.6. Family Code: CCE13
2.7. Family Code: CCE27
2.8. Family Code: ACR12
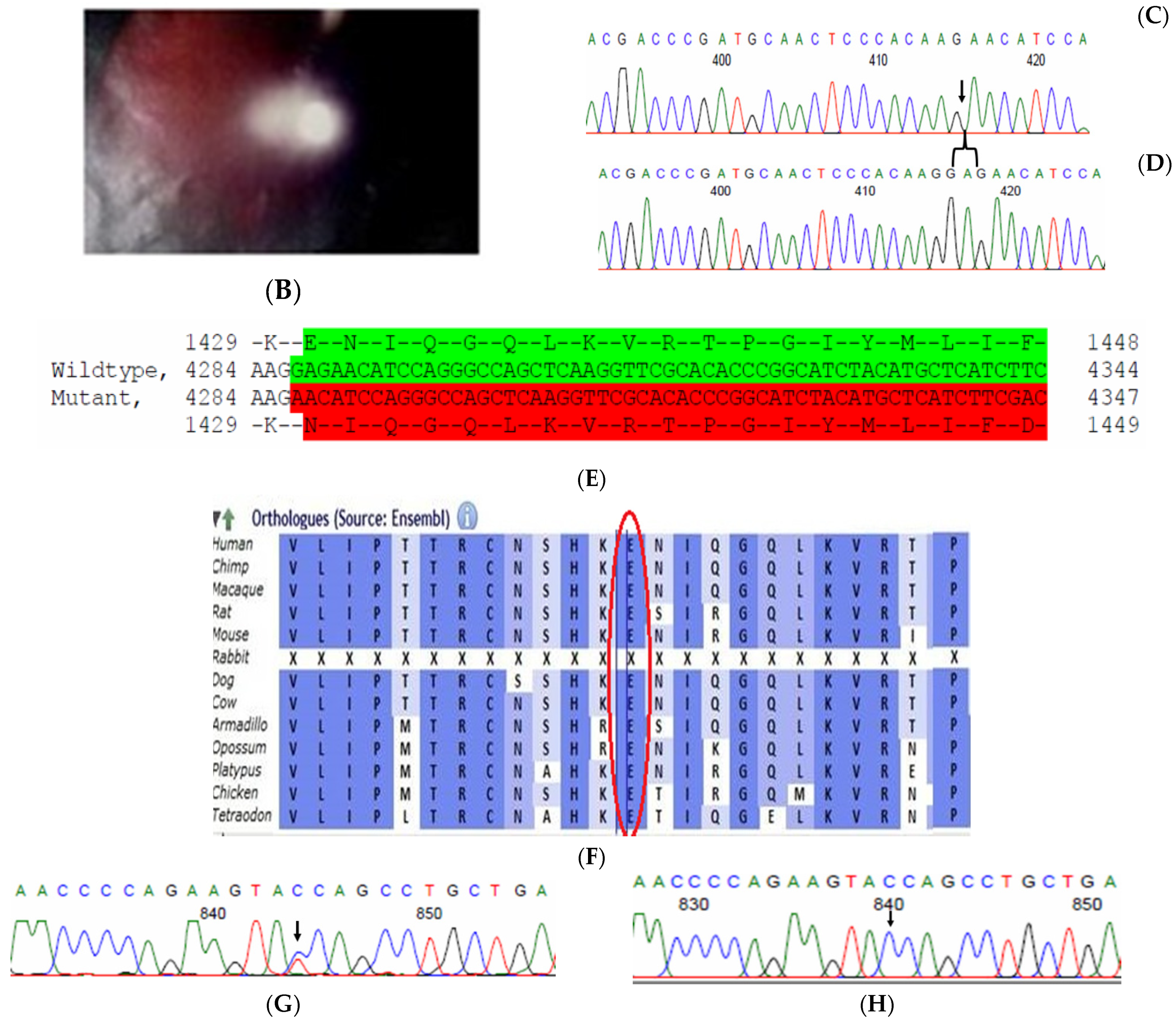
2.9. Family Code: BCC23
3. Discussion
4. Materials and Methods
4.1. Patients and Families
4.2. DNA Extraction and Quantification
4.3. Whole-Exome Sequencing and In Silico Analysis
4.4. Primer Design, PCR Amplification and Sanger Sequencing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, X.; Long, E.; Lin, H.; Liu, Y. Prevalence and Epidemiological Characteristics of Congenital Cataract: A Systematic Review and Meta-Analysis. Sci. Rep. 2016, 6, 28564. [Google Scholar] [CrossRef] [PubMed]
- Shiels, A.; Hejtmancik, J.F. Genetics of Human Cataract. Clin. Genet. 2013, 84, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Messina-Baas, O.; Cuevas-Covarrubias, S.A. Inherited Congenital Cataract: A Guide to Suspect the Genetic Etiology in the Cataract Genesis. Mol. Syndromol. 2017, 8, 58–78. [Google Scholar] [CrossRef] [PubMed]
- Self, J.E.; Taylor, R.; Solebo, A.L.; Biswas, S.; Parulekar, M.; Dev Borman, A.; Ashworth, J.; McClenaghan, R.; Abbott, J.; O’Flynn, E.; et al. Cataract Management in Children: A Review of the Literature and Current Practice across Five Large UK Centres. Eye 2020, 34, 2197–2218. [Google Scholar] [CrossRef]
- Birch, E.E.; Stager, D.R. The Critical Period for Surgical Treatment of Dense Congenital Unilateral Cataract. Investig. Ophthalmol. Vis. Sci. 1996, 37, 1532–1538. [Google Scholar] [CrossRef]
- Lloyd, I.C.; Ashworth, J.; Biswas, S.; Abadi, R.V. Advances in the Management of Congenital and Infantile Cataract. Eye (Lond.) 2007, 21, 1301–1309. [Google Scholar] [CrossRef]
- Duret, A.; Humphries, R.; Ramanujam, S.; Te Water Naudé, A.; Reid, C.; Allen, L.E. The Infrared Reflex: A Potential New Method for Congenital Cataract Screening. Eye (Lond.) 2019, 33, 1865–1870. [Google Scholar] [CrossRef]
- Mohammadpour, M.; Shaabani, A.; Sahraian, A.; Momenaei, B.; Tayebi, F.; Bayat, R.; Mirshahi, R. Updates on Managements of Pediatric Cataract. J. Curr. Ophthalmol. 2018, 31, 118–126. [Google Scholar] [CrossRef]
- Sun, W.; Xiao, X.; Li, S.; Guo, X.; Zhang, Q. Exome Sequencing of 18 Chinese Families with Congenital Cataracts: A New Sight of the NHS Gene. PLoS ONE 2014, 9, e100455. [Google Scholar] [CrossRef]
- Ma, A.S.; Grigg, J.R.; Ho, G.; Prokudin, I.; Farnsworth, E.; Holman, K.; Cheng, A.; Billson, F.A.; Martin, F.; Fraser, C.; et al. Sporadic and Familial Congenital Cataracts: Mutational Spectrum and New Diagnoses Using Next-Generation Sequencing. Hum. Mutat. 2016, 37, 371–384. [Google Scholar] [CrossRef]
- Zhai, Y.; Li, J.; Yu, W.; Zhu, S.; Yu, Y.; Wu, M.; Sun, G.; Gong, X.; Yao, K. Targeted Exome Sequencing of Congenital Cataracts Related Genes: Broadening the Mutation Spectrum and Genotype–Phenotype Correlations in 27 Chinese Han Families. Sci. Rep. 2017, 7, 1219. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wang, S.; Ye, H.; Tang, Y.; Qiu, X.; Fan, Q.; Rong, X.; Liu, X.; Chen, Y.; Yang, J.; et al. Distribution of Gene Mutations in Sporadic Congenital Cataract in a Han Chinese Population. Mol. Vis. 2016, 22, 589–598. [Google Scholar] [PubMed]
- Lachke, S.A.; Alkuraya, F.S.; Kneeland, S.C.; Ohn, T.; Aboukhalil, A.; Howell, G.R.; Saadi, I.; Cavallesco, R.; Yue, Y.; Tsai, A.C.-H.; et al. Mutations in the RNA Granule Component TDRD7 Cause Cataract and Glaucoma. Science 2011, 331, 1571–1576. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Hosokawa, M.; Vagin, V.V.; Reuter, M.; Hayashi, E.; Mochizuki, A.L.; Kitamura, K.; Yamanaka, H.; Kondoh, G.; Okawa, K.; et al. Tudor Domain Containing 7 (Tdrd7) Is Essential for Dynamic Ribonucleoprotein (RNP) Remodeling of Chromatoid Bodies during Spermatogenesis. Proc. Natl. Acad. Sci. USA 2011, 108, 10579–10584. [Google Scholar] [CrossRef]
- Parker, R.; Sheth, U. P Bodies and the Control of MRNA Translation and Degradation. Mol. Cell 2007, 25, 635–646. [Google Scholar] [CrossRef]
- Hanson, I.; Churchill, A.; Love, J.; Axton, R.; Moore, T.; Clarke, M.; Meire, F.; van Heyningen, V. Missense Mutations in the Most Ancient Residues of the PAX6 Paired Domain Underlie a Spectrum of Human Congenital Eye Malformations. Hum. Mol. Genet. 1999, 8, 165–172. [Google Scholar] [CrossRef]
- Hanson, I.M. PAX6 and Congenital Eye Malformations. Pediatr. Res. 2003, 54, 791–796. [Google Scholar] [CrossRef]
- Tang, H.K.; Chao, L.Y.; Saunders, G.F. Functional Analysis of Paired Box Missense Mutations in the PAX6 Gene. Hum. Mol. Genet. 1997, 6, 381–386. [Google Scholar] [CrossRef]
- Chao, L.-Y.; Mishra, R.; Strong, L.C.; Saunders, G.F. Missense Mutations in the DNA-Binding Region and Termination Codon in PAX6. Hum. Mutat. 2003, 21, 138–145. [Google Scholar] [CrossRef]
- Azuma, N.; Yamaguchi, Y.; Handa, H.; Tadokoro, K.; Asaka, A.; Kawase, E.; Yamada, M. Mutations of the PAX6 Gene Detected in Patients with a Variety of Optic-Nerve Malformations. Am. J. Hum. Genet. 2003, 72, 1565. [Google Scholar] [CrossRef]
- Aldahmesh, M.A.; Khan, A.O.; Mohamed, J.Y.; Hijazi, H.; Al-Owain, M.; Alswaid, A.; Alkuraya, F.S. Genomic Analysis of Pediatric Cataract in Saudi Arabia Reveals Novel Candidate Disease Genes. Genet. Med. 2012, 14, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Ricard-Blum, S. The Collagen Family. Cold Spring. Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Pankiv, S.; Johansen, T. FYCO1: Linking Autophagosomes to Microtubule plus End-Directing Molecular Motors. Autophagy 2010, 6, 550–552. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Ma, Z.; Jiao, X.; Fariss, R.; Kantorow, W.L.; Kantorow, M.; Pras, E.; Frydman, M.; Pras, E.; Riazuddin, S.; et al. Mutations in FYCO1 Cause Autosomal-Recessive Congenital Cataracts. Am. J. Hum. Genet. 2011, 88, 827–838. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, M.A.; Samuels, H.H. Nuclear Receptor Coactivator/Coregulator NCoA6(NRC) Is a Pleiotropic Coregulator Involved in Transcription, Cell Survival, Growth and Development. Nucl. Recept. Signal 2008, 6, e002. [Google Scholar] [CrossRef] [PubMed]
- Kuang, S.-Q.; Liao, L.; Zhang, H.; Pereira, F.A.; Yuan, Y.; DeMayo, F.J.; Ko, L.; Xu, J. Deletion of the Cancer-Amplified Coactivator AIB3 Results in Defective Placentation and Embryonic Lethality. J. Biol. Chem. 2002, 277, 45356–45360. [Google Scholar] [CrossRef]
- Antonson, P.; Schuster, G.U.; Wang, L.; Rozell, B.; Holter, E.; Flodby, P.; Treuter, E.; Holmgren, L.; Gustafsson, J.-Å. Inactivation of the Nuclear Receptor Coactivator RAP250 in Mice Results in Placental Vascular Dysfunction. Mol. Cell. Biol. 2003, 23, 1260–1268. [Google Scholar] [CrossRef][Green Version]
- Zhu, Y.-J.; Crawford, S.E.; Stellmach, V.; Dwivedi, R.S.; Rao, M.S.; Gonzalez, F.J.; Qi, C.; Reddy, J.K. Coactivator PRIP, the Peroxisome Proliferator-Activated Receptor-Interacting Protein, Is a Modulator of Placental, Cardiac, Hepatic, and Embryonic Development. J. Biol. Chem. 2003, 278, 1986–1990. [Google Scholar] [CrossRef]
- Mahajan, M.A.; Das, S.; Zhu, H.; Tomic-Canic, M.; Samuels, H.H. The Nuclear Hormone Receptor Coactivator NRC Is a Pleiotropic Modulator Affecting Growth, Development, Apoptosis, Reproduction, and Wound Repair. Mol. Cell. Biol. 2004, 24, 4994–5004. [Google Scholar] [CrossRef]
- Baek, H.J.; Malik, S.; Qin, J.; Roeder, R.G. Requirement of TRAP/Mediator for Both Activator-Independent and Activator-Dependent Transcription in Conjunction with TFIID-Associated TAFIIs. Mol. Cell. Biol. 2002, 22, 2842–2852. [Google Scholar] [CrossRef]
- Wang, W.-L.; Li, Q.; Xu, J.; Cvekl, A. Lens Fiber Cell Differentiation and Denucleation Are Disrupted through Expression of the N-Terminal Nuclear Receptor Box of Ncoa6 and Result in P53-Dependent and P53-Independent Apoptosis. MBoC 2010, 21, 2453–2468. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, S.K.; Anzick, S.L.; Choi, J.E.; Bubendorf, L.; Guan, X.Y.; Jung, Y.K.; Kallioniemi, O.P.; Kononen, J.; Trent, J.M.; Azorsa, D.; et al. A Nuclear Factor, ASC-2, as a Cancer-Amplified Transcriptional Coactivator Essential for Ligand-Dependent Transactivation by Nuclear Receptors in Vivo. J. Biol. Chem. 1999, 274, 34283–34293. [Google Scholar] [CrossRef] [PubMed]
- Shiels, A.; Bennett, T.M.; Hejtmancik, J.F. Cat-Map: Putting Cataract on the Map. Mol. Vis. 2010, 16, 2007. [Google Scholar] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and Guidelines for the Interpretation of Sequence Variants: A Joint Consensus Recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–423. [Google Scholar] [CrossRef]
- Kopanos, C.; Tsiolkas, V.; Kouris, A.; Chapple, C.E.; Albarca Aguilera, M.; Meyer, R.; Massouras, A. VarSome: The Human Genomic Variant Search Engine. Bioinformatics 2019, 35, 1978–1980. [Google Scholar] [CrossRef]















| Case Code | Age at Onset (Years) | POI | Sex (M/F) | Phenotype LE (Left Eye) | Phenotype RE (Right Eye) | Visual Acuity | Other Phenotypic Features if Any | Secondary Affected Relatives | Blood Samples Collected | |
|---|---|---|---|---|---|---|---|---|---|---|
| LE | RE | |||||||||
| DKEC4 | SB | AR | F | Posterior Sub capsular | Posterior Sub capsular | PL (NIP) | PL (NIP) | Grand father’s brother is infertile | Grandfather’s Brother | Proband, Mother, Father, Maternal Grandmother |
| C343 | SB | AD | F | Anterior Polar Cataract with Micro cornea | Anterior Polar Cataract with Micro cornea | NPL | NPL | Aniridia | Paternal Grand mother, Uncles, Aunt, Father, Cousin brothers, Cousin sister | 8 Family members (6 affected and 2 unaffected) – Refer Pedigree |
| CCE13 | SB | AR | M | Lamellar cataract + Micro cornea + Nystagmus | Lamellar cataract + Micro cornea + Nystagmus | PL | PL | Nil | Sibling | Proband, Mother, Sibling |
| CCE27 | SB | AR (PH) | - | Patchy, Total Cataract | Patchy, Total Cataract | 1/60-BCVA-6/60 | 6/36-BCVA-6/24 | Mother had milder developmental cataract phenotype | Sibling | Proband, Mother, Father, Sibling |
| ACR12 | 2.5 | AR (PH) | M | Lamellar Cataract | Posterior Cortical Cataract | 1/60-NIP | 1/60-NIP | Nil | Brother | Proband, Brother, Father, Mother |
| BCC23 | SB | ND (PH) | M | Dense Cortical cataract | Posterior sub capsular | PR | PR | Mother—Blue dot opacities; Proband and Sib—Deaf and Dumb | Two sibs, Mother | Two sibs and Father |
| Family-ID | POI | Consanguinity | Zygosity | Gene Variant | Genomic Co-Ordinate | Variant Identified | RS ID and Frequency | ACMG Criteria | Solved Cases |
|---|---|---|---|---|---|---|---|---|---|
| DKEC4 | AR | Y | HOM | TDRD7 | chr9-100249550-A- | TDRD7(NM_014290.3): c.3012delA (p.Val1005TyrfsTer4) | NA-Novel variant not reported before | PATH | S |
| C343 | AD | N | HET | PAX6 | chr11-31823275-C-A | PAX6(NM_001310158.2) c.233G > T (p.Gly78Val) | rs121907920-No frequency reported | LP | S |
| CCE13 | AR | Y | HOM | EPHA2 | chr1-16474911-C-T | EPHA2(NM_004431.5): c.785G > A (p.Cys262Tyr) | NA-Novel variant not reported before | VUS | S |
| CCE27 | AR | Y | HOM | P3H2 | chr3-189692382--C | P3H2(NM_018192.4): c.1417dupG (p.Glu473GlyfsTer19) | NA-Novel variant not reported before | PATH | S |
| ACR12 | AR | Y | HOM | FYCO1 | chr3-46007891-G-A | FYCO1(NM_024513.4): c.2935C > T (p.Gln979Ter) | NA-Novel variant not reported before | PATH | S |
| BCC23 | ND (PH) | Y | HOM | FYCO1 | chr3-45965219-CTC | FYCO1(NM_024513.4): c.4288_4290delGAG (p.Glu1430del) | rs774142616-gnomAD-0.0000239/ ISB Kaviar3-0.0000257 | VUS | S |
| Y | HET | NCOA6 | chr20-33338208-C-T | NCOA6(NM_014071.5): c.1790G > A (p.Gly597Asp) | rs1461340387-gnomAD-0.0000119 | LB | PS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kandaswamy, D.K.; Prakash, M.V.S.; Graw, J.; Koller, S.; Magyar, I.; Tiwari, A.; Berger, W.; Santhiya, S.T. Application of WES towards Molecular Investigation of Congenital Cataracts: Identification of Novel Alleles and Genes in a Hospital-Based Cohort of South India. Int. J. Mol. Sci. 2020, 21, 9569. https://doi.org/10.3390/ijms21249569
Kandaswamy DK, Prakash MVS, Graw J, Koller S, Magyar I, Tiwari A, Berger W, Santhiya ST. Application of WES towards Molecular Investigation of Congenital Cataracts: Identification of Novel Alleles and Genes in a Hospital-Based Cohort of South India. International Journal of Molecular Sciences. 2020; 21(24):9569. https://doi.org/10.3390/ijms21249569
Chicago/Turabian StyleKandaswamy, Dinesh Kumar, Makarla Venkata Sathya Prakash, Jochen Graw, Samuel Koller, István Magyar, Amit Tiwari, Wolfgang Berger, and Sathiyaveedu Thyagarajan Santhiya. 2020. "Application of WES towards Molecular Investigation of Congenital Cataracts: Identification of Novel Alleles and Genes in a Hospital-Based Cohort of South India" International Journal of Molecular Sciences 21, no. 24: 9569. https://doi.org/10.3390/ijms21249569
APA StyleKandaswamy, D. K., Prakash, M. V. S., Graw, J., Koller, S., Magyar, I., Tiwari, A., Berger, W., & Santhiya, S. T. (2020). Application of WES towards Molecular Investigation of Congenital Cataracts: Identification of Novel Alleles and Genes in a Hospital-Based Cohort of South India. International Journal of Molecular Sciences, 21(24), 9569. https://doi.org/10.3390/ijms21249569





