Dual Antibacterial Activities and Biofilm Eradication of a Marine Peptide-N6NH2 and Its Analogs against Multidrug-Resistant Aeromonas veronii
Abstract
:1. Introduction
2. Results
2.1. Design and Physicochemical Properties of N6NH2 and Its Analogs
2.2. N6NH2 and Its Analogs Showed Potent Antimicrobial Activity
2.3. N6NH2 and Its Analogs Showed Additive Effects with Antibiotics and Greater Post-Antibiotic Effect (PAE) Values
2.4. DN6NH2 and N6PNH2 Had Higher Stability in Different Conditions
2.5. All Analogs Exhibited No or Low Toxicity and Resistance
2.6. Antibacterial Mechanism of N6NH2 and Its Analogs
2.6.1. N6NH2 and Its Analogs Permeabilized the Outer Cell Membranes and Disrupted the Membrane Potentials
2.6.2. Peptides Exposure Affects Adenosine Triphosphate (ATP) Release from Cells
2.6.3. All Analogs of N6NH2 Bound to Bacterial Genomic DNA and Changed DNA Structure
2.7. All Analogs of N6NH2 Induced Morphological Changes in A. veronii
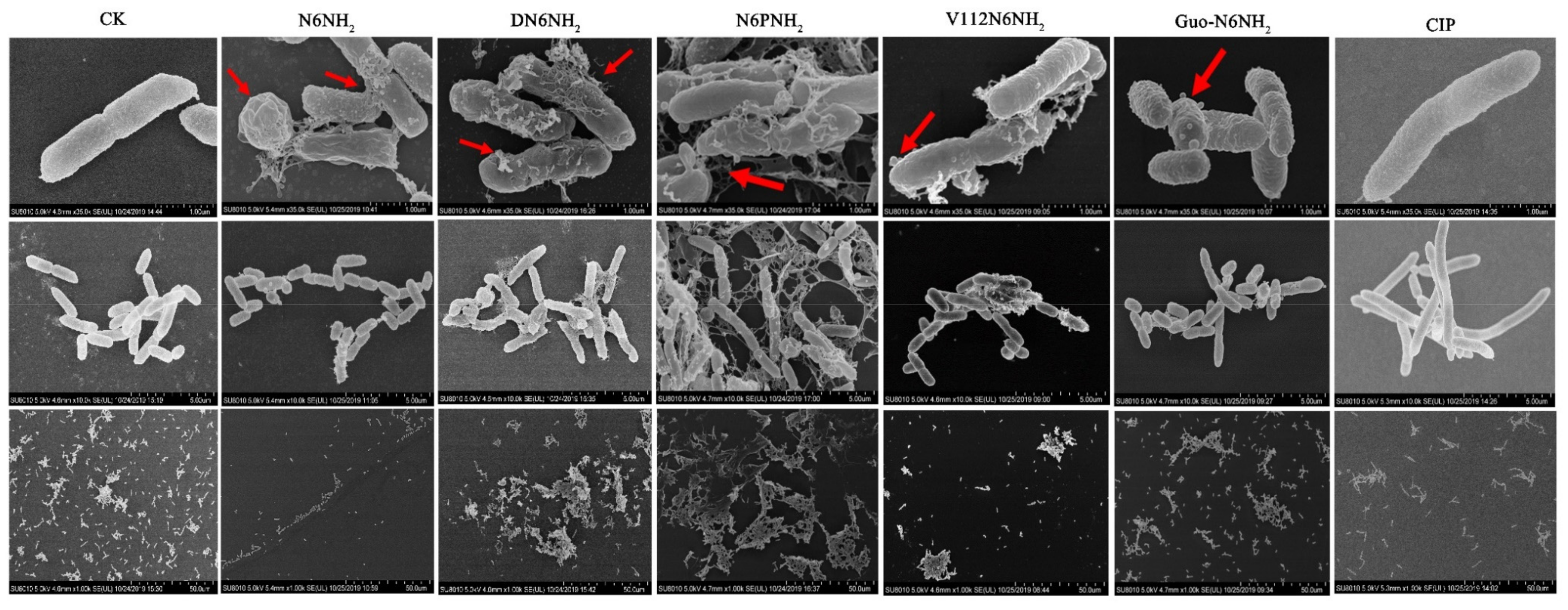
2.7.1. Scanning Electron Microscope (SEM) Observations
2.7.2. Transmission Electron Microscope (TEM) and Confocal Laser Scanning Microscope (CLSM) Observations
2.8. N6NH2 and Its Analogs Eliminated MDR A. veronii Biofilms and Persisters
2.8.1. Inhibition of Biofilm Formation
2.8.2. Eradication of Mature Biofilms
2.8.3. Killing Persisters in Biofilm
2.9. N6NH2 and Its Analogs Protected Mice from Catheter-Associated Biofilm Infection with MDR A. veronii
2.9.1. Protection of Biofilm-Infected Mice
2.9.2. Protection of Tissues from A. veronii ACCC61732 Biofilm
2.10. N6NH2 and Its Analogs Protected Mice from Bacterial Infection with MDR A. veronii
2.10.1. Protection of Mice
2.10.2. Inhibition of Bacterial Translocation
2.10.3. Regulation of Cytokines
2.10.4. Alleviation of the Organ Injury
3. Discussion
4. Materials and Methods
4.1. Reagents, Cell Lines, and Model Animals
4.2. Physiochemical Properties of N6NH2 and Its Analogs
4.3. Antibacterial Activities and Time-Killing Curves of N6NH2 and Its Analogs
4.4. Synergism and PAE of N6NH2 and Its Analogs against A. veronii
4.5. Stability of N6NH2 and Its Analogs
4.5.1. Temperature, pH, Salt, and Enzyme Sensitivity
4.5.2. Stability in Gastric/Intestinal Fluid and Serum
4.6. Hemolysis, Cytotoxicity, and Resistance of N6NH2 and Its Analogs
4.6.1. Hemolysis
4.6.2. Cytotoxicity
4.6.3. Resistance
4.7. Mechanism of N6NH2 and Its Analogs
4.7.1. Effects on the Cell Membrane and Membrane Potential
4.7.2. Measure of ATP Release
4.7.3. Effects on Bacterial Genomic DNA
4.7.4. Effects of N6NH2 and Its Analogs on Bacterial Morphology
4.8. Effects of N6NH2 and Its Analogs on Biofilms and Persisters of MDR A. veronii
4.8.1. Early and Mature Biofilms
4.8.2. Persisters in Biofilms
4.9. Efficacy of N6NH2 and Its Analogs in a Mouse Model of Catheter-Associated Biofilm Infection
4.9.1. Effects on Catheter-Associated Biofilms
4.9.2. Effects on Skin Ulcer in the Mouse Back
4.10. Efficacy of N6NH2 and Its Analogs in a Mouse Peritonitis Model
4.10.1. Survival of Mice
4.10.2. Effects on Bacterial Translocation and Cytokines
4.10.3. Effects on the Injury of Multiple Organs
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CIP | ciprofloxacin |
| AMP | antimicrobial peptide(s) |
| Orn | ornithine |
| MDR | multidrug resistant |
| MIC | minimum inhibitory concentration |
| MW | molecular weight |
| MS | mass spectroscopy |
| GRAVY | grand average of hydropathicity |
| AI | aliphatic index |
| BI | boman index |
| pI | isoelectric point |
| FICI | fractional inhibitory concentration index |
| PAE | postantibiotic effect |
| MTT | methylthiazolyldiphenyl-tetrazolium |
| SGF | simulated gastric fluid |
| SIF | simulated intestinal fluid |
| GEN | gentamicin |
| NPN | n-phenyl-1-naphthylamine |
| PI | propidium iodide |
| DISC3(5)) | 3,3′-dipropylthiadicarbocyanine iodide |
| ATP | adenosine triphosphate |
| CD | circular dichroism |
| SEM | scanning electron microscope |
| OMVs | outer membrane vesicles |
| PCR | polymerase chain reaction |
| TEM | transmission electron microscope |
| CLSM | confocal laser scanning microscope |
| TNF | tumor necrosis factor |
| IL | interleukin |
| ELISA | enzyme-linked immunosorbent assay |
| HEPES | n-2-hydroxyethylpiperazine-n-2-ethane sulfonic acid |
References
- Hoai, T.D.; Trang, T.T.; Van Tuyen, N.; Giang, N.T.H.; Van Van, K. Aeromonas veronii caused disease and mortality in channel catfish in Vietnam. Aquaculture 2019, 513, 734425. [Google Scholar] [CrossRef]
- Huang, H.; Zhou, P.; Chen, P.; Xia, L.; Hu, S.; Yi, G.; Lu, J.; Yang, S.; Xie, J.; Peng, J.; et al. Alteration of the gut microbiome and immune factors of grass carp infected with Aeromonas veronii and screening of an antagonistic bacterial strain (Streptomyces flavotricini). Microb. Pathog. 2020, 143, 104092. [Google Scholar] [CrossRef]
- Li, T.; Raza, S.H.A.; Yang, B.; Sun, Y.; Wang, G.-Q.; Sun, W.; Qian, A.; Wang, C.; Shan, X.; Shan, X. Aeromonas veronii Infection in Commercial Freshwater Fish: A Potential Threat to Public Health. Animal 2020, 10, 608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez-Serrano, C.J.; Santos, J.A.; Garcia-Lopez, M.L.; Otero, A. Virulence markers in Aeromonas hydrophila and Aeromonas veronii biovar sobria isolates from freshwater fish and from a diarrhoea case. J. Appl. Microbiol. 2002, 93, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Pearson, M.D.; Hirono, I.; Aoki, T.; Miranda, R.; Inglis, V. Virulence properties of motile aeromonads isolated from farmed frogs Rana tigerina and R. rugulosa. Dis. Aquat. Org. 2000, 40, 185–193. [Google Scholar] [CrossRef]
- Bhowmick, U.D.; Bhattacharjee, S. Bacteriological, Clinical and Virulence Aspects of Aeromonas-associated Diseases in Humans. Pol. J. Microbiol. 2018, 67, 137–149. [Google Scholar] [CrossRef]
- Pund, R.P.; Theegarten, D. The importance of aeromonads as a human pathogen. Bundesgesundheitsblatt Gesundh. Gesundh. 2008, 51, 569–576. [Google Scholar] [CrossRef]
- Hadi, N.; Mahmoodi, Z.; Emami, A.; Malekzadegan, Y.; Valadbeygi, T. Isolation and Molecular Identification of Aeromonas Wound Infection in Iranian Burn Patients. Infect. Disord. Drug Targets 2019, 19, 269–273. [Google Scholar] [CrossRef]
- Wu, C.-J.; Ko, W.-C.; Lee, N.-Y.; Su, S.-L.; Li, C.-W.; Li, M.-C.; Chen, Y.-W.; Su, Y.-C.; Shu, C.-Y.; Lin, Y.-T.; et al. Aeromonas Isolates from Fish and Patients in Tainan City, Taiwan: Genotypic and Phenotypic Characteristics. Appl. Environ. Microbiol. 2019, 85, e01360-19. [Google Scholar] [CrossRef]
- Lazado, C.C.; Zilberg, D. Pathogenic characteristics of Aeromonas veronii isolated from the liver of a diseased guppy (Poecilia reticulata). Lett. Appl. Microbiol. 2018, 67, 476–483. [Google Scholar] [CrossRef]
- Olsen, I. Biofilm-specific antibiotic tolerance and resistance. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Skwor, T.A.; Shinko, J.; Augustyniak, A.; Gee, C.; Andraso, G. Aeromonas hydrophila and Aeromonas veronii Predominate among Potentially Pathogenic Ciprofloxacin- and Tetracycline-Resistant Aeromonas Isolates from Lake Erie. Appl. Environ. Microbiol. 2014, 80, 841–848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vila, J.R.J.; Gallardo, F.; Vargas, M.; Soler, L.; Figueras, M.J.; Gascon, J. Aeromonas spp. and traveler’s diarrhea: Clinical features and antimicrobial resistance. Emerg. Infect. Dis. 2003, 9, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Haney, E.F.; Hancock, R.E. Peptide design for antimicrobial and immunomodulatory applications. Biopolymers 2013, 100, 572–583. [Google Scholar] [CrossRef]
- Thapa, R.K.; Diep, D.B.; Tønnesen, H.H. Topical antimicrobial peptide formulations for wound healing: Current developments and future prospects. Acta Biomater. 2020, 103, 52–67. [Google Scholar] [CrossRef]
- Jaśkiewicz, M.; Neubauer, D.; Kazor, K.; Bartoszewska, S.; Kamysz, W. Antimicrobial Activity of Selected Antimicrobial Peptides against Planktonic Culture and Biofilm of Acinetobacter baumannii. Probiotics Antimicrob. Proteins 2019, 11, 317–324. [Google Scholar] [CrossRef] [Green Version]
- Sieprawska-Lupa, M.; Mydel, P.; Krawczyk, K.; Wójcik, K.; Puklo, M.; Lupa, B.; Suder, P.; Silberring, J.; Reed, M.; Pohl, J.; et al. Degradation of Human Antimicrobial Peptide LL-37 by Staphylococcus aureus-Derived Proteinases. Antimicrob. Agents Chemother. 2004, 48, 4673–4679. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Jang, J.H.; Kim, S.C.; Cho, J.H. De novo generation of short antimicrobial peptides with enhanced stability and cell specificity. J. Antimicrob. Chemother. 2014, 69, 121–132. [Google Scholar] [CrossRef]
- Jia, F.; Wang, J.; Peng, J.; Zhao, P.; Kong, Z.; Wang, K.; Yan, W.; Wang, R. D-amino acid substitution enhances the stability of antimicrobial peptide polybia-CP. Acta Biochim. Biophys. Sin. 2017, 49, 916–925. [Google Scholar] [CrossRef] [Green Version]
- Bessalle, R.; Kapitkovsky, A.; Gorea, A.; Shalit, I.; Fridkin, M. All-D-magainin: Chirality, antimicrobial activity and proteolytic resistance. FEBS Lett. 1990, 274, 151–155. [Google Scholar] [CrossRef] [Green Version]
- Czihal, P.; Knappe, D.; Fritsche, S.; Zahn, M.; Berthold, N.; Piantavigna, S.; Müller, U.; Van Dorpe, S.; Herth, N.; Binas, A.; et al. Api88 Is a Novel Antibacterial Designer Peptide To Treat Systemic Infections with Multidrug-Resistant Gram-Negative Pathogens. ACS Chem. Biol. 2012, 7, 1281–1291. [Google Scholar] [CrossRef]
- Bluhm, M.E.; Knappe, D.; Hoffmann, R. Structure-activity relationship study using peptide arrays to optimize Api137 for an increased antimicrobial activity against Pseudomonas aeruginosa. Eur. J. Med. Chem. 2015, 103, 574–582. [Google Scholar] [CrossRef]
- López-Pérez, P.M.; Grimsey, E.; Bourne, L.; Mikut, R.; Hilpert, K. Screening and Optimizing Antimicrobial Peptides by Using SPOT-Synthesis. Front. Chem. 2017, 5, 25. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.; Knappe, D.; Wende, E.; Ostorhazi, E.; Hoffmann, R. In vivo Efficacy and Pharmacokinetics of Optimized Apidaecin Analogs. Front. Chem. 2017, 5, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Teng, D.; Mao, R.; Wang, X.; Hao, Y.; Wang, X.; Wang, J. Improved Antibacterial Activity of the Marine Peptide N6 against Intracellular Salmonella Typhimurium by Conjugating with the Cell-Penetrating Peptide Tat11 via a Cleavable Linker. J. Med. Chem. 2018, 61, 7991–8000. [Google Scholar] [CrossRef]
- Roier, S.; Zingl, F.G.; Cakar, F.; Durakovic, S.; Kohl, P.; Eichmann, T.O.; Klug, L.; Gadermaier, B.; Weinzerl, K.; Prassl, R.; et al. A novel mechanism for the biogenesis of outer membrane vesicles in Gram-negative bacteria. Nat. Commun. 2016, 7, 10515. [Google Scholar] [CrossRef] [Green Version]
- Avila-Calderon, E.D.; Otero-Olarra, J.E.; Flores-Romo, L.; Peralta, H.; Aguilera-Arreola, M.G.; Morales-Garcia, M.R.; Calderon-Amador, J.; Medina-Chavez, O.; Donis-Maturano, L.; Ruiz-Palma, M.D.S.; et al. The outer membrane vesicles of Aeromonas hydrophila ATCC((R)) 7966(TM): A proteomic analysis and effect on host cells. Front. Microbiol. 2018, 9, 2765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, W.C.; Yu, K.W.; Liu, C.Y.; Huang, C.T.; Leu, H.S.; Chuang, Y.C. Increasing antibiotic resistance in clinical isolates of aeromonas strains in Taiwan. Antimicrob. Agents Chemother. 1996, 40, 1260–1262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hossain, S.; De Silva, B.; Dahanayake, P.; Heo, G. Characterization of virulence properties and multi-drug resistance profiles in motile Aeromonas spp. isolated from zebrafish (Danio rerio). Lett. Appl. Microbiol. 2018, 67, 598–605. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, X.; Mao, R.; Hao, Y.; Yang, N.; Wang, X.; Li, Z.; Wang, X.; Wang, J.; Teng, D. Development of chimeric peptides to facilitate the neutralisation of lipopolysaccharides during bactericidal targeting of multidrug-resistant Escherichia coli. Commun. Biol. 2020, 3, 1–15. [Google Scholar] [CrossRef]
- Yang, N.; Liu, X.; Teng, D.; Li, Z.; Wang, X.; Mao, R.; Wang, X.; Hao, Y.; Wang, J. Antibacterial and detoxifying activity of NZ17074 analogues with multi-layers of selective antimicrobial actions against Escherichia coli and Salmonella enteritidis. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Bluhm, M.E.C.; Schneider, V.A.F.; Schäfer, I.; Piantavigna, S.; Goldbach, T.; Knappe, D.; Seibel, P.; Martin, L.L.; Veldhuizen, E.J.A.; Hoffmann, R. N-Terminal Ile-Orn- and Trp-Orn-Motif Repeats Enhance Membrane Interaction and Increase the Antimicrobial Activity of Apidaecins against Pseudomonas aeruginosa. Front. Cell Dev. Biol. 2016, 4, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wimley, W.C. Describing the Mechanism of Antimicrobial Peptide Action with the Interfacial Activity Model. ACS Chem. Biol. 2010, 5, 905–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, M.-F.; Kang, Y.-H.; Zhang, D.-X.; Chen, L.; Bi, J.-F.; Zhang, H.-P.; Zhang, L.; Qian, A.; Shan, X. Immunogenicity of extracellular products from an inactivated vaccine against Aeromonas veronii TH0426 in koi, Cyprinus carpio. Fish Shellfish. Immunol. 2018, 81, 176–181. [Google Scholar] [CrossRef]
- Roilides, E.; Simitsopoulou, M.; Katragkou, A.; Walsh, T.J. How Biofilms Evade Host Defenses. Microbiol. Spectr. 2015, 3, 287–300. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, C.J., Jr.; Mende, K.; Beckius, M.L.; Akers, K.S.; Romano, D.R.; Wenke, J.C.; Murray, C.K. Biofilm formation by clinical isolates and the implications in chronic infections. BMC Infect. Dis. 2013, 13, 47. [Google Scholar] [CrossRef] [Green Version]
- Zhao, F.; Yang, N.; Wang, X.; Mao, R.; Hao, Y.; Li, Z.; Wang, X.; Teng, D.; Fan, H.; Wang, J. In vitro/vivo Mechanism of Action of MP1102 with Low/Nonresistance against Streptococcus suis Type 2 Strain CVCC 3928. Front. Cell. Infect. Microbiol. 2019, 9, 48. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Yang, N.; Mao, R.; Teng, D.; Hao, Y.; Wang, X.; Wang, J. A new high-yielding antimicrobial peptide NZX and its antibacterial activity against Staphylococcus hyicus in vitro/vivo. Appl. Microbiol. Biotechnol. 2020, 104, 1555–1568. [Google Scholar] [CrossRef]
- White, R.L.B.D.S.; Manduru, M.; Bosso, J.A. Comparison of three different in vitro methods of detecting synergy: Time-kill, checkerboard, and E test. Antimicrob. Agents Chemother. 1996, 40, 1914–1918. [Google Scholar] [CrossRef] [Green Version]
- Giguère, S.; Lee, E.A.; Guldbech, K.M.; Berghaus, L.J. In vitro synergy, pharmacodynamics, and postantibiotic effect of 11 antimicrobial agents against Rhodococcus equi. Vet. Microbiol. 2012, 160, 207–213. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, W.; Dong, M.; Hang, B.; Sun, Y.; Wang, L.; Wang, Y.; Hu, J.; Zhang, W. HJH-1, a Broad-Spectrum Antimicrobial Activity and Low Cytotoxicity Antimicrobial Peptide. Molecules 2018, 23, 2026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, P.; Lai, Z.; Zhu, Y.; Shao, C.; Akhtar, M.U.; Li, W.; Zheng, X.; Shan, A. Multiple Strategy Optimization of Specifically Targeted Antimicrobial Peptide Based on Structure–Activity Relationships to Enhance Bactericidal Efficiency. ACS Biomater. Sci. Eng. 2019, 6, 398–414. [Google Scholar] [CrossRef]
- Li, B.; Yang, N.; Shan, Y.; Wang, X.; Hao, Y.; Mao, R.; Teng, D.; Fan, H.; Wang, J. Therapeutic potential of a designed CSalphabeta peptide ID13 in Staphylococcus aureus-induced endometritis of mice. Appl. Microbiol. Biotechnol. 2020, 104, 6693–6705. [Google Scholar] [CrossRef] [PubMed]
- Teng, D.; Wang, X.; Xi, D.; Mao, R.; Zhang, Y.; Guan, Q.; Zhang, J.; Wang, J. A dual mechanism involved in membrane and nucleic acid disruption of AvBD103b, a new avian defensin from the king penguin, against Salmonella enteritidis CVCC3377. Appl. Microbiol. Biotechnol. 2014, 98, 8313–8325. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Ji, Y.; Zhu, Y.; Wu, X.; Mei, L.; Zhang, H.; Deng, J.; Wang, S. Antibacterial effect of chitosan and its derivative on Enterococcus faecalis associated with endodontic infection. Exp. Ther. Med. 2020, 19, 3805–3813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elliott, A.G.; Huang, J.X.; Neve, S.; Zuegg, J.; Edwards, I.A.; Cain, A.K.; Boinett, C.J.; Barquist, L.; Lundberg, C.V.; Steen, J.; et al. An amphipathic peptide with antibiotic activity against multidrug-resistant Gram-negative bacteria. Nat. Commun. 2020, 11. [Google Scholar] [CrossRef]
- Hao, Y.; Yang, N.; Wang, X.; Teng, D.; Mao, R.; Wang, X.; Li, Z.; Wang, J. Killing of Staphylococcus aureus and Salmonella enteritidis and neutralization of lipopolysaccharide by 17-residue bovine lactoferricins: Improved activity of Trp/Ala-containing molecules. Sci. Rep. 2017, 7, srep44278. [Google Scholar] [CrossRef] [Green Version]
- Yang, N.; Teng, D.; Mao, R.; Hao, Y.; Wang, X.; Wang, Z.; Wang, X.; Wang, J. A recombinant fungal defensin-like peptide-P2 combats multidrug-resistant Staphylococcus aureus and biofilms. Appl. Microbiol. Biotechnol. 2019, 103, 5193–5213. [Google Scholar] [CrossRef]
- Mohamed, M.F.; Brezden, A.; Mohammad, H.; Chmielewski, J.; Seleem, M.N. A short D-enantiomeric antimicrobial peptide with potent immunomodulatory and antibiofilm activity against multidrug-resistant Pseudomonas aeruginosa and Acinetobacter baumannii. Sci. Rep. 2017, 7, 6953. [Google Scholar] [CrossRef]
- Asahi, Y.; Miura, J.; Tsuda, T.; Kuwabata, S.; Tsunashima, K.; Noiri, Y.; Sakata, T.; Ebisu, S.; Hayashi, M. Simple observation of Streptococcus mutans biofilm by scanning electron microscopy using ionic liquids. AMB Express 2015, 5. [Google Scholar] [CrossRef] [Green Version]
- Shan, Y.; Yang, N.; Teng, D.; Wang, X.; Mao, R.; Hao, Y.; Ma, X.; Fan, H.; Wang, J. Recombinant of the Staphylococcal Bacteriophage Lysin CHAPk and Its Elimination against Streptococcus agalactiae Biofilms. Microorganisms 2020, 8, 216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kadurugamuwa, J.L.; Sin, L.; Albert, E.; Yu, J.; Francis, K.; DeBoer, M.; Rubin, M.; Bellinger-Kawahara, C.; Parr, T.R., Jr.; Contag, P.R. Direct continuous method for monitoring biofilm infection in a mouse model. Infect. Immun. 2003, 71, 882–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Mao, R.; Teng, D.; Hao, Y.; Chen, H.; Wang, X.; Wang, X.; Yang, N.; Wang, J. Antibacterial and immunomodulatory activities of insect defensins-DLP2 and DLP4 against multidrug-resistant Staphylococcus aureus. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]







| Peptides | Amino Acid Sequences | Length | Theoretical MW (Da) | Measured MW (Da) | Charge (+) | GRAVY | AI | BI (kcal mol-1) | pI | Hydrophobicity | AH % | Extinction Coefficient (M−1 cm−1) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N6NH2 | GFAWNVCVYRNGVRVCHRRAN-NH2 | 21 | 2476.85 | 2476.8 | 5 | NP | NP | NP | 11.64 | 0.375 | NP | 6970 | |
| DN6NH2 | GfawnvcvyrnGvrvchrran-NH2 | 21 | 2476.85 | 2474.88 | 5 | NP | NP | NP | 11.64 | 0.375 | NP | 6970 | |
| N6PNH2 | GFAWNVCVYRNGVRVCHRPAN-NH2 | 21 | 2417.78 | 2415.81 | 4 | NP | NP | NP | 10.91 | 0.457 | NP | 6970 | |
| V112N6NH2 | VFAWNVCVYRNVVRVCHRRAN-NH2 | 21 | 2561.01 | 2559.04 | 5 | NP | NP | NP | 11.64 | 0.491 | NP | 6970 | |
| Guo-N6NH2 | Gu-OGFAWNVCVYRNGVRVCHRRAN-NH2 | 21 | NP | 2631.08 | 6 | NP | NP | NP | NP | NP | NP | NP | |
| N2143 | VCVYRGFAWNCHRRANNGVRV | 21 | 2477.85 | 2475.81 | 4 | −0.31 | 64.76 | 2.61 | 10.72 | 0.375 | 23.81% | 6970 | |
| N2413 | VCVYRCHRRANGFAWNNGVRV | 21 | 2477.85 | 2475.81 | 4 | −0.31 | 64.76 | 2.61 | 10.72 | 0.375 | 0.00% | 6970 | |
| SN1 | AACCGFWNVVYRNGVRVHRRN | 21 | 2477.85 | 2477.83 | 4 | −0.31 | 64.76 | 2.61 | 10.72 | 0.375 | 0.00% | 6970 | |
| SN3 | GFWNVVYRNGVAACCRVHRRN | 21 | 2477.85 | 2477.83 | 4 | −0.31 | 64.76 | 2.61 | 10.72 | 0.375 | 33.33% | 6970 |
| Species and Strains | MIC | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N6NH2 | DN6NH2 | N6PNH2 | V112N6NH2 | Guo-N6NH2 | CIP | |||||||
| μg/mL | μM | μg/mL | μM | μg/mL | μM | μg/mL | μM | μg/mL | μM | μg/mL | μM | |
| Gram-negative bacteria | ||||||||||||
| Aeromonas veronii ACCC61732 a | 4 | 1.61 | 4 | 1.62 | 16 | 6.62 | 16 | 6.25 | 8 | 3.04 | 0.125 | 0.38 |
| A. veronii X-1-06909 b | 32 | 12.9 | 32 | 12.93 | 64 | 26.49 | 32 | 12.51 | 16 | 6.08 | 0.25 | 0.75 |
| A. veronii CL0901 b | 16 | 6.46 | 32 | 12.93 | 64 | 26.49 | 32 | 12.51 | 16 | 6.08 | 0.0625 | 0.19 |
| A. veronii ATCC35624 | 8 | 3.23 | 64 | 25.86 | 64 | 26.49 | 16 | 6.25 | 32 | 12.16 | <0.0625 | <0.19 |
| Escherichia coli CVCC195 | 4 | 1.61 | 8 | 3.23 | 8 | 3.31 | 8 | 3.13 | 4 | 1.52 | <0.0625 | <0.19 |
| E. coli CVCC1515 | 2 | 0.81 | 4 | 1.62 | 4 | 1.66 | 4 | 1.56 | 2 | 0.76 | <0.0625 | <0.19 |
| E. coli CVCC25922 | 4 | 1.61 | 8 | 3.23 | 8 | 3.31 | 8 | 3.13 | 4 | 1.52 | <0.0625 | <0.19 |
| E. coli CVCCO157 | 4 | 1.61 | 4 | 1.62 | 8 | 3.31 | 8 | 3.13 | 4 | 1.52 | <0.0625 | <0.19 |
| Salmonella typhimurium ATCC14028 | 4 | 1.61 | 8 | 3.23 | 16 | 6.62 | 16 | 6.25 | 4 | 1.52 | 0.0625 | 0.19 |
| S. pullorum CVCC1809 | 4 | 1.61 | 8 | 3.23 | 8 | 3.31 | 8 | 3.13 | 4 | 1.52 | <0.0625 | <0.19 |
| S. pullorum CVCC1789 | 8 | 3.23 | 16 | 6.46 | 16 | 6.62 | 16 | 6.25 | 8 | 3.04 | <0.0625 | <0.19 |
| S. pullorum CVCC533 | 4 | 1.61 | 16 | 6.46 | 32 | 13.25 | 16 | 6.25 | 8 | 3.04 | <0.0625 | <0.19 |
| S. enteritidis CVCC3377 | 2 | 0.81 | 2 | 0.81 | 2 | 0.83 | 2 | 0.78 | 2 | 0.76 | <0.0625 | <0.19 |
| Pseudomonas aeruginosa CICC21630 | 64 | 25.8 | 8 | 3.23 | >64 | >26.49 | 32 | 12.5 | >64 | >24.3 | 0.125 | 0.38 |
| Gram-positive bacteria | ||||||||||||
| Staphylococcus aureus ATCC43300 | 16 | 6.46 | 4 | 1.62 | >64 | >26.49 | 32 | 12.5 | 16 | 6.08 | 0.25 | 0.75 |
| S. aureus ATCC546 | 16 | 6.46 | 16 | 6.46 | >64 | >26.49 | 16 | 6.25 | 16 | 6.08 | 0.125 | 0.38 |
| S. aureus ATCC25923 | 32 | 12.9 | 8 | 3.23 | >64 | >26.49 | 32 | 12.5 | 32 | 12.16 | 0.125 | 0.38 |
| S. hyicus NCTC10350 | 32 | 12.9 | 8 | 3.23 | >128 | >52.98 | 64 | 25.01 | 64 | 24.32 | 0.125 | 0.38 |
| S. hyicus 437-2 c | 32 | 12.9 | 8 | 3.23 | >64 | >26.49 | 32 | 12.5 | 32 | 12.16 | 8 | 24.14 |
| Fungus | ||||||||||||
| Candida albicans CMCC98001 | >128 | >52 | 128 | 51.72 | >128 | >52.98 | >128 | >50.02 | >128 | >48.6 | >128 | >386.3 |
| Peptides | PAE (h) | ||
|---|---|---|---|
| 1× MIC | 2× MIC | 4× MIC | |
| N6NH2 | 0.69 ± 0.01 | 0.7 ± 0.01 | 1.17 ± 0.22 |
| DN6NH2 | 0.68 ± 0.01 | 1.13 ± 0.11 | 3.36 ± 0.19 |
| N6PNH2 | 0.52 ± 0.01 | 0.62 ± 0.01 | 0.68 ± 0.01 |
| V112N6NH2 | 0.74 ± 0.02 | 1.48 ± 0.05 | 2.15 ± 0.13 |
| Guo-N6NH2 | 0.62 ± 0.01 | 1.32 ± 0.08 | 2.07 ± 0.08 |
| CIP | 0.6 ± 0.01 | 0.63 ± 0.1 | 0.67 ± 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, T.; Wang, Z.; Han, H.; Teng, D.; Mao, R.; Hao, Y.; Yang, N.; Wang, X.; Wang, J. Dual Antibacterial Activities and Biofilm Eradication of a Marine Peptide-N6NH2 and Its Analogs against Multidrug-Resistant Aeromonas veronii. Int. J. Mol. Sci. 2020, 21, 9637. https://doi.org/10.3390/ijms21249637
Li T, Wang Z, Han H, Teng D, Mao R, Hao Y, Yang N, Wang X, Wang J. Dual Antibacterial Activities and Biofilm Eradication of a Marine Peptide-N6NH2 and Its Analogs against Multidrug-Resistant Aeromonas veronii. International Journal of Molecular Sciences. 2020; 21(24):9637. https://doi.org/10.3390/ijms21249637
Chicago/Turabian StyleLi, Ting, Zhenlong Wang, Huihui Han, Da Teng, Ruoyu Mao, Ya Hao, Na Yang, Xiumin Wang, and Jianhua Wang. 2020. "Dual Antibacterial Activities and Biofilm Eradication of a Marine Peptide-N6NH2 and Its Analogs against Multidrug-Resistant Aeromonas veronii" International Journal of Molecular Sciences 21, no. 24: 9637. https://doi.org/10.3390/ijms21249637








