Molecular Rescue of Dyrk1A Overexpression Alterations in Mice with Fontup® Dietary Supplement: Role of Green Tea Catechins
Abstract
:1. Introduction
2. Results
2.1. Quantification of FontUp® Polyphenols
2.2. In Vitro Inhibition and Molecular Docking of DYRK1A
2.3. Effects of Low, Intermediate and High Doses of FontUp® Administration on Plasma Biomarkers Linked with DYRK1A
2.4. Effects of the Higher Dose of FontUp® Administration on Liver and Cardiac Biomarkers
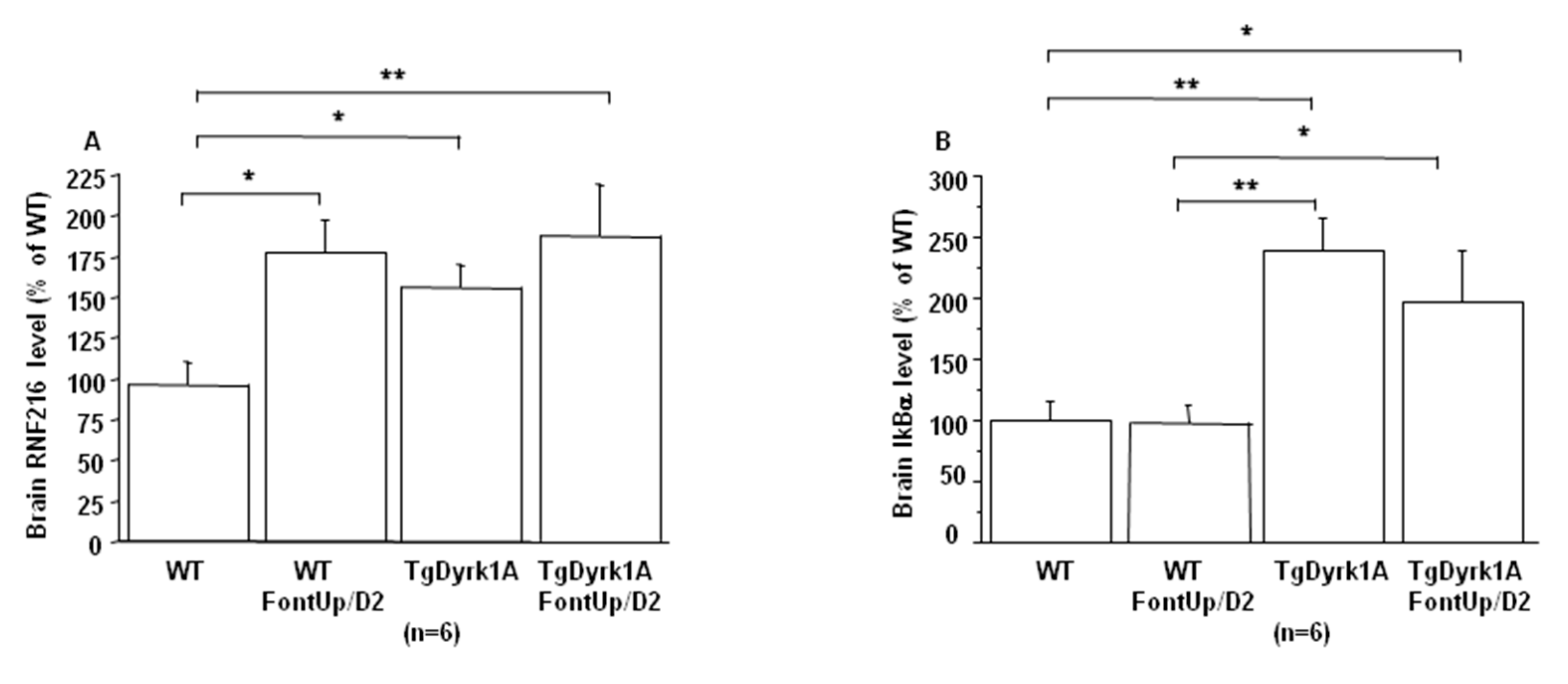
2.5. Effects of the Intermediate Dose of FontUp® Administration on Brain Biomarkers Linked with DYRK1A
3. Discussion
4. Materials and Methods
4.1. Quantification of FontUp® Polyphenols
4.2. Experimental Mice Model
4.3. FontUp® Treatment
4.4. Preparation of Serum Samples, Tissue Collection, and Plasma Assays
4.5. Protein Extraction and Analysis
4.6. Expression and Purification of Truncated DYRK1A (DYRK1A-ΔC)
4.7. DYRK1A Inhibition Assays
4.8. Quantification of FontUp® Polyphenols in Plasma and Brain Samples
4.9. Molecular Docking
4.10. Data Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Akt | Protein kinase B |
| ALT | Alanine aminotransferase |
| αSMA | α Smooth muscle actin |
| BBB | Blood-brain barrier |
| BDNF | Brain derived neurotrophic factor |
| DAD | Diode array detection |
| DS | Down syndrome |
| DYRK1A | Dual-specificity tyrosine phosphorylation-related kinase 1A |
| EC | Epicatechin |
| ECG | Epicatechin gallate |
| EGC | Epigallocatechin |
| EGCG | Epigallocatechin-3-gallate |
| Erk | Extracellular signal-regulated kinase |
| GSH | Glutathione |
| HPLC | High-performance liquid chromatography |
| hcy | Homocysteine |
| PAkt | Phosphor-Akt |
| Perk | Phosphor-Erk |
| RNF216 | Ring Finger Protein 216 |
| Truncated DYRK1A | Truncated dual-specificity tyrosine phosphorylation-related kinase 1A |
| WT | Wild type |
References
- Becker, W.; Joost, H.-G. Structural and functional characteristics of dyrk, a novel subfamily of protein kinases with dual specificity. Prog. Nucleic Acid. Res. Mol. Biol. 1998, 62, 1–17. [Google Scholar]
- Dierssen, M. Down syndrome: The brain in trisomic mode. Nat. Rev. Neurosci. 2012, 13, 844–858. [Google Scholar] [CrossRef]
- Tejedor, F.J.; Hämmerle, B. Mnb/dyrk1a as a multiple regulator of neuronal development. FEBS J. 2011, 278, 223–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duchon, A.Y.; Herault, Y. DYRK1A, a Dosage-Sensitive Gene Involved in Neurodevelopmental Disorders, Is a Target for Drug Development in Down Syndrome. Front. Behav. Neurosci. 2016, 10, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Becker, W.; Sippl, W. Activation, regulation, and inhibition of dyrk1a. FEBS J. 2011, 278, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Guedj, F.; Sébrié, C.; Rivals, I.S.; Ledru, A.; Paly, E.; Bizot, J.C.; Smith, D.; Rubin, E.; Gillet, B.; Arbones, M.; et al. Green tea polyphenols rescue of brain defects induced by overexpression of Dyrk1a. PLoS ONE 2009, 4, e4606. [Google Scholar] [CrossRef] [Green Version]
- Pons-Espinal, M.; Martinez de Lagran, M.; Dierssen, M. Environmental enrichment rescues dyrk1a activity and hippocampal adult neurogenesis in tgdyrk1a. Neurobiol. Dis. 2013, 60, 18–31. [Google Scholar] [CrossRef]
- De la Torre, R.; De Sola, S.; Pons, M.; Duchon, A.; de Lagran, M.M.; Farré, M.; Fitó, M.; Benejam, B.; Langohr, K.; Rodriguez, J.; et al. Epigallocatechin-3-gallate, a dyrk1a inhibitor, rescues cognitive deficits in down syndrome mouse models and in humans. Mol. Nut. Food Res. 2014, 58, 278–288. [Google Scholar] [CrossRef]
- Thomazeau, A.; Lassalle, O.; Iafrati, J.; Souchet, B.; Guedj, F.; Janel, N.; Chavis, P.; Delabar, J.; Manzoni, O.J. Prefrontal deficits in a murine model overexpressing the down syndrome candidate gene Dyrk1a. J Neurosci. 2014, 34, 1138–1147. [Google Scholar] [CrossRef] [Green Version]
- Catuara-Solarz, S.; Espinosa-Carrasco, J.; Erb, I.; Langohr, K.; Notredame, C.; Gonzalez, J.R.; Dierssen, M. Principal component analysis of the effects of environmental enrichment and (-)-epigallocatechin-3-gallate on age-associated learning deficits in a mouse model of down syndrome. Front. Behav. Neurosci. 2015, 9, 330. [Google Scholar] [CrossRef]
- Souchet, B.; Guedj, F.; Penke-Verdier, Z.; Daubigney, F.; Duchon, A.; Herault, Y.; Bizot, J.C.; Janel, N.; Créau, N.; Delatour, B.; et al. Pharmacological correction of excitation/inhibition imbalance in down syndrome mouse models. Front. Behav. Neurosci. 2015, 9, 267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stringer, M.; Abeysekera, I.; Dria, K.J.; Roper, R.J.; Goodlett, C.R. Low dose egcg treatment beginning in adolescence does not improve cognitive impairment in a down syndrome mouse model. Pharmacol. Biochem. Behav. 2015, 138, 70–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stagni, F.; Giacomini, A.; Emili, M.; Trazzi, S.; Guidi, S.; Sassi, M.; Ciani, E.; Rimondini, R.; Bartesaghi, R. Short- and long-term effects of neonatal pharmacotherapy with epigallocatechin-3-gallate on hippocampal development in the Ts65dn mouse model of down syndrome. Neurosci 2016, 333, 277–301. [Google Scholar] [CrossRef] [PubMed]
- Stringer, M.; Abeysekera, I.; Thomas, J.; LaCombe, J.; Stancombe, K.; Stewart, R.J.; Dria, K.J.; Wallace, J.M.; Goodlett, C.R.; Roper, R.J. Epigallocatechin-3-gallate (egcg) consumption in the ts65dn model of down syndrome fails to improve behavioral deficits and is detrimental to skeletal phenotypes. Physiol. Behav. 2017, 177, 230–241. [Google Scholar] [CrossRef]
- Souchet, B.; Duchon, A.; Gu, Y.; Dairou, J.; Chevalier, C.; Daubigney, F.; Nalesso, V.; Créau, N.; Yu, Y.; Janel, N.; et al. Prenatal treatment with EGCG enriched green tea extract rescues GAD67 related developmental and cognitive defects in Down syndrome mouse models. Sci. Rep. 2019, 9, 3914. [Google Scholar] [CrossRef] [Green Version]
- De la Torre, R.; de Sola, S.; Hernandez, G.; Farré, M.; Pujol, J.; Rodriguez, J.; Espadaler, J.M.; Langohr, K.; Cuenca-Royo, A.; Principe, A.; et al. Safety and efficacy of cognitive training plus epigallocatechin-3-gallate in young adults with down’s syndrome (tesdad): A double-blind, randomised, placebo-controlled, phase 2 trial. Lancet Neurol. 2016, 15, 801–810. [Google Scholar] [CrossRef]
- Bain, J.; Plater, L.; Elliott, M.; Shpiro, N.; Hastie, C.J.; McLauchlan, H.; Klevernic, I.; Arthur, J.S.; Alessi, D.R.; Cohen, P. The selectivity of protein kinase inhibitors: A further update. Biochem. J. 2007, 408, 297–315. [Google Scholar] [CrossRef] [Green Version]
- Abekhoukh, S.; Planque, C.; Ripoll, C.; Urbaniak, P.; Paul, J.L.; Delabar, J.M.; Janel, N. Dyrk1A, a serine/threonine kinase, is involved in ERK and Akt activation in brain of hyperhomocysteinemic mice. Mol. Neurobiol. 2013, 47, 105–116. [Google Scholar] [CrossRef]
- Noll, C.; Planque, C.; Ripoll, C.; Guedj, F.; Diez, A.; Ducros, V.; Belin, N.; Duchon, A.; Paul, J.L.; Badel, A.; et al. DYRK1A, a novel determinant of the methionine-homocysteine cycle in different mouse models overexpressing this Down-syndrome-associated kinase. PLoS ONE 2009, 4, e7540. [Google Scholar] [CrossRef]
- Renon, M.; Legrand, B.; Blanc, E.; Daubigney, F.; Bokobza, C.; Mortreux, M.; Paul, J.L.; Delabar, J.M.; Rouach, H.; Andreau, K.; et al. Impact of Dyrk1A concentration on alcohol metabolism. Biochim. Biophys. Acta 2016, 1862, 1495–1503. [Google Scholar] [CrossRef]
- Cai, Y.; He, S.Q.; Hong, H.Q.; Cai, Y.P.; Zhao, L.; Zhang, M. High doses of (-)-epigallocatechin-3-gallate from green tea induces cardiac fibrosis in mice. Biotechnol. Lett. 2015, 37, 2371–2377. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.S.; Beer, S.; Kearney, D.L.; Phillips, G.; Carter, B.A. green tea extract: A potential cause of acute liver failure. World J. Gastroenterol. 2013, 19, 5174–5177. [Google Scholar] [CrossRef] [PubMed]
- Raaf, L.; Noll, C.; Cherifi, M.; Benazzoug, Y.; Delabar, J.M.; Janel, N. Hyperhomocysteinemia-induced Dyrk1a downregulation results in cardiomyocyte hypertrophy in rats. Int. J. Cardiol. 2010, 145, 306–307. [Google Scholar] [CrossRef] [PubMed]
- Toiber, D.; Azkona, G.; Ben-Ari, S.; Torán, N.; Soreq, H.; Dierssen, M. Engineering DYRK1A overdosage yields Down syndrome-characteristic cortical splicing aberrations. Neurobiol. Dis. 2010, 40, 348–359. [Google Scholar] [CrossRef]
- Wang, L.; Yang, G.; Yuan, L.; Yang, Y.; Zhao, H.; Ho, C.T.; Li, S. Green tea catechins effectively altered hepatic fibrogenesis in rats by inhibiting ERK and Smad1/2 phosphorylation. J. Agric. Food Chem. 2019, 67, 5437–5445. [Google Scholar] [CrossRef]
- Latour, A.; Gu, Y.; Kassis, N.; Daubigney, F.; Colin, C.; Gausserès, B.; Middendorp, S.; Paul, J.L.; Hindié, V.; Rain, J.C.; et al. LPS-induced inflammation abolishes the effect of DYRK1A on IkB stability in the brain of mice. Mol. Neurobiol. 2019, 56, 963–975. [Google Scholar] [CrossRef]
- Gardiner, K.J. Pharmacological approaches to improving cognitive function in Down syndrome: Current status and considerations. Drug Des. Devel. Ther. 2015, 9, 103–125. [Google Scholar] [CrossRef] [Green Version]
- Abeysekera, I.; Thomas, J.; Georgiadis, T.M.; Berman, A.G.; Hammond, M.A.; Dria, K.J.; Wallace, J.M.; Roper, R.J. Differential effects of epigallocatechin-3-gallate containing supplements on correcting skeletal defects in a Down syndrome mouse model. Mol. Nutr. Food Res. 2016, 60, 717–726. [Google Scholar] [CrossRef] [Green Version]
- Hogas, S.; Bilha, S.C.; Branisteanu, D.; Hogas, D.M.; Gaipov, A.; Kanbay, M.; Covic, A. Potential novel biomarkers of cardiovascular dysfunction and disease: Cardiotrophin-1, adipokines and galectin-3. Arch. Med. Sci. 2017, 4, 897–913. [Google Scholar] [CrossRef]
- Muhammed, I.; Sankar, S.; Govindaraj, S. Ameliorative effect of epigallocatechin gallate on cardiac hypertrophy and fibrosis in aged rats. J. Cardiovasc. Pharmacol. 2018, 71, 65–75. [Google Scholar] [CrossRef]
- Tadano, N.; Du, C.K.; Yumoto, F.; Morimoto, S.; Ohta, M.; Xie, M.F.; Nagata, K.; Zhan, D.Y.; Lu, K.W.; Miwa, Y. Biological actions of green tea catechins on cardiac troponin C. Br. J. Pharmacol. 2010, 161, 1034–1043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chyu, K.Y.; Babbidge, S.M.; Zhao, X.; Dandillaya, R.; Rielveld, A.G.; Yano, J.; Dimayuga, P.; Cercek, B.; Shah, P.K. Differential effects of green tea-derived catechin on developing versus established atherosclerosis in apolipoprotein E-null mice. Circulation 2004, 109, 2448–2453. [Google Scholar] [CrossRef] [Green Version]
- Ludwig, A.; Lorenz, M.; Grimbo, N.; Steinle, F.; Meiners, S.; Bartsch, C.; Stangl, K.; Baumann, G.; Stangl, V. The tea flavonoid epigallocatechin-3-gallate reduces cytokine-induced VCAM-1 expression and monocyte adhesion to endothelial cells. Biochem. Biophys. Res. Commun. 2004, 316, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.C.; Wang, M.N.; Tseng, T.Y.; Sung, J.S.; Tsai, T.H. Pharmacokinetics of (−)-epigallocatechin-3-gallate in conscious and freely moving rats and its brain regional distribution. J. Agric. Food Chem. 2007, 55, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Pervin, M.; Unno, K.; Nakagawa, A.; Takahashi, Y.; Iguchi, K.; Yamamoto, H.; Hoshino, A.; Hara, A.; Takagaki, A.; Nanjo, F.; et al. Blood brain barrier permeability off (-)-epigallocatechin gallate, its proliferation-enhancing activity of human neuroblastoma SH-SY5Y cells, and its preventive effect on age-related cognitive dysfunction in mice. Biochem. Biophys. Rep. 2017, 9, 180–186. [Google Scholar] [PubMed]
- Unno, K.; Pervin, M.; Nakagawa, A.; Iguchi, K.; Hara, A.; Takagaki, A.; Nanjo, F.; Minami, A.; Nakamura, Y. Blood-brain barrier permeability of green tea catechin metabolites and their neuritogenic activity in human neuroblastoma SH-SY5Y cells. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Kang, Y.; Dong, X.X.; Zhou, Q.; Zhang, Y.; Cheng, R.; Hu, C.; Su, C.; Jin, H.; Liu, X.; Ma, D.; et al. Identification of novel candidate maternal serum protein markers for Down syndrome by integrated proteomic and bioinformatic analysis. Prenat. Diagn. 2012, 32, 284–292. [Google Scholar] [CrossRef]
- Zagrebelsky, M.; Korte, M. Form follows function: BDNF and its involvement in sculpting the function and structure of synapses. Neuropharmacology 2014, 76, 628–638. [Google Scholar] [CrossRef]
- Parrini, M.; Ghezzi, D.; Deidda, G.; Medrihan, L.; Castroflorio, E.; Alberti, M.; Baldelli, P.; Cancedda, L.; Contectabile, A. Aerobic exercice and a BDNF-mimetic therapy rescue learning and memory in a mouse model of Down syndrome. Sci. Rep. 2017, 7, 16825. [Google Scholar] [CrossRef]
- Shimizu, M.; Adachi, S.; Masuda, M.; Kozawa, O.; Moriwaki, H. Cancer chemoprevention with green tea catechins by targeting receptor tyrosine kinases. Mol. Nutr. Food Res. 2011, 55, 832–843. [Google Scholar] [CrossRef]
- Yang, C.S.; Lambert, J.D.; Ju, J.; Lu, G.; Sang, S. Tea and cancer prevention: Molecular mechanisms and human relevance. Toxicol. Appl. Pharmacol. 2007, 224, 265–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, F.; Oz, H.S.; Barve, S.; de Villiers, W.J.; McClain, C.J.; Varilek, G.W. The green tea polyphenol (-)-epigallocatechin-3-gallate blocks nuclear factor-kappa B activation by inhibiting I kappa B kinase activity in the intestinal epithelial cell line IEC-6. Mol. Pharmacol. 2001, 60, 528–533. [Google Scholar] [PubMed]
- Kumazoe, M.; Nakamura, Y.; Yamashita, M.; Suzuki, T.; Takamatsu, K.; Huang, Y.; Bae, J.; Yamashita, S.; Murata, M.; Yamada, S.; et al. Green Tea Polyphenol Epigallocatechin-3-gallate Suppresses Toll-like Receptor 4 Expression via Up-regulation of E3 Ubiquitin-protein Ligase RNF216. J. Biol. Chem. 2017, 292, 4077–4088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guedj, F.; Pereira, P.L.; Najas, S.; Barallobre, M.J.; Chabert, C.; Souchet, B.; Sebrie, C.; Verney, C.; Herault, Y.; Arbones, M.; et al. DYRK1A: A master regulatory protein controlling brain growth. Neurobiol. Dis. 2012, 46, 190–203. [Google Scholar] [CrossRef] [PubMed]
- Ducros, V.; Demuth, K.; Sauvant, M.; Quillard, M.; Caussé, E.; Candito, M.; Read, M.H.; Drai, J.; Garcia, I.; Gerhardt, M.F. SFBC Working group on homocysteine. French Society for Clinical Biology. Methods for homocysteine analysis and biological relevance of the results. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2002, 781, 207–226. [Google Scholar] [CrossRef]
- Bui, L.C.; Tabouy, L.; Busi, F.; Dupret, J.M.; Janel, N.; Planque, C.; Delabar, J.M.; Rodrigues-Lima, F.; Dairou, J. A high performance liquid chromatography assay for Dyrk1a, a Down syndrome-associated kinase. Anal. Biochem. 2014, 449, 172–178. [Google Scholar] [CrossRef]
- Maeda-Yamamoto, M.; Ema, K.; Monobe, M.; Tokuda, Y.; Tachibana, H. Epicatechin-3-O-(3″-O-methyl)-gallate content in various tea cultivars (Camellia sinensis L.) and its in vitro inhibitory effect on histamine release. Agric. Food Chem. 2012, 60, 2165–2170. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Burne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [Green Version]
- Olsson, M.H.; Søndergaard, C.R.; Rostkowski, M.; Jensen, J.H. PROPKA3: Consistent Treatment of Internal and Surface Residues in Empirical pKa Predictions. J. Chem. Theory Comput. 2011, 7, 525–537. [Google Scholar] [CrossRef]
- Koes, D.R.; Baumgartner, M.P.; Camacho, C.J. Lessons learned in empirical scoring with smina from the CSAR 2011 benchmarking exercise. J. Chem. Inf. Model 2013, 53, 1893–1904. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quiroga, R.; Villarreal, M.A. Vinardo: A Scoring Function Based on Autodock Vina Improves Scoring, Docking, and Virtual Screening. PLoS ONE 2016, 11, e0155183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delano, W.L. The PyMOL Molecular Graphics System; De Lano Scientific: San Carlos, CA, USA, 2002. [Google Scholar]






| Compound | Structure | DYRK1A-ΔC Remaining Activity at 0.1 µM (%) | DYRK1A-ΔC Remaining Activity at 1 µM (%) | DYRK1A-ΔC Remaining Activity at 10 µM (%) | DYRK1A-ΔC Remaining Activity at 100 µM (%) |
|---|---|---|---|---|---|
| ECG |  | 78.1 ± 3.6 (n = 8) | 27.1 ± 7.3 (n = 8) | 11.8 ± 4.2 (n = 8) | 7.6 ± 2.3 (n = 8) |
| EGC |  | 99.5 ± 3.2 (n = 4) | 93.6 ± 2.6 (n = 4) | 80.6 ± 11.4 (n = 4) | 81.4 ± 12.8 (n = 4) |
| EGCG |  | 71.3 ± 4.6 (n = 13) | 16.3 ± 2.3 (n = 13) | 6.4 ± 1.1 (n = 13) | 3.5 ± 0.8 (n = 13) |
| EC |  | 89.2 ± 9.5 (n = 4) | 84.1 ± 9.1 (n = 4) | 95.6 ± 8.1 (n = 4) | 88.7 ± 8.2 (n = 4) |
| Compound | Structure | DYRK1A Remaining Activity at 0.1 µM (%) | DYRK1A Remaining Activity at 1 µM (%) | DYRK1A Remaining Activity at 10 µM (%) | DYRK1A Remaining Activity at 100 µM (%) |
|---|---|---|---|---|---|
| ECG |  | 69.9 ± 3.9 (n = 4) | 12.5 ± 2.4 (n = 4) | 4.6 ± 0.9 (n = 4) | 2.3 ± 0.5 (n = 4) |
| EGCG |  | 67.5 ± 9.4 (n = 7) | 10 ± 2 (n = 7) | 3.4 ± 0.6 (n = 7) | 2.1 ± 0.5 (n = 7) |
| Compound | DYRK1A-ΔC Remaining Activity With 200 µM ATP (%) | DYRK1A-ΔC Remaining Activity with 400 µM ATP (%) | DYRK1A-ΔC Remaining Activity with 800 µM ATP (%) |
|---|---|---|---|
| ECG (10 µM) | 8.0 ± 0.8 (n = 4) | 6.5 ± 0.9 (n = 4) | 4.0 ± 0.5 (n = 4) |
| EGCG (10 µM) | 11.0 ± 3.1 (n = 4) | 9.6 ± 2.1 (n = 4) | 5.4 ± 1.3 (n = 4) |
| Compound | DYRK1A Remaining Activity with 200 µM ATP (%) | DYRK1A Remaining Activity with 400 µM ATP (%) | DYRK1A Remaining Activity with 800 µM ATP (%) |
|---|---|---|---|
| ECG (10 µM) | 9.4 ± 1.4 (n = 4) | 7.0 ± 1.1 (n = 4) | 5.4 ± 0.9 (n = 4) |
| EGCG (10 µM) | 6.3 ± 0.9 (n = 4) | 5.1 ± 0.9 (n = 4) | 4.0 ± 0.7 (n = 4) |
| WT CTL | WT D1 | WT D2 | WT D3 | |
|---|---|---|---|---|
| EGCG (nM) | 0 | 28 ± 0.0003 | 63 ± 10.8 | 104 ± 11.2 |
| hcy (µM) | 2.7 ± 0.1 | 2.9 ± 0.3 | 3.6 ± 0.3 * | 2.7 ± 0.2 |
| GSH (µM) | 66.4 ± 3.5 | 71.2 ± 2.6 | 75.2 ± 3.3 ** | 88.4 ± 2.7 *** |
| ALT (mU/mL) | 27.4 ± 7.1 | 17.1 ± 5 | 14.9 ± 4.3 * | 27.1 ± 4.9 |
| Galectin-3 (pg/mL) | 5.1 ± 0.3 | 5.2 ± 0.2 | 4.9 ± 0.25 | 5.1 ± 0.15 |
| WT | TgDyrk1A | |
|---|---|---|
| Plasma EGCG (µM) | 0.065 ± 0.023 | 0.036 ± 0.011 |
| Brain EGCG (µM) | 0.025 ± 0.007 | 0.050 ± 0. 01 $ |
| Kp | 1.2 ± 0.33 | 3.6 ± 0.82 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gu, Y.; Moroy, G.; Paul, J.-L.; Rebillat, A.-S.; Dierssen, M.; de la Torre, R.; Cieuta-Walti, C.; Dairou, J.; Janel, N. Molecular Rescue of Dyrk1A Overexpression Alterations in Mice with Fontup® Dietary Supplement: Role of Green Tea Catechins. Int. J. Mol. Sci. 2020, 21, 1404. https://doi.org/10.3390/ijms21041404
Gu Y, Moroy G, Paul J-L, Rebillat A-S, Dierssen M, de la Torre R, Cieuta-Walti C, Dairou J, Janel N. Molecular Rescue of Dyrk1A Overexpression Alterations in Mice with Fontup® Dietary Supplement: Role of Green Tea Catechins. International Journal of Molecular Sciences. 2020; 21(4):1404. https://doi.org/10.3390/ijms21041404
Chicago/Turabian StyleGu, Yuchen, Gautier Moroy, Jean-Louis Paul, Anne-Sophie Rebillat, Mara Dierssen, Rafael de la Torre, Cécile Cieuta-Walti, Julien Dairou, and Nathalie Janel. 2020. "Molecular Rescue of Dyrk1A Overexpression Alterations in Mice with Fontup® Dietary Supplement: Role of Green Tea Catechins" International Journal of Molecular Sciences 21, no. 4: 1404. https://doi.org/10.3390/ijms21041404






