Hybridization with Insect Cecropin A (1–8) Improve the Stability and Selectivity of Naturally Occurring Peptides
Abstract
1. Introduction
2. Results
2.1. Physicochemical Parameters of Peptides
2.2. Secondary Structures
2.3. Antimicrobial Activity
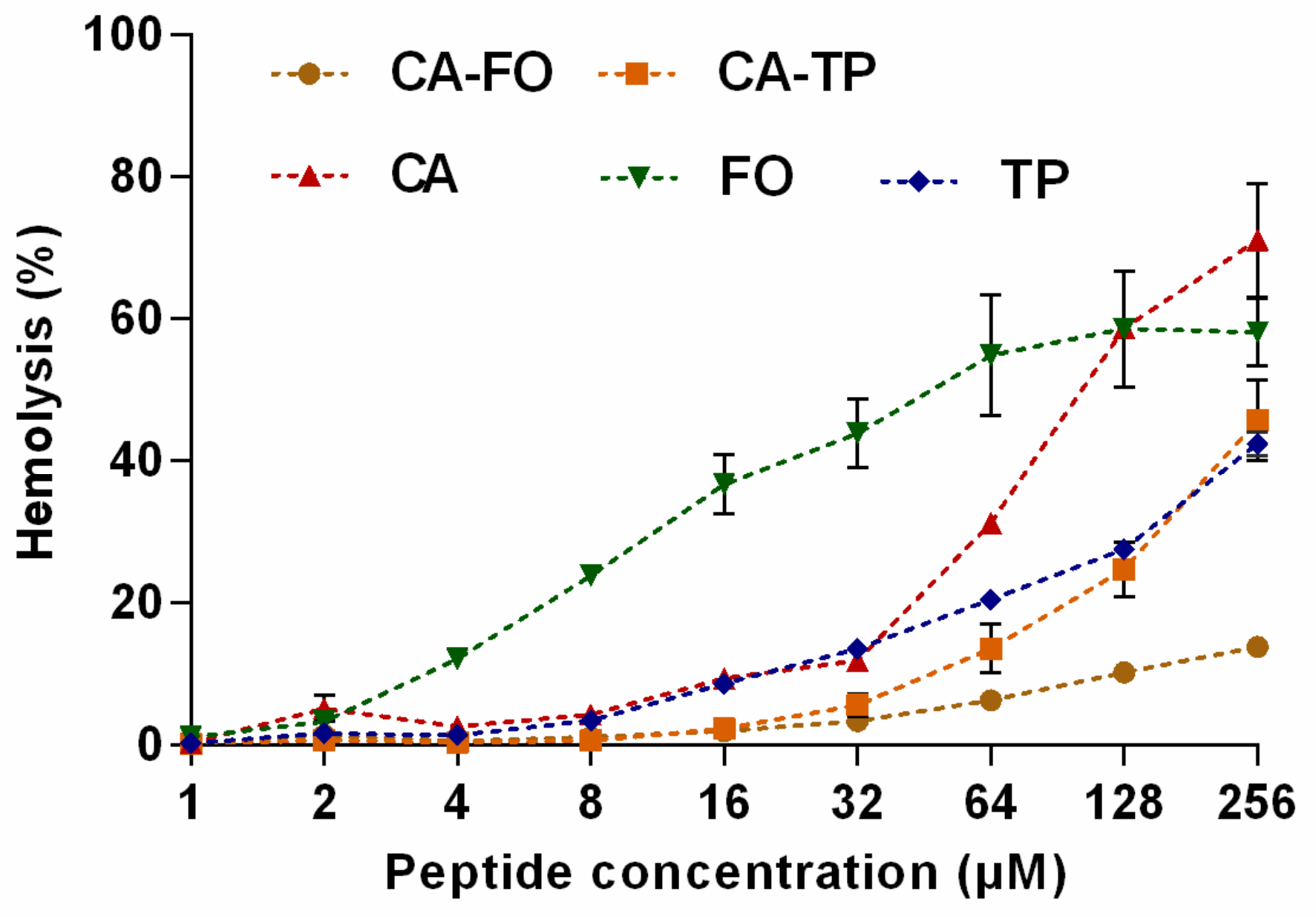
2.4. Hemolytic Activity and Cytotoxicity of the Peptides
2.5. Cell Selectivity of Peptides
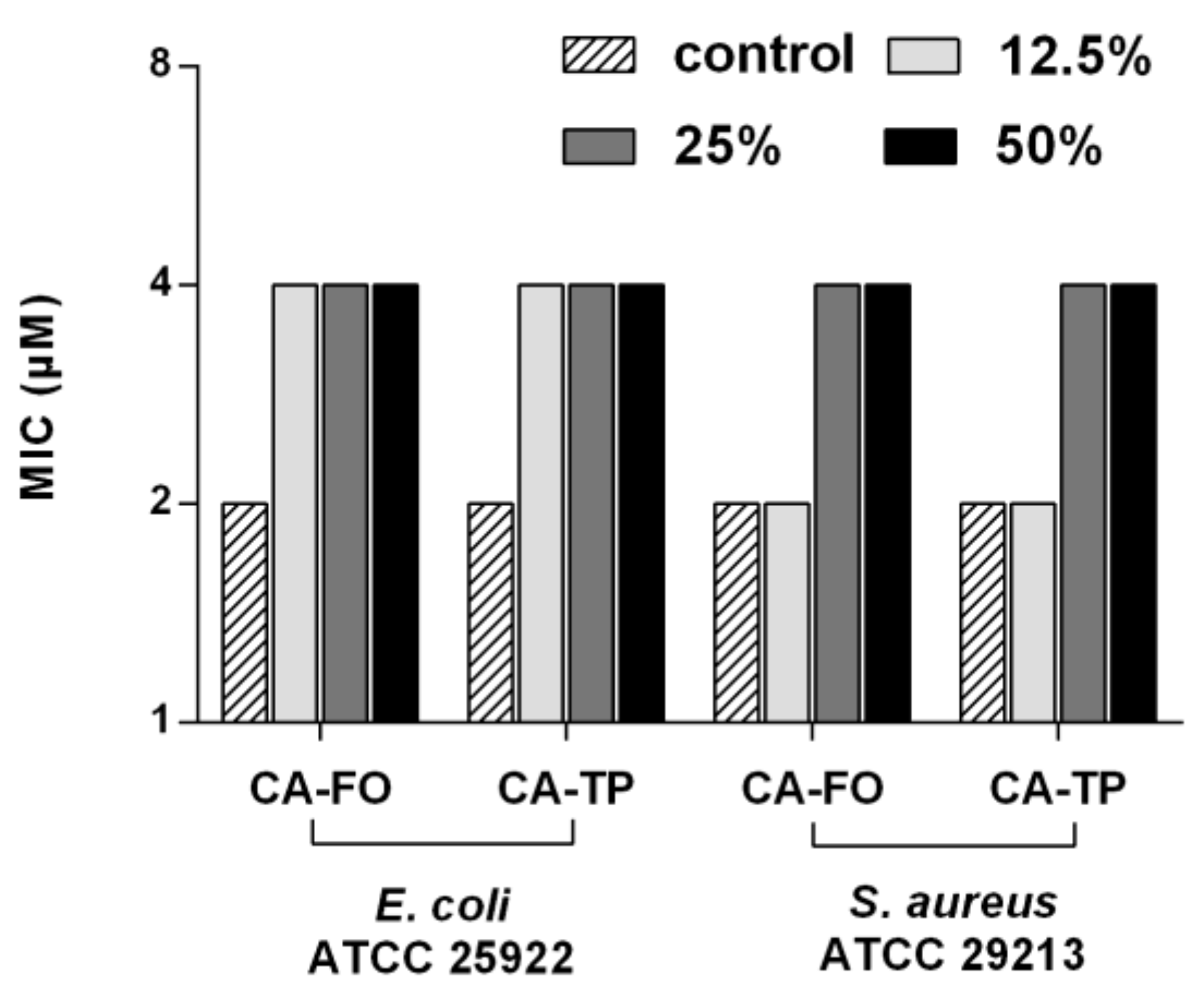
2.6. Salts and Serum Stability
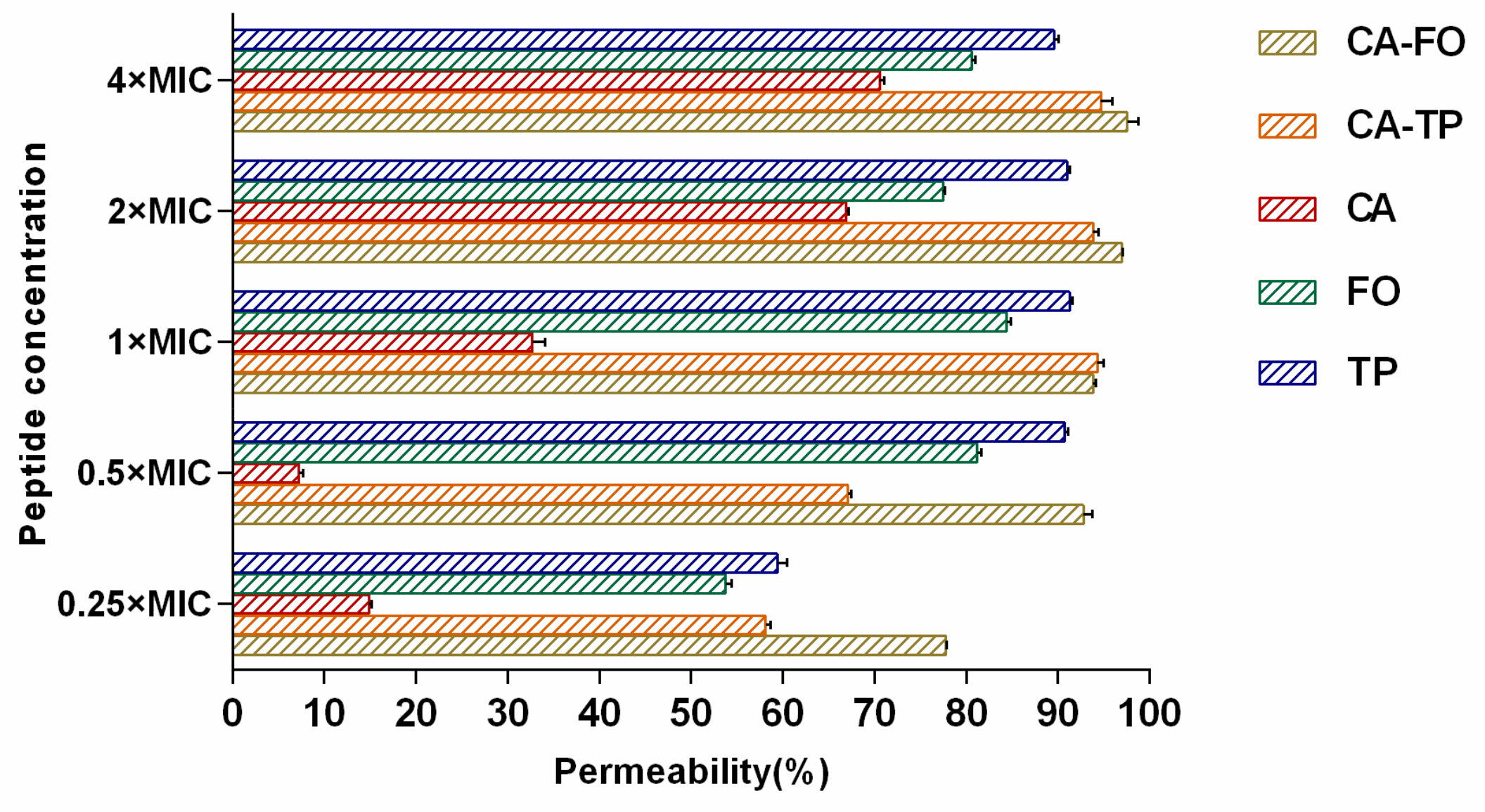
2.7. Outer Membrane Permeabilization
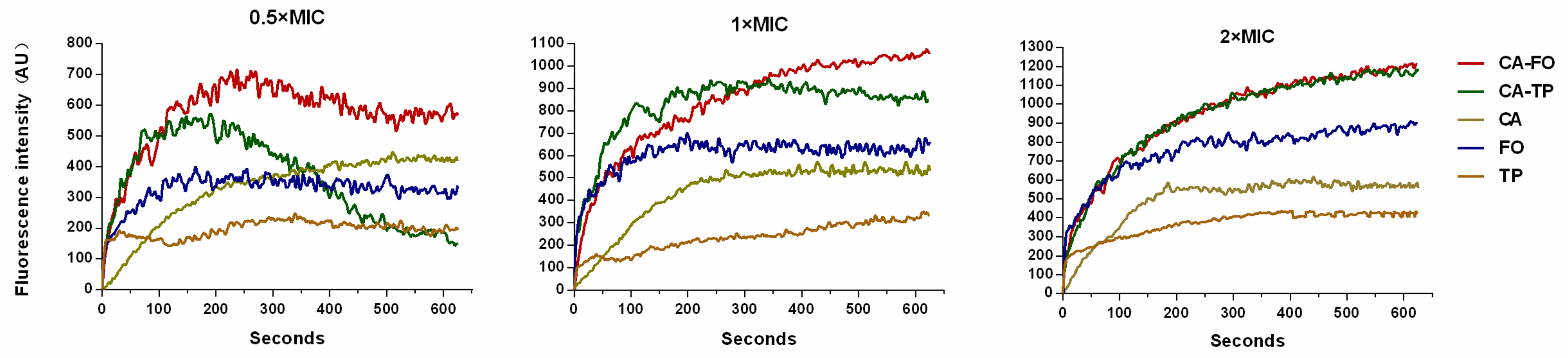
2.8. Cytoplasmic Membrane Depolarization
2.9. Membrane Morphological Analysis
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Mammalian Cells
4.2. Peptide Synthesis and Sequence Analysis
4.3. CD Spectroscopy
4.4. Antimicrobial Assays
4.5. Measurement of Hemolytic Activity
4.6. Cytotoxicity Assays
4.7. Calculation of the Therapeutic Index
4.8. Salt and Serum Stability
4.9. Outer Membrane Permeability Assay
4.10. Cytoplasmic Membrane Depolarization Assay
4.11. SEM and TEM Characterization
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AMR | Antimicrobial resistance |
| AMPs | Antimicrobial peptides |
| LPS | Lipopolysaccharides |
| LTA | Lipoteichoic acids |
| MALDI-TOF-MS | Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry |
| GRAVY | Grand average of hydropathicity |
| CD | Circular dichroism |
| PBS | Phosphate-buffered saline |
| TFE | Trifluoroethanol |
| MICs | The minimum inhibitory concentrations |
| MTT | 3-[4,5-dimethylthiozol-2-yl]-2,5-diphenyltetrazolium bromide |
| TI | Therapeutic index |
| NPN | 1-N-phenylnaphthylamine |
| DiSC3–5 | 3,3′-dipropylthiadicarbocyanine |
| RP-HPLC | Reverse-phase high performance liquid chromatography |
References
- World Health Organization. Antimicrobial Resistance and Primary Health Care: Brief; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Hofer, U. The cost of antimicrobial resistance. Nat. Rev. Microbiol. 2019, 17, 3. [Google Scholar] [CrossRef] [PubMed]
- Rončević, T.; Gajski, G.; Ilić, N.; Goić-Barišić, I.; Tonkić, M.; Zoranić, L.; Simunić, J.; Benincasa, M.; Mijaković, M.; Tossi, A. PGLa-H tandem-repeat peptides active against multidrug resistant clinical bacterial isolates. BBA Biomembr. 2017, 1859, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Wang, Y.; Wang, M.; Tian, Y.; Kang, W.; Liu, H.; Wang, H.; Dou, J.; Zhou, C. Effective antimicrobial activity of Cbf-14, derived from a cathelin-like domain, against penicillin-resistant bacteria. Biomaterials 2016, 87, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.; Haney, E.F.; Gill, E.E. The immunology of host defence peptides: Beyond antimicrobial activity. Nat. Rev. Immunol. 2016, 16, 321–334. [Google Scholar] [CrossRef]
- Hilchie, A.L.; Wuerth, K.; Hancock, R.E. Immune modulation by multifaceted cationic host defense (antimicrobial) peptides. Nat. Chem. Biol. 2013, 9, 761–768. [Google Scholar] [CrossRef]
- Noore, J.; Noore, A.; Li, B. Cationic antimicrobial peptide LL-37 is effective against both extra-and intracellular Staphylococcus aureus. Antimicrob. Agents Chemother. 2013, 57, 1283–1290. [Google Scholar] [CrossRef]
- Lee, D.G.; Kim, D.H.; Park, Y.; Kim, H.K.; Kim, H.N.; Shin, Y.K.; Choi, C.H.; Hahm, K.S. Fungicidal effect of antimicrobial peptide, PMAP-23, isolated from porcine myeloid against Candida albicans. Biochem. Biophys. Res. Commun. 2001, 282, 570–574. [Google Scholar] [CrossRef]
- Cochrane, S.A.; Findlay, B.; Bakhtiary, A.; Acedo, J.Z.; Rodriguez-Lopez, E.M.; Mercier, P.; Vederas, J.C. Antimicrobial lipopeptide tridecaptin A1 selectively binds to Gram-negative lipid II. Proc. Natl. Acad. Sci. USA 2016, 113, 11561–11566. [Google Scholar] [CrossRef]
- Li, G.R.; He, L.Y.; Liu, X.Y.; Liu, A.P.; Huang, Y.B.; Qiu, C.; Zhang, X.Y.; Xu, J.Q.; Yang, W.; Chen, Y.X. Rational design of peptides with anti-HCV/HIV activities and enhanced specificity. Chem. Biol. Drug Des. 2011, 78, 835–843. [Google Scholar] [CrossRef]
- Wang, G. Improved methods for classification, prediction and design of antimicrobial peptides. Methods Mol. Biol. 2015, 1268, 43–66. [Google Scholar]
- Hancock, R.E.; Sahl, H.G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 2006, 24, 1551–1557. [Google Scholar] [CrossRef] [PubMed]
- Koehbach, J.; Craik, D.J. The vast structural diversity of antimicrobial peptides. Trends Pharm. Sci. 2019, 40, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, J.P.; Cova, M.; Ferreira, R.; Vitorino, R. Antimicrobial peptides: An alternative for innovative medicines? Appl. Microbiol. Biotechnol. 2015, 99, 2023–2040. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Koh, J.J.; Liu, S.; Lakshminarayanan, R.; Verma, C.S.; Beuerman, R.W. Membrane active antimicrobial peptides: Translating mechanistic insights to design. Front. Neurosci. 2017, 11, 73. [Google Scholar] [CrossRef] [PubMed]
- Malanovic, N.; Lohner, K. Gram-positive bacterial cell envelopes: The impact on the activity of antimicrobial peptides. BBA Biomembr. 2016, 1858, 936–946. [Google Scholar] [CrossRef]
- Wang, J.; Dou, X.; Song, J.; Lyu, Y.; Zhu, X.; Xu, L.; Li, W.; Shan, A. Antimicrobial peptides: Promising alternatives in the post feeding antibiotic era. Med. Res. Rev. 2019, 39, 831–859. [Google Scholar] [CrossRef]
- Le, C.F.; Fang, C.M.; Sekaran, S.D. Intracellular targeting mechanisms by antimicrobial peptides. Antimicrob. Agents Chemother. 2017, 61, e02340-16. [Google Scholar] [CrossRef]
- Liou, J.W.; Hung, Y.J.; Yang, C.H.; Chen, Y.C. The antimicrobial activity of gramicidin A is associated with hydroxyl radical formation. PLoS ONE 2015, 10, e0117065. [Google Scholar] [CrossRef]
- Langham, A.A.; Khandelia, H.; Kaznessis, Y.N. How can a β-sheet peptide be both a potent antimicrobial and harmfully toxic? Molecular dynamics simulations of protegrin-1 in micelles. Biopolymers 2006, 84, 219–231. [Google Scholar] [CrossRef]
- Goldman, M.J.; Anderson, G.M.; Stolzenberg, E.D.; Kari, U.P.; Zasloff, M.; Wilson, J.M. Human β-defensin-1 is a salt-sensitive antibiotic in lung that is inactivated in cystic fibrosis. Cell 1997, 88, 553–560. [Google Scholar] [CrossRef]
- Lv, Y.; Wang, J.; Gao, H.; Wang, Z.; Dong, N.; Ma, Q.; Shan, A. Antimicrobial properties and membrane-active mechanism of a potential α-helical antimicrobial derived from cathelicidin PMAP-36. PLoS ONE 2014, 9, e86364. [Google Scholar] [CrossRef] [PubMed]
- Dong, N.; Wang, C.; Li, X.; Guo, Y.; Li, X. Simplified head-to-tail cyclic polypeptides as biomaterial-associated antimicrobials with endotoxin neutralizing and anti-Inflammatory capabilities. Int. J. Mol. Sci. 2019, 20, 5904. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhu, X.; Tan, T.; Li, W.; Shan, A. Design of embedded-hybrid antimicrobial peptides with enhanced cell selectivity and anti-biofilm activity. PLoS ONE 2014, 9, e98935. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.; Wu, D.; Li, W.; Zheng, X.; Li, W.; Shan, A. High specific selectivity and membrane-active mechanism of synthetic cationic hybrid antimicrobial peptides based on the peptide FV7. Int. J. Mol. Sci. 2017, 18, 339. [Google Scholar] [CrossRef]
- Steiner, H.; Hultmark, D.; Engström, Å.; Bennich, H.; Boman, H. Sequence and specificity of two antibacterial proteins involved in insect immunity. Nature 1981, 292, 246–248. [Google Scholar] [CrossRef]
- Boman, H.; Wade, D.; Boman, I.; Wåhlin, B.; Merrifield, R. Antibacterial and antimalarial properties of peptides that are cecropin-melittin hybrids. FEBS Lett. 1989, 259, 103–106. [Google Scholar] [CrossRef]
- Cao, Y.; Yu, R.Q.; Liu, Y.; Zhou, H.X.; Song, L.L.; Cao, Y.; Qiao, D.R. Design, recombinant expression and antibacterial activity of the cecropins–melittin hybrid antimicrobial peptides. Curr. Microbiol. 2010, 61, 169–175. [Google Scholar] [CrossRef]
- Shin, S.Y.; Lee, M.K.; KIM, K.L.; Hahm, K.S. Structure-antitumor and hemolytic activity relationships of synthetic peptides derived from cecropin A-magainin 2 and cecropin A-melittin hybrid peptides. J. Pept. Res. 1997, 50, 279–285. [Google Scholar] [CrossRef]
- Wei, X.B.; Wu, R.J.; Si, D.Y.; Liao, X.D.; Zhang, L.L.; Zhang, R.J. Novel hybrid peptide cecropin A (1–8)-LL37 (17–30) with potential antibacterial activity. Int. J. Mol. Sci. 2016, 17, 983. [Google Scholar] [CrossRef]
- Xiao, Y.; Cai, Y.; Bommineni, Y.R.; Fernando, S.C.; Prakash, O.; Gilliland, S.E.; Zhang, G. Identification and functional characterization of three chicken cathelicidins with potent antimicrobial activity. J. Biol. Chem. 2006, 281, 2858–2867. [Google Scholar] [CrossRef]
- Yang, S.T.; Shin, S.Y.; Kim, Y.C.; Kim, Y.; Hahm, K.S.; Kim, J.I. Conformation-dependent antibiotic activity of tritrpticin, a cathelicidin-derived antimicrobial peptide. Biochem. Biophys. Res. Commun. 2002, 296, 1044–1050. [Google Scholar] [CrossRef]
- Ma, Z.; Wei, D.; Yan, P.; Zhu, X.; Shan, A.; Bi, Z. Characterization of cell selectivity, physiological stability and endotoxin neutralization capabilities of α-helix-based peptide amphiphiles. Biomaterials 2015, 52, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; de Boer, L.; Zaat, S.A.; Vogel, H.J. Investigating the cationic side chains of the antimicrobial peptide tritrpticin: Hydrogen bonding properties govern its membrane-disruptive activities. BBA Biomembr. 2011, 1808, 2297–2303. [Google Scholar] [CrossRef] [PubMed]
- Arias, M.; Nguyen, L.; Kuczynski, A.; Lejon, T.; Vogel, H. Position-dependent influence of the three trp residues on the membrane activity of the antimicrobial peptide, tritrpticin. J. Antibiot. 2014, 3, 595–616. [Google Scholar] [CrossRef] [PubMed]
- Slochower, D.R.; Wang, Y.-H.; Tourdot, R.W.; Radhakrishnan, R.; Janmey, P.A. Counterion-mediated pattern formation in membranes containing anionic lipids. Adv. Colloid Interface Sci. 2014, 208, 177–188. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marr, A.K.; Gooderham, W.J.; Hancock, R.E. Antibacterial peptides for therapeutic use: Obstacles and realistic outlook. Curr. Opin. Pharmacol. 2006, 6, 468–472. [Google Scholar] [CrossRef]
- Travkova, O.G.; Moehwald, H.; Brezesinski, G. The interaction of antimicrobial peptides with membranes. Adv. Colloid Interface Sci. 2017, 247, 521–532. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, L.; Wang, J.; Ma, Z.; Xu, W.; Li, J.; Shan, A. Characterization of antimicrobial activity and mechanisms of low amphipathic peptides with different α-helical propensity. Acta Biomater. 2015, 18, 155–167. [Google Scholar] [CrossRef]
- Wiradharma, N.; Liu, S.Q.; Yang, Y.Y. Branched and 4-arm starlike α-helical peptide structures with enhanced antimicrobial potency and selectivity. Small 2012, 8, 362–366. [Google Scholar] [CrossRef]
- Wiradharma, N.; Khan, M.; Yong, L.-K.; Hauser, C.A.E.; Seow, S.V.; Zhang, S.; Yang, Y.-Y. The effect of thiol functional group incorporation into cationic helical peptides on antimicrobial activities and spectra. Biomaterials 2011, 32, 9100–9108. [Google Scholar] [CrossRef]
- Lyu, Y.; Chen, T.; Shang, L.; Yang, Y.; Li, Z.; Zhu, J.; Shan, A. Design of Trp-rich dodecapeptides with broad-spectrum antimicrobial potency and membrane-disruptive mechanism. J. Med. Chem. 2019, 62, 6941–6957. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Yang, J.; Han, J.; Gao, L.; Liu, H.; Lu, Z.; Zhao, H.; Bie, X. Insights into the antimicrobial activity and cytotoxicity of engineered α-helical peptide amphiphiles. J. Med. Chem. 2016, 59, 10946–10962. [Google Scholar] [CrossRef] [PubMed]
- Findlay, B.; Zhanel, G.G.; Schweizer, F. Cationic amphiphiles, a new generation of antimicrobials inspired by the natural antimicrobial peptide scaffold. Antimicrob. Agents Chemother. 2010, 54, 4049–4058. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Guarnieri, M.T.; Vasil, A.I.; Vasil, M.L.; Mant, C.T.; Hodges, R.S. Role of peptide hydrophobicity in the mechanism of action of α-helical antimicrobial peptides. Antimicrob. Agents Chemother. 2007, 51, 1398–1406. [Google Scholar] [CrossRef] [PubMed]
- Mattei, B.; Miranda, A.; Perez, K.R.; Riske, K.A. Structure-activity relationship of the antimicrobial peptide gomesin: The role of peptide hydrophobicity in its interaction with model membranes. Langmuir 2014, 30, 3513–3521. [Google Scholar] [CrossRef]
- Liu, Y.; Xia, X.; Xu, L.; Wang, Y. Design of hybrid β-hairpin peptides with enhanced cell specificity and potent anti-inflammatory activity. Biomaterials 2013, 34, 237–250. [Google Scholar] [CrossRef]
- Lee, E.; Shin, A.; Jeong, K.-W.; Jin, B.; Jnawali, H.N.; Shin, S.; Shin, S.Y.; Kim, Y. Role of phenylalanine and valine10 residues in the antimicrobial activity and cytotoxicity of piscidin-1. PLoS ONE 2014, 9, e114453. [Google Scholar] [CrossRef]
- Eckert, R. Road to clinical efficacy: Challenges and novel strategies for antimicrobial peptide development. Future Microbiol. 2011, 6, 635–651. [Google Scholar] [CrossRef]
- Huang, J.; Hao, D.; Chen, Y.; Xu, Y.; Tan, J.; Huang, Y.; Li, F.; Chen, Y. Inhibitory effects and mechanisms of physiological conditions on the activity of enantiomeric forms of an α-helical antibacterial peptide against bacteria. Peptides 2011, 32, 1488–1495. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, R.; Li, X.; Lim, K.; Mohanram, H.; Peng, L.; Mishra, B.; Basu, A.; Lee, J.M.; Bhattacharjya, S.; Leong, S.S.J. Design of short membrane selective antimicrobial peptides containing tryptophan and arginine residues for improved activity, salt-resistance and biocompatibility. Biotechnol. Bioeng. 2014, 111, 37–49. [Google Scholar] [CrossRef]
- Mingeot-Leclercq, M.-P.; Décout, J.-L. Bacterial lipid membranes as promising targets to fight antimicrobial resistance, molecular foundations and illustration through the renewal of aminoglycoside antibiotics and emergence of amphiphilic aminoglycosides. MedChemComm 2016, 7, 586–611. [Google Scholar] [CrossRef]
- Maisetta, G.; Di, L.M.; Esin, S.; Florio, W.; Brancatisano, F.L.; Bottai, D.; Campa, M.; Batoni, G. Evaluation of the inhibitory effects of human serum components on bactericidal activity of human beta defensin 3. Peptides 2008, 29, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Deslouches, B.; Islam, K.; Craigo, J.K.; Paranjape, S.M.; Montelaro, R.C.; Mietzner, T.A. Activity of the de novo engineered antimicrobial peptide WLBU2 against Pseudomonas aeruginosa in human serum and whole blood: Implications for systemic applications. Antimicrob. Agents Chemother. 2005, 49, 3208–3216. [Google Scholar] [CrossRef] [PubMed]
- Irazazabal, L.N.; Porto, W.F.; Ribeiro, S.M.; Casale, S.; Humblot, V.; Ladram, A.; Franco, O.L. Selective amino acid substitution reduces cytotoxicity of the antimicrobial peptide mastoparan. BBA Biomembr. 2016, 1858, 2699–2708. [Google Scholar] [CrossRef] [PubMed]
- Juba, M.L.; Porter, D.K.; Williams, E.H.; Rodriguez, C.A.; Barksdale, S.M.; Bishop, B.M. Helical cationic antimicrobial peptide length and its impact on membrane disruption. BBA Biomembr. 2015, 1848, 1081–1091. [Google Scholar] [CrossRef]
- Zhu, X.; Shan, A.; Ma, Z.; Xu, W.; Wang, J.; Chou, S.; Cheng, B. Bactericidal efficiency and modes of action of the novel antimicrobial peptide T9W against Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2015, 59, 3008–3017. [Google Scholar] [CrossRef]
- Lyu, Y.; Yang, Y.; Lyu, X.; Dong, N.; Shan, A. Antimicrobial activity, improved cell selectivity and mode of action of short PMAP-36-derived peptides against bacteria and Candida. Sci. Rep. 2016, 6, 27258. [Google Scholar] [CrossRef]
- Wang, J.; Chou, S.; Xu, L.; Zhu, X.; Dong, N.; Shan, A.; Chen, Z. High specific selectivity and membrane-active mechanism of the synthetic centrosymmetric α-helical peptides with Gly-Gly pairs. Sci. Rep. 2015, 5, 15963. [Google Scholar] [CrossRef]
- Srisook, K.; Han, S.-S.; Choi, H.-S.; Li, M.-H.; Ueda, H.; Kim, C.; Cha, Y.-N. CO from enhanced HO activity or from CORM-2 inhibits both O2− and NO production and downregulates HO-1 expression in LPS-stimulated macrophages. Biochem. Pharmacol. 2006, 71, 307–318. [Google Scholar] [CrossRef]
- Zhu, X.; Dong, N.; Wang, Z.; Ma, Z.; Zhang, L.; Ma, Q.; Shan, A. Design of imperfectly amphipathic α-helical antimicrobial peptides with enhanced cell selectivity. Acta Biomater. 2014, 10, 244–257. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, Z.; Feng, F.; Zhu, W.; Guang, H.; Liu, J.; He, W.; Chi, L.; Li, Z.; Yu, H. Molecular cloning and characterization of novel cathelicidin-derived myeloid antimicrobial peptide from Phasianus colchicus. Dev. Comp. Immunol. 2011, 35, 314–322. [Google Scholar] [CrossRef] [PubMed]


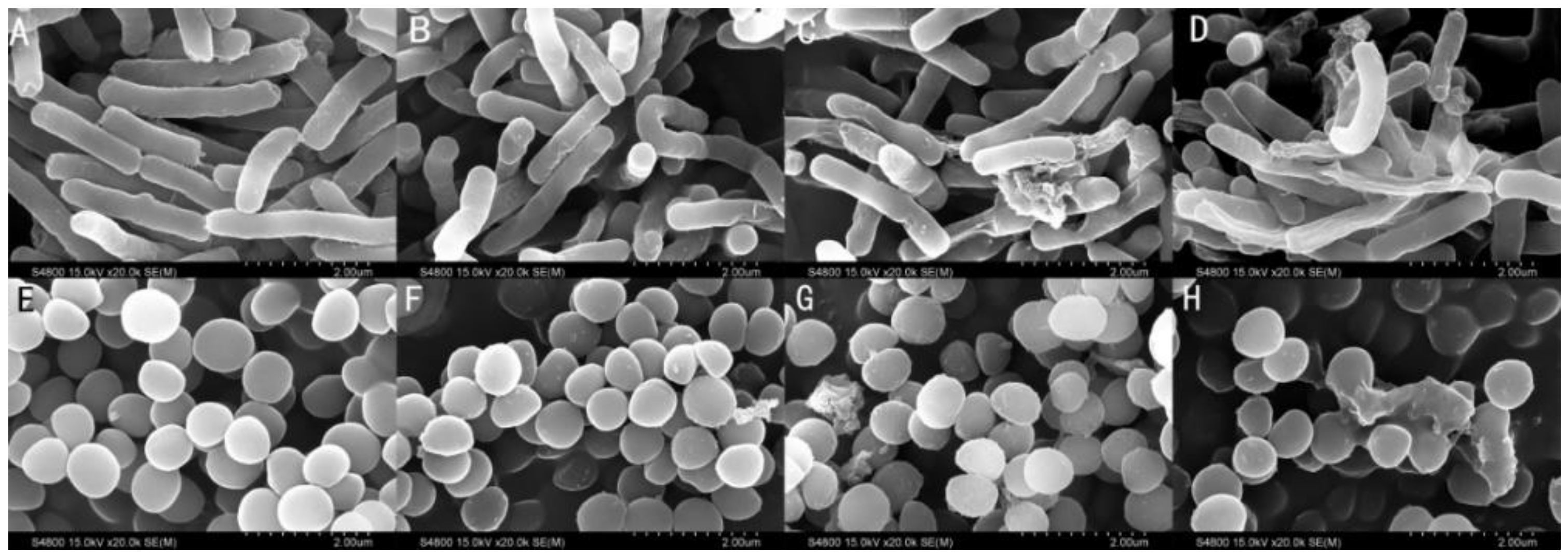
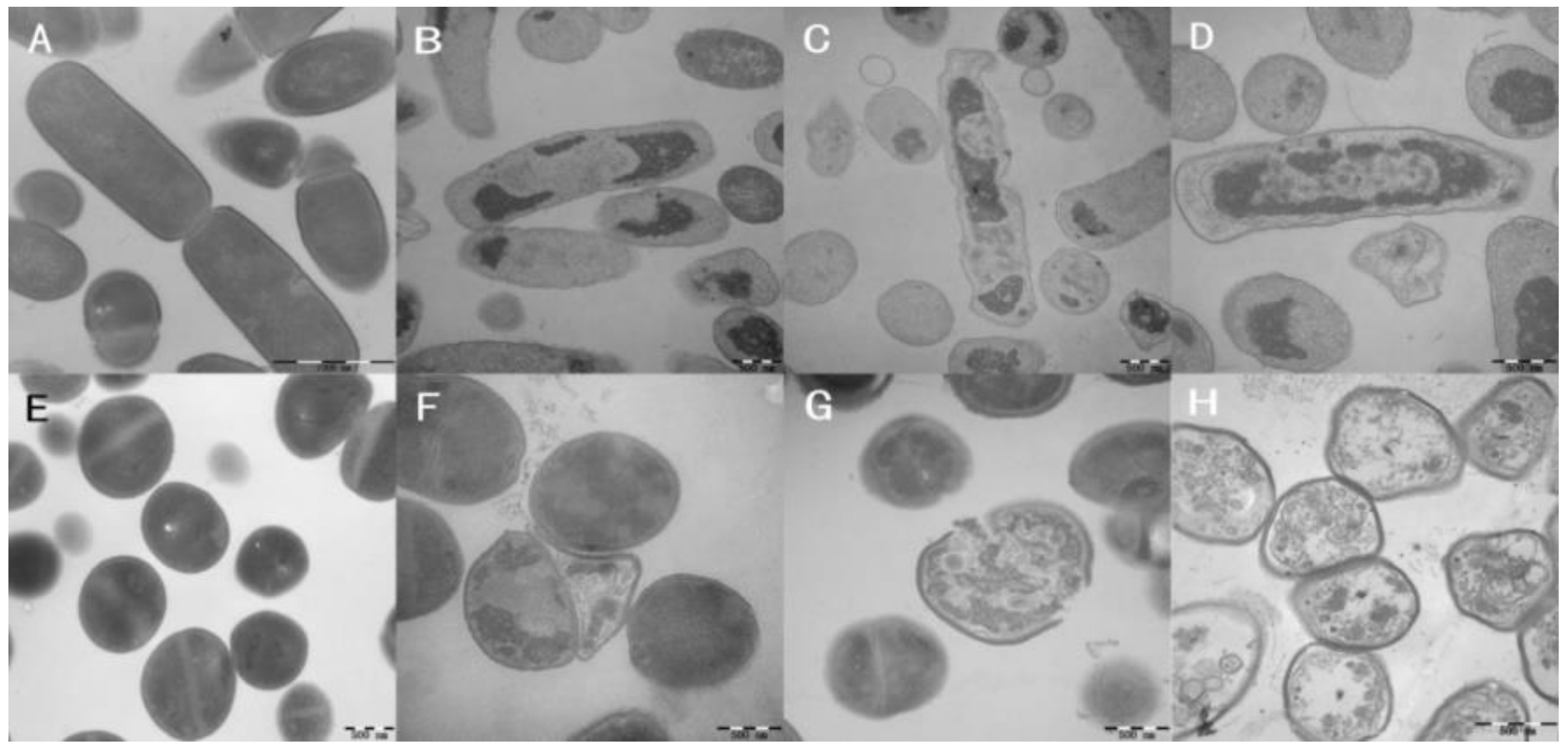
| Peptide | Sequence | Theoretical Mav (u) | Measured Mav (u) 1 | Purity 2 (%) | Charge | GRAVY 3 |
|---|---|---|---|---|---|---|
| CA-FO | KWKLFKKIRFGRFLRKIRRFRPK-NH2 | 3104.88 | 3104.24 | 95.72% | +13 | −1.109 |
| CA-TP | KWKLFKKIWWPFLRR-NH2 | 2131.66 | 2132.61 | 97.42% | +7 | −0.747 |
| CA | KWKLFKKI-NH2 | 1089.42 | 1089.89 | 95.74% | +5 | −0.675 |
| FO | RFGRFLRKIRRFRPK-NH2 | 2032.49 | 2032.59 | 99.25% | +9 | −1.340 |
| TP | WWPFLRR-NH2 | 1059.27 | 1059.79 | 98.33% | +3 | −0.829 |
| MIC (µM) 1 | CA | FO | TP | CA-FO | CA-TP |
|---|---|---|---|---|---|
| Gram-negative bacteria | |||||
| E. coli ATCC25922 | 64 | 32 | 128 | 2 | 2 |
| E. coli UB 1005 | >128 | 16 | 128 | 2 | 4 |
| S. typhimurium C7731 | >128 | 32 | 64 | 4 | 2 |
| S. typhimurium ATCC14028 | >128 | 32 | 32 | 4 | 4 |
| P. aeruginosa ATCC27853 | >128 | 32 | 64 | 4 | 4 |
| S. pullorum C7913 | >128 | 16 | 64 | 8 | 4 |
| Gram-positive bacteria | |||||
| S. aureus ATCC29213 | 128 | 64 | 64 | 2 | 2 |
| S. epidermidis ATCC12228 | >128 | 16 | 32 | 4 | 2 |
| S. faecalis ATCC29212 | >128 | 16 | >128 | 4 | 2 |
| Peptide | GM (μM) 1 | HC10 2 | Therapeutic Index (TI) 3 |
|---|---|---|---|
| CA | 203.19 | 15.54 | 0.08 |
| FO | 25.40 | 2.78 | 0.11 |
| TP | 74.66 | 25.06 | 0.34 |
| CA-FO | 3.43 | 143.34 | 41.79 |
| CA-TP | 2.72 | 49.66 | 18.26 |
| NaCl 1 | KCl 1 | NH4Cl 1 | MgCl2 1 | ZnCl2 1 | CaCl2 1 | FeCl3 1 | Mix 2 | Control 1 | |
|---|---|---|---|---|---|---|---|---|---|
| E. coli ATCC 25922 | |||||||||
| CA-FO | 32 | 128 | 16 | 8 | 16 | >128 | 8 | >128 | 2 |
| CA-TP | 4 | 4 | 1 | 2 | 2 | >128 | 4 | >128 | 2 |
| CA | 64 | >128 | >128 | 128 | >128 | >128 | >128 | >128 | 64 |
| FO | 32 | 32 | 32 | 16 | 64 | >128 | >128 | >128 | 32 |
| TP | 128 | 64 | 128 | 64 | 128 | 64 | 128 | 128 | 64 |
| S. aureus ATCC 29213 | |||||||||
| CA-FO | 4 | 4 | 2 | 2 | 2 | 1 | 2 | 4 | 2 |
| CA-TP | 2 | 2 | 2 | 2 | 2 | 0.25 | 2 | 4 | 2 |
| CA | 128 | 128 | >128 | 64 | 64 | 64 | 64 | 64 | 128 |
| FO | 32 | 32 | 32 | 16 | 64 | 64 | 64 | 64 | 64 |
| TP | 128 | 64 | 128 | 64 | 128 | 64 | 128 | 128 | 64 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Wu, D.; Wang, C.; Shan, A.; Bi, C.; Li, Y.; Gan, W. Hybridization with Insect Cecropin A (1–8) Improve the Stability and Selectivity of Naturally Occurring Peptides. Int. J. Mol. Sci. 2020, 21, 1470. https://doi.org/10.3390/ijms21041470
Yang Y, Wu D, Wang C, Shan A, Bi C, Li Y, Gan W. Hybridization with Insect Cecropin A (1–8) Improve the Stability and Selectivity of Naturally Occurring Peptides. International Journal of Molecular Sciences. 2020; 21(4):1470. https://doi.org/10.3390/ijms21041470
Chicago/Turabian StyleYang, Yang, Di Wu, Chenxi Wang, Anshan Shan, Chongpeng Bi, Yanbing Li, and Wenping Gan. 2020. "Hybridization with Insect Cecropin A (1–8) Improve the Stability and Selectivity of Naturally Occurring Peptides" International Journal of Molecular Sciences 21, no. 4: 1470. https://doi.org/10.3390/ijms21041470
APA StyleYang, Y., Wu, D., Wang, C., Shan, A., Bi, C., Li, Y., & Gan, W. (2020). Hybridization with Insect Cecropin A (1–8) Improve the Stability and Selectivity of Naturally Occurring Peptides. International Journal of Molecular Sciences, 21(4), 1470. https://doi.org/10.3390/ijms21041470




