Anemarrhenae asphodeloides rhizoma Extract Enriched in Mangiferin Protects PC12 Cells against a Neurotoxic Agent-3-Nitropropionic Acid
Abstract
:1. Introduction
2. Results
2.1. Effects of 3-NP on the PC12 Cells Viability
2.2. Effects of XF on PC12 Cells Viability
2.3. Effect of XF on the PC12 Cells under 3-NP-Induced Cytotoxicity
2.4. Effects of Combined XF Pre-Incubation and Co-Incubation XF
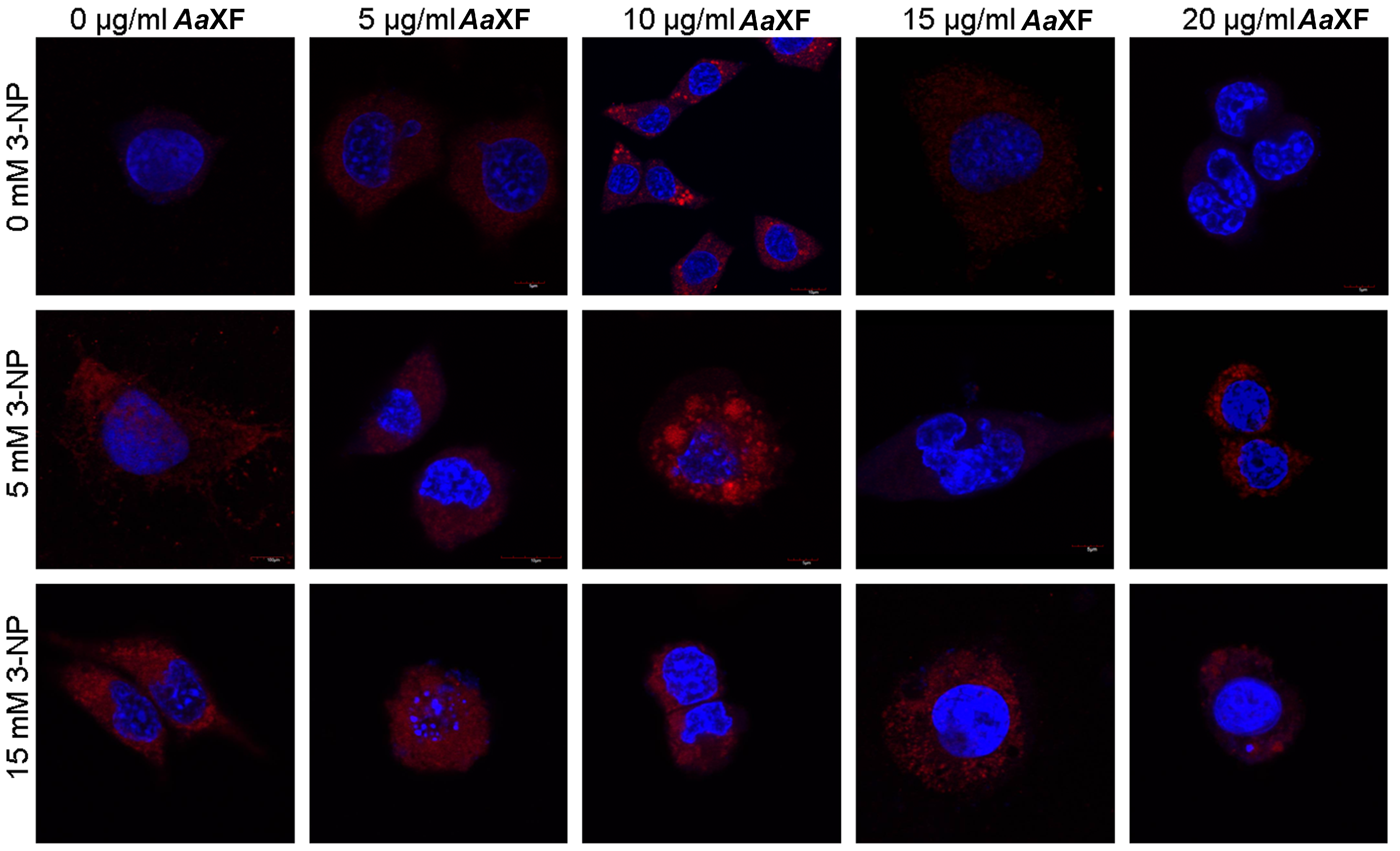
2.5. Confocal Scanning Laser Microscopy for Detecting the Evaluation and Intracellular Distribution of Mitochondria
2.6. Determination of Polyhydroxyxanthone Glycosides
3. Discussion
4. Materials and Methods
4.1. Preparation of Anemarrhena Asphodeloides Extract
4.1.1. Isolation Procedure
4.1.2. Analysis of Extract Composition
4.2. Cell Culture
4.3. Cell Viability Assay
4.4. 3-NP-Induced Cytotoxicity in PC12 Cells
4.5. Evaluation of Protective Activity against 3-NP–Induced Cytotoxicity in PC12 Cells
4.5.1. Experimental Models
4.5.2. Confocal Scanning Laser Microscopy Study
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Peters, R.; Ee, N.; Peters, J.; Booth, A.; Mudway, I.; Anstey, K.J. Air Pollution and Dementia: A Systematic Review. J. Alzheimer’s Dis. 2019, 70, S145–S163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grandjean, P.; Landrigan, P.J. Developmental neurotoxicity of industrial chemicals. Lancet 2006, 368, 2167–2178. [Google Scholar] [CrossRef]
- Heyer, D.B.; Meredith, R.M. Environmental toxicology: Sensitive periods of development and neurodevelopmental disorders. Neurotoxicology 2017, 58, 23–41. [Google Scholar] [CrossRef] [PubMed]
- Cheignon, C.; Tomas, M.; Bonnefont-Rousselot, D.; Faller, P.; Hureau, C.; Collin, F. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Biol. 2018, 14, 450–464. [Google Scholar] [CrossRef] [PubMed]
- Wortmann, M. Dementia: A global health priority—Highlights from an ADI and World Health Organization report. Alzheimer’s Res. Ther. 2012, 4, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho, Y.S.; So, K.F.; Chang, R.C. Drug discovery from Chinese medicine against neurodegeneration in Alzheimer’s and vascular dementia. Chin. Med. 2011, 6, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Z.K.; Yang, H.Q.; Chen, S.D. Traditional Chinese medicine: A promising candidate for the treatment of Alzheimer’s disease. Transl. Neurodegener. 2013, 2, 6. [Google Scholar] [CrossRef] [Green Version]
- Karanian, D.A.; Baude, A.S.; Brown, Q.B.; Parsons, C.G.; Bahr, B.A. 3-Nitropropionic acid toxicity in hippocampus: Protection through N-methyl-D-aspartate receptor antagonism. Hippocampus 2006, 16, 834–842. [Google Scholar] [CrossRef]
- Penel, A.J.; Kosikowski, F.V. Beta-Nitropropionic Acid Production by Aspergillus oryzae in Selected High Protein and Carbohydrate-rich Foods. J. Food Prot. 1990, 53, 321–323. [Google Scholar] [CrossRef]
- Burdock, G.A.; Carabin, I.G.; Soni, M.G. Safety assessment of B-nitropropionic acid: A monograph in support of an acceptable daily intake in humans. Food Chem. 2001, 75, 1–27. [Google Scholar] [CrossRef]
- Szabó, A.; Papp, A.; Nagymajtényi, L. Effects of 3-nitropropionic acid in rats: General toxicity and functional neurotoxicity. Arh. Hig. Rada Toksikol. 2005, 56, 297–302. [Google Scholar] [PubMed]
- Jardim, F.R.; de Rossi, F.T.; Nascimento, M.X.; da Silva Barros, R.G.; Borges, P.A.; Prescilio, I.C.; de Oliveira, M.R. Resveratrol and Brain Mitochondria: A Review. Mol. Neurobiol. 2018, 55, 2085–2101. [Google Scholar] [CrossRef]
- Kulasekaran, G.; Ganapasam, S. Neuroprotective efficacy of naringin on 3-nitropropionic acid-induced mitochondrial dysfunction through the modulation of Nrf2 signaling pathway in PC12 cells. Mol. Cell. Biochem. 2015, 409, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Mandavilli, B.S.; Boldogh, I.; Van Houten, B. 3-Nitropropionic acid-induced hydrogen peroxide, mitochondrial DNA damage, and cell death are attenuated by Bcl-2 overexpression in PC12 cells. Mol. Brain. Res. 2005, 133, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Brownell, A.L.; Chen, Y.I.; Yu, M.; Wang, X.; Dedeoglu, A.; Cicchetti, F.; Jenkins, B.G.; Beal, M.F. 3-Nitropropionic acid-induced neurotoxicity—Assessed by ultra high-resolution positron emission tomography with comparison to magnetic resonance spectroscopy. J. Neurochem. 2004, 89, 1206–1214. [Google Scholar] [CrossRef]
- Al Mutairy, A.; Al Kadasah, S.; Elfaki, I.; Arshaduddin, M.; Malik, D.; Al Moutaery, K.; Tariq, M. Trolox ameliorates 3-nitropropionic acid-induced neurotoxicity in rats. Neurotoxicol. Teratol. 2010, 32, 226–233. [Google Scholar] [CrossRef]
- Miller, P.J.; Zaborszky, L. 3-Nitropropionic acid neurotoxicity: Visualization by silver staining and implications for use as an animal model of Huntington’s disease. Exp. Neurol. 1997, 146, 212–229. [Google Scholar] [CrossRef] [Green Version]
- Uddin, M.S.; Al Mamun, A.; Kabir, M.T.; Jakaria, M.; Mathew, B.; Barreto, G.E.; Ashraf, G.M. Nootropic and Anti-Alzheimer’s Actions of Medicinal Plants: Molecular Insight into Therapeutic Potential to Alleviate Alzheimer’s Neuropathology. Mol. Neurobiol. 2019, 56, 4925–4944. [Google Scholar] [CrossRef]
- Kubis, A.M.; Janusz, M. Alzheimer’s disease: New prospects in therapy and applied experimental models. Postepy Hig. Med. Dosw. 2008, 62, 372–392. [Google Scholar]
- Ong, W.Y.; Farooqui, T.; Koh, H.L.; Farooqui, A.A.; Ling, E.A. Protective effects of ginseng on neurological disorders. Front. Aging Neurosci. 2015, 7, 129. [Google Scholar] [CrossRef] [Green Version]
- Oh, J.K.; Hyun, S.Y.; Oh, H.R.; Jung, J.W.; Park, C.; Lee, S.Y.; Park, J.H.; Kim, S.Y.; Kim, K.H.; Kim, Y.K.; et al. Effects of Anemarrhena asphodeloides on focal ischemic brain injury induced by middle cerebral artery occlusion in rats. Biol. Pharm. Bull. 2007, 30, 38–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Won, J.B.; Ma, C.J. Neuroprotective activities of some medicinal plants against glutamate-induced neurotoxicity in primary cultures of rat cortical cells. Nat. Prod. Sci. 2009, 15, 125–129. [Google Scholar]
- Wang, Y.; Dan, Y.; Yang, D.; Hu, Y.; Zhang, L.; Zhang, C.; Zhu, H.; Cui, Z.; Li, M.; Liu, Y. The genus Anemarrhena Bunge: A review on ethnopharmacology, phytochemistry and pharmacology. J. Ethnopharmacol. 2014, 153, 42–60. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.K.; Chen, T.T. Chinese Medical Herbology and Pharmacology; Art of Medicine Press: City of Industry, CA, USA, 2004; pp. 111–113. [Google Scholar]
- Yan, H.; Li, L.; Tang, X.C. Treating senile dementia with traditional Chinese medicine. Clin. Interv. Aging 2007, 2, 201–208. [Google Scholar] [PubMed]
- Wang, Z.; Cai, J.; Fu, Q.; Cheng, L.; Wu, L.; Zhang, W.; Zhang, Y.; Jin, Y.; Zhang, C. Anti-Inflammatory Activities of Compounds Isolated from the Rhizome of Anemarrhena asphodeloides. Molecules 2018, 23, 2631. [Google Scholar] [CrossRef] [Green Version]
- Ji, K.Y.; Kim, K.M.; Kim, Y.H.; Im, A.R.; Lee, J.Y.; Park, B.; Na, M.; Chae, S. The enhancing immune response and anti-inflammatory effects of Anemarrhena asphodeloides extract in RAW 264.7 cells. Phytomedicine 2019, 59, 152789. [Google Scholar] [CrossRef]
- Ji, D.; Huang, Z.Y.; Fei, C.H.; Xue, W.W.; Lu, T.L. Comprehensive profiling and characterization of chemical constituents of rhizome of Anemarrhena asphodeloides Bge. J. Chromatogr. B 2017, 1060, 355–366. [Google Scholar] [CrossRef]
- Greene, L.; Sobeih, M.; Teng, K.K. Culturing nerve cells: Methodologies for the culture and experimental use of the PC12 rat pheochromocytoma cell line. In Culturing Nerve Cells; Banker, G., Goslin, K., Eds.; MIT Press: Cambridge, MA, USA, 1991; pp. 207–226. [Google Scholar]
- Yakovlev, A.G.; Faden, A.I. Mechanisms of neural cell death: Implications for development of neureprotective treatment strategies. NeuroRx 2004, 1, 5–16. [Google Scholar] [CrossRef]
- Boczek, T.; Radzik, T.; Ferenc, B.; Zylinska, L. The Puzzling Role of Neuron-Specific PMCA Isoforms in the Aging Process. Int. J. Mol. Sci. 2019, 20, 6338. [Google Scholar] [CrossRef] [Green Version]
- Limonta, P.; Moretti, R.M.; Marzagalli, M.; Fontana, F.; Raimondi, M.; Montagnani Marelli, M. Role of Endoplasmic Reticulum Stress in the Anticancer Activity of Natural Compounds. Int. J. Mol. Sci. 2019, 20, 961. [Google Scholar] [CrossRef] [Green Version]
- Jomová, K.; Hudecova, L.; Lauro, P.; Simunkova, M.; Alwasel, S.H.; Alhazza, I.M.; Valko, M. A Switch between Antioxidant and Prooxidant Properties of the Phenolic Compounds Myricetin, Morin, 3′,4′-Dihydroxyflavone, Taxifolin and 4-Hydroxy-Coumarin in the Presence of Copper(II) Ions: A Spectroscopic, Absorption Titration and DNA Damage Study. Molecules 2019, 24, 4335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, F.Y.; Song, X.Y.; Chen, J.J.; Yao, G.D.; Song, S.J. Timosaponin AIII: A novel potential anti-tumor compound from Anemarrhena asphodeloides. Steroids 2018, 140, 125–130. [Google Scholar] [CrossRef] [PubMed]
- MarElia, C.B.; Sharp, A.E.; Shemwell, T.A.; Clare Zhang, Y.; Burkhardt, B.R. Anemarrhena asphodeloides Bunge and its constituent timosaponin-AIII induce cell cycle arrest and apoptosis in pancreatic cancer cells. FEBS Open Bio 2018, 8, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.D.; Wu, C.L.; Hwang, W.C.; Yang, D.I. More Insight into BDNF against Neurodegeneration: Anti-Apoptosis, Anti-Oxidation, and Suppression of Autophagy. Int. J. Mol. Sci. 2017, 18, 545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade, S.; Ramalho, M.J.; Loureiro, J.A.; Pereira, M.C. Natural Compounds for Alzheimer’s Disease Therapy: A Systematic Review of Preclinical and Clinical Studies. Int. J. Mol. Sci. 2019, 20, 2313. [Google Scholar] [CrossRef] [Green Version]
- Ma, H.; Johnson, S.L.; Liu, W.; DaSilva, N.A.; Meschwitz, S.; Dain, J.A.; Seeram, N.P. Evaluation of Polyphenol Anthocyanin-Enriched Extracts of Blackberry, Black Raspberry, Blueberry, Cranberry, Red Raspberry, and Strawberry for Free Radical Scavenging, Reactive Carbonyl Species Trapping, Anti-Glycation, Anti-β-Amyloid Aggregation, and Microglial Neuroprotective Effects. Int. J. Mol. Sci. 2018, 19, 461. [Google Scholar] [CrossRef] [Green Version]
- Habtemariam, S. Molecular Pharmacology of Rosmarinic and Salvianolic Acids: Potential Seeds for Alzheimer’s and Vascular Dementia Drugs. Int. J. Mol. Sci. 2018, 19, 458. [Google Scholar] [CrossRef] [Green Version]
- Márquez-Valadez, B.; Lugo-Huitrón, R.; Valdivia-Cerda, V.; Miranda-Ramírez, L.R.; Pérez-De La Cruz, V.; González-Cuahutencos, O.; Rivero-Cruz, I.; Mata, R.; Santamaría, A.; Pedraza-Chaverrí, J. The natural xanthone α-mangostin reduces oxidative damage in rat brain tissue. Nutr. Neurosci. 2009, 12, 35–42. [Google Scholar] [CrossRef]
- Pedraza-Chaverrí, J.; Reyes-Fermín, L.M.; Nolasco-Amaya, E.G.; Orozco-Ibarra, M.; Medina-Campos, O.N.; González-Cuahutencos, O.; Rivero-Cruz, I.; Mata, R. ROS scavenging capacity and neuroprotective effect of alpha-mangostin against 3-nitropropionic acid in cerebellar granule neurons. Exp. Toxicol. Pathol. 2009, 61, 491–501. [Google Scholar] [CrossRef]
- Peng, S.; Hou, Y.; Yao, J.; Fang, J. Neuroprotection of mangiferin against oxidative damage via arousing Nrf2 signaling pathway in PC12 cells. Biofactors 2019, 45, 381–392. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, L.; Dang, X.; Liu, J.; Ito, Y.; Sun, W. Neuroprotective effects of total steroid saponins on cerebral ischemia injuries in an animal model of focal ischemia/reperfusion. Planta Med. 2014, 80, 637–644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.Y.; Guo, Z.; Liu, Y.M.; Liu, X.M.; Chang, Q.; Liao, Y.H.; Pan, R.L. Neuroprotective effects of Total Saikosaponins of Bupleurum yinchowense on corticosterone-induced apoptosis in PC12 cells. J. Ethnopharmacol. 2013, 148, 794–803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsukamoto, S.; Wakana, T.; Koimaru, K.; Yoshida, T.; Sato, M.; Ohta, T. 7-hydroxy-3-(4-hydroxybenzyl)chroman and broussonin b: Neurotrophic compounds, isolated from Anemarrhena asphodeloides BUNGE, function as proteasome inhibitors. Biol. Pharm. Bull. 2005, 28, 1798–1800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xi, J.S.; Wang, Y.F.; Long, X.X.; Ma, Y. Mangiferin Potentiates Neuroprotection by Isoflurane in Neonatal Hypoxic Brain Injury by Reducing Oxidative Stress and Activation of Phosphatidylinositol-3-Kinase/Akt/Mammalian Target of Rapamycin (PI3K/Akt/mTOR) Signaling. Med. Sci. Monit. 2018, 24, 7459–7468. [Google Scholar] [CrossRef]
- Zhou, B.O.; Li, H. Mangiferin Improves Memory Deficits followed Cerebral Ischemia Reperfusion in Mice through Attenuation of Oxidative Stress and AChE Activity. Lat. Am. J. Pharm. 2015, 34, 1229–1238. [Google Scholar]
- Zajac, D.; Stasinska, A.; Delgado, R.; Pokorski, M. Mangiferin and its traversal into the brain. Adv. Exp. Med. Biol. 2013, 756, 105–111. [Google Scholar] [CrossRef]
- Schiera, G.; Di Liegro, C.M.; Di Liegro, I. Cell-to-Cell Communication in Learning and Memory: From Neuro- and Glio-Transmission to Information Exchange Mediated by Extracellular Vesicles. Int. J. Mol. Sci. 2020, 21, 266. [Google Scholar] [CrossRef] [Green Version]
- Peng, Y.; Zhao, L.; Lin, D.; Liu, Y.; Zhang, M.; Song, S. Determination of the chemical constituents of the different processed products of Anemarrhena asphodeloides Rhizomes by high-performance liquid chromatography quadrupole time-of-flight mass spectrometry. Biomed. Chromatogr. 2016, 30, 508–519. [Google Scholar] [CrossRef]
- Nowak, B.; Matuszewska, A.; Szandruk, M.; Matkowski, A.; Woźniak, D.; Zduniak, K.; Rzeszutko, M.; Landwójtowicz, M.; Jędrzejuk, D.; Piasecki, T.; et al. Effect of long-term administration of mangiferin from Belamcanda chinensis on bone metabolism in ovariectomized rats. J. Funct. Food 2018, 46, 12–18. [Google Scholar] [CrossRef]
- Kulbacka, J.; Kulbacki, M.; Segen, J.; Choromańska, A.; Saczko, J.; Dubińska-Magiera, M.; Kotulska, M. Automated Analysis of Images from Confocal Laser Scanning Microscopy Applied to Observation of Calcium. In Intelligent Information and Database Systems; ACIIDS Lecture Notes in Computer, Science; Nguyen, N., Trawiński, B., Kosala, R., Eds.; Springer: Cham, Germany, 2015; pp. 297–306. [Google Scholar] [CrossRef]
- Novickij, V.; Rembialkowska, N.; Staigvila, G.; Kulbacka, J. Effects of extracellular medium conductivity on cell response in the context of sub-microsecond range calcium electroporation. Sci. Rep. 2020, 10, 3718. [Google Scholar] [CrossRef] [Green Version]
- Colle, D.; Santos, D.B.; Hartwig, J.M.; Godoi, M.; Engel, D.F.; de Bem, A.F.; Braga, A.L.; Farina, M. Succinobucol, a Lipid-Lowering Drug, Protects Against 3-Nitropropionic Acid-Induced Mitochondrial Dysfunction and Oxidative Stress in SH-SY5Y Cells via Upregulation of Glutathione Levels and Glutamate Cysteine Ligase Activity. Mol. Neurobiol. 2016, 53, 1280–1295. [Google Scholar] [CrossRef] [PubMed]






© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piwowar, A.; Rembiałkowska, N.; Rorbach-Dolata, A.; Garbiec, A.; Ślusarczyk, S.; Dobosz, A.; Długosz, A.; Marchewka, Z.; Matkowski, A.; Saczko, J. Anemarrhenae asphodeloides rhizoma Extract Enriched in Mangiferin Protects PC12 Cells against a Neurotoxic Agent-3-Nitropropionic Acid. Int. J. Mol. Sci. 2020, 21, 2510. https://doi.org/10.3390/ijms21072510
Piwowar A, Rembiałkowska N, Rorbach-Dolata A, Garbiec A, Ślusarczyk S, Dobosz A, Długosz A, Marchewka Z, Matkowski A, Saczko J. Anemarrhenae asphodeloides rhizoma Extract Enriched in Mangiferin Protects PC12 Cells against a Neurotoxic Agent-3-Nitropropionic Acid. International Journal of Molecular Sciences. 2020; 21(7):2510. https://doi.org/10.3390/ijms21072510
Chicago/Turabian StylePiwowar, Agnieszka, Nina Rembiałkowska, Anna Rorbach-Dolata, Arnold Garbiec, Sylwester Ślusarczyk, Agnieszka Dobosz, Anna Długosz, Zofia Marchewka, Adam Matkowski, and Jolanta Saczko. 2020. "Anemarrhenae asphodeloides rhizoma Extract Enriched in Mangiferin Protects PC12 Cells against a Neurotoxic Agent-3-Nitropropionic Acid" International Journal of Molecular Sciences 21, no. 7: 2510. https://doi.org/10.3390/ijms21072510






