Human DENND1A.V2 Drives Cyp17a1 Expression and Androgen Production in Mouse Ovaries and Adrenals
Abstract
:1. Introduction
2. Results
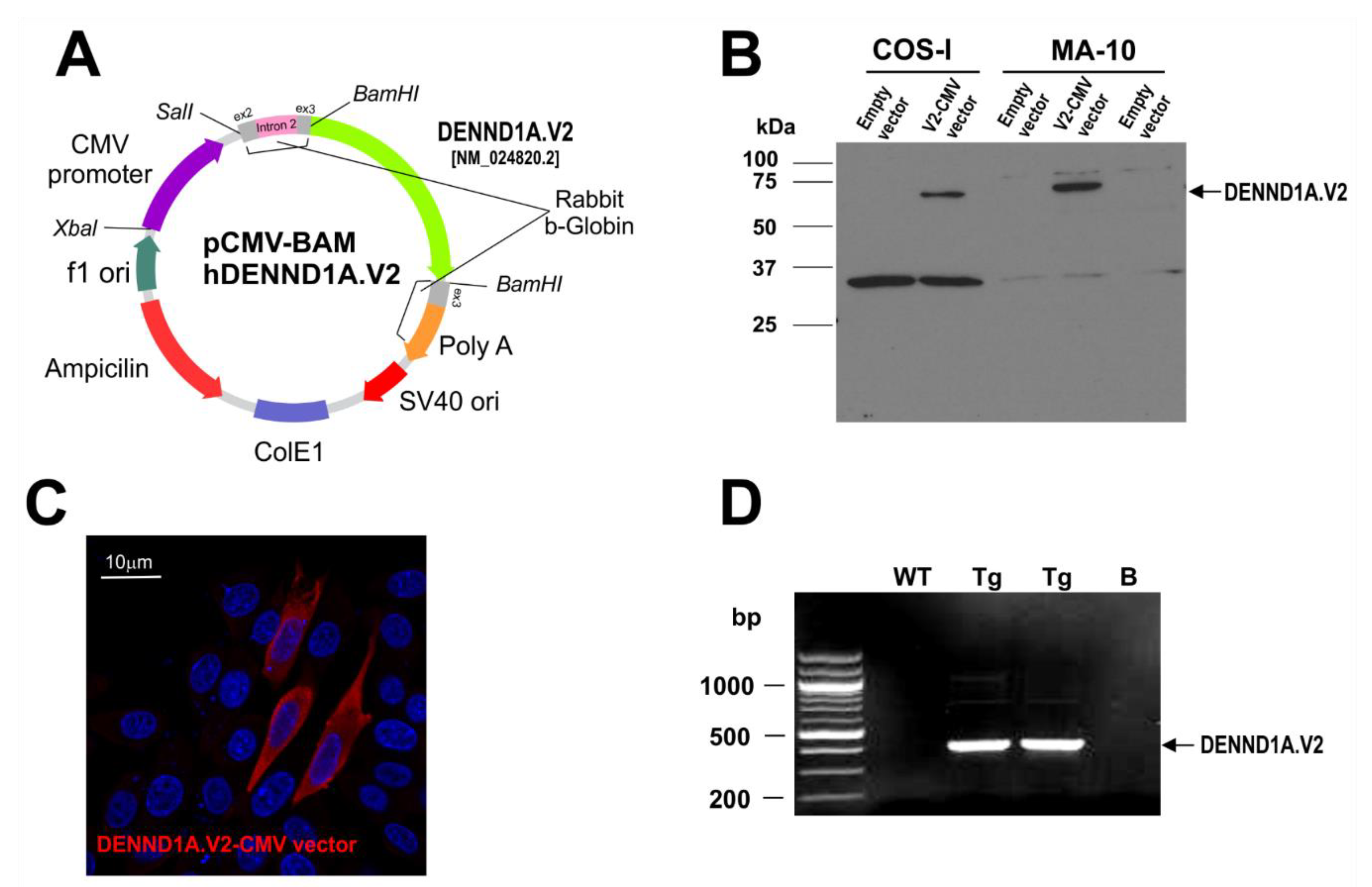
2.1. hDENND1A.V2 Induces a PCOS Phenotype in Mouse Leydig MA-10 Cells—Evidence to Support a hDENND1A.V2 Mouse Model for PCOS
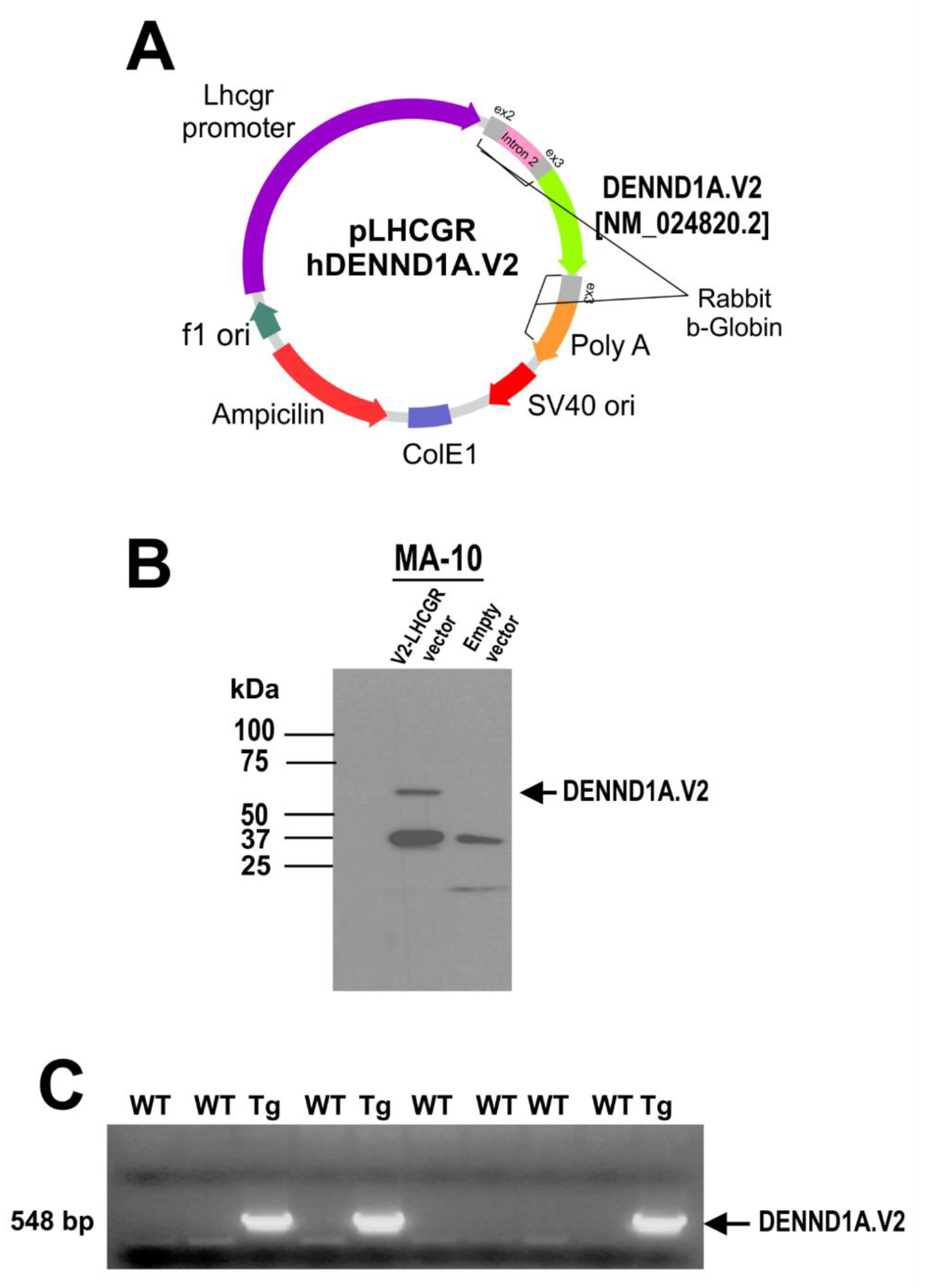
2.2. Generation of hDENND1A.V2 Transgenic Mice
3. Discussion
4. Materials and Methods
4.1. Adenoviral Expression of hDENND1A in Mouse MA-10 Leydig Cells
4.2. Animals
4.3. Generation of CMV-hDENND1A.V2 Transgenic Mice
4.4. Generation of Lhcgr-hDENND1A.V2 Transgenic Mice
4.5. Generation of TetOn-hDENND1A.V2 Transgenic Mice
4.6. RT-PCR
4.7. Quantitative Real-Time qRT-PCR Analyses of DENND1A.V2 and Cyp17a1
4.8. Western Blot and Immunodetection
4.9. Cell Transfection
4.10. Immunofluorescence Detection of hDENND1A.V2
4.11. Immunohistochemical Detection of hDENND1A.V2
4.12. Theca-Interstitial Cell (TIC) and Granulosa Cell (GC) Isolation
4.13. Quantitation of Progesterone (P4), 17α-hydroxyprogesterone (17OHP4), and Androstenedione (adione) by ELISA
4.14. Statistical Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PCOS | polycystic ovary syndrome |
| GWAS | Genome Wide Association Study |
| DENND1A | DENN (Differentially Expressed in Neoplastic vs. Normal cells) Domain Containing 1A |
| hDENND1A.V2 | hDENND1A variant 2 (a truncated splice variant of the DENND1A gene) |
| Lhcgr | Luteinizing Hormone/Choriogonadotropin Receptor |
| Cyp17 | mouse cytochrome P450 17-hydroxylase |
| P4 | progesterone |
| 17OHP4 | 17-hydroxyprogesterone |
References
- Cadagan, D.; Khan, R.; Amer, S. Thecal cell sensitivity to luteinizing hormone and insulin in polycystic ovarian syndrome. Reprod. Biol. 2016, 16, 53–60. [Google Scholar] [CrossRef] [PubMed]
- McAllister, J.M.; Legro, R.S.; Modi, B.P.; Strauss, J.F., 3rd. Functional genomics of PCOS: From GWAS to molecular mechanisms. Trends Endocrinol. Metab. 2015, 26, 118–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Z.-J.; Zhao, H.; He, L.; Shi, Y.; Qin, Y.; Shi, Y.; Li, Z.; You, L.; Zhao, J.; Liu, J.; et al. Genome-wide association study identifies susceptibility loci for polycystic ovary syndrome on chromosome 2p16.3, 2p21 and 9q33.3. Nat. Genet. 2011, 43, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.G.; Urbanek, M.; Ehrmann, D.A.; Armstrong, L.L.; Lee, J.Y.; Sisk, R.; Karaderi, T.; Barber, T.M.; McCarthy, M.I.; Franks, S.; et al. Genome-wide association of polycystic ovary syndrome implicates alterations in gonadotropin secretion in European ancestry populations. Nat Commun. 2015, 6, 7502–7515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Day, F.; Karaderi, T.; Jones, M.R.; Meun, C.; He, C.; Drong, A.; Kraft, P.; Lin, N.; Huang, H.; Broer, L.; et al. Large-scale genome-wide meta-analysis of polycystic ovary syndrome suggests shared genetic architecture for different diagnosis criteria. PLoS Genet. 2018, 14, e1007813. [Google Scholar] [CrossRef] [Green Version]
- Marat, A.L.; McPherson, P.S. The connecdenn family, Rab35 guanine nucleotide exchange factors interfacing with the clathrin machinery. J. Biol. Chem. 2010, 285, 10627–10637. [Google Scholar] [CrossRef] [Green Version]
- Kaksonen, M.; Roux, A. Mechanisms of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell Biol. 2018, 19, 313–326. [Google Scholar] [CrossRef]
- McAllister, J.M.; Modi, B.; Miller, B.A.; Biegler, J.; Bruggeman, R.; Legro, R.S.; Strauss, J.F. Overexpression of a DENND1A isoform produces a polycystic ovary syndrome theca phenotype. Proc. Natl. Acad. Sci. USA 2014, 111, E1519–E1527. [Google Scholar] [CrossRef] [Green Version]
- Kulkarni, R.; Teves, M.E.; Han, A.X.; McAllister, J.M.; Strauss, J.F., 3rd. Colocalization of Polycystic Ovary Syndrome Candidate Gene Products in Theca Cells Suggests Novel Signaling Pathways. J. Endocr. Soc. 2019, 3, 2204–2223. [Google Scholar] [CrossRef] [Green Version]
- Elia, E.; Sander, V.; Luchetti, C.G.; Solano, M.E.; Di Girolamo, G.; Gonzalez, C.; Motta, A.B. The mechanisms involved in the action of metformin in regulating ovarian function in hyperandrogenized mice. Molecular Human Reproduction. MHR Basic Sci. Reprod. Med. 2006, 12, 475–481. [Google Scholar] [CrossRef] [Green Version]
- Heber, M.; Velez, L.; Herreira, S.; Amalfi, S.; Motta, A. The role of prenatal hyperandrogenism on lipid metabolism during adult life in a rat model. Medicina 2012, 72, 389–392. [Google Scholar] [PubMed]
- Osuka, S.; Nakanishi, N.; Murase, T.; Nakamura, T.; Goto, M.; Iwase, A.; Kikkawa, F. Animal models of polycystic ovary syndrome: A review of hormone-induced rodent models focused on hypothalamus-pituitary-ovary axis and neuropeptides. Reprod. Med. Biol. 2018, 18, 151–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbott, D.H.; Dumesic, D.A.; Eisner, J.R.; Colman, R.J.; Kemnitz, J.W. Insights into the Development of Polycystic Ovary Syndrome (PCOS) from Studies of Prenatally Androgenized Female Rhesus Monkeys. Trends Endocrinol. Metab. 1998, 9, 62–67. [Google Scholar] [CrossRef]
- Padmanabhan, V.; Veiga-Lopez, A. Sheep models of polycystic ovary syndrome phenotype. Mol. Cell. Endocrinol. 2013, 373, 8–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tee, M.K.; Speek, M.; Legeza, B.; Modi, B.; Teves, M.E.; McAllister, J.M.; Strauss, J.F., III; Miller, W.L. Alternative splicing of DENND1A, a PCOS candidate gene, generates variant 2. Mol. Cell. Endocrinol. 2016, 434, 25–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ascoli, M. Regulation of gonadotropin receptors and gonadotropin responses in a clonal strain of Leydig tumor cells by epidermal growth factor. J. Biol. Chem. 1981, 256, 179–183. [Google Scholar]
- Ascoli, M. Characterization of Several Clonal Lines of Cultured Ley dig Tumor Cells: Gonadotropin Receptors and Steroidogenic Responses. Endocrinology 1981, 108, 88–95. [Google Scholar] [CrossRef]
- Dumontet, T.; Sahut-Barnola, I.; Septier, A.; Montanier, N.; Plotton, I.; Roucher-Boulez, F.; Ducros, V.; Lefrançois-Martinez, A.M.; Pointud, J.C.; Zubair, M.; et al. Adrenocortical development: Lessons from mouse models. Ann. d’Endocrinologie 2018, 79, 95–97. [Google Scholar] [CrossRef]
- Shi, Y.; Zhao, H.; Shi, Y.; Cao, Y.; Yang, D.; Li, Z.; Zhang, B.; Liang, X.; Li, T.; Chen, J.; et al. Genome-wide association study identifies eight new risk loci for polycystic ovary syndrome. Nat. Genet. 2012, 44, 1020–1025. [Google Scholar] [CrossRef]
- Liu, H.; Zhao, H.; Chen, Z.-J. Genome-Wide Association Studies for Polycystic Ovary Syndrome. Semin. Reprod. Med. 2016, 34, 224–229. [Google Scholar] [CrossRef]
- Chen, L.; Hu, L.-M.; Wang, Y.-F.; Yang, H.-Y.; Huang, X.-Y.; Zhou, W.; Sun, H.X. Genome-wide association study for SNPs associated with PCOS in human patients. Exp. Ther. Med. 2017, 14, 4896–4900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, J.; Gao, Q.; Cao, Y.; Fu, J. Dennd1a, a susceptibility gene for polycystic ovary syndrome, is essential for mouse embryogenesis. Dev. Dyn. 2019, 248, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Nelson-Degrave, V.L.; Wickenheisser, J.K.; Hendricks, K.L.; Asano, T.; Fujishiro, M.; Legro, R.S.; Kimball, S.R.; Strauss, J.F., III; McAllister, J.M. Alterations in Mitogen-Activated Protein Kinase Kinase and Extracellular Regulated Kinase Signaling in Theca Cells Contribute to Excessive Androgen Production in Polycystic Ovary Syndrome. Mol. Endocrinol. 2005, 19, 379–390. [Google Scholar] [CrossRef] [Green Version]
- Tian, Y.; Shen, W.; Lai, Z.; Shi, L.; Yang, S.; Ding, T.; Wang, S.; Luo, A. Isolation and identification of ovarian theca-interstitial cells and granulose cells of immature female mice. Cell Biol. Int. 2015, 39, 584–590. [Google Scholar] [CrossRef] [PubMed]
- DiRienzo, J.; Balzarini, M.; Gonzalez, L.; Tablada, M.; Robledo, C. InfoStat Versión 2018. Centro de Transferencia InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. 2018. Available online: http://www.infostat.com.ar (accessed on 27 January 2019).








© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teves, M.E.; Modi, B.P.; Kulkarni, R.; Han, A.X.; Marks, J.S.; Subler, M.A.; Windle, J.; Newall, J.M.; McAllister, J.M.; Strauss, J.F., III. Human DENND1A.V2 Drives Cyp17a1 Expression and Androgen Production in Mouse Ovaries and Adrenals. Int. J. Mol. Sci. 2020, 21, 2545. https://doi.org/10.3390/ijms21072545
Teves ME, Modi BP, Kulkarni R, Han AX, Marks JS, Subler MA, Windle J, Newall JM, McAllister JM, Strauss JF III. Human DENND1A.V2 Drives Cyp17a1 Expression and Androgen Production in Mouse Ovaries and Adrenals. International Journal of Molecular Sciences. 2020; 21(7):2545. https://doi.org/10.3390/ijms21072545
Chicago/Turabian StyleTeves, Maria E., Bhavi P. Modi, Rewa Kulkarni, Angela X. Han, Jamaia S. Marks, Mark A. Subler, Jolene Windle, Jordan M. Newall, Jan M. McAllister, and Jerome F. Strauss, III. 2020. "Human DENND1A.V2 Drives Cyp17a1 Expression and Androgen Production in Mouse Ovaries and Adrenals" International Journal of Molecular Sciences 21, no. 7: 2545. https://doi.org/10.3390/ijms21072545





