Low-Molecular-Weight Heparin Reduces Ventilation-Induced Lung Injury through Hypoxia Inducible Factor-1α in a Murine Endotoxemia Model
Abstract
:1. Introduction
2. Results
2.1. The Effects of Endotoxin Stimulation on VILI Are Partially Inversed by Enoxaparin
2.2. The Effects of Endotoxin-Stimulated MV-Induced Neutrophil Sequestration, Oxygen Radicals and HIF-1α mRNA Activation Are Partially Inhibited by Enoxaparin
2.3. The Effects of Endotoxin-Stimulated MV-Induced HIF-1α Expression Are Partially Reduced by Enoxaparin
2.4. The Effects of Endotoxin-Stimulated MV-Induced Lung Injuries, Hypoxemia and Impaired Respiratory Function Are Partially Inhibited in HIF-1α Deficient Mice
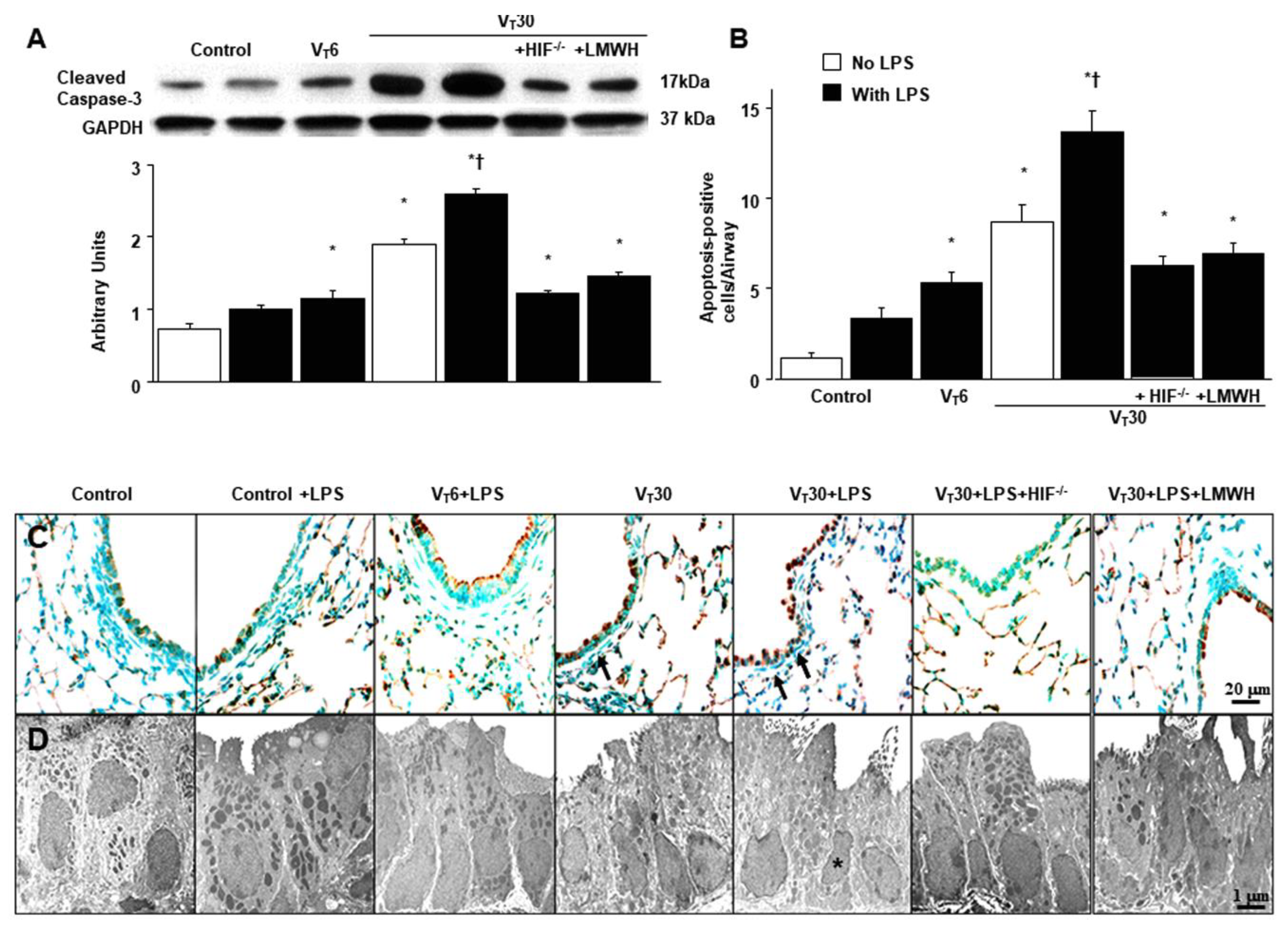
2.5. The Effects of Endotoxin-Stimulated MV-Induced Expression of Caspase-3 and Epithelial Apoptosis Are Partially Suppressed in HIF-1α Deficient Mice and Enoxaparin
3. Discussion
4. Materials and Methods
4.1. Ethics of Experimental Animals
4.2. Experimental Groups
4.3. Ventilator Protocol
4.4. Lipopolysaccharide Administration
4.5. Enoxaparin Administration
4.6. Whole Body Plethysmography
4.7. Measurement of Inflammatory Cytokines
4.8. Real Time Polymerase Chain Reaction
4.9. Immunoblot Analysis
4.10. Immunohistochemistry
4.11. Histopathologic Grading of VILI
4.12. Analysis of Data
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ALI | acute lung injury |
| BAL | bronchoalveolar lavage |
| FiO2 | fraction of inspired oxygen |
| HIF | hypoxia-inducible factor |
| IL | interleukin |
| LMWH | low-molecular-weight heparin |
| LPS | lipopolysaccharide |
| MIP-2 | macrophage inflammatory protein-2 |
| MPO | myeloperoxidase |
| MV | mechanical ventilation |
| ROS | reactive oxygen species |
| SOD | sodium dismutase |
| TEM | transmission electron microscopy |
| TNF-α | tumor necrosis factor-α |
| TUNEL | terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end-labeling assay |
| VEGF | vascular endothelial growth factor |
| VILI | ventilator-induced lung injury |
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Genga, K.R.; Russell, J.A. Update of Sepsis in the Intensive Care Unit. J. Innate. Immun. 2017, 9, 441–455. [Google Scholar] [CrossRef]
- Hattori, Y.; Hattori, K.; Suzuki, T.; Matsuda, N. Recent advances in the pathophysiology and molecular basis of sepsis-associated organ dysfunction: Novel therapeutic implications and challenges. Pharmacol. Ther. 2017, 177, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Lee, C.H.; Dedaj, R.; Zhao, H.; Mrabat, H.; Sheidlin, A.; Syrkina, O.; Huang, P.M.; Garg, H.G.; Hales, C.A.; et al. High-molecular-weight hyaluronan—A possible new treatment for sepsis-induced lung injury: A preclinical study in mechanically ventilated rats. Crit. Care 2008, 12, R102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cornet, A.D.; Smit, E.G.; Beishuizen, A.; Groeneveld, A.B. The role of heparin and allied compounds in the treatment of sepsis. Thromb. Haemost. 2007, 98, 579–586. [Google Scholar] [CrossRef] [Green Version]
- Zarychanski, R.; Abou-Setta, A.M.; Kanji, S.; Turgeon, A.F.; Kumar, A.; Houston, D.S.; Rimmer, E.; Houston, B.L.; McIntyre, L.; Fox-Robichaud, A.E.; et al. The efficacy and safety of heparin in patients with sepsis: A systematic review and metaanalysis. Crit. Care Med. 2015, 43, 511–518. [Google Scholar] [CrossRef]
- Fan, Y.; Jiang, M.; Gong, D.; Zou, C. Efficacy and safety of low-molecular-weight heparin in patients with sepsis: A meta-analysis of randomized controlled trials. Sci. Rep. 2016, 6, 25984. [Google Scholar] [CrossRef] [Green Version]
- Li, L.F.; Huang, C.C.; Lin, H.C.; Tsai, Y.H.; Quinn, D.A.; Liao, S.K. Unfractionated heparin and enoxaparin reduce high-stretch ventilation augmented lung injury: A prospective, controlled animal experiment. Crit. Care 2009, 13, R108. [Google Scholar] [CrossRef] [Green Version]
- Bogdanovski, D.A.; DiFazio, L.T.; Bogdanovski, A.K.; Csóka, B.; Jordan, G.B.; Paul, E.R.; Antonioli, L.; Pilip, S.A.; Nemeth, Z.H. Hypoxia-inducible-factor-1 in trauma and critical care. J. Crit. Care 2017, 42, 207–212. [Google Scholar] [CrossRef]
- Epstein, A.C.; Gleadle, J.M.; McNeill, L.A.; Hewitson, K.S.; O’Rourke, J.; Mole, D.R.; Mukherji, M.; Metzen, E.; Wilson, M.I.; Dhanda, A.; et al. elegans EGL-9 and mammalian homologs define a family of dioxygenases that regulate HIF by prolyl hydroxylation. Cell 2001, 107, 43–54. [Google Scholar] [CrossRef] [Green Version]
- Peyssonnaux, C.; Cejudo-Martin, P.; Doedens, A.; Zinkernagel, A.S.; Johnson, R.S.; Nizet, V. Cutting edge: Essential role of hypoxia inducible factor-1alpha in development of lipopolysaccharide-induced sepsis. J. Immunol. 2007, 178, 7516–7519. [Google Scholar] [CrossRef]
- Jung, Y.; Isaacs, J.S.; Lee, S.; Trepel, J.; Liu, Z.G.; Neckers, L. Hypoxia-inducible factor induction by tumour necrosis factor in normoxic cells requires receptor-interacting protein-dependent nuclear factor kappa B activation. Biochem. J. 2003, 370, 1011–1017. [Google Scholar] [CrossRef]
- Eckle, T.; Brodsky, K.; Bonney, M.; Packard, T.; Han, J.; Borchers, C.H.; Mariani, T.J.; Kominsky, D.J.; Mittelbronn, M.; Eltzschig, H.K. HIF1A reduces acute lung injury by optimizing carbohydrate metabolism in the alveolar epithelium. PLoS Biol. 2013, 11, e1001665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirota, K. Involvement of hypoxia-inducible factors in the dysregulation of oxygen homeostasis in sepsis. Cardiovasc. Hematol. Disord. Drug Targets 2015, 15, 29–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeh, C.H.; Cho, W.; So, E.C.; Chu, C.C.; Lin, M.C.; Wang, J.J.; Hsing, C.H. Propofol inhibits lipopolysaccharide-induced lung epithelial cell injury by reducing hypoxia-inducible factor-1alpha expression. Br. J. Anaesth. 2011, 106, 590–599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Görlach, A.; Bonello, S. The cross-talk between NF-kappaB and HIF-1: Further evidence for a significant liaison. Biochem. J. 2008, 412, e17–e19. [Google Scholar] [CrossRef]
- Li, L.F.; Huang, C.C.; Liu, Y.Y.; Lin, H.C.; Kao, K.C.; Yang, C.T.; Liao, S.K. Hydroxyethyl starch reduces high stretch ventilation-augmented lung injury via vascular endothelial growth factor. Transl. Res. 2011, 157, 293–305. [Google Scholar] [CrossRef]
- Camprubí-Rimblas, M.; Tantinyà, N.; Bringué, J.; Guillamat-Prats, R.; Artigas, A. Anticoagulant therapy in acute respiratory distress syndrome. Ann. Transl. Med. 2018, 6, 36. [Google Scholar] [CrossRef]
- Tuinman, P.R.; Dixon, B.; Levi, M.; Juffermans, N.P.; Schultz, M.J. Nebulized anticoagulants for acute lung injury—A systematic review of preclinical and clinical investigations. Crit. Care 2012, 16, R70. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.Y.; Chen, N.H.; Chang, C.H.; Lin, S.W.; Kao, K.C.; Hu, H.C.; Chang, G.J.; Li, L.F. Ethyl pyruvate attenuates ventilation-induced diaphragm dysfunction through high-mobility group box-1 in a murine endotoxaemia model. J. Cell. Mol. Med. 2019, 23, 5679–5691. [Google Scholar] [CrossRef] [Green Version]
- Altemeier, W.A.; Matute-Bello, G.; Frevert, C.W.; Kawata, Y.; Kajikawa, O.; Martin, T.R.; Glenny, R.W. Mechanical ventilation with moderate tidal volumes synergistically increases lung cytokine response to systemic endotoxin. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L533–L542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altemeier, W.A.; Matute-Bello, G.; Gharib, S.A.; Glenny, R.W.; Martin, T.R.; Liles, W.C. Modulation of lipopolysaccharide-induced gene transcription and promotion of lung injury by mechanical ventilation. J. Immunol. 2005, 175, 3369–3376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bregeon, F.; Delpierre, S.; Chetaille, B.; Kajikawa, O.; Martin, T.R.; Autillo-Touati, A.; Jammes, Y.; Pugin, J. Mechanical ventilation affects lung function and cytokine production in an experimental model of endotoxemia. Anesthesiology 2005, 102, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Bruells, C.S.; Maes, K.; Rossaint, R.; Thomas, D.; Cielen, N.; Bleilevens, C.; Bergs, I.; Loetscher, U.; Dreier, A.; Gayan-Ramirez, G.; et al. Prolonged mechanical ventilation alters the expression pattern of angio-neogenetic factors in a pre-clinical rat model. PLoS ONE 2013, 8, e70524. [Google Scholar] [CrossRef] [Green Version]
- Haddad, J.J. Science review: Redox and oxygen-sensitive transcription factors in the regulation of oxidant-mediated lung injury: Role for hypoxia-inducible factor-1alpha. Crit. Care 2003, 7, 47–54. [Google Scholar] [CrossRef]
- Santos, S.A.D.; Andrade, D.R.J. HIF-1alpha and infectious diseases: A new frontier for the development of new therapies. Rev. Inst. Med. Trop. Sao Paulo 2017, 59, e92. [Google Scholar] [CrossRef]
- El Awad, B.; Kreft, B.; Wolber, E.M.; Hellwig-Bürgel, T.; Metzen, E.; Fandrey, J.; Jelkmann, W. Hypoxia and interleukin-1beta stimulate vascular endothelial growth factor production in human proximal tubular cells. Kidney Int. 2000, 58, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Vohwinkel, C.U.; Hoegl, S.; Eltzschig, H.K. Hypoxia signaling during acute lung injury. J. Appl. Physiol. (1985) 2015, 119, 1157–1163. [Google Scholar] [CrossRef] [Green Version]
- Rius, J.; Guma, M.; Schachtrup, C.; Akassoglou, K.; Zinkernagel, A.S.; Nizet, V.; Johnson, R.S.; Haddad, G.G.; Karin, M. NF-kappaB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1alpha. Nature 2008, 453, 807–811. [Google Scholar] [CrossRef] [Green Version]
- Shalova, I.N.; Lim, J.Y.; Chittezhath, M.; Zinkernagel, A.S.; Beasley, F.; Hernández-Jiménez, E.; Toledano, V.; Cubillos-Zapata, C.; Rapisarda, A.; Chen, J.; et al. Human monocytes undergo functional re-programming during sepsis mediated by hypoxia-inducible factor-1α. Immunity 2015, 42, 484–498. [Google Scholar] [CrossRef] [Green Version]
- Yano, K.; Liaw, P.C.; Mullington, J.M.; Shih, S.C.; Okada, H.; Bodyak, N.; Kang, P.M.; Toltl, L.; Belikoff, B.; Buras, J.; et al. Vascular endothelial growth factor is an important determinant of sepsis morbidity and mortality. J. Exp. Med. 2006, 203, 1447–1458. [Google Scholar] [CrossRef] [PubMed]
- Mura, M.; dos Santos, C.C.; Stewart, D.; Liu, M. Vascular endothelial growth factor and related molecules in acute lung injury. J. Appl. Physiol. (1985) 2004, 97, 1605–1617. [Google Scholar] [CrossRef] [PubMed]
- Becker, P.M.; Alcasabas, A.; Yu, A.Y.; Semenza, G.L.; Bunton, T.E. Oxygen-independent upregulation of vascular endothelial growth factor and vascular barrier dysfunction during ventilated pulmonary ischemia in isolated ferret lungs. Am. J. Respir. Cell Mol. Biol. 2000, 22, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Jeong, J.S.; Kim, S.R.; Park, S.Y.; Chae, H.J.; Lee, Y.C. Inhibition of endoplasmic reticulum stress alleviates lipopolysaccharide-induced lung inflammation through modulation of NF-kappaB/HIF-1alpha signaling pathway. Sci. Rep. 2013, 3, 1142. [Google Scholar] [CrossRef]
- Li, M.; Li, G.; Yu, B.; Luo, Y.; Li, Q. Activation of hypoxia-inducible factor -1α via succinate dehydrogenase pathway during acute lung injury induced by trauma/hemorrhagic shock. Shock 2019, 53, 208–216. [Google Scholar] [CrossRef]
- Sun, H.D.; Liu, Y.J.; Chen, J.; Chen, M.Y.; Ouyang, B.; Guan, X.D. The pivotal role of HIF-1α in lung inflammatory injury induced by septic mesenteric lymph. Biomed. Pharmacother. 2017, 91, 476–484. [Google Scholar] [CrossRef]
- Suresh, M.V.; Balijepalli, S.; Zhang, B.; Singh, V.V.; Swamy, S.; Panicker, S.; Dolgachev, V.A.; Subramanian, C.; Ramakrishnan, S.K.; Thomas, B.; et al. Hypoxia-Inducible Factor (HIF)-1α Promotes Inflammation and Injury Following Aspiration-Induced Lung Injury in Mice. Shock 2019, 52, 612–621. [Google Scholar] [CrossRef]
- Textoris, J.; Beaufils, N.; Quintana, G.; Ben Lassoued, A.; Zieleskiewicz, L.; Wiramus, S.; Blasco, V.; Lesavre, N.; Martin, C.; Gabert, J.; et al. Hypoxia-inducible factor (HIF1α) gene expression in human shock states. Crit. Care 2012, 16, R120. [Google Scholar] [CrossRef] [Green Version]
- Ning, F.; Wang, X.; Shang, L.; Wang, T.; Lv, C.; Qi, Z.; Wu, D. Low molecular weight heparin may prevent acute lung injury induced by sepsis in rats. Gene 2015, 557, 88–91. [Google Scholar] [CrossRef]
- Luan, Z.G.; Naranpurev, M.; Ma, X.C. Treatment of low molecular weight heparin inhibits systemic inflammation and prevents endotoxin-induced acute lung injury in rats. Inflammation 2014, 37, 924–932. [Google Scholar] [CrossRef]
- Levi, M.; van der Poll, T. Inflammation and coagulation. Crit. Care Med. 2010, 38 (Suppl. 2), S26–S34. [Google Scholar] [CrossRef]
- Li, J.; Guo, Z.Y.; Gao, X.H.; Bian, Q.; Jia, M.; Lai, X.L.; Wang, T.Y.; Bian, X.L.; Wang, H.Y. Low molecular weight heparin (LMWH) improves peritoneal function and inhibits peritoneal fibrosis possibly through suppression of HIF-1α, VEGF and TGF-β1. PLoS ONE 2015, 10, e0118481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, R.F.; Ward, P.A. Role of oxidants in lung injury during sepsis. Antioxid. Redox Signal. 2007, 9, 1991–2002. [Google Scholar] [CrossRef] [PubMed]
- Bardales, R.H.; Xie, S.S.; Schaefer, R.F.; Hsu, S.M. Apoptosis is a major pathway responsible for the resolution of type II pneumocytes in acute lung injury. Am. J. Pathol. 1996, 149, 845–852. [Google Scholar]
- Li, W.; Pauluhn, J. Phosgene-induced acute lung injury (ALI): Differences from chlorine-induced ALI and attempts to translate toxicology to clinical medicine. Clin. Transl. Med. 2017, 6, 19. [Google Scholar] [CrossRef]
- Görlach, A.; Diebold, I.; Schini-Kerth, V.B.; Berchner-Pfannschmidt, U.; Roth, U.; Brandes, R.P.; Kietzmann, T.; Busse, R. Thrombin activates the hypoxia-inducible factor-1 signaling pathway in vascular smooth muscle cells: Role of the p22(phox)-containing NADPH oxidase. Circ. Res. 2001, 89, 47–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buyue, Y.; Misenheimer, T.M.; Sheehan, J.P. Low molecular weight heparin inhibits plasma thrombin generation via direct targeting of factor IXa: Contribution of the serpin-independent mechanism. J. Thromb. Haemost. 2012, 10, 2086–2098. [Google Scholar] [CrossRef] [Green Version]
- Wilson, M.R.; Patel, B.V.; Takata, M. Ventilation with “clinically relevant” high tidal volumes does not promote stretch-induced injury in the lungs of healthy mice. Crit. Care Med. 2012, 40, 2850–2857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dang, E.V.; Barbi, J.; Yang, H.Y.; Jinasena, D.; Yu, H.; Zheng, Y.; Borgman, Z.; Fu, J.; Kim, Y.; Yen, H.R.; et al. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. Cell 2011, 146, 772–784. [Google Scholar] [CrossRef] [Green Version]
- Harris, T.J.; Grosso, J.F.; Yen, H.R.; Xin, H.; Kortylewski, M.; Albesiano, E.; Hipkiss, E.L.; Getnet, D.; Goldberg, M.V.; Maris, C.H. Cutting edge: An in vivo requirement for STAT3 signaling in TH17 development and TH17-dependent autoimmunity. J. Immunol. 2007, 179, 4313–4317. [Google Scholar] [CrossRef] [Green Version]
- Savov, J.D.; Whitehead, G.S.; Wang, J.; Liao, G.; Usuka, J.; Peltz, G.; Foster, W.M.; Schwartz, G.A. Ozone-induced acute pulmonary injury in inbred mouse strain. Am. J. Respir. Cell. Mol. Biol. 2004, 31, 69–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]







© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.-F.; Liu, Y.-Y.; Lin, S.-W.; Chang, C.-H.; Chen, N.-H.; Hung, C.-Y.; Lee, C.-S. Low-Molecular-Weight Heparin Reduces Ventilation-Induced Lung Injury through Hypoxia Inducible Factor-1α in a Murine Endotoxemia Model. Int. J. Mol. Sci. 2020, 21, 3097. https://doi.org/10.3390/ijms21093097
Li L-F, Liu Y-Y, Lin S-W, Chang C-H, Chen N-H, Hung C-Y, Lee C-S. Low-Molecular-Weight Heparin Reduces Ventilation-Induced Lung Injury through Hypoxia Inducible Factor-1α in a Murine Endotoxemia Model. International Journal of Molecular Sciences. 2020; 21(9):3097. https://doi.org/10.3390/ijms21093097
Chicago/Turabian StyleLi, Li-Fu, Yung-Yang Liu, Shih-Wei Lin, Chih-Hao Chang, Ning-Hung Chen, Chen-Yiu Hung, and Chung-Shu Lee. 2020. "Low-Molecular-Weight Heparin Reduces Ventilation-Induced Lung Injury through Hypoxia Inducible Factor-1α in a Murine Endotoxemia Model" International Journal of Molecular Sciences 21, no. 9: 3097. https://doi.org/10.3390/ijms21093097






