Assessment of Glial Activation Response in the Progress of Natural Scrapie after Chronic Dexamethasone Treatment
Abstract
1. Introduction
2. Results
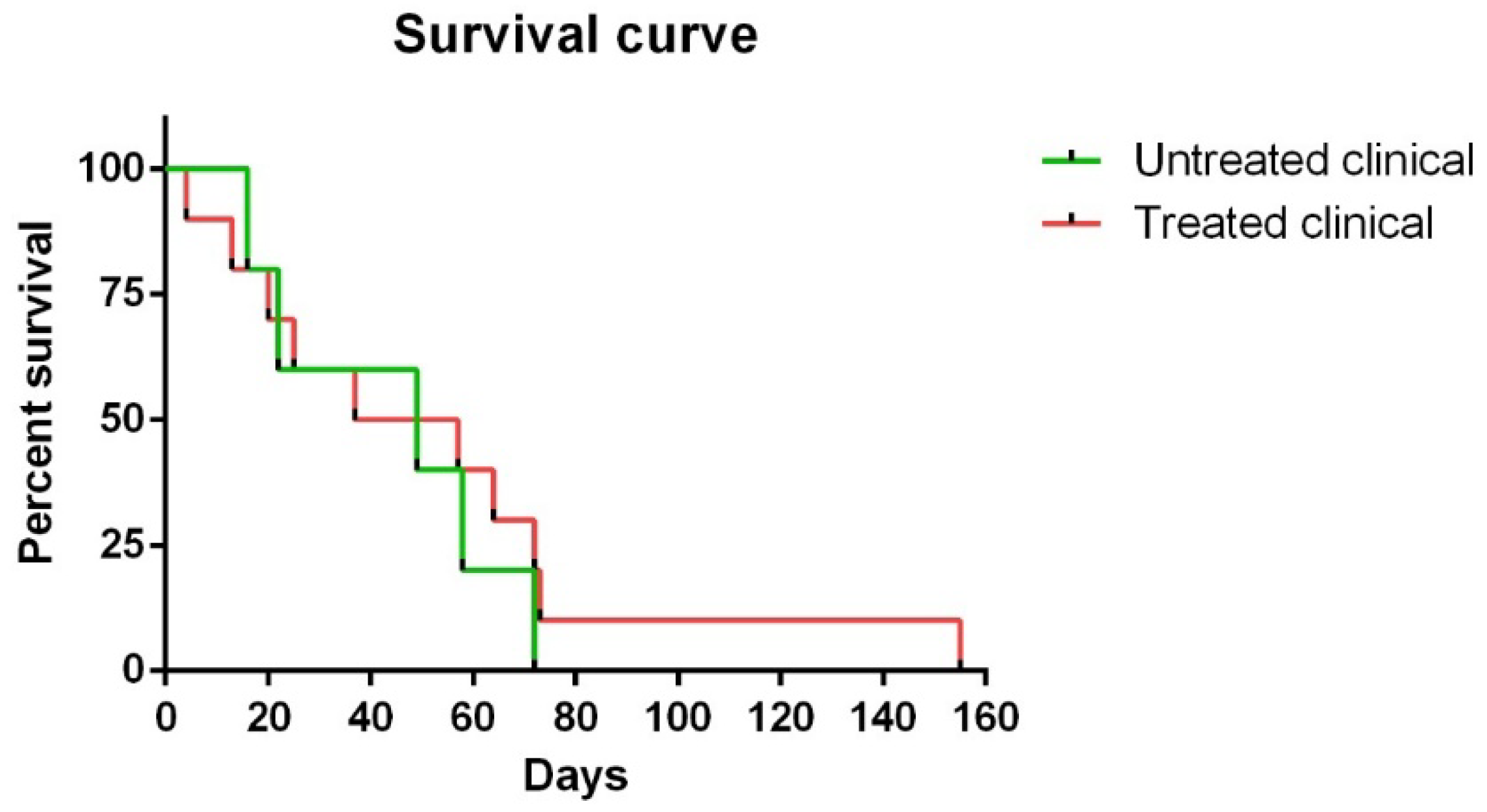
2.1. Clinical Signs
2.2. Histopathological Findings (H-E)
2.3. Immunohistochemical (IHC) Findings
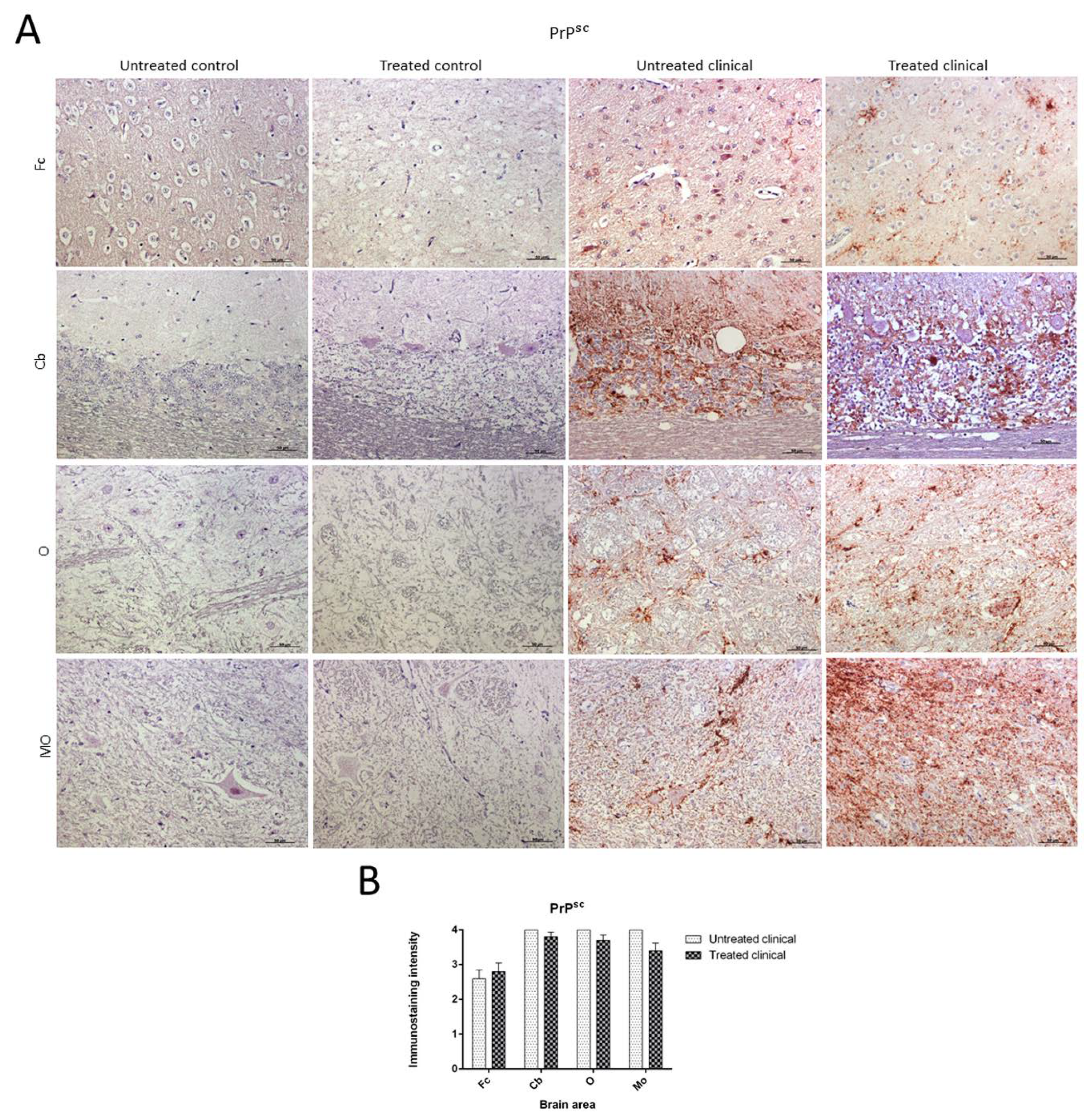
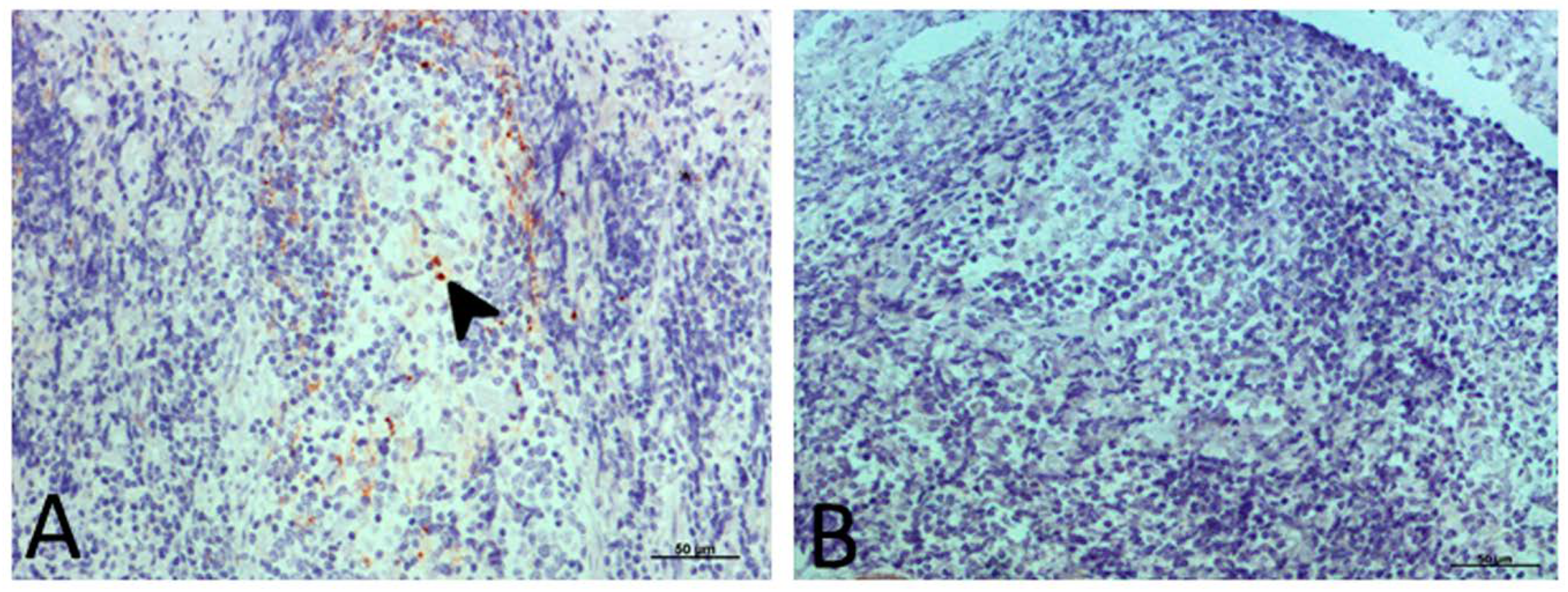
2.3.1. PrPsc Accumulation
2.3.2. GFAP
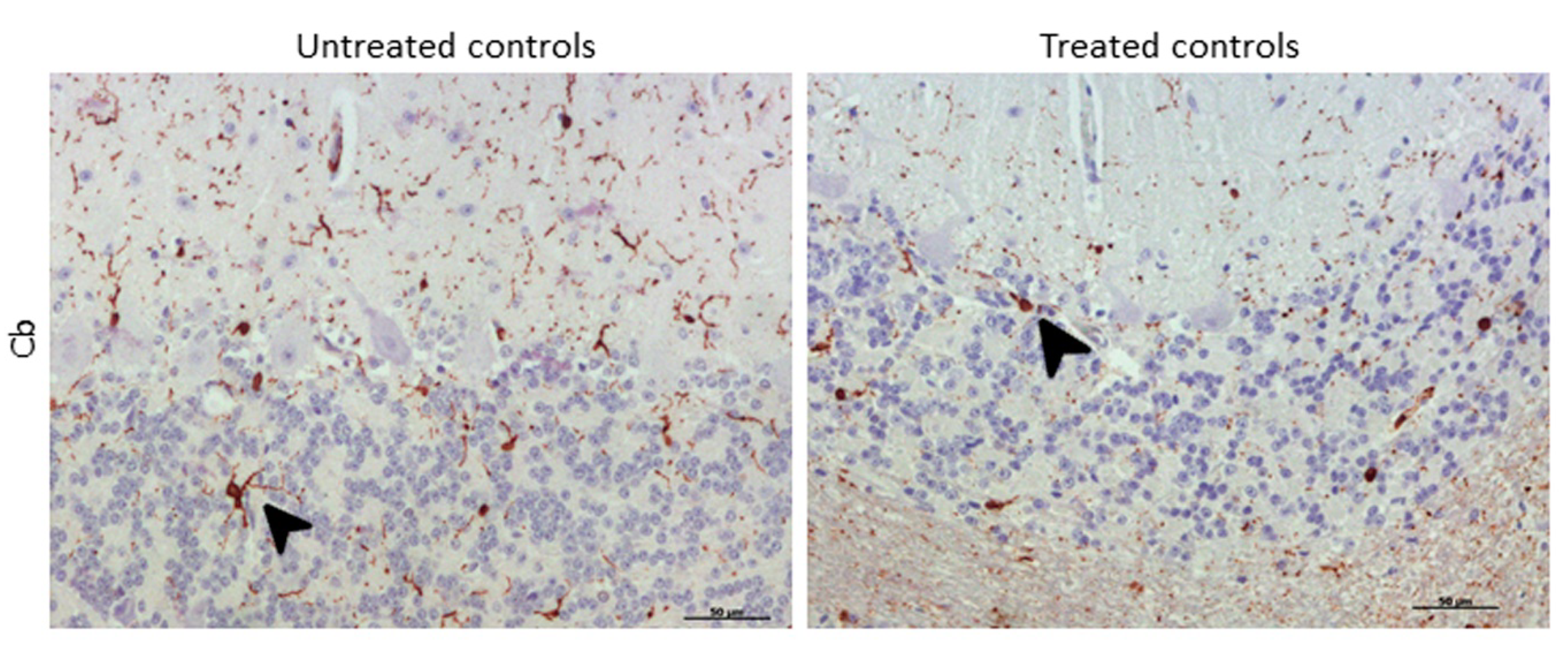
2.3.3. IBA-1
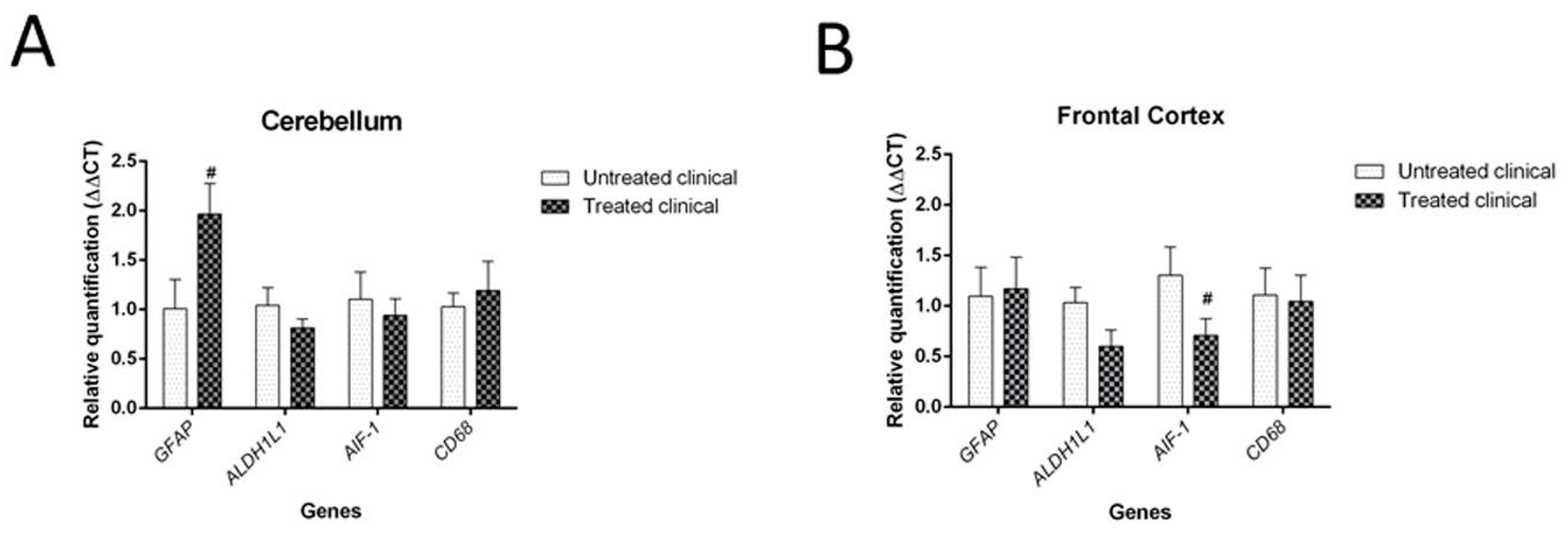
2.3.4. RT-qPCR
3. Discussion
4. Material and Methods
4.1. Histopathological Studies (H-E)
4.2. Immunohistochemical (IHC) Techniques
4.2.1. PrPsc Detection
4.2.2. Glial Fibrillary Acidic Protein (GFAP) Detection for Astrogliosis
4.2.3. Ionized Calcium-Binding Adaptor Molecule-1 (IBA-1) Detection for Microgliosis
4.2.4. RT-qPCR
4.2.5. RNA Purification
4.2.6. Retrotranscription
4.2.7. RT-qPCR
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- van Keulen, L.J.; Schreuder, B.E.; Vromans, M.E.; Langeveld, J.P.; Smits, M.A. Pathogenesis of natural scrapie in sheep. Arch. Virol. Suppl. 2000, 16, 57–71. [Google Scholar]
- González, L.; Jeffrey, M.; Sisó, S.; Martin, S.; Bellworthy, S.J.; Stack, M.J.; Chaplin, M.J.; Davis, L.; Dagleish, M.P.; Reid, H.W. Diagnosis of preclinical scrapie in samples of rectal mucosa. Vet. Rec. 2005, 156, 846–847. [Google Scholar] [CrossRef] [PubMed]
- Raymond, C.R.; Mabbott, N.A. Assessing the involvement of migratory dendritic cells in the transfer of the scrapie agent from the immune to peripheral nervous systems. J. Neuroimmunol. 2007, 187, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Tabouret, G.; Lacroux, C.; Lugan, S.; Costes, P.; Corbière, F.; Weisbecker, J.L.; Schelcher, F.; Andréoletti, O. Relevance of oral experimental challenge with classical scrapie in sheep. J. Gen. Virol. 2010, 91, 2139–2144. [Google Scholar] [CrossRef] [PubMed]
- Aisen, P.S.; Schafer, K.A.; Grundman, M.; Pfeiffer, E.; Sano, M.; Davis, K.L.; Farlow, M.R.; Jin, S.; Thomas, R.G.; Thal, L.J. Effects of rofecoxib or naproxen vs placebo on Alzheimer disease progression, a randomized controlled trial. JAMA 2003, 289, 2819–2826. [Google Scholar] [CrossRef] [PubMed]
- Gordon, P.H.; Moore, D.H.; Miller, R.G.; Florence, J.M.; Verheijde, J.L.; Doorish, C.; Hilton, J.F.; Spitalny, G.M.; MacArthur, R.B.; Mitsumoto, H.; et al. Western. Efficacy of minocycline in patients with amyotrophic lateral sclerosis, a phase III randomised trial. Lancet Neurol. 2007, 6, 1045–1053. [Google Scholar] [CrossRef]
- Barcikowska, M.; Liberski, P.P.; Boellaard, J.W.; Brown, P.; Gajdusek, D.C.; Budka, H. Microglia is a component of the prion protein amyloid plaque in the Gerstmann-Straussler-Scheinker syndrome. Acta. Neuropathol. 1993, 85, 623–627. [Google Scholar] [CrossRef]
- Liberski, P.P.; Ironside, J.W. An outline of the neuropathology of transmissible spongiform encephalopathies (prion diseases). Folia. Neuropathol. 2004, 42, 39–58. [Google Scholar]
- Ironside, J.W.; Ritchie, D.L.; Head, M.W. Phenotypic variability in human prion diseases. Neuropathol. Appl. Neurobiol. 2005, 31, 565–579. [Google Scholar] [CrossRef]
- DeArmond, S.J. Alzheimer’s disease and Creutzfeldt-Jakob disease, overlap of pathogenic mechanisms. Curr. Opin. Neurol. 1993, 6, 872–881. [Google Scholar] [CrossRef]
- DeArmond, S.J.; Yang, S.L.; Lee, A.; Bowler, R.; Taraboulos, A.; Groth, D.; Prusiner, S.B. Three scrapie prion isolates exhibit different accumulation patterns of the prion protein scrapie isoform. Proc. Natl. Acad. Sci. USA 1993, 90, 6449–6453. [Google Scholar] [CrossRef] [PubMed]
- Soto, C. Unfolding the role of protein misfolding in neurodegenerative diseases. Nat. Rev. Neurosci. 2003, 4, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Prusiner, S.B. Biology and genetics of prions causing neurodegeneration. Annu. Rev. Genet. 2013, 47, 601–623. [Google Scholar] [CrossRef] [PubMed]
- Frost, B.; Diamond, M.I. Prion-like mechanisms in neurodegenerative diseases. Nat. Rev. Neurosci. 2010, 11, 155–159. [Google Scholar] [CrossRef]
- Duyckaerts, C.; Clavaguera, F.; Potier, M.C. The prion-like propagation hypothesis in Alzheimer’s and Parkinson’s disease. Curr. Opin. Neurol. 2019, 32, 266–271. [Google Scholar] [CrossRef]
- Ransohoff, R.M. How neuroinflammation contributes to neurodegeneration. Science 2016, 353, 777–783. [Google Scholar] [CrossRef]
- Akiyama, H. Inflammatory response in Alzheimer’s disease. Tohoku. J. Exp. Med. 1994, 174, 295–303. [Google Scholar] [CrossRef]
- Aguzzi, A.; Falsig, J. Prion propagation, toxicity and degradation. Nat. Neurosci. 2012, 15, 936–939. [Google Scholar] [CrossRef]
- Xie, W.L.; Shi, Q.; Zhang, J.; Zhang, B.Y.; Gong, H.S.; Guo, Y.; Wang, S.B.; Xu, Y.; Wang, K.; Chen, C.; et al. Abnormal activation of microglia accompanied with disrupted CX3CR1/CX3CL1 pathway in the brains of the hamsters infected with scrapie agent 263K. J. Mol. Neurosci. 2013, 51, 919–932. [Google Scholar] [CrossRef]
- Carroll, J.A.; Striebel, J.F.; Rangel, A.; Woods, T.; Phillips, K.; Peterson, K.E.; Race, B.; Chesebro, B. Prion Strain Differences in Accumulation of PrPSc on Neurons and Glia Are Associated with Similar Expression Profiles of Neuroinflammatory Genes, Comparison of Three Prion Strains. PLoS Pathog. 2016, 12, e1005551. [Google Scholar] [CrossRef]
- Iaccarino, L.; Moresco, R.M.; Presotto, L.; Bugiani, O.; Iannaccone, S.; Giaccone, G.; Tagliavini, F.; Perani, D. An In Vivo (11)C-(R)-PK11195 PET and In Vitro Pathology Study of Microglia Activation in Creutzfeldt-Jakob Disease. Mol. Neurobiol. 2018, 55, 2856–2868. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Kummer, M.P.; Latz, E. Innate immune activation in neurodegenerative disease. Nat. Rev. Immunol. 2014, 14, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.E.; Lawson, L.J.; Perry, V.H.; Fraser, H. Characterization of the microglial response in murine scrapie. Neuropathol. Appl. Neurobiol. 1994, 20, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Liberski, P.P.; Sklodowski, P.; Klimek, A. Creutzfeldt-Jakob disease, ultrastructural study of brain biopsy, unusual interaction between astrocytes and oligo- and microglia. Folia. Neuropathol. 1995, 33, 85–92. [Google Scholar]
- Kordek, R.; Nerurkar, V.R.; Liberski, P.P.; Isaacson, S.; Yanagihara, R.; Gajdusek, D.C. Heightened expression of tumor necrosis factor alpha, interleukin 1 alpha, and glial fibrillary acidic protein in experimental Creutzfeldt-Jakob disease in mice. Proc. Natl. Acad. Sci. USA 1996, 93, 9754–9758. [Google Scholar] [CrossRef]
- Williams, A.; Lucassen, P.J.; Ritchie, D.; Bruce, M. PrP deposition, microglial activation, and neuronal apoptosis in murine scrapie. Exp. Neurol. 1997, 144, 433–438. [Google Scholar] [CrossRef]
- Riemer, C.; Queck, I.; Simon, D.; Kurth, R.; Baier, M. Identification of upregulated genes in scrapie-infected brain tissue. J. Virol. 2000, 74, 10245–10248. [Google Scholar] [CrossRef]
- Marella, M.; Chabry, J. Neurons and astrocytes respond to prion infection by inducing microglia recruitment. J. Neurosci. 2004, 24, 620–627. [Google Scholar] [CrossRef]
- Gomez-Nicola, D.; Fransen, N.L.; Suzzi, S.; Perry, V.H. Regulation of microglial proliferation during chronic neurodegeneration. J. Neurosci. 2013, 33, 2481–2493. [Google Scholar] [CrossRef]
- Schwartz, M.; Deczkowska, A. Neurological Disease as a Failure of Brain-Immune Crosstalk, The Multiple Faces of Neuroinflammation. Trends Immunol. 2016, 37, 668–679. [Google Scholar] [CrossRef]
- Chesebro, B.; Race, R.; Kercher, L. Scrapie pathogenesis in brain and retina, effects of prion protein expression in neurons and astrocytes. J. Neurovirol. 2005, 11, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Sarasa, R.; Martinez, A.; Monleon, E.; Bolea, R.; Vargas, A.; Badiola, J.J.; Monzon, M. Involvement of astrocytes in transmissible spongiform encephalopathies, a confocal microscopy study. Cell Tissue Res. 2012, 350, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, R.S.; Sarasa, R.; Toledano, A.; Badiola, J.J.; Monzon, M. Morphological approach to assess the involvement of astrocytes in prion propagation. Cell Tissue Res. 2014, 358, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Hollister, J.R.; Lee, K.S.; Dorward, D.W.; Baron, G.S. Efficient uptake and dissemination of scrapie prion protein by astrocytes and fibroblasts from adult hamster brain. PLoS ONE 2015, 10, e0115351. [Google Scholar] [CrossRef]
- Victoria, G.S.; Arkhipenko, A.; Zhu, S.; Syan, S.; Zurzolo, C. Astrocyte-to-neuron intercellular prion transfer is mediated by cell-cell contact. Sci. Rep. 2016, 6, 20762. [Google Scholar] [CrossRef]
- Monzón, M.; Hernández, R.S.; Garcés, M.; Sarasa, R.; Badiola, J.J. Glial alterations in human prion diseases, a correlative study of astroglia, reactive microglia, protein deposition, and neuropathological lesions. Medicine (Baltimore) 2018, 97, e0320. [Google Scholar] [CrossRef]
- Carroll, J.A.; Chesebro, B. Neuroinflammation, Microglia, and Cell-Association during Prion Disease. Viruses 2019, 11, 65. [Google Scholar] [CrossRef]
- Pekny, M.; Pekna, M. Reactive gliosis in the pathogenesis of CNS diseases. Biochim. Biophys. Acta. 2016, 1862, 483–491. [Google Scholar] [CrossRef]
- Liu, B.; Teschemacher, A.G.; Kasparov, S. Astroglia as a cellular target for neuroprotection and treatment of neuro-psychiatric disorders. Glia 2017, 65, 1205–1226. [Google Scholar] [CrossRef]
- Garcés, M.; Guijarro, M.I.; Vargas, A.; Badiola, J.J.; Monzón, M. Neuroglial patterns are shared by cerebella from prion and prion-like disorder affected patients. Mech. Ageing Dev. 2019, 184, 111176. [Google Scholar] [CrossRef]
- Guitart, K.; Loers, G.; Buck, F.; Bork, U.; Schachner, M.; Kleene, R. Improvement of neuronal cell survival by astrocyte-derived exosomes under hypoxic and ischemic conditions depends on prion protein. Glia 2016, 64, 896–910. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, U.K.; Kettenmann, H. Microglia, active sensor and versatile effector cells in the normal and pathologic brain. Nat. Neurosci. 2007, 10, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- Falsig, J.; Julius, C.; Margalith, I.; Schwarz, P.; Heppner, F.L.; Aguzzi, A. A versatile prion replication assay in organotypic brain slices. Nat. Neurosci. 2008, 11, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Herrmann, U.S.; Falsig, J.; Abakumova, I.; Nuvolone, M.; Schwarz, P.; Frauenknecht, K.; Rushing, E.J.; Aguzzi, A. A neuroprotective role for microglia in prion diseases. J. Exp. Med. 2016, 213, 1047–1059. [Google Scholar] [CrossRef]
- Carroll, J.A.; Race, B.; Williams, K.; Striebel, J.; Chesebro, B. Microglia Are Critical in Host Defense against Prion Disease. J. Virol. 2018, 92, e00549-18. [Google Scholar] [CrossRef]
- Sierra, A.; Gottfried-Blackmore, A.; Milner, T.A.; McEwen, B.S.; Bulloch, K. Steroid ormone receptor expression and function in microglia. Glia 2008, 56, 659–674. [Google Scholar] [CrossRef]
- Beeri, M.S.; Schmeidler, J.; Lesser, G.T.; Maroukian, M.; West, R.; Leung, S.; Wysocki, M.; Perl, D.P.; Purohit, D.P.; Haroutunian, V. Corticosteroids, but not NSAIDs, are associated with less Alzheimer neuropathology. Neurobiol. Aging 2012, 33, 1258–1264. [Google Scholar] [CrossRef]
- Aguzzi, A.; Zhu, C. Microglia in prion diseases. J. Clin. Invest. 2017, 127, 3230–3239. [Google Scholar] [CrossRef]
- Kimberlin, R.H.; Walker, C.A. Antiviral compound effective against experimental scrapie. Lancet 1979, 2, 591–592. [Google Scholar] [CrossRef]
- Kimberlin, R.H.; Walker, C.A. The antiviral compound HPA-23 can prevent scrapie when administered at the time of infection. Arch. Virol. 1983, 78, 9–18. [Google Scholar] [CrossRef]
- White, A.R.; Enever, P.; Tayebi, M.; Mushens, R.; Linehan, J.; Brandner, S.; Anstee, D.; Collinge, J.; Hawke, S. Monoclonal antibodies inhibit prion replication and delay the development of prion disease. Nature 2003, 422, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Grilli, M.; Memo, M. Nuclear factor-kappaB/Rel proteins, a point of convergence of signalling pathways relevant in neuronal function and dysfunction. Biochem. Pharmacol. 1999, 57, 1–7. [Google Scholar] [CrossRef]
- Efremova, L.; Chovancova, P.; Adam, M.; Gutbier, S.; Schildknecht, S.; Leist, M. Switching from astrocytic neuroprotection to neurodegeneration by cytokine stimulation. Arch. Toxicol. 2017, 91, 231–246. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kurkowska-Jastrzebska, I.; Litwin, T.; Joniec, I.; Ciesielska, A.; Przybylkowski, A.; Czlonkowski, A.; Czlonkowska, A. Dexamethasone protects against dopaminergic neurons damage in a mouse model of Parkinson’s disease. Int. Immunopharmacol. 2004, 4, 1307–1318. [Google Scholar] [CrossRef] [PubMed]
- Nerius, M.; Haenisch, B.; Gomm, M.; Doblhammer, G.; Schneider, A. Glucocorticoid Therapy is Associated with a Lower Risk of Dementia. J. Alzheimers Dis. 2020, 73, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Green, K.N.; Billings, L.M.; Roozendaal, B.; McGaugh, J.L.; LaFerla, F.M. Glucocorticoids increase amyloid-beta and tau pathology in a mouse model of Alzheimer’s disease. J. Neurosci. 2006, 26, 9047–9056. [Google Scholar] [CrossRef] [PubMed]
- Outram, G.W.; Dickinson, A.G.; Fraser, H. Reduced susceptibility to scrapie in mice after steroid administration. Nature 1974, 249, 855–856. [Google Scholar] [CrossRef]
- Riemer, C.; Burwinkel, M.; Schwarz, A.; Gultner, S.; Mok, S.W.F.; Heise, I.; Holtkamp, N.; Baier, M. Evaluation of drugs for treatment of prion infections of the central nervous system. J. Gen. Virol. 2008, 89, 594–597. [Google Scholar] [CrossRef]
- Imbimbo, B.P. An update on the efficacy of non-steroidal anti-inflammatory drugs in Alzheimer’s disease. Expert Opin. Investig. Drugs 2009, 18, 1147–1168. [Google Scholar] [CrossRef]
- Tischner, D.; Reichardt, H.M. Glucocorticoids in the control of neuroinflammation. Mol. Cell Endocrinol. 2007, 275, 62–70. [Google Scholar] [CrossRef]
- Min, L.; Hodi, F.S.; Kaiser, U.B. Corticosteroids and immune checkpoint blockade. Aging (Albany NY) 2015, 7, 521–522. [Google Scholar] [CrossRef] [PubMed]
- Munck, A.; Naray-Fejes-Toth, A. The ups and downs of glucocorticoid physiology. Permissive and suppressive effects revisited. Mol. Cell Endocrinol. 1992, 90, C1–C4. [Google Scholar] [CrossRef]
- Filaretova, L.; Podvigina, T.; Bagaeva, T.; Morozova, O. Dual action of glucocorticoid hormones on the gastric mucosa, how the gastroprotective action can be transformed to the ulcerogenic one. Inflammopharmacology 2009, 17, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Sorrells, S.F.; Caso, J.R.; Munhoz, C.D.; Sapolsky, R.M. The stressed CNS, when glucocorticoids aggravate inflammation. Neuron 2009, 64, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Spies, C.M.; Strehl, C.; van der Goes, M.C.; Bijlsma, J.W.; Buttgereit, F. Glucocorticoids. Best Pract Res. Clin. Rheumatol. 2011, 25, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, J.R. Glucocorticoid resistance in patients with rheumatoid arthritis. Scand. J. Rheumatol. 2007, 36, 165–166. [Google Scholar] [CrossRef]
- De Kloet, E.R.; Vreugdenhil, E.; Oitzl, M.S.; Joels, M. Brain corticosteroid receptor balance in health and disease. Endocr. Rev. 1998, 19, 269–301. [Google Scholar] [CrossRef]
- Meijer, O.C.; de Lange, E.C.; Breimer, D.D.; de Boer, A.G.; Workel, J.O.; de Kloet, E.R. Penetration of dexamethasone into brain glucocorticoid targets is enhanced in mdr1A P-glycoprotein knockout mice. Endocrinology 1998, 139, 1789–1793. [Google Scholar] [CrossRef]
- Hueston, C.M.; Deak, T. The inflamed axis, the interaction between stress, hormones, and the expression of inflammatory-related genes within key structures comprising the hypothalamic-pituitary-adrenal axis. Physiol. Behav. 2014, 124, 77–91. [Google Scholar] [CrossRef]
- Giles, A.J.; Hutchinson, M.N.D.; Sonnemann, H.M.; Jung, J.; Fecci, P.E.; Ratnam, N.M.; Zhang, W.; Song, H.; Bailey, R.; Davis, D.; et al. Dexamethasone-induced immunosuppression, mechanisms and implications for immunotherapy. J. Immunother. Cancer 2018, 6, 51. [Google Scholar] [CrossRef]
- Weindl, G.; Schaller, M.; Schafer-Korting, M.; Korting, H.C. Hyaluronic acid in the treatment and prevention of skin diseases, molecular biological, pharmaceutical and clinical aspects. Skin Pharmacol. Physiol. 2004, 17, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, M.W. Anti-inflammatory glucocorticoid drugs, reflections after 60 years. Inflammopharmacology 2011, 19, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Sarna, J.R.; Hawkes, R. Patterned Purkinje cell death in the cerebellum. Prog. Neurobiol. 2003, 70, 473–507. [Google Scholar] [CrossRef]
- Sarasa, R.; Junquera, C.; Toledano, A.; Badiola, J.J.; Monzón, M. Ultrastructural changes in the progress of natural Scrapie regardless fixation protocol. Histochem. Cell Biol. 2015, 144, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Lezmi, S.; Seuberlich, T.; Oevermann, A.; Baron, T.; Bencsik, A. Comparison of brain PrPd distribution in ovine BSE and scrapie. Vet. Pathol. 2011, 48, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Alibhai, J.; Blanco, R.A.; Barria, M.A.; Piccardo, P.; Caughey, B.; Perry, V.H.; Freeman, T.C.; Manson, J.C. Distribution of Misfolded Prion Protein Seeding Activity Alone Does Not Predict Regions of Neurodegeneration. PLoS Biol. 2016, 14, e1002579. [Google Scholar] [CrossRef]
- Akiyama, H.; Barger, S.; Barnum, S.; Bradt, B.; Bauer, J.; Cole, G.M.; Cooper, N.R.; Eikelenboom, P.; Emmerling, M.; Fiebich, B.L.; et al. Inflammation and Alzheimer’s disease. Neurobiol. Aging 2000, 21, 383–421. [Google Scholar] [CrossRef]
- Verkhratsky, A.; Marutle, A.; Rodriguez-Arellano, J.J.; Nordberg, A. Glial Asthenia and Functional Paralysis, A New Perspective on Neurodegeneration and Alzheimer’s Disease. Neuroscientist 2015, 21, 552–568. [Google Scholar] [CrossRef]
- Verkhratsky, A.; Rodrigues, J.J.; Pivoriunas, A.; Zorec, R.; Semyanov, A. Astroglial atrophy in Alzheimer’s disease. Pflugers Arch. 2019, 471, 1247–1261. [Google Scholar] [CrossRef]
- Fraser, H.; Brown, K.L.; Stewart, K.; McConnell, I.; McBride, P.; Williams, A. Replication of scrapie in spleens of SCID mice follows reconstitution with wild-type mouse bone marrow. J. Gen. Virol. 1996, 77, 1935–1940. [Google Scholar] [CrossRef]
- Berciano, J.; Berciano, M.T.; Polo, J.M.; Figols, J.; Ciudad, J.; Lafarga, M. Creutzfeldt-Jakob disease with severe involvement of cerebral white matter and cerebellum. Virchows Arch. Pathol. Anat. Histopathol. 1990, 417, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, R.A.; Ironside, J.W.; Lantos, P.L.; Cairns, N.J. A quantitative study of the pathological changes in the cerebellum in 15 cases of variant Creutzfeldt-Jakob disease (vCJD). Neuropathol. Appl. Neurobiol. 2009, 35, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Cali, I.; Miller, C.J.; Parisi, J.E.; Geschwind, M.D.; Gambetti, P.; Schonberger, L.B. Distinct pathological phenotypes of Creutzfeldt-Jakob disease in recipients of prion-contaminated growth hormone. Acta. Neuropathol. Commun. 2015, 3, 37. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.I.; Rivas, L.; Lacruz, C.; Toledano, A. Astroglial cell subtypes in the cerebella of normal adults, elderly adults, and patients with Alzheimer’s disease, a histological and immunohistochemical comparison. Glia 2015, 63, 287–312. [Google Scholar] [CrossRef] [PubMed]
- Zeug, A.; Muller, F.E.; Anders, S.; Herde, M.K.; Minge, D.; Ponimaskin, E.; Henneberger, C. Control of astrocyte morphology by Rho GTPases. Brain Res. Bull. 2018, 136, 44–53. [Google Scholar] [CrossRef]
- Perry, V.H.; Cunningham, C.; Boche, D. Atypical inflammation in the central nervous system in prion disease. Curr. Opin. Neurol. 2002, 15, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Perry, V.H.; O’Connor, V. The role of microglia in synaptic stripping and synaptic degeneration, a revised perspective. ASN Neuro 2010, 2, e00047. [Google Scholar] [CrossRef]
- Meneses, G.; Gevorkian, G.; Florentino, A.; Bautista, M.A.; Espinosa, A.; Acero, G.; Diaz, G.; Fleury, A.; Perez Osorio, I.N.; Del Rey, A.; et al. Intranasal delivery of dexamethasone efficiently controls LPS-induced murine neuroinflammation. Clin. Exp. Immunol. 2017, 190, 304–314. [Google Scholar] [CrossRef]
- Hui, B.; Yao, X.; Zhang, L.; Zhou, Q. Dexamethasone sodium phosphate attenuates lipopolysaccharide-induced neuroinflammation in microglia BV2 cells. Naunyn Schmiedebergs Arch. Pharmacol. 2020. [CrossRef]
- Duque Ede, A.; Munhoz, C.D. The Pro-inflammatory Effects of Glucocorticoids in the Brain. Front. Endocrinol (Lausanne) 2016, 7, 78. [Google Scholar] [CrossRef]
- Ransohoff, R.M.; Perry, V.H. Microglial physiology, unique stimuli, specialized responses. Annu. Rev. Immunol. 2009, 27, 119–145. [Google Scholar] [CrossRef] [PubMed]
- Boche, D.; Perry, V.H.; Nicoll, J.A. Review, activation patterns of microglia and their identification in the human brain. Neuropathol. Appl. Neurobiol. 2013, 39, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Streit, W.J.; Xue, Q.S.; Tischer, J.; Bechmann, I. Microglial pathology. Acta Neuropathol. Commun. 2014, 2, 142. [Google Scholar] [CrossRef]
- Kim, S.U.; de Vellis, J. Microglia in health and disease. J. Neurosci. Res. 2005, 81, 302–313. [Google Scholar] [CrossRef]
- Muhleisen, H.; Gehrmann, J.; Meyermann, R. Reactive microglia in Creutzfeldt-Jakob disease. Neuropathol. Appl. Neurobiol. 1995, 21, 505–517. [Google Scholar] [CrossRef] [PubMed]
- Llorens, F.; Lopez-Gonzalez, I.; Thune, K.; Carmona, M.; Zafar, S.; Andreoletti, O.; Zerr, I.; Ferrer, I. Subtype and regional-specific neuroinflammation in sporadic creutzfeldt-jakob disease. Front. Aging. Neurosci. 2014, 6, 198. [Google Scholar] [CrossRef] [PubMed]
- Norden, D.M.; Trojanowski, P.J.; Villanueva, E.; Navarro, E.; Godbout, J.P. Sequential activation of microglia and astrocyte cytokine expression precedes increased Iba-1 or GFAP immunoreactivity following systemic immune challenge. Glia 2016, 64, 300–316. [Google Scholar] [CrossRef]
- DiSabato, D.J.; Quan, N.; Godbout, J.P. Neuroinflammation, the devil is in the details. J. Neurochem. 2016, 139, 136–153. [Google Scholar] [CrossRef]
- Obst, J.; Simon, E.; Mancuso, R.; Gomez-Nicola, D. The Role of Microglia in Prion Diseases, A Paradigm of Functional Diversity. Front. Aging Neurosci. 2017, 9, 207. [Google Scholar] [CrossRef]
- Buffo, A.; Rolando, C.; Ceruti, S. Astrocytes in the damaged brain, molecular and cellular insights into their reactive response and healing potential. Biochem. Pharmacol. 2010, 79, 77–89. [Google Scholar] [CrossRef]
- Garwood, C.J.; Pooler, A.M.; Atherton, J.; Hanger, D.P.; Noble, W. Astrocytes are important mediators of Abeta-induced neurotoxicity and tau phosphorylation in primary culture. Cell Death Dis. 2011, 2, e167. [Google Scholar] [CrossRef] [PubMed]
- Liddelow, S.A.; Barres, B.A. Reactive Astrocytes, Production, Function, and Therapeutic Potential. Immunity 2017, 46, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Liddelow, S.A.; Guttenplan, K.A.; Clarke, L.E.; Bennett, F.C.; Bohlen, C.J.; Schirmer, L.; Bennett, M.L.; Munch, A.E.; Chung, W.S.; Peterson, T.C.; et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 2017, 541, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Kirkley, K.S.; Popichak, K.A.; Afzali, M.F.; Legare, M.E.; Tjalkens, R.B. Microglia amplify inflammatory activation of astrocytes in manganese neurotoxicity. J. Neuroinflammation 2017, 14, 99. [Google Scholar] [CrossRef]
- Espinosa, J.C.; Nonno, R.; Di Bari, M.; Aguilar-Calvo, P.; Pirisinu, L.; Fernandez-Borges, N.; Vanni, I.; Vaccari, G.; Marin-Moreno, A.; Frassanito, P.; et al. PrPC Governs Susceptibility to Prion Strains in Bank Vole, While Other Host Factors Modulate Strain Features. J. Virol. 2016, 90, 10660–10669. [Google Scholar] [CrossRef]
- Mathiason, C.K. Scrapie, CWD, and Transmissible Mink Encephalopathy. Prog. Mol. Biol. Transl. Sci. 2017, 150, 267–292. [Google Scholar]
- Kriz, J.; Nguyen, M.D.; Julien, J.P. Minocycline slows disease progression in a mouse model of amyotrophic lateral sclerosis. Neurobiol. Dis. 2002, 10, 268–278. [Google Scholar] [CrossRef]
- Choi, J.K.; Jenkins, B.G.; Carreras, I.; Kaymakcalan, S.; Cormier, K.; Kowall, N.W.; Dedeoglu, A. Anti-inflammatory treatment in AD mice protects against neuronal pathology. Exp. Neurol. 2010, 223, 377–384. [Google Scholar] [CrossRef][Green Version]
- Burwinkel, M.; Riemer, C.; Schwarz, A.; Schultz, J.; Neidhold, S.; Bamme, T.; Baier, M. Role of cytokines and chemokines in prion infections of the central nervous system. Int. J. Dev. Neurosci. 2004, 22, 497–505. [Google Scholar] [CrossRef]
- Hoes, J.N.; Jacobs, J.W.; Verstappen, S.M.; Bijlsma, J.W.; Van der Heijden, G.J. Adverse events of low- to medium-dose oral glucocorticoids in inflammatory diseases, a meta-analysis. Ann. Rheum. Dis. 2009, 68, 1833–1838. [Google Scholar] [CrossRef]
- Dickinson, A.G. Scrapie in sheep and goats. Front. Biol. 1976, 44, 209–241. [Google Scholar] [PubMed]
- Hadlow, W.J.; Kennedy, R.C.; Race, R.E. Natural infection of Suffolk sheep with scrapie virus. J. Infect. Dis. 1982, 146, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Bellworthy, S.J.; Dexter, G.; Stack, M.; Chaplin, M.; Hawkins, S.A.; Simmons, M.M.; Jeffrey, M.; Martin, S.; Gonzalez, L.; Martin, S.; et al. Oral transmission of BSE to VRQ/VRQ sheep in an experimental flock. Vet. Rec. 2008, 162, 130–131. [Google Scholar] [CrossRef] [PubMed]
- Monleon, E.; Monzon, M.; Hortells, P.; Vargas, A.; Acin, C.; Badiola, J.J. Detection of PrPsc on lymphoid tissues from naturally affected scrapie animals, comparison of three visualization systems. J. Histochem. Cytochem. 2004, 52, 145–151. [Google Scholar] [CrossRef] [PubMed]



| Group | Sheep No | Main Clinical Signs |
|---|---|---|
| Clinical non-treated | 11 | Tremors, pruritus, ataxia, lost look |
| 12 | Pruritus with skin lesions, alopecia, hyperexcitation | |
| 13 | Pruritus, alopecia, prostration | |
| 14 | Tremors, alopecia, prostration | |
| 15 | Pruritus | |
| Clinical DEX-treated | 16 | Pruritus, ataxia, tremors |
| 17 | Pruritus, alopecia | |
| 18 | Tremors, ataxia, hyperexcitation | |
| 19 | Pruritus, alopecia, hyperexcitation | |
| 20 | Tremors, intense widespread alopecia, prostration | |
| 21 | Hyperexcitation, prostration | |
| 22 | Tremors, constant pruritus, bruxism | |
| 23 | Scarce pruritus | |
| 24 | Intense pruritus with skin lesions | |
| 25 | Pruritus, alopecia, prostration |
| Sheep No | PRNP Genotype | Age (Years) | Group | Treatment and Duration |
|---|---|---|---|---|
| 1 | ARQ/ARQ | 9 | Control | Untreated (13 months) |
| 2 | ARQ/ARH | 10 | Control | Untreated (16 months) |
| 3 | ARQ/ARQ | 7 | Control | Untreated (17 months) |
| 4 | ARQ/ARQ | 4 | Control | Untreated (17 months) |
| 5 | ARQ/ARQ | 4 | Control | Untreated (16 months) |
| 6 | ARQ/ARQ | 8 | Control | Untreated (9.5 months) |
| 7 | ARQ/ARQ | 8 | Control | Treated (10 months) |
| 8 | ARQ/ARQ | 5 | Control | Treated (16 months) |
| 9 | ARQ/ARQ | 8 | Control | Treated (16 months) |
| 10 | ARQ/ARQ | 4 | Control | Treated (16 months) |
| 11 | ARQ/ARQ | 9 | Clinical | Untreated (2 months) |
| 12 | ARQ/ARQ | 4 | Clinical | Untreated (<1 month) |
| 13 | ARQ/ARQ | 5 | Clinical | Untreated (1.5 months) |
| 14 | ARQ/ARQ | 4 | Clinical | Untreated (<1 month) |
| 15 | ARQ/ARQ | 5 | Clinical | Untreated (2.5 months) |
| 16 | ARQ/ARQ | 6 | Clinical | Treated (<1 month) |
| 17 | ARQ/ARQ | 4 | Clinical | Treated (<1 month) |
| 18 | ARQ/ARQ | 4 | Clinical | Treated (<1 month) |
| 19 | ARQ/ARQ | 4 | Clinical | Treated (<1 month) |
| 20 | ARQ/ARQ | 5 | Clinical | Treated (1 month) |
| 21 | ARQ/ARQ | 4 | Clinical | Treated (2 months) |
| 22 | ARQ/ARQ | 7 | Clinical | Treated (2 months) |
| 23 | ARQ/ARQ | 4 | Clinical | Treated (2 months) |
| 24 | ARQ/ARQ | 4 | Clinical | Treated (2 months) |
| 25 | ARQ/ARQ | 4 | Clinical | Treated (5 months) |
| Antibody | Antigen | Type | Dilution | Retrieval Method | Source |
|---|---|---|---|---|---|
| L42 | PrPsc | Monoclonal | 1:500 | Formic acid, 15 min Proteinase K, 15 min Heat treatment, 20 min Peroxidase blocking | DAKO |
| Anti-GFAP | GFAP | Polyclonal | 1:500 | Peroxidase blocking | DAKO |
| Anti-IBA-1 | IBA-1 | Polyclonal | 1:1000 | Heat treatment, 20 min Peroxidase blocking | WAKO |
| Gene | Full Name | Reference | Source |
|---|---|---|---|
| AIF-1 | Allograft inflammatory factor-1 | Oa03222904_g1 | Thermo Fisher |
| ALDH1L1 | Aldehyde dehydrogenase 1 family member L1 | Oa03267152_m1 | Thermo Fisher |
| CD68 | CD68 molecule | Oa04741636_g1 | Thermo Fisher |
| GFAP | Glial fibrillary acidic protein | Oa03251662_m1 | Thermo Fisher |
| GUS-β | β-Glucuronidase (reference gene) | Oa04828868_m1 | Thermo Fisher |
| HPRT-1 | Hypoxanthine phosphoribosyl transferase-1 (reference gene) | Oa04825272_gH | Thermo Fisher |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guijarro, I.M.; Garcés, M.; Andrés-Benito, P.; Marín, B.; Otero, A.; Barrio, T.; Carmona, M.; Ferrer, I.; Badiola, J.J.; Monzón, M. Assessment of Glial Activation Response in the Progress of Natural Scrapie after Chronic Dexamethasone Treatment. Int. J. Mol. Sci. 2020, 21, 3231. https://doi.org/10.3390/ijms21093231
Guijarro IM, Garcés M, Andrés-Benito P, Marín B, Otero A, Barrio T, Carmona M, Ferrer I, Badiola JJ, Monzón M. Assessment of Glial Activation Response in the Progress of Natural Scrapie after Chronic Dexamethasone Treatment. International Journal of Molecular Sciences. 2020; 21(9):3231. https://doi.org/10.3390/ijms21093231
Chicago/Turabian StyleGuijarro, Isabel M., Moisés Garcés, Pol Andrés-Benito, Belén Marín, Alicia Otero, Tomás Barrio, Margarita Carmona, Isidro Ferrer, Juan J. Badiola, and Marta Monzón. 2020. "Assessment of Glial Activation Response in the Progress of Natural Scrapie after Chronic Dexamethasone Treatment" International Journal of Molecular Sciences 21, no. 9: 3231. https://doi.org/10.3390/ijms21093231
APA StyleGuijarro, I. M., Garcés, M., Andrés-Benito, P., Marín, B., Otero, A., Barrio, T., Carmona, M., Ferrer, I., Badiola, J. J., & Monzón, M. (2020). Assessment of Glial Activation Response in the Progress of Natural Scrapie after Chronic Dexamethasone Treatment. International Journal of Molecular Sciences, 21(9), 3231. https://doi.org/10.3390/ijms21093231







