Spatiotemporal Regulation of Signaling: Focus on T Cell Activation and the Immunological Synapse
Abstract
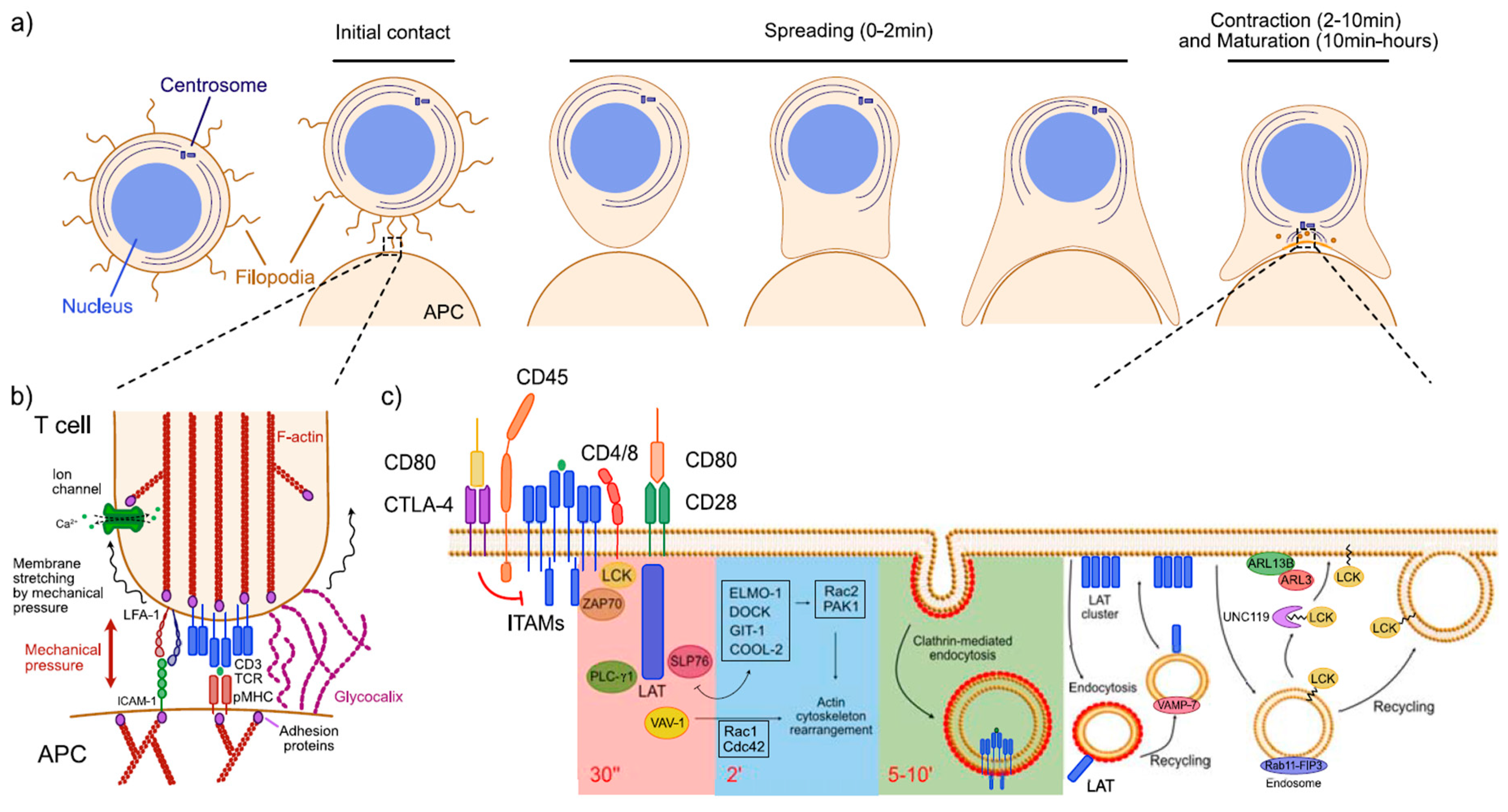
:1. Introduction
2. Temporal Regulation of Signaling during Early T Cell Activation
3. Spatial Regulation of Signaling during Early T Cell Activation
3.1. Generation of Specialized Membrane Domains at the Immunological Synapse
3.2. Dynamic Behavior of Molecules at the Immunological Synapse: Barriers to Free Diffusion
4. Conclusion and Future Perspectives
Funding
Conflicts of Interest
Abbreviations
| APC | Antigen Presenting Cell |
| ARL3 | GTPase ADP-ribosylation factor-like protein 3 |
| ARL13B | GTPase ADP-ribosylation factor-like protein 13B |
| DOCK | Dedicator of Cytokinesis |
| IFT | Intraflagellar Transport |
| IL-2 | Interleukin-2 |
| INPP5E/F | Inositol polyphosphate 5-phosphatase E/F |
| IS | Immunological Synapse |
| ITK | Interleukin-2-inducible T-cell Kinase |
| LAT | Linker for Activation of T cells |
| MAL | Myelin and Lymphocytes protein |
| MHC | Major Histocompatibility Complex |
| PDE6δ | Phosphodiesterase 6δ |
| PI3K | Phosphoinositide 3-Kinase |
| PKC-θ | Protein Kinase C-θ |
| PLC-γ | Phospholipase C-γ |
| PTPN6 | Tyrosine-protein Phosphatase Nonreceptor type 6 |
| v-SNARE | v-SNAP Receptor |
| SLP-76 | SH2 domain containing Leukocyte Protein 76 |
| TCR | T Cell Receptor |
| SMAC | Supramacromolecular activation center |
| TIRF | Total internal reflection fluorescence |
| UNC | Uncoordinated |
| VAMP-7 | Vesicle Associated Membrane Protein 7 |
| WASP | Wiskott–Aldrich Syndrome Protein |
| ZAP-70 | Zeta-chain-Associated Protein kinase 70 |
References
- Grecco, H.E.; Schmick, M.; Bastiaens, P.I. Signaling from the living plasma membrane. Cell 2011, 144, 897–909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friedl, P.; den Boer, A.T.; Gunzer, M. Tuning immune responses: Diversity and adaptation of the immunological synapse. Nat. Rev. Immunol. 2005, 5, 532–545. [Google Scholar] [CrossRef] [PubMed]
- Thauland, T.J.; Parker, D.C. Diversity in immunological synapse structure. Immunology 2010, 131, 466–472. [Google Scholar] [CrossRef]
- Dustin, M.L. T-cell activation through immunological synapses and kinapses. Immunol. Rev. 2008, 221, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Torralba, D.; Martin-Cofreces, N.B.; Sanchez-Madrid, F. Mechanisms of polarized cell-cell communication of T lymphocytes. Immunol. Lett. 2019, 209, 11–20. [Google Scholar] [CrossRef]
- Alarcon, B.; Mestre, D.; Martinez-Martin, N. The immunological synapse: A cause or consequence of T-cell receptor triggering? Immunology 2011, 133, 420–425. [Google Scholar] [CrossRef]
- Soares, H.; Lasserre, R.; Alcover, A. Orchestrating cytoskeleton and intracellular vesicle traffic to build functional immunological synapses. Immunol. Rev. 2013, 256, 118–132. [Google Scholar] [CrossRef]
- Monks, C.R.; Freiberg, B.A.; Kupfer, H.; Sciaky, N.; Kupfer, A. Three-dimensional segregation of supramolecular activation clusters in T cells. Nature 1998, 395, 82–86. [Google Scholar] [CrossRef]
- Grakoui, A.; Bromley, S.K.; Sumen, C.; Davis, M.M.; Shaw, A.S.; Allen, P.M.; Dustin, M.L. The immunological synapse: A molecular machine controlling T cell activation. Science 1999, 285, 221–227. [Google Scholar] [CrossRef] [Green Version]
- Iwashima, M.; Irving, B.A.; van Oers, N.S.; Chan, A.C.; Weiss, A. Sequential interactions of the TCR with two distinct cytoplasmic tyrosine kinases. Science 1994, 263, 1136–1139. [Google Scholar] [CrossRef]
- Davis, S.J.; van der Merwe, P.A. The kinetic-segregation model: TCR triggering and beyond. Nat. Immunol. 2006, 7, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Sieh, M.; Bolen, J.B.; Weiss, A. CD45 specifically modulates binding of Lck to a phosphopeptide encompassing the negative regulatory tyrosine of Lck. EMBO J. 1993, 12, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Weiss, A. T cell antigen receptor signal transduction: A tale of tails and cytoplasmic protein-tyrosine kinases. Cell 1993, 73, 209–212. [Google Scholar] [CrossRef]
- Beach, D.; Gonen, R.; Bogin, Y.; Reischl, I.G.; Yablonski, D. Dual role of SLP-76 in mediating T cell receptor-induced activation of phospholipase C-gamma1. J. Biol. Chem. 2007, 282, 2937–2946. [Google Scholar] [CrossRef] [Green Version]
- Berg, L.J.; Finkelstein, L.D.; Lucas, J.A.; Schwartzberg, P.L. Tec family kinases in T lymphocyte development and function. Annu. Rev. Immunol. 2005, 23, 549–600. [Google Scholar] [CrossRef]
- Lo, W.L.; Shah, N.H.; Ahsan, N.; Horkova, V.; Stepanek, O.; Salomon, A.R.; Kuriyan, J.; Weiss, A. Lck promotes Zap70-dependent LAT phosphorylation by bridging Zap70 to LAT. Nat. Immunol. 2018, 19, 733–741. [Google Scholar] [CrossRef]
- Gonzalez-Amaro, R.; Cortes, J.R.; Sanchez-Madrid, F.; Martin, P. Is CD69 an effective brake to control inflammatory diseases? Trends Mol. Med. 2013, 19, 625–632. [Google Scholar] [CrossRef] [Green Version]
- Boyman, O.; Sprent, J. The role of interleukin-2 during homeostasis and activation of the immune system. Nat. Rev. Immunol. 2012, 12, 180–190. [Google Scholar] [CrossRef]
- Yokosuka, T.; Saito, T. The immunological synapse, TCR microclusters, and T cell activation. Curr. Top. Microbiol. Immunol. 2010, 340, 81–107. [Google Scholar]
- Na, S.; Collin, O.; Chowdhury, F.; Tay, B.; Ouyang, M.; Wang, Y.; Wang, N. Rapid signal transduction in living cells is a unique feature of mechanotransduction. Proc. Natl. Acad. Sci. USA 2008, 105, 6626–6631. [Google Scholar] [CrossRef] [Green Version]
- Janmey, P.A.; Miller, R.T. Mechanisms of mechanical signaling in development and disease. J. Cell Sci. 2011, 124, 9–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borovsky, Z.; Mishan-Eisenberg, G.; Yaniv, E.; Rachmilewitz, J. Serial triggering of T cell receptors results in incremental accumulation of signaling intermediates. J. Biol. Chem. 2002, 277, 21529–21536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heinrich, R.; Neel, B.G.; Rapoport, T.A. Mathematical models of protein kinase signal transduction. Mol. Cell 2002, 9, 957–970. [Google Scholar] [CrossRef]
- Morishita, Y.; Kobayashi, T.J.; Aihara, K. An optimal number of molecules for signal amplification and discrimination in a chemical cascade. Biophys. J. 2006, 91, 2072–2081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fooksman, D.R.; Vardhana, S.; Vasiliver-Shamis, G.; Liese, J.; Blair, D.A.; Waite, J.; Sacristan, C.; Victora, G.D.; Zanin-Zhorov, A.; Dustin, M.L. Functional anatomy of T cell activation and synapse formation. Annu. Rev. Immunol. 2010, 28, 79–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poh, Y.C.; Na, S.; Chowdhury, F.; Ouyang, M.; Wang, Y.; Wang, N. Rapid activation of Rac GTPase in living cells by force is independent of Src. PLoS ONE 2009, 4, e7886. [Google Scholar] [CrossRef] [Green Version]
- Wang, N.; Butler, J.P.; Ingber, D.E. Mechanotransduction across the cell surface and through the cytoskeleton. Science 1993, 260, 1124–1127. [Google Scholar] [CrossRef]
- Cohen, B.E. Membrane Thickness as a Key Factor Contributing to the Activation of Osmosensors and Essential Ras Signaling Pathways. Front. Cell Dev. Biol. 2018, 6, 76. [Google Scholar] [CrossRef] [Green Version]
- Verstraeten, S.V.; Mackenzie, G.G.; Oteiza, P.I. The plasma membrane plays a central role in cells response to mechanical stress. Biochim. Biophys. Acta 2010, 1798, 1739–1749. [Google Scholar] [CrossRef] [Green Version]
- Le Roux, A.L.; Quiroga, X.; Walani, N.; Arroyo, M.; Roca-Cusachs, P. The plasma membrane as a mechanochemical transducer. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2019, 374, 20180221. [Google Scholar] [CrossRef]
- Cox, C.D.; Bavi, N.; Martinac, B. Biophysical Principles of Ion-Channel-Mediated Mechanosensory Transduction. Cell Rep. 2019, 29, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hivroz, C.; Saitakis, M. Biophysical Aspects of T Lymphocyte Activation at the Immune Synapse. Front. Immunol. 2016, 7, 46. [Google Scholar] [CrossRef] [Green Version]
- Roy, N.H.; Burkhardt, J.K. The Actin Cytoskeleton: A Mechanical Intermediate for Signal Integration at the Immunological Synapse. Front. Cell Dev. Biol. 2018, 6, 116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Comrie, W.A.; Burkhardt, J.K. Action and Traction: Cytoskeletal Control of Receptor Triggering at the Immunological Synapse. Front. Immunol. 2016, 7, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, S.; Di Bartolo, V.; Tubul, L.; Shimoni, E.; Kartvelishvily, E.; Dadosh, T.; Feigelson, S.W.; Alon, R.; Alcover, A.; Haran, G. ERM-Dependent Assembly of T Cell Receptor Signaling and Co-stimulatory Molecules on Microvilli prior to Activation. Cell Rep. 2020, 30, 3434–3447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Razvag, Y.; Neve-Oz, Y.; Sajman, J.; Reches, M.; Sherman, E. Nanoscale kinetic segregation of TCR and CD45 in engaged microvilli facilitates early T cell activation. Nat. Commun. 2018, 9, 732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, E.; Marchuk, K.; Beemiller, P.; Beppler, C.; Rubashkin, M.G.; Weaver, V.M.; Gerard, A.; Liu, T.L.; Chen, B.C.; Betzig, E.; et al. Visualizing dynamic microvillar search and stabilization during ligand detection by T cells. Science 2017, 356, eaal3118. [Google Scholar] [CrossRef] [Green Version]
- Jung, Y.; Riven, I.; Feigelson, S.W.; Kartvelishvily, E.; Tohya, K.; Miyasaka, M.; Alon, R.; Haran, G. Three-dimensional localization of T-cell receptors in relation to microvilli using a combination of superresolution microscopies. Proc. Natl. Acad. Sci. USA 2016, 113, E5916–E5924. [Google Scholar] [CrossRef] [Green Version]
- Gaus, K.; Gratton, E.; Kable, E.P.; Jones, A.S.; Gelissen, I.; Kritharides, L.; Jessup, W. Visualizing lipid structure and raft domains in living cells with two-photon microscopy. Proc. Natl. Acad. Sci. USA 2003, 100, 15554–15559. [Google Scholar] [CrossRef] [Green Version]
- James, J.R.; Vale, R.D. Biophysical mechanism of T-cell receptor triggering in a reconstituted system. Nature 2012, 487, 64–69. [Google Scholar] [CrossRef]
- Ostrowski, P.P.; Grinstein, S.; Freeman, S.A. Diffusion Barriers, Mechanical Forces, and the Biophysics of Phagocytosis. Dev. Cell 2016, 38, 135–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varga, G.; Nippe, N.; Balkow, S.; Peters, T.; Wild, M.K.; Seeliger, S.; Beissert, S.; Krummen, M.; Roth, J.; Sunderkotter, C.; et al. LFA-1 contributes to signal I of T-cell activation and to the production of T(h)1 cytokines. J. Invest. Dermatol. 2010, 130, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Garcia, A.J.; Mould, A.P.; Humphries, M.J.; Zhu, C. Demonstration of catch bonds between an integrin and its ligand. J. Cell Biol. 2009, 185, 1275–1284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, F.; Li, Z.; Parks, W.M.; Dumbauld, D.W.; Garcia, A.J.; Mould, A.P.; Humphries, M.J.; Zhu, C. Cyclic mechanical reinforcement of integrin-ligand interactions. Mol. Cell 2013, 49, 1060–1068. [Google Scholar] [CrossRef] [Green Version]
- Wenning, A.S.; Neblung, K.; Strauss, B.; Wolfs, M.J.; Sappok, A.; Hoth, M.; Schwarz, E.C. TRP expression pattern and the functional importance of TRPC3 in primary human T-cells. Biochim. Biophys. Acta 2011, 1813, 412–423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pottosin, I.; Delgado-Enciso, I.; Bonales-Alatorre, E.; Nieto-Pescador, M.G.; Moreno-Galindo, E.G.; Dobrovinskaya, O. Mechanosensitive Ca(2)(+)-permeable channels in human leukemic cells: Pharmacological and molecular evidence for TRPV2. Biochim. Biophys. Acta 2015, 1848, 51–59. [Google Scholar] [CrossRef] [Green Version]
- Schamel, W.W.; Alarcon, B.; Minguet, S. The TCR is an allosterically regulated macromolecular machinery changing its conformation while working. Immunol. Rev. 2019, 291, 8–25. [Google Scholar] [CrossRef]
- Malissen, B.; Bongrand, P. Early T cell activation: Integrating biochemical, structural, and biophysical cues. Annu. Rev. Immunol. 2015, 33, 539–561. [Google Scholar] [CrossRef]
- Ma, Z.; Janmey, P.A.; Finkel, T.H. The receptor deformation model of TCR triggering. FASEB J. 2008, 22, 1002–1008. [Google Scholar] [CrossRef] [Green Version]
- Husson, J.; Chemin, K.; Bohineust, A.; Hivroz, C.; Henry, N. Force generation upon T cell receptor engagement. PLoS ONE 2011, 6, e19680. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.H.; Reinherz, E.L. The structural basis of alphabeta T-lineage immune recognition: TCR docking topologies, mechanotransduction, and co-receptor function. Immunol. Rev. 2012, 250, 102–119. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Discher, D.E.; Finkel, T.H. Mechanical force in T cell receptor signal initiation. Front. Immunol. 2012, 3, 217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Z.; Finkel, T.H. T cell receptor triggering by force. Trends Immunol. 2010, 31, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.T.; Takeuchi, K.; Sun, Z.Y.; Touma, M.; Castro, C.E.; Fahmy, A.; Lang, M.J.; Wagner, G.; Reinherz, E.L. The alphabeta T cell receptor is an anisotropic mechanosensor. J. Biol. Chem. 2009, 284, 31028–31037. [Google Scholar] [CrossRef] [Green Version]
- Lim, T.S.; Mortellaro, A.; Lim, C.T.; Hammerling, G.J.; Ricciardi-Castagnoli, P. Mechanical interactions between dendritic cells and T cells correlate with T cell responsiveness. J. Immunol. 2011, 187, 258–265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, Y.; Brazin, K.N.; Kobayashi, E.; Mallis, R.J.; Reinherz, E.L.; Lang, M.J. Mechanosensing drives acuity of alphabeta T-cell recognition. Proc. Natl. Acad. Sci. USA 2017, 114, E8204–E8213. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.; Chen, W.; Evavold, B.D.; Zhu, C. Accumulation of dynamic catch bonds between TCR and agonist peptide-MHC triggers T cell signaling. Cell 2014, 157, 357–368. [Google Scholar] [CrossRef] [Green Version]
- Santos, A.M.; Ponjavic, A.; Fritzsche, M.; Fernandes, R.A.; de la Serna, J.B.; Wilcock, M.J.; Schneider, F.; Urbancic, I.; McColl, J.; Anzilotti, C.; et al. Capturing resting T cells: The perils of PLL. Nat. Immunol. 2018, 19, 203–205. [Google Scholar] [CrossRef]
- Huang, J.; Brameshuber, M.; Zeng, X.; Xie, J.; Li, Q.J.; Chien, Y.H.; Valitutti, S.; Davis, M.M. A single peptide-major histocompatibility complex ligand triggers digital cytokine secretion in CD4(+) T cells. Immunity 2013, 39, 846–857. [Google Scholar] [CrossRef] [Green Version]
- O’Donoghue, G.P.; Pielak, R.M.; Smoligovets, A.A.; Lin, J.J.; Groves, J.T. Direct single molecule measurement of TCR triggering by agonist pMHC in living primary T cells. Elife 2013, 2, e00778. [Google Scholar] [CrossRef]
- Evavold, B.D.; Sloan-Lancaster, J.; Allen, P.M. Tickling the TCR: Selective T-cell functions stimulated by altered peptide ligands. Immunol. Today 1993, 14, 602–609. [Google Scholar] [CrossRef]
- van Panhuys, N.; Klauschen, F.; Germain, R.N. T-cell-receptor-dependent signal intensity dominantly controls CD4(+) T cell polarization In Vivo. Immunity 2014, 41, 63–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tubo, N.J.; Pagan, A.J.; Taylor, J.J.; Nelson, R.W.; Linehan, J.L.; Ertelt, J.M.; Huseby, E.S.; Way, S.S.; Jenkins, M.K. Single naive CD4+ T cells from a diverse repertoire produce different effector cell types during infection. Cell 2013, 153, 785–796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manz, B.N.; Jackson, B.L.; Petit, R.S.; Dustin, M.L.; Groves, J. T-cell triggering thresholds are modulated by the number of antigen within individual T-cell receptor clusters. Proc. Natl. Acad. Sci. USA 2011, 108, 9089–9094. [Google Scholar] [CrossRef] [Green Version]
- Eggena, M.P.; Walker, L.S.; Nagabhushanam, V.; Barron, L.; Chodos, A.; Abbas, A.K. Cooperative roles of CTLA-4 and regulatory T cells in tolerance to an islet cell antigen. J. Exp. Med. 2004, 199, 1725–1730. [Google Scholar] [CrossRef]
- Lovatt, M.; Filby, A.; Parravicini, V.; Werlen, G.; Palmer, E.; Zamoyska, R. Lck regulates the threshold of activation in primary T cells, while both Lck and Fyn contribute to the magnitude of the extracellular signal-related kinase response. Mol. Cell. Biol. 2006, 26, 8655–8665. [Google Scholar] [CrossRef] [Green Version]
- Brockmeyer, C.; Paster, W.; Pepper, D.; Tan, C.P.; Trudgian, D.C.; McGowan, S.; Fu, G.; Gascoigne, N.R.; Acuto, O.; Salek, M. T cell receptor (TCR)-induced tyrosine phosphorylation dynamics identifies THEMIS as a new TCR signalosome component. J. Biol. Chem. 2011, 286, 7535–7547. [Google Scholar] [CrossRef] [Green Version]
- Nika, K.; Soldani, C.; Salek, M.; Paster, W.; Gray, A.; Etzensperger, R.; Fugger, L.; Polzella, P.; Cerundolo, V.; Dushek, O.; et al. Constitutively active Lck kinase in T cells drives antigen receptor signal transduction. Immunity 2010, 32, 766–777. [Google Scholar] [CrossRef] [Green Version]
- Moogk, D.; Zhong, S.; Yu, Z.; Liadi, I.; Rittase, W.; Fang, V.; Dougherty, J.; Perez-Garcia, A.; Osman, I.; Zhu, C.; et al. Constitutive Lck Activity Drives Sensitivity Differences between CD8+ Memory T Cell Subsets. J. Immunol. 2016, 197, 644–654. [Google Scholar] [CrossRef] [Green Version]
- Ballek, O.; Valecka, J.; Manning, J.; Filipp, D. The pool of preactivated Lck in the initiation of T-cell signaling: A critical re-evaluation of the Lck standby model. Immunol. Cell Biol. 2015, 93, 384–395. [Google Scholar] [CrossRef]
- Iezzi, G.; Karjalainen, K.; Lanzavecchia, A. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 1998, 8, 89–95. [Google Scholar] [CrossRef] [Green Version]
- Schneider, H.; Martin, M.; Agarraberes, F.A.; Yin, L.; Rapoport, I.; Kirchhausen, T.; Rudd, C.E. Cytolytic T lymphocyte-associated antigen-4 and the TCR zeta/CD3 complex, but not CD28, interact with clathrin adaptor complexes AP-1 and AP-2. J. Immunol. 1999, 163, 1868–1879. [Google Scholar] [PubMed]
- Liu, H.; Rhodes, M.; Wiest, D.L.; Vignali, D.A. On the dynamics of TCR:CD3 complex cell surface expression and downmodulation. Immunity 2000, 13, 665–675. [Google Scholar] [CrossRef] [Green Version]
- Finetti, F.; Patrussi, L.; Galgano, D.; Cassioli, C.; Perinetti, G.; Pazour, G.J.; Baldari, C.T. The small GTPase Rab8 interacts with VAMP-3 to regulate the delivery of recycling T-cell receptors to the immune synapse. J. Cell Sci. 2015, 128, 2541–2552. [Google Scholar] [CrossRef] [Green Version]
- Redpath, G.M.I.; Ecker, M.; Kapoor-Kaushik, N.; Vartoukian, H.; Carnell, M.; Kempe, D.; Biro, M.; Ariotti, N.; Rossy, J. Flotillins promote T cell receptor sorting through a fast Rab5-Rab11 endocytic recycling axis. Nat. Commun. 2019, 10, 4392. [Google Scholar] [CrossRef] [Green Version]
- Anton, O.; Batista, A.; Millan, J.; Andres-Delgado, L.; Puertollano, R.; Correas, I.; Alonso, M.A. An essential role for the MAL protein in targeting Lck to the plasma membrane of human T lymphocytes. J. Exp. Med. 2008, 205, 3201–3213. [Google Scholar] [CrossRef] [Green Version]
- Pfisterer, K.; Forster, F.; Paster, W.; Supper, V.; Ohradanova-Repic, A.; Eckerstorfer, P.; Zwirzitz, A.; Donner, C.; Boulegue, C.; Schiller, H.B.; et al. The late endosomal transporter CD222 directs the spatial distribution and activity of Lck. J. Immunol. 2014, 193, 2718–2732. [Google Scholar] [CrossRef] [Green Version]
- Gorska, M.M.; Liang, Q.; Karim, Z.; Alam, R. Uncoordinated 119 protein controls trafficking of Lck via the Rab11 endosome and is critical for immunological synapse formation. J. Immunol. 2009, 183, 1675–1684. [Google Scholar] [CrossRef] [Green Version]
- Stephen, L.A.; ElMaghloob, Y.; McIlwraith, M.J.; Yelland, T.; Castro Sanchez, P.; Roda-Navarro, P.; Ismail, S. The Ciliary Machinery Is Repurposed for T Cell Immune Synapse Trafficking of LCK. Dev. Cell 2018, 47, 122–132. [Google Scholar] [CrossRef] [Green Version]
- Stephen, L.A.; Ismail, S. Shuttling and sorting lipid-modified cargo into the cilia. Biochem. Soc. Trans. 2016, 44, 1273–1280. [Google Scholar] [CrossRef] [Green Version]
- Fansa, E.K.; Kosling, S.K.; Zent, E.; Wittinghofer, A.; Ismail, S. PDE6delta-mediated sorting of INPP5E into the cilium is determined by cargo-carrier affinity. Nat. Commun. 2016, 7, 11366. [Google Scholar] [CrossRef] [Green Version]
- Brignatz, C.; Restouin, A.; Bonello, G.; Olive, D.; Collette, Y. Evidences for ubiquitination and intracellular trafficking of LAT, the linker of activated T cells. Biochim. Biophys. Acta 2005, 1746, 108–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larghi, P.; Williamson, D.J.; Carpier, J.M.; Dogniaux, S.; Chemin, K.; Bohineust, A.; Danglot, L.; Gaus, K.; Galli, T.; Hivroz, C. VAMP7 controls T cell activation by regulating the recruitment and phosphorylation of vesicular Lat at TCR-activation sites. Nat. Immunol. 2013, 14, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Vivar, O.I.; Masi, G.; Carpier, J.M.; Magalhaes, J.G.; Galgano, D.; Pazour, G.J.; Amigorena, S.; Hivroz, C.; Baldari, C.T. IFT20 controls LAT recruitment to the immune synapse and T-cell activation in vivo. Proc. Natl. Acad. Sci. USA 2016, 113, 386–391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carpier, J.M.; Zucchetti, A.E.; Bataille, L.; Dogniaux, S.; Shafaq-Zadah, M.; Bardin, S.; Lucchino, M.; Maurin, M.; Joannas, L.D.; Magalhaes, J.G.; et al. Rab6-dependent retrograde traffic of LAT controls immune synapse formation and T cell activation. J. Exp. Med. 2018, 215, 1245–1265. [Google Scholar] [CrossRef] [PubMed]
- Balagopalan, L.; Yi, J.; Nguyen, T.; McIntire, K.M.; Harned, A.S.; Narayan, K.; Samelson, L.E. Plasma membrane LAT activation precedes vesicular recruitment defining two phases of early T-cell activation. Nat. Commun. 2018, 9, 2013. [Google Scholar] [CrossRef] [Green Version]
- Yudushkin, I.A.; Vale, R.D. Imaging T-cell receptor activation reveals accumulation of tyrosine-phosphorylated CD3zeta in the endosomal compartment. Proc. Natl. Acad. Sci. USA 2010, 107, 22128–22133. [Google Scholar] [CrossRef] [Green Version]
- Willinger, T.; Staron, M.; Ferguson, S.M.; De Camilli, P.; Flavell, R.A. Dynamin 2-dependent endocytosis sustains T-cell receptor signaling and drives metabolic reprogramming in T lymphocytes. Proc. Natl. Acad. Sci. USA 2015, 112, 4423–4428. [Google Scholar] [CrossRef] [Green Version]
- Kumari, S.; Colin-York, H.; Irvine, D.J.; Fritzsche, M. Not All T Cell Synapses Are Built the Same Way. Trends Immunol. 2019, 40, 977–980. [Google Scholar] [CrossRef] [Green Version]
- Richie, L.I.; Ebert, P.J.; Wu, L.C.; Krummel, M.F.; Owen, J.J.; Davis, M.M. Imaging synapse formation during thymocyte selection: Inability of CD3zeta to form a stable central accumulation during negative selection. Immunity 2002, 16, 595–606. [Google Scholar] [CrossRef] [Green Version]
- Dustin, M.L.; Cooper, J.A. The immunological synapse and the actin cytoskeleton: Molecular hardware for T cell signaling. Nat. Immunol. 2000, 1, 23–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumari, S.; Vardhana, S.; Cammer, M.; Curado, S.; Santos, L.; Sheetz, M.P.; Dustin, M.L. T Lymphocyte Myosin IIA is Required for Maturation of the Immunological Synapse. Front. Immunol. 2012, 3, 230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammer, J.A.; Wang, J.C.; Saeed, M.; Pedrosa, A.T. Origin, Organization, Dynamics, and Function of Actin and Actomyosin Networks at the T Cell Immunological Synapse. Annu. Rev. Immunol. 2019, 37, 201–224. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto-Tane, A.; Sakuma, M.; Ike, H.; Yokosuka, T.; Kimura, Y.; Ohara, O.; Saito, T. Micro-adhesion rings surrounding TCR microclusters are essential for T cell activation. J. Exp. Med. 2016, 213, 1609–1625. [Google Scholar] [CrossRef] [PubMed]
- Varma, R.; Campi, G.; Yokosuka, T.; Saito, T.; Dustin, M.L. T cell receptor-proximal signals are sustained in peripheral microclusters and terminated in the central supramolecular activation cluster. Immunity 2006, 25, 117–127. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.H.; Holdorf, A.D.; Dustin, M.L.; Chan, A.C.; Allen, P.M.; Shaw, A.S. T cell receptor signaling precedes immunological synapse formation. Science 2002, 295, 1539–1542. [Google Scholar] [CrossRef] [Green Version]
- Freiberg, B.A.; Kupfer, H.; Maslanik, W.; Delli, J.; Kappler, J.; Zaller, D.M.; Kupfer, A. Staging and resetting T cell activation in SMACs. Nat. Immunol. 2002, 3, 911–917. [Google Scholar] [CrossRef]
- Cemerski, S.; Das, J.; Giurisato, E.; Markiewicz, M.A.; Allen, P.M.; Chakraborty, A.K.; Shaw, A.S. The balance between T cell receptor signaling and degradation at the center of the immunological synapse is determined by antigen quality. Immunity 2008, 29, 414–422. [Google Scholar] [CrossRef] [Green Version]
- Geiger, B.; Rosen, D.; Berke, G. Spatial relationships of microtubule-organizing centers and the contact area of cytotoxic T lymphocytes and target cells. J. Cell Biol. 1982, 95, 137–143. [Google Scholar] [CrossRef]
- Kupfer, A.; Dennert, G.; Singer, S.J. Polarization of the Golgi apparatus and the microtubule-organizing center within cloned natural killer cells bound to their targets. Proc. Natl. Acad. Sci. USA 1983, 80, 7224–7228. [Google Scholar] [CrossRef] [Green Version]
- Quann, E.J.; Merino, E.; Furuta, T.; Huse, M. Localized diacylglycerol drives the polarization of the microtubule-organizing center in T cells. Nat. Immunol. 2009, 10, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Dinic, J.; Riehl, A.; Adler, J.; Parmryd, I. The T cell receptor resides in ordered plasma membrane nanodomains that aggregate upon patching of the receptor. Sci. Rep. 2015, 5, 10082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beck-Garcia, K.; Beck-Garcia, E.; Bohler, S.; Zorzin, C.; Sezgin, E.; Levental, I.; Alarcon, B.; Schamel, W.W. Nanoclusters of the resting T cell antigen receptor (TCR) localize to non-raft domains. Biochim. Biophys. Acta 2015, 1853, 802–809. [Google Scholar] [CrossRef] [Green Version]
- Xavier, R.; Brennan, T.; Li, Q.; McCormack, C.; Seed, B. Membrane compartmentation is required for efficient T cell activation. Immunity 1998, 8, 723–732. [Google Scholar] [CrossRef] [Green Version]
- Montixi, C.; Langlet, C.; Bernard, A.M.; Thimonier, J.; Dubois, C.; Wurbel, M.A.; Chauvin, J.P.; Pierres, M.; He, H.T. Engagement of T cell receptor triggers its recruitment to low-density detergent-insoluble membrane domains. EMBO J. 1998, 17, 5334–5348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schamel, W.W.; Arechaga, I.; Risueno, R.M.; van Santen, H.M.; Cabezas, P.; Risco, C.; Valpuesta, J.M.; Alarcon, B. Coexistence of multivalent and monovalent TCRs explains high sensitivity and wide range of response. J. Exp. Med. 2005, 202, 493–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaus, K.; Chklovskaia, E.; Fazekas de St Groth, B.; Jessup, W.; Harder, T. Condensation of the plasma membrane at the site of T lymphocyte activation. J. Cell Biol. 2005, 171, 121–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Trible, R.P.; Samelson, L.E. LAT palmitoylation: Its essential role in membrane microdomain targeting and tyrosine phosphorylation during T cell activation. Immunity 1998, 9, 239–246. [Google Scholar] [CrossRef] [Green Version]
- Yasuda, K.; Nagafuku, M.; Shima, T.; Okada, M.; Yagi, T.; Yamada, T.; Minaki, Y.; Kato, A.; Tani-Ichi, S.; Hamaoka, T.; et al. Cutting edge: Fyn is essential for tyrosine phosphorylation of Csk-binding protein/phosphoprotein associated with glycolipid-enriched microdomains in lipid rafts in resting T cells. J. Immunol. 2002, 169, 2813–2817. [Google Scholar] [CrossRef] [Green Version]
- Filipp, D.; Zhang, J.; Leung, B.L.; Shaw, A.; Levin, S.D.; Veillette, A.; Julius, M. Regulation of Fyn through translocation of activated Lck into lipid rafts. J. Exp. Med. 2003, 197, 1221–1227. [Google Scholar] [CrossRef]
- Anton, O.M.; Andres-Delgado, L.; Reglero-Real, N.; Batista, A.; Alonso, M.A. MAL protein controls protein sorting at the supramolecular activation cluster of human T lymphocytes. J. Immunol. 2011, 186, 6345–6356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reales, E.; Bernabe-Rubio, M.; Casares-Arias, J.; Rentero, C.; Fernandez-Barrera, J.; Rangel, L.; Correas, I.; Enrich, C.; Andres, G.; Alonso, M.A. The MAL protein is crucial for proper membrane condensation at the ciliary base, which is required for primary cilium elongation. J. Cell Sci. 2015, 128, 2261–2270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gawden-Bone, C.M.; Griffiths, G.M. Phospholipids: Pulling Back the Actin Curtain for Granule Delivery to the Immune Synapse. Front. Immunol. 2019, 10, 700. [Google Scholar] [CrossRef] [PubMed]
- Gawden-Bone, C.M.; Frazer, G.L.; Richard, A.C.; Ma, C.Y.; Strege, K.; Griffiths, G.M. PIP5 Kinases Regulate Membrane Phosphoinositide and Actin Composition for Targeted Granule Secretion by Cytotoxic Lymphocytes. Immunity 2018, 49, 427–437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McLaughlin, S.; Murray, D. Plasma membrane phosphoinositide organization by protein electrostatics. Nature 2005, 438, 605–611. [Google Scholar] [CrossRef]
- Le Floc’h, A.; Tanaka, Y.; Bantilan, N.S.; Voisinne, G.; Altan-Bonnet, G.; Fukui, Y.; Huse, M. Annular PIP3 accumulation controls actin architecture and modulates cytotoxicity at the immunological synapse. J. Exp. Med. 2013, 210, 2721–2737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minagawa, T.; Ijuin, T.; Mochizuki, Y.; Takenawa, T. Identification and characterization of a sac domain-containing phosphoinositide 5-phosphatase. J. Biol. Chem. 2001, 276, 22011–22015. [Google Scholar] [CrossRef] [Green Version]
- Kisseleva, M.V.; Wilson, M.P.; Majerus, P.W. The isolation and characterization of a cDNA encoding phospholipid-specific inositol polyphosphate 5-phosphatase. J. Biol. Chem. 2000, 275, 20110–20116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chavez, M.; Ena, S.; Van Sande, J.; de Kerchove d’Exaerde, A.; Schurmans, S.; Schiffmann, S.N. Modulation of Ciliary Phosphoinositide Content Regulates Trafficking and Sonic Hedgehog Signaling Output. Dev. Cell 2015, 34, 338–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garcia-Gonzalo, F.R.; Phua, S.C.; Roberson, E.C.; Garcia, G., 3rd; Abedin, M.; Schurmans, S.; Inoue, T.; Reiter, J.F. Phosphoinositides Regulate Ciliary Protein Trafficking to Modulate Hedgehog Signaling. Dev. Cell 2015, 34, 400–409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prosseda, P.P.; Luo, N.; Wang, B.; Alvarado, J.A.; Hu, Y.; Sun, Y. Loss of OCRL increases ciliary PI(4,5)P2 in Lowe oculocerebrorenal syndrome. J. Cell Sci. 2017, 130, 3447–3454. [Google Scholar] [CrossRef] [Green Version]
- Dyson, J.M.; Conduit, S.E.; Feeney, S.J.; Hakim, S.; DiTommaso, T.; Fulcher, A.J.; Sriratana, A.; Ramm, G.; Horan, K.A.; Gurung, R.; et al. INPP5E regulates phosphoinositide-dependent cilia transition zone function. J. Cell Biol. 2017, 216, 247–263. [Google Scholar] [CrossRef]
- Marat, A.L.; Haucke, V. Phosphatidylinositol 3-phosphates-at the interface between cell signaling and membrane traffic. EMBO J. 2016, 35, 561–579. [Google Scholar] [CrossRef] [PubMed]
- Bernardino de la Serna, J.; Schutz, G.J.; Eggeling, C.; Cebecauer, M. There Is No Simple Model of the Plasma Membrane Organization. Front. Cell Dev. Biol. 2016, 4, 106. [Google Scholar] [CrossRef] [Green Version]
- DeMond, A.L.; Mossman, K.D.; Starr, T.; Dustin, M.L.; Groves, J.T. T cell receptor microcluster transport through molecular mazes reveals mechanism of translocation. Biophys. J. 2008, 94, 3286–3292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, J.; Wu, X.S.; Crites, T.; Hammer, J.A., 3rd. Actin retrograde flow and actomyosin II arc contraction drive receptor cluster dynamics at the immunological synapse in Jurkat T cells. Mol. Biol. Cell 2012, 23, 834–852. [Google Scholar] [CrossRef]
- Kaizuka, Y.; Douglass, A.D.; Varma, R.; Dustin, M.L.; Vale, R.D. Mechanisms for segregating T cell receptor and adhesion molecules during immunological synapse formation in Jurkat T cells. Proc. Natl. Acad. Sci. USA 2007, 104, 20296–20301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartman, N.C.; Nye, J.A.; Groves, J.T. Cluster size regulates protein sorting in the immunological synapse. Proc. Natl. Acad. Sci. USA 2009, 106, 12729–12734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trimble, W.S.; Grinstein, S. Barriers to the free diffusion of proteins and lipids in the plasma membrane. J. Cell Biol. 2015, 208, 259–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujiwara, T.; Ritchie, K.; Murakoshi, H.; Jacobson, K.; Kusumi, A. Phospholipids undergo hop diffusion in compartmentalized cell membrane. J. Cell Biol. 2002, 157, 1071–1081. [Google Scholar] [CrossRef] [Green Version]
- Ritchie, K.; Iino, R.; Fujiwara, T.; Murase, K.; Kusumi, A. The fence and picket structure of the plasma membrane of live cells as revealed by single molecule techniques (Review). Mol. Membr. Biol. 2003, 20, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Kusumi, A.; Ike, H.; Nakada, C.; Murase, K.; Fujiwara, T. Single-molecule tracking of membrane molecules: Plasma membrane compartmentalization and dynamic assembly of raft-philic signaling molecules. Semin. Immunol. 2005, 17, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, F.; Vogel, S.K.; Schwille, P. Lateral membrane diffusion modulated by a minimal actin cortex. Biophys. J. 2013, 104, 1465–1475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade, D.M.; Clausen, M.P.; Keller, J.; Mueller, V.; Wu, C.; Bear, J.E.; Hell, S.W.; Lagerholm, B.C.; Eggeling, C. Cortical actin networks induce spatio-temporal confinement of phospholipids in the plasma membrane--a minimally invasive investigation by STED-FCS. Sci. Rep. 2015, 5, 11454. [Google Scholar] [CrossRef] [Green Version]
- He, L.; Kooistra, R.; Das, R.; Oudejans, E.; van Leen, E.; Ziegler, J.; Portegies, S.; de Haan, B.; van Regteren Altena, A.; Stucchi, R.; et al. Cortical anchoring of the microtubule cytoskeleton is essential for neuron polarity. Elife 2020, 9, e55111. [Google Scholar] [CrossRef] [PubMed]
- Owen, D.M.; Oddos, S.; Kumar, S.; Davis, D.M.; Neil, M.A.; French, P.M.; Dustin, M.L.; Magee, A.I.; Cebecauer, M. High plasma membrane lipid order imaged at the immunological synapse periphery in live T cells. Mol. Membr. Biol. 2010, 27, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Janes, P.W.; Ley, S.C.; Magee, A.I. Aggregation of lipid rafts accompanies signaling via the T cell antigen receptor. J. Cell Biol. 1999, 147, 447–461. [Google Scholar] [CrossRef] [Green Version]
- Burack, W.R.; Lee, K.H.; Holdorf, A.D.; Dustin, M.L.; Shaw, A.S. Cutting edge: Quantitative imaging of raft accumulation in the immunological synapse. J. Immunol. 2002, 169, 2837–2841. [Google Scholar] [CrossRef] [PubMed] [Green Version]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia, E.; Ismail, S. Spatiotemporal Regulation of Signaling: Focus on T Cell Activation and the Immunological Synapse. Int. J. Mol. Sci. 2020, 21, 3283. https://doi.org/10.3390/ijms21093283
Garcia E, Ismail S. Spatiotemporal Regulation of Signaling: Focus on T Cell Activation and the Immunological Synapse. International Journal of Molecular Sciences. 2020; 21(9):3283. https://doi.org/10.3390/ijms21093283
Chicago/Turabian StyleGarcia, Esther, and Shehab Ismail. 2020. "Spatiotemporal Regulation of Signaling: Focus on T Cell Activation and the Immunological Synapse" International Journal of Molecular Sciences 21, no. 9: 3283. https://doi.org/10.3390/ijms21093283




