Coloured Rice Phenolic Extracts Increase Expression of Genes Associated with Insulin Secretion in Rat Pancreatic Insulinoma β-cells
Abstract
:1. Introduction
2. Results
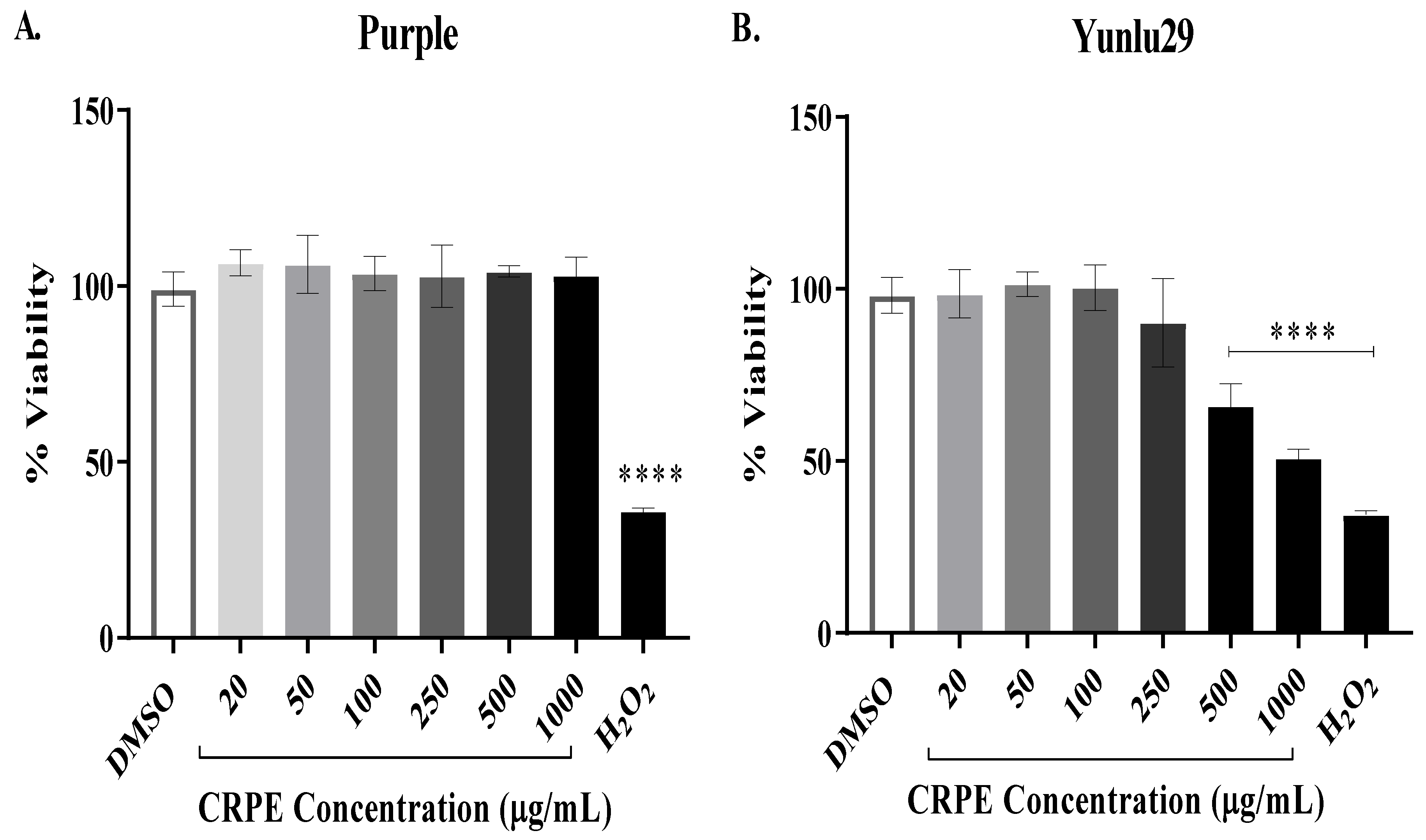
2.1. Cytotoxicity of Coloured-Rice Phenolic Extracts (CRPE) on INS-1E Cells
2.2. Coloured Rice Phenolic Extracts Modulate the Expression of Genes Associated with β-cell Function
2.3. Expression of Glucose Transporter 2 (Glut2) Gene
2.4. Expression of Silent Mating Type Information Regulation 2 Homolog 1 (Sirt1) Gene
2.5. Expression of Mitochondrial Transcription Factor A (Tfam) Gene
2.6. Expression of Pancreatic and Duodenal Homeobox-1 (Pdx-1) Gene
2.7. Expression of Insulin 1 (Ins1) Gene
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Coloured Rice Sampling and Phenolic Extraction
4.3. Cells and Cell Culture Conditions
4.4. Cytotoxicity Assay
4.5. Experimental Design and High Glucose Stress Induction
4.6. Ribonucleic Acid (RNA) Extraction
4.7. Complementary Deoxyribonucleic Acid (cDNA) Synthesis
4.8. Quantitative Polymerase Chain Reaction (qPCR)
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATP | Adenosine triphosphate |
| CRPE | Coloured rice phenolic extract |
| DMSO | Dimethyl sulfoxide |
| Glut2 | Glucose transporter 2 |
| GSIS | Glucose stimulated insulin secretion |
| HGSC | High glucose-induced stress condition |
| mtDNA | Mitochondrial DNA |
| NAD | Nicotinamide adenine dinucleotide |
| NF-kB | Nuclear factor kappa light chain enhancer of activated B cells |
| NGC | Normal glucose condition |
| Pdx-1 | Pancreatic and duodenal homeobox-1 |
| RNA | Ribonucleic acid |
| ROS | Reactive oxygen species |
| Sirt1 | Silent mating type information regulation 2 homolog 1 |
| T2DM | Type 2 diabetes mellitus |
| Tfam | Mitochondrial transcription factor A |
References
- Martin, C. The physiology of amylin and insulin: Maintaining the balance between glucose secretion and glucose uptake. Diabetes Educ. 2006, 32 (Suppl. 3), 101s. [Google Scholar] [CrossRef]
- Rösen, P.; Nawroth, P.P.; King, G.; Möller, W.; Tritschler, H.-J.; Packer, L. The role of oxidative stress in the onset and progression of diabetes and its complications: Asummary of a Congress Series sponsored byUNESCO-MCBN, the American Diabetes Association and the German Diabetes Society. Diabetes/Metab. Res. Rev. 2001, 17, 189. [Google Scholar] [CrossRef] [PubMed]
- Migdal, C.; Serres, M. Reactive oxygen species and oxidative stress. Med. Sci. 2011, 27, 405. [Google Scholar]
- Wiederkehr, A.; Wollheim, C.B. Minireview: Implication of mitochondria in insulin secretion and action. Endocrinology 2006, 147, 2643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ighodaro, O.M. Molecular pathways associated with oxidative stress in diabetes mellitus. Biomed. Pharmacother. 2018, 108, 656. [Google Scholar] [CrossRef]
- Rodgers, J.T.; Lerin, C.; Haas, W.; Gygi, S.P.; Spiegelman, B.M.; Puigserver, P. Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature 2005, 434, 113. [Google Scholar] [CrossRef]
- Luu, L.; Dai, F.F.; Prentice, K.J.; Huang, X.; Hardy, A.B.; Hansen, J.B.; Liu, Y.; Joseph, J.W.; Wheeler, M.B. The loss of Sirt1 in mouse pancreatic beta cells impairs insulin secretion by disrupting glucose sensing. Diabetologia 2013, 56, 2010. [Google Scholar] [CrossRef]
- Bartoli-Leonard, F.; Wilkinson, F.L.; Schiro, A.; Inglott, F.S.; Alexander, M.Y.; Weston, R. Suppression of SIRT1 in diabetic conditions induces osteogenic differentiation of human vascular smooth muscle cells via RUNX2 signalling. Sci. Rep. 2019, 9, 878. [Google Scholar] [CrossRef]
- Campbell, C.T.; Kolesar, J.E.; Kaufman, B.A. Mitochondrial transcription factor A regulates mitochondrial transcription initiation, DNA packaging, and genome copy number. Biochim. Biophys. Acta Gene Regul. Mech. 2012, 1819, 921. [Google Scholar] [CrossRef]
- Brun, T.; Li, N.; Jourdain, A.A.; Gaudet, P.; Duhamel, D.; Meyer, J.; Bosco, D.; Maechler, P. Diabetogenic milieus induce specific changes in mitochondrial transcriptome and differentiation of human pancreatic islets. Hum. Mol. Genet. 2015, 24, 5270. [Google Scholar] [CrossRef] [Green Version]
- Scarpulla, R.C. Transcriptional paradigms in mammalian mitochondrial biogenesis and function. Physiol. Rev. 2008, 88, 611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakrabarti, S.K.; James, J.C.; Mirmira, R.G. Quantitative assessment of gene targeting in vitroand in vivo by the pancreatic transcription factor, Pdx1 importance of chromatin structure in directing promoter binding. J. Biol. Chem. 2002, 277, 13286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahn, Y.; Xu, G.; Marselli, L.; Toschi, E.; Sharma, A.; Bonner-Weir, S.; Sgroi, D.; Weir, G. Changes in gene expression in beta cells after islet isolation and transplantation using laser-capture microdissection. Diabetologia 2007, 50, 334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roche, E.; Maestre, I.; Martin, F.; Fuentes, E.; Casero, J.; Reig, J.A.; Soria, B. Nutrient toxicity in pancreatic beta-cell dysfunction. .J Physiol. Biochem. 2000, 56, 119. [Google Scholar] [CrossRef]
- Kang, G.G.; Francis, N.; Hill, R.; Waters, D.; Blanchard, C.; Santhakumar, A.B. Dietary polyphenols and gene expression in molecular pathways associated with Type 2 diabetes mellitus: A Review. Int. J. Mol. Sci. 2020, 21, 140. [Google Scholar] [CrossRef] [Green Version]
- Dragan, S.; Andrica, F.; Serban, M.-C.; Timar, R. Polyphenols-rich natural products for treatment of diabetes. Curr. Med. Chem. 2015, 22, 14. [Google Scholar] [CrossRef]
- Callcott, E.T.; Blanchard, C.L.; Oli, P.; Santhakumar, A.B. Pigmented rice-derived phenolic compounds reduce biomarkers of oxidative stress and inflammation in human umbilical vein endothelial cells. Mol. Nutr. Food Res. 2018, 62, 1800840. [Google Scholar] [CrossRef]
- Watanabe, M. Effects of black rice containing anthocyanins on plasma and hepatic parameters in type 2 diabetic db/db mice. Food Sci. Technol. Res. 2016, 22, 719. [Google Scholar] [CrossRef] [Green Version]
- Imam, M.U.; Musa, S.N.A.; Azmi, N.H.; Ismail, M. Effects of white rice, brown rice and germinated brown rice on antioxidant status of type 2 diabetic rats. Int. J. Mol. Sci. 2012, 13. [Google Scholar] [CrossRef]
- Vetterli, L.; Brun, T.; Giovannoni, L.; Bosco, D.; Maechler, P. Resveratrol potentiates glucose-stimulated insulin secretion in INS-1E beta-cells and human islets through a SIRT1-dependent mechanism. J. Biol. Chem. 2011, 286, 6049. [Google Scholar] [CrossRef] [Green Version]
- Cai, E.P.; Lin, J.-K. Epigallocatechin Gallate (EGCG) and rutin suppress the glucotoxicity through activating IRS2 and AMPK signaling in rat pancreatic β cells. J. Agric. Food Chem. 2009, 57, 9817. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Oksbjerg, N.; Young, J.F.; Jeppesen, P.B. Caffeic acid, naringenin and quercetin enhance glucose-stimulated insulin secretion and glucose sensitivity in INS-1E cells. Diabetes Obes. Metab. 2014, 16, 602. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.D.; Zhang, B.; Zhang, J.K.; Xu, C.J.; Wu, Y.L.; Li, X.; Chen, K.S. Cyanidin-3-glucoside-rich extract from Chinese bayberry fruit protects pancreatic beta cells and ameliorates hyperglycemia in streptozotocin-induced diabetic mice. J. Med. Food 2012, 15, 288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sameermahmood, Z.; Raji, L.; Saravanan, T.; Vaidya, A.; Mohan, V.; Balasubramanyam, M. Gallic acid protects RINm5F β-cells from glucolipotoxicity by its antiapoptotic and insulin-secretagogue actions. Phytother. Res. 2010, 24, S83. [Google Scholar] [CrossRef]
- Rowley, T.J.; Bitner, B.F.; Ray, J.D.; Lathen, D.R.; Smithson, A.T.; Dallon, B.W.; Plowman, C.J.; Bikman, B.T.; Hansen, J.M.; Dorenkott, M.R.; et al. Monomeric cocoa catechins enhance β-cell function by increasing mitochondrial respiration. J. Nutr. Biochem. 2017, 49, 30. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, J.; Yang, L.; Chen, R.; Yang, R.; Zhang, H.; Cai, D.; Chen, H. The cytotoxic role of intermittent high glucose on apoptosis and cell viability in pancreatic beta cells. J. Diabetes Res. 2014, 2014, 712781. [Google Scholar] [CrossRef]
- Rao, S.; Callcott, E.T.; Santhakumar, A.B.; Chinkwo, K.A.; Vanniasinkam, T.; Luo, J.; Blanchard, C.L. Profiling polyphenol composition and antioxidant activity in Australian-grown rice using UHPLC Online-ABTS system. J. Cereal Sci. 2018, 80, 174. [Google Scholar] [CrossRef]
- Chen, K.; Zhou, Y.-X.; Li, K.; Qi, L.-X.; Zhang, Q.-F.; Wang, M.-C.; Xiao, J.-H. A novel three-round multiplex PCR for SNP genotyping with next generation sequencing. Anal. Bioanal. Chem. 2016, 408, 4371. [Google Scholar] [CrossRef]
- Mennen, L.I.; Walker, R.; Bennetau-Pelissero, C.; Scalbert, A. Risks and safety of polyphenol consumption. Am. J. Clin. Nutr. 2005, 81, 326S. [Google Scholar] [CrossRef] [Green Version]
- Callcott, E.T.; Blanchard, C.L.; Snell, P.; Santhakumar, A.B. The anti-inflammatory and antioxidant effects of pigmented rice consumption in an obese cohort. Food Funct. 2019, 10, 8016. [Google Scholar] [CrossRef]
- Suantawee, T.; Elazab, S.T.; Hsu, W.H.; Yao, S.; Cheng, H.; Adisakwattana, S. Cyanidin Stimulates Insulin Secretion and Pancreatic β-Cell Gene Expression through Activation of l-type Voltage-Dependent Ca(2+) Channels. Nutrients 2017, 9, 814. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, A.; Chinnaiyan, M.; Karundevi, B. Ferulic acid regulates hepatic GLUT2 gene expression in high fat and fructose-induced type-2 diabetic adult male rat. Eur. J. Pharmacol. 2015, 761, 391. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Baldwin, L.A. The hormetic dose-response model is more common than the threshold model in toxicology. Toxicol. Sci. 2003, 71, 246. [Google Scholar] [CrossRef] [PubMed]
- Renaud, J.; Martinoli, M.-G. Considerations for the use of polyphenols as therapies in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 1883. [Google Scholar] [CrossRef] [Green Version]
- Aragonès, G.; Suárez, M.; Ardid-Ruiz, A.; Vinaixa, M.; Rodríguez, M.A.; Correig, X.; Arola, L.; Bladé, C. Dietary proanthocyanidins boost hepatic NAD+ metabolism and SIRT1 expression and activity in a dose-dependent manner in healthy rats. Sci. Rep. 2016, 6, 24977. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.-J.; Ryu, G.R.; Chung, J.-S.; Sim, S.S.; Min, D.S.; Rhie, D.-J.; Yoon, S.H.; Hahn, S.J.; Kim, M.-S.; Jo, Y.-H. Protective effects of epicatechin against the toxic effects of streptozotocin on rat pancreatic islets: in vivo and in vitro. Pancreas 2003, 26, 292. [Google Scholar] [CrossRef]
- Bernatoniene, J.; Kopustinskiene, D.M. The role of catechins in cellular responses to oxidative stress. Molecules 2018, 23, 965. [Google Scholar] [CrossRef] [Green Version]
- Cao, M.-M.; Lu, X.; Liu, G.-D.; Su, Y.; Li, Y.-B.; Zhou, J. Resveratrol attenuates type 2 diabetes mellitus by mediating mitochondrial biogenesis and lipid metabolism via Sirtuin type 1. Exp. Ther. Med. 2018, 15, 576. [Google Scholar] [CrossRef]
- Rutanen, J.; Yaluri, N.; Modi, S.; Pihlajamäki, J.; Vänttinen, M.; Itkonen, P.; Kainulainen, S.; Yamamoto, H.; Lagouge, M.; Sinclair, D.A.; et al. SIRT1 mRNA expression may be associated with energy expenditure and insulin sensitivity. Diabetes 2010, 59, 829. [Google Scholar] [CrossRef] [Green Version]
- Gauthier, B.R.; Wiederkehr, A.; Baquié, M.; Dai, C.; Powers, A.C.; Kerr-Conte, J.; Pattou, F.; MacDonald, R.J.; Ferrer, J.; Wollheim, C.B. PDX1 Deficiency Causes Mitochondrial Dysfunction and Defective Insulin Secretion through TFAM Suppression. Cell Metab. 2009, 10, 110. [Google Scholar] [CrossRef] [Green Version]
- Puddu, A.; Sanguineti, R.; Mach, F.; Dallegri, F.; Viviani, G.L.; Montecucco, F. Update on the protective molecular pathways improving pancreatic beta-cell dysfunction. Mediat. Inflamm. 2013, 2013, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luna-Vital, D.A.; Gonzalez de Mejia, E. Anthocyanins from purple corn activate free fatty acid-receptor 1 and glucokinase enhancing in vitro insulin secretion and hepatic glucose uptake. PLoS ONE 2018, 13, e0200449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, E.H.; Sung, R.K.; Hwang, I.K.; Tae, Y.H. Hypoglycemic effects of a phenolic acid fraction of rice bran and ferulic acid in C57BL/KsJ-db/db mice. J. Agric. Food Chem. 2007, 55, 9800. [Google Scholar] [CrossRef] [PubMed]
- Rugina, D.; Diaconeasa, Z.; Coman, C.; Bunea, A.; Socaciu, C.; Pintea, A. Chokeberry anthocyanin extract as pancreatic b-Cell protectors in two models of induced oxidative stress. Oxid. Med. Cell. Longev. 2015, 2015, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jayaprakasam, B.; Vareed, S.K.; Olson, L.K.; Nair, M.G. Insulin secretion by bioactive anthocyanins and anthocyanidins present in fruits. J. Agric. Food Chem. 2005, 53, 28. [Google Scholar] [CrossRef]
- Johnson, M.H.; de Mejia, E.G. phenolic compounds from fermented berry beverages modulated gene and protein expression to increase insulin secretion from pancreatic β-cells in vitro. J. Agric. Food Chem. 2016, 64, 2569. [Google Scholar] [CrossRef]
- Francis, N.; Rao, S.; Blanchard, C.; Santhakumar, A.J.M. Black sorghum phenolic extract regulates expression of genes associated with oxidative stress and inflammation in human endothelial cells. Molecules 2019, 24, 3321. [Google Scholar] [CrossRef] [Green Version]
- Bei, W.; Wang, Y.; Chen, J.; Zhang, J.; Wang, L.; Gu, Z.; Hu, Y.; Huang, Y.; Xu, W.; Lei, Z.; et al. Chinese medicine FTZ recipe protects against high-glucose-induced beta cell injury through alleviating oxidative stress. J. Evid. Based Complement. Altern. Med. 2019, 2019, 14. [Google Scholar] [CrossRef] [Green Version]
- Hohmeier, H.E.; Mulder, H.; Chen, G.; Henkel-Rieger, R.; Prentki, M.; Newgard, C.B. Isolation of INS-1-derived cell lines with robust ATP-sensitive K+ channel-dependent and -independent glucose-stimulated insulin secretion. Diabetes 2000, 49, 424. [Google Scholar] [CrossRef] [Green Version]
- Muller, P.Y.; Janovjak, H.; Miserez, A.R.; Dobbie, Z. Processing of gene expression data generated by quantitative real-time RT-PCR. Biotechniques 2002, 32, 1372. [Google Scholar]





| Genes | Origin | Sense (5’-3’) | Antisense (3’-5’) |
|---|---|---|---|
| Glut2 | Rat | TCAGCCAGCCTGTGTATGCA | TCCACAAGCAGCACAGAGACA |
| Sirt1 | Rat | CAGTGTCATGGTTCCTTTGC | CACCGAGGAACTACCTGA T |
| Tfam | Rat | GGGAAGAGCAAATGGCTGAA | TCACACTGCGACGGATGA GA |
| Pdx-1 | Rat | CCGCGTTCATCTCCCTTT C | CTCCTGCCCACTGGCTTT T |
| Ins1 | Rat | TGCTCACCCGCGACCTT | GTTCATATGCACCACTGGACTGAA |
| TfIIb | Rat | GTTCTGCTCCAACCTTTGCCT | TGTGTAGCTGCCATCTGCACT T |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, G.G.; Francis, N.; Hill, R.; LE Waters, D.; Blanchard, C.L.; Santhakumar, A.B. Coloured Rice Phenolic Extracts Increase Expression of Genes Associated with Insulin Secretion in Rat Pancreatic Insulinoma β-cells. Int. J. Mol. Sci. 2020, 21, 3314. https://doi.org/10.3390/ijms21093314
Kang GG, Francis N, Hill R, LE Waters D, Blanchard CL, Santhakumar AB. Coloured Rice Phenolic Extracts Increase Expression of Genes Associated with Insulin Secretion in Rat Pancreatic Insulinoma β-cells. International Journal of Molecular Sciences. 2020; 21(9):3314. https://doi.org/10.3390/ijms21093314
Chicago/Turabian StyleKang, Gideon Gatluak, Nidhish Francis, Rodney Hill, Daniel LE Waters, Christopher L. Blanchard, and Abishek Bommannan Santhakumar. 2020. "Coloured Rice Phenolic Extracts Increase Expression of Genes Associated with Insulin Secretion in Rat Pancreatic Insulinoma β-cells" International Journal of Molecular Sciences 21, no. 9: 3314. https://doi.org/10.3390/ijms21093314






