The Role of Nuclear Factor of Activated T Cells 5 in Hyperosmotic Stress-Exposed Human Lens Epithelial Cells
Abstract
1. Introduction
2. Results
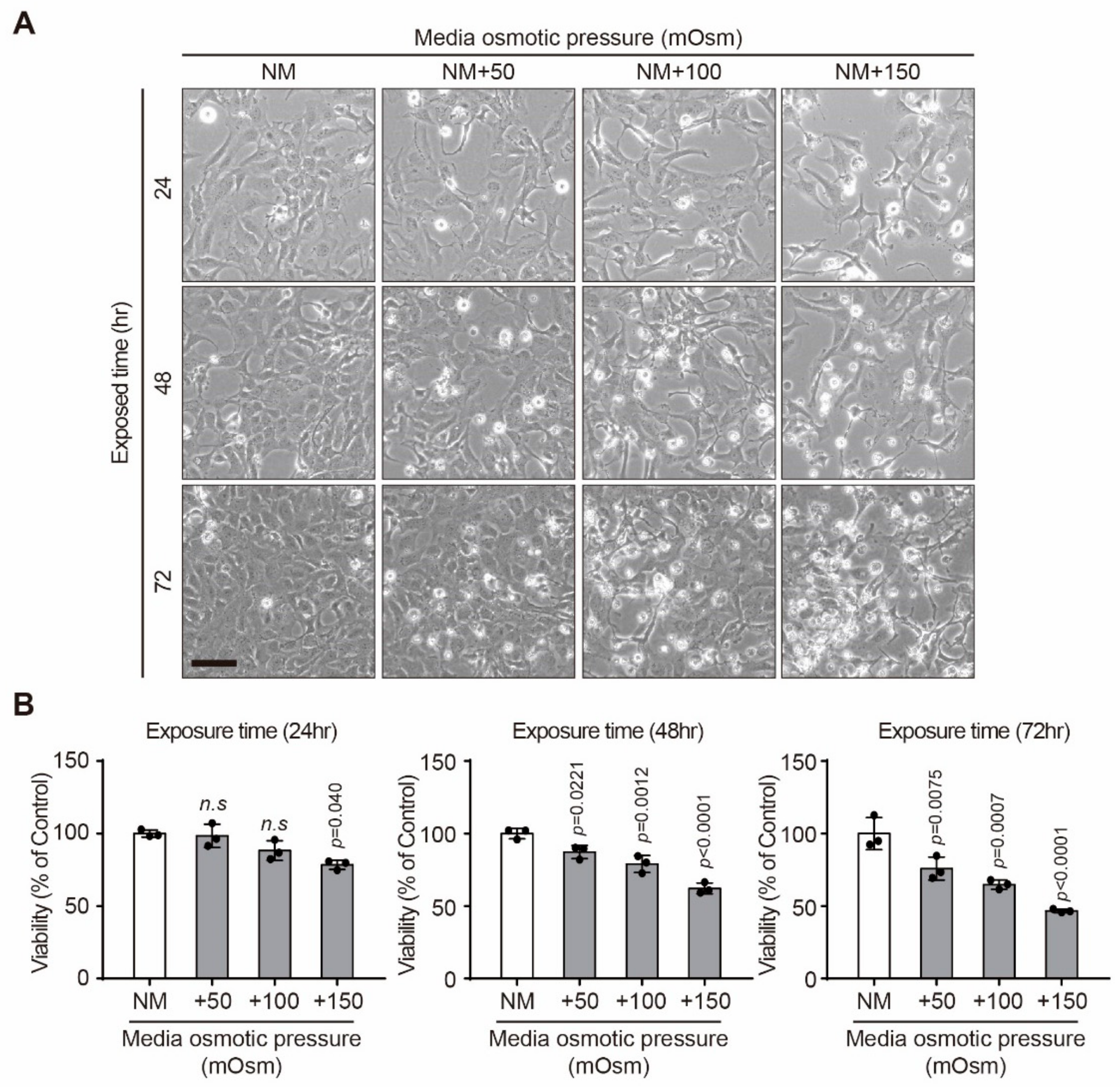
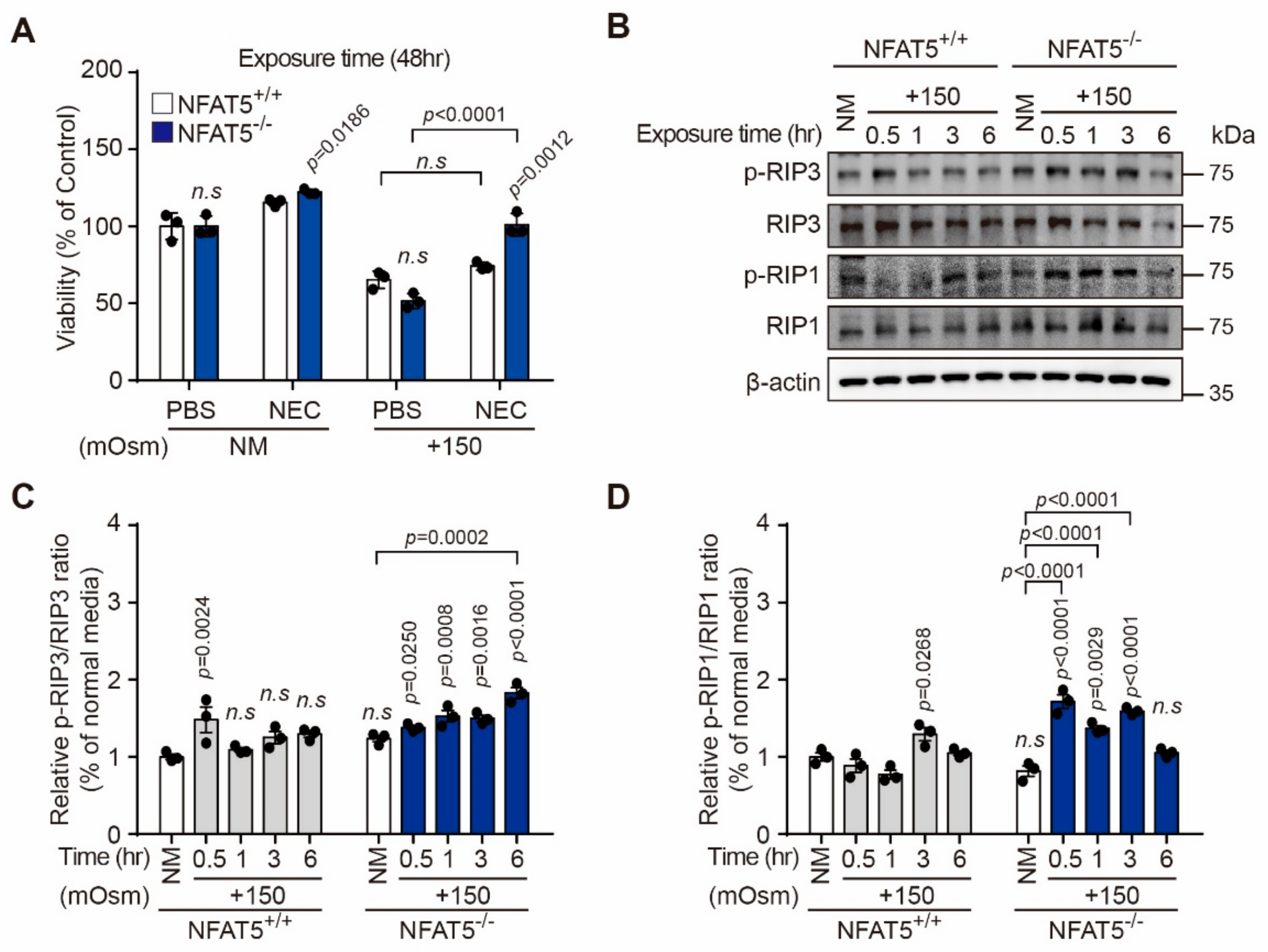
2.1. Effect of Hyperosmotic Stress on the Viability of Human Lens Epithelial B-3 (HLE-B3) Cells
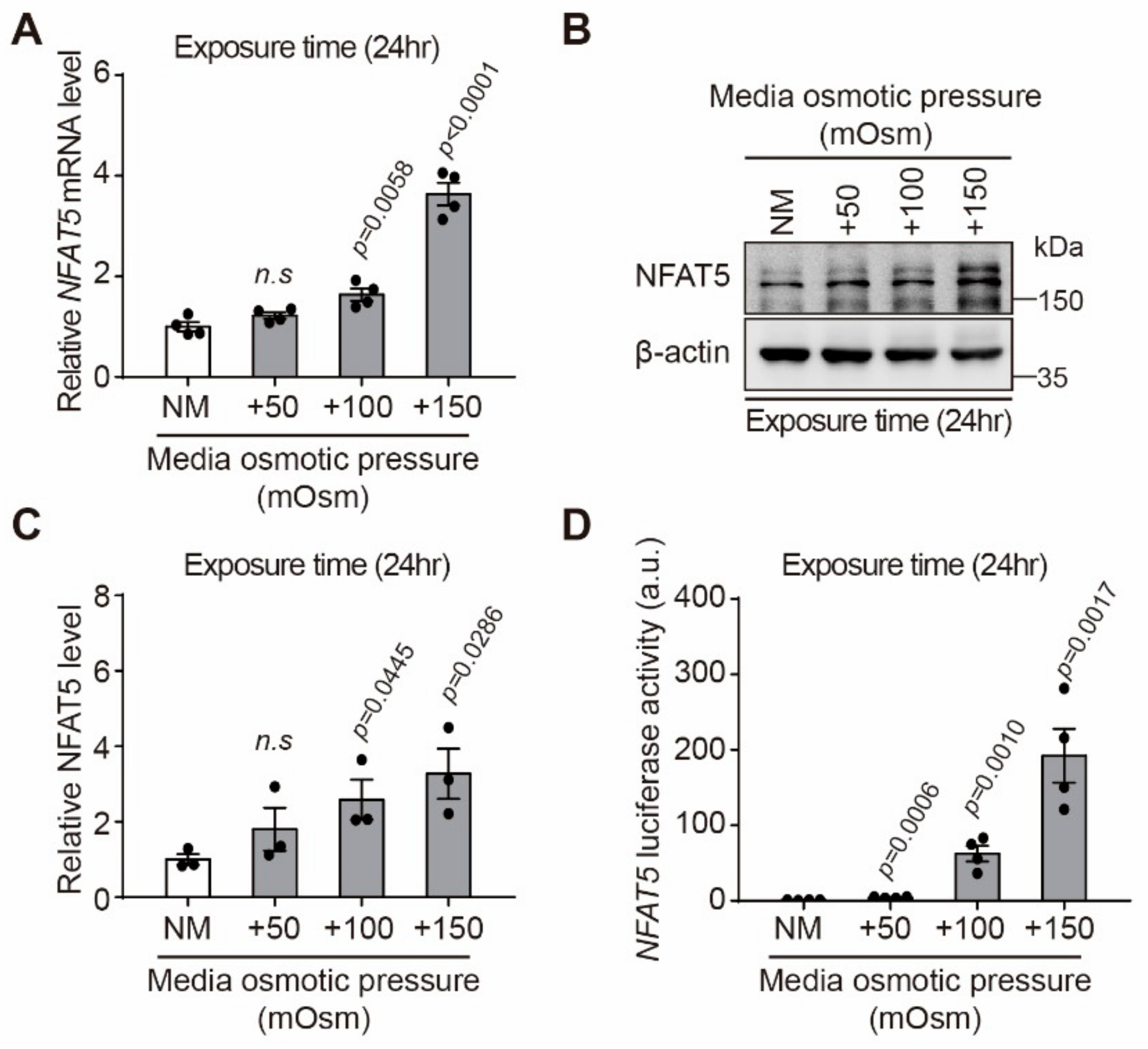
2.2. Effect of Hyperosmotic Stress on Expression of NFAT5 in HLE-B3 Cells
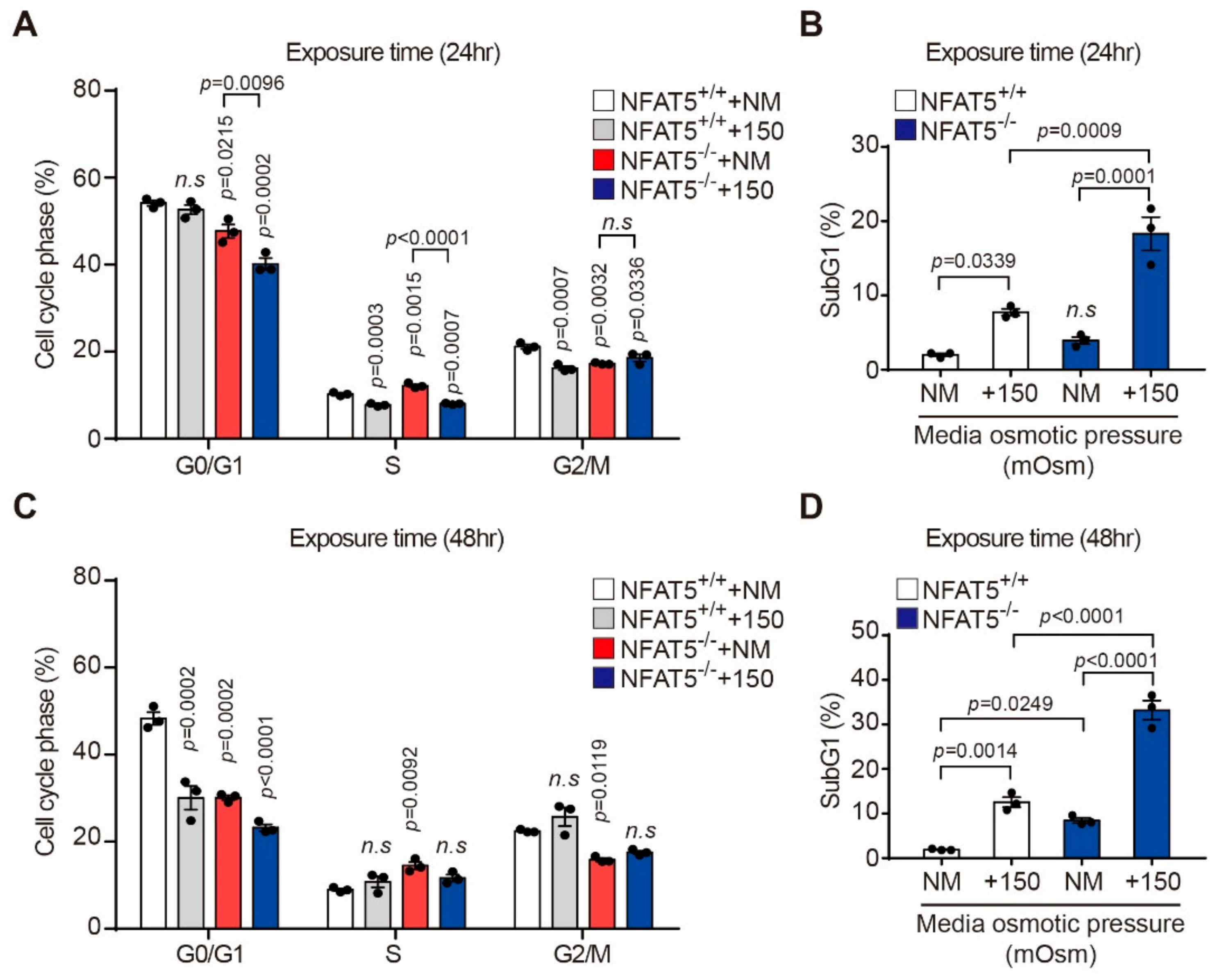
2.3. NFAT5 Knockout Cell Production and Changes of Cell Viability after Hyperosmotic Stress in HLE-B3 Cells
2.4. Effect of Hyperosmotic Stress on the Cell Cycle in HLE-B3 Cells
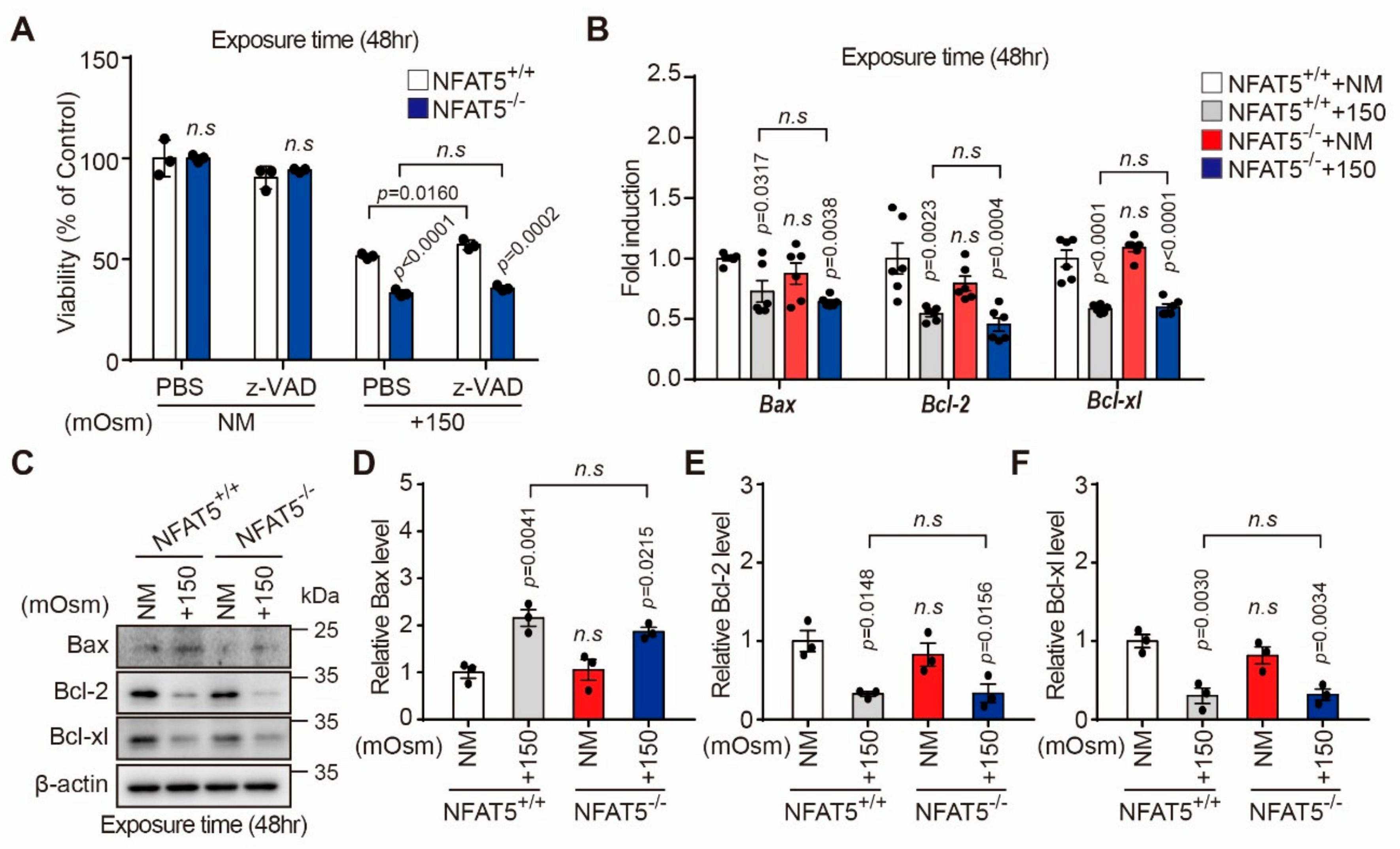
2.5. Characterization of Hyperosmotic Stress-Induced Death of Lens Epithelial Cells
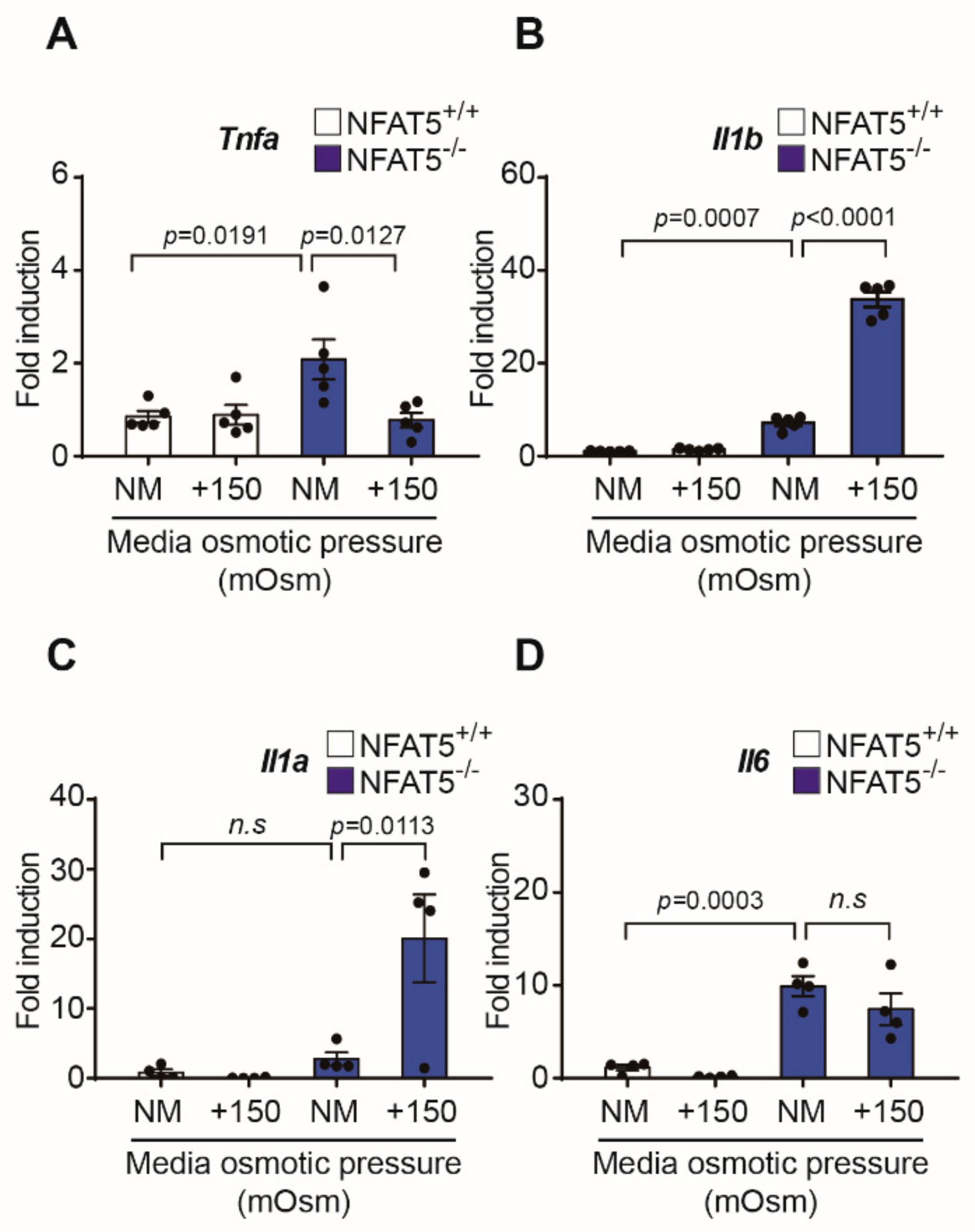
2.6. Changes in Inflammatory Cytokines by qPCR after Exposure to Hypertonic Conditions
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Treatment
4.2. Measurement of Cell Viability
4.3. Flow Cytometric Analysis
4.4. Transfection and Luciferase Assays
4.5. Generation of the NFAT5 KO Cell Line
4.6. Quantitative Real-Time PCR
4.7. Western Blot Analysis
4.8. DCFDA/H2DCFDA Cellular ROS Assay
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Leske, M.C.; Sperduto, R.D. The epidemiology of senile cataracts: A review. Am. J. Epidemiol. 1983, 118, 152–165. [Google Scholar] [CrossRef] [PubMed]
- Ghanavati, M.; Behrooz, M.; Rashidkhani, B.; Ashtray-Larky, D.; Zameni, S.D.; Alipour, M. Healthy Eating Index in Patients with Cataract: A Case-Control Study. Iran. Red Crescent Med. J. 2015, 17, e22490. [Google Scholar] [CrossRef] [PubMed]
- Tavani, A.; Negri, E.; La Vecchia, C. Food and nutrient intake and risk of cataract. Ann. Epidemiol. 1996, 6, 41–46. [Google Scholar] [CrossRef]
- Cumming, R.G.; Mitchell, P.; Smith, W. Dietary sodium intake and cataract: The Blue Mountains Eye Study. Am. J. Epidemiol. 2000, 151, 624–626. [Google Scholar] [CrossRef]
- Bae, J.H.; Shin, D.S.; Lee, S.C.; Hwang, I.C. Sodium Intake and Socioeconomic Status as Risk Factors for Development of Age-Related Cataracts: The Korea National Health and Nutrition Examination Survey. PLoS ONE 2015, 10, e0136218. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Sargent, C.; Berrios, G.; Irrizarry, J.E.; Estape, E.S.; Cangiano, J.L.; Martinez-Maldonado, M. Prevention and reversal of cataracts in genetically hypertensive rats through sodium restriction. Investig. Ophthalmol. Vis. Sci. 1989, 30, 2356–2360. [Google Scholar]
- Appel, L.J.; Brands, M.W.; Daniels, S.R.; Karanja, N.; Elmer, P.J.; Sacks, F.M.; American Heart, A. Dietary approaches to prevent and treat hypertension: A scientific statement from the American Heart Association. Hypertension 2006, 47, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Stookey, J.D.; Pieper, C.F.; Cohen, H.J. Is the prevalence of dehydration among community-dwelling older adults really low? Informing current debate over the fluid recommendation for adults aged 70+years. Public Health Nutr. 2005, 8, 1275–1285. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, N.B. Dehydration in the elderly: Insidious and manageable. Geriatrics 1991, 46, 35–38. [Google Scholar]
- Matz, R. Dehydration in older adults. JAMA 1996, 275, 911–912. [Google Scholar] [CrossRef]
- O’Neill, P.A.; Davies, I.; Wears, R.; Barrett, J.A. Elderly female patients in continuing care: Why are they hyperosmolar? Gerontology 1989, 35, 205–209. [Google Scholar] [CrossRef]
- Aramburu, J.; Drews-Elger, K.; Estrada-Gelonch, A.; Minguillon, J.; Morancho, B.; Santiago, V.; Lopez-Rodriguez, C. Regulation of the hypertonic stress response and other cellular functions by the Rel-like transcription factor NFAT5. Biochem. Pharmacol. 2006, 72, 1597–1604. [Google Scholar] [CrossRef] [PubMed]
- Ko, B.C.; Lam, A.K.; Kapus, A.; Fan, L.; Chung, S.K.; Chung, S.S. Fyn and p38 signaling are both required for maximal hypertonic activation of the osmotic response element-binding protein/tonicity-responsive enhancer-binding protein (OREBP/TonEBP). J. Biol. Chem. 2002, 277, 46085–46092. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rodriguez, C.; Aramburu, J.; Jin, L.; Rakeman, A.S.; Michino, M.; Rao, A. Bridging the NFAT and NF-kappaB families: NFAT5 dimerization regulates cytokine gene transcription in response to osmotic stress. Immunity 2001, 15, 47–58. [Google Scholar] [PubMed]
- Li, D.Q.; Luo, L.; Chen, Z.; Kim, H.S.; Song, X.J.; Pflugfelder, S.C. JNK and ERK MAP kinases mediate induction of IL-1beta, TNF-alpha and IL-8 following hyperosmolar stress in human limbal epithelial cells. Exp. Eye Res. 2006, 82, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Maeno, E.; Ishizaki, Y.; Kanaseki, T.; Hazama, A.; Okada, Y. Normotonic cell shrinkage because of disordered volume regulation is an early prerequisite to apoptosis. Proc. Natl. Acad. Sci. USA 2000, 97, 9487–9492. [Google Scholar] [CrossRef]
- Cheung, C.Y.; Ko, B.C. NFAT5 in cellular adaptation to hypertonic stress—regulations and functional significance. J. Mol. Signal. 2013, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.N. Intracellular water homeostasis and the mammalian cellular osmotic stress response. J. Cell Physiol. 2006, 206, 9–15. [Google Scholar] [CrossRef]
- Neuhofer, W. Role of NFAT5 in inflammatory disorders associated with osmotic stress. Curr. Genom. 2010, 11, 584–590. [Google Scholar] [CrossRef]
- Alfieri, R.R.; Petronini, P.G. Hyperosmotic stress response: Comparison with other cellular stresses. Pflug. Archiv. Eur. J. Physiol. 2007, 454, 173–185. [Google Scholar] [CrossRef]
- Burg, M.B.; Ferraris, J.D.; Dmitrieva, N.I. Cellular response to hyperosmotic stresses. Physiol. Rev. 2007, 87, 1441–1474. [Google Scholar] [CrossRef] [PubMed]
- Pflugfelder, S.C. Tear dysfunction and the cornea: LXVIII Edward Jackson Memorial Lecture. Am. J. Ophthalmol. 2011, 152, 900–909.e1. [Google Scholar] [CrossRef] [PubMed]
- Libert, S.; Willermain, F.; Weber, C.; Bryla, A.; Salik, D.; Gregoire, F.; Bolaky, N.; Caspers, L.; Perret, J.; Delporte, C. Involvement of TonEBP/NFAT5 in osmoadaptative response of human retinal pigmented epithelial cells to hyperosmolar stress. Mol. Vis. 2016, 22, 100–115. [Google Scholar]
- Andley, U.P.; Hebert, J.S.; Morrison, A.R.; Reddan, J.R.; Pentland, A.P. Modulation of lens epithelial cell proliferation by enhanced prostaglandin synthesis after UVB exposure. Investig. Ophthalmol. Vis. Sci. 1994, 35, 374–381. [Google Scholar]
- Hightower, K.R.; Reddan, J.R.; McCready, J.P.; Dziedzic, D.C. Lens epithelium: A primary target of UVB irradiation. Exp. Eye Res. 1994, 59, 557–564. [Google Scholar] [CrossRef]
- Hightower, K.R. The role of the lens epithelium in development of UV cataract. Curr. Eye Res. 1995, 14, 71–78. [Google Scholar] [CrossRef]
- Li, W.C.; Spector, A. Lens epithelial cell apoptosis is an early event in the development of UVB-induced cataract. Free Radic. Biol. Med. 1996, 20, 301–311. [Google Scholar] [CrossRef]
- Halterman, J.A.; Kwon, H.M.; Wamhoff, B.R. Tonicity-independent regulation of the osmosensitive transcription factor TonEBP (NFAT5). Am. J. Physiol. Cell Physiol. 2012, 302, C1–C8. [Google Scholar] [CrossRef] [PubMed]
- Miyakawa, H.; Woo, S.K.; Dahl, S.C.; Handler, J.S.; Kwon, H.M. Tonicity-responsive enhancer binding protein, a rel-like protein that stimulates transcription in response to hypertonicity. Proc. Natl. Acad. Sci. USA 1999, 96, 2538–2542. [Google Scholar] [CrossRef] [PubMed]
- Vandenabeele, P.; Galluzzi, L.; Vanden Berghe, T.; Kroemer, G. Molecular mechanisms of necroptosis: An ordered cellular explosion. Nat. Rev. Mol. Cell Biol. 2010, 11, 700–714. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Sun, L.; Su, L.; Rizo, J.; Liu, L.; Wang, L.F.; Wang, F.S.; Wang, X. Mixed lineage kinase domain-like protein MLKL causes necrotic membrane disruption upon phosphorylation by RIP3. Mol. Cell 2014, 54, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Robinson, K.R.; Patterson, J.W. Localization of steady currents in the lens. Curr. Eye Res. 1982, 2, 843–847. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.B.; Rajagopala, M.; Ravishankar, B. Etiopathogenesis of cataract: An appraisal. Indian J. Ophthalmol. 2014, 62, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Osnes-Ringen, O.; Berg, K.H.; Moe, M.C.; Zetterstrom, C.; Roger, M.; Nicolaissen, B. Cell death pattern in lens epithelium of cataract patients. Acta Ophthalmol. 2016, 94, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Charakidas, A.; Kalogeraki, A.; Tsilimbaris, M.; Koukoulomatis, P.; Brouzas, D.; Delides, G. Lens epithelial apoptosis and cell proliferation in human age-related cortical cataract. Eur. J. Ophthalmol. 2005, 15, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, M.; Im, Y.S.; Choi, W.; Byeon, S.H.; Lee, H.K. NFAT5 induction and its role in hyperosmolar stressed human limbal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1827–1835. [Google Scholar] [CrossRef] [PubMed]
- Arsenijevic, T.; Vujovic, A.; Libert, F.; Op de Beeck, A.; Hebrant, A.; Janssens, S.; Gregoire, F.; Lefort, A.; Bolaky, N.; Perret, J.; et al. Hyperosmotic stress induces cell cycle arrest in retinal pigmented epithelial cells. Cell Death Dis. 2013, 4, e662. [Google Scholar] [CrossRef]
- Wang, L.; Dai, W.; Lu, L. Osmotic stress-induced phosphorylation of H2AX by polo-like kinase 3 affects cell cycle progression in human corneal epithelial cells. J. Biol. Chem. 2014, 289, 29827–29835. [Google Scholar] [CrossRef]
- Chan, F.K.; Luz, N.F.; Moriwaki, K. Programmed necrosis in the cross talk of cell death and inflammation. Annu. Rev. Immunol. 2015, 33, 79–106. [Google Scholar] [CrossRef]
- Kimberly, M.B.; Jake, D.C.; Nicholas, A.S.; Julianna, M.B.; Mohammed, A.; Kara, L.L.; Jonathan, M.F.; Kira, L.L.; Elliot, C.; Deanna, M.S.; et al. Blowing epithelial bubbles with GumB:shlA-family pore-forming toxins induce blebbing and rapid cellular death in corneal epithelial cells. PLoS Pathog. 2019, 16, e1007825. [Google Scholar]
- Yani, L.; Miaomiao, Y.; Tingjun, F. Insights in to mechanisms of pranoprofen-induced apoptosis and necroptosis in human corneal stromal cells. Toxicol. Lett. 2020, 320, 9–18. [Google Scholar]
- Jing, J.P.; Wei, T.S.; Fei, Y.; Xuan, Z.; Jun, P.; Xiu, J.L.; Xiao, B.X. Involvement of regulated nacrosis in blinding diseases: Focus on necroptosis and ferroptosis. Exp. Eye Res. 2020, 191, 107922. [Google Scholar]
- Roth, I.; Leproy, V.; Kwon, H.M.; Martin, P.Y.; Feraille, E.; Hasler, U. Osmoprotective transcription factor NFAT5/TonEBP modulates nuclear factor-kappaB activity. Mol. Biol. Cell 2010, 21, 3459–3474. [Google Scholar] [CrossRef] [PubMed]
- Blanchett, S.; Boal-Carvalho, I.; Layzell, S.; Seddon, B. NF-κB and extrinsic cell death pathways-Entwined Do-or-Die decisions for T cells. Trends Immunol. 2021, 42, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; He, Y.; Wang, L.; Mo, C.; Zhang, W.; Li, J.; Liao, Z.; Tang, X.; Xiao, H. D-galactose induces necroptotic cell death in neuroblastoma cell lines. J. Cell Biochem. 2011, 112, 3834–3844. [Google Scholar] [CrossRef] [PubMed]

| Gene | Primer | Sequences (5′–3′) | |
|---|---|---|---|
| 1 | Nfat5 | Sense | GACCAUGGUCCAAAUGCAA |
| Antisense | UUGCAUUUGGACCAUGGUC | ||
| 2 | Nfat5 | Sense | CUGGAUAACAGUCGGAUGU |
| Antisense | ACAUCCGACUGUUAUCCAG |
| Gene | Primer | Sequences (5′–3′) | |
|---|---|---|---|
| 1 | Bcl-2 | Forward | CAT GCG GCC TCT GTT TGA TT |
| Reverse | TCACTTGTGGCCCAGATAGG | ||
| 2 | Bcl-x1 | Forward | GGT CGC ATT GTG GCC TTT TT |
| Reverse | CGT CGA TCC GAC TCA CCA AT | ||
| 3 | Bax | Forward | TGA GCA GAT CAT GAA GAC AGG G |
| Reverse | TTG AGA CAC TCG CTC AGC TT | ||
| 4 | tnf-α | Forward | CCCTCACACTCAGATCATCTTCT |
| Reverse | GCTACGACGTGGGCTACAG | ||
| 5 | il-1α | Forward | GCACCTTACACCTACCAGAGT |
| Reverse | AAACTTCTGCCTGACGAGCTT | ||
| 6 | il-1β | Forward | GCTCGCCAGTGAAATGATGG |
| Reverse | GTCCTGGAAGGAGCACTTCAT | ||
| 7 | il-6 | Forward | TAGTCCTTCCTACCCCAATTTCC |
| Reverse | TTGGTCCTTAGCCACTCCTTC | ||
| 8 | Nfat1 | Forward | AGAATCCATCCTGCTGGTTC |
| Reverse | TCCATGTAGCCATGGAGCTG | ||
| 9 | Nfat2 | Forward | TCATTGACTGTGCCGGAATC |
| Reverse | AAGTTGTGGCCAGACAGGAC | ||
| 10 | Nfat3 | Forward | AGAACTGGACTCAGAGGATG |
| Reverse | ATGGAGGTGATGCGGATG | ||
| 11 | Nfat5 | Forward | TACCTCAGTCACCGACAGCAAG |
| Reverse | CGACTGTTATCCAGCAAGTC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, G.-N.; Hah, Y.-S.; Seong, H.; Yoo, W.-S.; Choi, M.-Y.; Cho, H.-Y.; Yun, S.P.; Kim, S.-J. The Role of Nuclear Factor of Activated T Cells 5 in Hyperosmotic Stress-Exposed Human Lens Epithelial Cells. Int. J. Mol. Sci. 2021, 22, 6296. https://doi.org/10.3390/ijms22126296
Kim G-N, Hah Y-S, Seong H, Yoo W-S, Choi M-Y, Cho H-Y, Yun SP, Kim S-J. The Role of Nuclear Factor of Activated T Cells 5 in Hyperosmotic Stress-Exposed Human Lens Epithelial Cells. International Journal of Molecular Sciences. 2021; 22(12):6296. https://doi.org/10.3390/ijms22126296
Chicago/Turabian StyleKim, Gyu-Nam, Young-Sool Hah, Hyemin Seong, Woong-Sun Yoo, Mee-Young Choi, Hee-Young Cho, Seung Pil Yun, and Seong-Jae Kim. 2021. "The Role of Nuclear Factor of Activated T Cells 5 in Hyperosmotic Stress-Exposed Human Lens Epithelial Cells" International Journal of Molecular Sciences 22, no. 12: 6296. https://doi.org/10.3390/ijms22126296
APA StyleKim, G.-N., Hah, Y.-S., Seong, H., Yoo, W.-S., Choi, M.-Y., Cho, H.-Y., Yun, S. P., & Kim, S.-J. (2021). The Role of Nuclear Factor of Activated T Cells 5 in Hyperosmotic Stress-Exposed Human Lens Epithelial Cells. International Journal of Molecular Sciences, 22(12), 6296. https://doi.org/10.3390/ijms22126296







