Early Changes in Crayfish Hemocyte Proteins after Injection with a β-1,3-glucan, Compared to Saline Injected and Naive Animals
Abstract
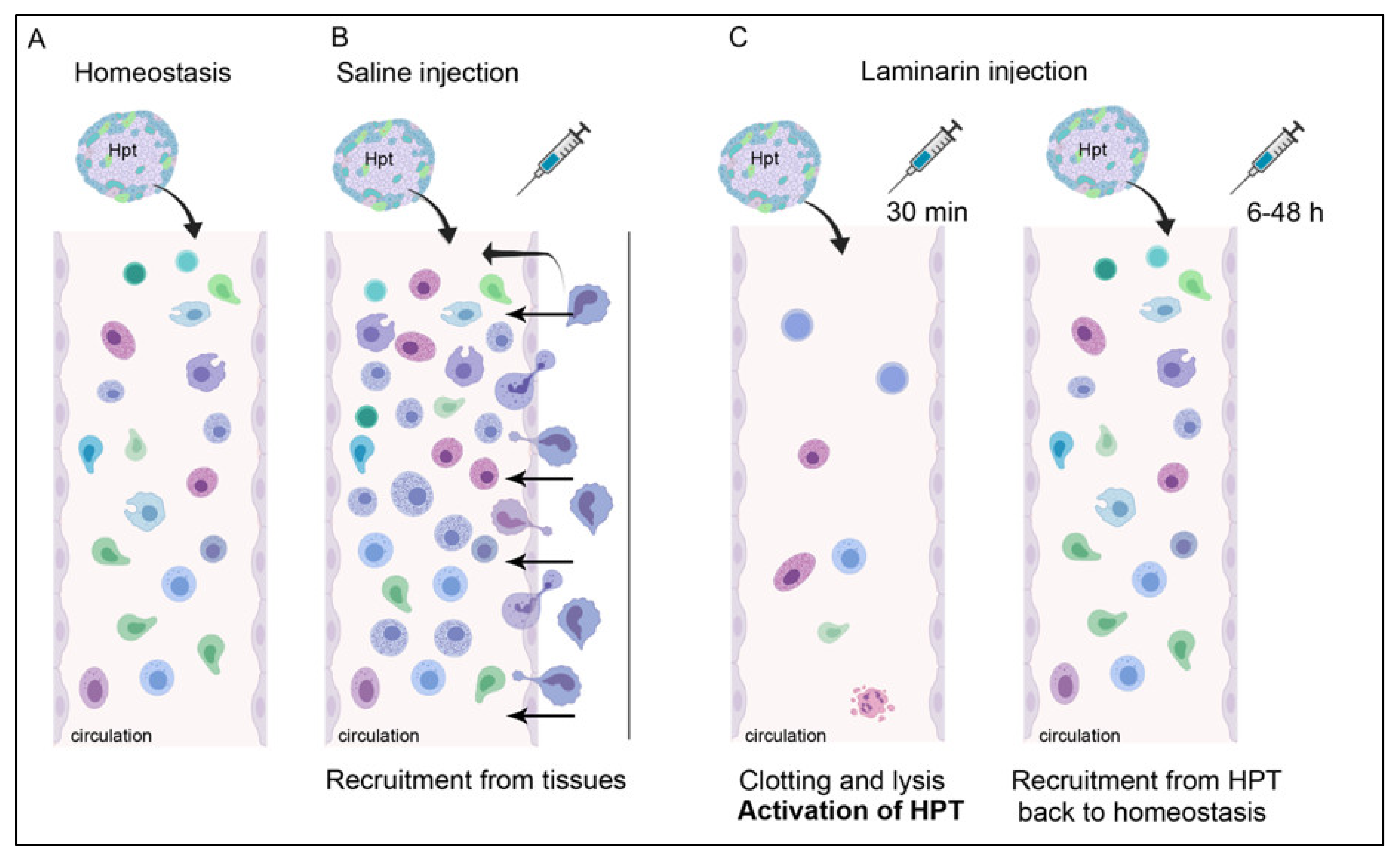
:1. Introduction
2. Results
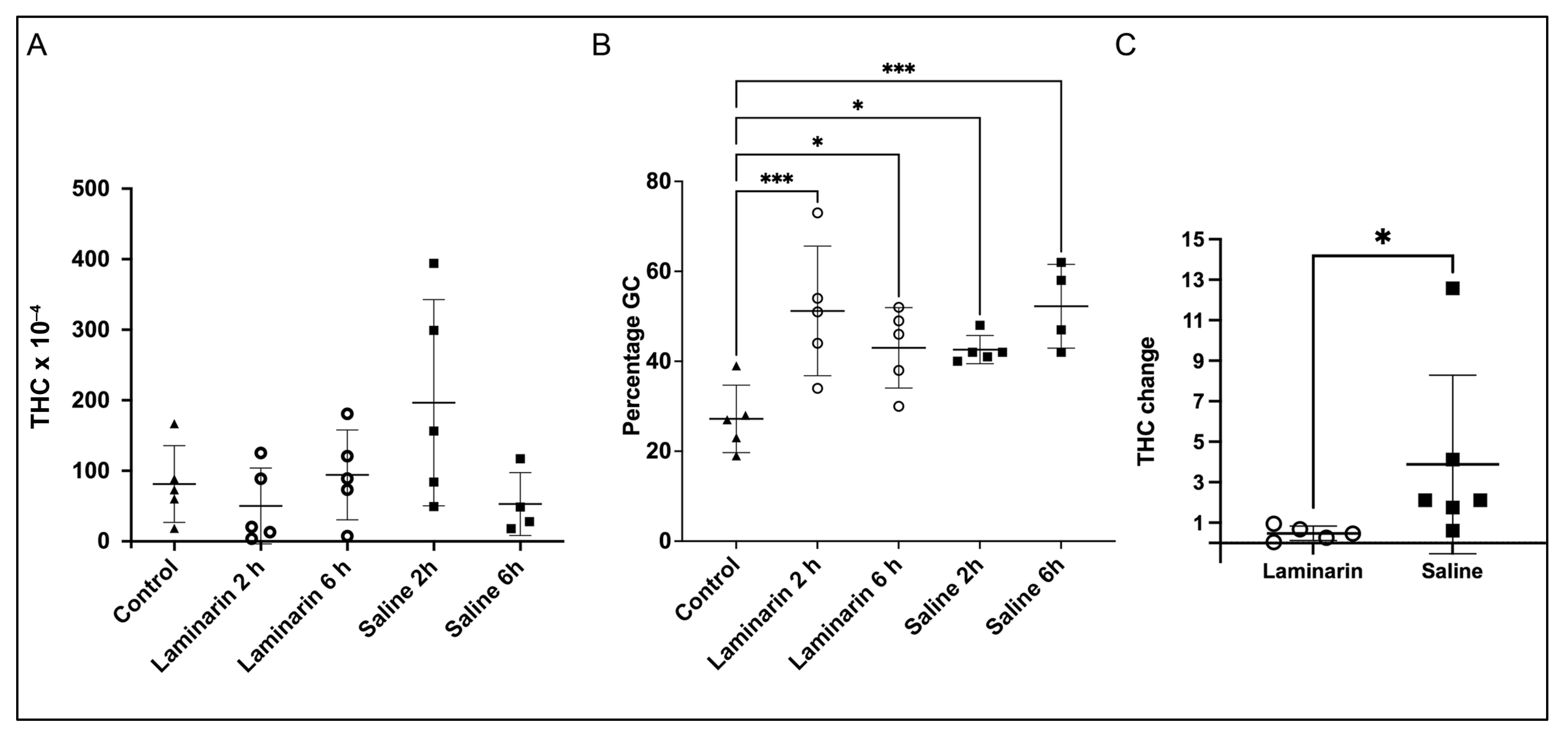
2.1. Differential Cell Counts
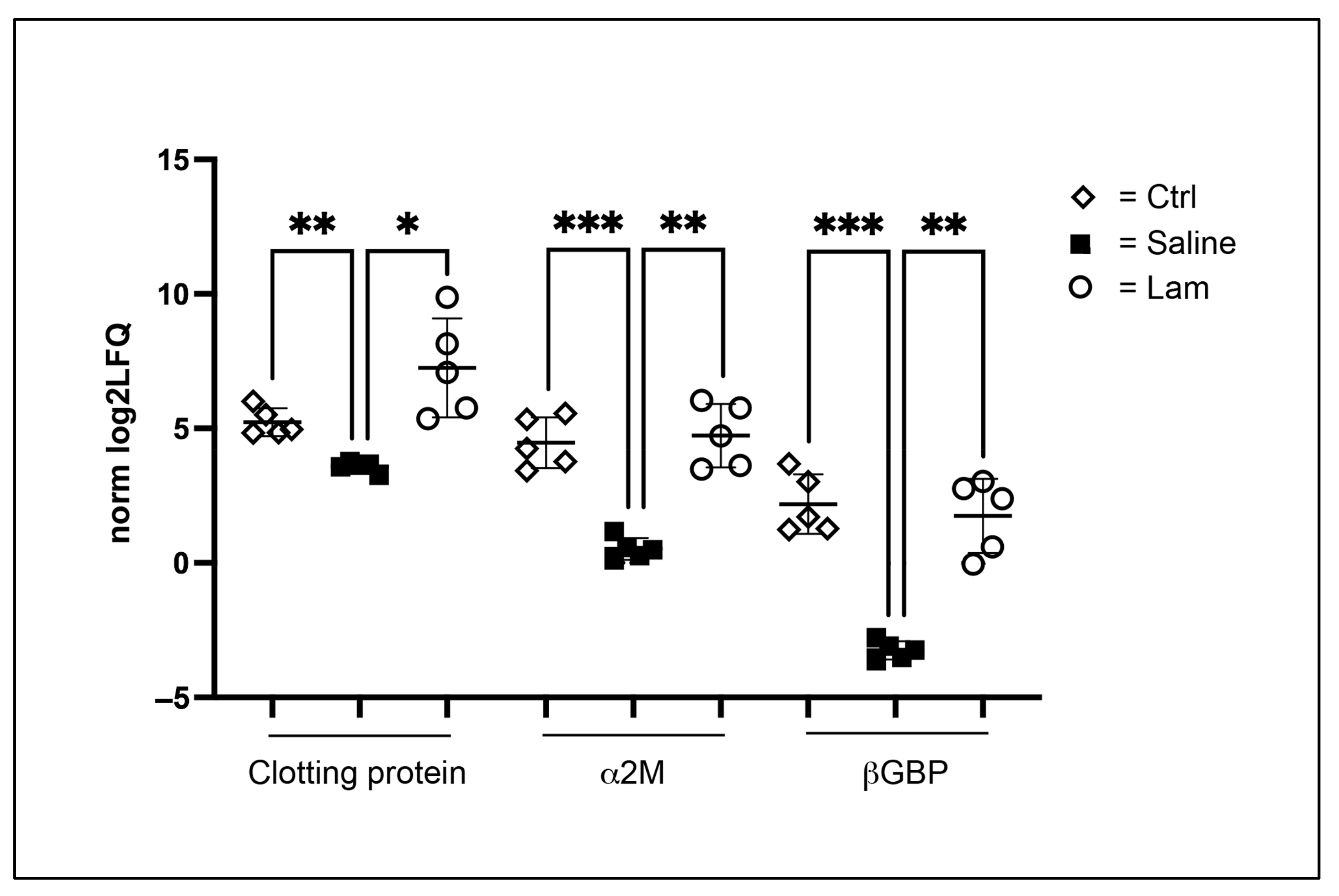
2.2. Proteomic Analysis
2.3. mRNA Expression
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Injections and Cell Preparation
4.3. Proteomic Analysis
4.3.1. Chemicals and Reagents
4.3.2. Protein Extraction and Quantification
4.3.3. On-Filter Tryptic Digestion of Proteins
4.3.4. NanoLC-MS/MS for Protein Identification
4.3.5. Mass Spectrometry Data Handling
4.4. Qualitative Data Analysis
4.5. RNA Extraction and mRNA Expression Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| α2M | Alfa-2-macroglobulin |
| AMP | antimicrobial peptide |
| βGBP | Beta-1,3-glucan binding protein |
| proPO | prophenoloxidase |
| SPH | serine protease homologue |
| VMO1 | vitelline membrane outer layer 1 homologue |
References
- Cerenius, L.; Jiravanichpaisal, P.; Liu, H.; Söderhäll, I. Crustacean Immunity. In Invertebrate Immunity; Söderhäll, K., Ed.; Springer: Boston, MA, USA, 2010; Volume 708, pp. 239–259. ISBN 9781441980588. [Google Scholar]
- Cerenius, L.; Lee, B.L.; Söderhäll, K. The ProPO-System: Pros and cons for its role in invertebrate immunity. Trends Immunol. 2008, 29, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Cerenius, L.; Söderhäll, K. Crayfish immunity—Recent findings. Dev. Comp. Immunol. 2018, 80, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ren, Q. Research progress in innate immunity of freshwater crustaceans. Dev. Comp. Immunol. 2020, 104, 103569. [Google Scholar] [CrossRef]
- Duvic, B.; Söderhäll, K. Purification and characterization of a Beta-1,3-Glucan binding protein from plasma of the crayfish Pacifastacus leniusculus. J. Biol. Chem. 1990, 265, 9327–9332. [Google Scholar] [CrossRef]
- Duvic, B.; Söderhäll, K. Purification and partial characterization of a Beta-1,3-glucan-binding-protein membrane receptor from blood cells of the crayfish Pacifastacus leniusculus. Eur. J. Biochem. 1992, 207, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Seki, N.; Muta, T.; Oda, T.; Iwaki, D.; Kuma, K.; Miyata, T.; Iwanaga, S. Horseshoe Crab (1,3)-Beta-D-Glucan-Sensitive Coagulation Factor G. a serine protease zymogen heterodimer with similarities to beta-glucan-binding proteins. J. Biol. Chem. 1994, 269, 1370–1374. [Google Scholar] [CrossRef]
- Ma, C.; Kanost, M.R. A Beta-1,3-Glucan-Binding Protein from Manduca sexta. Adv. Exp. Med. Biol. 2001, 484, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Amparyup, P.; Sutthangkul, J.; Charoensapsri, W.; Tassanakajon, A. Pattern Recognition Protein Binds to Lipopolysaccharide and β-1,3-Glucan and Activates Shrimp Prophenoloxidase System. J. Biol. Chem. 2012, 287, 10060–10069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, X.; Xia, Y. β-1,3-Glucan Recognition Protein (ΒGRP) Is Essential for Resistance against Fungal Pathogen and Opportunistic Pathogenic Gut Bacteria in Locusta migratoria Manilensis. Dev. Comp. Immunol. 2012, 36, 602–609. [Google Scholar] [CrossRef]
- Söderhäll, K.; Smith, V.J. Separation of the Haemocyte Populations of Carcinus maenas and Other Marine Decapods, and Prophenoloxidase Distribution. Dev. Comp. Immunol. 1983, 7, 229–239. [Google Scholar] [CrossRef]
- Johansson, M.W.; Keyser, P.; Sritunyalucksana, K.; Söderhäll, K. Crustacean Haemocytes and Haematopoiesis. Aquaculture 2000, 191, 45–52. [Google Scholar] [CrossRef]
- Sequeira, T.; Tavares, D.; Arala-Chaves, M. Evidence for Circulating Hemocyte Proliferation in the Shrimp Penaeus japonicus. Dev. Comp. Immunol. 1996, 20, 97–104. [Google Scholar] [CrossRef]
- Zhou, Y.-L.; Gu, W.-B.; Tu, D.-D.; Zhu, Q.-H.; Zhou, Z.-K.; Chen, Y.-Y.; Shu, M.-A. Hemocytes of the Mud Crab Scylla Paramamosain: Cytometric, Morphological Characterization and Involvement in Immune Responses. Fish Shellfish Immunol. 2018, 72, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Van de Braak, C.B.T.; Taverne, N.; Botterblom, M.H.A.; van der Knaap, W.P.W.; Rombout, J.H.W.M. Characterisation of Different Morphological Features of Black Tiger Shrimp (Penaeus monodon) Haemocytes Using Monoclonal Antibodies. Fish Shellfish Immunol. 2000, 10, 515–530. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Zhan, W.; Li, Q.; Zhang, Z.; Wei, X.; Sheng, X. Ontogenesis of Haemocytes in Shrimp (Fenneropenaeus chinensis) Studied with Probes of Monoclonal Antibody. Dev. Comp. Immunol. 2007, 31, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Chang, Y.; Tang, X.; Sheng, X.; Zhan, W. Separation of Haemocyte Subpopulations in Shrimp Fenneropenaeus chinensis by Immunomagnetic Bead Using Monoclonal Antibody against Granulocytes. Fish Shellfish Immunol. 2017, 60, 114–118. [Google Scholar] [CrossRef]
- Wu, C.; Söderhäll, I.; Kim, Y.-A.; Liu, H.; Söderhäll, K. Hemocyte-Lineage Marker Proteins in a Crustacean, the Freshwater Crayfish, Pacifastacus leniusculus. Proteomics 2008, 8, 4226–4235. [Google Scholar] [CrossRef] [PubMed]
- Söderhäll, I.; Junkunlo, K. A Comparative Global Proteomic Analysis of the Hematopoietic Lineages in the Crustacean Pacifastacus leniusculus. Dev. Comp. Immunol. 2019, 92, 170–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorenzon, S.; de Guarrini, S.; Smith, V.J.; Ferrero, E.A. Effects of LPS Injection on Circulating Haemocytes in Crustaceansin Vivo. Fish Shellfish Immunol. 1999, 9, 31–50. [Google Scholar] [CrossRef]
- Smith, V.J.; Söderhäll, K.; Hamilton, M. β 1,3-Glucan Induced Cellular Defence Reactions in the Shore Crab, Carcinus maenas. Comp. Biochem. Physiol. A Physiol. 1984, 77, 635–639. [Google Scholar] [CrossRef]
- Söderhäll, I.; Bangyeekhun, E.; Mayo, S.; Söderhäll, K. Hemocyte Production and Maturation in an Invertebrate Animal; Proliferation and Gene Expression in Hematopoietic Stem Cells of Pacifastacus leniusculus. Dev. Comp. Immunol. 2003, 27, 661–672. [Google Scholar] [CrossRef]
- Hammond, J.A.; Smith, V.J. Lipopolysaccharide Induces DNA-Synthesis in a Sub-Population of Hemocytes from the Swimming Crab, Liocarcinus depurator. Dev. Comp. Immunol. 2002, 26, 227–236. [Google Scholar] [CrossRef]
- Söderhäll, I.; Kim, Y.-A.; Jiravanichpaisal, P.; Lee, S.-Y.; Söderhäll, K. An Ancient Role for a Prokineticin Domain in Invertebrate Hematopoiesis. J. Immunol. 2005, 174, 6153–6160. [Google Scholar] [CrossRef] [Green Version]
- Fan, L.; Wang, A.; Wu, Y. Comparative Proteomic Identification of the Hemocyte Response to Cold Stress in White Shrimp, Litopenaeus vannamei. J. Proteomics 2013, 80, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tang, X.; Xing, J.; Sheng, X.; Zhan, W. Proteomic Analysis of Differentially Expressed Proteins in Fenneropenaeus chinensis Hemocytes upon White Spot Syndrome Virus Infection. PLoS ONE 2014, 9, e89962. [Google Scholar] [CrossRef] [Green Version]
- Havanapan, P.; Mangkalanan, S.; Phungthanom, N.; Krittanai, C. Proteomic Analysis and White Spot Syndrome Virus Interaction of Mud Crab (Scylla olivacea) Revealed Responsive Roles of the Hemocytes. Fish Shellfish Immunol. 2019, 89, 458–467. [Google Scholar] [CrossRef]
- Sun, B.; Wang, Z.; Wang, Z.; Ma, X.; Zhu, F. A Proteomic Study of Hemocyte Proteins from Mud Crab (Scylla paramamosain) Infected with White Spot Syndrome Virus or Vibrio alginolyticus. Front. Immunol. 2017, 8, 468. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Xiu, Y.; Wang, J.; Liu, X.; Liu, Y.; Gu, W.; Wang, W.; Meng, Q. ITRAQ-Based Quantitative Proteomic Analysis of Macrobrachium rosenbergii Hemocytes during Spiroplasma eriocheiris Infection. J. Proteomics 2016, 136, 112–122. [Google Scholar] [CrossRef]
- Hou, L.; Zhou, H.; Wan, H.; Liu, Z.; Wang, L.; Cheng, Y.; Wu, X.; Gu, W.; Wang, W.; Meng, Q. TMT-Based Quantitative Proteomic Analysis of Eriocheir sinensis Hemocytes and Thoracic Ganglion during Spiroplasma eriocheiris Infection. Fish Shellfish Immunol. 2020, 96, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Wang, R.; Söderhäll, K. A Lipopolysaccharide- and Beta-1,3-Glucan-Binding Protein from Hemocytes of the Freshwater Crayfish Pacifastacus leniusculus. Purification, Characterization, and CDNA Cloning. J. Biol. Chem. 2000, 275, 1337–1343. [Google Scholar] [CrossRef] [Green Version]
- Lorenzon, S.; Giulianini, P.G.; Ferrero, E.A. Lipopolysaccharide-Induced Hyperglycemia Is Mediated by CHH Release in Crustaceans. Gen. Comp. Endocrinol. 1997, 108, 395–405. [Google Scholar] [CrossRef]
- Okumura, T. Effects of Lipopolysaccharide on Gene Expression of Antimicrobial Peptides (Penaeidins and Crustin), Serine Proteinase and Prophenoloxidase in Haemocytes of the Pacific White Shrimp, Litopenaeus vannamei. Fish Shellfish Immunol. 2007, 22, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Xian, J.-A.; Zhang, X.-X.; Guo, H.; Wang, D.-M.; Wang, A.-L. Cellular Responses of the Tiger Shrimp Penaeus monodon Haemocytes after Lipopolysaccharide Injection. Fish Shellfish Immunol. 2016, 54, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Persson, M.; Cerenius, L.; Söderhäll, K. The Influence of Haemocyte Number on the Resistance of the Freshwater Crayfish, Pacifastacus leniusculus Dana, to the Parasitic Fungus Aphanomyces Astaci. J. Fish Dis. 1987, 10, 471–477. [Google Scholar] [CrossRef]
- Sricharoen, S.; Kim, J.J.; Tunkijjanukij, S.; Söderhäll, I. Exocytosis and Proteomic Analysis of the Vesicle Content of Granular Hemocytes from a Crayfish. Dev. Comp. Immunol. 2005, 29, 1017–1031. [Google Scholar] [CrossRef]
- Cerenius, L.; Liu, H.; Zhang, Y.; Rimphanitchayakit, V.; Tassanakajon, A.; Gunnar Andersson, M.; Söderhäll, K.; Söderhäll, I. High Sequence Variability among Hemocyte-Specific Kazal-Type Proteinase Inhibitors in Decapod Crustaceans. Dev. Comp. Immunol. 2010, 34, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Jearaphunt, M.; Noonin, C.; Jiravanichpaisal, P.; Nakamura, S.; Tassanakajon, A.; Söderhäll, I.; Söderhäll, K. Caspase-1-like regulation of the ProPO-system and role of PpA and Caspase-1-like cleaved peptides from ProPO in innate immunity. PLoS Pathog. 2014, 10, e1004059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.Y.; Söderhäll, K. Characterization of a pattern recognition protein, a masquerade-like protein, in the freshwater crayfish Pacifastacus leniusculus. J. Immunol. Baltim. Md 1950 2001, 166, 7319–7326. [Google Scholar] [CrossRef] [Green Version]
- Jiravanichpaisal, P.; Lee, S.Y.; Kim, Y.-A.; Andrén, T.; Söderhäll, I. Antibacterial peptides in hemocytes and hematopoietic tissue from freshwater crayfish Pacifastacus leniusculus: Characterization and Expression Pattern. Dev. Comp. Immunol. 2007, 31, 441–455. [Google Scholar] [CrossRef]
- Hergenhahn, H.G.; Hall, M.; Söderhäll, K. Purification and characterization of an Alpha 2-Macroglobulin-like proteinase inhibitor from plasma of the crayfish Pacifastacus leniusculus. Biochem. J. 1988, 255, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Kopácek, P.; Hall, M.; Söderhäll, K. Characterization of a clotting protein, isolated from plasma of the freshwater crayfish Pacifastacus leniusculus. Eur. J. Biochem. 1993, 213, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Pérez, A.; Zamora-Briseño, J.A.; Ruiz-May, E.; Pereira-Santana, A.; Elizalde-Contreras, J.M.; Pozos-González, S.; Torres-Irineo, E.; Hernández-López, J.; Gaxiola-Cortés, M.G.; Rodríguez-Canul, R. Proteomic profiling of the white shrimp Litopenaeus vannamei (Boone, 1931) hemocytes infected with white spot syndrome virus reveals the induction of allergy-related proteins. Dev. Comp. Immunol. 2019, 91, 37–49. [Google Scholar] [CrossRef]
- Zhu, L.; Tang, X.; Xing, J.; Sheng, X.; Zhan, W. Differential proteome of haemocyte subpopulations responded to white spot syndrome virus infection in chinese shrimp Fenneropenaeus chinensis. Dev. Comp. Immunol. 2018, 84, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Sirikharin, R.; Noonin, C.; Junkunlo, K.; Söderhäll, K.; Söderhäll, I. Astakine1 Forms Protein Complex in Plasma. Fish Shellfish Immunol. 2019, 94, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Callewaert, L.; Michiels, C.W. Lysozymes in the Animal Kingdom. J. Biosci. 2010, 35, 127–160. [Google Scholar] [CrossRef]
- Smith, V.J.; Fernandes, J.M.O.; Kemp, G.D.; Hauton, C. Crustins: Enigmatic WAP domain-containing antibacterial proteins from crustaceans. Dev. Comp. Immunol. 2008, 32, 758–772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramond, E.; Dudzic, J.P.; Lemaitre, B. Comparative RNA-Seq Analyses of drosophila plasmatocytes reveal gene specific signatures in response to clean injury and septic injury. PLoS ONE 2020, 15, e0235294. [Google Scholar] [CrossRef] [PubMed]
- Rimphanitchayakit, V.; Tassanakajon, A. Structure and function of invertebrate kazal-type serine proteinase inhibitors. Dev. Comp. Immunol. 2010, 34, 377–386. [Google Scholar] [CrossRef]
- Johansson, M.W.; Keyser, P.; Söderhäll, K. Purification and cDNA cloning of a four-domain kazal proteinase inhibitor from crayfish blood cells. Eur. J. Biochem. 1994, 223, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Donpudsa, S.; Söderhäll, I.; Rimphanitchayakit, V.; Cerenius, L.; Tassanakajon, A.; Söderhäll, K. Proteinase inhibitory activities of two two-domain kazal proteinase inhibitors from the freshwater crayfish Pacifastacus leniusculus and the importance of the P2 position in proteinase inhibitory activity. Fish Shellfish Immunol. 2010, 29, 716–723. [Google Scholar] [CrossRef]
- Wang, B.; Zhao, J.; Song, L.; Zhang, H.; Wang, L.; Li, C.; Zheng, P.; Zhu, L.; Qiu, L.; Xing, K. Molecular cloning and expression of a novel kazal-type serine proteinase inhibitor gene from zhikong scallop Chlamys farreri, and the inhibitory activity of its recombinant domain. Fish Shellfish Immunol. 2008, 24, 629–637. [Google Scholar] [CrossRef]
- Kong, H.J.; Cho, H.K.; Park, E.-M.; Hong, G.-E.; Kim, Y.-O.; Nam, B.-H.; Kim, W.-J.; Lee, S.-J.; Han, H.S.; Jang, I.-K. Molecular Cloning of kazal-type proteinase inhibitor of the shrimp Fenneropenaeus chinensis. Fish Shellfish Immunol. 2009, 26, 109–114. [Google Scholar] [CrossRef]
- Donpudsa, S.; Tassanakajon, A.; Rimphanitchayakit, V. Domain inhibitory and bacteriostatic activities of the five-domain kazal-type serine proteinase inhibitor from black tiger shrimp Penaeus monodon. Dev. Comp. Immunol. 2009, 33, 481–488. [Google Scholar] [CrossRef]
- Augustin, R.; Franke, A.; Khalturin, K.; Kiko, R.; Siebert, S.; Hemmrich, G.; Bosch, T.C.G. Dickkopf related genes are components of the positional value gradient in hydra. Dev. Biol. 2006, 296, 62–70. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, G.; Cheng, X.; Wang, Q.; Wang, B.; Wang, B.; Zhang, H.; He, Q.; Zhang, L. Antimicrobial activity of a newly identified kazal-type serine proteinase inhibitor, CcKPI1, from the Jellyfish Cyanea capillata. Int. J. Biol. Macromol. 2018, 107, 1945–1955. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y. Procambarin: A glycine-rich peptide found in the haemocytes of red swamp crayfish Procambarus clarkii and its response to white spot syndrome virus challenge. Fish Shellfish Immunol. 2013, 35, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Wan, H.; Zeng, X.; Zhang, Z.; Wang, Y. Characterization and antimicrobial evaluation of a New Spgly-AMP, glycine-rich antimicrobial peptide from the mud crab Scylla paramamosain. Fish Shellfish Immunol. 2020, 106, 384–392. [Google Scholar] [CrossRef]
- Lorenzini, D.M.; da Silva, P.I.; Fogaça, A.C.; Bulet, P.; Daffre, S. Acanthoscurrin: A novel glycine-rich antimicrobial peptide constitutively expressed in the hemocytes of the spider Acanthoscurria gomesiana. Dev. Comp. Immunol. 2003, 27, 781–791. [Google Scholar] [CrossRef]
- Verdon, J.; Coutos-Thevenot, P.; Rodier, M.-H.; Landon, C.; Depayras, S.; Noel, C.; La Camera, S.; Moumen, B.; Greve, P.; Bouchon, D.; et al. Armadillidin H, a glycine-rich peptide from the terrestrial crustacean Armadillidium vulgare, displays an unexpected wide antimicrobial spectrum with membranolytic activity. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Jesus Oliveira, T.; de Oliveira, U.C.; da Silva Junior, P.I. Serrulin: A glycine-rich bioactive peptide from the hemolymph of the yellow Tityus serrulatus Scorpion. Toxins 2019, 11, 517. [Google Scholar] [CrossRef] [Green Version]
- Junkunlo, K.; Söderhäll, K.; Söderhäll, I. Clotting Protein—An Extracellular Matrix (ECM) protein involved in crustacean hematopoiesis. Dev. Comp. Immunol. 2018, 78, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Chaikeeratisak, V.; Somboonwiwat, K.; Tassanakajon, A. Shrimp Alpha-2-Macroglobulin prevents the bacterial escape by inhibiting fibrinolysis of blood clots. PLoS ONE 2012, 7, e47384. [Google Scholar] [CrossRef]
- Jahed, Z.; Shams, H.; Mehrbod, M.; Mofrad, M.R.K. Mechanotransduction pathways linking the extracellular matrix to the nucleus. Int. Rev. Cell Mol. Biol. 2014, 310, 171–220. [Google Scholar] [CrossRef]
- Gordon, O.; Henry, C.M.; Srinivasan, N.; Ahrens, S.; Franz, A.; Deddouche, S.; Chakravarty, P.; Phillips, D.; George, R.; Kjaer, S.; et al. α-actinin accounts for the bioactivity of actin preparations in inducing STAT target genes in Drosophila melanogaster. eLife 2018, 7. [Google Scholar] [CrossRef]
- Srinivasan, N.; Gordon, O.; Ahrens, S.; Franz, A.; Deddouche, S.; Chakravarty, P.; Phillips, D.; Yunus, A.A.; Rosen, M.K.; Valente, R.S.; et al. Actin is an evolutionarily-conserved damage-associated molecular pattern that signals tissue injury in Drosophila melanogaster. eLife 2016, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rock, K.L.; Latz, E.; Ontiveros, F.; Kono, H. The sterile inflammatory response. Annu. Rev. Immunol. 2010, 28, 321–342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zelenay, S.; Reis e Sousa, C. Adaptive Immunity after Cell Death. Trends Immunol. 2013, 34, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Weiner, A.K.; Sidoli, S.; Diskin, S.J.; Garcia, B.A. Graphical interpretation and analysis of proteins and their ontologies (GiaPronto): A one-click graph visualization software for proteomics data Sets. Mol. Cell. Proteomics MCP 2018, 17, 1426–1431. [Google Scholar] [CrossRef] [Green Version]

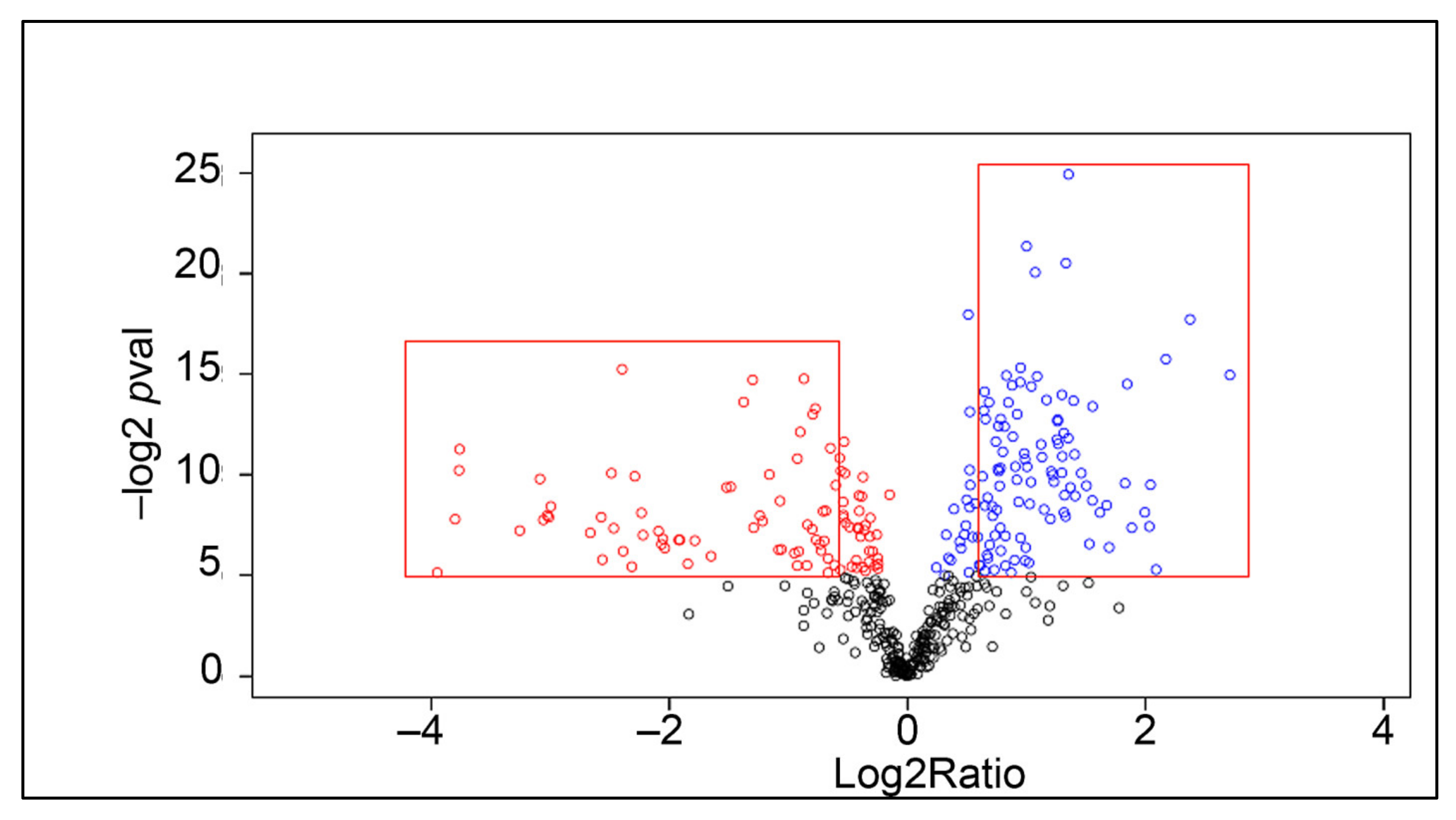
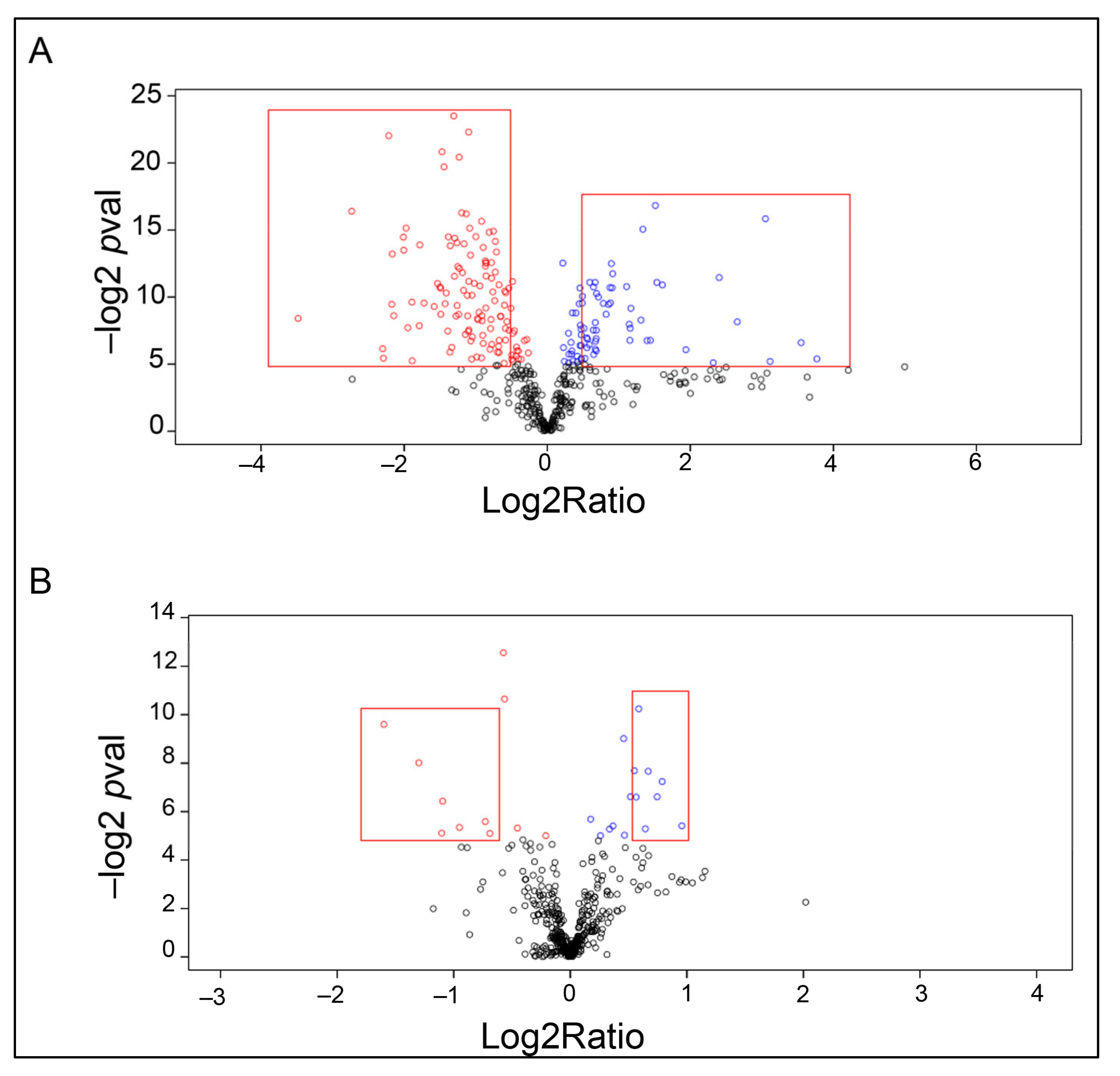

| Accession Number | Protein Name | Fold Change, Log2ratio | p-Value (−log) |
|---|---|---|---|
| GBYW01031372.1 | Glutathione peroxidase 3 (GPX) | 2.708 | 14.95 |
| GBYW01022610.1 GBYW01021121.1 GBYW01005009.1 | Heat shock cognate 71kDa protein | 2.374 | 17.72 |
| GBYW01026927.1 | Trypsin-like serine protease | 2.170 | 15.740 |
| GBYW01024713.1 | CLIP-domain serin protease | 2.088 | 5.28 |
| GBYW01017647.1 GBYW01041647.1 | Flotillin-like protein | 2.041 | 9.49 |
| GBYW01021864.1 | PDGF/VEGF domain protein | 2.031 | 7.41 |
| GBYW01011929.1 | Peroxidase | 1.994 | 8.12 |
| GBYW01037257.1 | Vitelline membrane outer layer protein 1 (VMO1) | 1.884 | 7.35 |
| GBYW01020841.1 GBYW01022391.1 | Delta-1-pyrroline-5-carboxylate dehydrogenase | 1.845 | 14.50 |
| GBYW01031650.1 | Unknown | 1.694 | 6.34 |
| GBYW01028873.1 | i-type lysozyme | 1.673 | 8.474 |
| GBYW01030110.1 GBYW01030111.1 | Unknown | 1.617 | 8.11 |
| Accession Number | Protein Name | Biomarker Value | Fold Change Log2Ratio | p-Value |
|---|---|---|---|---|
| GBYW01037257.1 | VMO1 | 6.656 | 1.8836 | 0.006 |
| EF523612.1 | Crustin 1 | 7.838 | 1.319 | 0.002 |
| AY861652.1 | SPH1 | 5.418 | 1.404 | 0.0005 |
| GBYW01031372.1 | Glutathione peroxidase (GPX) | 1.767 | 2.708 | 3,15199E-05 |
| AY861653.1 | Mannose binding lectin | 5.133 | 1.1447 | 0.003 |
| Y11145.2 | Masquerade | 7.177 | 0.652 | 0.003 |
| GBYW01028010.1 | Kazal-type PI one domain | 5.682 | 0.949 | 0.009 |
| GBYW01031855.1 | Kazal-type PI Agrin-like | 6.029 | 0.823 | 0.022 |
| Proteins Upregulated | Accession Number | Proteins Upregulated | Accession Number |
|---|---|---|---|
| In Laminarin and Saline Injection Compared to Uninjected | In Laminarin Compared to Uninjected | ||
| i-type lysozyme | GBYW01028873.1 | Glycine-rich peptide | GBYW01011676.1 |
| Crustin -like AMP | GBYW01028095.1 | Kazal-type | GBYW01031549.1 |
| Masquerade | Y11145.2 | Putative chitin binding protein | GBYW01037126.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ekblom, C.; Söderhäll, K.; Söderhäll, I. Early Changes in Crayfish Hemocyte Proteins after Injection with a β-1,3-glucan, Compared to Saline Injected and Naive Animals. Int. J. Mol. Sci. 2021, 22, 6464. https://doi.org/10.3390/ijms22126464
Ekblom C, Söderhäll K, Söderhäll I. Early Changes in Crayfish Hemocyte Proteins after Injection with a β-1,3-glucan, Compared to Saline Injected and Naive Animals. International Journal of Molecular Sciences. 2021; 22(12):6464. https://doi.org/10.3390/ijms22126464
Chicago/Turabian StyleEkblom, Charlotta, Kenneth Söderhäll, and Irene Söderhäll. 2021. "Early Changes in Crayfish Hemocyte Proteins after Injection with a β-1,3-glucan, Compared to Saline Injected and Naive Animals" International Journal of Molecular Sciences 22, no. 12: 6464. https://doi.org/10.3390/ijms22126464





