Effect of Fibrin Concentration on the In Vitro Production of Dermo-Epidermal Equivalents
Abstract
1. Introduction
2. Results and Discussion
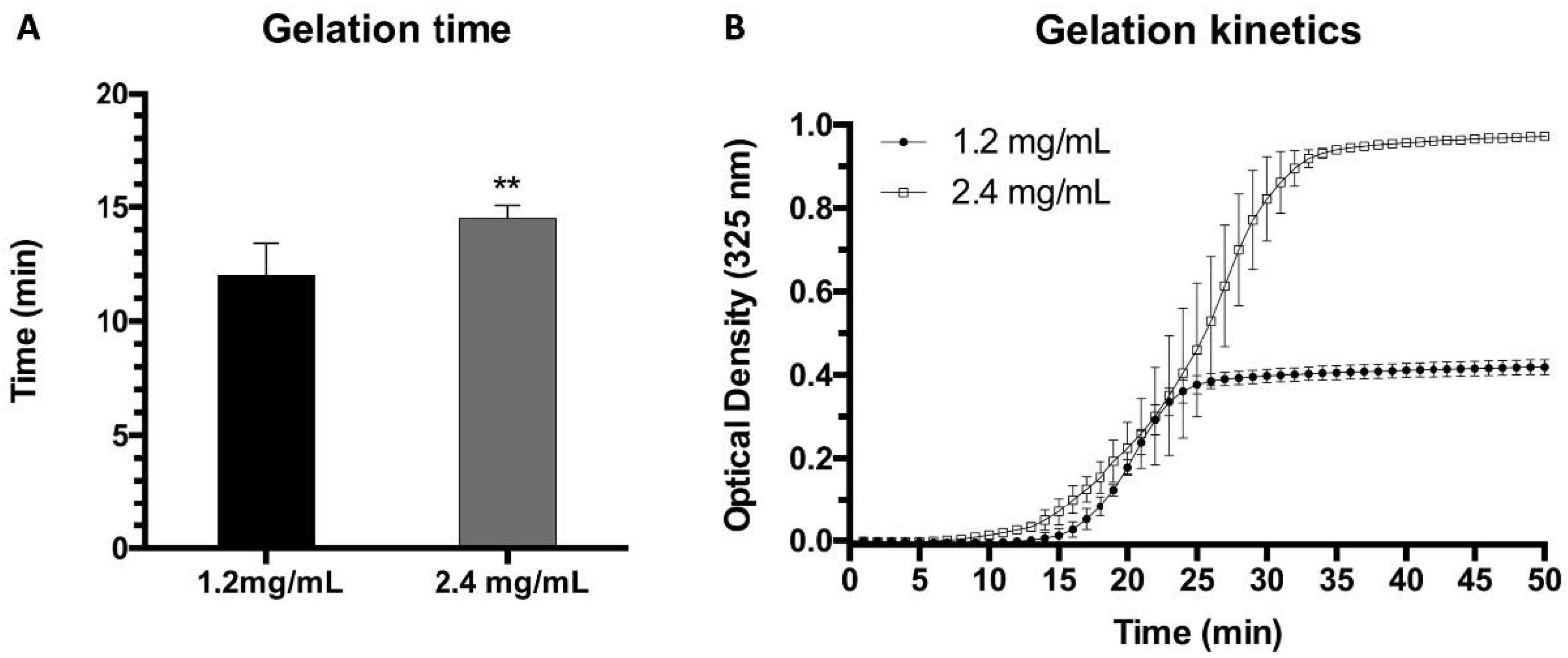
2.1. Gelation Time and Kinetics
2.2. Mechanical Characterization
2.3. Structural Characterization
2.4. hFB-Mediated Matrix Contraction, Proliferation, and Live/Dead Assays
2.5. hKC Proliferation Assay
2.6. Contraction of Organotypic Skin Construct (Z Axis)
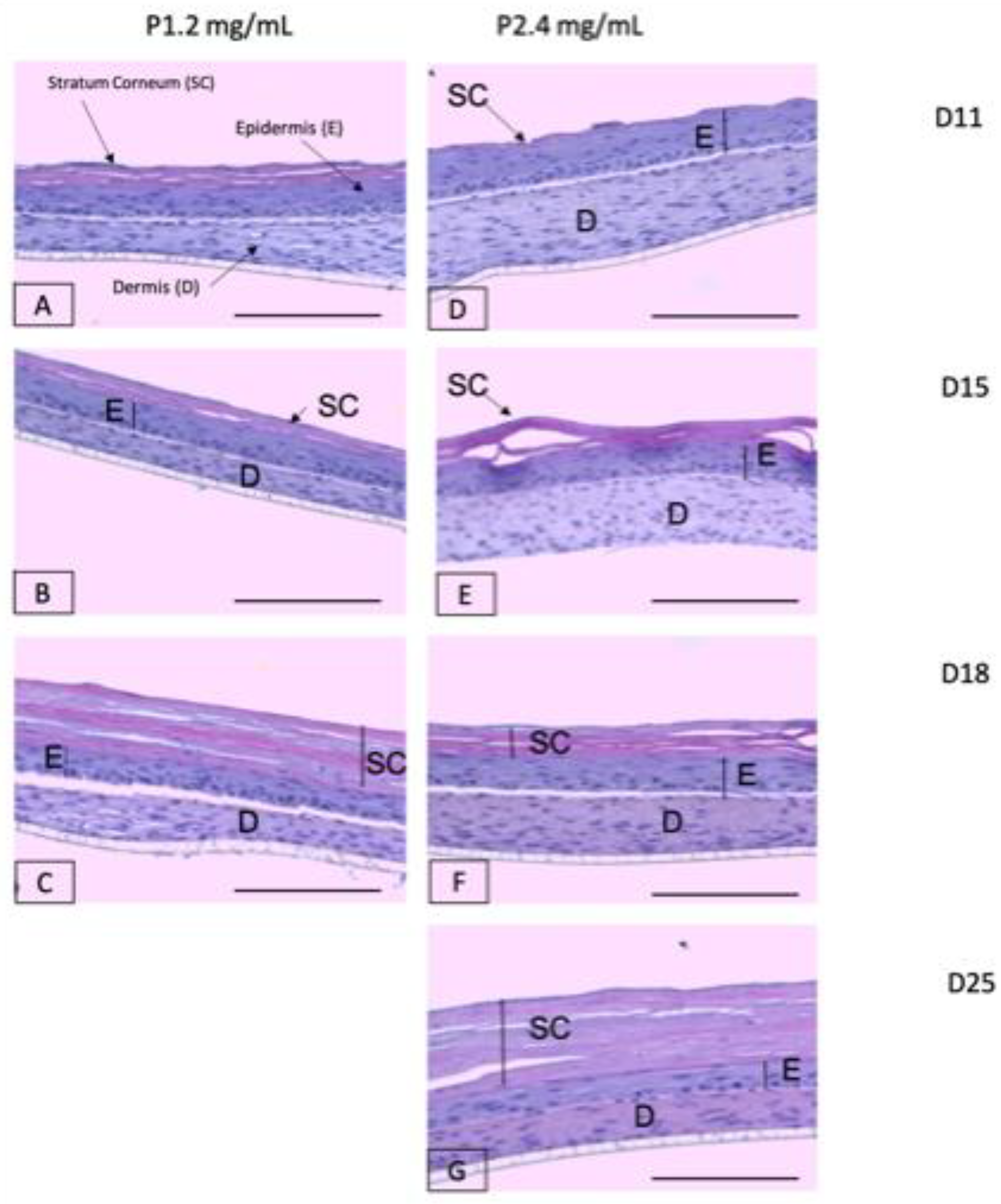
2.7. Study of Organotypic Skin Cultures
3. Materials and Methods
3.1. Gelation Time and Kinetics
3.1.1. Plasma-Derived Fibrin Hydrogel Preparation
3.1.2. Gelation Time and Kinetics
3.1.3. Mechanical Characterization: Compression Test
3.1.4. Structural Characterization (SEM)
3.1.5. Primary hKCs and hFBs Culture
3.1.6. hFBs-Mediated Contraction
3.1.7. hFB Proliferation and Life/Death Assay
3.1.8. Keratinocyte Proliferation Assay (MTS)
3.1.9. Contraction of Plasma-Derived Fibrin Hydrogels in the Z Axis under In Vitro Skin Culture Conditions
3.2. Organotypic Skin Cultures
3.2.1. Study of Organotypic Skin Cultures at Different Timepoints
3.2.2. Histological and Immunofluorescence Analyses
3.3. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Berthiaume, F.; Maguire, T.J.; Yarmush, M.L. Tissue Engineering and Regenerative Medicine: History, Progress, and Challenges. Annu. Rev. Chem. Biomol. Eng. 2011, 2, 403–430. [Google Scholar] [CrossRef]
- Brohem, C.A.; Cardeal, L.B.D.S.; Tiago, M.; Soengas, M.; Barros, S.B.D.M.; Maria-Engler, S.S. Artificial skin in perspective: Concepts and applications. Pigment. Cell Melanoma Res. 2010, 24, 35–50. [Google Scholar] [CrossRef]
- Vig, K.; Chaudhari, A.; Tripathi, S.; Dixit, S.; Sahu, R.; Pillai, S.; Dennis, V.A.; Singh, S.R. Advances in Skin Regeneration Using Tissue Engineering. Int. J. Mol. Sci. 2017, 18, 789. [Google Scholar] [CrossRef] [PubMed]
- Abd, E.; A Yousuf, S.; Pastore, M.N.; Telaprolu, K.; Mohammed, Y.H.; Namjoshi, S.; E Grice, J.; Roberts, M.S. Skin models for the testing of transdermal drugs. Clin. Pharmacol. Adv. Appl. 2016, 8, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Mathes, S.H.; Ruffner, H.; Graf-Hausner, U. The use of skin models in drug development. Adv. Drug Deliv. Rev. 2014, 69–70, 81–102. [Google Scholar] [CrossRef]
- Priya, S.G.; Jungvid, H.; Kumar, A. Skin Tissue Engineering for Tissue Repair and Regeneration. Tissue Eng. Part B Rev. 2008, 14, 105–118. [Google Scholar] [CrossRef]
- Pereira, R.F.; Sousa, A.; Barrias, C.C.; Bayat, A.; Granja, P.L.; Bártolo, P.J. Advances in bioprinted cell-laden hydrogels for skin tissue engineering. Biomanuf. Rev. 2017, 2, 1. [Google Scholar] [CrossRef]
- Ng, W.L.; Wang, S.; Yeong, W.Y.; Naing, M.W. Skin Bioprinting: Impending Reality or Fantasy? Trends Biotechnol. 2016, 34, 689–699. [Google Scholar] [CrossRef] [PubMed]
- Cubo, N.; Garcia, M.; Del Cañizo, J.F.; Velasco, D.; Jorcano, J.L. 3D bioprinting of functional human skin: Production and in vivo analysis. Biofabrication 2016, 9, 015006. [Google Scholar] [CrossRef]
- Stojic, M.; López, V.; Montero, A.; Quílez, C.; de Aranda Izuzquiza, G.; Vojtova, L.; Jorcano, J.L.; Velasco, D. Skin tissue Engineering. In Biomaterials for Skin Repair and Regeneration; Garcia-Gareta, E., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 59–100. [Google Scholar]
- Chaudhari, A.A.; Vig, K.; Baganizi, D.R.; Sahu, R.; Dixit, S.; Dennis, V.; Singh, S.R.; Pillai, S.R. Future prospects for scaffolding methods and biomaterials in skin tissue engineering: A review. Int. J. Mol. Sci. 2016, 17, 1974. [Google Scholar] [CrossRef]
- Jeong, K.-H.; Park, D.; Lee, Y.-C. Polymer-based hydrogel scaffolds for skin tissue engineering applications: A mini-review. J. Polym. Res. 2017, 24, 112. [Google Scholar] [CrossRef]
- Norouzi, M.; Boroujeni, S.M.; Omidvarkordshouli, N.; Soleimani, M. Advances in Skin Regeneration: Application of Electrospun Scaffolds. Adv. Healthc. Mater. 2015, 4, 1114–1133. [Google Scholar] [CrossRef] [PubMed]
- MacNeil, S. Biomaterials for tissue engineering of skin. Mater. Today 2008, 11, 26–35. [Google Scholar] [CrossRef]
- Llames, S.; Del Rio, M.; Larcher, F.; García, E.; García, M.; Escámez, M.J.; Jorcano, J.L.; Holguín, P.; Meana, A. Human plasma as a dermal scaffold for the generation of a completely autologous bioengineered skin. Transplantation 2004, 77, 350–355. [Google Scholar] [CrossRef]
- Mazlyzam, A.; Aminuddin, B.; Fuzina, N.; Norhayati, M.; Fauziah, O.; Isa, M.; Saim, L.; Ruszymah, B. Reconstruction of living bilayer human skin equivalent utilizing human fibrin as a scaffold. Burns 2007, 33, 355–363. [Google Scholar] [CrossRef]
- Montero, A.; Acosta, S.; Hernández, R.; Elvira, C.; Jorcano, J.L.; Velasco, D. Contraction of fibrin-derived matrices and its implications for in vitro human skin bioengineering. J. Biomed. Mater. Res. Part A 2021, 109, 500–514. [Google Scholar] [CrossRef]
- Marck, R.E.; Middelkoop, E.; Breederveld, R.S. Considerations on the Use of Platelet-Rich Plasma, Specifically for Burn Treatment. J. Burn. Care Res. 2014, 35, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Hellstern, P. Solvent/detergent-treated plasma: Composition, efficacy, and safety. Curr. Opin. Hematol. 2004, 11, 346–350. [Google Scholar] [CrossRef]
- Geer, D.J.; Swartz, D.D.; Andreadis, S.T. Fibrin Promotes Migration in a Three-Dimensional in Vitro Model of Wound Regeneration. Tissue Eng. 2002, 8, 787–798. [Google Scholar] [CrossRef]
- Gil Park, Y.; Lee, I.H.; Park, E.S.; Kim, J.Y. Hydrogel and Platelet-Rich Plasma Combined Treatment to Accelerate Wound Healing in a Nude Mouse Model. Arch. Plast. Surg. 2017, 44, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Aurora, A.; Wrice, N.; Walters, T.J.; Christy, R.J.; Natesan, S. A PEGylated platelet free plasma hydrogel based composite scaffold enables stable vascularization and targeted cell delivery for volumetric muscle loss. Acta Biomater. 2018, 65, 150–162. [Google Scholar] [CrossRef]
- Chellini, F.; Tani, A.; Zecchi-Orlandini, S.; Sassoli, C. Influence of Platelet-Rich and Platelet-Poor Plasma on endogenous mechanisms of skeletal muscle repair/regeneration. Int. J. Mol. Sci. 2019, 20, 683. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.J.; Gray, W.D.; Hayek, S.S.; Ko, Y.A.; Thomas, S.; Rooney, K.; Searles, C.D. Platelets confound the measurement of extracellular miRNA in archived plasma. Sci. Rep. 2016, 6, 32651. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, R.; Judicone, C.; Mooberry, M.; Boucekine, M.; Key, N.S.; Dignat-George, F. Standardization of pre-analytical variables in plasma microparticle determination: Results of the International Society on Thrombosis and Haemostasis SSC Collaborative workshop. J. Thromb. Haemost. 2013, 11, 1190–1193. [Google Scholar] [CrossRef] [PubMed]
- Marx, R.E. Platelet-Rich Plasma (PRP): What Is PRP and What Is Not PRP? Implant. Dent. 2001, 10, 225–228. [Google Scholar] [CrossRef]
- Jalowiec, J.M.; D’Este, M.; Bara, J.J.; Denom, J.; Menzel, U.; Alini, M.; Verrier, S.; Herrmann, M. AnIn VitroInvestigation of Platelet-Rich Plasma-Gel as a Cell and Growth Factor Delivery Vehicle for Tissue Engineering. Tissue Eng. Part C Methods 2016, 22, 49–58. [Google Scholar] [CrossRef]
- Zahn, J.; Loibl, M.; Sprecher, C.; Nerlich, M.; Alini, M.; Verrier, S.; Herrmann, M. Platelet-Rich Plasma as an Autologous and Proangiogenic Cell Delivery System. Mediat. Inflamm. 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Furuya, H.; Tabata, Y. Enhancement of bone regeneration by dual release of a macrophage recruitment agent and platelet-rich plasma from gelatin hydrogels. Biomaterials 2014, 35, 214–224. [Google Scholar] [CrossRef]
- Jain, E.; Chinzei, N.; Blanco, A.; Case, N.; Sandell, L.J.; Sell, S.; Rai, M.F.; Zustiak, S.P. Platelet-Rich Plasma Released From Polyethylene Glycol Hydrogels Exerts Beneficial Effects on Human Chondrocytes. J. Orthop. Res. 2019, 37, 2401–2410. [Google Scholar] [CrossRef]
- Growney, E.A.; Linder, H.R.; Garg, K.; Bledsoe, J.G.; Sell, S.A. Bio-conjugation of platelet-rich plasma and alginate through carbodiimide chemistry for injectable hydrogel therapies. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 1972–1984. [Google Scholar] [CrossRef]
- Gao, X.; Gao, L.; Groth, T.; Liu, T.; He, D.; Wang, M.; Gong, F.; Chu, J.; Zhao, M. Fabrication and properties of an injectable sodium alginate/PRP composite hydrogel as a potential cell carrier for cartilage repair. J. Biomed. Mater. Res. Part A 2019, 107, 2076–2087. [Google Scholar] [CrossRef] [PubMed]
- Nimal, T.R.; Baranwal, G.; Bavya, M.C.; Biswas, R.; Jayakumar, R. Anti-staphylococcal Activity of Injectable Nano Tigecycline/Chitosan-PRP Composite Hydrogel Using Drosophila melanogaster Model for Infectious Wounds. ACS Appl. Mater. Interfaces 2016, 8, 22074–22083. [Google Scholar] [CrossRef] [PubMed]
- Samberg, M.; Stone, R.; Natesan, S.; Kowalczewski, A.; Becerra, S.; Wrice, N.; Cap, A.; Christy, R. Platelet rich plasma hydrogels promote in vitro and in vivo angiogenic potential of adipose-derived stem cells. Acta Biomater. 2019, 87, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Bennardo, F.; Bennardo, L.; Del Duca, E.; Patruno, C.; Fortunato, L.; Giudice, A.; Nisticò, S.P. Autologous platelet-rich fibrin injections in the management of facial cutaneous sinus tracts secondary to medication-related osteonecrosis of the jaw. Dermatol. Ther. 2020, 33, e13334. [Google Scholar] [CrossRef]
- Lang, S.; Loibl, M.; Herrmann, M. Platelet-Rich Plasma in Tissue Engineering: Hype and Hope. Eur. Surg. Res. 2018, 59, 265–275. [Google Scholar] [CrossRef]
- Jain, E.; Sheth, S.; Dunn, A.; Zustiak, S.P.; Sell, S.A. Sustained release of multicomponent platelet-rich plasma proteins from hydrolytically degradable PEG hydrogels. J. Biomed. Mater. Res. Part A 2017, 105, 3304–3314. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, M.S.; Patel, S.; Bansal, T. Improvising PRP for use in osteoarthritis knee- upcoming trends and futuristic view. J. Clin. Orthop. Trauma 2019, 10, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, M.S.; Patel, S.; John, R. PRP in OA knee—Update, current confusions and future options. SICOT J. 2017, 3, 27. [Google Scholar] [CrossRef]
- Natesan, S.; Stone, R.; Coronado, R.E.; Wrice, N.L.; Kowalczewski, A.C.; Zamora, D.O.; Christy, R.J. PEGylated Platelet-Free Blood Plasma-Based Hydrogels for Full-Thickness Wound Regeneration. Adv. Wound Care 2019, 8, 323–340. [Google Scholar] [CrossRef]
- Ii, R.S.; Wall, J.T.; Natesan, S.; Christy, R.J.; Stone, R. PEG-Plasma Hydrogels Increase Epithelialization Using a Human Ex Vivo Skin Model. Int. J. Mol. Sci. 2018, 19, 3156. [Google Scholar] [CrossRef]
- Burmeister, D.M.; Roy, D.C.; Becerra, S.C.; Natesan, S.; Christy, R.J. In Situ Delivery of Fibrin-Based Hydrogels Prevents Contraction and Reduces Inflammation. J. Burns Care Res. 2017, 39, 40–53. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Gan, S.; Chu, S.; Maldonado, M.; Tang, S.; Ma, J.; Zhou, J.; Ma, L. Platelet poor plasma gel combined with amnion improves the therapeutic effects of human umbilical cord-derived mesenchymal stem cells on wound healing in rats. Mol. Med. Rep. 2017, 16, 3494–3502. [Google Scholar] [CrossRef] [PubMed]
- Meana, A.; Iglesias, J.; Del Rio, M.; Larcher, F.; Madrigal, B.; Fresno, M.F.; Tevar, F. Large surface of cultured human epithelium obtained on a dermal matrix based on live fibroblast-containing fibrin gels. Burns 1998, 24, 621–630. [Google Scholar] [CrossRef]
- Llames, S.; García, E.; García, V.; Del Rio, M.; Larcher, F.; Jorcano, J.L.; López, E.; Holguín, P.; Miralles, F.; Otero, J.; et al. Clinical Results of an Autologous Engineered Skin. Cell Tissue Bank. 2006, 7, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Gómez, C.; Galán, J.M.; Torrero, V.; Ferreiro, I.; Pérez, D.; Palao, R.; Holguín, P. Use of an autologous bioengineered composite skin in extensive burns: Clinical and functional outcomes. A multicentric study. Burns 2011, 37, 580–589. [Google Scholar] [CrossRef]
- Guerrero-Aspizua, S.; García, M.; Murillas, R.; Retamosa, L.; Illera, N.; Duarte, B.; Holguín, A.; Puig, S.; Hernández, M.I.; Meana, A.; et al. Development of a Bioengineered Skin-Humanized Mouse Model for Psoriasis: Dissecting Epidermal-Lymphocyte Interacting Pathways. Am. J. Pathol. 2010, 177, 3112–3124. [Google Scholar] [CrossRef]
- Martínez-Santamaría, L.; Conti, C.J.; Llames, S.; García, E.; Retamosa, L.; Holguin, A.; Illera, N.; Duarte, B.; Camblor, L.; Llaneza, J.M.; et al. The regenerative potential of fibroblasts in a new diabetes-induced delayed humanised wound healing model. Exp. Dermatol. 2013, 22, 195–201. [Google Scholar] [CrossRef]
- Carretero, M.; Guerrero-Aspizua, S.; Illera, N.; Galvez, V.; Navarro, M.; García-García, F.; Dopazo, J.; Jorcano, J.L.; Larcher, F.; Del Rio, M. Differential Features between Chronic Skin Inflammatory Diseases Revealed in Skin-Humanized Psoriasis and Atopic Dermatitis Mouse Models. J. Investig. Dermatol. 2016, 136, 136–145. [Google Scholar] [CrossRef]
- Burmeister, D.M.; Stone, R.; Wrice, N.L.; Becerra, S.C.; Natesan, S.; Christy, R.J. Fibrin hydrogels prevent contraction and deliver adipose stem cells to debrided deep partial thickness burns for accelerated angiogenesis. FASEB J. 2016, 30, 1300–1307. [Google Scholar] [CrossRef]
- Carriel, V.; Garzón, I.; Jiménez, J.M.; Arias-Santiago, S.; Campos, A.; Sánchez-Quevedo, M.C.; Alaminos, M. Epithelial and stromal developmental patterns in a novel substitute of the human skin generated with fibrin-agarose biomaterials. Cells Tissues Organs 2012, 196, 1–12. [Google Scholar] [CrossRef]
- Buis, B.; Koomen GC, M.; Imholz AL, T.; Struijk, D.G.; Reddingius, R.E.; Arisz, L.; Krediet, R.T. Effect of electric charge on the transperitoneal transport of plasma proteins during CAPD. Nephrol. Dial. Transplant. 1996, 11, 1113–1120. [Google Scholar] [CrossRef]
- Arnaud, P. The Plasma Proteins (Structure, Function and Genetic Control), 2nd ed.; Putnam, F.W., Ed.; Academic Press: New York, NY, USA, 1977. [Google Scholar]
- Turner, P.; McLennan, A.; Bates, A.; White, M. BIOS Instant Notes in Molecular Biology; Garland Science: New York, NY, USA, 2007. [Google Scholar]
- Weigandt, K.M.; White, N.; Chung, D.; Ellingson, E.; Wang, Y.; Fu, X.; Pozzo, D.C. Fibrin Clot Structure and Mechanics Associated with Specific Oxidation of Methionine Residues in Fibrinogen. Biophys. J. 2012, 103, 2399–2407. [Google Scholar] [CrossRef]
- Wedgwood, J.; Freemont, A.J.; Tirelli, N. Rheological and Turbidity Study of Fibrin Hydrogels. Macromol. Symp. 2013, 334, 117–125. [Google Scholar] [CrossRef]
- Chernysh, N.; Weisel, J.W. Dynamic imaging of fibrin network formation correlated with other measures of polymerization. Blood 2008, 111, 4854–4861. [Google Scholar] [CrossRef]
- Ferri, F.; Greco, M.; Arcovito, G.; Bassi, F.A.; De Spirito, M.; Paganini, E.; Rocco, M. Growth kinetics and structure of fibrin gels. Phys. Rev. E 2001, 63, 031401. [Google Scholar] [CrossRef] [PubMed]
- Blombäck, B.; Carlsson, K.; Hessel, B.; Liljeborg, A.; Procyk, R.; Åslund, N. Native fibrin gel networks observed by 3D microscopy, permeation and turbidity. Biochim. Biophys. Acta (BBA) Protein Struct. Mol. Enzym. 1989, 997, 96–110. [Google Scholar] [CrossRef]
- Duong, H.; Wu, B.; Tawil, B. Modulation of 3D Fibrin Matrix Stiffness by Intrinsic Fibrinogen–Thrombin Compositions and by Extrinsic Cellular Activity. Tissue Eng. Part A 2009, 15, 1865–1876. [Google Scholar] [CrossRef]
- Weng, S.; Fu, J. Synergistic regulation of cell function by matrix rigidity and adhesive pattern. Biomaterials 2011, 32, 9584–9593. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, G.; Luo, X.; Qiu, J.; Tang, C. Substrate stiffness regulates the proliferation, migration, and differentiation of epidermal cells. Burns 2012, 38, 414–420. [Google Scholar] [CrossRef]
- Smithmyer, M.E.; Sawicki, L.A.; Kloxin, A.M. Hydrogel scaffolds as in vitro models to study fibroblast activation in wound healing and disease. Biomater. Sci. 2014, 2, 634–650. [Google Scholar] [CrossRef]
- Zarkoob, H.; Chinnathambi, S.; Selby, J.C.; Sander, E.A. Substrate deformations induce directed keratinocyte migration. J. R. Soc. Interface 2018, 15, 20180133. [Google Scholar] [CrossRef] [PubMed]
- Sage, H. Collagens of Basement Membranes. J. Investig. Dermatol. 1982, 79, 51–59. [Google Scholar] [CrossRef][Green Version]
- Pöschl, E.; Schlötzer-Schrehardt, U.; Brachvogel, B.; Saito, K.; Ninomiya, Y.; Mayer, U. Collagen IV is essential for basement membrane stability but dispensable for initiation of its assembly during early development. Development 2004, 131, 1619–1628. [Google Scholar] [CrossRef]
- Evans, M.J.; Fanucchi, M.V.; Miller, L.A.; Carlson, M.A.; Nishio, S.J.; Hyde, D.M. Reduction of collagen VII anchoring fibrils in the airway basement membrane zone of infant rhesus monkeys exposed to house dust mite. Am. J. Physiol. Cell. Mol. Physiol. 2010, 298, L543–L547. [Google Scholar] [CrossRef]
- Ockleford, C.; McCracken, S.; Rimmington, L.; Hubbard, A.; Bright, N.; Cockcroft, N.; Jefferson, T.; Waldron, E.; D’Lacey, C. Type VII collagen associated with the basement membrane of amniotic epithelium forms giant anchoring rivets which penetrate a massive lamina reticularis. Placenta 2013, 34, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Stark, H.-J.; Boehnke, K.; Mirancea, N.; Willhauck, M.J.; Pavesio, A.; Fusenig, N.E.; Boukamp, P. Epidermal Homeostasis in Long-Term Scaffold-Enforced Skin Equivalents. J. Investig. Dermatol. Symp. Proc. 2006, 11, 93–105. [Google Scholar] [CrossRef]
- Baltazar, T.; Merola, J.; Catarino, C.; Xie, C.B.; Kirkiles-Smith, N.C.; Lee, V.; Hotta, S.; Dai, G.; Xu, X.; Ferreira, F.C.; et al. Three Dimensional Bioprinting of a Vascularized and Perfusable Skin Graft Using Human Keratinocytes, Fibroblasts, Pericytes, and Endothelial Cells. Tissue Eng. Part A 2020, 26, 227–238. [Google Scholar] [CrossRef]
- Stark, H.-J.; Willhauck, M.J.; Mirancea, N.; Boehnke, K.; Nord, I.; Breitkreutz, D.; Pavesio, A.; Boukamp, P.; Fusenig, N.E. Authentic fibroblast matrix in dermal equivalents normalises epidermal histogenesis and dermo-epidermal junction in organotypic co-culture. Eur. J. Cell Biol. 2004, 83, 631–645. [Google Scholar] [CrossRef]
- Faust, R.J. Technical Manual of the American Association of Blood Banks. J. Am. Soc. Anesthesiol. 1986, 65, 242. [Google Scholar] [CrossRef]
- Bray, D. Critical Point Drying of Biological Specimens for Scanning Electron Microscopy BT. In Supercritical Fluid Methods and Protocols; Williams, J.R., Clifford, A.A., Eds.; Humana Press: Totowa, NJ, USA, 2000; pp. 235–243. [Google Scholar]
- Rheinwatd, J.G.; Green, H. Seria cultivation of strains of human epidemal keratinocytes: The formation keratinizin colonies from single cell is. Cell 1975, 6, 331–343. [Google Scholar] [CrossRef]
- Del Rio, M.; Larcher, F.; Serrano, F.; Meana, A.; Muñoz, M.; García, M.; Munoz, E.; Martin, C.; Bernad, A.; Jorcano, J.L. A Preclinical Model for the Analysis of Genetically Modified Human Skin In Vivo. Hum. Gene Ther. 2002, 13, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Cardiff, R.D.; Miller, C.H.; Munn, R.J. Manual Hematoxylin and Eosin Staining of Mouse Tissue Sections. Cold Spring Harb. Protoc. 2014, 2014, 655–658. [Google Scholar] [CrossRef] [PubMed]
- Student. The Probable Error of a Mean. Biometrika 1908, 6, 1–25. [Google Scholar] [CrossRef]








Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montero, A.; Quílez, C.; Valencia, L.; Girón, P.; Jorcano, J.L.; Velasco, D. Effect of Fibrin Concentration on the In Vitro Production of Dermo-Epidermal Equivalents. Int. J. Mol. Sci. 2021, 22, 6746. https://doi.org/10.3390/ijms22136746
Montero A, Quílez C, Valencia L, Girón P, Jorcano JL, Velasco D. Effect of Fibrin Concentration on the In Vitro Production of Dermo-Epidermal Equivalents. International Journal of Molecular Sciences. 2021; 22(13):6746. https://doi.org/10.3390/ijms22136746
Chicago/Turabian StyleMontero, Andrés, Cristina Quílez, Leticia Valencia, Paula Girón, José Luis Jorcano, and Diego Velasco. 2021. "Effect of Fibrin Concentration on the In Vitro Production of Dermo-Epidermal Equivalents" International Journal of Molecular Sciences 22, no. 13: 6746. https://doi.org/10.3390/ijms22136746
APA StyleMontero, A., Quílez, C., Valencia, L., Girón, P., Jorcano, J. L., & Velasco, D. (2021). Effect of Fibrin Concentration on the In Vitro Production of Dermo-Epidermal Equivalents. International Journal of Molecular Sciences, 22(13), 6746. https://doi.org/10.3390/ijms22136746






