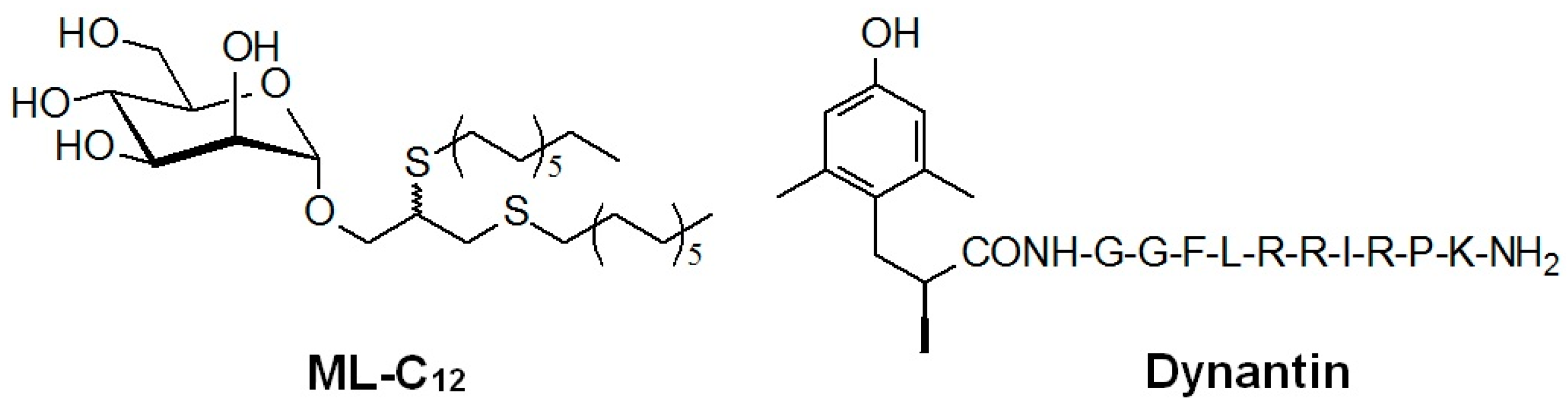
Improving the Utility of a Dynorphin Peptide Analogue Using Mannosylated Glycoliposomes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Entrapment and Plasma Stability
2.2. In Vitro Dopamine Modulation
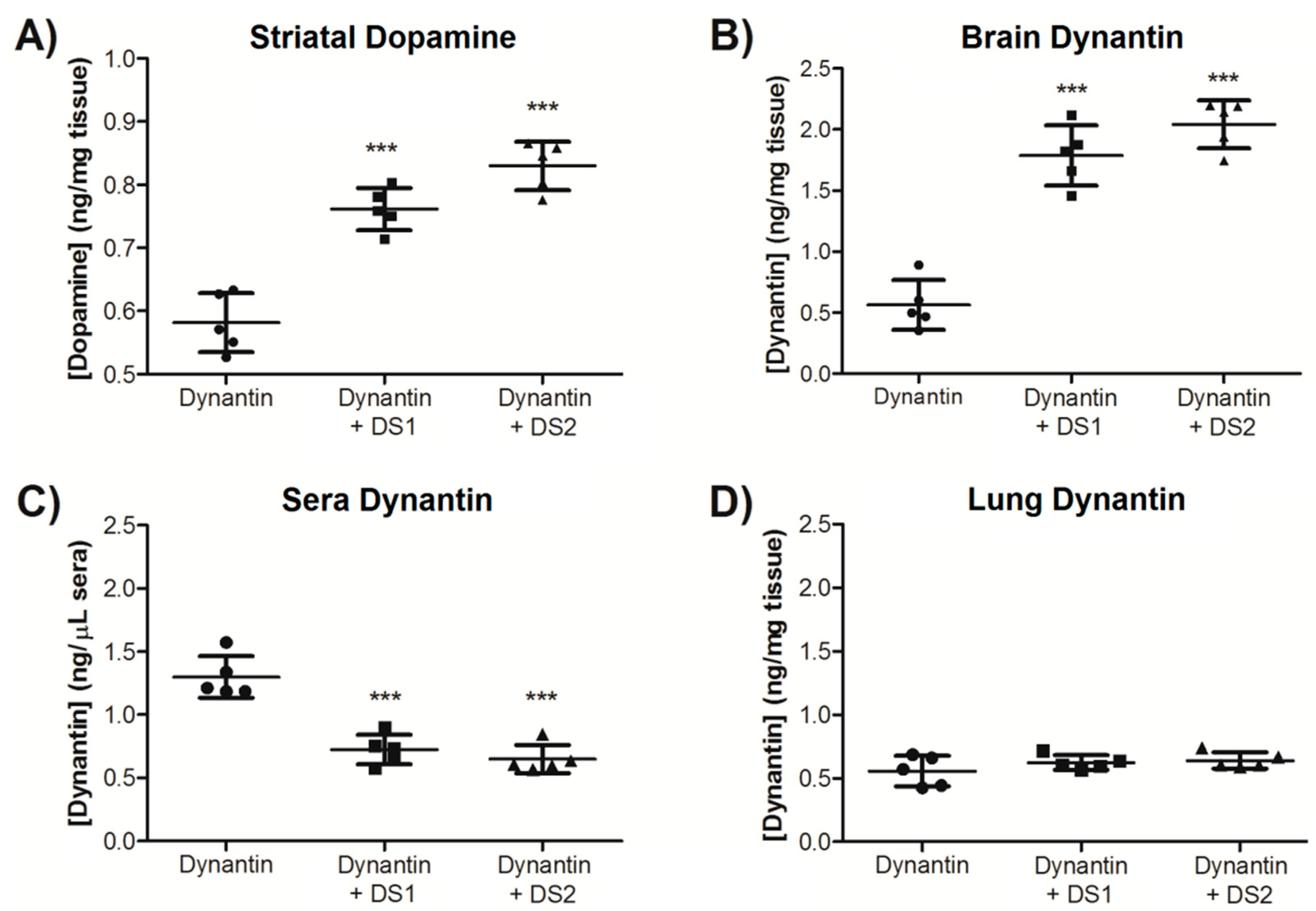
2.3. In Vivo Dynantin Distribution and Dopamine Modulation
3. Materials and Methods
3.1. Reagents
3.2. Human Plasma Collection
3.3. HPLC Conditions
3.4. LC/MS Conditions
3.5. Entrapment and Plasma Stability
3.6. Animals and Husbandry
3.7. In Vitro Dopamine Modulation
3.8. In Vivo Dynantin Distribution and Dopamine Modulation
3.9. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Erak, M.; Bellmann-Sickert, K.; Els-Heindl, S.; Beck-Sickinger, A.G. Peptide chemistry toolbox- transforming natural peptides into peptide therapeutics. Bioorganic Med. Chem. 2018, 10, 2759–2765. [Google Scholar] [CrossRef]
- Uhlig, T.; Kyprianou, T.; Martinelli, F.G.; Oppici, C.A.; Heilgers, D.; Hills, D.; Calvo, X.R.; Verhaert, P. The emergence of peptides in the pharmaceutical business: From exploration to exploitation. EuPA Open Proteom. 2014, 4, 58–69. [Google Scholar] [CrossRef] [Green Version]
- Dioa, L.; Meibohm, B. Pharmacokinetics and pharmacokinetic-pharmacodynamic correlations of therapeutic peptide. Clin. Pharmacokinet. 2013, 52, 855–868. [Google Scholar] [CrossRef]
- Kaspar, A.A.; Reichert, J.M. Future directions for peptide therapeutics development. Drug Discov. Today 2013, 18, 807–817. [Google Scholar] [CrossRef]
- De la Torre, B.G.; Albericio, F. Peptide Therapeutics 2.0. Molecules 2020, 25, 2293. [Google Scholar] [CrossRef]
- Morimoto, H. Therapeutic peptides for CNS indications: Progress and challenges. Bioorganic Med. Chem. 2018, 26, 2859–2862. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A. The CNS as a target of peptides and peptide-based drugs. Expert Opin. Drug Deliv. 2016, 3, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Begley, D.J. Delivery of therapeutic agents to the central nervous system: The problems and the possibilities. Pharmacol. Ther. 2004, 104, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Salameh, T.S.; Banks, W.A. Delivery of Therapeutic Peptides and Proteins to the CNS. Adv. Pharmacol. 2014, 71, 277–299. [Google Scholar]
- Keaney, J.; Campbell, M. The dynamic blood-brain barrier. FEBS J. 2015, 282, 4067–4079. [Google Scholar] [CrossRef]
- Vlieghe, P.; Khrestchatisky, M. Medicinal chemistry based approaches and nanotechnology-based systems to improve CNS drug targeting and delivery. Med. Res. Rev. 2013, 33, 457–516. [Google Scholar] [CrossRef]
- Bhaskar, S.; Tian, F.; Stoeger, T.; Kreyling, W.; de la Fuente, J.M.; Grazú, V.; Borm, P.; Estrada, G.; Ntziachristos, V.; Razansky, D. Multifunctional nanocarriers for diagnostics, drug delivery and targeted treatment across blood-brain barrier: Perspectives on tracking and neuroimaging. Part. Fibre Toxicol. 2010, 7, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saeedi, M.; Eslamifar, M.; Khezri, K.; Dizaj, S.M. Applications in drug delivery to the central nervous system. Biomed. Pharamcother. 2019, 111, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Tosi, G.; Duskey, J.T.; Kreuter, J. Nanoparticles as carriers for drug delivery of macromolecules across the blood-brain barrier. Expert Opin. Drug Deliv. 2020, 17, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Khan, A.I.; Cai, X.; Song, Y.; Lyu, Z.; Du, D.; Dutta, P.; Lin, Y. Overcoming blood-brain barrier transport: Advances in nanoparticle-based drug delivery strategies. Mater. Today 2020, 37, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Vieira, D.B.; Gamarra, L.F. Getting into the brain: Liposome-based strategies for effective drug delivery across the blood-brain barrier. Int. J. Nanomed. 2016, 11, 5381–5414. [Google Scholar] [CrossRef] [Green Version]
- Padridge, W.M. Drug transport across the blood-brain barrier. J. Cereb. Blood Flow Metab. 2012, 32, 1959–1972. [Google Scholar] [CrossRef]
- Asthana, G.S.; Asthana, A.; Kohli, D.V.; Vyas, S.P. Mannosylated chitosan nanoparticles for delivery of antisense oligonucleotides for macrophage targeting. Biomed. Res. Int. 2014, 2014, 526391. [Google Scholar]
- Carrillo-Conde, B.; Song, E.-H.; Chavez-Santoscoy, A.; Phanse, Y.; Ramer-Tait, A.E.; Pohl, N.L.; Wannemuehler, M.J.; Bellaire, B.H.; Narasimhan, B. Mannose-functionalized “pathogen-like” polyanhydride nanoparticles target C-type lectin receptors on dendritic cells. Mol. Pharm. 2011, 8, 1877–1886. [Google Scholar] [CrossRef] [Green Version]
- Costa, A.; Sarmento, B.; Seabra, V. Mannose-functionalized solid lipid nanoparticles are effective in targeting alveolar macrophages. Eur. J. Pharm. Sci. 2018, 114, 103–113. [Google Scholar] [CrossRef] [PubMed]
- SÏtimac, A.; CvitasÏ, J.T.; Frkanec, L.; Vugrek, O.; Frkanex, R. Design and syntheses of mono and multivalent mannosyl-lipoconjugates for targeted liposomal drug delivery. Int. J. Pharm. 2016, 511, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.C.; Chaves, L.L.; Pinheiro, M.; Ferreira, D.; Sarmento, B.; Reis, S. Design and statistical modeling of mannose-decorated dapsone-containing nanoparticles as a strategy of targeting intestinal M-cells. Int. J. Nanomed. 2016, 11, 2601–2617. [Google Scholar]
- Vieira, A.C.; Chaves, L.; Pinheiro, M.; Costa Lima, S.A.; Ferreira, D.; Sarmento, B. Mannosylated solid lipid nanoparticles for the selective delivery of rifampicin to macrophages. Artif. Cells Nanomed. Biotechnol. 2018, 46, 653–663. [Google Scholar] [CrossRef]
- Wijagkanalan, W.; Kawakami, S.; Takenaga, M.; Igarashi, R.; Yamashita, F.; Hashida, M. Efficient targeting to alveolar macrophages by intratracheal administration of mannosylated liposomes in rats. J. Control. Release 2008, 125, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Zhang, Q.; Wang, S.; Bharate, P.; Varela-Aramburu, S.; Lu, M. Tumour-targeted drug delivery with mannose-functionalized nanoparticles self-assembled from Amphiphilic B-cyclodextrins. Chemistry 2016, 22, 15216–15221. [Google Scholar] [CrossRef] [PubMed]
- Regnier-Vigoroux, A. The mannose receptor in the brain. Int. Rev. Cytol. 2003, 226, 321–342. [Google Scholar]
- Umezawa, F.; Eto, Y. Liposome targeting to mouse brain: Mannose as a recognition marker. Biochem. Biophys. Res. Commun. 1998, 153, 1038–1044. [Google Scholar] [CrossRef]
- Irache, J.M.; Salman, H.H.; Gamazo, C.; Espuelsas, S. Mannose targeted systems for the delivery of therapeutics. Expert Opin. Drug Deliv. 2008, 6, 703–724. [Google Scholar] [CrossRef]
- Ghosh, S.; Das, N.; Mandal, A.K.; Dungdung, S.R.; Sarkar, S. Mannosylated liposomal cytidine 5′ diphosphocholine prevent age related global moderate cerebral ischemia reperfusion induced mitochondrial cytochrome C release in aged rat brain. Neuroscience 2010, 171, 1287–1299. [Google Scholar] [CrossRef]
- Li, X.Y.; Zhao, Y.; Sun, M.G.; Shi, J.F.; Ju, R.J.; Zhang, C.X.; Li, X.T.; Zhao, W.Y.; Mu, L.M.; Zeng, F.; et al. Multifunctional liposomes loaded with paclitaxel and artemether for treatment of invasive brain glioma. Biomaterials 2014, 35, 5591–5604. [Google Scholar] [CrossRef]
- Goyard, D.; Shiao, T.C.; Fraleigh, N.L.; Vu, H.-Y.; Lee, H.; Diaz-Mitoma, F.; Le, H.-T.; Roy, R. Expedient synthesis of functional single-component glycoliposomes using thiol-yne chemistry. J. Mater. Chem. B 2016, 4, 4227–4233. [Google Scholar] [CrossRef]
- Lewicky, J.D.; Martel, A.L.; Fraleigh, N.L.; Boraman, A.; Nguyen, T.M.-D.; Schiller, P.W.; Shiao, T.C.; Roy, R.; Le, H.-T. Strengthening peptide-based drug activity with novel glyconanoparticle. PLoS ONE 2018, 13, e0204472. [Google Scholar] [CrossRef]
- Lewicky, J.D.; Fraleigh, N.L.; Boraman, A.; Martel, A.L.; Nguyen, T.M.-D.; Schiller, P.W.; Shiao, T.C.; Roy, R.; Montaut, S.; Le, H.-T. Mannosylated glycoliposomes for the delivery of a peptide kappa opioid receptor antagonist to the brain. Eur. J. Pharm. Biopharm. 2020, 154, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Born, J.; Lange, T.; Kern, W.; McGregor, G.P.; Bickel, U.; Fehm, H.L. Sniffing neuropeptide: A transnasal approach to the human brain. Nat. Neurosci. 2002, 5, 514–516. [Google Scholar] [CrossRef]
- Dufes, C.; Olivier, J.-C.; Gaillard, F.; Gaillard, A.; Couet, W.; Muller, J.-M. Brain delivery of vasoactive intestinal peptide (VIP) following nasal administration to rats. Int. J. Pharm. 2003, 255, 87–97. [Google Scholar] [CrossRef] [Green Version]
- Banks, W.A.; During, M.J.; Niehoff, M.L. Brain uptake of glucagon-like peptide 1 antagonist extendin(9–39) after intranasal administration. J. Pharmacol. Exp. Ther. 2004, 309, 469–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, Y.; Nguyen, T.M.-D.; Weltrowska, G.; Berezowska, I.; Lemieux, C.; Chung, N.N.; Schiller, P.W. [2′,6′-Dimethyltyrosine] dynorphin A(1–11)-NH2 analogues lacking an N-terminal amino group: Potent and selective κ opioid antagonists. J. Med. Chem. 2001, 44, 3048–3053. [Google Scholar] [CrossRef] [PubMed]
- Brujinzeel, A.W. Kappa-opioid receptor signalling and brain reward function. Brain Res. Rev. 2009, 62, 127–146. [Google Scholar] [CrossRef] [Green Version]
- Maisonneuve, I.M.; Archer, S.; Glick, S.D. U50,488, a kappa opioid receptor agonist, attenuates cocaine induced increases in extracellular dopamine in the nucleus accumbens of rats. Neurosci. Lett. 1994, 181, 57–60. [Google Scholar] [CrossRef]
- Koob, G.F. Neurobiological substrates for the dark side of compulsivity in addiction. Neuropharmacology 2009, 56, 18–31. [Google Scholar] [CrossRef] [Green Version]
- Lalanne, L.; Ayranci, G.; Kieffler, B.L.; Luts, P.-E. The kappa opioid receptor from addiction to depression and back. Front. Psychiatry 2014, 5, 170. [Google Scholar] [CrossRef] [Green Version]
- Spanagel, R.; Herz, A.; Shippenberg, T.S. Opposing tonically active endogenous opioid systems modulate the mesolimbic dopaminergic pathway. Proc. Natl. Acad. Sci. USA 1992, 89, 2046–2050. [Google Scholar] [CrossRef] [Green Version]
- Chavkin, C.; Koob, G.F. Dynorphin, dysphoria, and dependence: The stress of addiction. Neuropsychopharmacology 2016, 41, 373–374. [Google Scholar] [CrossRef]
- Page, S.; Mavrikaki, M.M.; Lintz, T.; Puttick, D.; Roberts, E.; Rosen, H.; Carrol, F.I.; Carlezon, W.A.J. Chrathoff. Behavioral pharmacology of novel kappa opioid receptor antagonists in rats. Int. J. Neuropsychopharmacol. 2019, 22, 735–745. [Google Scholar]
- Tejada, H.A.; Natvidad, L.A.; Orfila, J.E.; Torres, O.V.; O’Dell, L.E. Dysregulation of kappa-opioid receptor systems by chronic nicotine modulate the nicotine withdrawal syndrome in an age-dependent manner. Psychopharmacology 2012, 224, 289–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, B.M.; Zorrilla, E.P.; Koob, G.F. Systemic kappa-opioid receptor antagonism by nor-binaltorphimine reduces dependence-induced excessive alcohol self-administration in rats. Addict. Biol. 2011, 16, 116–119. [Google Scholar] [CrossRef] [Green Version]
- Bruchas, M.R.; Yang, T.; Schreiber, S.; Defino, M.; Kwan, S.C.; Li, S. Long-acting kappa opioid antagonists disrupt signaling and produce non-competitive effects by activating c-Jun N-terminal kinase. J. Biol. Chem. 2007, 282, 29803–29811. [Google Scholar] [CrossRef] [Green Version]
- Beardsley, P.M.; Howard, J.L.; Skelton, K.L.; Carroll, F.I. Differential effects of the novel kappa opioid receptor antagonist, JDTic, on reinstatement of cocaine-seeking induced by footshock stressors vs. cocaine primes and its antidepressant-like effects in rats. Psychopharmacology 2005, 183, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Jarman, S.K.; Haney, A.M.; Valdez, G.R. Kappa opioid regulation of depressive-like behavior during acute withdrawal and protracted abstinence from ethanol. PLoS ONE 2018, 13, e0205016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whitfield, T.W.J.; Schlosburg, J.E.; Wee, S.; Gould, A.; George, O.; Grant, Y. Kappa opioid receptor in the nucleus accumbens shell mediate escalation of methamphetamine intake. J. Neurosci. 2015, 35, 4296–4305. [Google Scholar] [CrossRef] [Green Version]
- Munro, T.A.; Berry, L.M.; Van’t Veer, A.; Béguin, C.; Carroll, F.I.; Zhao, Z.; Carlezon, W.A.J.; Cohen, B.M. Long-acting κ opioid antagonists nor-BNI, GNTI and JDTic: Pharmacokinetics in mice and lipophilicity. BMC Pharmacol. 2012, 12, 5. [Google Scholar] [CrossRef] [Green Version]
- Carroll, F.I.; Carelzon, W.A.J. Development of κ opioid receptor antagonists. J. Med. Chem. 2013, 56, 2178–2195. [Google Scholar] [CrossRef] [PubMed]
- Chavkin, C.; Martinez, D. Kappa antagonist JDTic in phase 1 clinical trial. Neuropsychopharmacology 2015, 40, 2057–2058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butelman, E.R.; Negus, S.S.; Ai, Y.; de Costa, B.R.; Woods, J.H. Kappa opioid antagonist effects of systemically administered norbinaltorphimine in a thermal antinociception assay in rhesus monkeys. J. Pharmacol. Exp. Ther. 1993, 267, 1269–1276. [Google Scholar]
- Carroll, I.; Thomas, J.B.; Dykstra, L.A.; Granger, A.L.; Allen, R.M.; Howard, J.L.; Pollard, G.T.; Aceto, M.D.; Harris, L.S. Pharmacological properties of JDTic: A novel kappa-opioid receptor antagonist. Eur. J. Pharmacol. 2004, 501, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Stahl, E.L.; Lovell, K.M.; Frankowski, K.J.; Prisinzano, T.E.; Aube, J.; Bohn, L.M. Characterization of kappa opioid receptor mediated, dynorphin stimulated [35S]GTPγS binding in mouse striatum for the evaluation of selective KOR ligands in an endogenous setting. Neuropharmacology 2015, 99, 131–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reed, B.; Butelman, E.R.; Kreek, M.J. Kappa opioid receptor antagonists as potential therapeutics for mood substance use disorders. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Tamiaki, H.; Azefu, Y.; Shibata, R.; Sato, R.; Toma, K. Oligomethylene spacer length dependent interaction of synthetic galactolipids incorporated in phospholipid layers with ricin. Colloids Surf. B Biointerfaces 2006, 53, 87–93. [Google Scholar] [CrossRef]
- Huang, F.; Li, J.; Shi, H.-L.; Wang, T.-T.; Muhtar, W.; Du, M.; Zhang, B.-B.; Wu, H.; Yang, L.; Hu, Z.-B.; et al. Simultaneous quantification of seven hippocampal neurotransmitters in depression mice by LC-MS/MS. J. Neurosci. Methods 2014, 229, 8–14. [Google Scholar] [CrossRef]
- González, R.R.; Fernández, R.F.; Vidal, J.L.M.; Frenich, A.G.; Pérez, M.L.G. Development and validation of an ultra-high performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS) method for the simultaneous determination of neurotransmitters in rat brain sample. J. Neurosci. Methods 2011, 198, 187–194. [Google Scholar] [CrossRef]
- Sørensen, L.K.; Johannsen, M. Sensitive determination of monoamine neurotransmitters, their main metabolites and precursor amino acids in different mouse brain components by liquid chromatography-electrospray tandem mass spectrometry after selective sample clean-up. Biomed. Chromatogr. BMC 2019, 33, e4479. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-H.; Choi, J.; Kim, H.-G.; Kim, H.R. Quantification of neurotransmitters in mouse brain tissue by liquid chromatography coupled electrospray tandem mass spectrometry. J. Anal. Methods Chem. 2014, 2014, 506870. [Google Scholar] [CrossRef] [PubMed]
- Fuertig, R.; Ceci, A.; Camus, S.M.; Bezard, E.; Luippol, A.H.; Hengerer, B. LC-MS based quantification of kynurenine metabolites, tryptophan, monoamines and neopterin in plasma, cerebrospinal fluid and brain. Bioanalysis 2016, 8, 1903–1917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valenza, M.; Butelman, E.R.; Kreek, M.J. Effects of the novel relatively short-acting kappa opioid receptor antagonist LY2444296 in behaviors observed after chronic extended-access cocaine self-administration in rats. Psychopharmacology 2017, 234, 2219–2231. [Google Scholar] [CrossRef] [PubMed]
- Dhuria, S.V.; Hanson, L.R.; Frey, W.H. Intranasal delivery to the central nervous system: Mechanisms and experimental considerations. J. Pharm. Sci. 2010, 99, 1654–1673. [Google Scholar] [CrossRef]
- Hoekman, J.D.; Ho, R.Y.J. Enhanced analgesic responses after preferential delivery of morphine and fentanyl to the olfactory epithelium in rats. Anesth. Analg. 2011, 113, 641–651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wojnicz, A.; Ortiz, J.A.; Casas, A.I.; Freitas, A.E.; Lopez, M.G.; Ruiz-Nuño, A. Simultaneous determination of 8 neurotransmitters and their metabolite levels in rat brain using liquid chromatography in tandem with mass spectrometry: Application to the murine Nrf2 model of depression. Clin. Chim. Acta 2016, 453, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates, 3rd ed.; Academic Press: San Diego, CA, USA, 2007. [Google Scholar]
- Mikhaleva, A.; Kannan, M.; Wagner, C.; Yalcin, B. Histomorphological phenotyping of the adult mouse brain. Curr. Protoc. Mouse Biol. 2016, 6, 307–332. [Google Scholar] [CrossRef] [PubMed]



| Brain (%) | Sera (%) | Lung (%) | |
|---|---|---|---|
| Dynantin | 8.15 ± 3.06 | 21.66 ± 3.99 | 4.93 ± 0.89 |
| Dynantin + DS1 | 24.89 ± 4.39 | 14.03 ± 3.02 | 5.05 ± 0.98 |
| Dynantin + DS2 | 28.03 ± 3.51 | 11.68 ± 1.18 | 5.50 ± 0.53 |
| Time (min) | Solvent | |
|---|---|---|
| A (%) | B (%) | |
| 0 | 100 | 0 |
| 15 | 40 | 60 |
| 18 | 20 | 80 |
| 26 | 20 | 80 |
| 30 | 100 | 0 |
| 40 | 100 | 0 |
| Time (min) | Solvent | |
|---|---|---|
| A (%) | B (%) | |
| 0 | 100 | 0 |
| 15 | 40 | 60 |
| 18 | 20 | 80 |
| 26 | 20 | 80 |
| 30 | 100 | 0 |
| 40 | 100 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewicky, J.D.; Fraleigh, N.L.; Martel, A.L.; Nguyen, T.M.-D.; Schiller, P.W.; Mousavifar, L.; Roy, R.; Le, A.D.; Funk, D.; Le, H.-T. Improving the Utility of a Dynorphin Peptide Analogue Using Mannosylated Glycoliposomes. Int. J. Mol. Sci. 2021, 22, 7996. https://doi.org/10.3390/ijms22157996
Lewicky JD, Fraleigh NL, Martel AL, Nguyen TM-D, Schiller PW, Mousavifar L, Roy R, Le AD, Funk D, Le H-T. Improving the Utility of a Dynorphin Peptide Analogue Using Mannosylated Glycoliposomes. International Journal of Molecular Sciences. 2021; 22(15):7996. https://doi.org/10.3390/ijms22157996
Chicago/Turabian StyleLewicky, Jordan D., Nya L. Fraleigh, Alexandrine L. Martel, Thi M.-D. Nguyen, Peter W. Schiller, Leila Mousavifar, René Roy, Anh Dzung Le, Douglas Funk, and Hoang-Thanh Le. 2021. "Improving the Utility of a Dynorphin Peptide Analogue Using Mannosylated Glycoliposomes" International Journal of Molecular Sciences 22, no. 15: 7996. https://doi.org/10.3390/ijms22157996








